Introduction
In Chile, the landscape of clinical trial outsourcing offers MedTech and Biopharma companies a strategic advantage to expedite their research efforts. With a regulatory framework that enables swift approvals – often within 30 business days – sponsors can significantly enhance their operational efficiency and reduce costs. Yet, selecting the right trial sites and implementing effective patient recruitment strategies that meet local regulations and community needs can be challenging. What strategies can sponsors adopt to navigate these advantages and tackle the challenges in this evolving landscape?
Understand the Regulatory Landscape for Clinical Trials in Chile
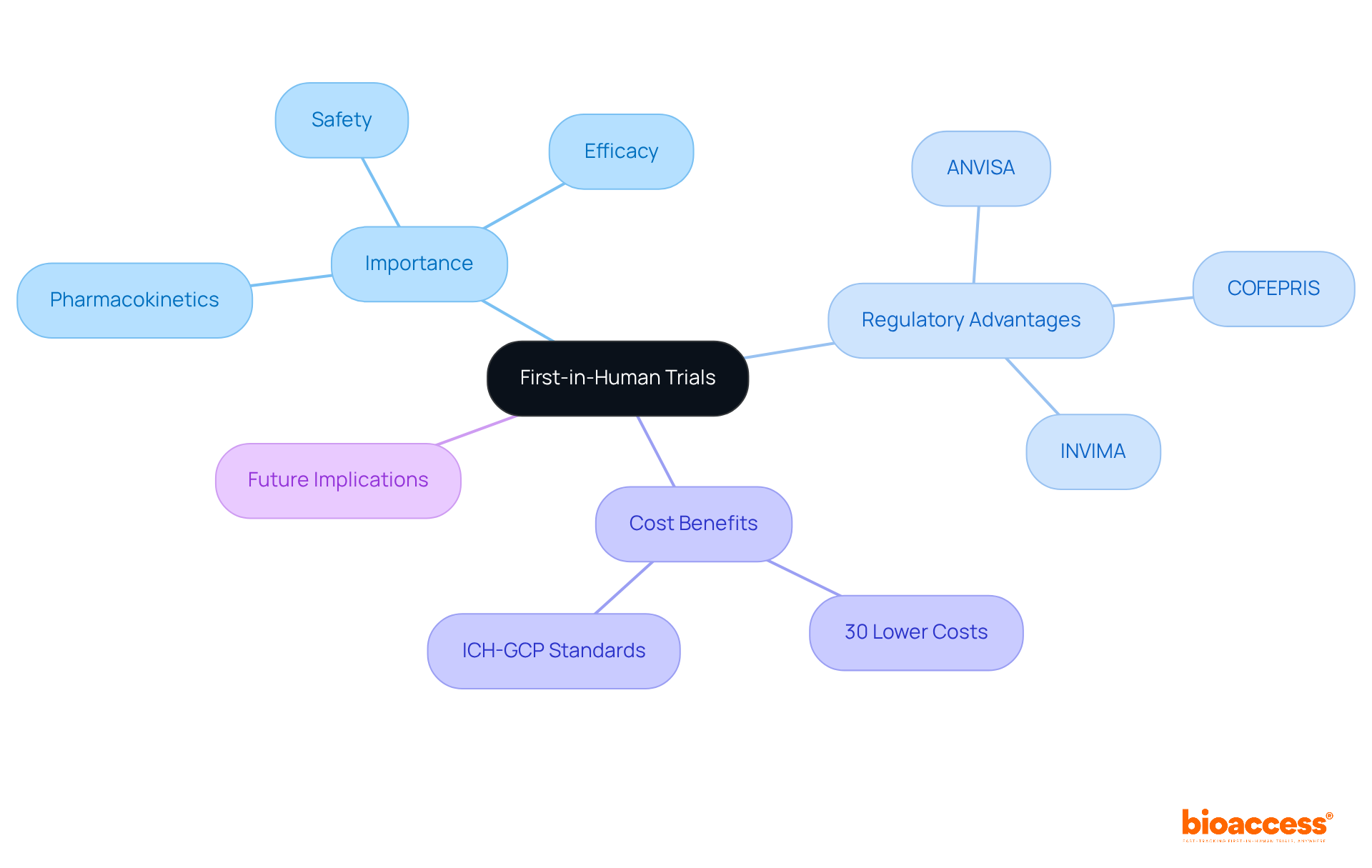
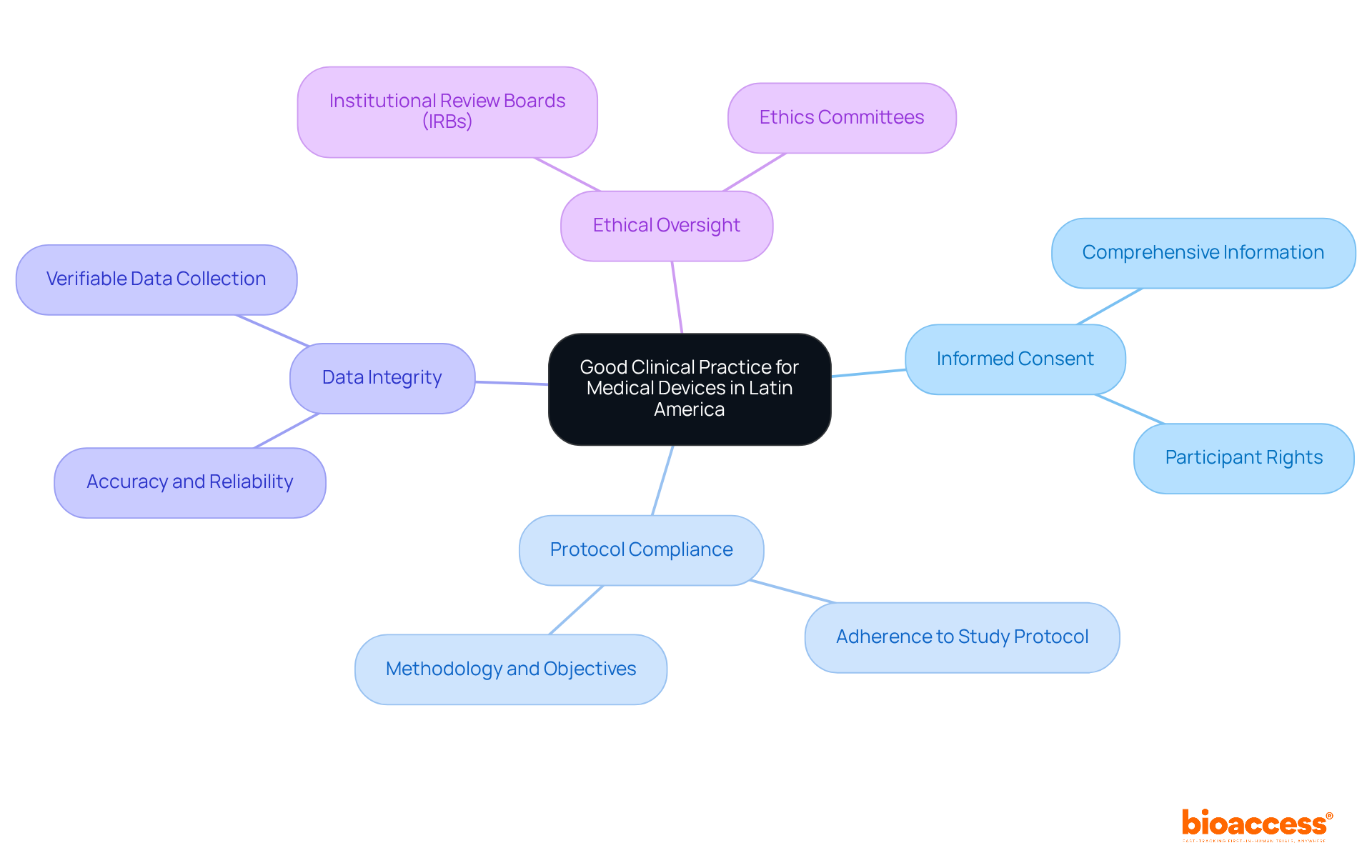
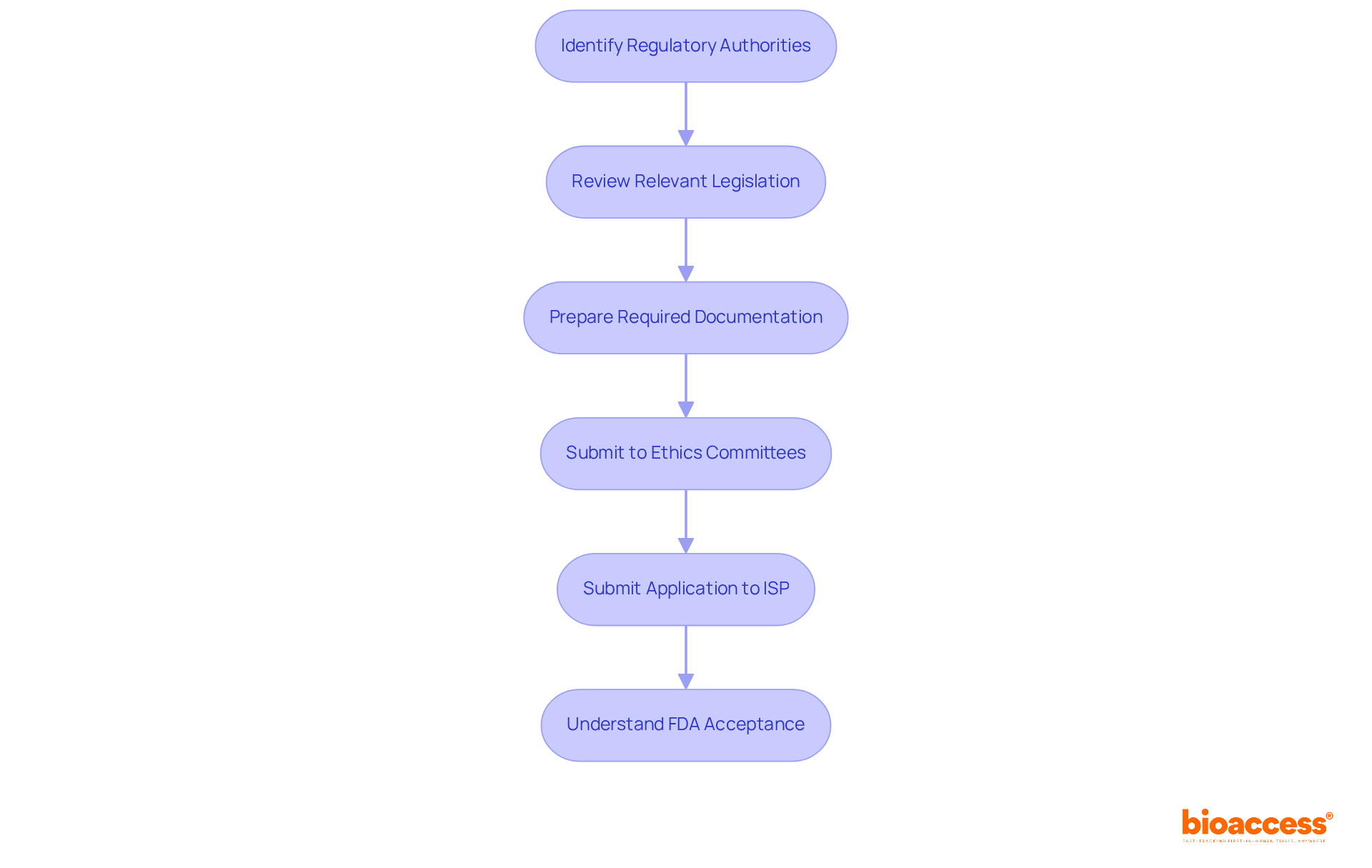
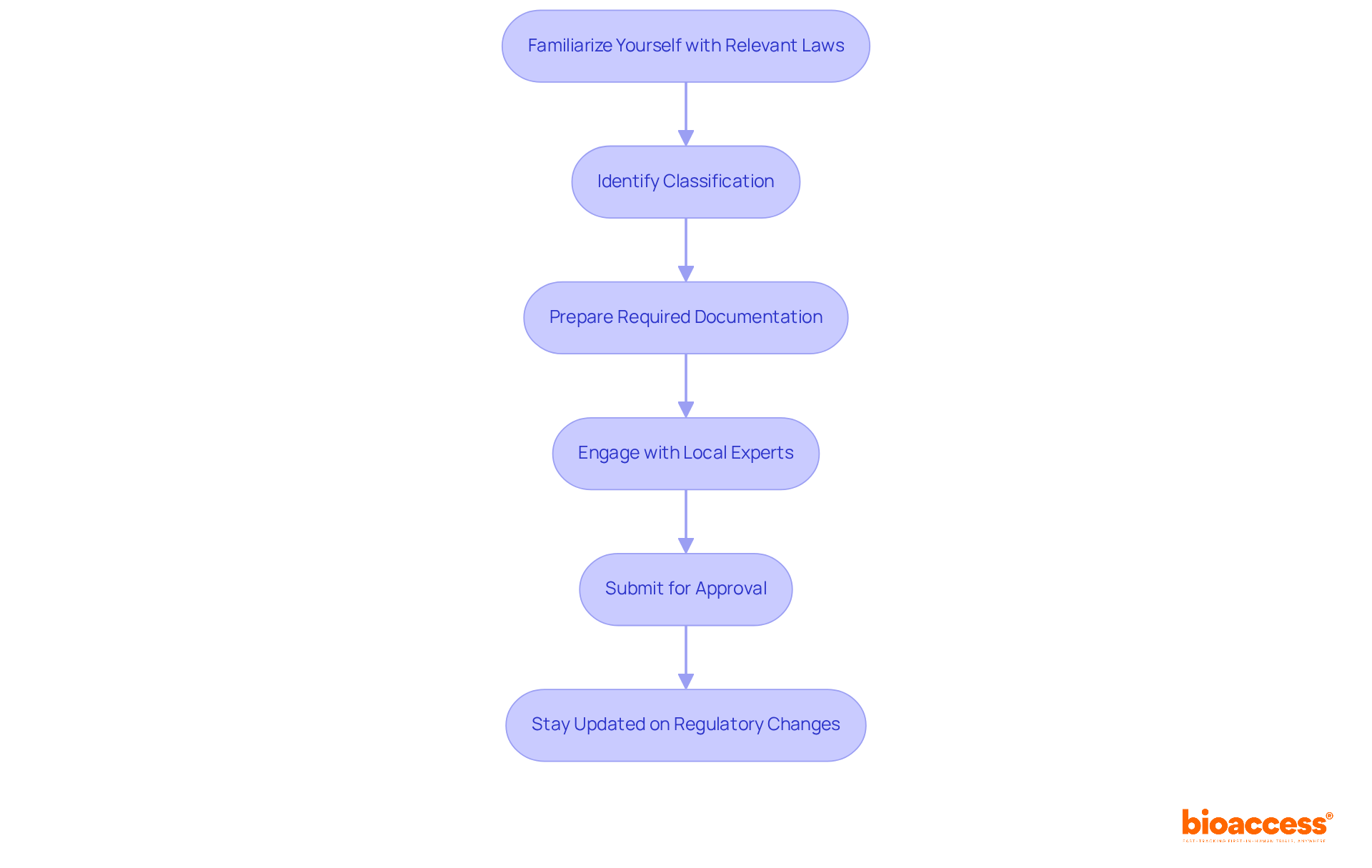
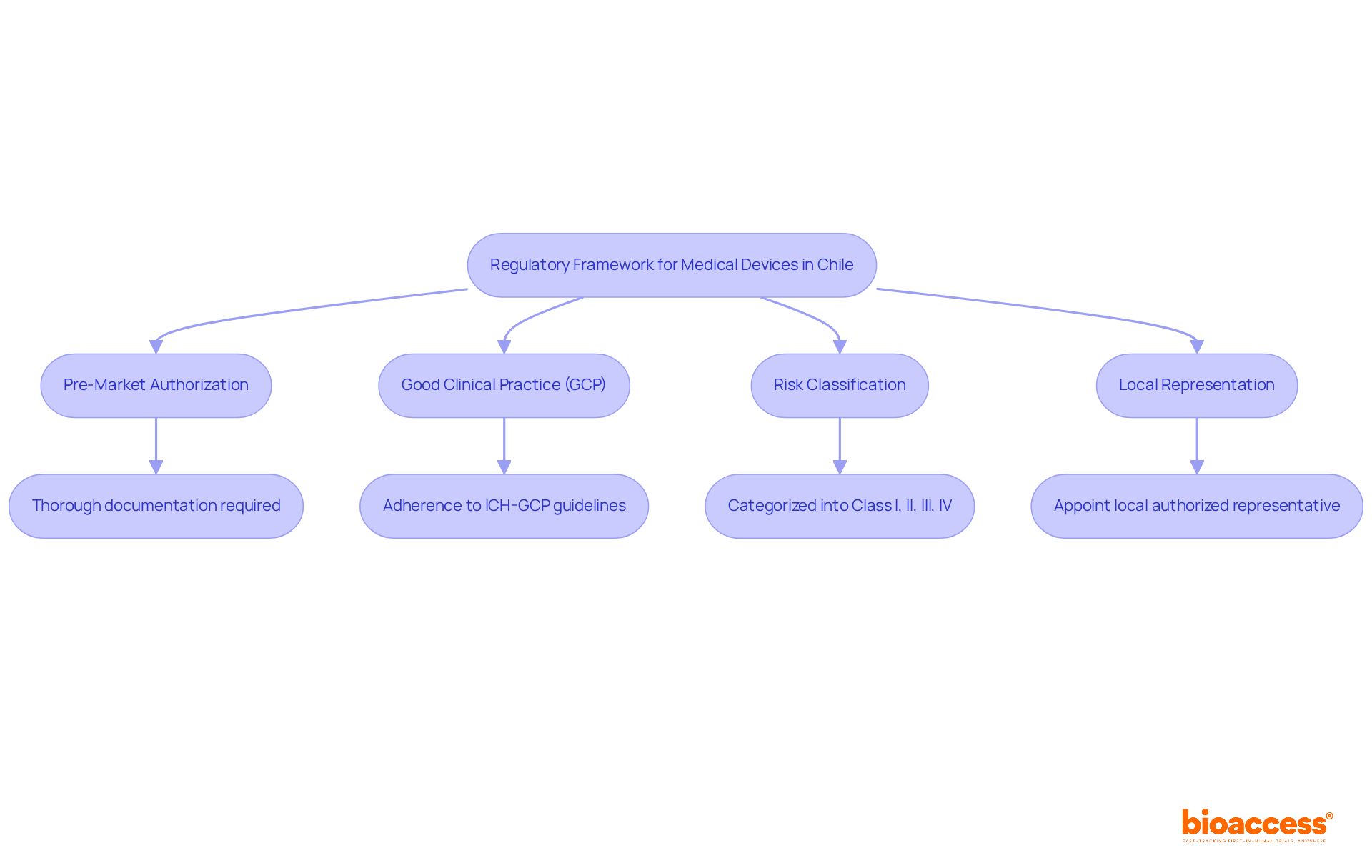
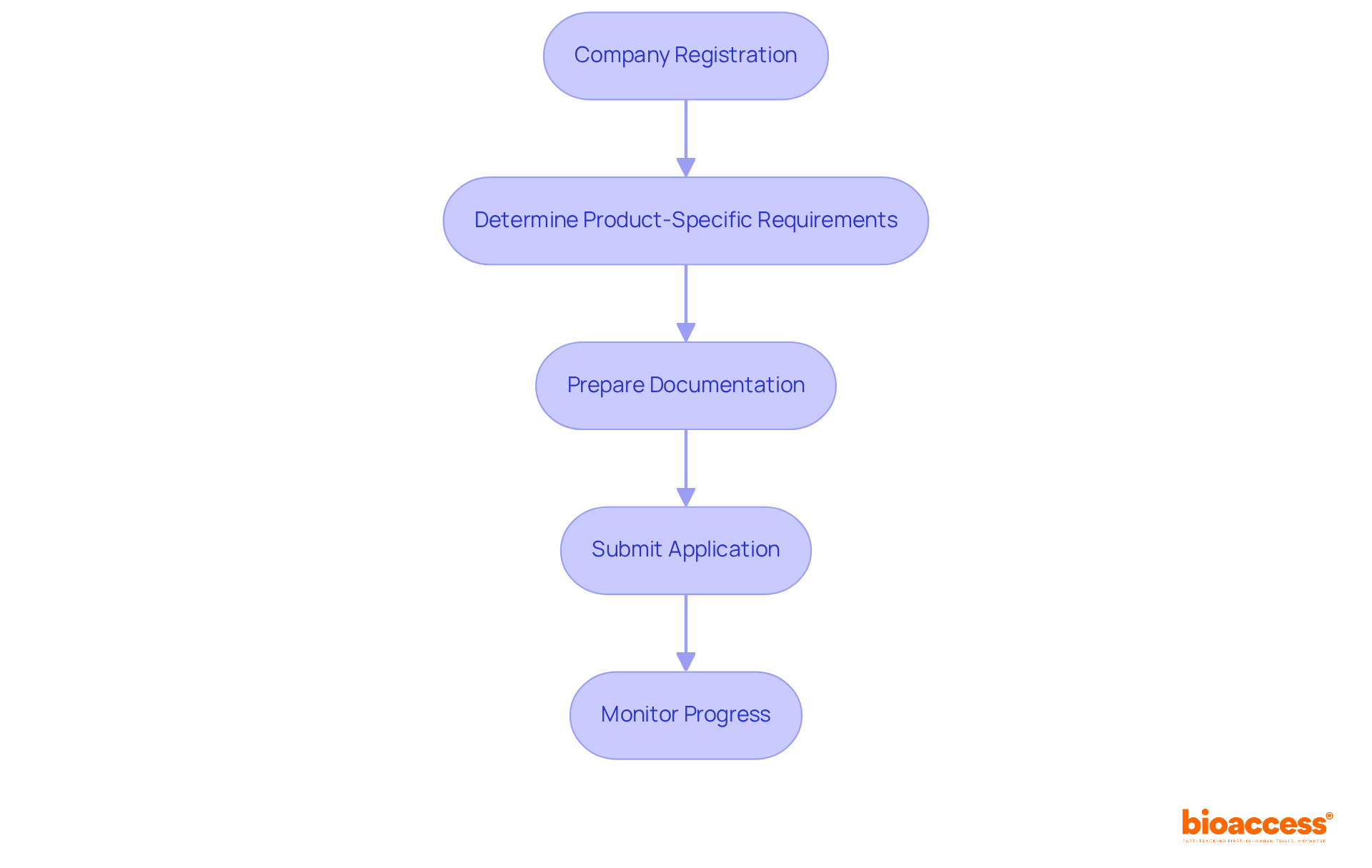
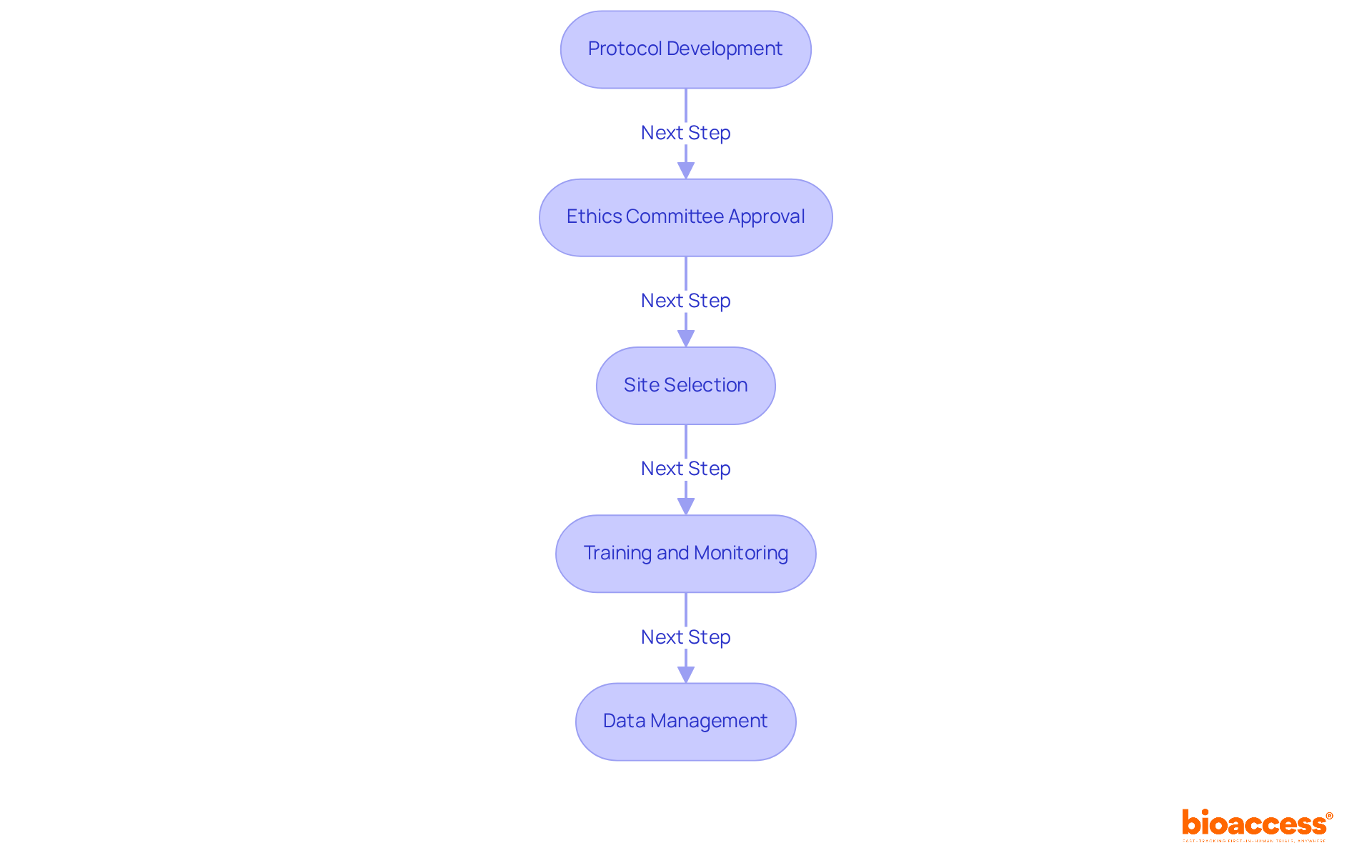
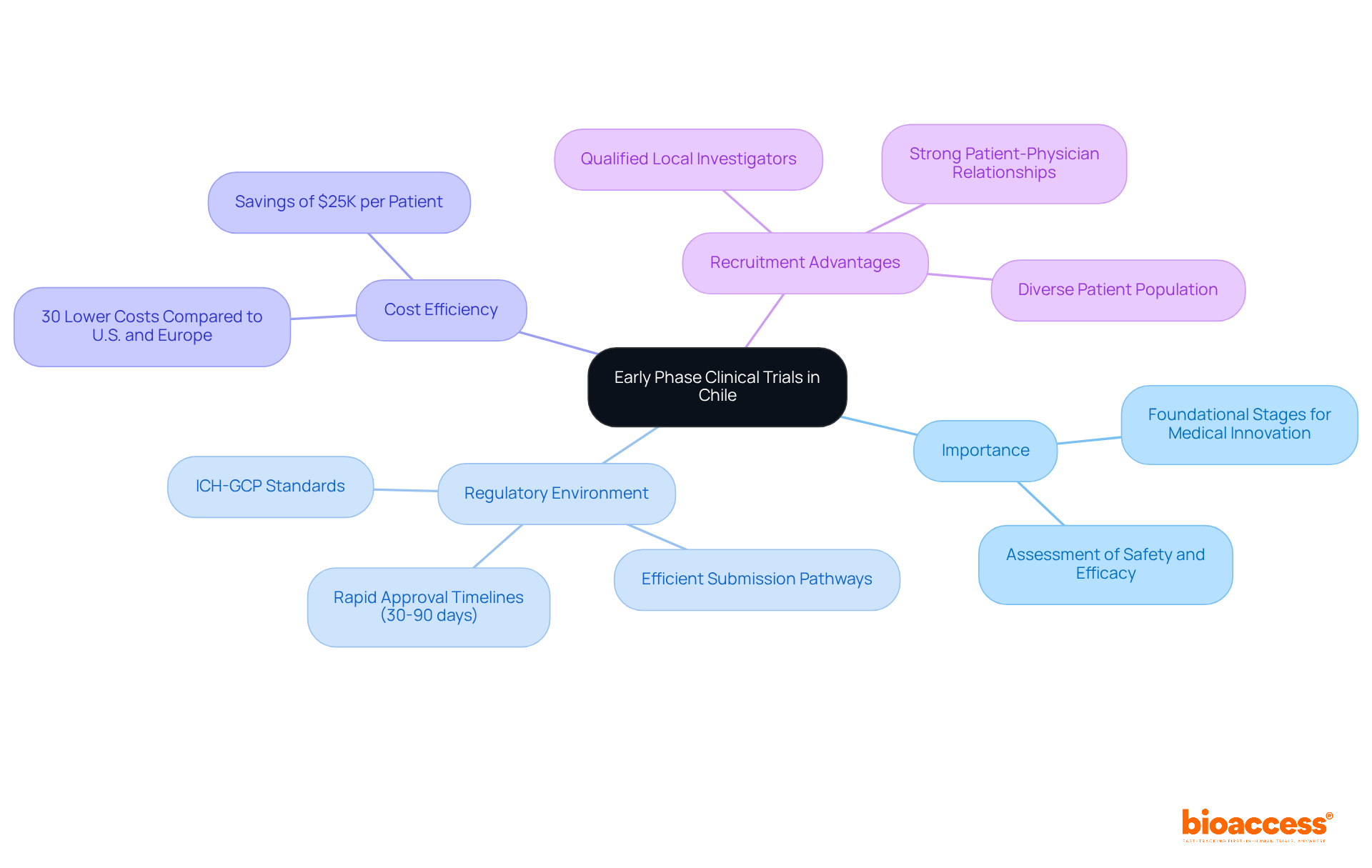
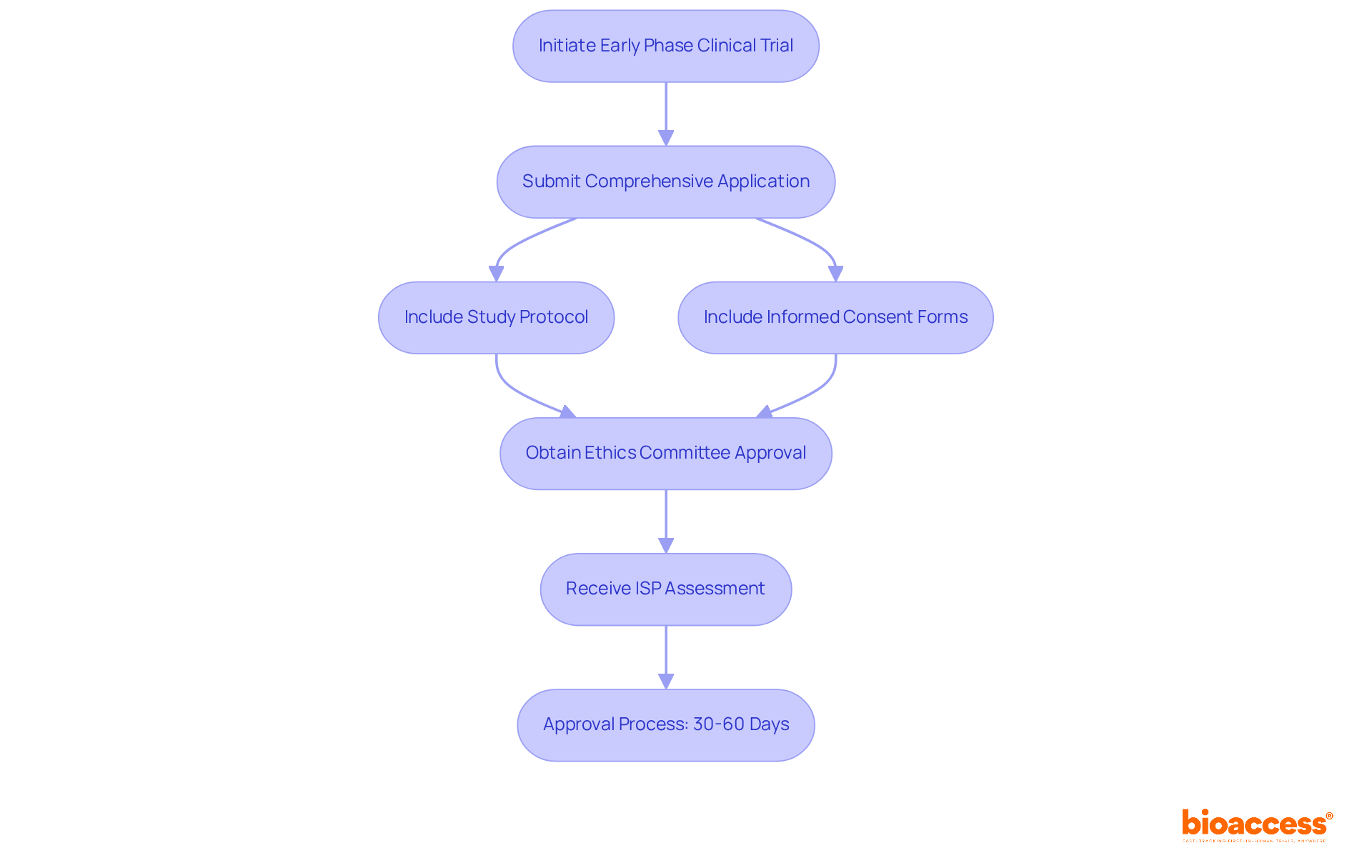
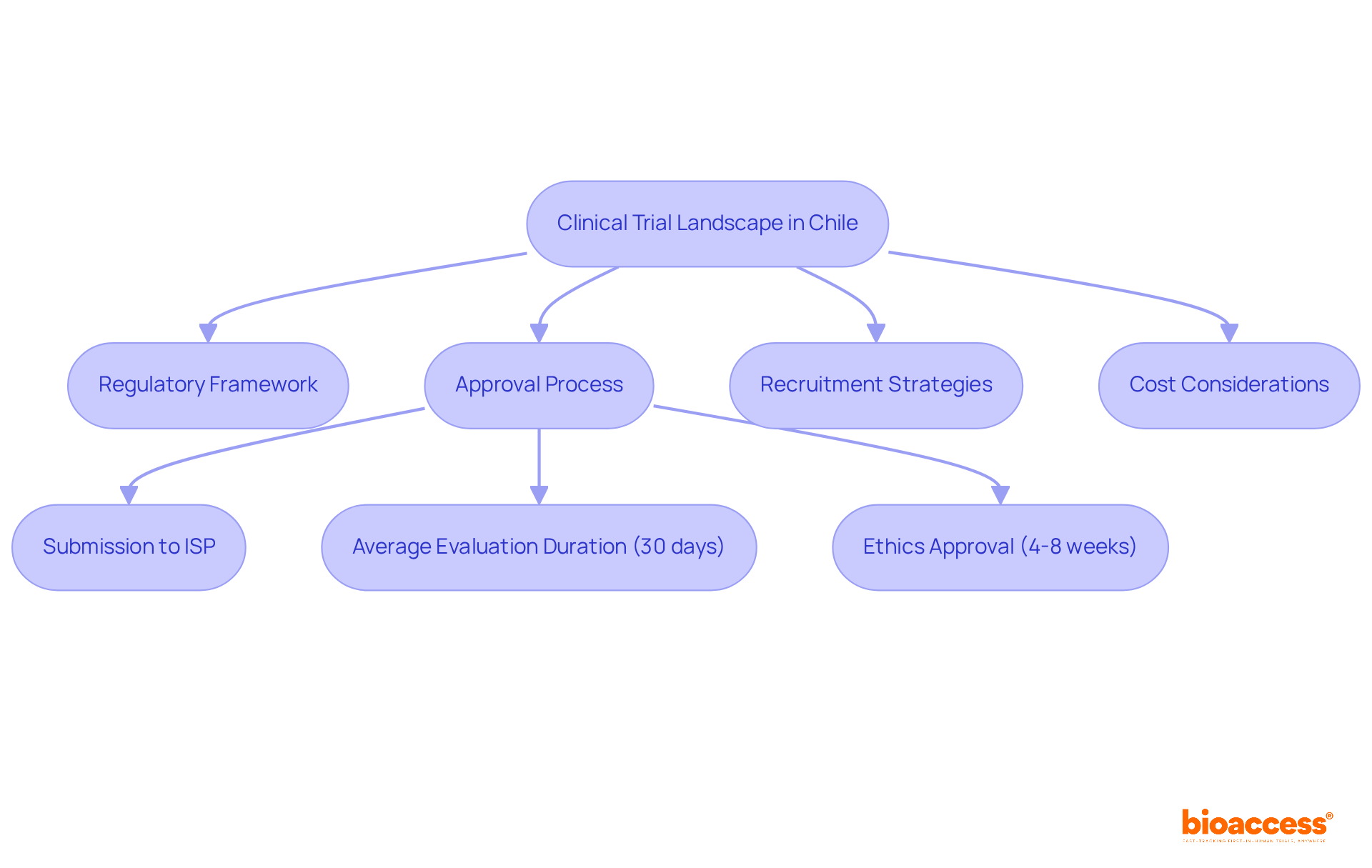
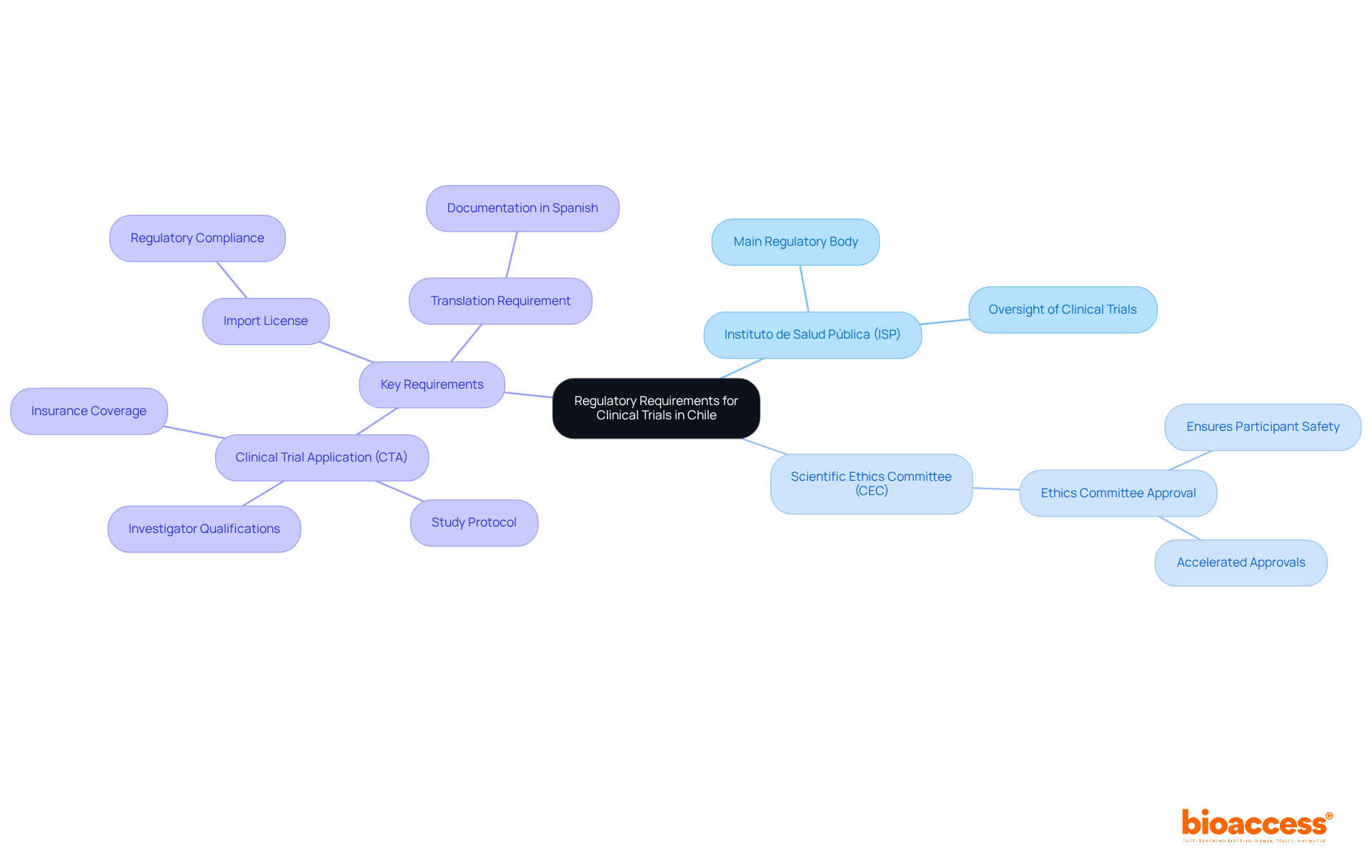
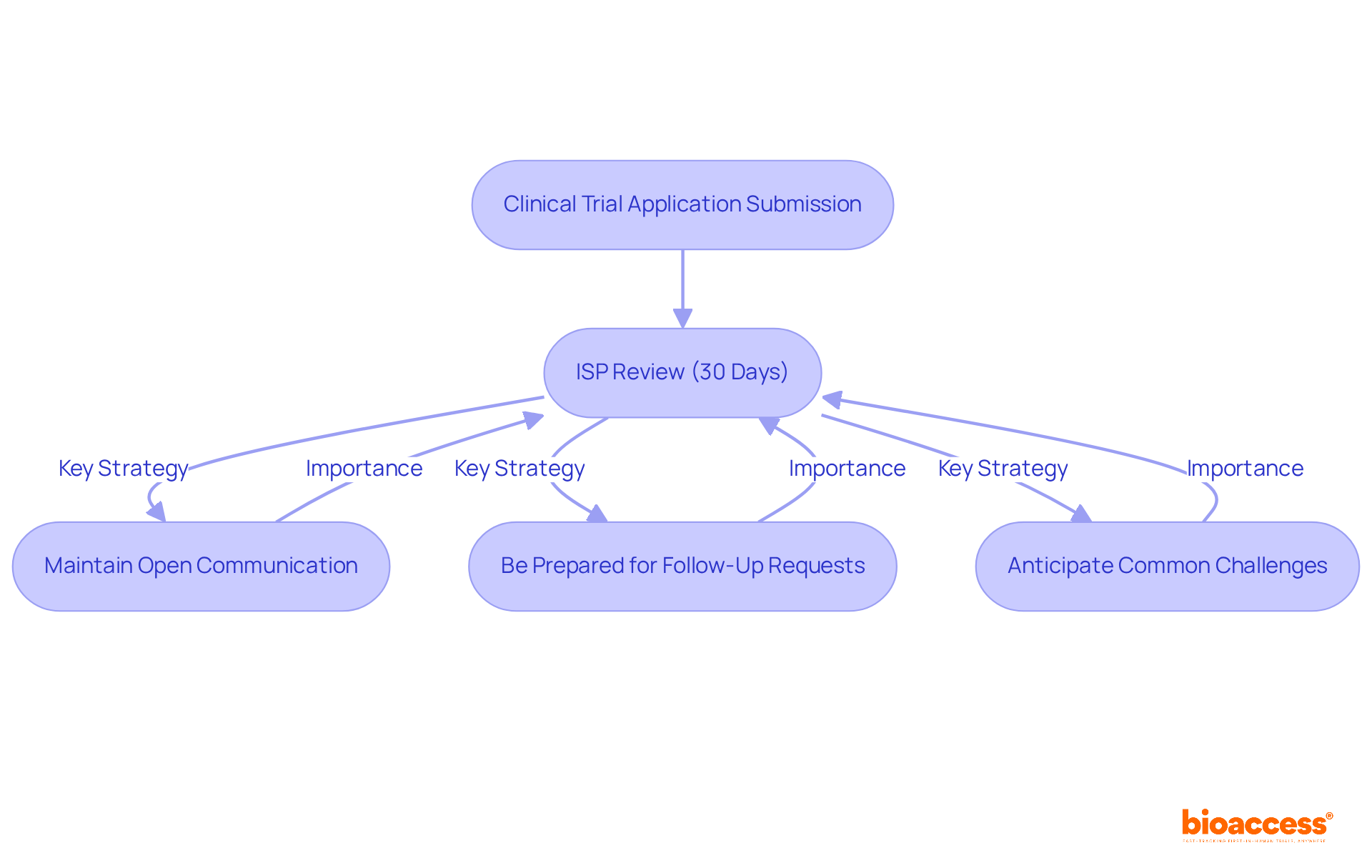
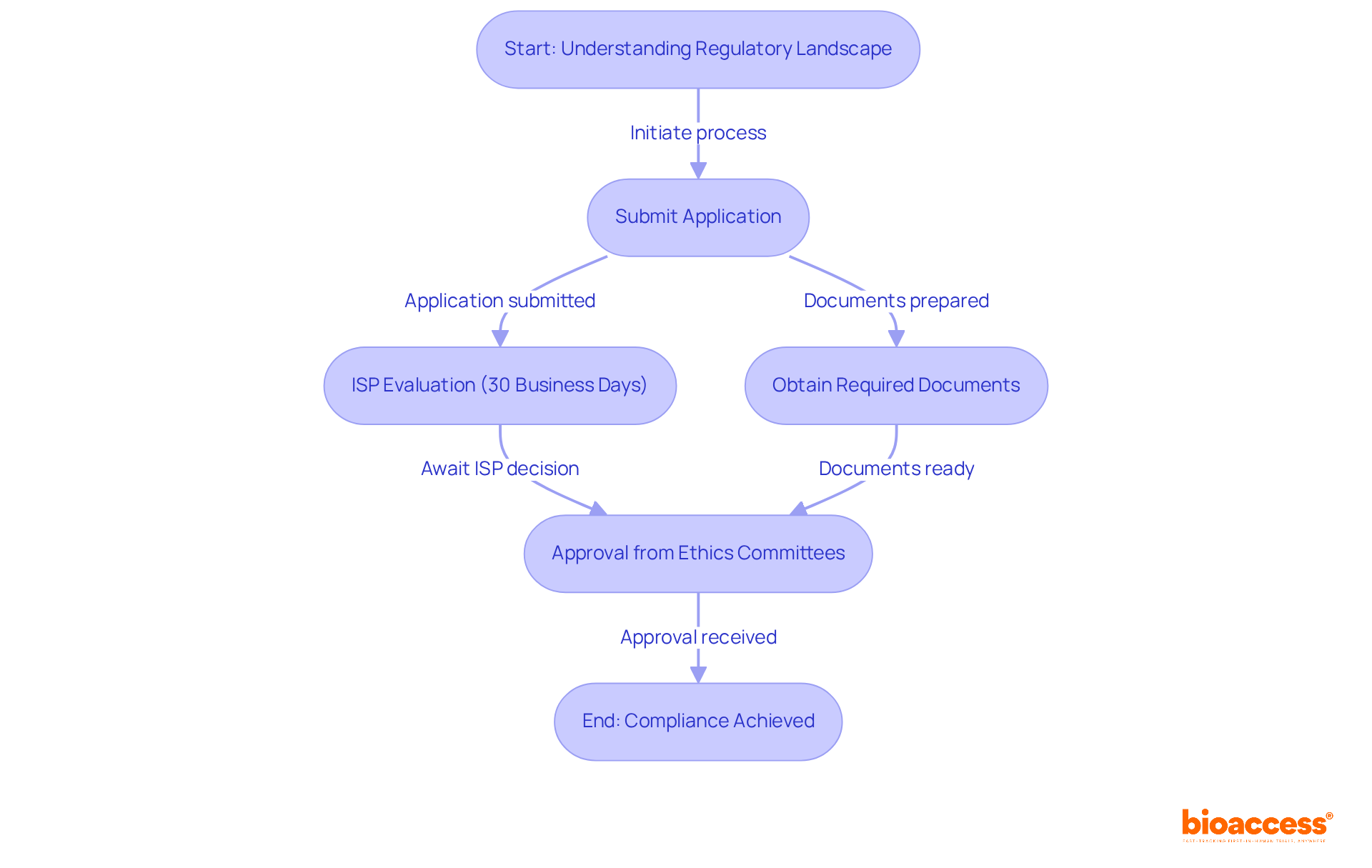
Navigating Chile’s regulatory landscape is critical for sponsors aiming to conduct clinical research effectively. The Instituto de Salud Pública (ISP) oversees the regulatory framework for research studies, ensuring adherence to national health regulations. For sponsors intending to carry out first-in-human (FIH) studies, understanding the specific requirements for research application submissions is non-negotiable. The ISP generally evaluates applications within 30 business days, establishing Chile as one of the quicker regions in Latin America for clinical study approvals.
Key documents required for submission include:
- The study protocol
- Informed consent forms
- Qualifications of the investigators
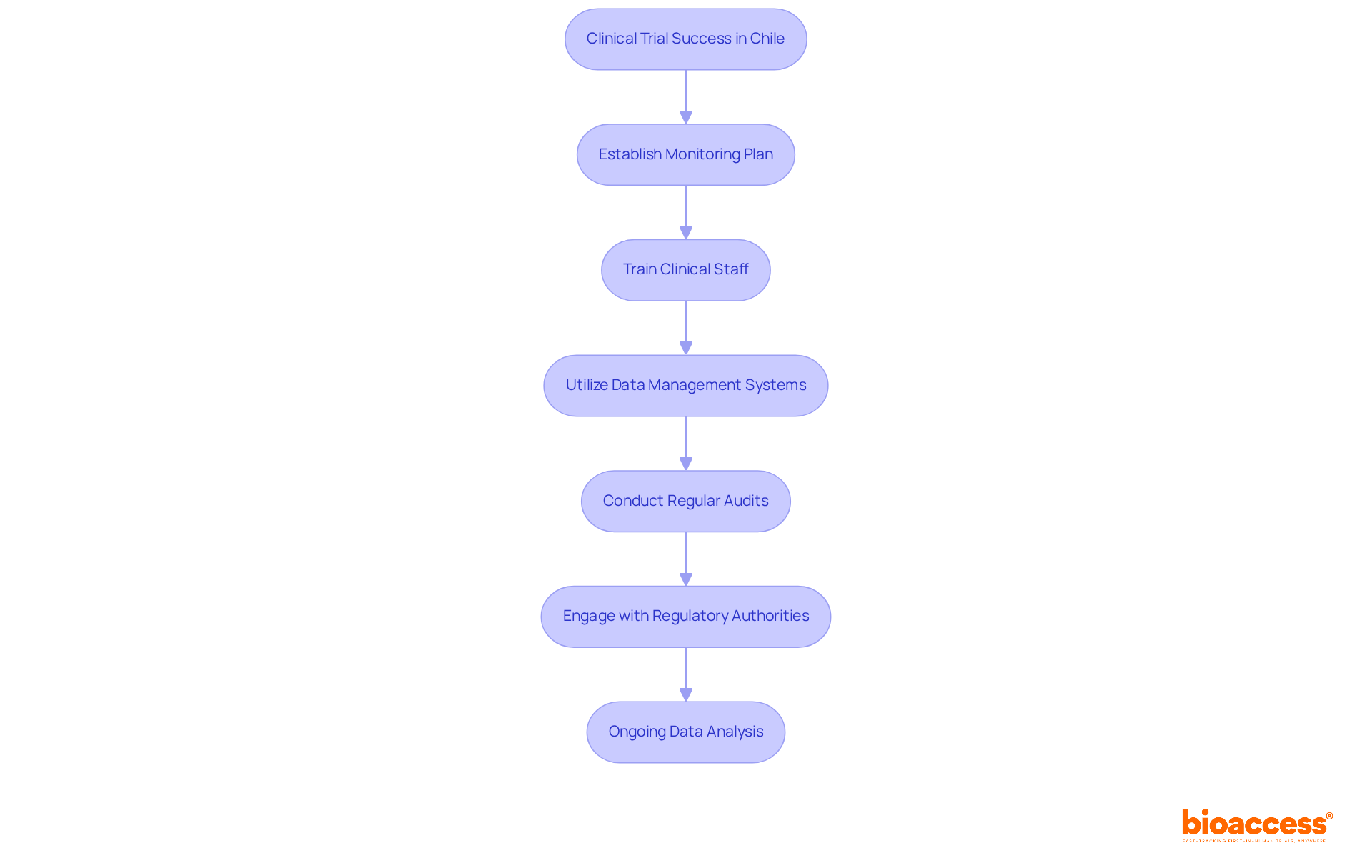
Compliance with ICH-GCP standards is essential, guaranteeing that studies are carried out ethically and that data integrity is maintained. Furthermore, obtaining approval from regional ethics committees is necessary and can influence overall timelines. Familiarizing themselves with these regulatory nuances empowers sponsors to streamline their study initiation processes and sidestep common pitfalls that lead to delays. This understanding can be the difference between a successful trial and a costly delay.
Select Optimal Clinical Trial Sites for Enhanced Efficiency
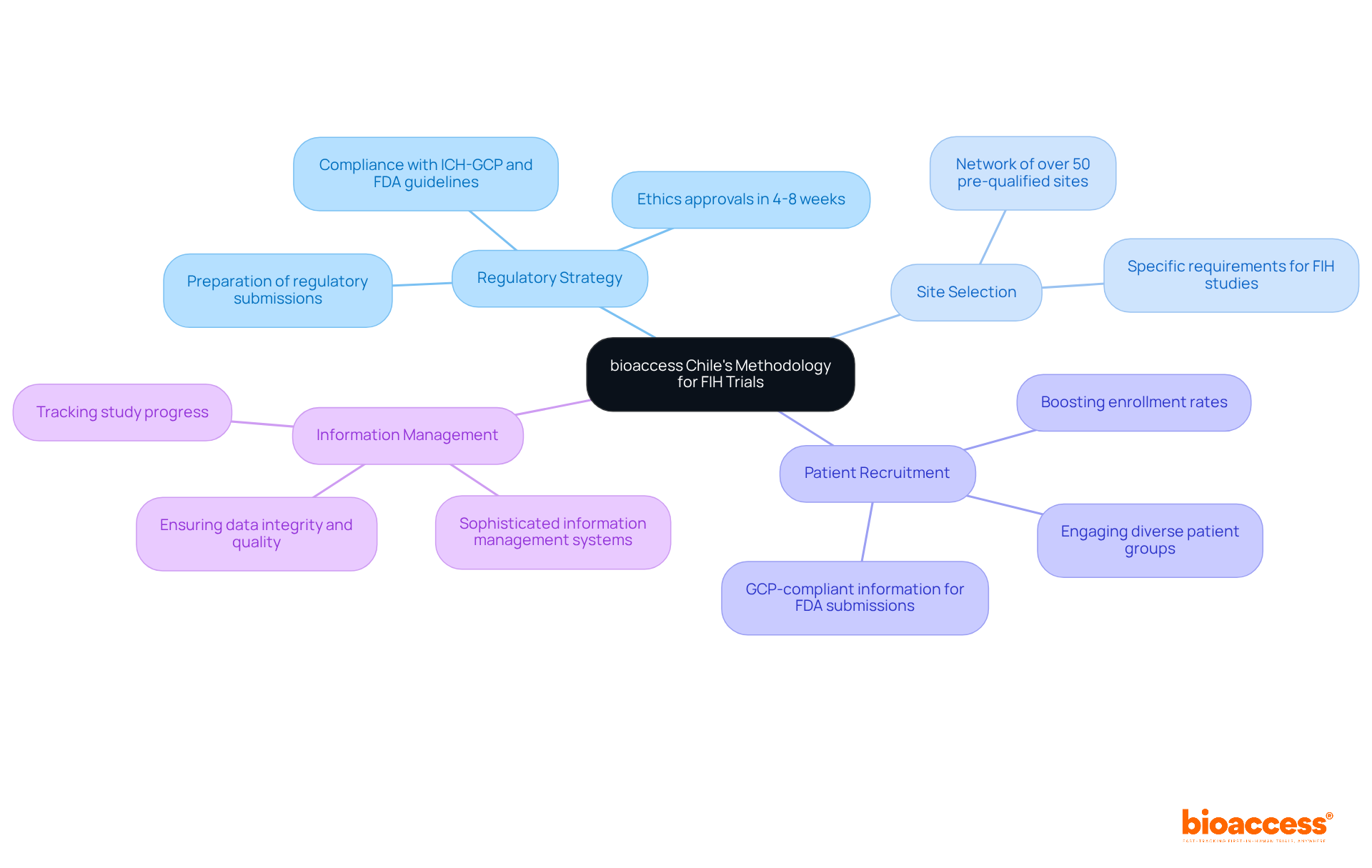
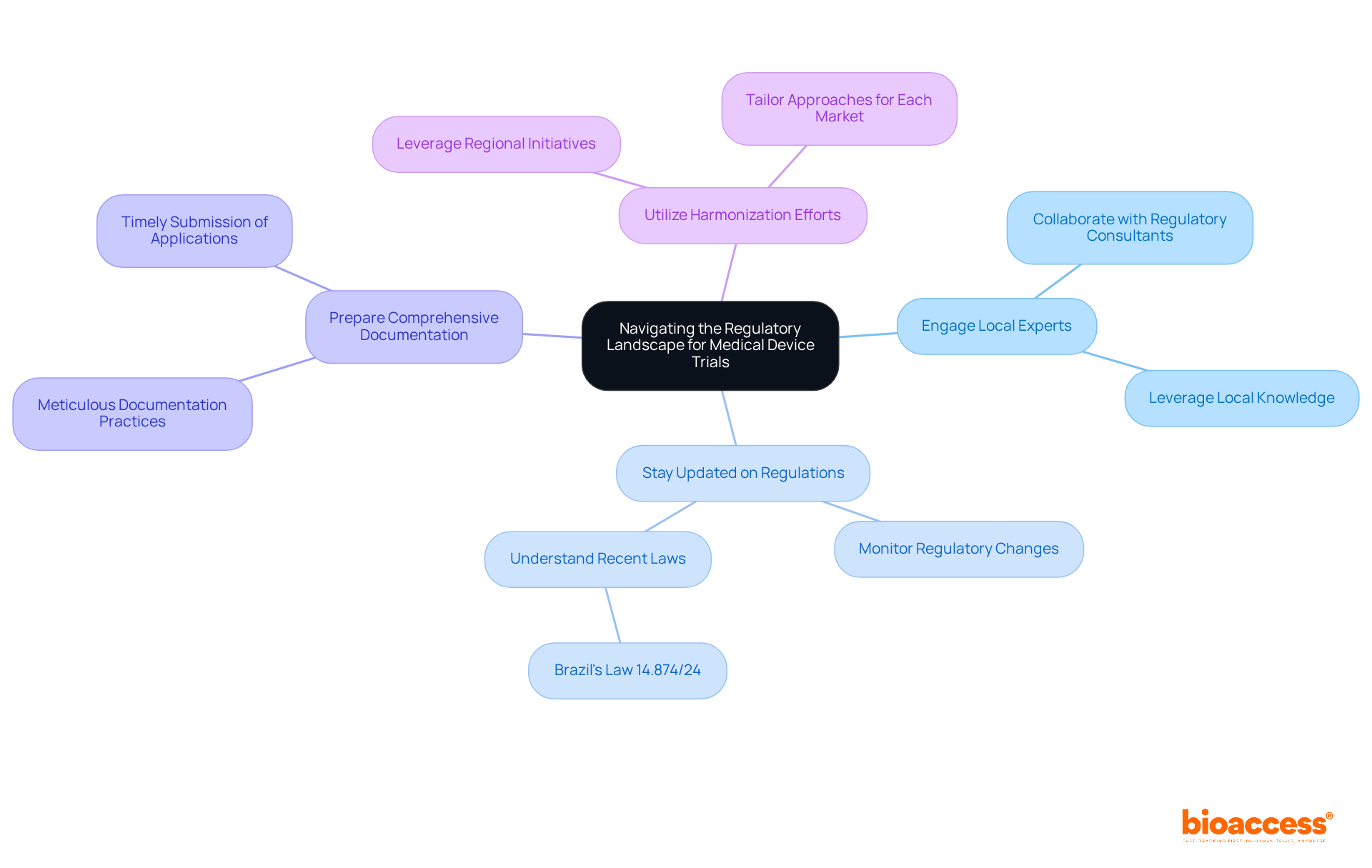
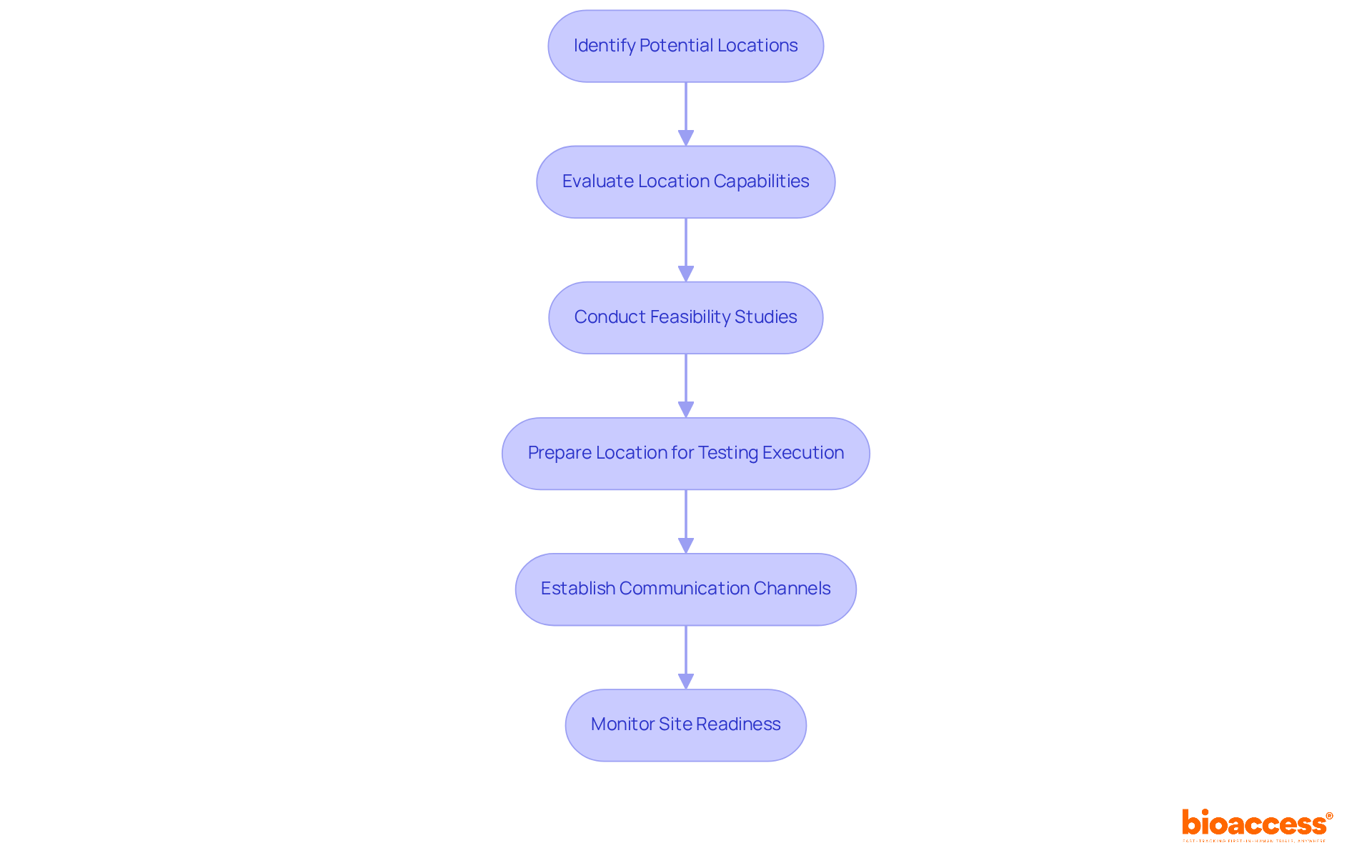
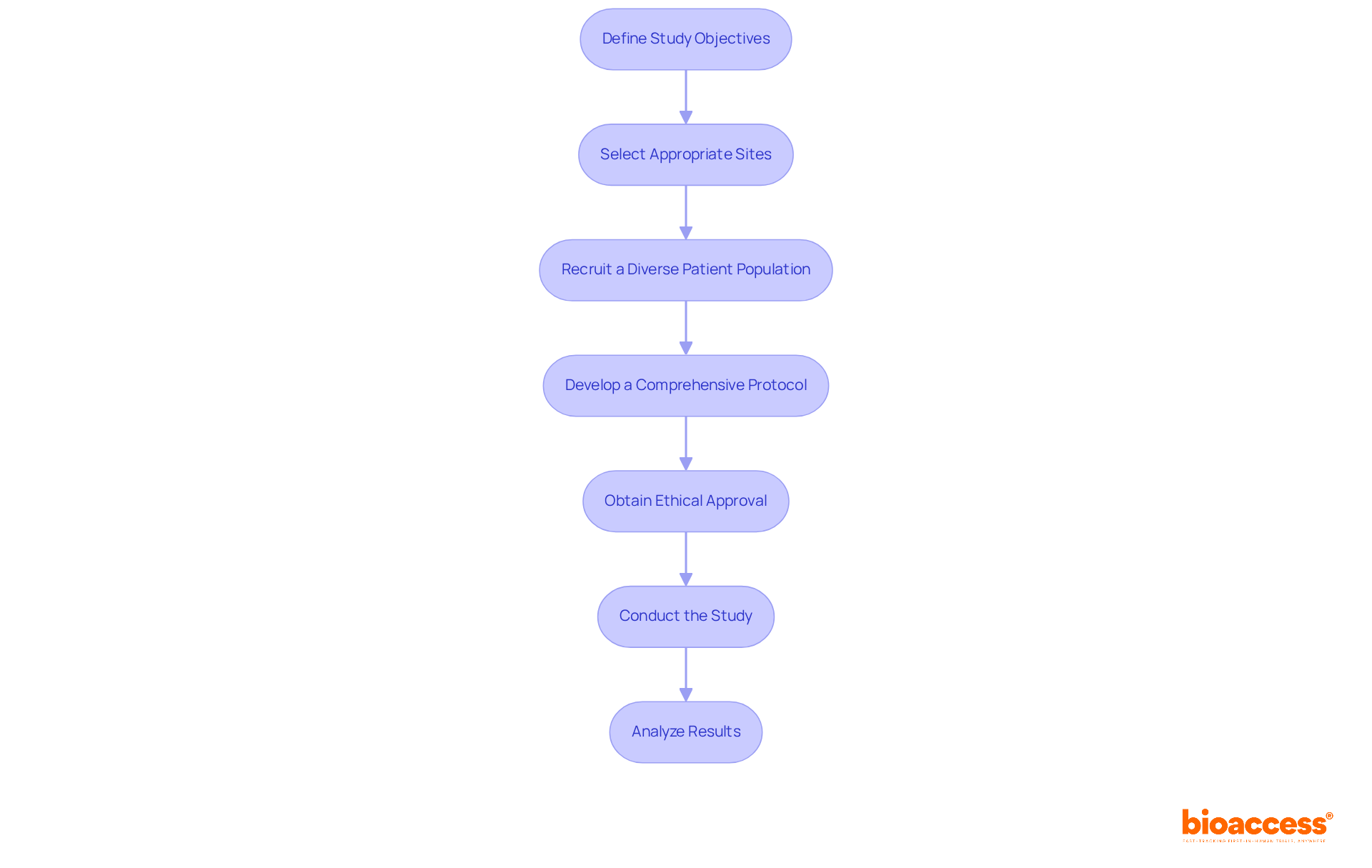
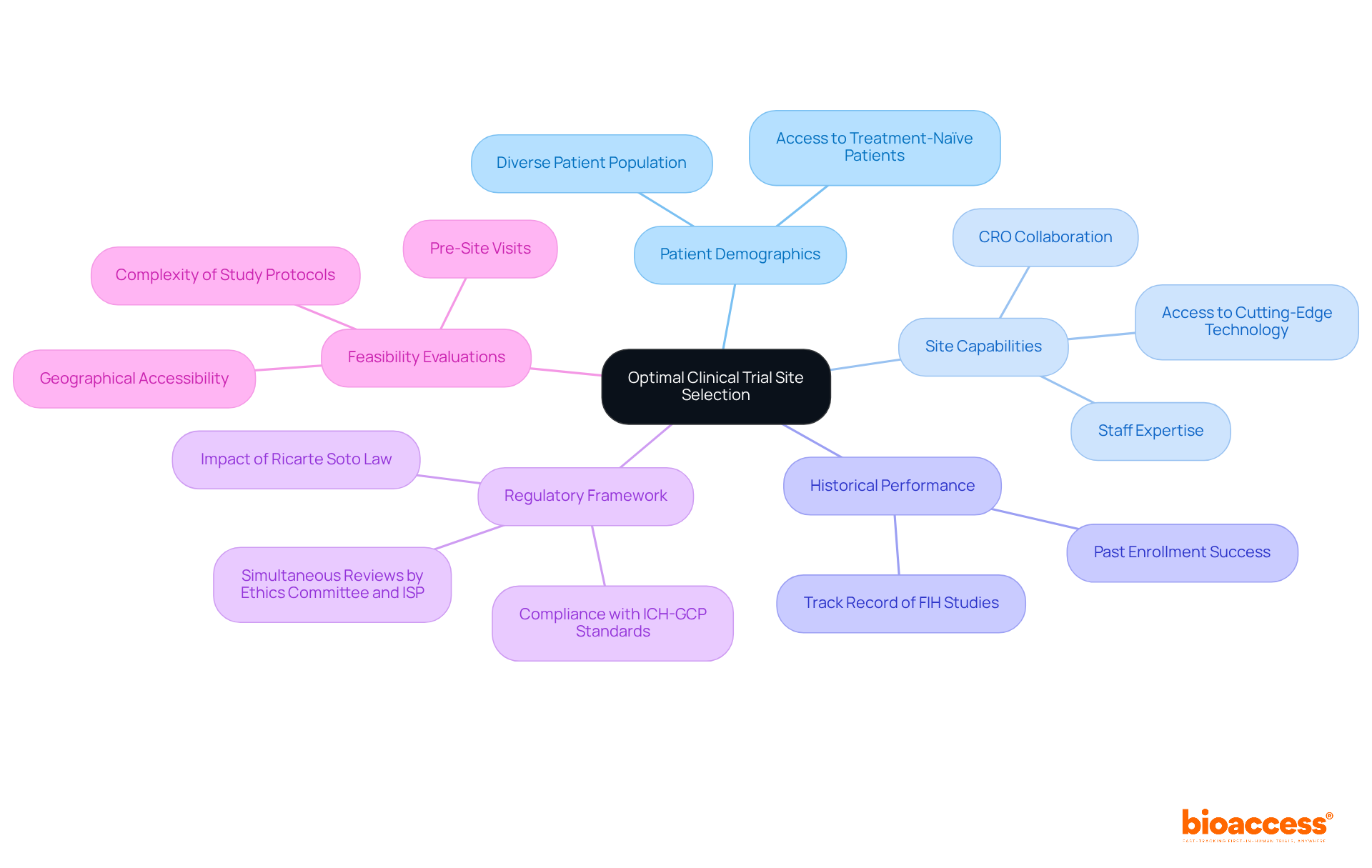
Selecting the right clinical trial outsourcing in Chile is a complex task that demands careful consideration of multiple factors, including patient demographics, site capabilities, and historical performance metrics. Sponsors should prioritize sites with a proven track record of successfully conducting first-in-human (FIH) studies and those that can access a diverse patient population. Collaborating with local contract research organizations (CROs) like bioaccess® offers sponsors crucial insights into site capabilities and helps identify locations that align with the specific goals of the study.
bioaccess® accelerates development through its Innovation Runway, designed to expedite the pathway from prototype to milestones, enabling MedTech, Biopharma, and Radiopharma startups to achieve their goals 40% faster. With access to 80% treatment-naïve patient populations and the capability to enroll participants 50% faster, bioaccess® enhances the efficiency of clinical studies. Academic institutions frequently serve as key locations due to their access to cutting-edge technology and skilled researchers, which can significantly improve the quality of studies. Moreover, it is crucial to perform comprehensive feasibility evaluations that consider geographical accessibility, compliance with regulations, and the intricacy of study protocols. This approach not only promotes efficient patient recruitment but also guarantees that selected sites are prepared to meet the rigorous demands of first-in-human studies.
In Chile, understanding the regulatory framework is essential for clinical trial outsourcing, as entities like the Instituto de Salud Pública (ISP) supervise adherence to ICH-GCP standards to accelerate approvals. As noted by Francisca Rodríguez, Manager of Innovation and Health at CIF, “It is crucial to allow simultaneous reviews by the Ethics Committee and the ISP, maintaining fluid communication between both.” By utilizing regional expertise and following optimal methods in site selection, including the audit-ready nature of bioaccess® assessments, sponsors can enhance their study processes, ultimately resulting in quicker timelines and lower expenses. Ultimately, the right site selection can be the difference between success and failure in clinical trials.
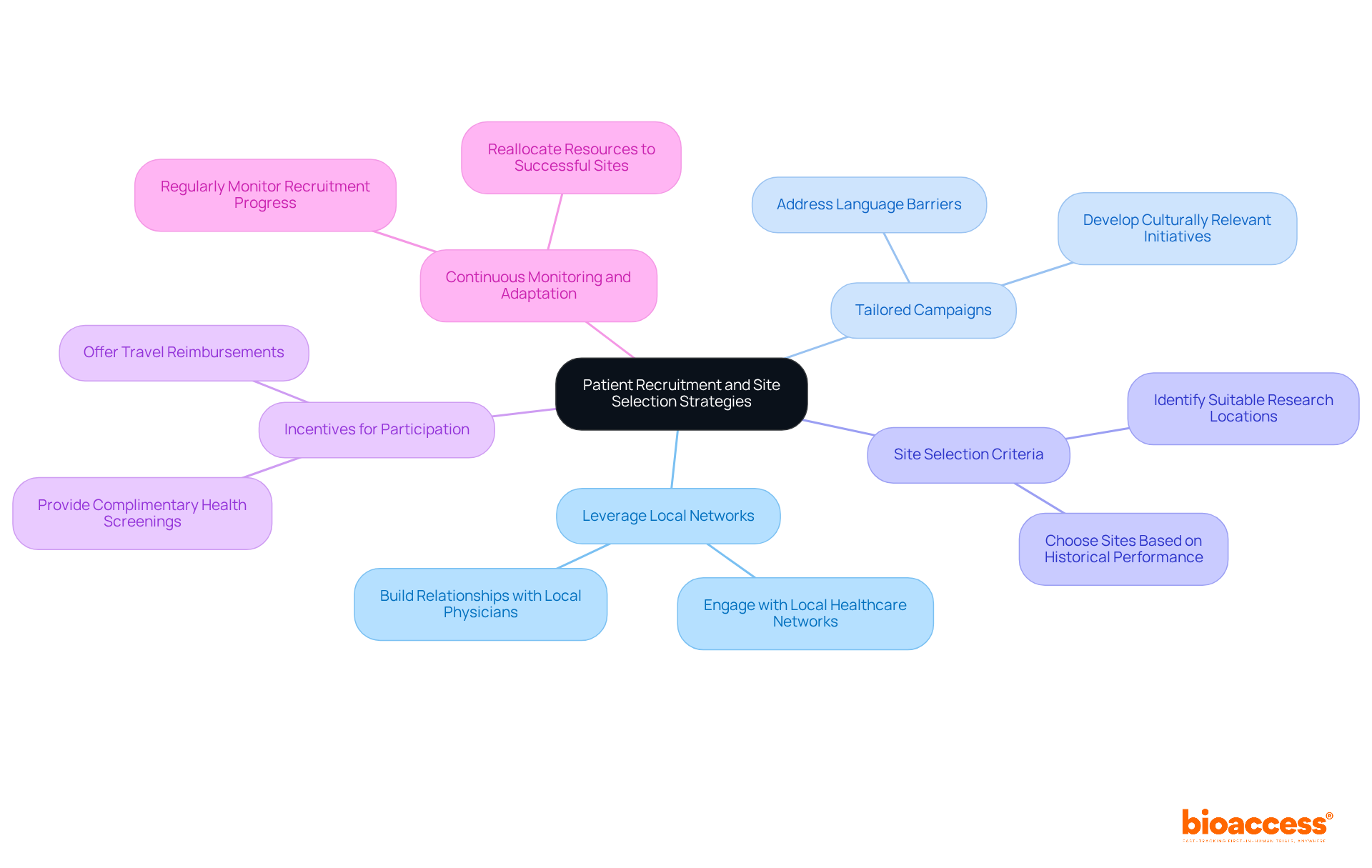
Implement Effective Patient Recruitment Strategies
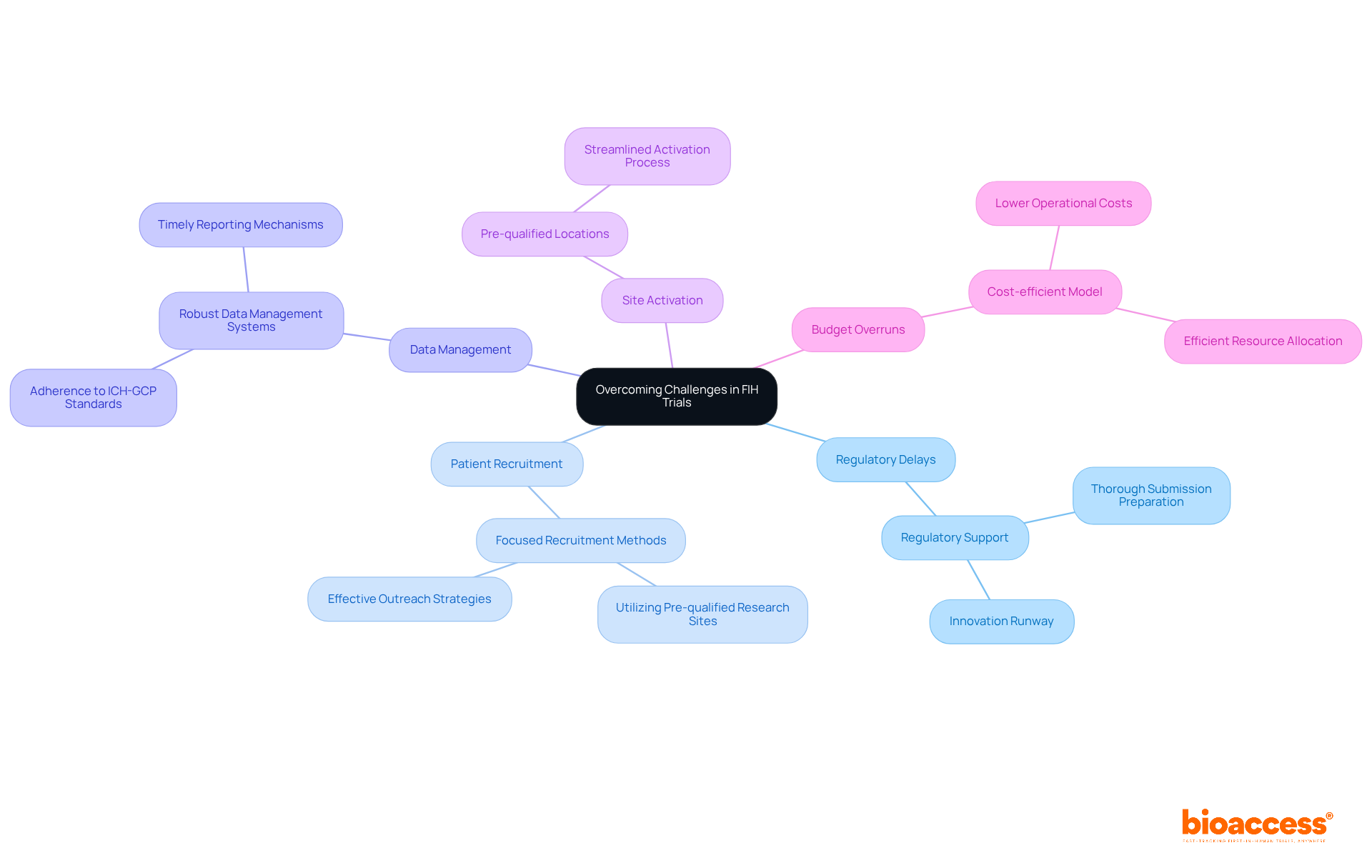
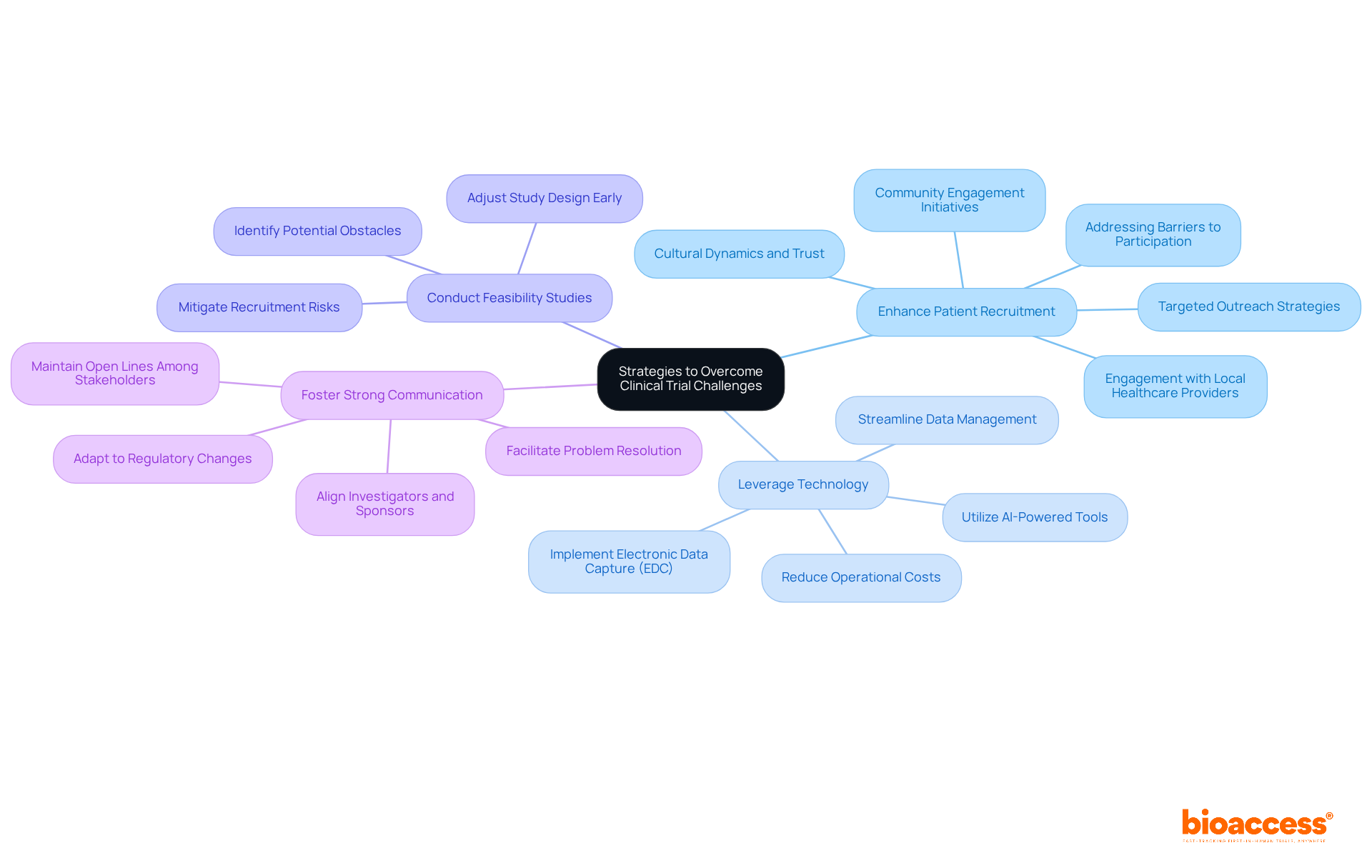
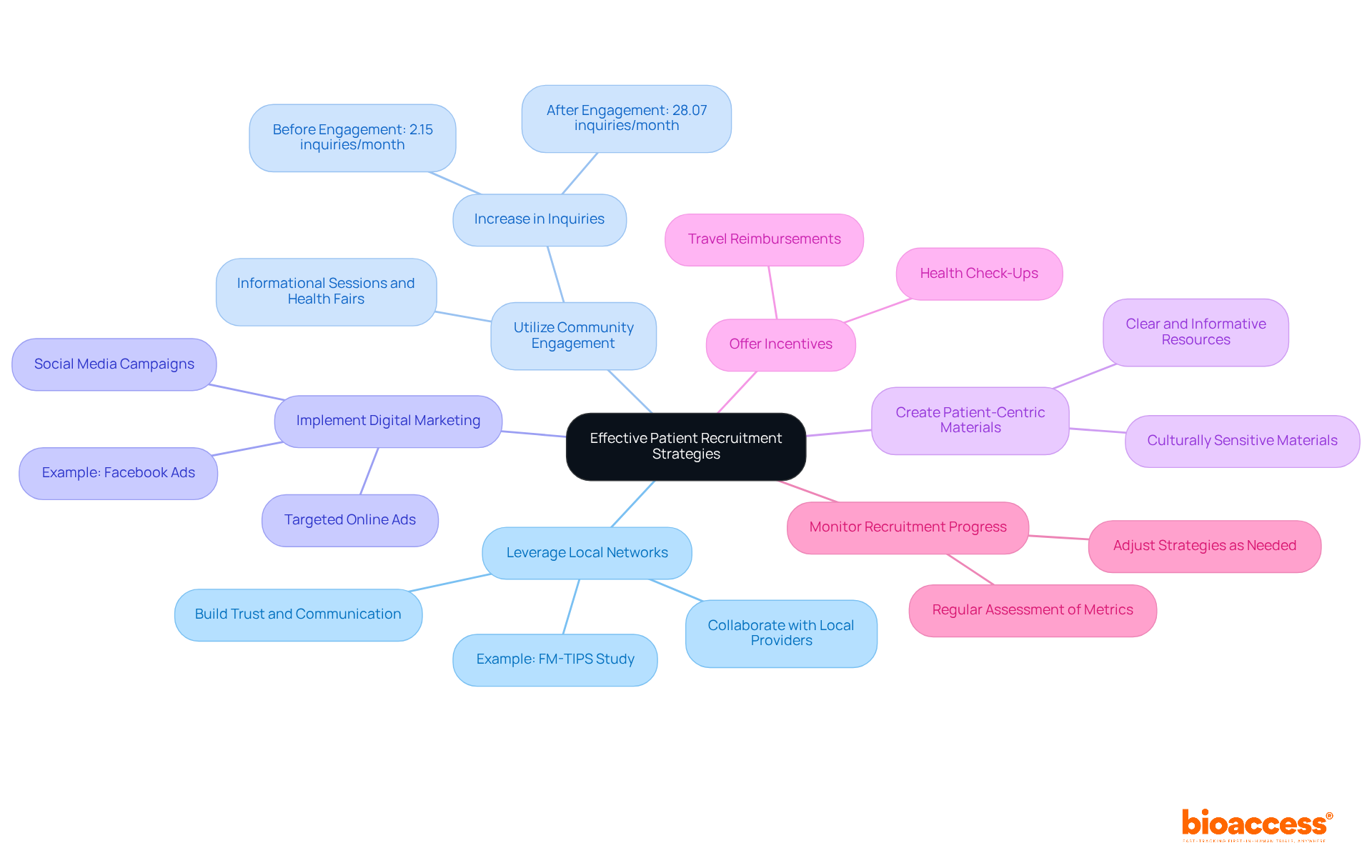
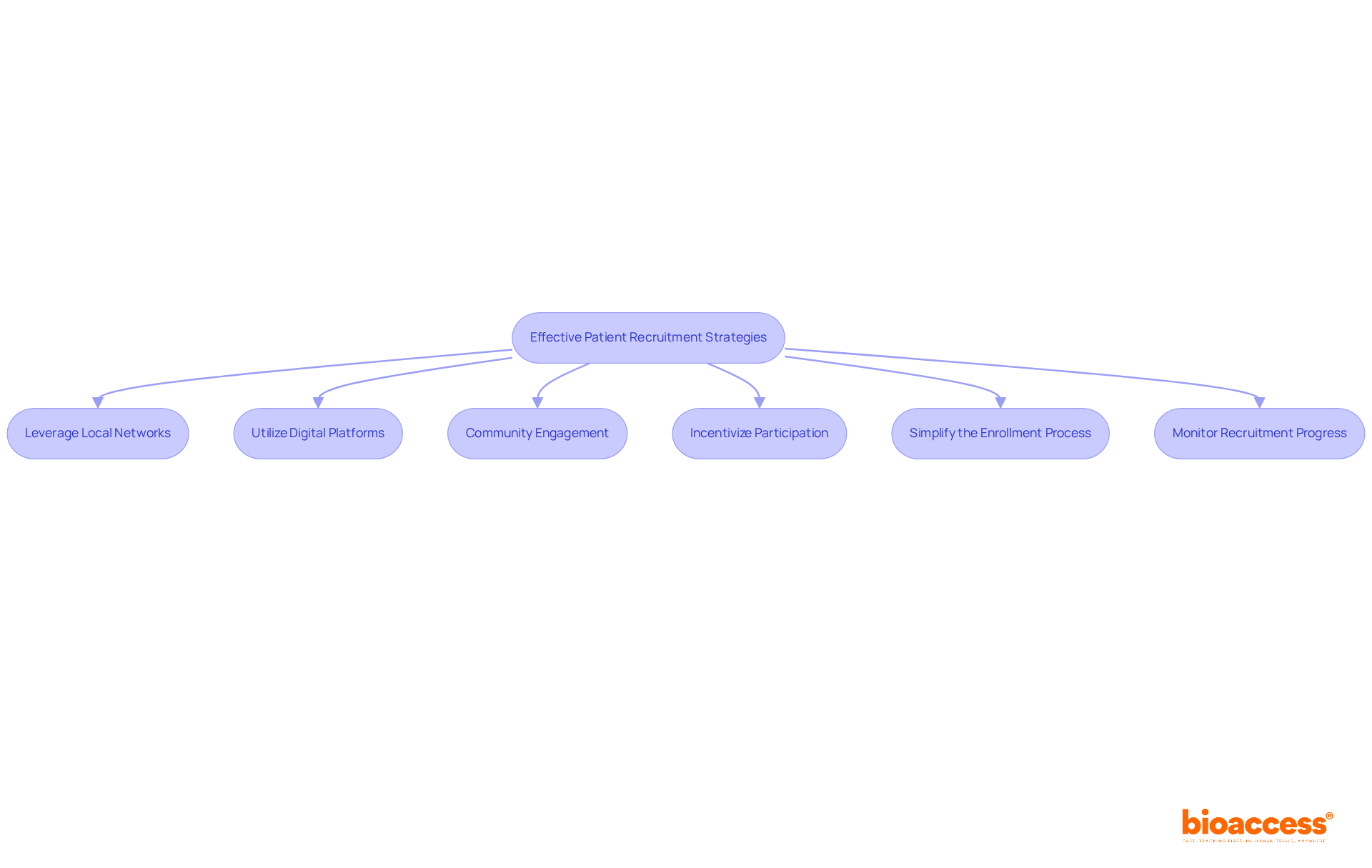
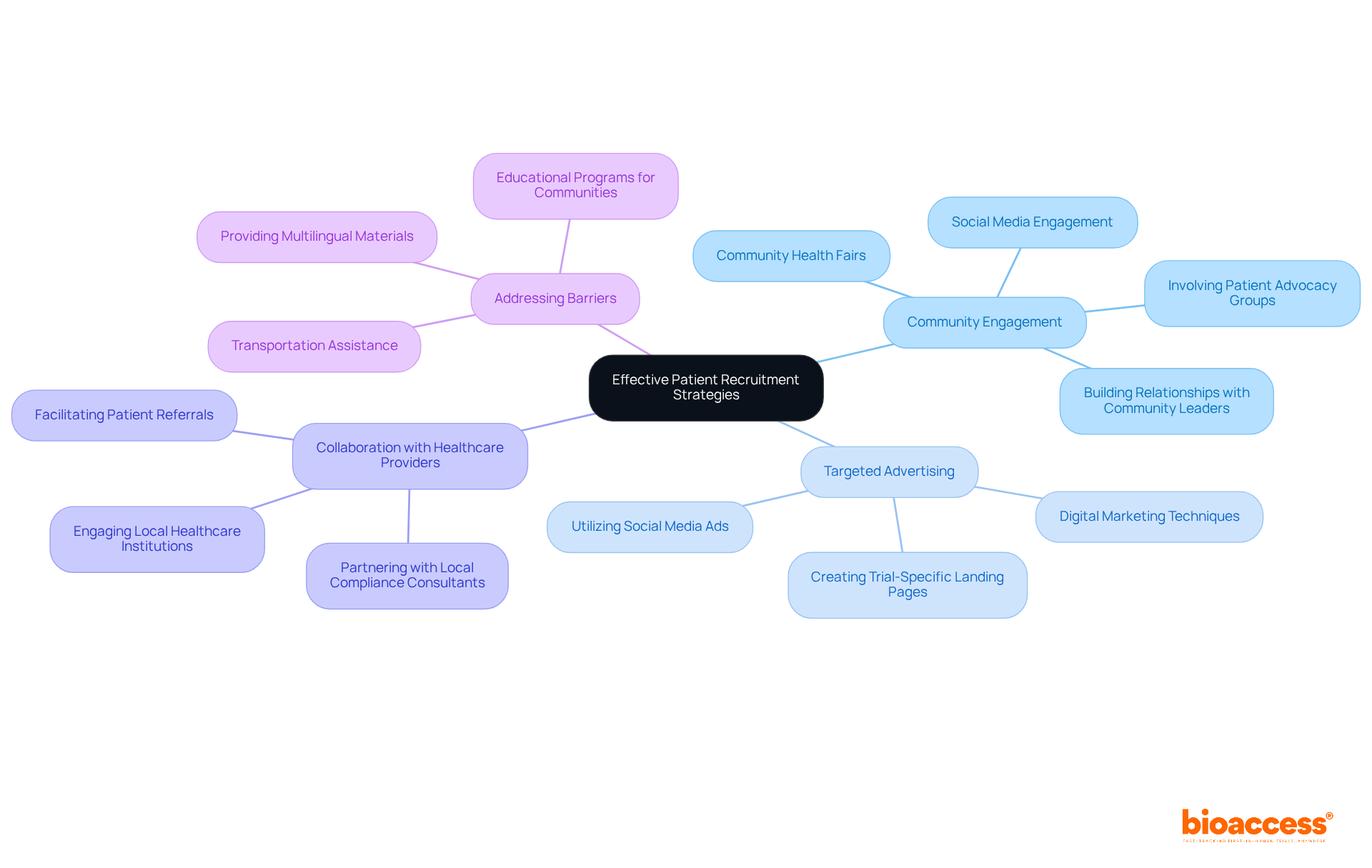
Recruiting participants for clinical trial outsourcing in Chile presents unique challenges that require a strategic approach. To effectively recruit participants, sponsors must implement a multifaceted strategy that prioritizes:
- Community engagement
- Targeted advertising
- Collaboration with regional healthcare providers
Leveraging social media platforms and arranging community health fairs greatly enhances awareness about research studies, drawing in potential participants. Forge strong relationships with community leaders and patient advocacy groups to build trust and drive participation, which is essential for successful recruitment.
Addressing barriers such as transportation challenges and language differences is crucial. Many potential participants face obstacles that hinder their involvement in clinical studies. Providing resources, such as transportation assistance and multilingual materials, can help mitigate these issues and make participation more accessible. Additionally, involving local communities through educational programs clarifies medical studies, improving their attractiveness and accessibility.
By implementing these strategies, sponsors enhance enrollment rates and ensure a diverse participant pool, crucial for credible study outcomes. Community involvement not only boosts recruitment strategies but also aids in creating a more inclusive research atmosphere. Ultimately, overcoming these barriers is essential for ensuring that clinical trial outsourcing in Chile is effective and representative.
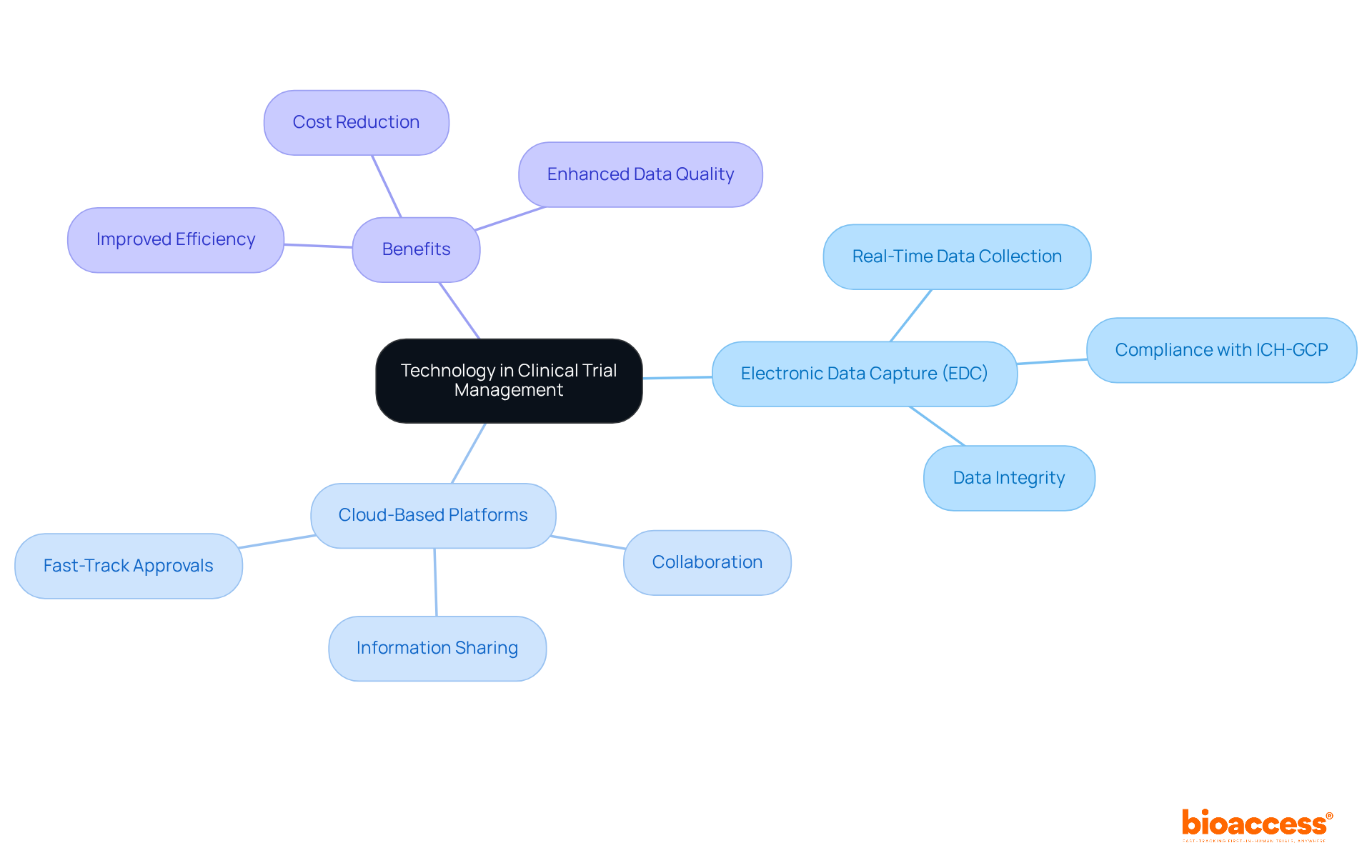
Leverage Technology for Streamlined Trial Management
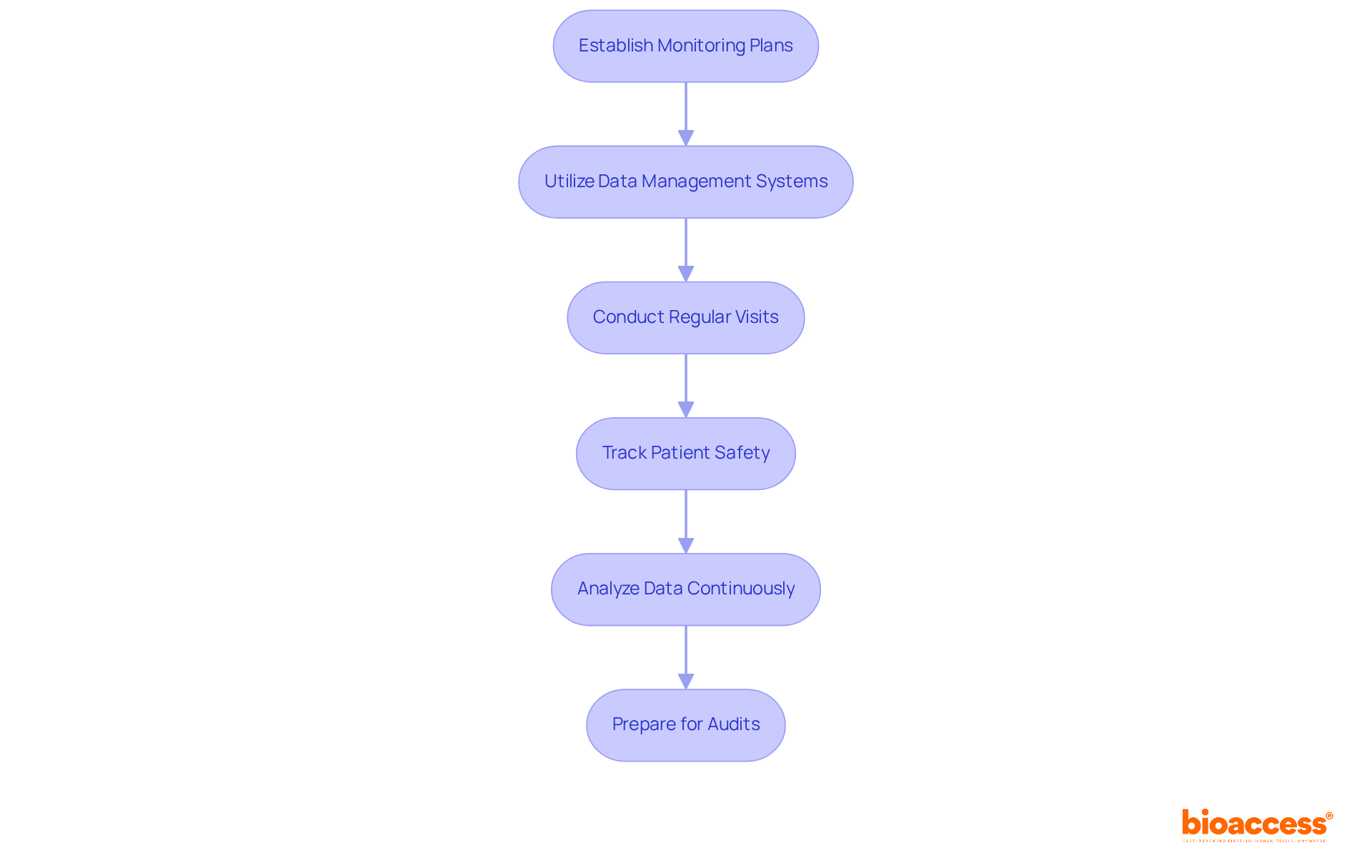
In the rapidly evolving landscape of clinical trials, the integration of technology is not just beneficial; it’s essential for success, particularly in first-in-human (FIH) trials in Latin America. Incorporating technology into clinical trial management significantly enhances both efficiency and data integrity. Electronic data capture (EDC) systems play a crucial role by enabling real-time data collection and analysis, which is essential for fulfilling compliance requirements established by authorities such as ANVISA and COFEPRIS. These systems streamline data management processes, reducing the administrative burden on research teams and improving patient compliance and retention rates.
Cloud-based platforms not only facilitate seamless collaboration among stakeholders but also ensure everyone stays in the loop with the latest information. This is especially advantageous in fast-track jurisdictions like Chile and Brazil for clinical trial outsourcing Chile, where approval timelines can be as brief as 30 to 90 days. By utilizing these technological advancements, sponsors can not only lower expenses but also expedite their journey to regulatory approval, ultimately improving the overall success of their trials. The integration of EDC systems ensures compliance with ICH-GCP standards and provides a robust framework for data integrity, making them indispensable tools for MedTech, Biopharma, and Radiopharma companies navigating the complexities of early-stage clinical research in Latin America.
As the MedTech and Biopharma sectors continue to evolve, leveraging these technological advancements will be crucial for staying ahead in the competitive landscape of clinical research.
Conclusion
Navigating the complexities of clinical trial outsourcing in Chile presents both challenges and opportunities for MedTech, Biopharma, and Radiopharmaceutical companies. Understanding the regulatory landscape and selecting optimal trial sites are crucial steps. By implementing effective patient recruitment strategies and leveraging technology, sponsors can significantly enhance their operational efficiency and success rates in early-stage clinical research.
It’s crucial to be familiar with the Instituto de Salud Pública’s requirements, as this knowledge, combined with strategic site selection and robust community engagement, can streamline processes and mitigate delays. Utilizing local expertise, such as that provided by organizations like bioaccess®, allows sponsors to tap into valuable resources for patient recruitment and site capabilities, ultimately leading to quicker approvals and reduced costs. Furthermore, the integration of technology, such as electronic data capture systems, is essential for maintaining data integrity and ensuring compliance with ICH-GCP standards.
In conclusion, embracing these strategies not only positions sponsors for success but also transforms the clinical trial landscape in Latin America. By adopting these best practices, sponsors can capitalize on the unique opportunities presented by the Latin American market, driving innovation and improving patient outcomes in the clinical trial landscape.
Frequently Asked Questions
What is the role of the Instituto de Salud Pública (ISP) in clinical trials in Chile?
The Instituto de Salud Pública (ISP) oversees the regulatory framework for clinical research studies in Chile, ensuring compliance with national health regulations.
What is the typical timeline for application evaluations by the ISP?
The ISP generally evaluates clinical trial applications within 30 business days, making Chile one of the quicker regions in Latin America for clinical study approvals.
What key documents are required for submitting a clinical trial application in Chile?
Key documents required for submission include the study protocol, informed consent forms, and qualifications of the investigators.
Why is compliance with ICH-GCP standards important for clinical trials in Chile?
Compliance with ICH-GCP standards is essential to ensure that studies are conducted ethically and that data integrity is maintained throughout the clinical trial process.
Are regional ethics committee approvals necessary for clinical trials in Chile?
Yes, obtaining approval from regional ethics committees is necessary and can influence the overall timelines for clinical trial initiation.
How can understanding the regulatory landscape benefit sponsors conducting clinical trials in Chile?
Familiarizing themselves with the regulatory nuances allows sponsors to streamline their study initiation processes and avoid common pitfalls that can lead to delays, ultimately contributing to the success of their trials.
List of Sources
- Understand the Regulatory Landscape for Clinical Trials in Chile
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Navigating Clinical Research Regulations for Medical Devices in Chile | Julio G. Martinez-Clark (https://linkedin.com/posts/juliomartinezclark_navigating-clinical-research-regulations-activity-7265280286836322305-4OxJ)
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- Clinical trials by start date Chile 1999-2024| Statista (https://statista.com/statistics/1560150/chile-number-new-clinical-trials?srsltid=AfmBOooWmpb3fNKp75jpPsSx8zCza-J3u-qq_39bsUJzfW9qylZ-YSdl)
- Select Optimal Clinical Trial Sites for Enhanced Efficiency
- Relatório revela que o Chile é o país que mais realiza Ensaios Clínicos na América Latina (https://saludyfarmacos.org/lang/es/boletin-farmacos/boletines/nov202406pt/05_re)
- Best Practices for Clinical Site Selection | CITI Program (https://about.citiprogram.org/blog/best-practices-for-clinical-site-selection)
- Trends in clinical trials performed in Chile (https://scielo.cl/scielo.php?pid=S0034-98872021000100110&script=sci_abstract&tlng=en)
- Implement Effective Patient Recruitment Strategies
- Clinical Trials Patient Recruitment in Latin America | H Clinical (https://hclinical.com/patient-recruitment)
- Patient Recruitment Strategies for Trials | CCRPS (https://ccrps.org/clinical-research-blog/patient-recruitment-strategies-for-clinical-trials)
- Patient Recruitment Strategies for Clinical Trials (https://freyrsolutions.com/blog/patient-recruitment-strategies-for-clinical-trials)
- Navigate Medical Device Clinical Trials in Chile: A Step-by-Step Guide – cms.bioaccessla.com (https://cms.bioaccessla.com/blog/navigate-medical-device-clinical-trials-in-chile-a-step-by-step-guide)
- Leverage Technology for Streamlined Trial Management
- Careset.com – CareSet: Medicare Data Analysis & Patient Care Insights (https://careset.com/10-benefits-of-edc-electronic-data-capture-for-clinical-trials)
- Electronic Data Capture Systems for Clinical Trials and Research (https://egnyte.com/guides/life-sciences/electronic-data-capture)
- Electronic Data Capture (EDC) System: 5 Key Benefits (https://clinion.com/insight/benefits-of-electronic-data-capture-system)
- Electronic Data Capture In Clinical Trials | Credevo Articles (https://credevo.com/articles/2024/05/05/electronic-data-capture-in-clinical-trials-impact-advantages)
- How Clinical Trial Technology Increases Speed and Efficiency (https://datacubed.com/resources/the-impact-of-clinical-trial-technology-on-speed-and-efficiency-in-research)