Introduction
While Chile’s regulatory framework offers promising approval timelines for first-in-human biopharma trials, the path to success is fraught with challenges that sponsors must navigate. As a strategic hub for clinical research in Latin America, Chile presents significant opportunities. Yet, the complexities of compliance, patient recruitment, and site selection present significant hurdles that can hinder progress.
What strategies can sponsors implement to not only leverage Chile’s advantages but also navigate these obstacles for successful trial outcomes?
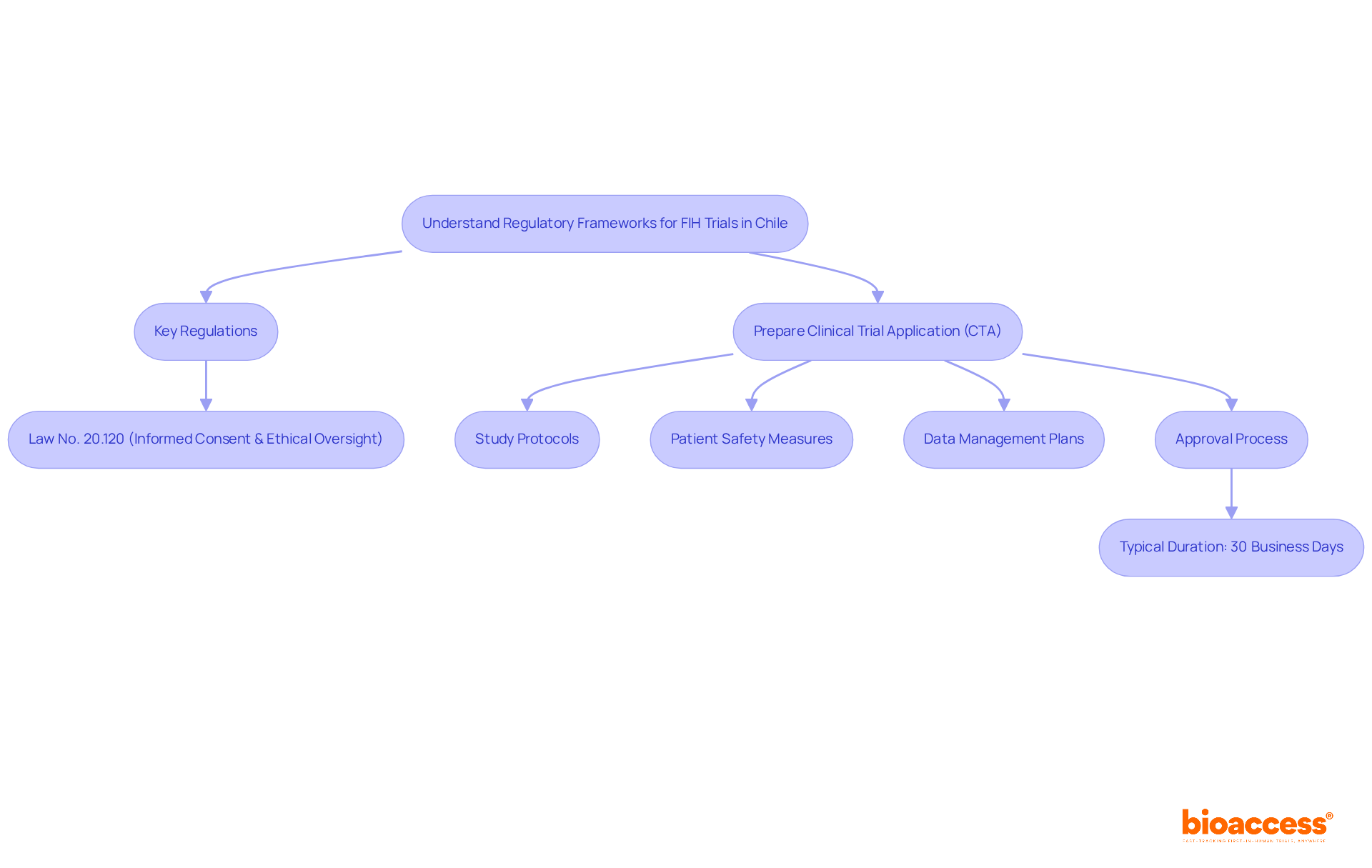
Understand Regulatory Frameworks for FIH Trials in Chile
Understanding the regulatory landscape for first in human biopharma Chile is crucial for sponsors aiming to expedite their clinical research efforts. Conducting FIH studies requires a thorough understanding of the frameworks established by the Instituto de Salud Pública (ISP) and strict adherence to ICH-GCP standards. The approval process is efficient, typically taking around 30 business days, positioning Chile as an attractive destination for sponsors seeking expedited timelines.
Key regulations include:
- Law No. 20.120, which emphasizes the necessity of informed consent and ethical oversight through accredited Scientific Ethics Committees (CECs).
However, many sponsors find the regulatory landscape daunting due to its complexity and the need for meticulous preparation. To navigate this landscape effectively, sponsors need to prepare a detailed Clinical Trial Application (CTA). This should include:
- Thorough study protocols
- Patient safety measures
- Robust data management plans
Acquaintance with these guidelines not only simplifies the approval process but also enhances the credibility of study data, which is crucial for subsequent submissions to authorities such as the FDA. As of 2024, Chile ranks fourth among Latin American nations for research studies, highlighting its growing importance. This shift in research activity underscores the need for sponsors to act swiftly and strategically to capitalize on Chile’s favorable conditions for successful first in human biopharma Chile studies.
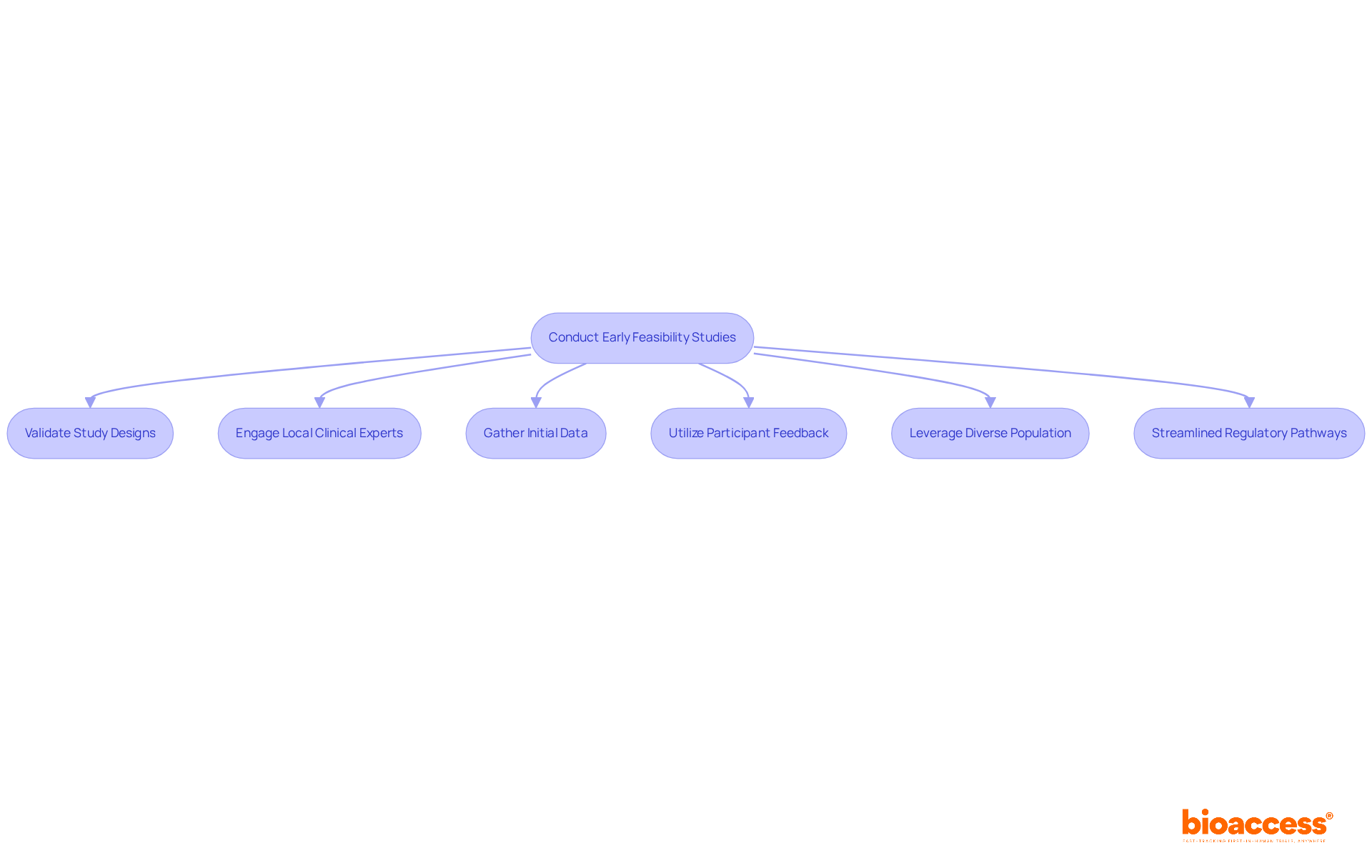
Conduct Early Feasibility Studies to Validate Trial Designs
Early feasibility studies (EFS) are not just a formality; they are essential for validating study designs and ensuring project success in clinical research. In Chile, EFS offers vital insights into patient safety and device functionality, contributing to the advances of first in human biopharma Chile. These insights are crucial for navigating the complex compliance landscape. Typically involving a small cohort of participants – often ten or fewer – EFS focus on gathering initial data that can inform larger trials.
To really make EFS work, sponsors need to connect with local clinical experts who know the ins and outs of compliance, including adherence to ICH-GCP standards and the requirements set forth by local authorities like the Instituto de Salud Pública (ISP). Utilizing feedback from participants during these studies is also essential, as it aids in refining protocols and improving the overall quality of the data gathered.
Additionally, leveraging Chile’s diverse population can greatly enhance the generalizability of findings, making the data more reliable for future submissions. The streamlined regulatory pathways in Latin America, along with the capacity to obtain regulatory approval in as few as 4-8 weeks, position EFS as a strategic asset for early-stage trials, particularly as the first in human biopharma Chile. Moreover, sponsors can realize significant cost savings, estimated at $25K per individual, which can be reinvested into research and development or future funding milestones. By leveraging EFS effectively, sponsors can not only enhance their study designs but also secure a competitive edge in the clinical research landscape.
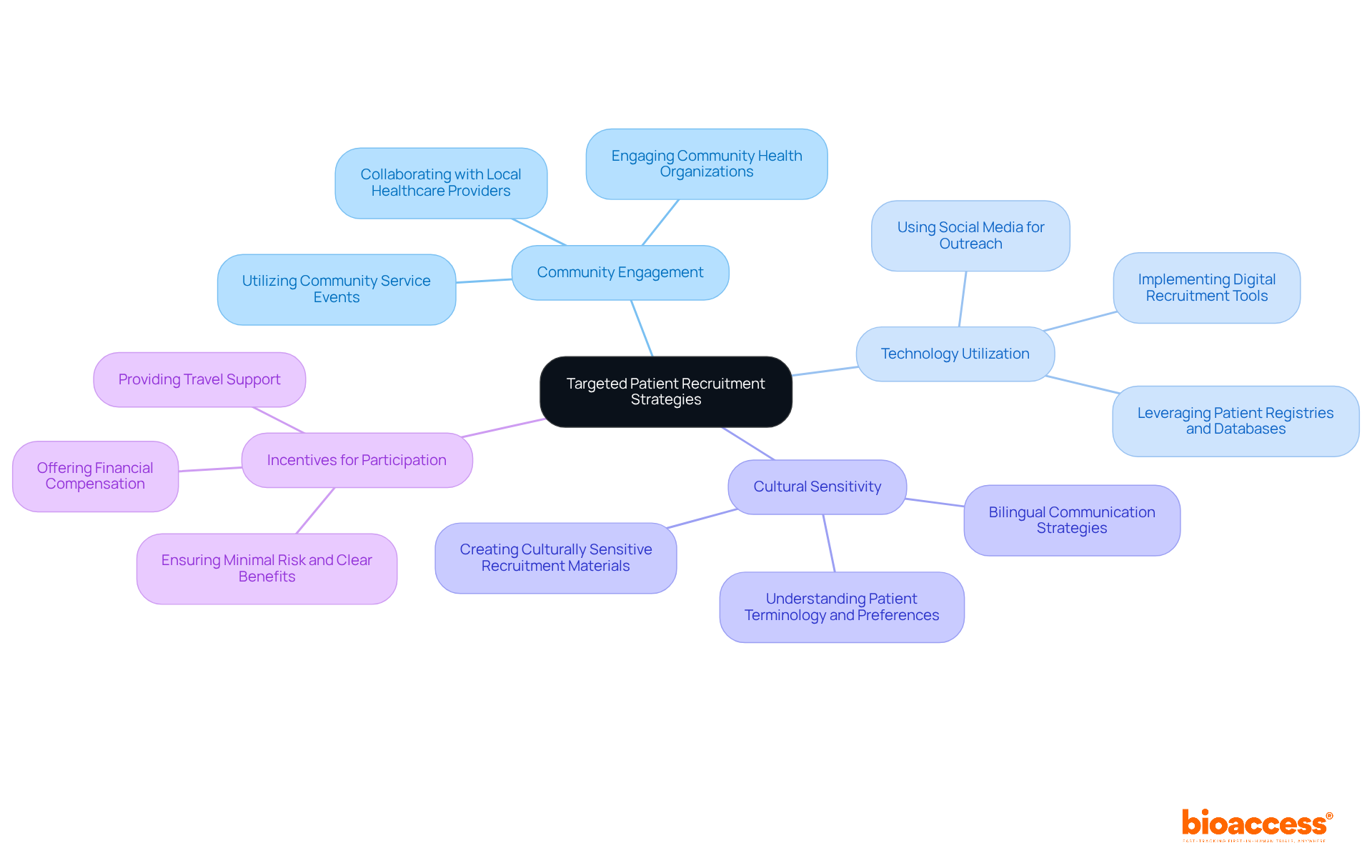
Implement Targeted Patient Recruitment Strategies
Effective recruitment of participants is not just important; it’s essential for the success of first in human biopharma Chile studies. Sponsors should implement targeted strategies that resonate with the local population, including engaging community health organizations and local healthcare providers. Utilizing social media platforms for outreach can significantly enhance visibility and participation. Offering incentives for participation, such as financial compensation for time and travel, can further motivate potential participants. Additionally, ensuring that recruitment materials are culturally sensitive and accessible is crucial for maximizing enrollment rates.
Patient registries and databases can help identify individuals who meet study criteria. This enhances the recruitment process. In Chile, where 37% of study centers fail to meet their recruitment targets, these strategies are vital for achieving enrollment goals. Without effective recruitment, studies may face delays or fail altogether. Furthermore, the integration of bilingual U.S. board-certified physicians in the recruitment process enhances communication and trust, which are critical for participant retention. By implementing these thorough recruitment strategies, sponsors can guarantee a diverse and representative sample, ultimately enhancing the credibility of study results.
At bioaccess®, our founders-distinguished specialists like Dr. Martinez-Clark and Dr. O’Neill-bring their extensive expertise to expedite studies and tackle compliance hurdles in Latin America. Their dedication to closing the divide between medical innovation and research ensures that our clients can efficiently carry out studies in first in human biopharma Chile, benefiting from the strategic advantages of the region. Furthermore, adherence to local oversight bodies such as INVIMA is crucial to navigate the operational landscape effectively, ensuring that all recruitment strategies align with compliance requirements. By prioritizing these strategies, sponsors can not only enhance enrollment but also contribute to the future of medical research in Latin America.
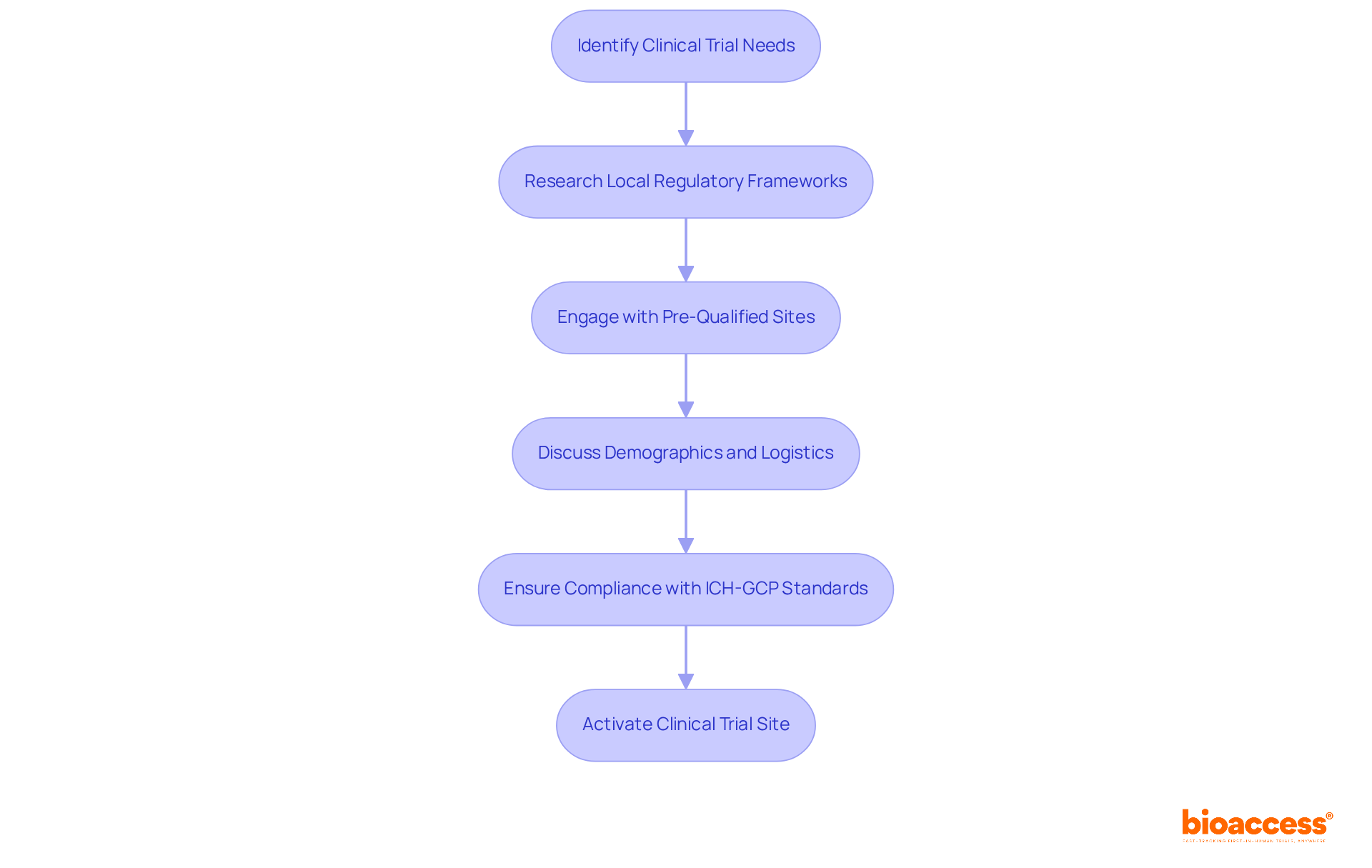
Select and Activate Clinical Trial Sites Efficiently
Selecting the right clinical trial sites is crucial for the success of first in human biopharma Chile trials, yet many sponsors overlook this vital step. Sponsors should prioritize sites that have a solid understanding of local regulatory frameworks and a proven track record in conducting FIH studies, especially those that are first in human biopharma Chile and governed by the Instituto de Salud Pública (ISP). Engaging with these sites early in the planning phase leads to smoother activation. It opens the door for discussions about demographics, logistical considerations, and resource availability. Utilizing a network of pre-qualified sites can significantly expedite the activation process, reducing the time to first patient in, which typically lasts about 30 days according to industry standards.
In Latin America, the benefits of conducting studies related to first in human biopharma Chile are substantial, with accelerated ethics approvals often achievable within 4-8 weeks. Make sure that sites are well-staffed and equipped to meet the study’s specific requirements and comply with ICH-GCP standards. Furthermore, sponsors should be aware of the submission routes for approvals, including the necessary documentation and timelines for compliance with the ISP and other relevant authorities such as INVIMA, ANVISA, and COFEPRIS. By concentrating on efficient site selection and activation strategies, sponsors can markedly enhance the overall timeline and success rate of their clinical trials, ultimately leading to faster regulatory approvals and improved patient outcomes.
Conclusion
Navigating the complexities of first in human biopharma trials in Chile can be daunting, yet it presents unique opportunities for those who are prepared. A strategic approach is essential, encompassing:
- Understanding regulatory frameworks
- Conducting early feasibility studies
- Implementing effective patient recruitment strategies
- Selecting optimal clinical trial sites
These best practices help streamline the research process. They also position sponsors to leverage Chile’s advantageous clinical landscape, characterized by efficient approval timelines and a growing reputation as a hub for biopharma research in Latin America.
Key insights from this article highlight the importance of familiarizing oneself with the regulatory environment established by the Instituto de Salud Pública (ISP) and adhering to ICH-GCP standards. Early feasibility studies play a critical role in validating trial designs and ensuring patient safety. Targeted recruitment strategies are essential for achieving enrollment goals. Additionally, the careful selection and activation of clinical trial sites can significantly enhance the speed and success of studies, ultimately leading to faster regulatory approvals and improved patient outcomes.
In conclusion, the strategic advantages of conducting first in human biopharma trials in Chile cannot be overstated. By embracing these best practices, sponsors can navigate the regulatory landscape more effectively and capitalize on the region’s unique strengths, such as diverse patient populations and rapid approval processes. This proactive approach is key to advancing medical innovation and making sure clinical research in Latin America thrives, ultimately benefiting the global healthcare community.
Frequently Asked Questions
What is the importance of understanding regulatory frameworks for first in human (FIH) trials in Chile?
Understanding the regulatory landscape in Chile is crucial for sponsors to expedite their clinical research efforts, as it allows for efficient navigation of the approval process established by the Instituto de Salud Pública (ISP) and adherence to ICH-GCP standards.
How long does the approval process for FIH trials typically take in Chile?
The approval process for FIH trials in Chile typically takes around 30 business days, making it an attractive destination for sponsors seeking expedited timelines.
What key regulations must sponsors be aware of when conducting FIH trials in Chile?
Key regulations include Law No. 20.120, which emphasizes the necessity of informed consent and ethical oversight through accredited Scientific Ethics Committees (CECs).
What are the essential components of a Clinical Trial Application (CTA) for FIH studies in Chile?
A detailed Clinical Trial Application (CTA) should include thorough study protocols, patient safety measures, and robust data management plans.
How does familiarity with regulatory guidelines benefit sponsors conducting FIH trials in Chile?
Familiarity with regulatory guidelines simplifies the approval process and enhances the credibility of study data, which is crucial for subsequent submissions to authorities such as the FDA.
What is Chile’s current ranking in Latin America regarding research studies as of 2024?
As of 2024, Chile ranks fourth among Latin American nations for research studies, highlighting its growing importance in the field.
Why is it essential for sponsors to act swiftly and strategically in Chile’s regulatory environment?
The shift in research activity towards Chile underscores the need for sponsors to capitalize on the favorable conditions for successful first in human biopharma studies, ensuring they can leverage the country’s advantages in speed, cost efficiency, and patient recruitment.
List of Sources
- Understand Regulatory Frameworks for FIH Trials in Chile
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Clinical trials by start date Chile 1999-2024| Statista (https://statista.com/statistics/1560150/chile-number-new-clinical-trials?srsltid=AfmBOooTXyjSwEH4_QafBwJInZYKr08bzEQj6pveus93SvpyjGJzdo9s)
- Navigate First in Human Medical Device Trials in Chile: A Step-by-Step Guide | bioaccess® (https://bioaccessla.com/blog/navigate-first-in-human-medical-device-trials-in-chile-a-step-by-step-guide)
- Master Early Phase Clinical Trials in Chile: Key Strategies and Insights | bioaccess® (https://bioaccessla.com/blog/master-early-phase-clinical-trials-in-chile-key-strategies-and-insights)
- Conduct Early Feasibility Studies to Validate Trial Designs
- Inderes — Connecting Investors and Companies (https://inderes.dk/en/articles/probabilities-of-success-in-drug-development)
- Early feasibility studies on devices: “doing it sooner” to avoid trial failure | Meditrial (https://meditrial.net/2022/09/early-feasibility-studies-on-devices-doing-it-sooner-to-avoid-trial-failure)
- Why Are Clinical Development Success Rates Falling? (https://insights.citeline.com/IV154612/Why-Are-Clinical-Development-Success-Rates-Falling)
- Early Feasibility Studies: Top 6 Considerations | MED Institute (https://medinstitute.com/blog/early-feasibility-studies-top-6-considerations)
- Implement Targeted Patient Recruitment Strategies
- Patient Recruitment and Retention in Clinical Trials: Strategies and Challenges (https://mdgroup.com/blog/patient-recruitment-and-retention-in-clinical-trials-strategies-and-challenges)
- 3 Patient Recruitment Strategies in Clinical Trials (https://clariness.com/resource/3-patient-recruitment-strategies-in-clinical-trials)
- Successes and Challenges in Clinical Trial Recruitment: The Experience of a New Study Team – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC11348161)
- Latin America: A Compelling Region To Conduct Your Clinical Trials (https://clinicalleader.com/doc/latin-america-a-compelling-region-to-conduct-your-clinical-trials-0001)
- Patient Recruitment for Clinical Trials: Strategies That Actually Work (https://kapsuletech.com/blog/patient-recruitment-clinical-trials)
- Select and Activate Clinical Trial Sites Efficiently
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- Clinical trials by start date Chile 1999-2024| Statista (https://statista.com/statistics/1560150/chile-number-new-clinical-trials?srsltid=AfmBOorIgx9EusEA1UXDnMlDFfoY348h5b3ULrkxL-dycroOn2uLWUjd)
- Master Early Phase Clinical Trials in Chile: Key Strategies and Insights | bioaccess® (https://bioaccessla.com/blog/master-early-phase-clinical-trials-in-chile-key-strategies-and-insights)
- 70 Research Quotes to Inspire Your Work – Qualtrics (https://qualtrics.com/articles/strategy-research/research-quotes)
- Clinical trials: Investment in Chile grew by 62% in three years (https://blog.investchile.gob.cl/clinical-trials-investment-in-chile)

Leave a Reply