Introduction
In the fast-paced world of clinical research, securing Institutional Review Board (IRB) approval in Chile is a critical milestone that can define the success of a trial. This process ensures ethical treatment of participants while boosting the credibility and success of clinical trials. For researchers engaged in first-in-human studies, mastering the IRB approval process is crucial. It directly influences both the timeline and integrity of their research.
How can researchers effectively tackle the challenges of IRB approval to ensure their studies stay on track?
Understand the Importance of IRB Approval in Chile
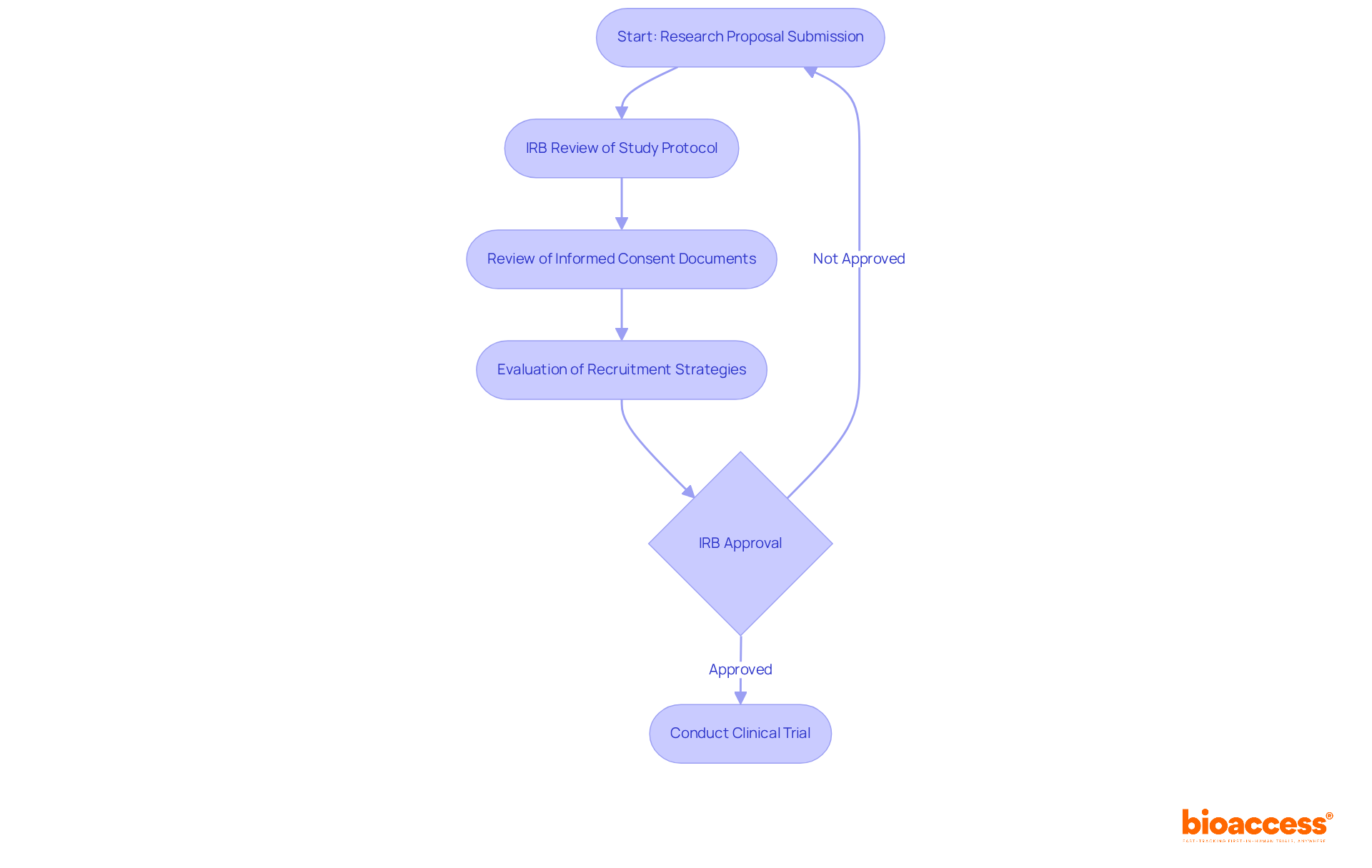
In the realm of clinical research in Chile, the role of Institutional Review Boards (IRBs) transcends mere regulation; they are pivotal in upholding ethical standards and protecting participant rights. The IRB review process is a critical step that not only enhances research integrity but also safeguards participant welfare.
The IRB meticulously reviews study protocols, informed consent documents, and recruitment strategies to ensure that risks are minimized and that participants are adequately informed about the study’s nature and potential risks. While other regions may face prolonged IRB clearance times, Chile’s 30-day average presents a significant advantage for researchers. This efficiency highlights how crucial thorough preparation and strict adherence to ethical standards are for the success of clinical trials.
Furthermore, successful clinical trials in Chile frequently depend on acquiring timely IRB consent, as it aids in adhering to local regulations and boosts the credibility of the research findings. Expert opinions emphasize that robust ethical oversight by IRBs is critical in fostering trust among participants and stakeholders, ultimately contributing to higher success rates in clinical trials. Navigating the IRB process effectively can be the difference between success and delays in clinical trials. Understanding and navigating the IRB approval Chile journey is therefore essential for researchers intending to conduct first-in-human trials, particularly when utilizing the strategic benefits that bioaccess® provides regarding speed and regulatory efficiency. Bioaccess® collaborates with regulatory agencies such as ISP (Instituto de Salud Pública) to ensure compliance and expedite the approval process. For researchers, mastering the IRB process is not just beneficial; it is essential for ensuring the success of their clinical trials in Chile.
Gather Required Documentation for IRB Submission
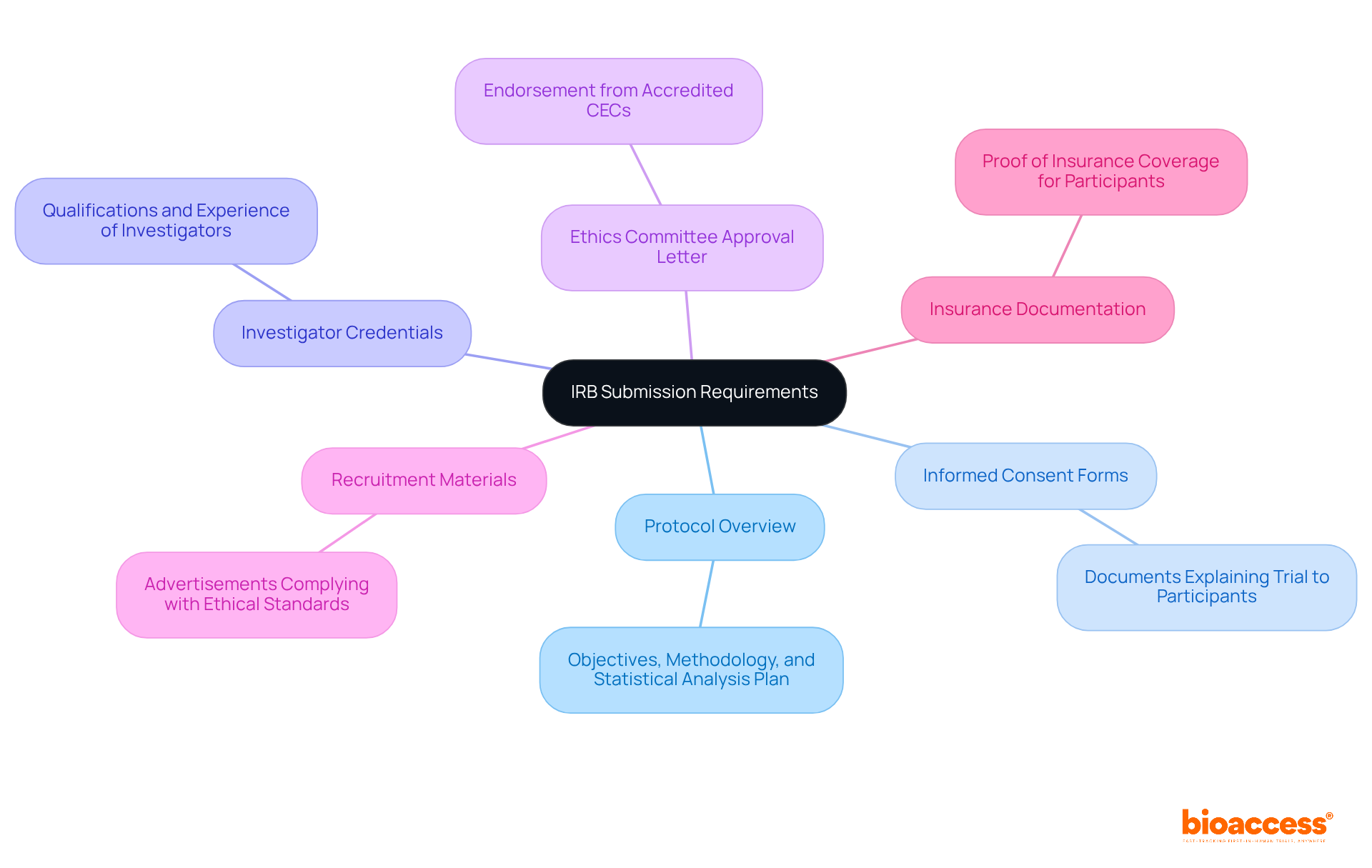
Submitting an irb approval chile application can be a complex process, but with the right preparation, researchers can successfully navigate it. To ensure a smooth submission, researchers must compile a comprehensive set of documents. The following checklist outlines the essential items required:
- Protocol Overview: A comprehensive outline of the objectives, methodology, and statistical analysis plan of the research, ensuring alignment with ICH-GCP standards.
- Informed Consent Forms: Clear and concise documents that explain the trial to participants, ensuring they understand their rights and the nature of the research.
- Investigator Credentials: Documentation of the qualifications and experience of the principal investigator and any co-investigators, demonstrating their capability to conduct the trial.
- Ethics Committee Approval Letter: Evidence of endorsement from one of the more than 60 accredited Scientific Ethical Committees (CECs) in Chile, which is mandatory before submitting to the IRB.
- Recruitment Materials: Any advertisements or materials that will be used to recruit participants, ensuring they comply with ethical standards.
- Insurance Documentation: Proof of insurance coverage for participants, which is often required to mitigate risks associated with the trial.
By thoroughly preparing and organizing these documents, researchers can streamline the submission timeline. This significantly enhances their chances of obtaining timely irb approval chile. Navigating the lengthy irb approval chile process can pose a significant hurdle for researchers. Have you considered how thorough preparation might ease this challenge? Understanding these requirements not only expedites the process but also sets the stage for successful clinical trials. Typically, the Instituto de Salud Pública (ISP) takes about 30 business days to review applications, underscoring the importance of accurate and complete submissions.
Navigate the IRB Submission Process in Chile
Successfully navigating the IRB submission process in Chile is crucial for the efficiency of your clinical trial, especially for First-in-Human (FIH) studies:
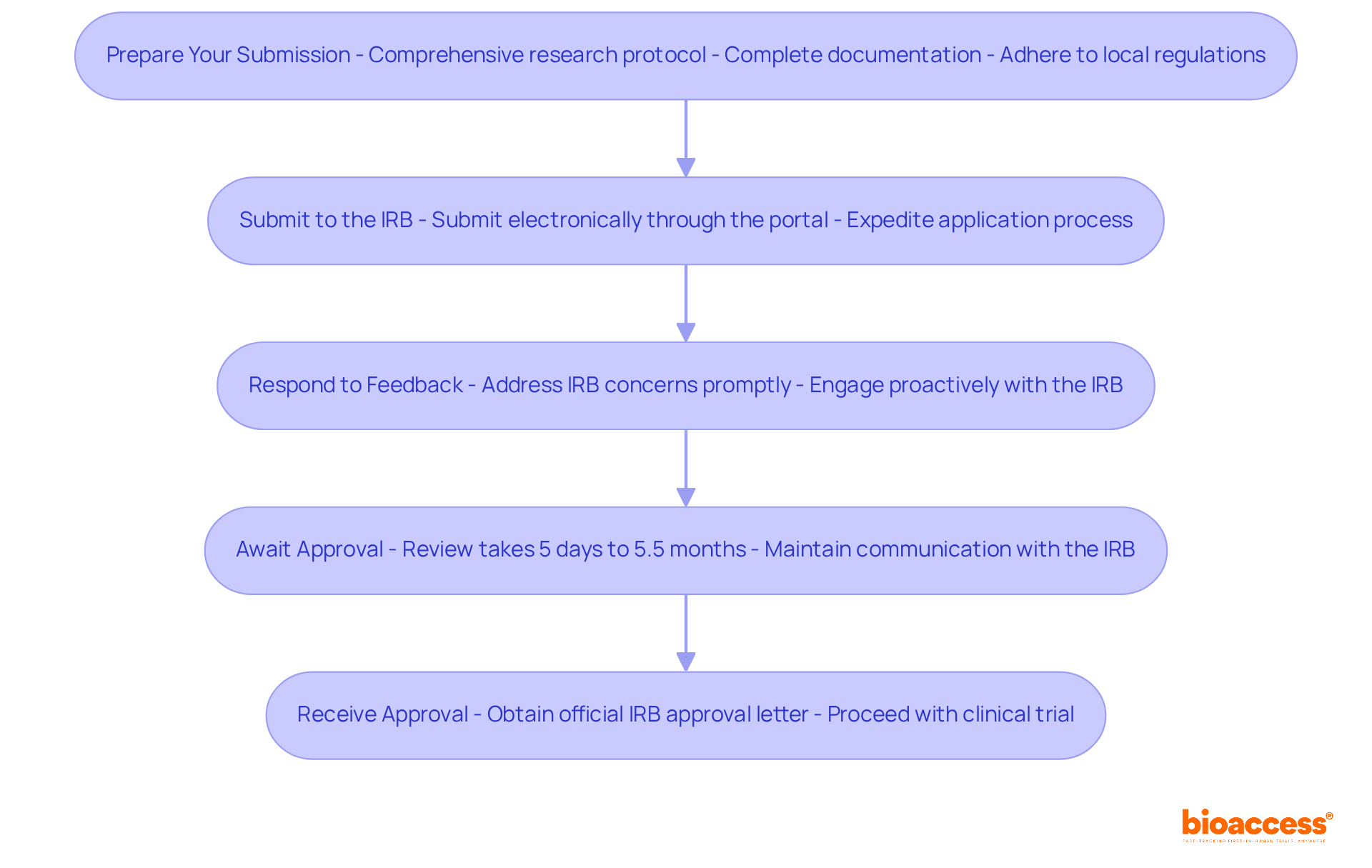
- Prepare Your Submission: This requires a comprehensive research protocol that clearly defines your project’s objectives, methodologies, and ethical considerations. Ensure that all required documentation is complete and organized, adhering to local regulations and ethical guidelines set forth by the Instituto de Salud Pública (ISP) and relevant ethics committees. Thorough preparation is essential, as it can help mitigate risks in medical device development and enable quicker authorizations, particularly through early feasibility studies.
- Submit to the IRB: When your documents are ready, take the crucial step of submitting them electronically through the IRB’s designated submission portal. This streamlined method is vital for expediting your application, particularly in the context of FIH trials, where time is of the essence.
- Respond to Feedback: After submission, the IRB may provide feedback or request modifications. Be prepared to address any concerns promptly and thoroughly. Proactively engaging with the IRB is essential for ensuring a smooth review process, which is vital for accelerating your clinical trial timelines.
- Await Approval: The IRB review typically takes about 30 days, although it can range from 5 days to 5.5 months depending on various factors, including the completeness of your submission and the experience level of the investigator. Maintaining open communication with the IRB during this period is essential to address any questions they may have. Utilizing the swift ethics endorsement timelines available in Latin America can offer a strategic benefit for your FIH studies.
- Receive Approval: Once your application is approved, ensure you obtain the official IRB approval letter, which is necessary for proceeding with your clinical trial.
By following these steps, researchers can effectively navigate the IRB submission journey for IRB approval in Chile, minimizing delays and ensuring adherence to ethical standards. Navigating the IRB submission process can be fraught with challenges, particularly for those unfamiliar with the regulatory landscape. Common pitfalls include incomplete documentation and misinterpretations of regulatory requirements, which can lead to significant delays. This disparity highlights the critical need for seasoned guidance in navigating the complexities of IRB submissions. Interestingly, less seasoned investigators may find the undertaking more difficult, with a subjective difficulty level rated at 4 out of 10, in contrast to 2 out of 10 for seasoned investigators. Understanding these nuances can be the difference between a timely approval and significant delays in your clinical research journey.
Identify and Overcome Common Challenges in IRB Approval
Navigating the irb approval chile process can be challenging, but understanding common pitfalls can help streamline your research efforts.
The IRB approval process in Chile, while generally efficient, presents several common challenges that researchers must navigate:
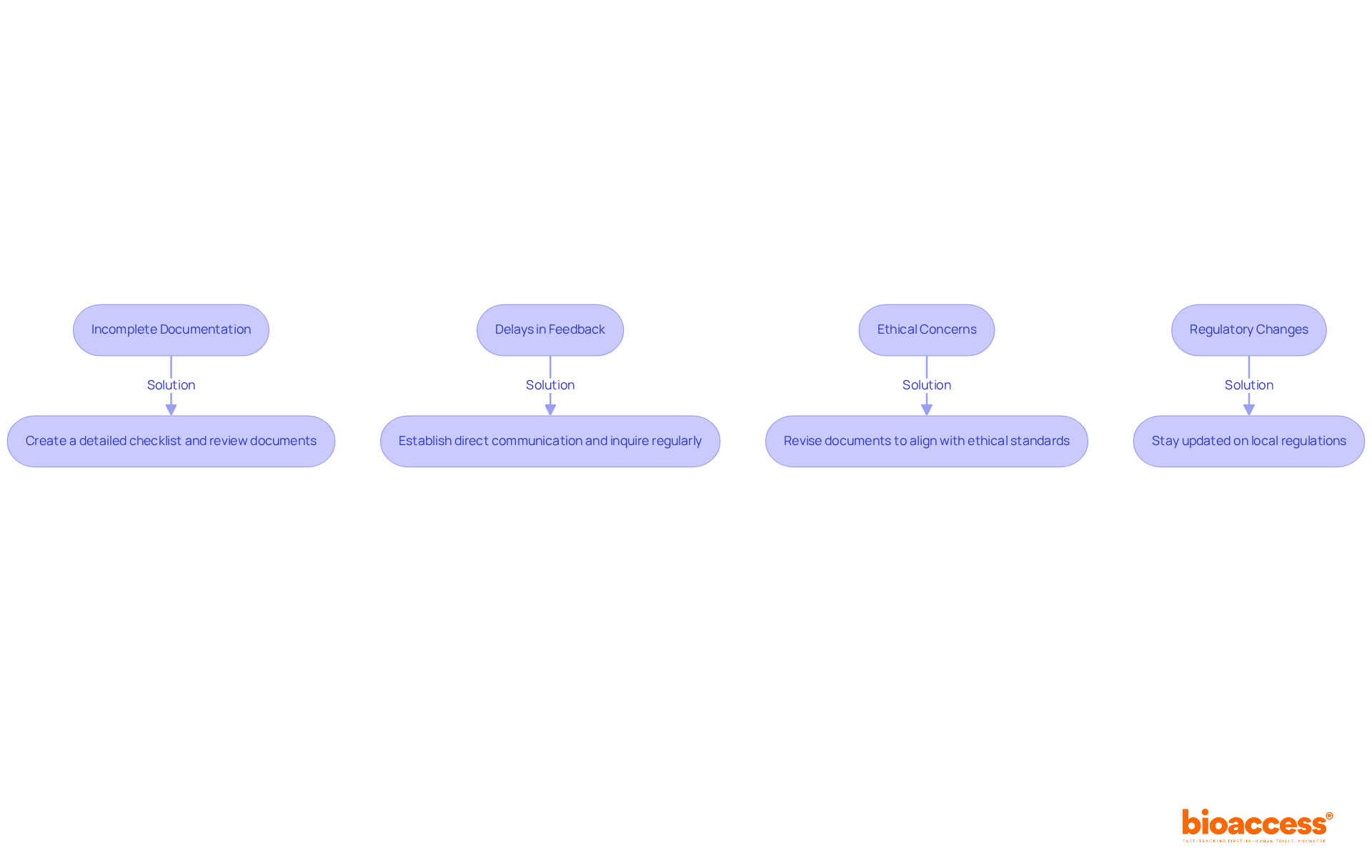
- Incomplete Documentation: Submitting incomplete or poorly organized documentation is a frequent issue. Create a detailed checklist of all required documents, including informed consent forms and study protocols, to avoid incomplete submissions. Ensure that multiple team members review these documents for completeness and clarity before submission. A comprehensive proposal decreases the chances of delays in the IRB review stage.
- Delays in Feedback: Have you ever faced delays in receiving feedback from the IRB? This can stall your research progress and lead to frustration. The duration required for the investigator to reply to communications from the IRB Analyst or the Board represents a crucial area for time loss in the review timeline. To address this, establish a direct line of communication regarding your IRB approval in Chile and regularly inquire about the status of your submission. This proactive approach can assist in identifying any potential issues early in the review stage.
- Ethical Concerns: If the IRB raises ethical concerns, be prepared to respond thoroughly. This may involve revising your informed consent documents or trial protocols to better align with ethical standards, such as those outlined by the Chilean regulatory authorities, including the Instituto de Salud Pública (ISP) and the Agencia Nacional de Medicamentos (ANAMED). Make sure your revisions tackle all the IRB’s concerns to help speed up the review process.
- Regulatory Changes: Stay updated on any changes in local regulations that may impact your study. Regularly consult resources from Chile’s regulatory bodies to ensure compliance with current requirements. There are substantial direct and indirect costs associated with IRB oversight of research, which should be factored into your planning.
By anticipating these challenges and implementing strategic solutions, you can transform potential setbacks into opportunities for success in your clinical trials.
Conclusion
Mastering the IRB approval process in Chile is not just a regulatory hurdle; it’s a critical step that defines the success of clinical trials in the MedTech and biopharma sectors. IRB approval is more than just a compliance requirement; it reflects a commitment to ethical standards and participant safety, which are crucial for the integrity of research findings. By mastering the IRB process, researchers can enhance the likelihood of timely approvals and successful trial outcomes.
Key insights highlighted throughout the article emphasize the importance of thorough preparation, including:
- The meticulous gathering of required documentation
- Proactive engagement with the IRB
- Submitting a comprehensive protocol overview
- Addressing feedback promptly
Each step plays a crucial role in minimizing delays and ensuring a smooth review process. Navigating the IRB process can be daunting, with numerous requirements and potential setbacks. However, successfully navigating these challenges can significantly enhance the chances of timely approvals and successful trial outcomes.
In conclusion, the IRB approval process in Chile represents a vital component of clinical research that cannot be overlooked. Leveraging the region’s strategic advantages, including swift approval timelines and robust regulatory frameworks, positions researchers for success in their studies. By embracing these best practices, researchers not only secure IRB approval but also contribute to the evolution of healthcare innovation in Chile.
Frequently Asked Questions
What is the role of Institutional Review Boards (IRBs) in clinical research in Chile?
IRBs in Chile are essential for upholding ethical standards and protecting participant rights. They review study protocols, informed consent documents, and recruitment strategies to ensure risks are minimized and participants are well-informed about the study.
How long does the IRB approval process typically take in Chile?
The average IRB approval process in Chile takes about 30 days, which is relatively efficient compared to other regions.
Why is timely IRB consent important for clinical trials in Chile?
Timely IRB consent is crucial as it helps researchers adhere to local regulations and enhances the credibility of the research findings, contributing to the overall success of clinical trials.
What impact does IRB oversight have on clinical trials?
Robust ethical oversight by IRBs fosters trust among participants and stakeholders, which is vital for achieving higher success rates in clinical trials.
How can researchers effectively navigate the IRB process in Chile?
Researchers can navigate the IRB process effectively by preparing thoroughly and adhering strictly to ethical standards, which can prevent delays and ensure the success of their clinical trials.
What strategic advantages does bioaccess® provide regarding the IRB approval process?
Bioaccess® collaborates with regulatory agencies like the Instituto de Salud Pública (ISP) to ensure compliance and expedite the IRB approval process, which is particularly beneficial for first-in-human trials.
What should researchers focus on to ensure the success of their clinical trials in Chile?
Researchers should focus on mastering the IRB process, as it is essential for compliance with local regulations and for the overall success of their clinical trials in Chile.
List of Sources
- Understand the Importance of IRB Approval in Chile
- Trends in clinical trials performed in Chile (https://scielo.cl/article_plus.php?pid=S0034-98872021000100110&tlng=en&lng=es)
- “If I am here, it is because the system failed”: a critical qualitative study of global commercial clinical trials for advanced cancer in Chile – Globalization and Health (https://link.springer.com/article/10.1186/s12992-025-01148-x)
- Challenges in institutional ethical review process and approval for international multicenter clinical studies in lower and middle-income countries: the case of PARITY study – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC11577162)
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Gather Required Documentation for IRB Submission
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- How the Scientific Ethical Committees work in Chile (https://linkedin.com/pulse/how-scientific-ethical-committees-work-chile-bopalcro-sigce)
- Refugee Appeals by Country of Alleged Persecution – 2024
- Immigration and Refugee Board of Canada (https://irb-cisr.gc.ca/en/statistics/appeals/Pages/RADStat2024.aspx?=undefined&wbdisable=true)
- Required Documents for IRB Submissions – Solutions IRB (https://solutionsirb.com/required-documents-for-irb-submissions)
- Navigate the IRB Submission Process in Chile
- Navigate Clinical Trial Approval in Chile: 4 Essential Steps | bioaccess® (https://bioaccessla.com/blog/navigate-clinical-trial-approval-in-chile-4-essential-steps)
- Navigating the Institutional Review Board Approval Process in a Multicenter Observational Critical Care Study – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC3989388)
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- Navigate Biopharma Clinical Trials in Chile: A Step-by-Step Guide | bioaccess® (https://bioaccessla.com/blog/navigate-biopharma-clinical-trials-in-chile-a-step-by-step-guide)
- Identify and Overcome Common Challenges in IRB Approval
- Common Problems with IRB Applications (https://montclair.edu/institutional-review-board/faqs/common-problems-with-irb-applications)
- Institutional Review Boards: Purpose and Challenges – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC4631034)
- Top 10 Common Problems to Avoid with IRB Protocol Submissions | Ohio University (https://ohio.edu/research/compliance/human-subjects/top10)
- Challenges in institutional ethical review process and approval for international multicenter clinical studies in lower and middle-income countries: the case of PARITY study – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC11577162)
- Ethical Considerations for Trials in Chile: Key Insights and Challenges | bioaccess® (https://bioaccessla.com/blog/ethical-considerations-for-trials-in-chile-key-insights-and-challenges)

Leave a Reply