Introduction
While Chile presents unique advantages for MedTech clinical trials, the complexities of navigating its landscape can pose significant challenges. By understanding the regulatory frameworks and leveraging local expertise, sponsors can streamline processes and reduce costs, making Chile an attractive destination for clinical research. Despite the advantages, sponsors often struggle with patient recruitment and trial execution. Addressing these challenges is crucial for sponsors aiming to thrive in a competitive clinical research environment.
Understand Regulatory Frameworks for MedTech Trials in Chile
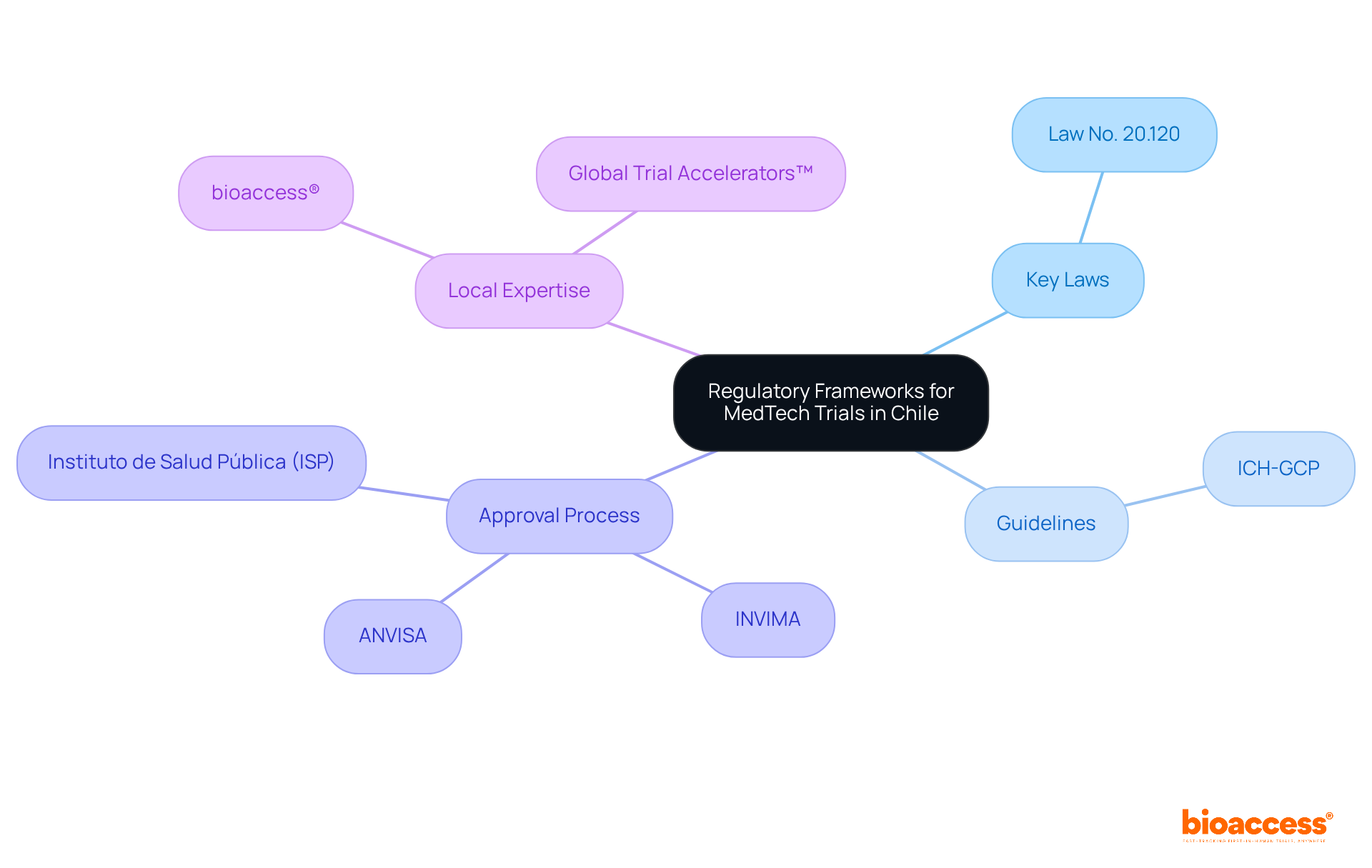
Navigating the regulatory landscape for medtech clinical trial Chile is crucial for the success of clinical trials. Chile’s framework for MedTech research activities is governed by several key laws and guidelines, including Law No. 20.120, which outlines the ethical and scientific standards for human research. Adhering to the International Council for Harmonisation Good Clinical Practice (ICH-GCP) is essential. It ensures that studies are conducted ethically and with scientific rigor. The Instituto de Salud Pública (ISP) oversees the approval process, which typically takes about 30 days, allowing for rapid initiation of studies. Moreover, governing bodies like INVIMA and ANVISA play essential roles in the approval processes for studies in the region.
Understanding these regulations facilitates smoother study execution. It also enhances the credibility of the data, making it more acceptable to global regulatory agencies like the FDA. By tapping into local expertise through bioaccess® and the Global Trial Accelerators™, you can speed up the approval process and gain valuable clinical insights. This approach further enhances operational efficiency and market access strategies for MedTech startups in Latin America. By leveraging local expertise, MedTech startups can significantly enhance their chances of success in the medtech clinical trial Chile market.
Implement Early Feasibility Studies to Validate Concepts
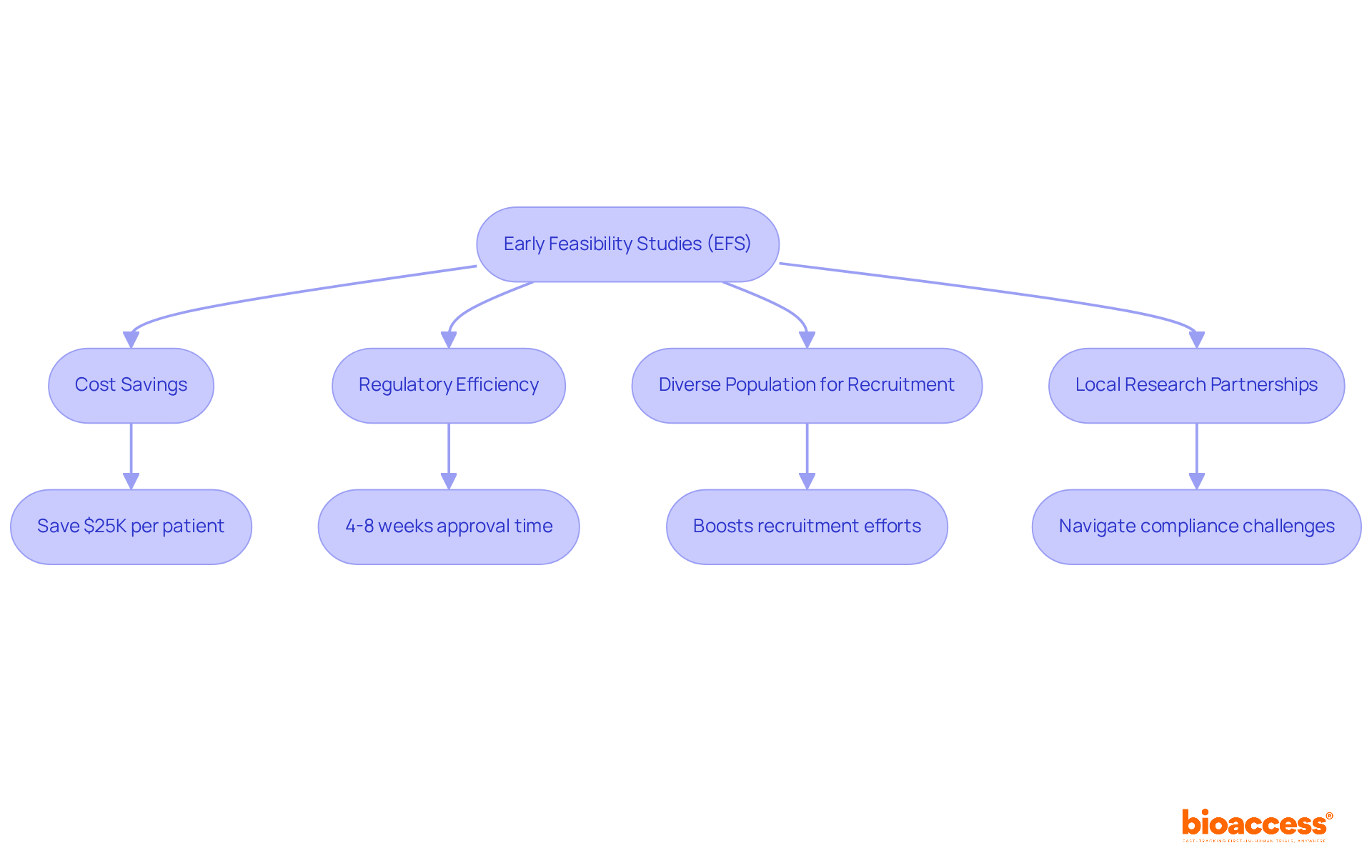
Early feasibility studies (EFS) are a critical step in the MedTech device development process, providing essential insights before larger trials commence. Conducting a medtech clinical trial in Chile presents significant advantages. The regulatory process is streamlined, resulting in an average approval time of just 4 to 8 weeks. This is significantly faster than the six to nine months required in other Latin American countries, such as Brazil and Colombia. Plus, the diverse population in Chile boosts recruitment efforts and enriches the data collected.
Key advantages of conducting EFS in Chile include:
- Cost Savings: Sponsors can save approximately $25K per patient with pre-negotiated site contracts, leading to 30% lower overall trial costs.
- Regulatory Efficiency: The expedited approval process allows for quicker access to medical data, facilitating timely investor meetings or FDA Pre-Submissions.
EFS studies generally involve a small cohort of participants, often ten or fewer, focusing on the initial performance and safety profile of the device. Partnering with local research facilities that understand the compliance environment helps sponsors navigate the challenges of early feasibility studies, ensuring they gather significant data to guide future study designs. This proactive approach not only enhances the quality of medical evidence but also aligns with the rapid pace of MedTech innovation.
Moreover, the FDA’s guidelines for EFS allow for studies to be initiated even when preclinical testing has not provided complete information, facilitating a more flexible and adaptive research environment. This flexibility is crucial for early-stage MedTech companies aiming to validate their concepts swiftly and efficiently. By utilizing Chile’s advantageous regulatory framework and proficient healthcare specialists, along with bioaccess®’s knowledge in expediting studies, sponsors can improve their likelihood of success in early feasibility assessments, ultimately fostering progress in medtech clinical trial Chile. By leveraging Chile’s regulatory advantages and local expertise, sponsors can significantly enhance their chances of success in the competitive MedTech landscape.
Develop Targeted Patient Recruitment Strategies for Success
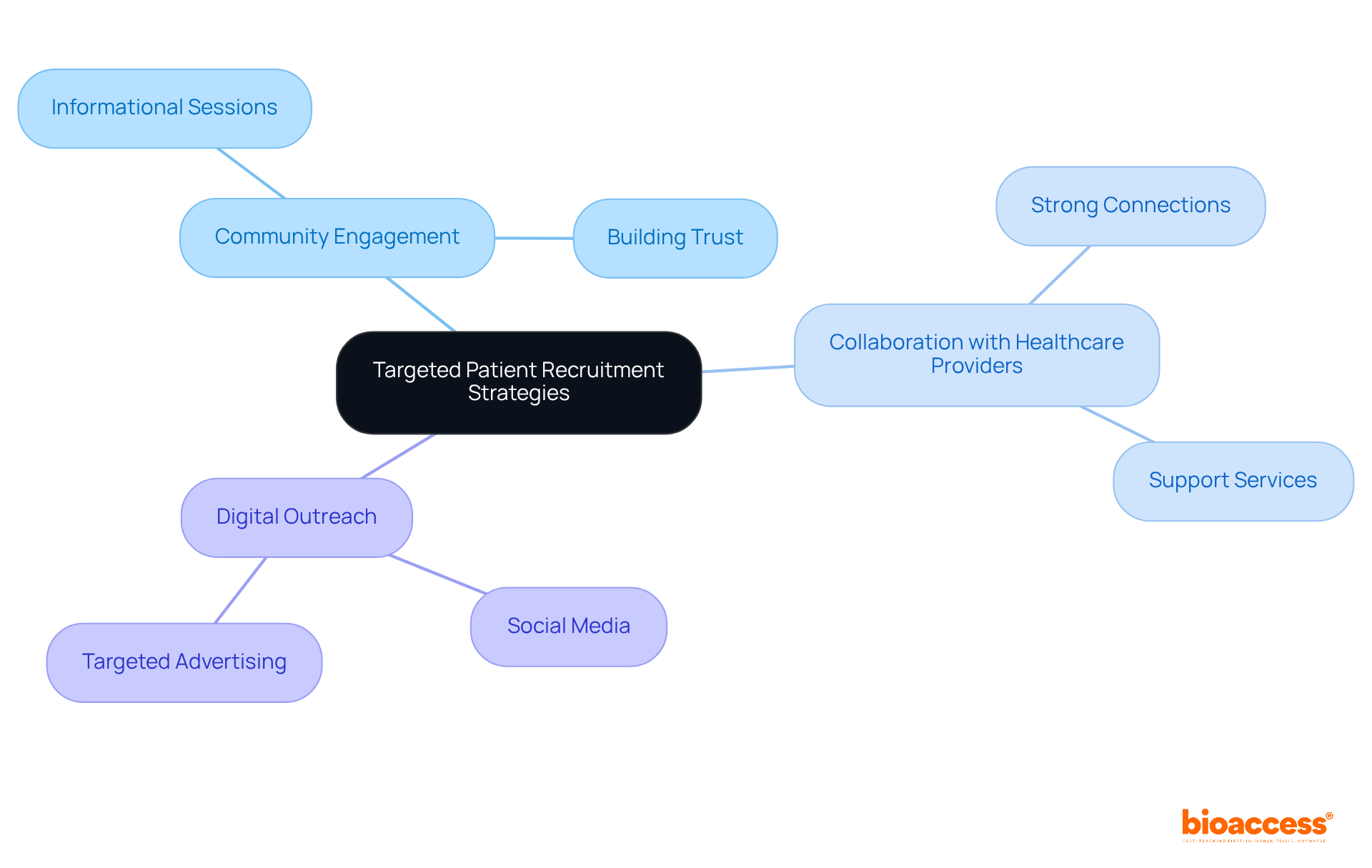
Recruitment challenges often hinder the success of medtech clinical trial Chile, particularly in the dynamic MedTech landscape. Sponsors can enhance recruitment by implementing a multifaceted strategy that focuses on community engagement, collaborating with local healthcare providers, and leveraging digital platforms for outreach. Tailoring these strategies to the demographics and cultural nuances of the local population can significantly boost enrollment rates. For instance, utilizing registries and organizing informational sessions in community centers builds trust and raises awareness about the study. Additionally, employing social media and targeted advertising can effectively reach potential participants.
Have you considered how dedicated support services and proactive communication could help you achieve a retention rate of 95%? Statistics from successful studies in the region suggest this is possible. A Phase III study for a novel radiopharmaceutical in Brazil achieved an impressive 96% retention rate, highlighting the effectiveness of localized support for individuals and community outreach initiatives.
Experts agree that building strong connections with healthcare providers is crucial, as individuals are more inclined to engage when they feel supported and informed. Engaging with local communities not only enhances recruitment but also guarantees a diverse and representative patient population, which is essential for the validity of study outcomes. By prioritizing these strategies, sponsors can not only enhance recruitment but also ensure the integrity and success of their clinical research efforts.
Leverage Local Clinical Trial Sites for Efficient Execution
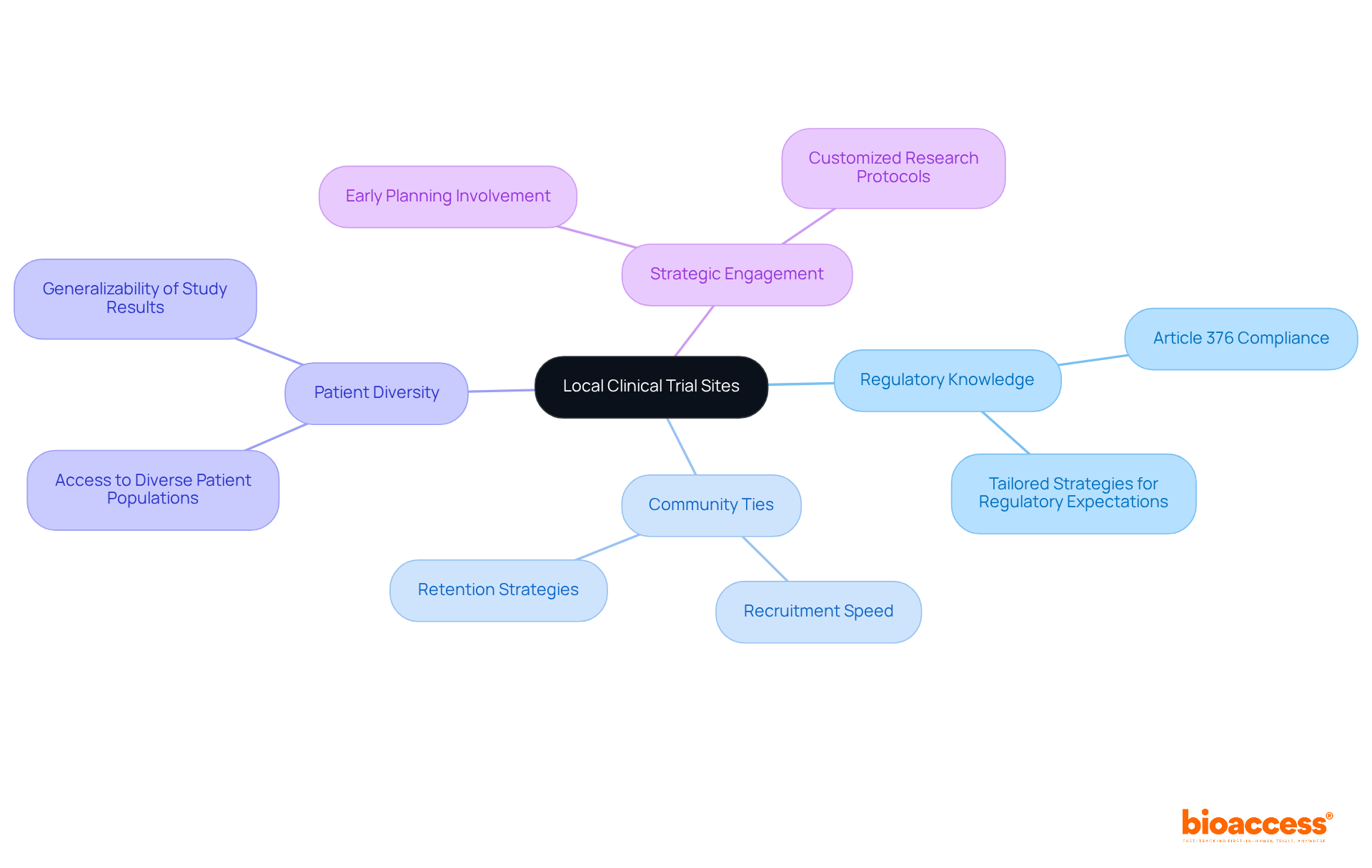
Navigating the complexities of medtech clinical trial Chile demands collaboration with local sites to ensure efficiency and compliance. Local sites are well-versed in regulatory requirements, including Article 376, and have strong community ties that speed up recruitment and retention. Choosing sites with a solid history in similar studies empowers sponsors to navigate local regulations effectively and refine research protocols for the medtech clinical trial Chile.
Moreover, these locations provide access to diverse patient populations, which is essential for the generalizability of study results. Engaging with these sites early in the planning process allows for tailored strategies that align with regulatory expectations from authorities such as INVIMA and COFEPRIS, ultimately leading to a more streamlined execution of studies.
Without these insights, sponsors risk delays that could jeopardize their competitive edge in the rapidly evolving MedTech landscape.
Conclusion
Navigating the complexities of MedTech clinical trials in Chile presents both challenges and remarkable opportunities for innovation. By understanding the regulatory frameworks, sponsors can streamline their processes and enhance the credibility of their studies. The emphasis on early feasibility studies, targeted patient recruitment strategies, and collaboration with local clinical trial sites positions Chile as a strategic advantage in the competitive MedTech landscape. Have you considered how these factors could impact your own trials?
Key insights from the article highlight the importance of adhering to established regulations, such as ICH-GCP and local laws, to ensure ethical and scientifically sound trials. The rapid approval timelines of 30 days for regulatory processes, coupled with significant cost savings and diverse patient populations, make Chile an attractive location for early-stage MedTech trials. Additionally, effective recruitment strategies that engage local communities can lead to improved retention rates and more representative data.
As the MedTech sector continues to evolve, leveraging Chile’s regulatory advantages and local expertise will be crucial for success. This strategic approach not only streamlines processes but also positions sponsors for greater success in the MedTech arena. Now is the moment to capitalize on Chile’s strategic advantages, positioning your clinical trials for success in this dynamic market.
Frequently Asked Questions
What is the regulatory framework for MedTech clinical trials in Chile?
The regulatory framework for MedTech clinical trials in Chile is governed by several key laws and guidelines, including Law No. 20.120, which outlines the ethical and scientific standards for human research. Compliance with the International Council for Harmonisation Good Clinical Practice (ICH-GCP) is also essential.
Who oversees the approval process for MedTech trials in Chile?
The approval process for MedTech trials in Chile is overseen by the Instituto de Salud Pública (ISP). This process typically takes about 30 days, facilitating the rapid initiation of studies.
What are the roles of INVIMA and ANVISA in the context of MedTech trials?
INVIMA and ANVISA are governing bodies that play essential roles in the approval processes for studies in the region, ensuring compliance with local regulations.
How does understanding the regulatory landscape benefit MedTech startups?
Understanding the regulatory landscape facilitates smoother study execution and enhances the credibility of the data, making it more acceptable to global regulatory agencies like the FDA.
What resources can MedTech startups utilize to expedite the approval process in Chile?
MedTech startups can tap into local expertise through bioaccess® and the Global Trial Accelerators™ to speed up the approval process and gain valuable clinical insights.
Why is Latin America considered a strategic advantage for early-stage clinical trials?
Latin America is considered a strategic advantage for early-stage clinical trials due to its speed, cost efficiency, patient recruitment capabilities, and streamlined regulatory pathways, which enhance operational efficiency and market access for MedTech startups.
List of Sources
- Understand Regulatory Frameworks for MedTech Trials in Chile
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Trends in clinical trials performed in Chile (https://scielo.cl/article_plus.php?pid=S0034-98872021000100110&tlng=en&lng=es)
- Clinical trials by start date Chile 1999-2024| Statista (https://statista.com/statistics/1560150/chile-number-new-clinical-trials?srsltid=AfmBOoozGi_4P4QFETF42cQnc6mHGXW2ggkjhiDWQ_9OWTBopTaFFB7Y)
- Navigate First in Human Medical Device Trials in Chile: A Step-by-Step Guide | bioaccess® (https://bioaccessla.com/blog/navigate-first-in-human-medical-device-trials-in-chile-a-step-by-step-guide)
- Master Early Phase Clinical Trials in Chile: Key Strategies and Insights | bioaccess® (https://bioaccessla.com/blog/master-early-phase-clinical-trials-in-chile-key-strategies-and-insights)
- Implement Early Feasibility Studies to Validate Concepts
- Early feasibility studies on devices: “doing it sooner” to avoid trial failure | Meditrial (https://meditrial.net/2022/09/early-feasibility-studies-on-devices-doing-it-sooner-to-avoid-trial-failure)
- How Chile Is Shaping Medical Device Clinical Trials In Latin America (https://meddeviceonline.com/doc/how-chile-is-shaping-medical-device-clinical-trials-in-latin-america-0001)
- Master Early Phase Clinical Trials in Chile: Key Strategies and Insights | bioaccess® (https://bioaccessla.com/blog/master-early-phase-clinical-trials-in-chile-key-strategies-and-insights)
- Develop Targeted Patient Recruitment Strategies for Success
- Patient Recruitment and Retention Services Market Report 2026 (https://researchandmarkets.com/reports/5939968/patient-recruitment-retention-services-market?srsltid=AfmBOooA7aRRS8bN1pKuk5TL3N5y-vSUVQHUep2nr8SSurLJN5My5TJD)
- 5 Key Strategies for Clinical Trial Patient Recruitment (https://proximacro.com/news/5-key-strategies-for-clinical-trial-patient-recruitment)
- Achieving A 95 Patient Retention Rate In LATAM… | bioaccess® (https://bioaccessla.com/blog/achieving-a-95-patient-retention-rate-in-latam-clinical-trials)
- Enrollment in Clinical Trials: Statistics and Patient Recruitment Strategies | Power (https://withpower.com/guides/enrollment-in-clinical-trials-statistics-and-patient-recruitment-strategies)
- Early Adopter of Breast Conservation, Surgical Oncologist Augusto Leon, MD, Reflects on Cancer Care in Chile (https://ascopost.com/issues/july-10-2021/early-adopter-of-breast-conservation-surgical-oncologist-augusto-leon-md-reflects-on-cancer-care-in-chile)
- Leverage Local Clinical Trial Sites for Efficient Execution
- Clinical trials by start date Chile 1999-2024| Statista (https://statista.com/statistics/1560150/chile-number-new-clinical-trials?srsltid=AfmBOoruktLQtHUaq9iIluBHsTG7FN_4w4USL4SbpVAEGLCfFcxiEIB3)
- Chile Clinical Trial Supply & Logistics Market Size & Outlook, 2030 (https://grandviewresearch.com/horizon/outlook/clinical-trial-supply-logistics-market/chile)
- 25+ useful clinical trial recruitment statistics for better results (https://antidote.me/blog/25-useful-clinical-trial-recruitment-statistics-for-better-results)
- Changes in key recruitment performance metrics from 2008–2019 in industry-sponsored phase III clinical trials registered at ClinicalTrials.gov – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC9321424)

Leave a Reply