Introduction
In Chile, the clinical trial landscape is a complex arena filled with both significant opportunities and formidable challenges for sponsors. With a regulatory framework that emphasizes participant rights and safety, understanding these requirements can significantly streamline the approval process and enhance data credibility.
So, what strategies can sponsors use to tackle these hurdles and ensure their trials succeed in this ever-changing landscape? By exploring best practices for clinical trial sites in Chile, sponsors can uncover actionable insights that pave the way for efficient operations and better outcomes in the growing Latin American market.
Understand Regulatory Requirements for Clinical Trials in Chile
Navigating the regulatory landscape in Chile is essential for successful research at a clinical trial site in Chile, yet many sponsors face significant hurdles. To effectively carry out research studies at a clinical trial site in Chile, it is crucial to navigate the regulatory framework established by the Instituto de Salud Pública (ISP) and other pertinent authorities. A cornerstone of this framework is Law No. 20.120, which governs human research and outlines the ethical considerations and requirements for studies. This law guarantees that proceedings are carried out with the utmost respect for participant rights and safety.
Additionally, Decree No. 114 (2010) offers extensive guidelines on the execution of studies, highlighting the importance of ethical committee approval before starting any research. The approval process for clinical trial site in Chile usually takes between 30 and 90 days. This timeframe depends on the study’s complexity and how complete the submitted documentation is. To facilitate a smooth approval process, it is crucial to prepare a thorough Clinical Trial Application (CTA) that includes:
- Study protocol
- Informed consent forms
- Investigator brochures
- Safety and efficacy data
It’s essential to comply with ICH-GCP standards, ensuring that trials are conducted ethically and that data integrity is upheld. Have you considered that understanding these regulatory requirements can streamline the approval process and enhance the credibility of your data for future submissions, including to the FDA?
Furthermore, the Chilean supply and logistics market for medical research is anticipated to reach USD 71.5 million by 2030, emphasizing the increasing significance of logistics and distribution in the supply chain for medical studies. Conducting studies at a clinical trial site in Chile can be up to 30% more affordable than in North America or Europe. The country boasts high retention rates, surpassing 85% in research assessments. However, sponsors should also be mindful of potential challenges in the approval process, such as the need for thorough documentation and timely communication with oversight bodies. By leveraging the insights and market access strategies provided by bioaccess® through their Global Trial Accelerators™, sponsors can navigate these challenges effectively. This strategic approach positions Chile as an appealing clinical trial site in Chile for early-stage studies, capitalizing on its efficient regulatory pathways and robust healthcare infrastructure. By embracing these strategies, sponsors can not only streamline their research efforts but also unlock the potential of Chile’s burgeoning clinical landscape.
Evaluate Site Selection Criteria and Performance Metrics
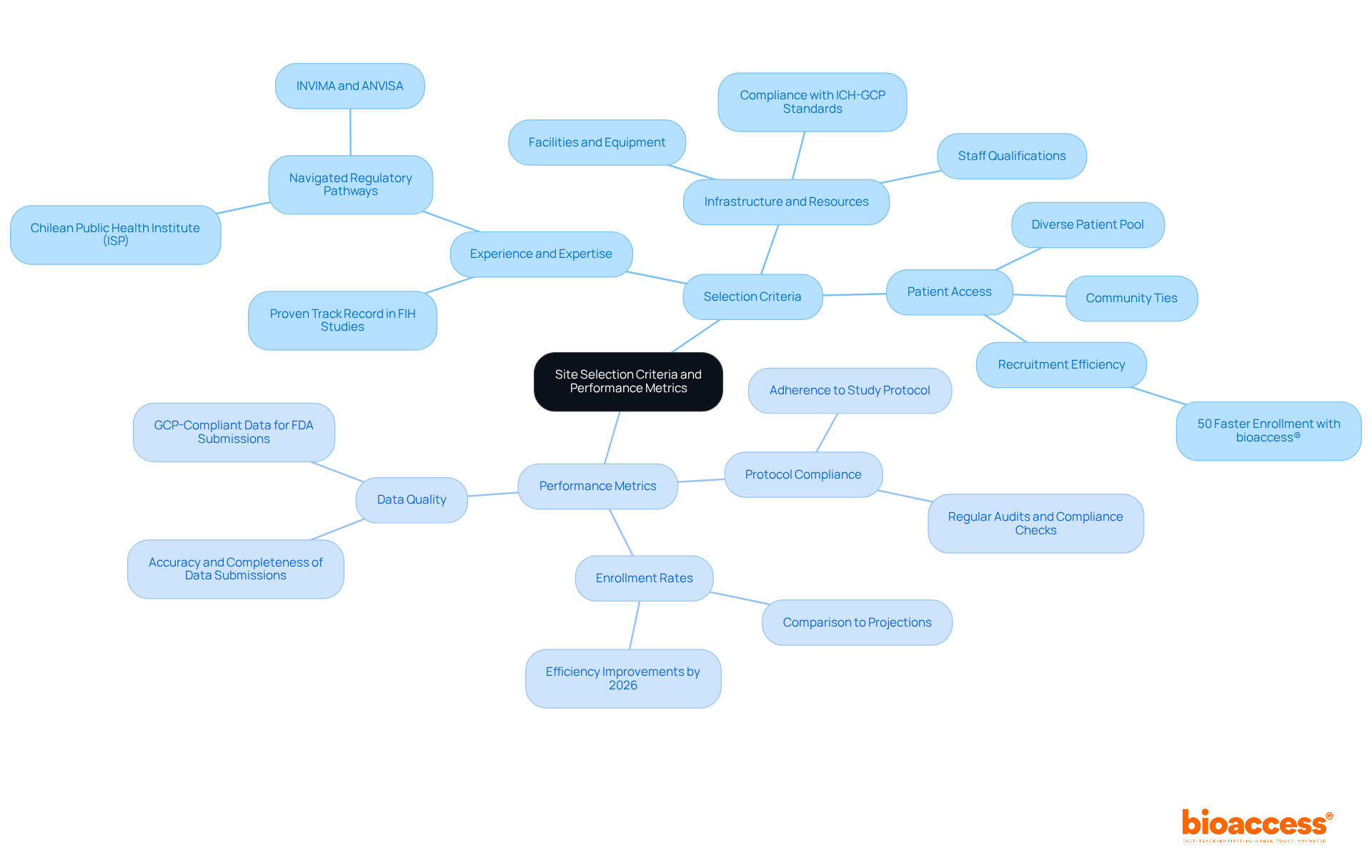
Selecting the right clinical trial site in Chile is crucial for the success of first-in-human (FIH) studies in the dynamic landscape of Chile. Key criteria to consider include:
- Experience and Expertise: Prioritize sites with a proven track record in conducting FIH studies, particularly within the MedTech and Biopharma sectors. Sites that have effectively navigated regulatory pathways with authorities such as the Chilean Public Health Institute (ISP) and other pertinent bodies like INVIMA and ANVISA will be more skilled at managing the complexities of a clinical trial site in Chile.
- Infrastructure and Resources: It’s essential to evaluate the facilities, equipment, and staff qualifications to ensure they meet your study’s demands. Adequate infrastructure is vital for compliance with ICH-GCP standards and for facilitating efficient trial execution.
- Patient Access: How can a facility’s community ties and diverse patient pool enhance your recruitment efforts? Evaluate the facility’s capability to effectively recruit participants, considering local demographics and common health conditions pertinent to the study. A location with strong community ties and a diverse patient pool can significantly enhance recruitment efforts. With bioaccess®, companies can benefit from 50% faster enrollment through pre-qualified networks, tapping into treatment-naïve patient populations that enhance recruitment efficiency.
Performance metrics to monitor include:
- Enrollment Rates: Track how quickly sites can enroll participants compared to projections. In 2026, sponsors will expect locations to document efficiency improvements, including hours spent per enrollment, as a key performance indicator. bioaccess®’s Innovation Runway is designed to facilitate faster enrollment, helping startups reach clinical milestones 40% faster.
- Protocol Compliance: Ensure that sites adhere strictly to the study protocol, which is critical for maintaining data integrity and fulfilling compliance requirements. Regular audits and compliance checks are integral to bioaccess®’s operational strategy.
- Data Quality: Regularly review data submissions for accuracy and completeness. High-quality data is crucial for acceptance by authorities and for supporting future submissions to the FDA or other oversight bodies. bioaccess® leverages GCP-compliant data to ensure that submissions meet FDA requirements, streamlining the pathway for regulatory approvals.
By establishing clear performance metrics and regularly evaluating capabilities, sponsors can enhance their selection process, leading to more efficient execution and improved outcomes. By refining their selection process, sponsors can not only meet enrollment goals but also secure a competitive edge in future studies across Latin America.
Foster Relationships with Clinical Trial Sites for Enhanced Collaboration
To ensure the success of first-in-human studies, establishing robust connections with a clinical trial site in Chile is non-negotiable. Here are strategies to enhance collaboration:
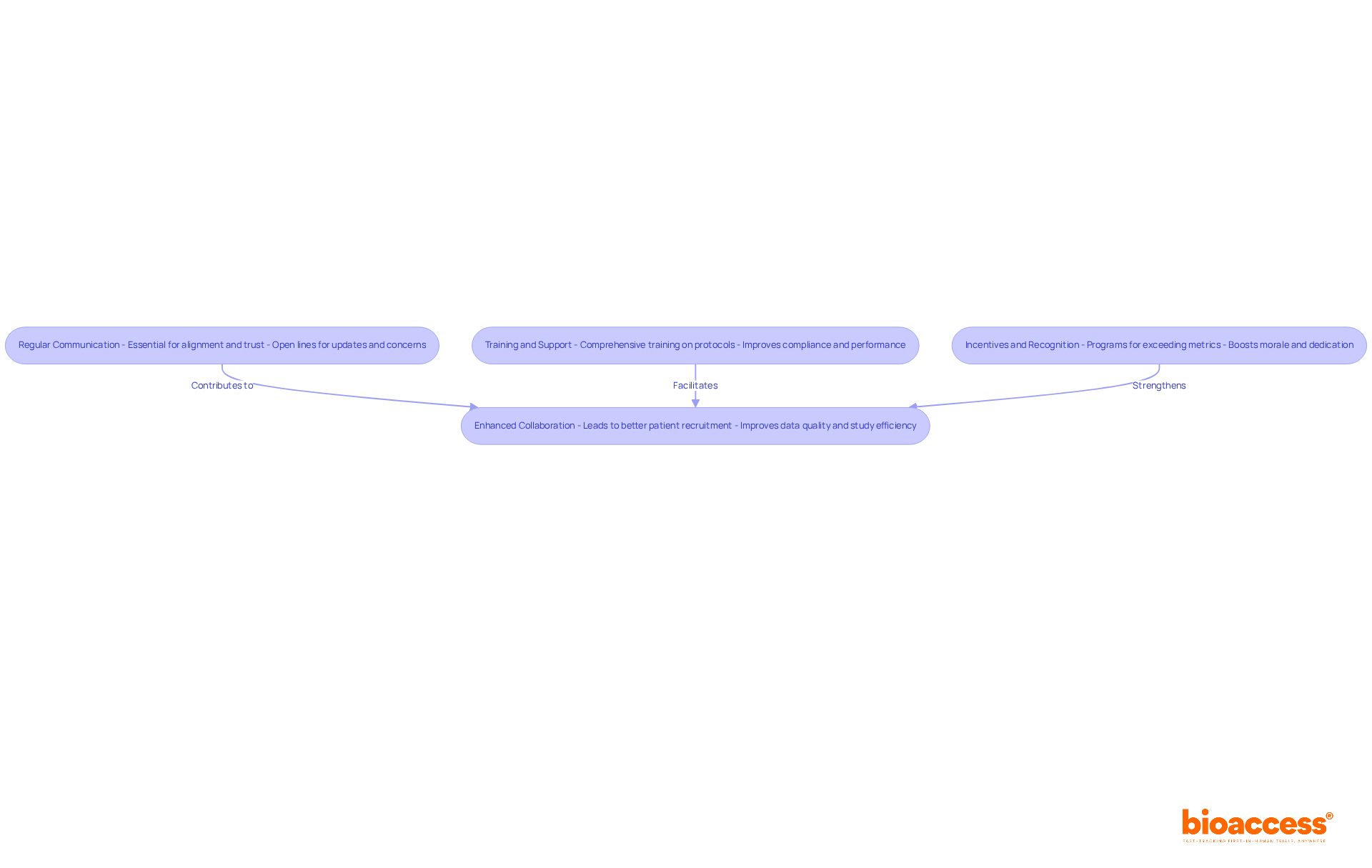
- Regular Communication: Regular communication is essential for maintaining alignment and fostering trust, both of which are crucial for effective collaboration. Establishing open lines of communication with site staff allows for discussions about trial progress, addressing concerns, and sharing updates. As communication specialists emphasize, effective teamwork hinges on communication, underscoring its importance in achieving study objectives. At bioaccess®, we prioritize this communication to ensure that our partners are well-informed and engaged throughout the study process. For instance, one of our clients highlighted how consistent updates helped them navigate challenges effectively, leading to a smoother trial execution.
- Training and Support: How can we ensure that our personnel are fully equipped to meet compliance requirements? Providing comprehensive training sessions on the study protocol is key. This guarantees that everyone is aligned and can perform their roles effectively, ultimately improving performance metrics. Our experience in accelerating clinical trials in Latin America, particularly at clinical trial site Chile, has shown that well-trained staff are essential for navigating the complexities of regulatory compliance and achieving timely approvals. A recent testimonial from a director emphasized that the training provided by bioaccess® significantly improved their team’s understanding of compliance requirements, resulting in faster approval timelines.
- Incentives and Recognition: Implementing incentive programs for locations that meet or exceed performance metrics can inspire staff and strengthen their dedication to the study. Research indicates that companies promoting a collaborative culture are five times more likely to be high-performing, highlighting the value of recognition in fostering a productive environment. At bioaccess®, we honor the achievements of our research partners, emphasizing the significance of their contributions to study success. This recognition not only boosts morale but also cultivates a culture of excellence that drives performance and success in clinical trials.
By fostering a collaborative environment, sponsors can improve site engagement, leading to better patient recruitment and data quality. This collaborative strategy not only improves study efficiency but also establishes a basis for future alliances, guaranteeing that investigations at the clinical trial site Chile are conducted promptly and economically. For instance, RDI achieved 100% of their original and latest enrollment target goals, demonstrating the effectiveness of these strategies in real-world applications, supported by bioaccess®’s expertise in navigating the regulatory landscape.
Implement Effective Patient Recruitment Strategies in Chile
In Chile, the challenge of efficient patient recruitment at a clinical trial site can significantly impact the success of clinical studies, particularly first-in-human research. Here are key strategies to enhance recruitment efforts:
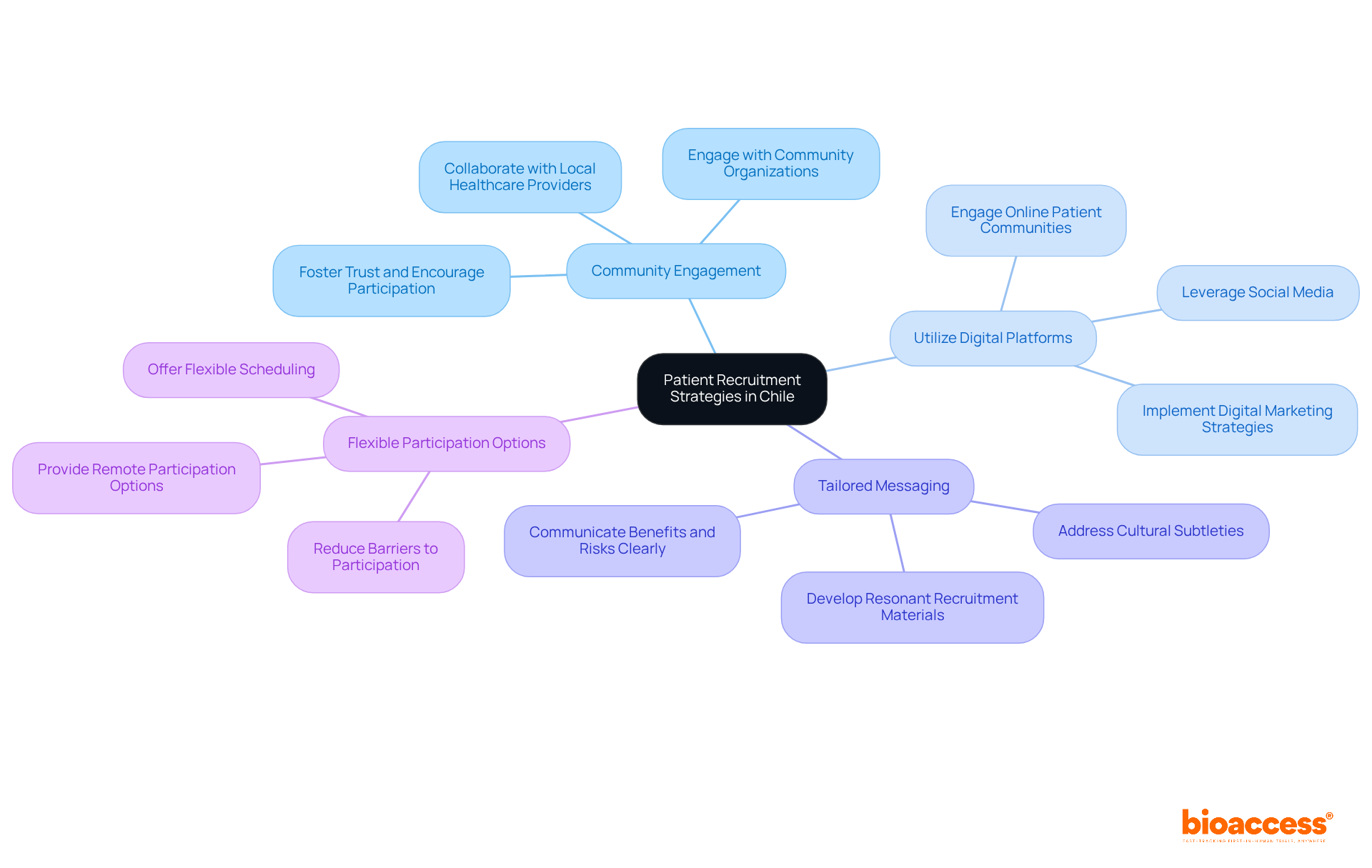
- Community Engagement: Collaborate with local healthcare providers and community organizations to raise awareness about the trial. Engaging with the community fosters trust and encourages participation, which is critical in a region where personal relationships significantly influence patient decisions.
- Utilize Digital Platforms: Leverage social media and online patient communities to reach potential participants. Digital marketing strategies can broaden your reach and attract diverse patient populations, particularly in urban areas where internet access is prevalent.
- Tailored Messaging: Develop recruitment materials that resonate with the target patient population. Clear communication regarding the study’s benefits and risks can alleviate concerns and promote enrollment. This is especially significant in Chile, where cultural subtleties can influence perceptions of research in the medical field.
- Flexible Participation Options: Offer flexible scheduling and remote participation options when possible. This can reduce barriers to participation and improve retention rates, especially for patients who may have difficulty attending in-person visits.
Implementing these strategies allows sponsors to boost patient recruitment efforts. This ensures studies are adequately powered and completed on time. This strategic approach boosts enrollment rates and plays a crucial role in the trial’s overall success at a clinical trial site in Chile, aligning with regulatory requirements established by authorities such as the Instituto de Salud Pública de Chile (ISP) and adhering to ICH-GCP standards. Furthermore, leveraging the rapid ethics approvals and early feasibility studies available in Latin America can lead to faster approval timelines and a more efficient pathway to market for innovative therapies, supported by FDA/EMA-ready clinical data.
Conclusion
Navigating the complexities of clinical trials in Chile can be daunting without a clear strategy that encompasses regulatory understanding and collaboration. By addressing these areas strategically, sponsors can significantly enhance the efficiency and success of their studies, especially for first-in-human trials in the MedTech and Biopharma sectors.
Key strategies discussed include:
- Familiarizing oneself with the essential regulatory framework established by the Instituto de Salud Pública.
- Adhering to ICH-GCP standards to ensure ethical compliance and data integrity.
- Selecting sites based on their experience, infrastructure, and patient access to significantly impact recruitment and overall trial performance.
- Establishing strong relationships with clinical trial sites through effective communication, training, and recognition to foster collaboration that is crucial for timely and successful study execution.
By embracing these strategies, sponsors not only enhance their trial success but also contribute to the growth of innovative therapies in Latin America, shaping the future of healthcare in the region.
Frequently Asked Questions
What are the key regulatory requirements for conducting clinical trials in Chile?
The key regulatory requirements for conducting clinical trials in Chile include compliance with Law No. 20.120, which governs human research and ensures participant rights and safety, as well as obtaining approval from ethical committees as outlined in Decree No. 114 (2010).
What is the typical approval timeline for clinical trials in Chile?
The approval process for clinical trials in Chile typically takes between 30 and 90 days, depending on the complexity of the study and the completeness of the submitted documentation.
What documentation is necessary for a Clinical Trial Application (CTA) in Chile?
A thorough Clinical Trial Application (CTA) in Chile should include the study protocol, informed consent forms, investigator brochures, and safety and efficacy data.
What standards must be followed during clinical trials in Chile?
Clinical trials in Chile must comply with International Conference on Harmonisation – Good Clinical Practice (ICH-GCP) standards to ensure ethical conduct and data integrity.
How can understanding regulatory requirements benefit sponsors conducting trials in Chile?
Understanding regulatory requirements can streamline the approval process and enhance the credibility of data for future submissions, including to the FDA.
What are the advantages of conducting clinical trials in Chile compared to North America or Europe?
Conducting clinical trials in Chile can be up to 30% more affordable than in North America or Europe, and the country has high retention rates, exceeding 85% in research assessments.
What challenges might sponsors face during the approval process in Chile?
Sponsors may face challenges such as the need for thorough documentation and the importance of timely communication with oversight bodies during the approval process.
How can bioaccess® and Global Trial Accelerators™ assist sponsors in navigating the regulatory landscape in Chile?
Bioaccess® and their Global Trial Accelerators™ provide insights and market access strategies that help sponsors effectively navigate regulatory challenges, making Chile an appealing site for early-stage studies.
What is the projected growth of the Chilean supply and logistics market for medical research?
The Chilean supply and logistics market for medical research is anticipated to reach USD 71.5 million by 2030, highlighting the importance of logistics and distribution in the supply chain for medical studies.
List of Sources
- Understand Regulatory Requirements for Clinical Trials in Chile
- Chile Clinical Trial Supply & Logistics Market Size & Outlook, 2030 (https://grandviewresearch.com/horizon/outlook/clinical-trial-supply-logistics-market/chile)
- Master Early Phase Clinical Trials in Chile: Key Strategies and Insights | bioaccess® (https://bioaccessla.com/blog/master-early-phase-clinical-trials-in-chile-key-strategies-and-insights)
- How Chile Is Shaping Medical Device Clinical Trials In Latin America (https://meddeviceonline.com/doc/how-chile-is-shaping-medical-device-clinical-trials-in-latin-america-0001)
- Clinical trials by start date Chile 1999-2024| Statista (https://statista.com/statistics/1560150/chile-number-new-clinical-trials?srsltid=AfmBOoqM-CQfRkaZO9eWlHXHETblV2mY_d-Xkr-4RN9I5wCD0jMwAMiA)
- Evaluate Site Selection Criteria and Performance Metrics
- Clinical Trial Success Rates by Therapeutic Area 2026-27 Data Analysis (https://ccrps.org/clinical-research-blog/clinical-trial-success-rates-by-therapeutic-area-2026-27-data-analysis)
- Top 5 Clinical Trial Enrollment Metrics to Watch in 2026 (https://bekhealth.com/blog/clinical-trial-enrollment-metrics-2026)
- New York Times Article on Clinical Trials Features Quote from the Alliance for Aging Research – Alliance for Aging Research (https://agingresearch.org/blog/new-york-times-article-on-clinical-trials-features-quote-from-the-alliance-for-aging-research)
- Clinical Trial Site Selection: Key Factors & Best Practices | IntuitionLabs (https://intuitionlabs.ai/articles/clinical-trial-site-selection)
- Quotes About Medical Research (https://changingthepresent.org/pages/quotes-about-medical-research)
- Foster Relationships with Clinical Trial Sites for Enhanced Collaboration
- 4 Metrics That Define a High-Performing Clinical Trial Site (https://remdavis.com/news/clinical-trial-site-performance)
- 35 Quotes about Communication to Inspire Collaboration (https://vibe.us/blog/35-quotes-about-communication?srsltid=AfmBOor7xO3dD4gz4K7lOHAtbEtFL2-tIhRFSVmsWSfvsSWfqDXnfEaI)
- Patient Engagement Quotes: For Every Purpose & Audience (https://nclusiv.co.uk/edi-consulting/f/patient-engagement-quotes-for-every-purpose-audience)
- Elevate Training to Overcome Site Burden and Reduce Protocol Deviations by 35-50% | WCG (https://wcgclinical.com/insights/elevate-training-to-overcome-site-burden-and-reduce-protocol-deviations-by-35-50)
- Implement Effective Patient Recruitment Strategies in Chile
- Clinical Trial Patient Recruitment | Curebase (https://curebase.ai/resources/articles/clinical-trial-patient-recruitment-guide)
- Community engagement is key to clinical trial recruitment and diversity (https://statnews.com/2019/08/23/clinical-trial-recruitment-diversity-community-engagement)
- Recruitment Strategies for High-Performing Trials in 2026 | Brian Vogtsberger posted on the topic | LinkedIn (https://linkedin.com/posts/brian-vogtsberger_clinicaltrials-patientrecruitment-siteengagement-activity-7414795069326708736-J0HJ)
- Patient Recruitment for Clinical Trials: Strategies That Actually Work (https://kapsuletech.com/blog/patient-recruitment-clinical-trials)
- Precision, Relevance and Prediction: The Future of Clinical Trial Recruitment (https://gcihealth.com/2026/01/22/precision-relevance-and-prediction-the-future-of-clinical-trial-recruitment)

Leave a Reply