Introduction
Choosing the right investigator site for clinical trials in Chile is not just important; it can make or break the success of first-in-human studies. With a regulatory landscape that offers efficient approval timelines and a wealth of local expertise, Chile presents unique opportunities for sponsors aiming to streamline their research processes. Yet, navigating the complexities of site selection can be daunting, especially when it comes to evaluating geographic advantages and fostering strong relationships with local investigators. Are sponsors truly leveraging these elements to secure successful clinical outcomes in this dynamic environment?
Understand the Regulatory Landscape in Chile
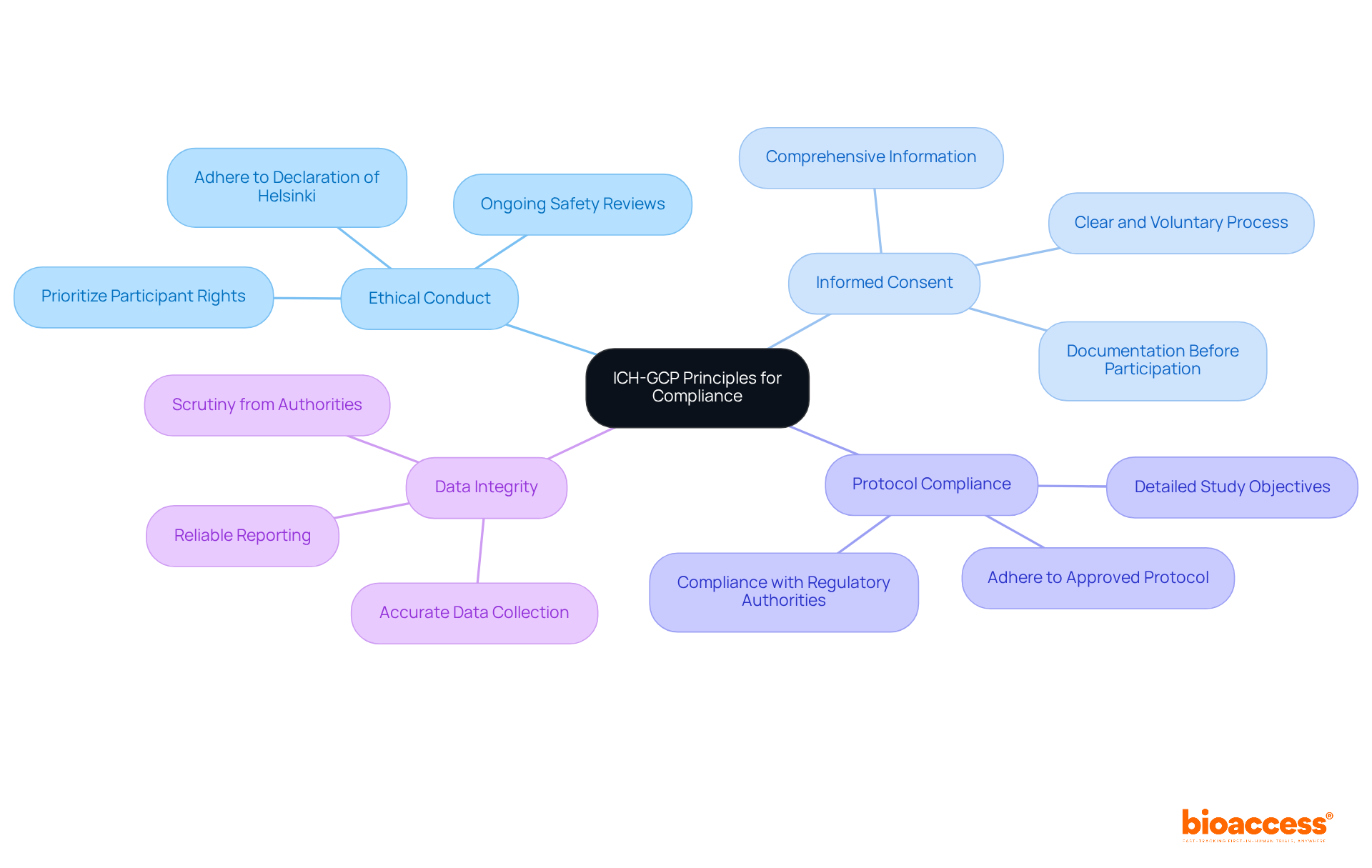
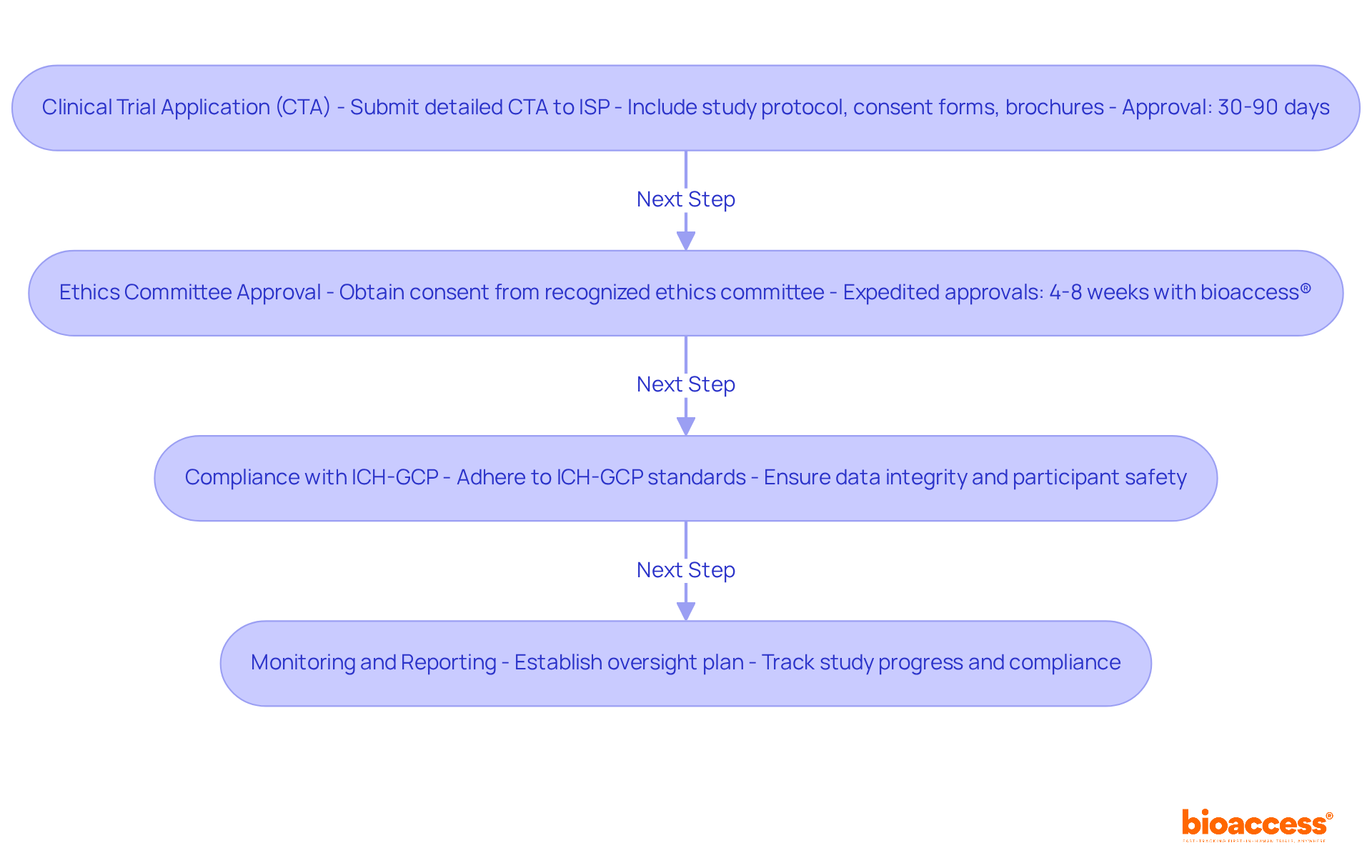
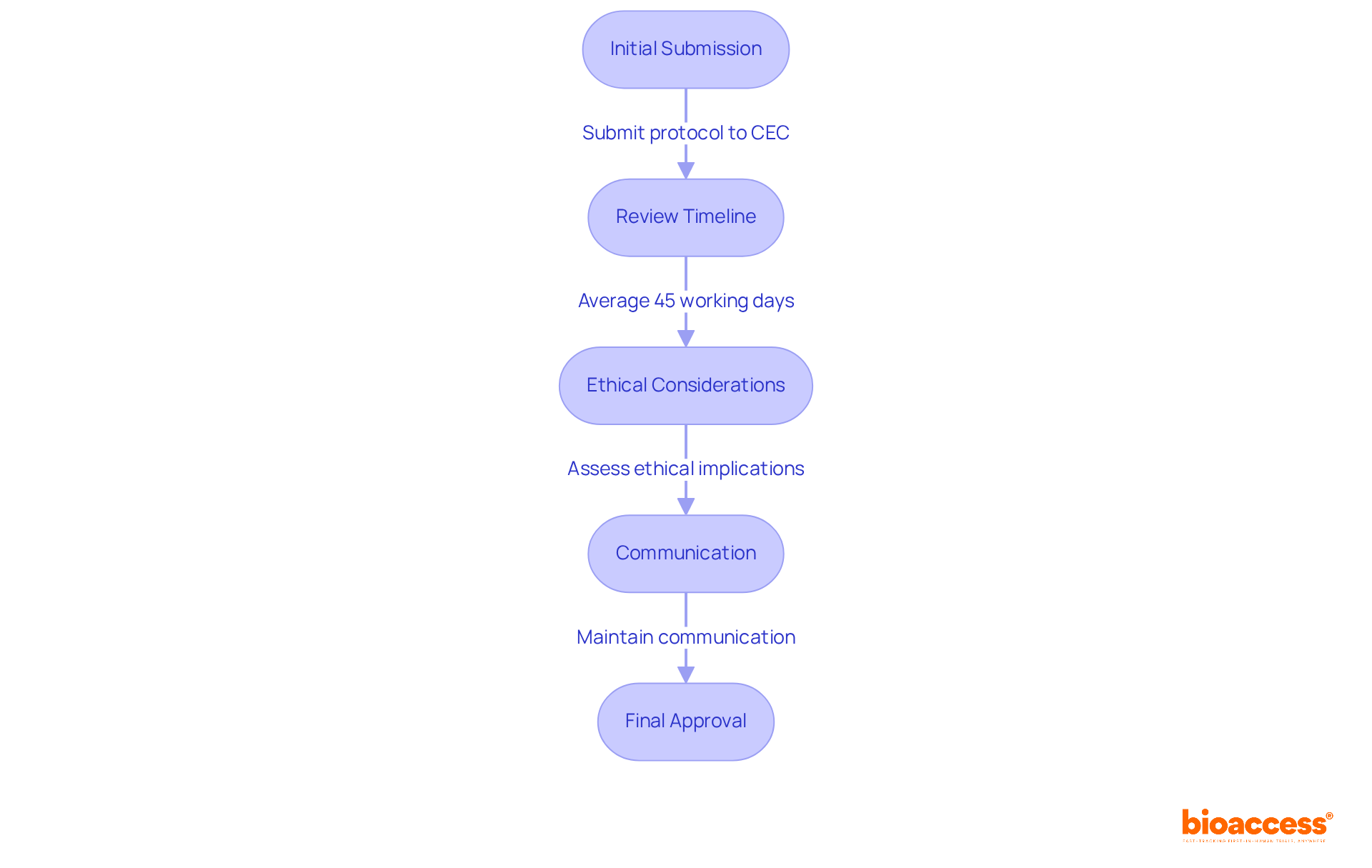
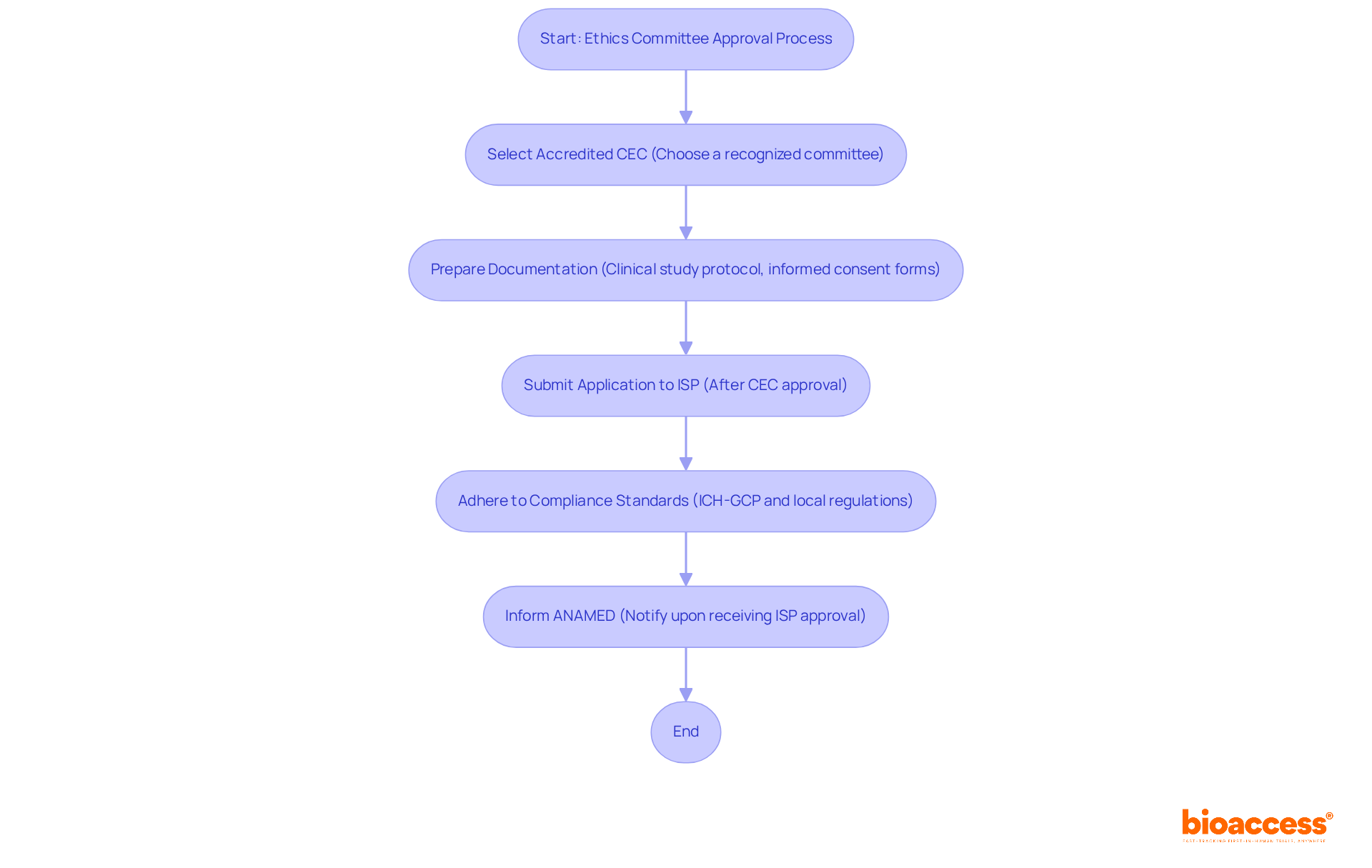
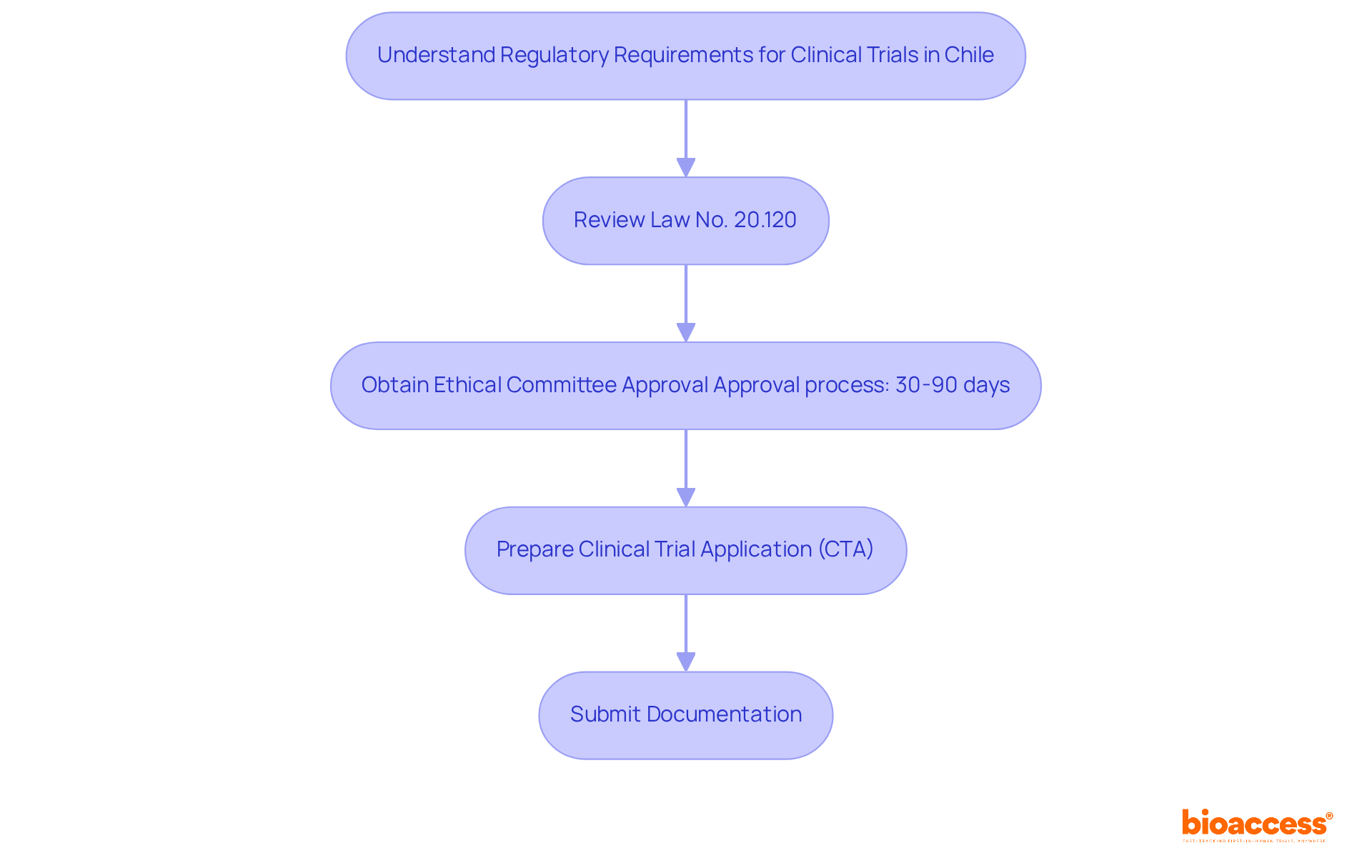
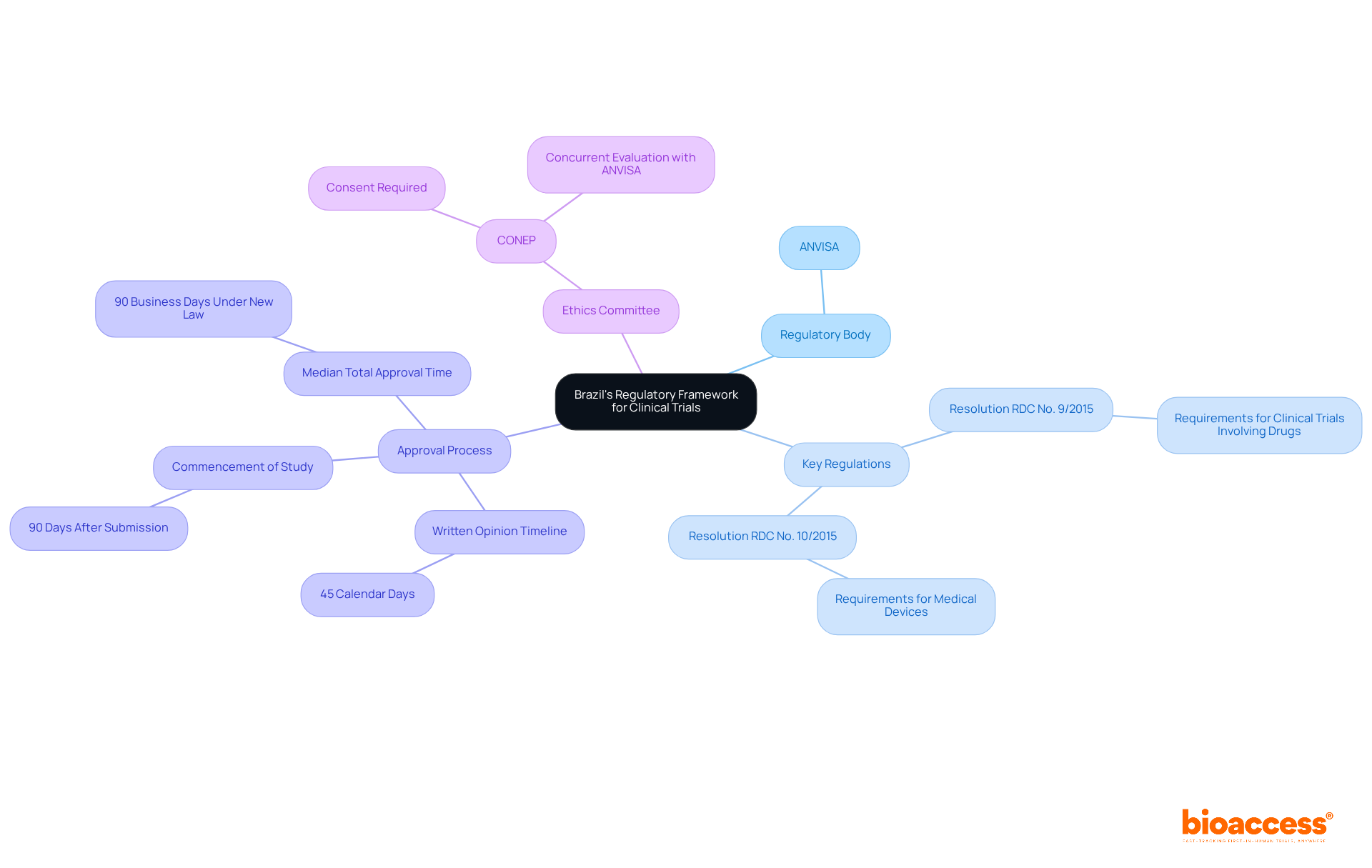
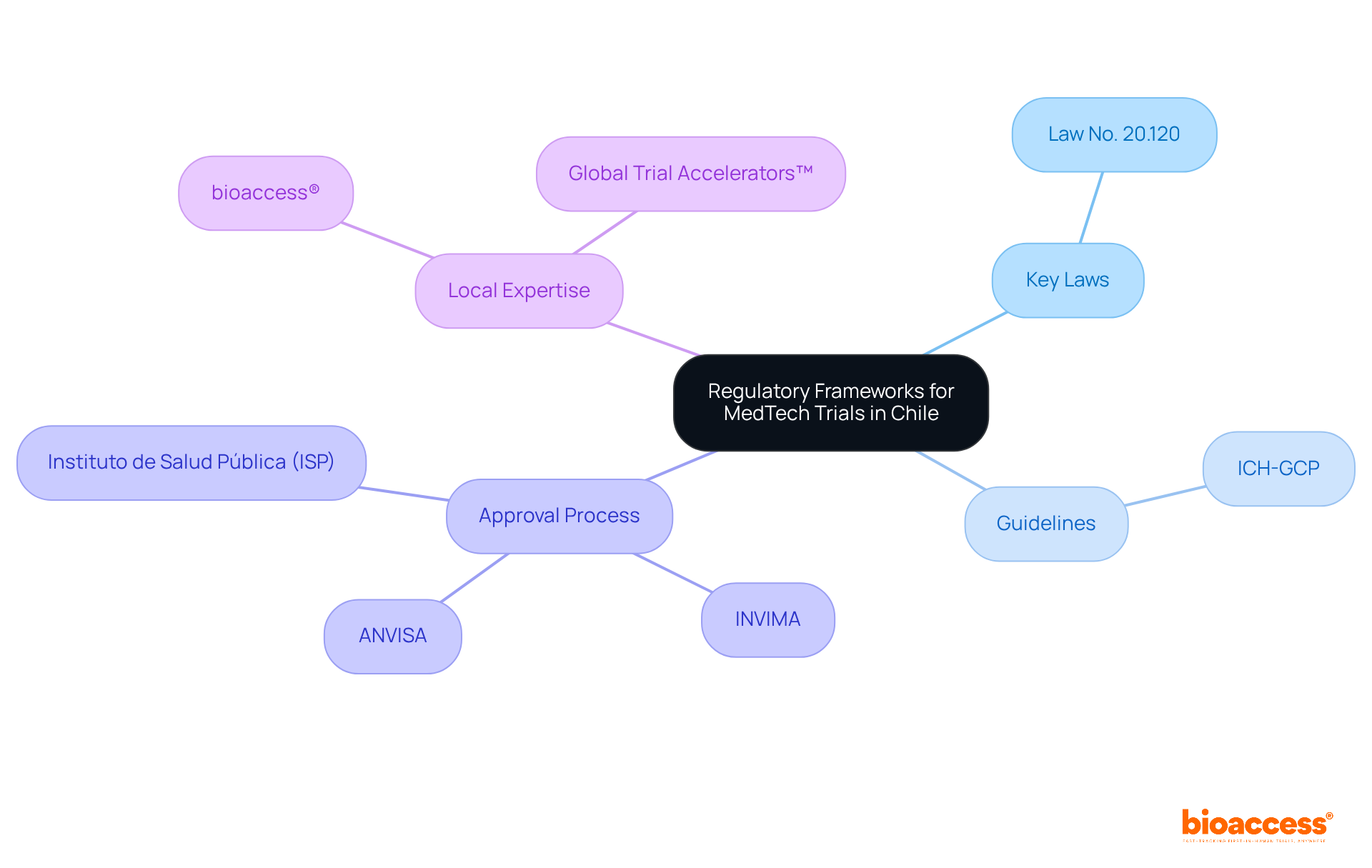
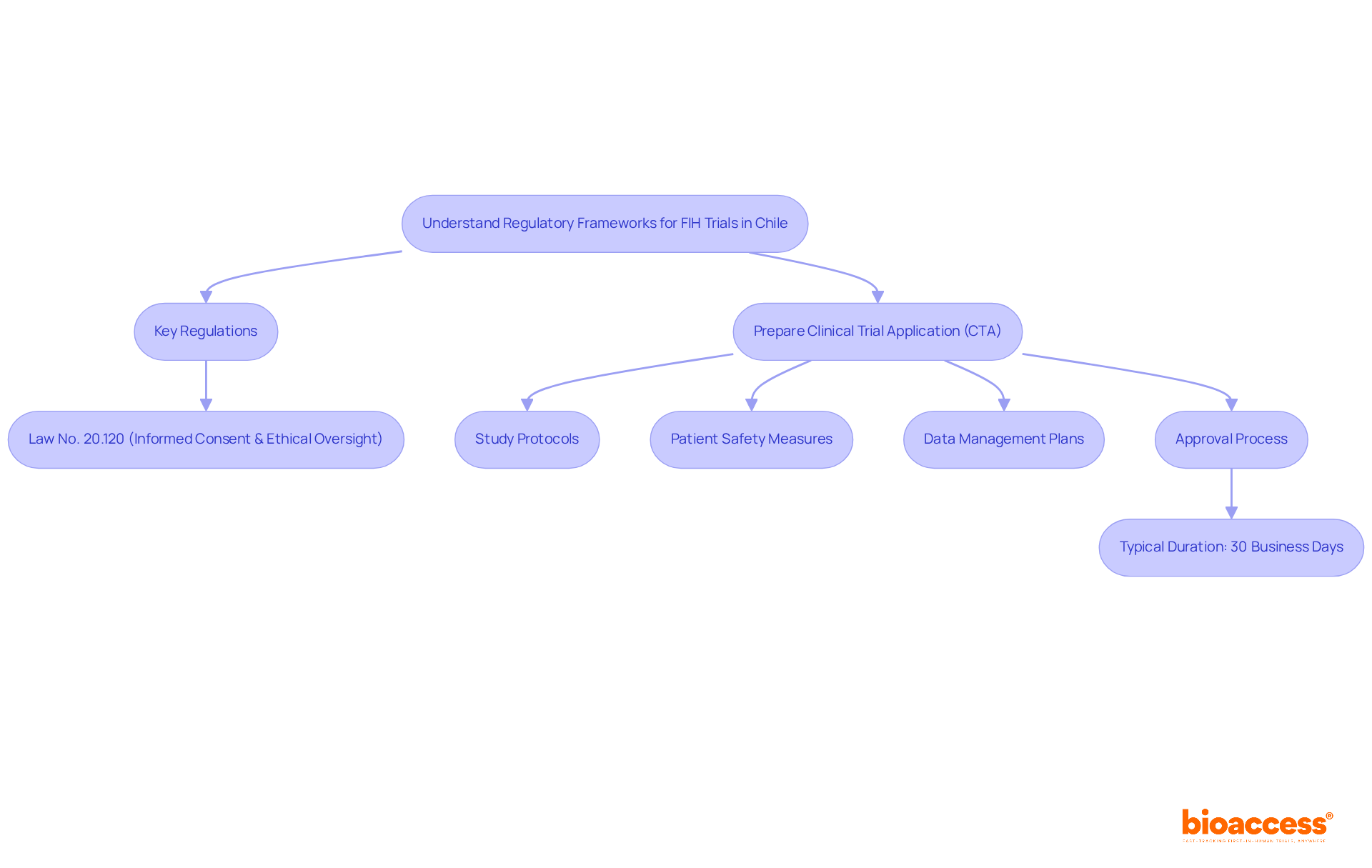
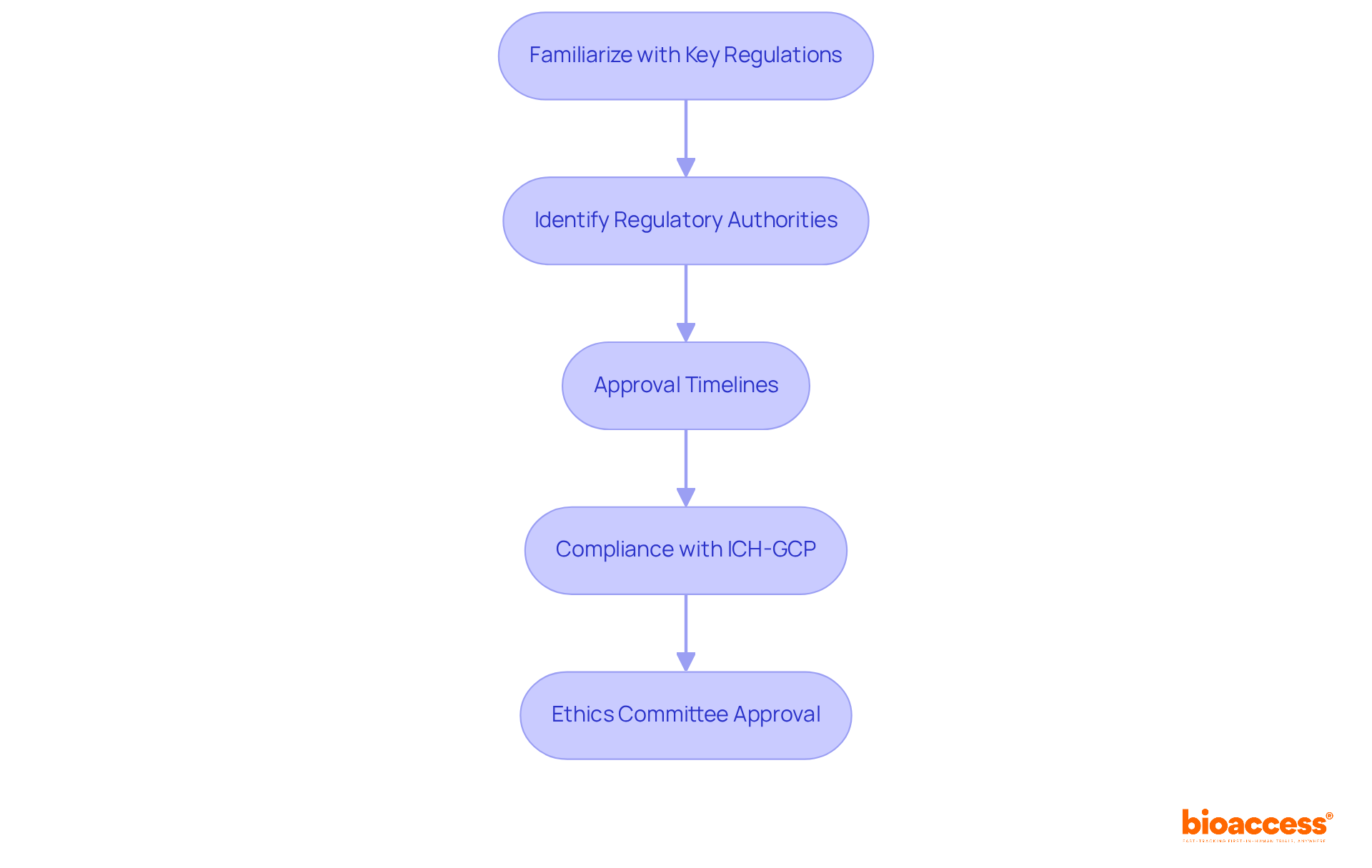
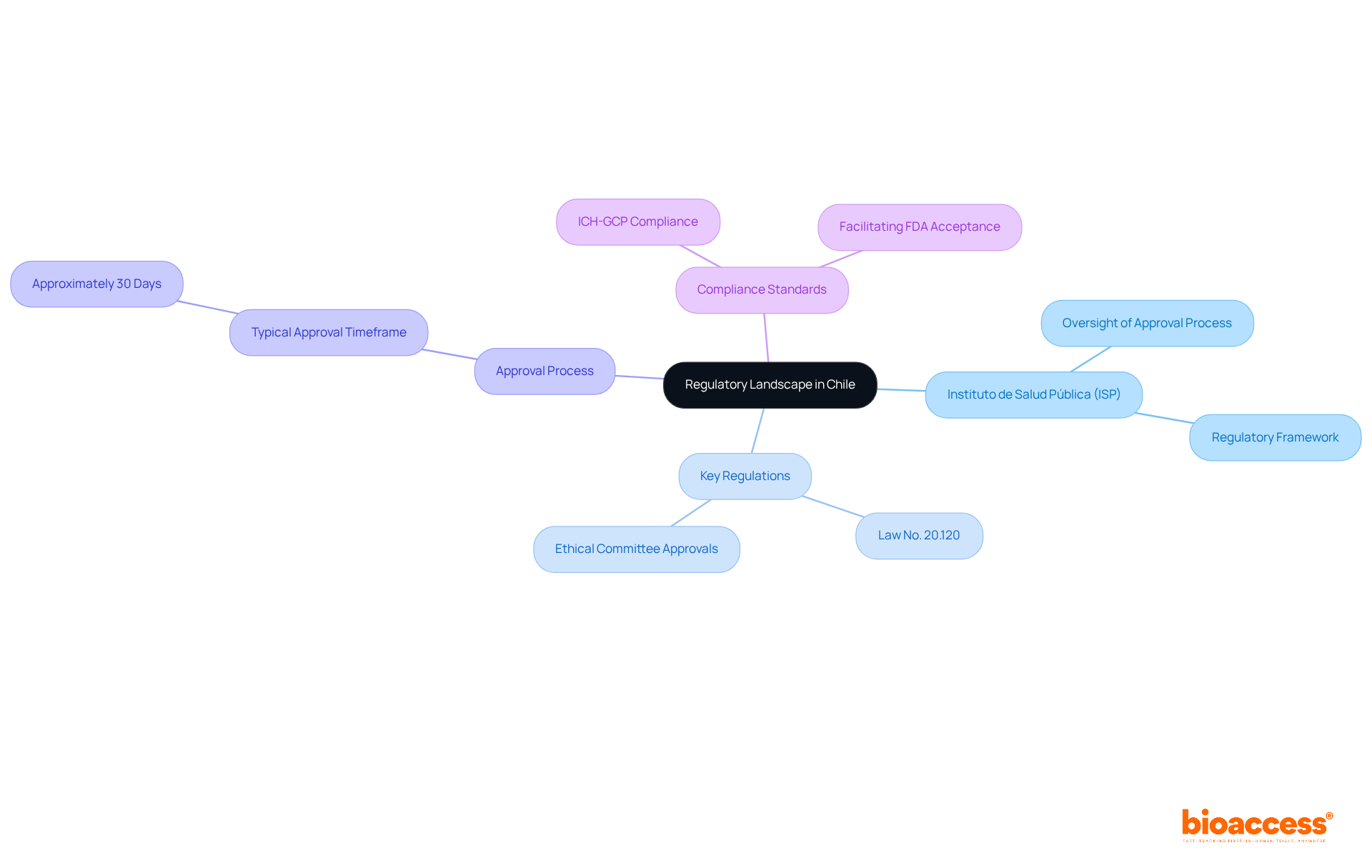
Understanding Chile’s regulatory framework is crucial for successful clinical research, especially for first-in-human studies. The Instituto de Salud Pública (ISP) oversees the approval process for research projects, making it essential to navigate this regulatory landscape effectively. Key regulations, such as Law No. 20.120, outline the requirements for conducting human research and mandate ethical committee approvals. In Chile, studies typically receive approval in about 30 days. This efficiency stands out compared to other regions. To facilitate FDA acceptance of data generated during studies, sponsors must ensure compliance with ICH-GCP standards. By mastering these regulations, researchers can streamline their processes and enhance their chances of success, establishing Chile as a strategic center for research in Latin America.
Evaluate Site Expertise and Geographic Location
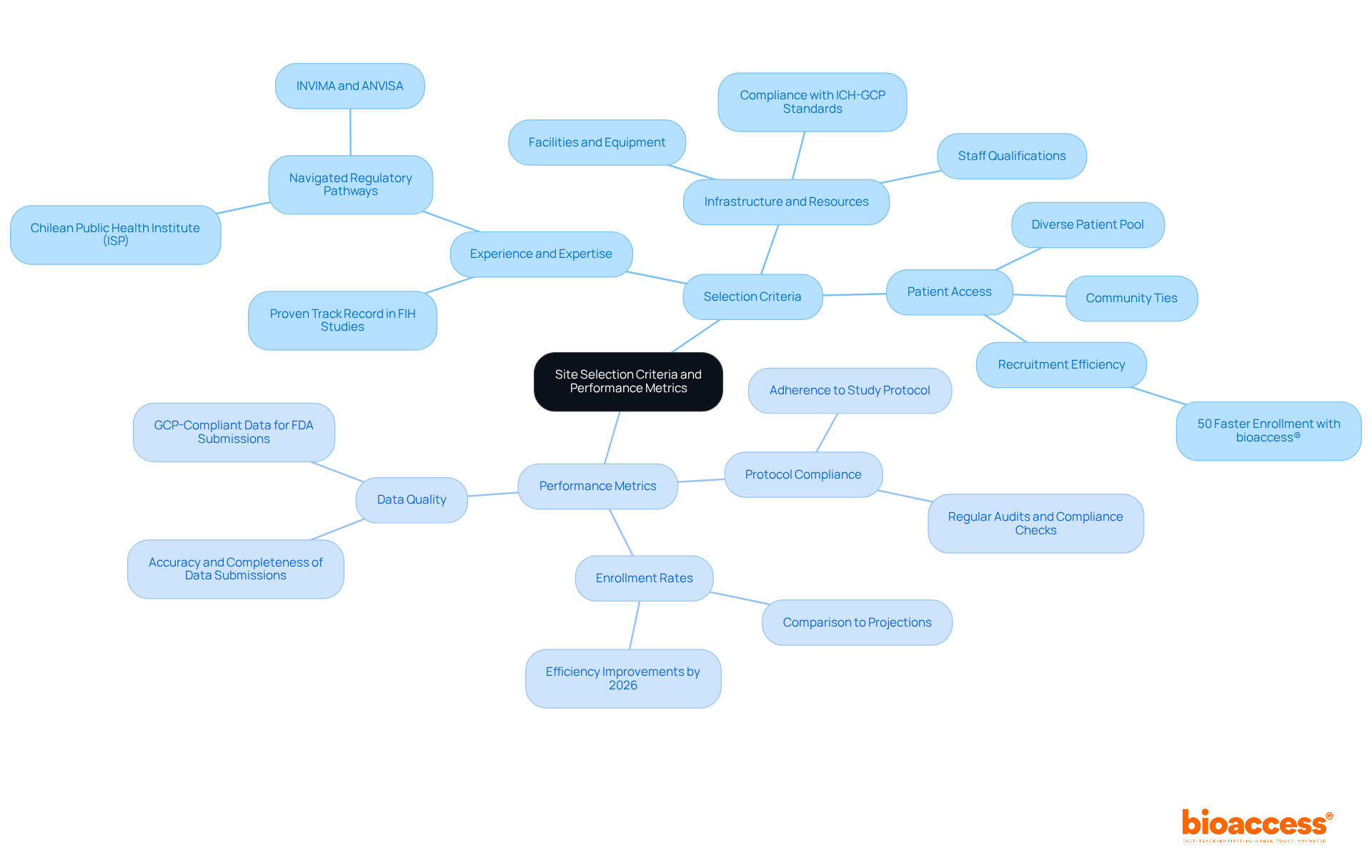
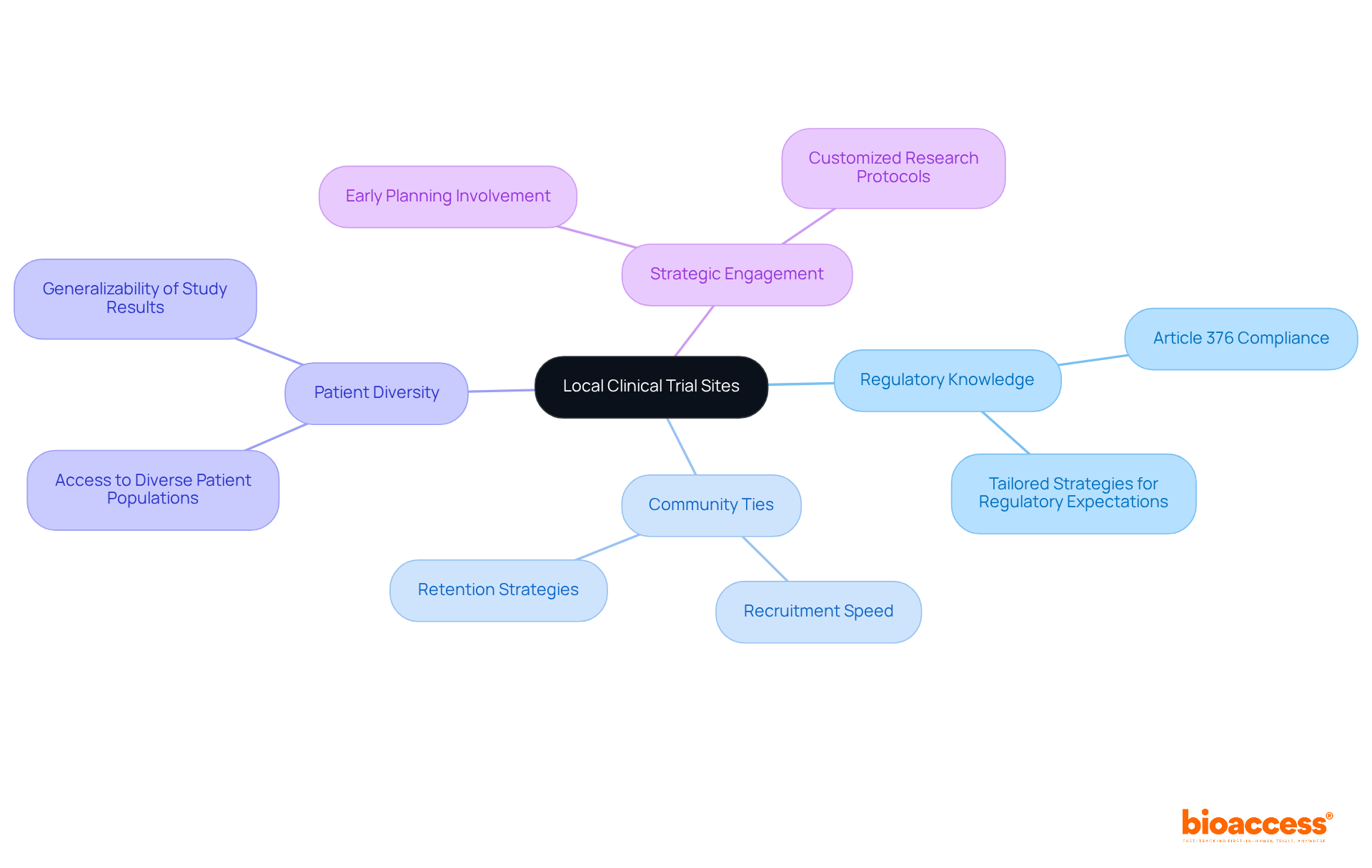
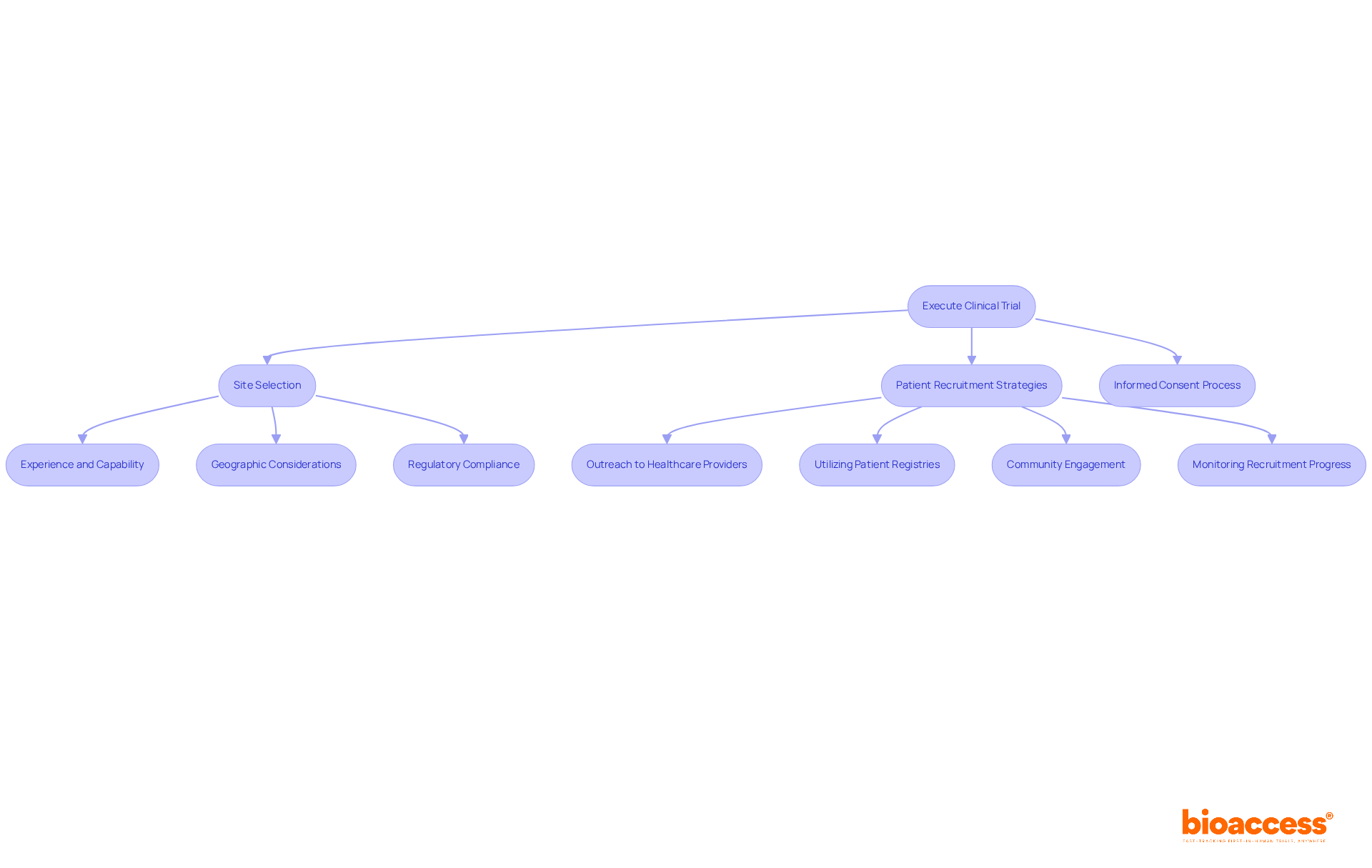
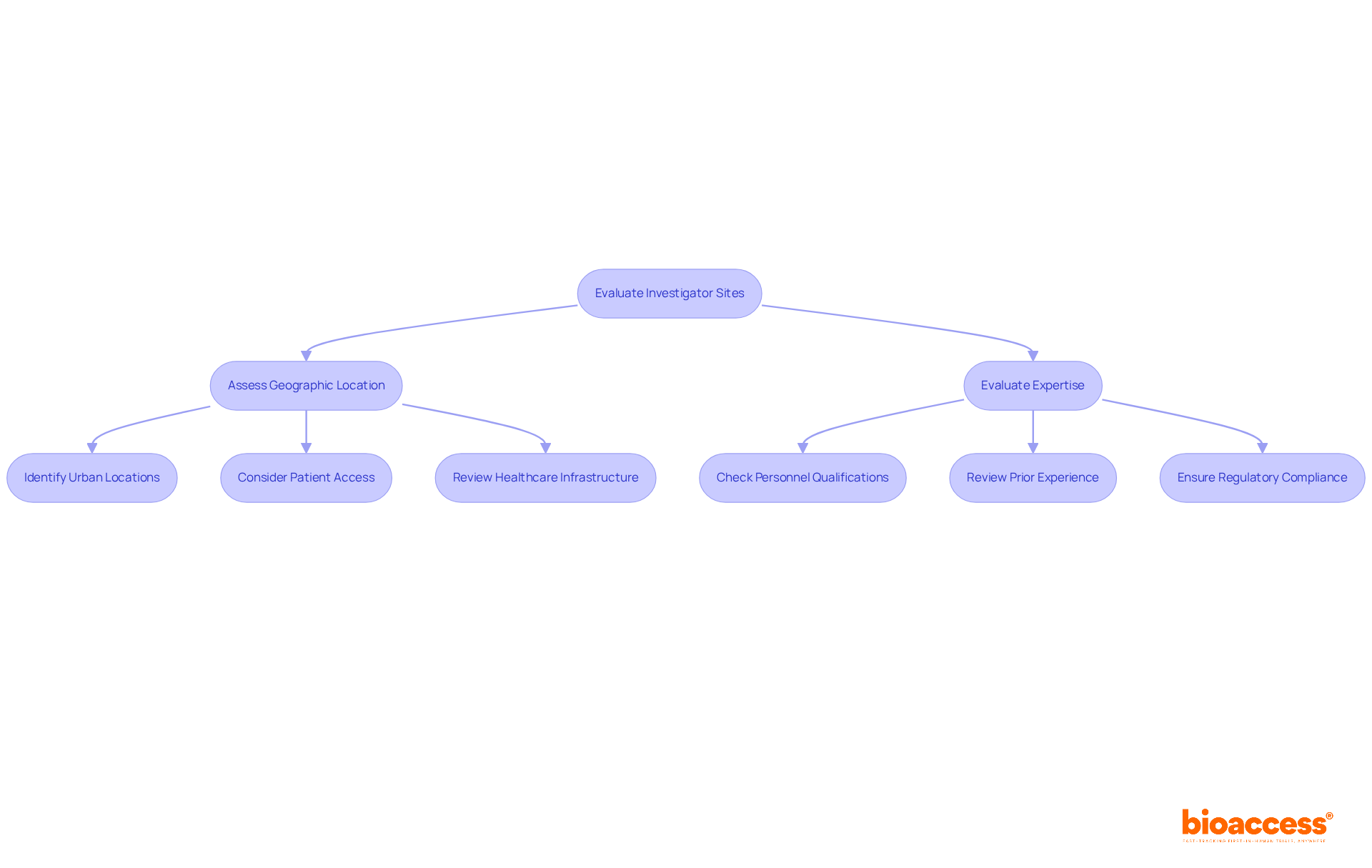
Selecting the right investigator site in Chile for clinical trial first-in-human (FIH) studies is a critical decision that can determine the success of your research. A comprehensive assessment of both expertise and geographic positioning is essential. Urban locations are vital because they offer better access to diverse patient groups, which is essential for successful recruitment. Centers like Santiago and Valparaíso are often preferred due to their advanced healthcare infrastructure and higher patient availability, enhancing recruitment efficiency.
In addition to geographic advantages, evaluating the qualifications of personnel and their prior experience with similar studies is crucial. Adhering to regulatory standards, such as ICH-GCP and FDA acceptance, ensures that the location can meet the stringent requirements of the study. The Chilean Ministry of Health oversees the approval process, and delays in regulatory approvals can significantly impact timelines, posing challenges for sponsors. By prioritizing the investigator site Chile clinical trial with established expertise and strategic locations, sponsors can not only enhance recruitment but also streamline the overall research process.
Moreover, utilizing bioaccess®’s Innovation Runway can significantly expedite the FIH study process. This pathway is designed to accelerate medical development, enabling MedTech, Biopharma, and Radiopharma startups to achieve milestones 40% faster. Additionally, the regulatory landscape in Chile, including the approval timelines set by the Chilean Ministry of Health, can be navigated more efficiently with the support of bioaccess®, which facilitates fast ethics approvals and early feasibility studies. By leveraging bioaccess®’s resources and focusing on strategic site selection, sponsors can navigate the complexities of FIH studies in Chile with confidence.
Foster Relationships with Local Investigators
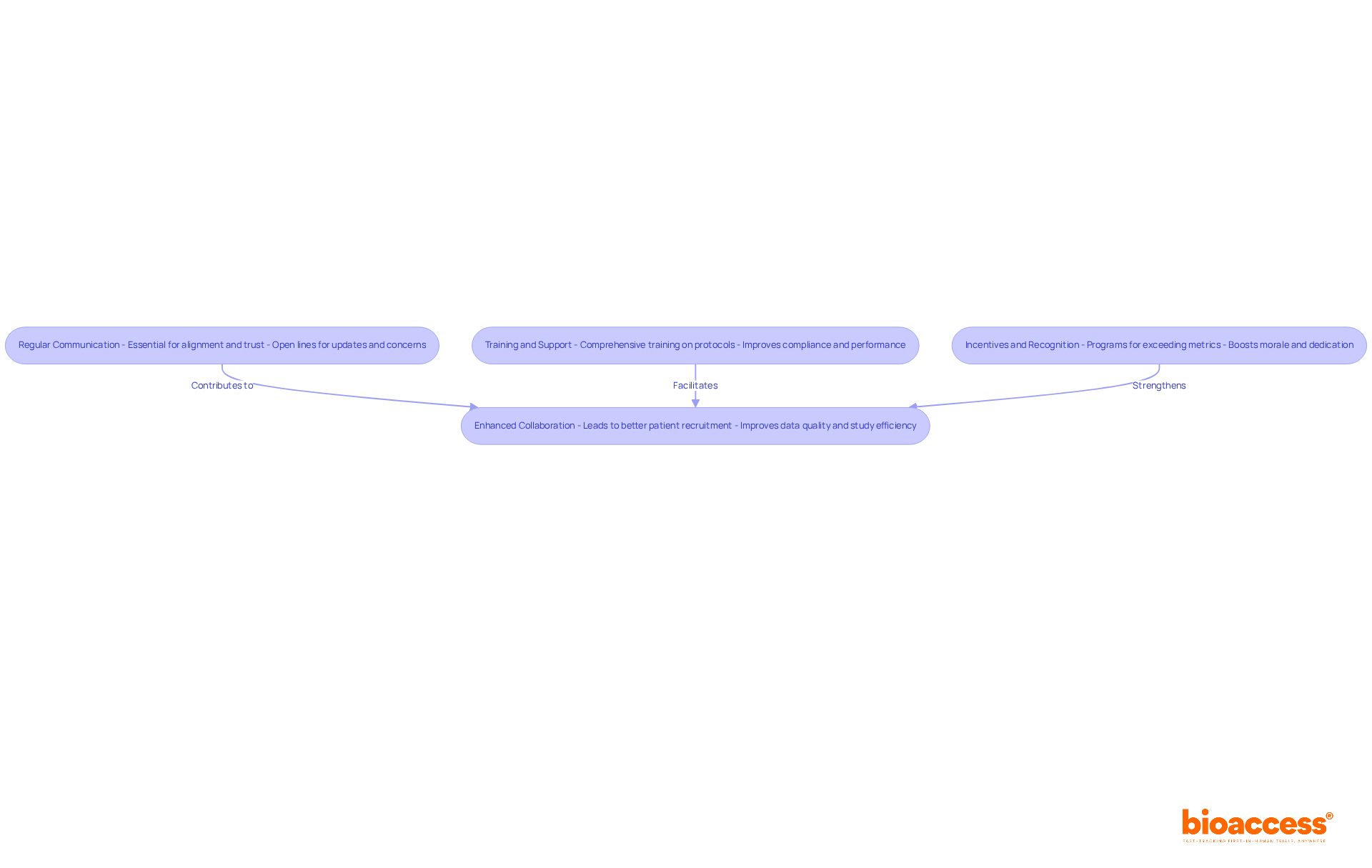
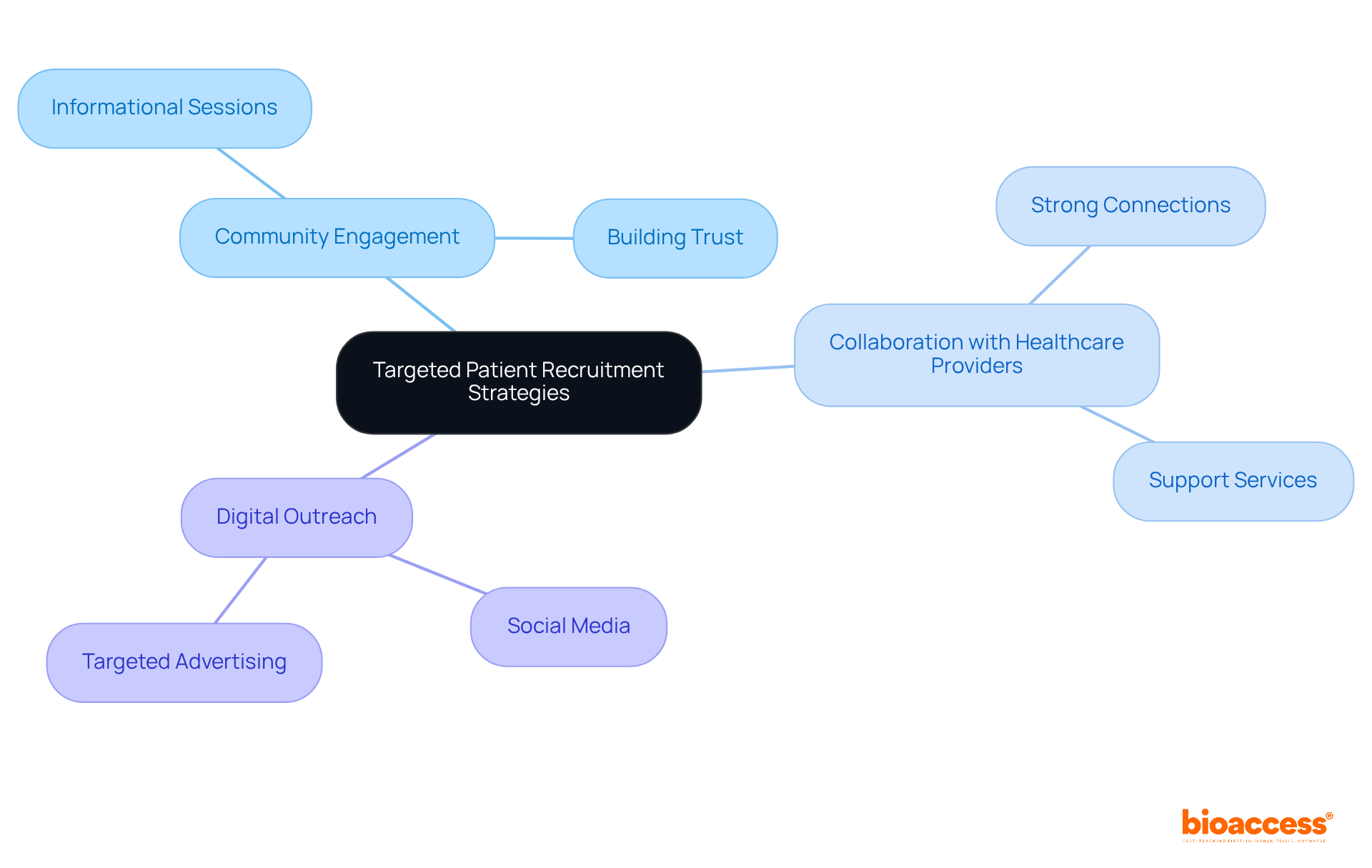
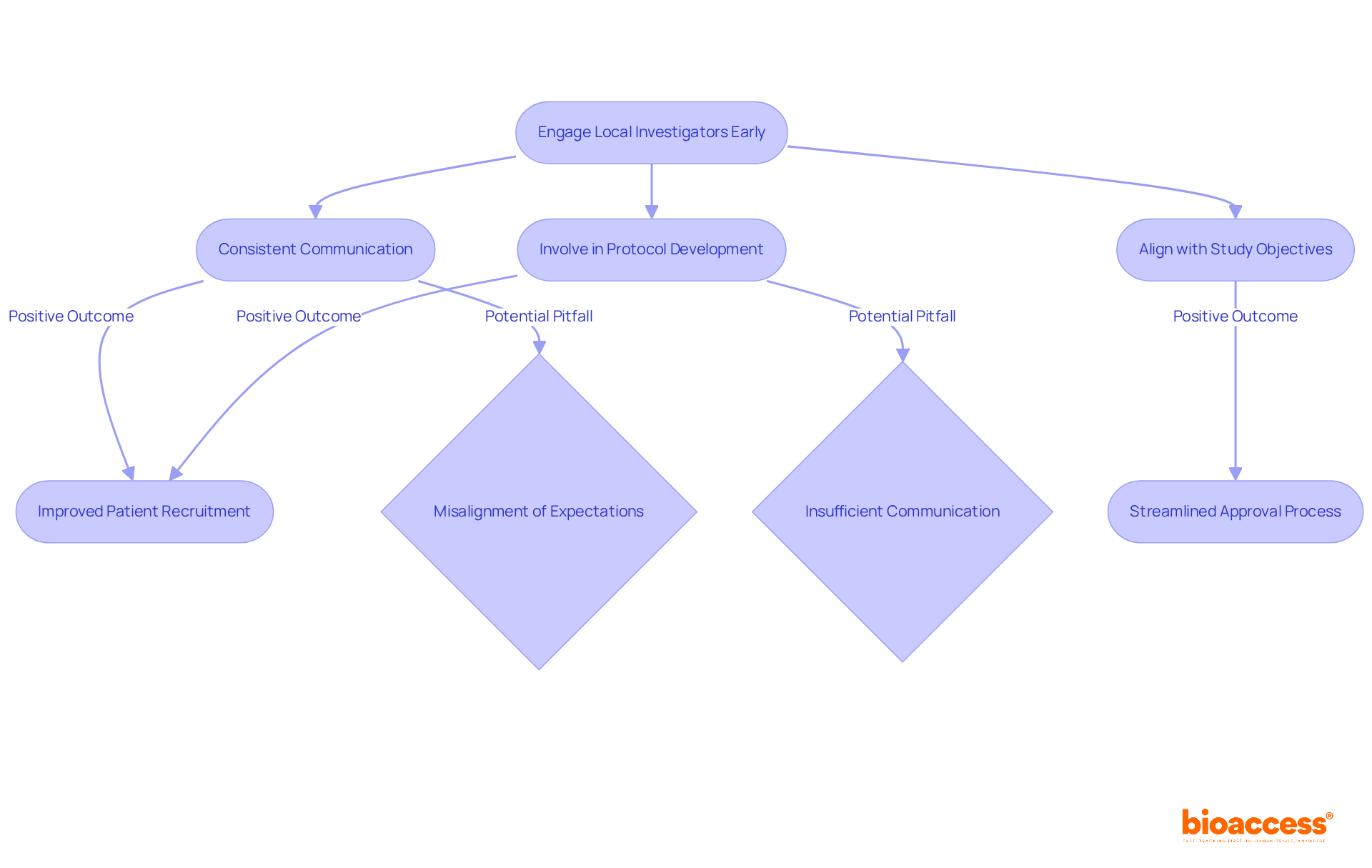
In the realm of medical research in Chile, the strength of local investigator connections at the investigator site Chile clinical trial can significantly impact the success of a study. Building strong relationships with these investigators is essential for success. They possess profound insights into the patient population and the regulatory environment, which can significantly enhance study design and execution. Engaging with them early in the clinical study process fosters collaboration. It also ensures alignment with the study’s objectives.
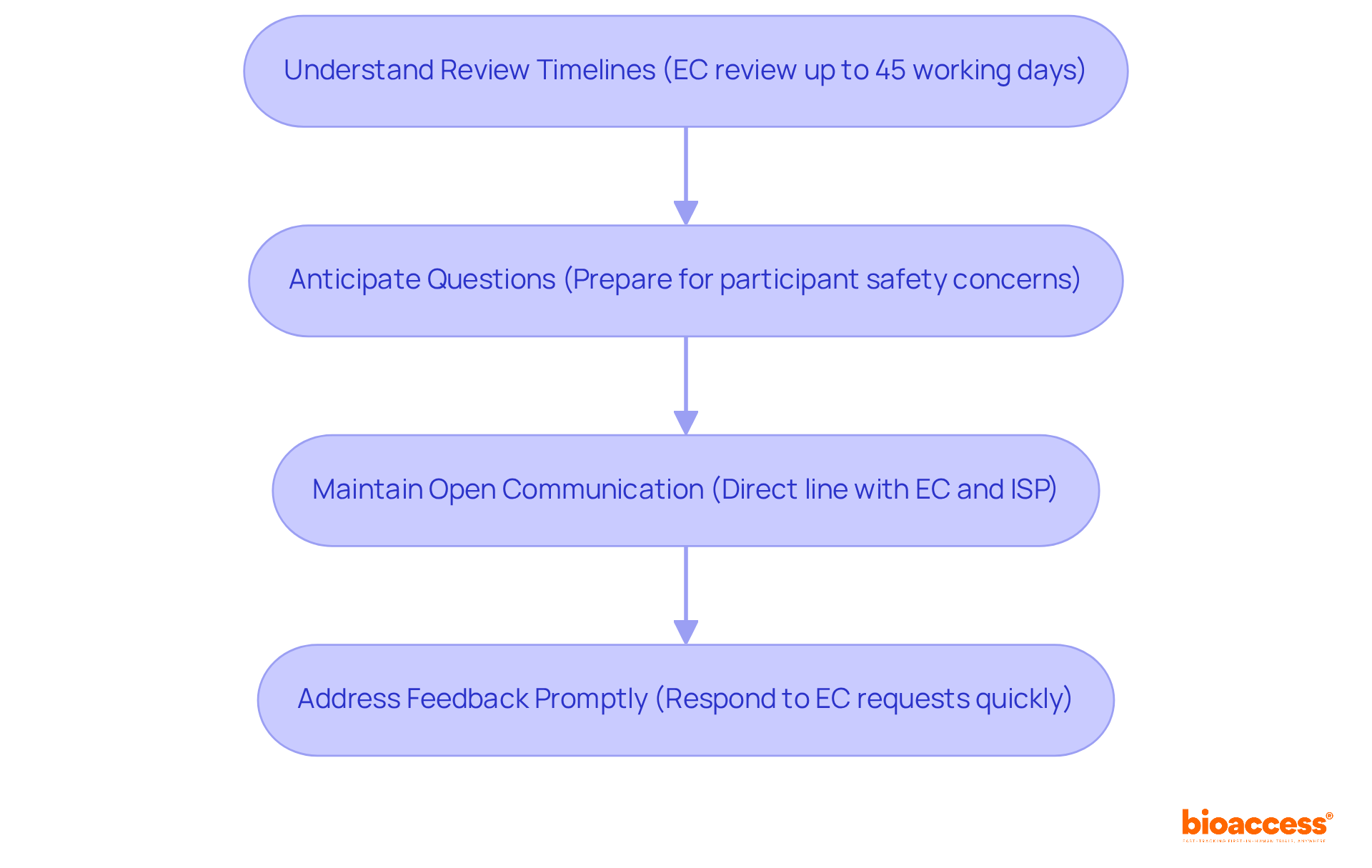
Consistent communication, including updates and meetings, is crucial for tackling any issues that may arise during the study. For instance, involving local investigators in the protocol development phase can lead to more practical and culturally relevant study designs, ultimately improving patient recruitment and retention. This proactive approach streamlines the approval process with regulatory authorities like the Instituto de Salud Pública (ISP). It also aligns with ICH-GCP standards, ensuring that studies are conducted efficiently and ethically.
By utilizing local knowledge, organizations can navigate the complexities of the investigator site Chile clinical trial environment more effectively, enhancing the likelihood of successful results. Misalignment of expectations can derail even the best-laid plans. Recognizing potential pitfalls, like misalignment of expectations or insufficient communication, helps stakeholders avoid common errors and nurture stronger collaborations with investigators.
With the support of bioaccess®, which has successfully accelerated over 50 MedTech and Biopharma companies through its deep understanding of the local landscape, sponsors can further enhance their research strategies and market entry in Latin America. Testimonials from clients emphasize how bioaccess®’s customized strategy has resulted in enhanced patient engagement and accelerated study timelines, illustrating the concrete advantages of working with local investigators. By leveraging local insights, sponsors can not only avoid common pitfalls but also pave the way for groundbreaking advancements in patient care.
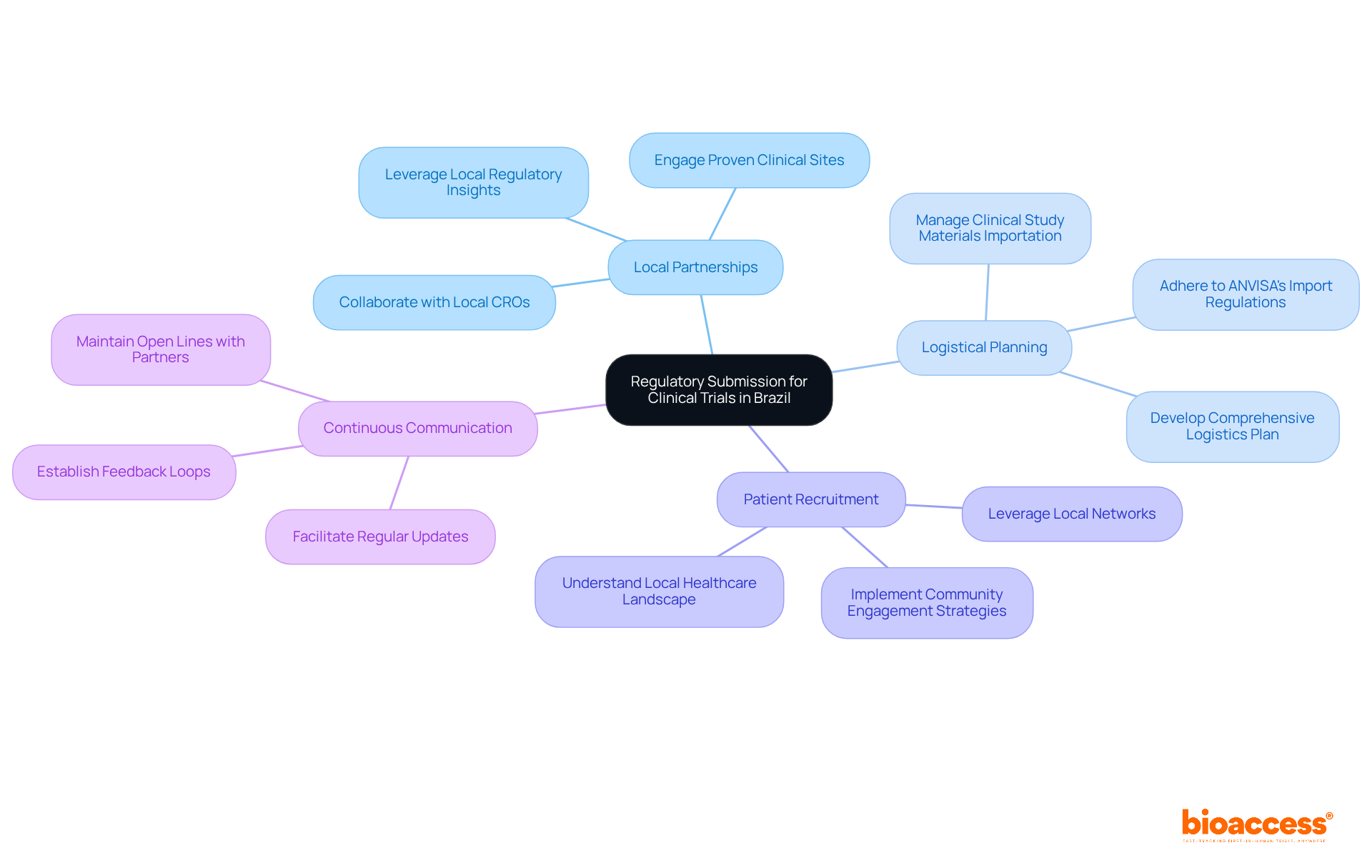
Utilize Pre-Qualified Clinical Trial Sites
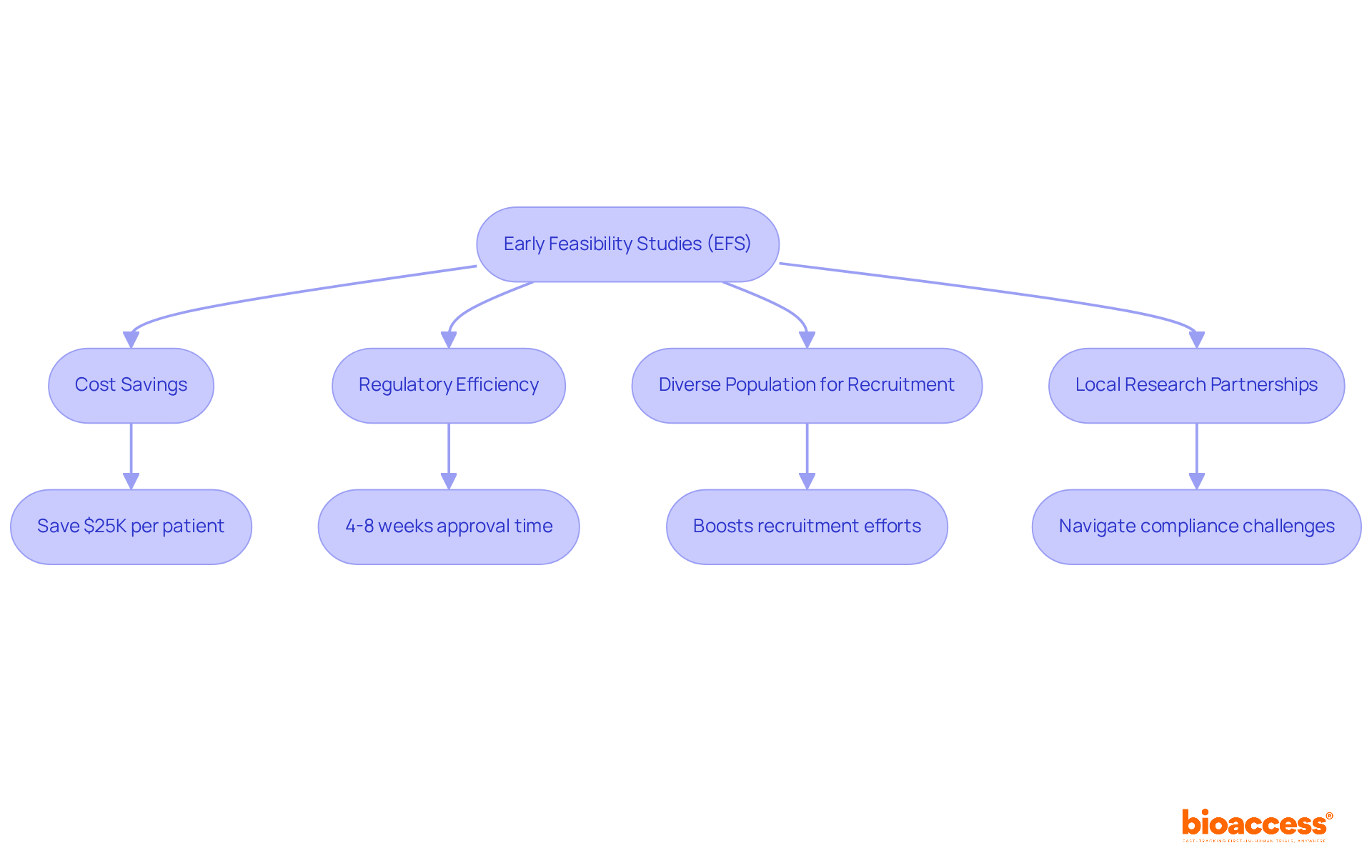
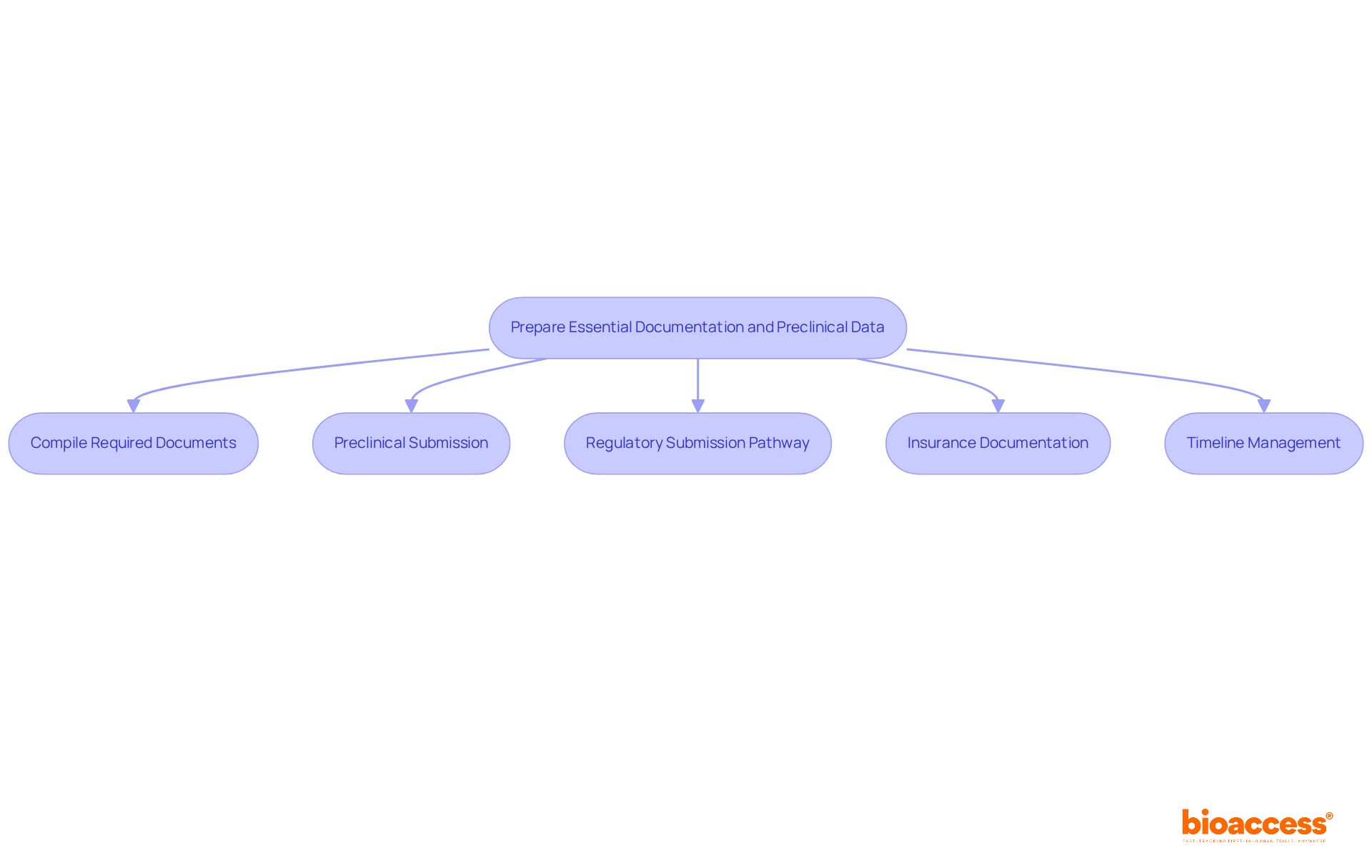
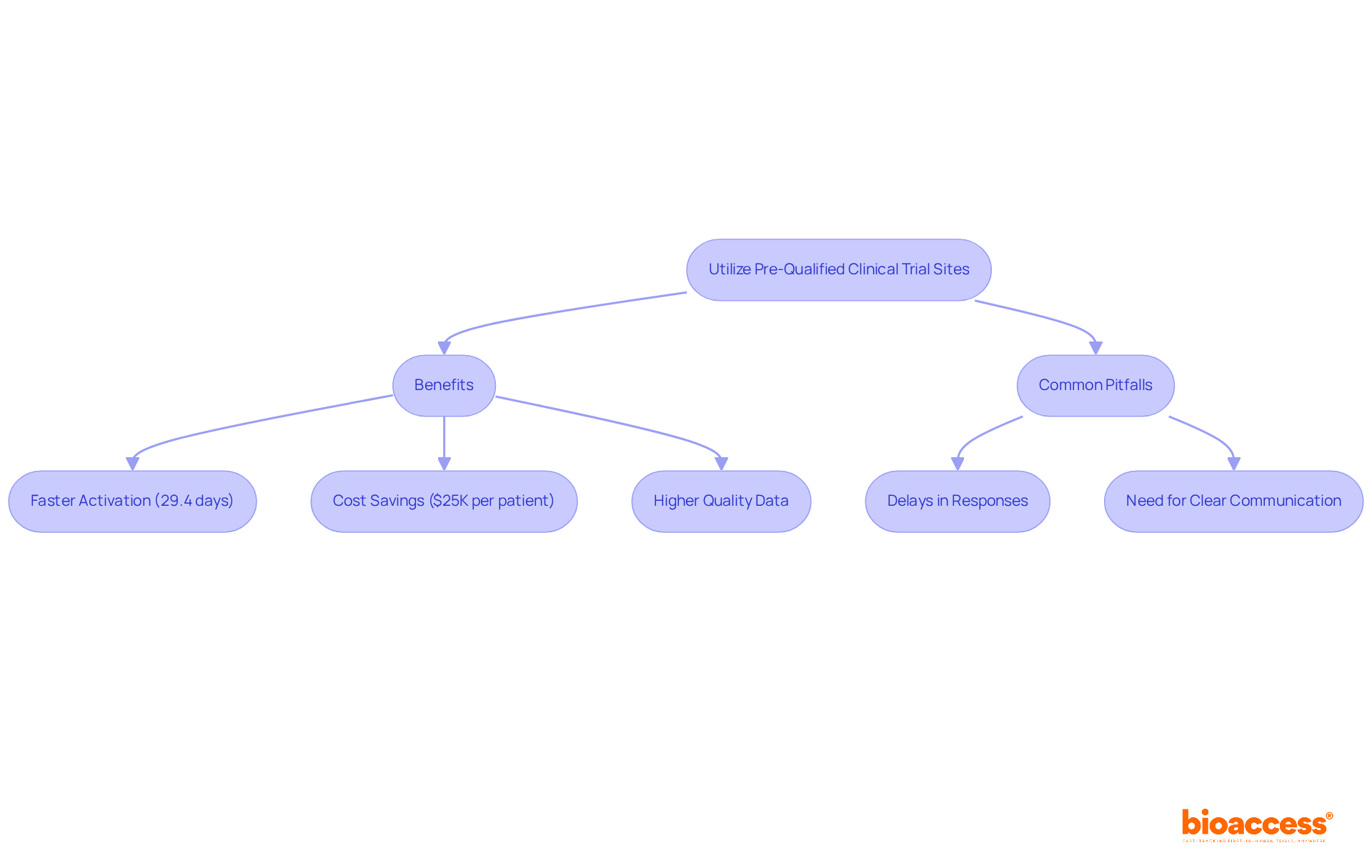
In the competitive landscape of clinical research in Chile, leveraging pre-qualified investigator site Chile clinical trial locations is not just beneficial; it’s essential for success. Utilizing pre-qualified locations allows organizations to significantly cut activation timelines, achieving readiness in about 29.4 days, while non-qualified locations often take much longer.
For instance, bioaccess®’s network of over 50 pre-qualified investigator site Chile clinical trial locations enables swift initiation and execution of studies, allowing sponsors to navigate the regulatory framework more efficiently, particularly with the Instituto de Salud Pública (ISP) managing compliance requirements.
Furthermore, these sites typically possess established patient recruitment strategies and experienced staff, which can lead to faster enrollment and higher quality data. Bioaccess® offers compelling solutions that save $25K per patient and enable enrollment to occur 50% faster through its pre-qualified networks. This strategic choice not only accelerates the clinical development process but also enhances the likelihood of successful outcomes.
However, sponsors must remain vigilant about common pitfalls, including:
- Delays in responses
- The necessity for clear communication with site staff to align on trial objectives, crucial for regulatory compliance.
Conclusion
Selecting the right investigator sites in Chile is not just important; it’s a decisive factor that can make or break first-in-human (FIH) studies. Understanding the regulatory landscape and evaluating site expertise are just the beginning. By fostering relationships with local investigators and utilizing pre-qualified sites, sponsors can ensure effective and efficient research outcomes. These factors are crucial when it comes to navigating the complexities of clinical trials in Chile, establishing the country as a prime location for innovative medical research.
The article emphasizes the importance of mastering Chile’s regulatory framework, which allows for expedited approval processes compared to other regions. Additionally, the significance of selecting urban sites with experienced personnel cannot be overstated, as these factors enhance patient recruitment and streamline compliance with ICH-GCP standards. Building strong local investigator relationships further enriches the study design and execution, ensuring alignment with cultural and regulatory expectations. Utilizing pre-qualified sites accelerates timelines and reduces costs, making them an essential part of a successful strategy.
In conclusion, leveraging the unique advantages of Chile’s clinical research environment is essential for sponsors looking to conduct FIH trials. Implementing best practices in site selection helps organizations avoid common pitfalls and paves the way for groundbreaking advancements in patient care. Ultimately, the right strategies can transform challenges into opportunities, positioning sponsors at the forefront of medical innovation in Latin America.
Frequently Asked Questions
What is the role of the Instituto de Salud Pública (ISP) in Chile’s clinical research?
The Instituto de Salud Pública (ISP) oversees the approval process for clinical research projects in Chile, making it essential for researchers to navigate this regulatory framework effectively.
What key regulation governs human research in Chile?
Law No. 20.120 outlines the requirements for conducting human research in Chile and mandates that ethical committee approvals are obtained before proceeding with studies.
How long does it typically take to receive approval for clinical studies in Chile?
Clinical studies in Chile typically receive approval in about 30 days, which is considered efficient compared to other regions.
What compliance standards must sponsors adhere to for FDA acceptance of study data?
Sponsors must ensure compliance with International Council for Harmonisation Good Clinical Practice (ICH-GCP) standards to facilitate FDA acceptance of data generated during studies.
How can mastering Chile’s regulatory landscape benefit researchers?
By mastering the regulatory landscape in Chile, researchers can streamline their processes, enhance their chances of success, and establish Chile as a strategic center for clinical research in Latin America.
List of Sources
- Understand the Regulatory Landscape in Chile
- Navigate Clinical Trial Approval in Chile: 4 Essential Steps | bioaccess® (https://bioaccessla.com/blog/navigate-clinical-trial-approval-in-chile-4-essential-steps)
- Clinical Trials in Latin America (https://languageconnections.com/clinical-trials-in-latin-america)
- How to Conduct First-in-Human Trials in Chile: A Step-by-Step Guide | bioaccess® (https://bioaccessla.com/blog/how-to-conduct-first-in-human-trials-in-chile-a-step-by-step-guide)
- 4 Best Practices for First in Human Biopharma Trials in Chile | bioaccess® (https://bioaccessla.com/blog/4-best-practices-for-first-in-human-biopharma-trials-in-chile)
- Evaluate Site Expertise and Geographic Location
- Clinical trials: Investment in Chile grew by 62% in three years (https://blog.investchile.gob.cl/clinical-trials-investment-in-chile)
- Clinical trials by start date Chile 1999-2024| Statista (https://statista.com/statistics/1560150/chile-number-new-clinical-trials?srsltid=AfmBOorAtaWb4DJ2kU5zMZM5mmVUIDc3D5viq7Qpne_1W-SXBbgyC1P1)
- Clinical Trials Quotes (2 quotes) (https://goodreads.com/quotes/tag/clinical-trials)
- TOP 10 CLINICAL TRIALS QUOTES | A-Z Quotes (https://azquotes.com/quotes/topics/clinical-trials.html)
- Considerations for an urban health perspective in Chile from the “Quiero Mi Barrio” program – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10118418)
- Foster Relationships with Local Investigators
- Tips For Clinical Trial Sponsors To Cultivate Meaningful Relationships With Sites (https://clinicalleader.com/doc/tips-for-clinical-trial-sponsors-to-cultivate-meaningful-relationships-with-sites-0001)
- The Importance of Site Relationships in Clinical Research (https://catawbaresearch.com/importance-of-site-relationships-in-clinical-research)
- Do investigator meetings improve recruitment rates in clinical trials? A retrospective before-and-after study of data from nine multi-centre clinical trials – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC7288550)
- Investigator Engagement: The Key to Solving the Clinical Trial Enrollment Crisis | Applied Clinical Trials Online (https://appliedclinicaltrialsonline.com/view/investigator-engagement-key-solving-clinical-trial-enrollment-crisis)
- It’s Still All About Relationships: Why Sponsors, CROs, Patients, and Study Sites Are Key to Improving Performance (https://worldwide.com/blog/2020/08/its-still-all-about-relationships-why-sponsors-cros-patients-and-study-sites-are-key-to-improving-performance)
- Utilize Pre-Qualified Clinical Trial Sites
- Master Early Phase Clinical Trials in Chile: Key Strategies and Insights | bioaccess® (https://bioaccessla.com/blog/master-early-phase-clinical-trials-in-chile-key-strategies-and-insights)
- Benchmarking the Investigative Site Qualification Process | Applied Clinical Trials Online (https://appliedclinicaltrialsonline.com/view/benchmarking-the-investigative-site-qualification-process)
- Clinical Trials Quotes (2 quotes) (https://goodreads.com/quotes/tag/clinical-trials)
- 4 Best Practices for First in Human Biopharma Trials in Chile | bioaccess® (https://bioaccessla.com/blog/4-best-practices-for-first-in-human-biopharma-trials-in-chile)