Introduction
In the dynamic realm of clinical research, the significance of trial agreements is paramount. These agreements form the foundation for successful device and drug trials, ensuring compliance and clarity among all stakeholders involved. By delving into ten essential trial agreement templates, readers will discover how these tools can streamline processes, enhance collaboration, and ultimately expedite the journey from innovation to patient access.
What challenges might emerge when navigating the complexities of these agreements? The right templates can provide effective solutions.
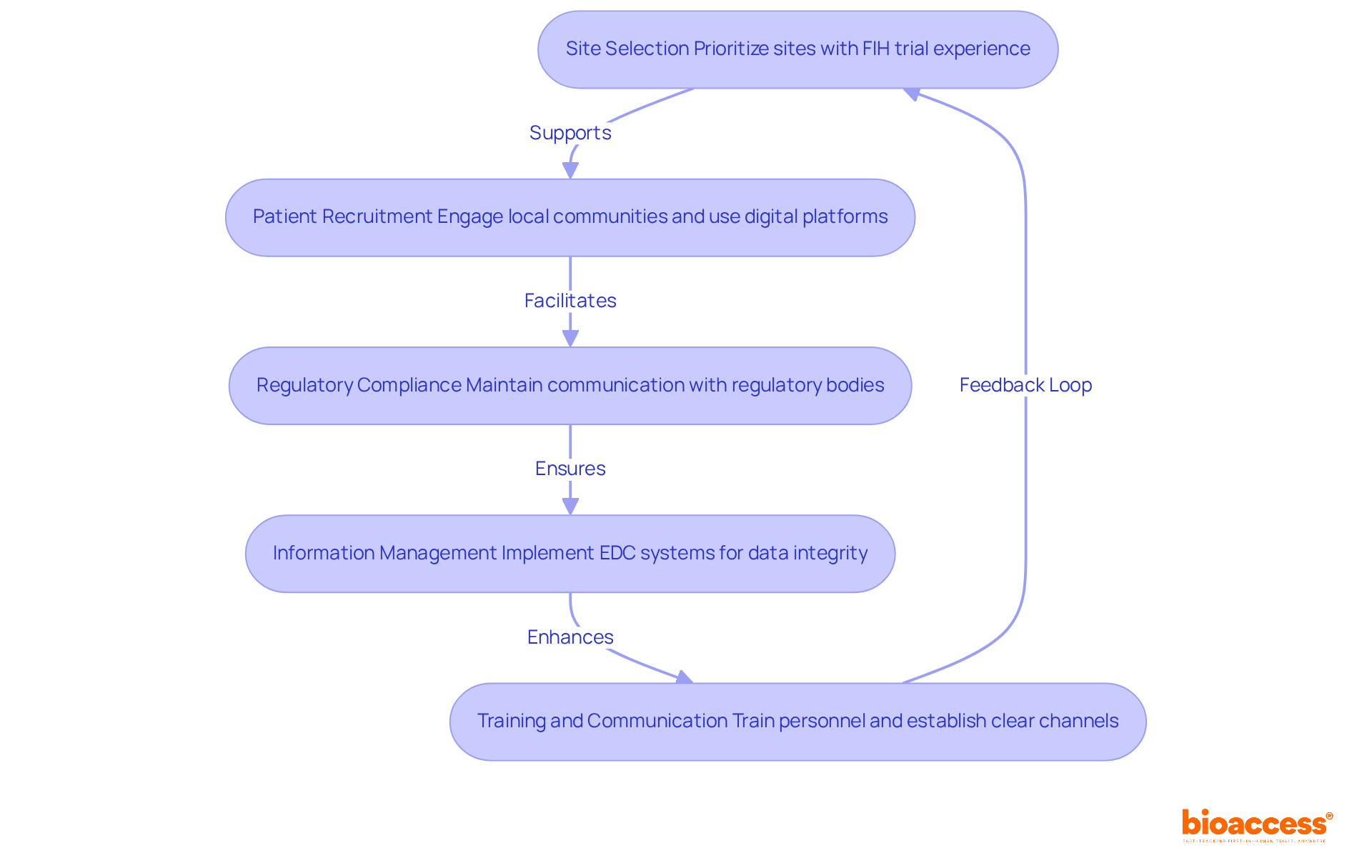
bioaccess® Clinical Trial Agreement Template for Medtech Innovations
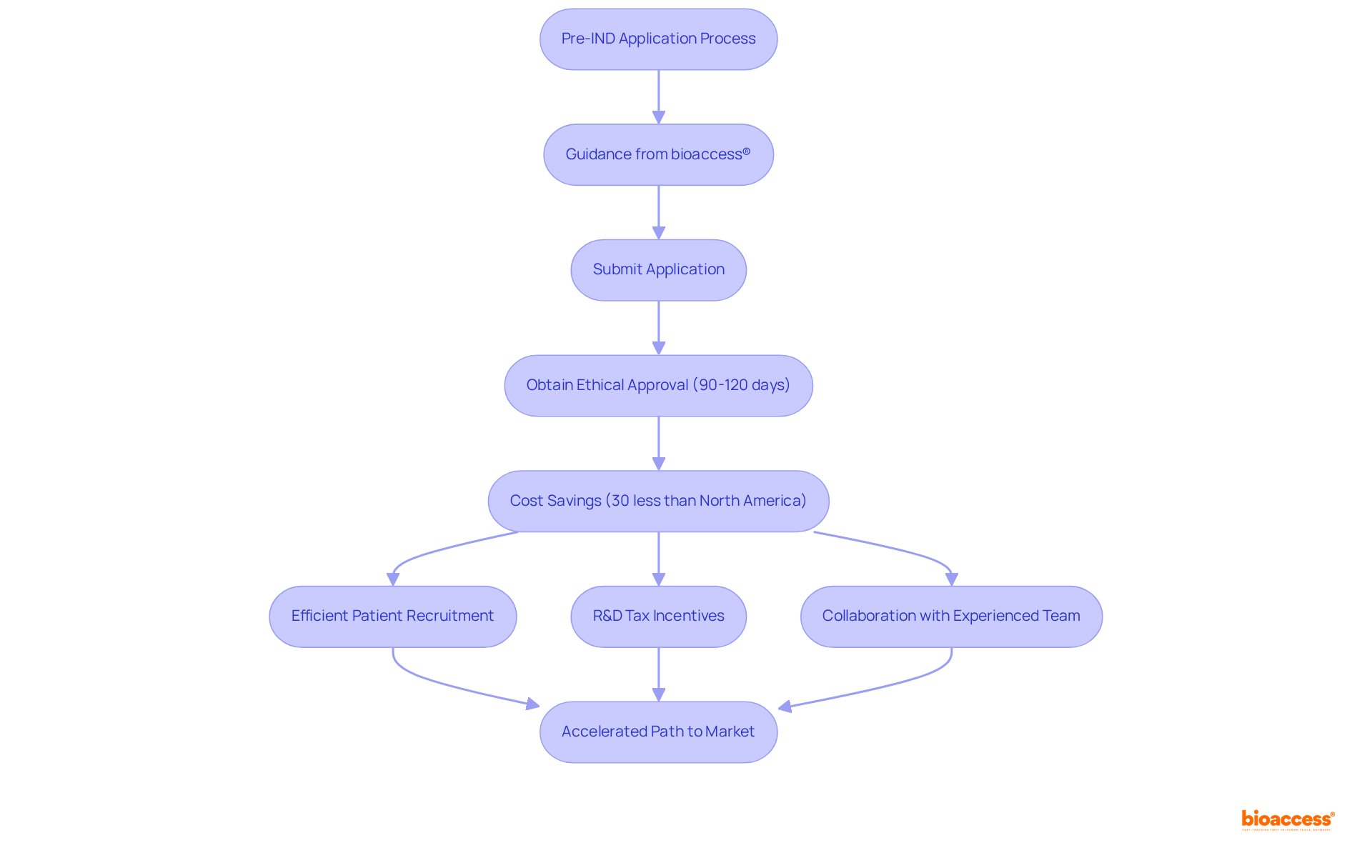
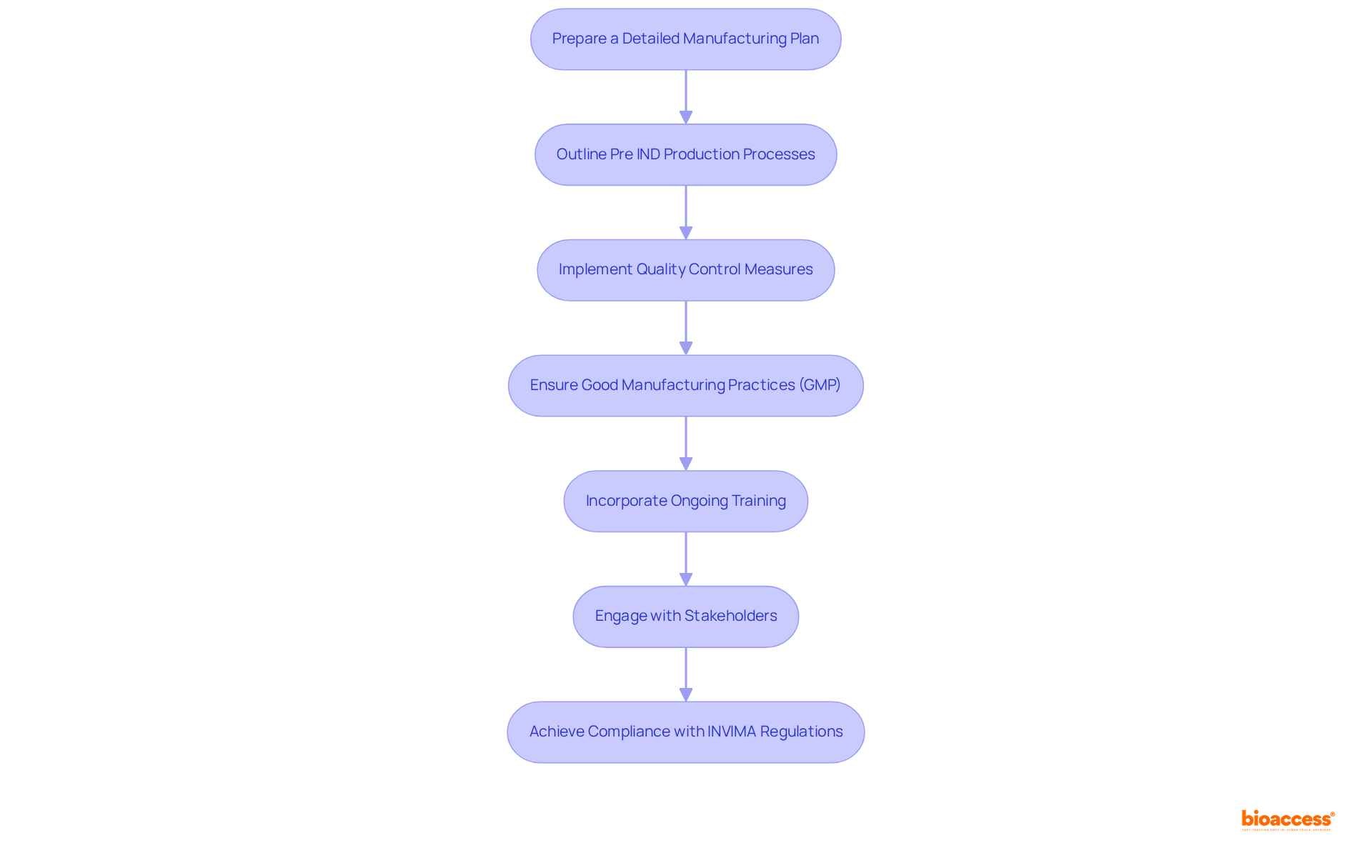
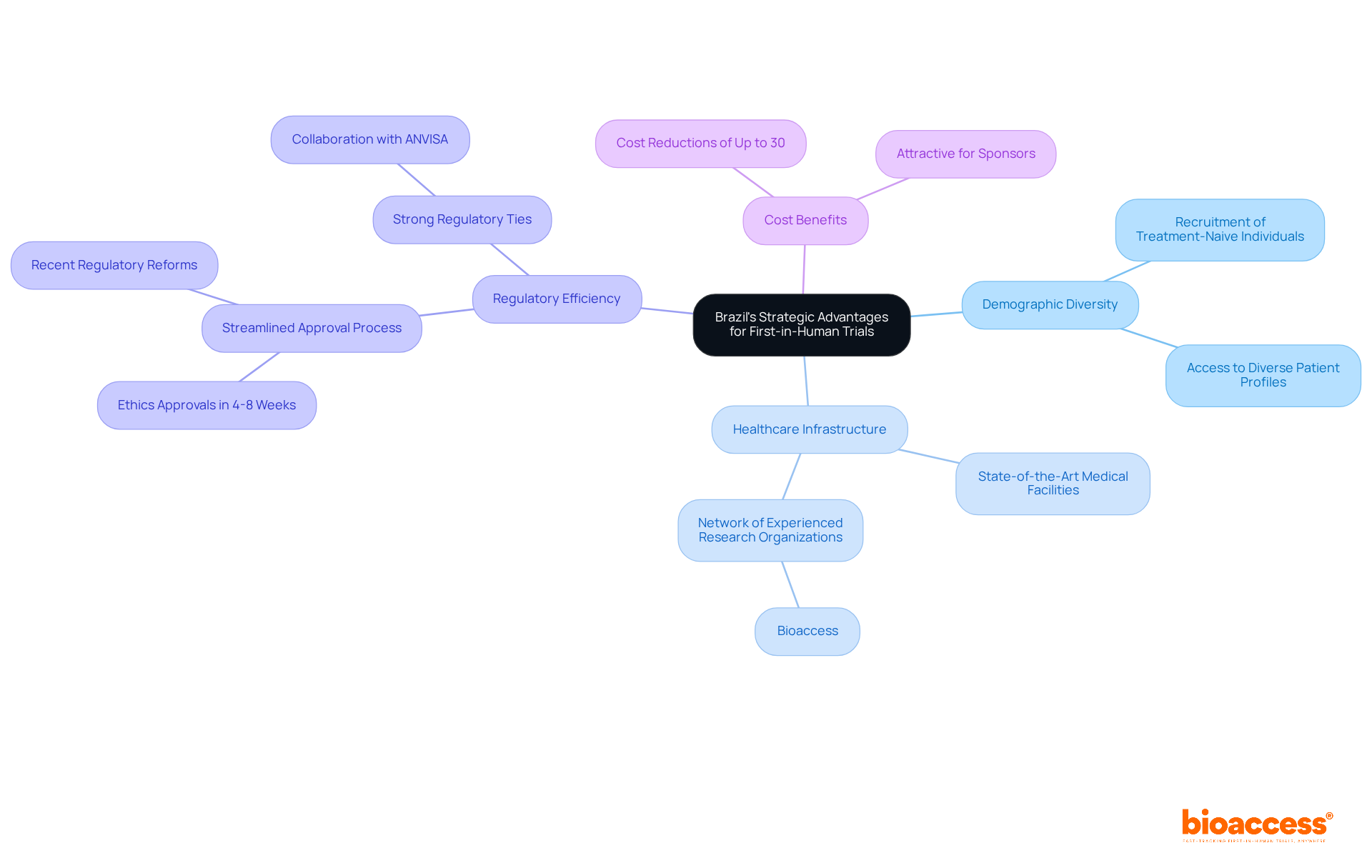
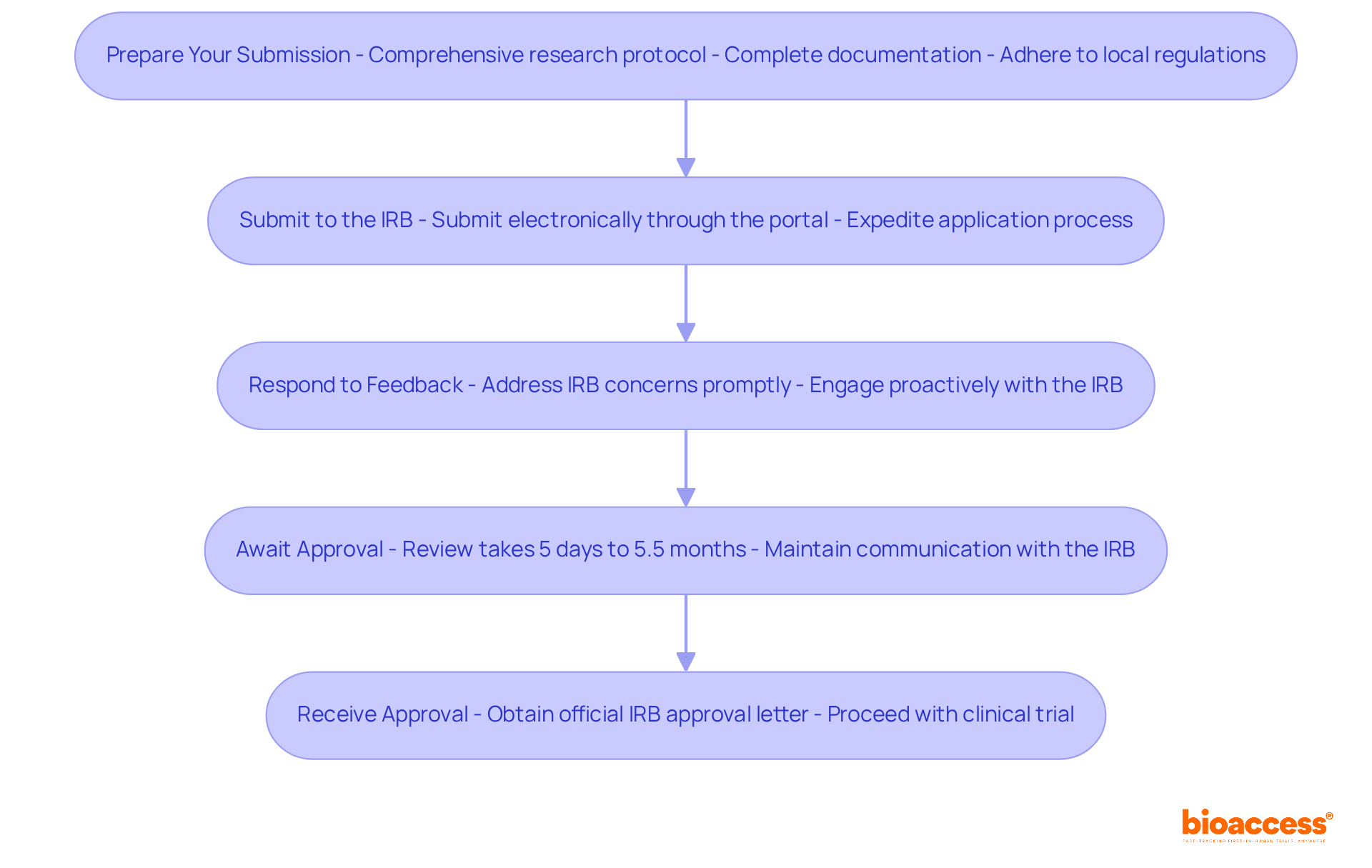
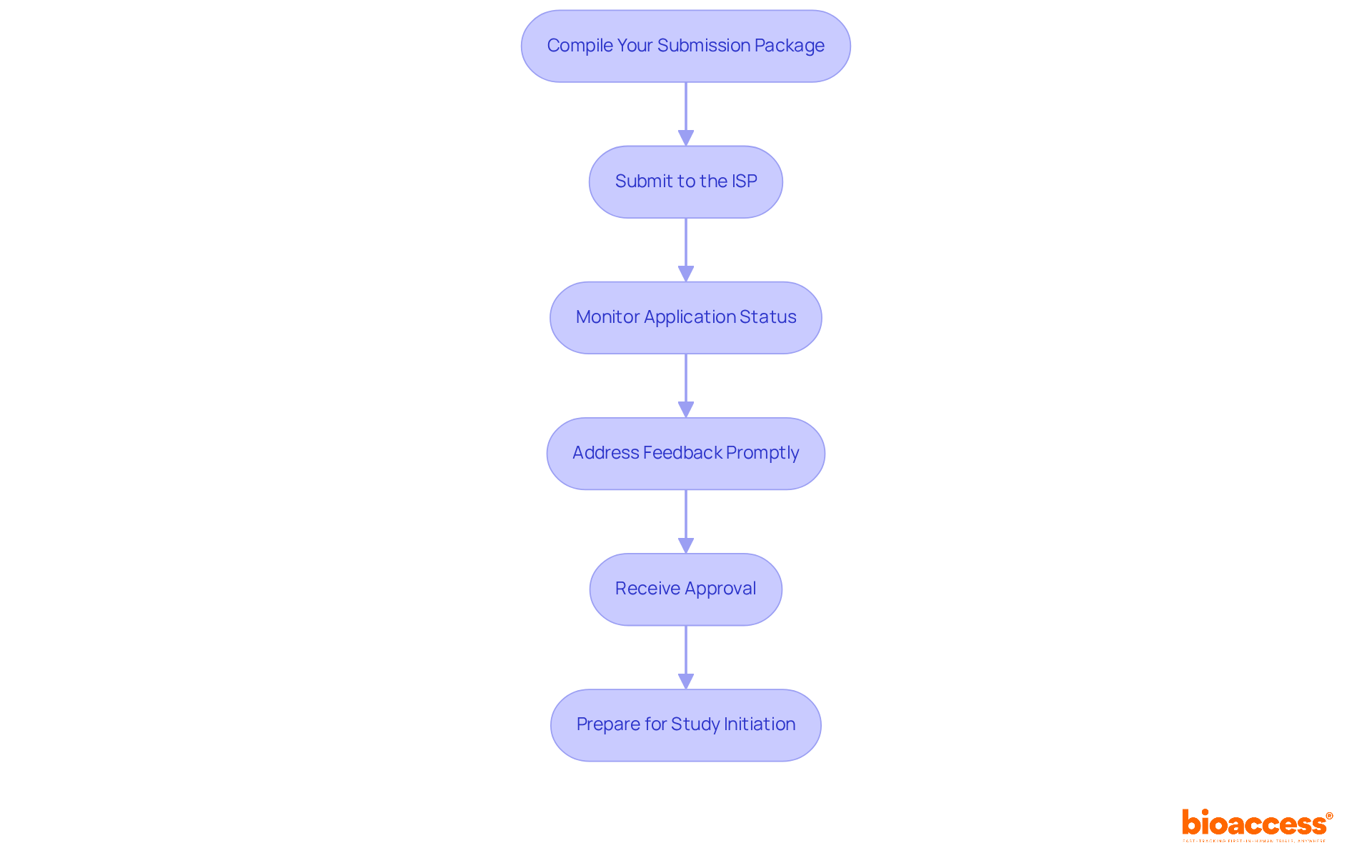
The bioaccess® is a pivotal resource for Medtech innovators, designed to significantly enhance efficiency in by utilizing . This template includes that ensure compliance with local regulations while streamlining the approval process. It clearly delineates roles, responsibilities, and timelines, fostering clarity and .
By leveraging this model, sponsors can anticipate a more efficient negotiation process and quicker study initiation, ultimately leading to to innovative medical devices. This approach aligns with the latest trends in research agreements, underscoring the need for agility and compliance in an increasingly competitive landscape.
With bioaccess®’s extensive expertise in managing Early-Feasibility, , Pilot, Pivotal, and Post-Market Follow-Up Studies, their accelerated patient enrollment process-achieving 50% faster enrollment and $25K savings with FDA-ready data-makes this template an indispensable tool for navigating the complexities of research in .
In a field where collaboration is key, utilizing can empower sponsors to tackle the challenges of head-on.
Ontario Universal Agreement for Clinical Trials (UACT)
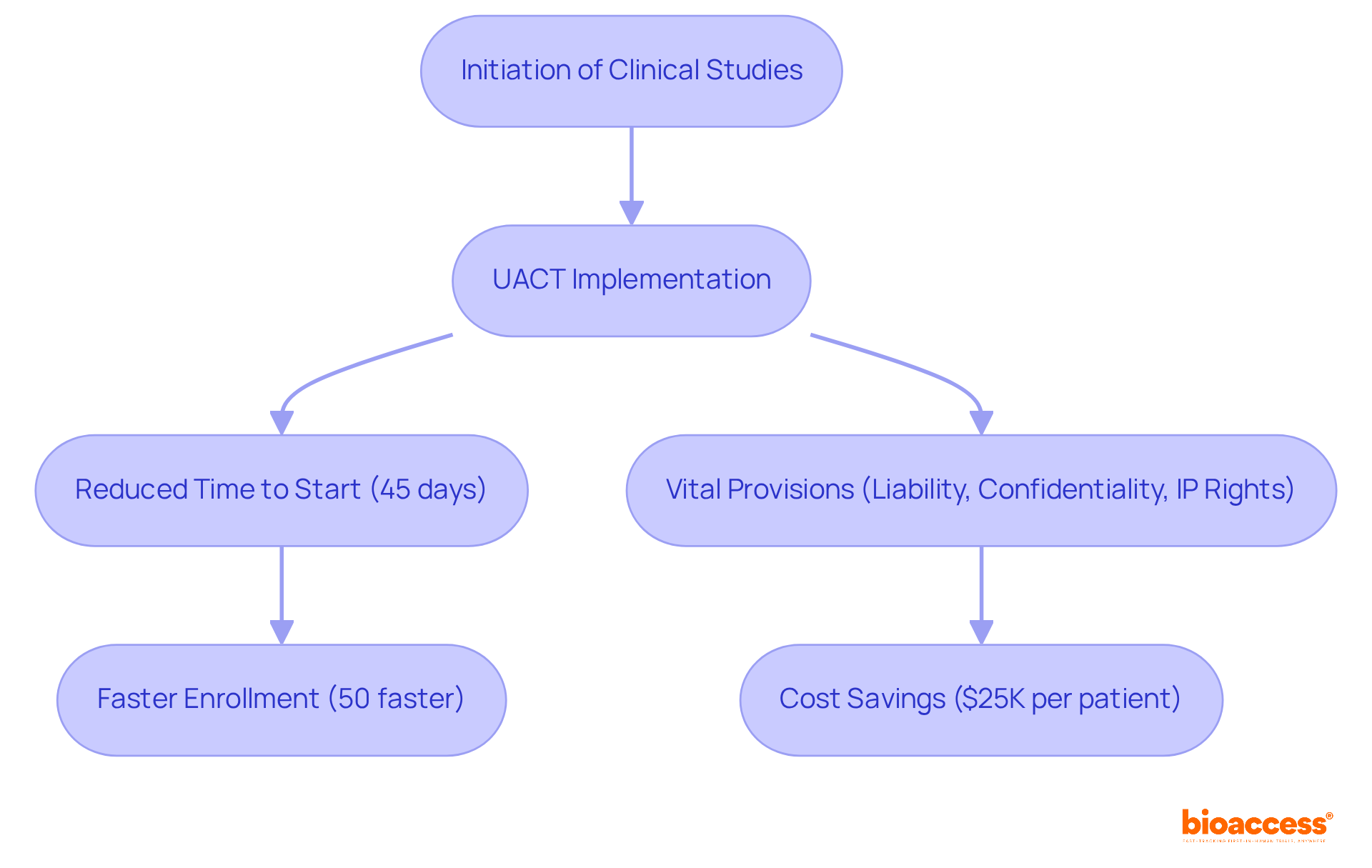
The Ontario Universal Agreement for (UACT) is a pivotal contract designed to streamline the initiation of throughout Ontario. By significantly reducing the time and resources typically associated with contract negotiations, the UACT , allowing studies to commence in as little as 45 days. This agreement includes vital provisions regarding liability, confidentiality, and intellectual property rights, making it an and research institutions.
The implementation of the UACT has led to a remarkable decrease in the time required to initiate studies, fostering quicker connections between patients and innovative therapies. This shift has transformed wait times from months or even years to mere weeks. Currently, 21 organizations have adopted the UACT, reflecting its growing impact on research initiation in Ontario. Furthermore, this collaboration with bioaccess® enhances the enrollment of , achieving a 50% faster enrollment rate compared to traditional Western sites. This efficiency translates into substantial cost savings of $25K per patient, thanks to that eliminates rework and delays.
Ultimately, the synergy between the UACT and bioaccess®’s services not only but also significantly benefits the broader research landscape. As we move forward, embracing such collaborative frameworks will be crucial in addressing the challenges faced in clinical research.
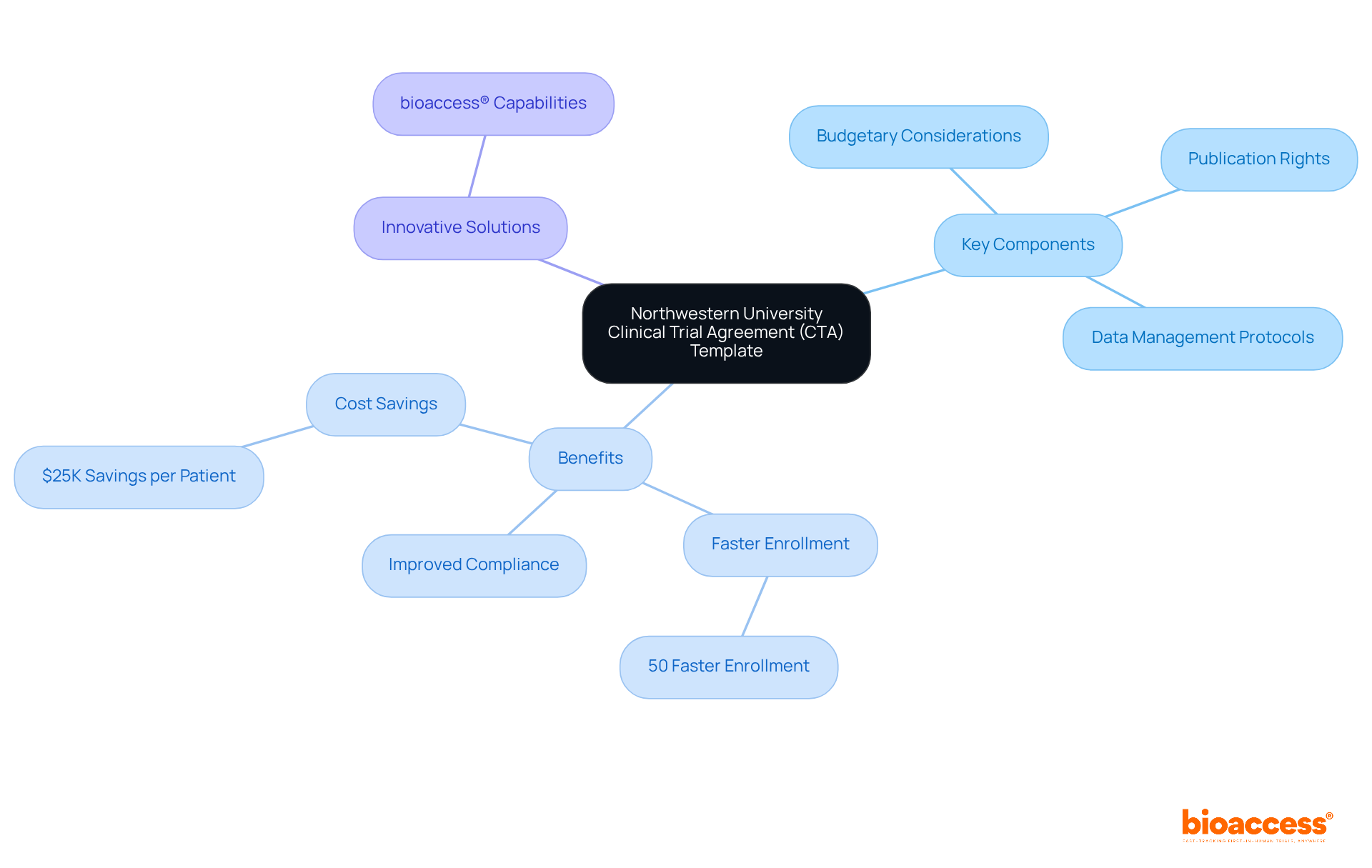
Northwestern University Clinical Trial Agreement (CTA) Template
The serves as a vital document for conducting . It clearly outlines the responsibilities of both the sponsor and the institution, ensuring compliance with federal regulations and institutional policies. Key components of this framework include:
- Budgetary considerations
- Publication rights
- Data management protocols
By utilizing this template, researchers can align their studies with established . This alignment not only improves compliance rates but also promotes . Moreover, leveraging bioaccess®’s capabilities significantly enhances study efficiency. For instance, can be enrolled 50% faster than through conventional methods. This acceleration leads to , with that eliminates rework and delays.
In the evolving Medtech landscape, collaboration is essential. By embracing innovative solutions like the and bioaccess®, researchers can navigate challenges more effectively and drive impactful results.
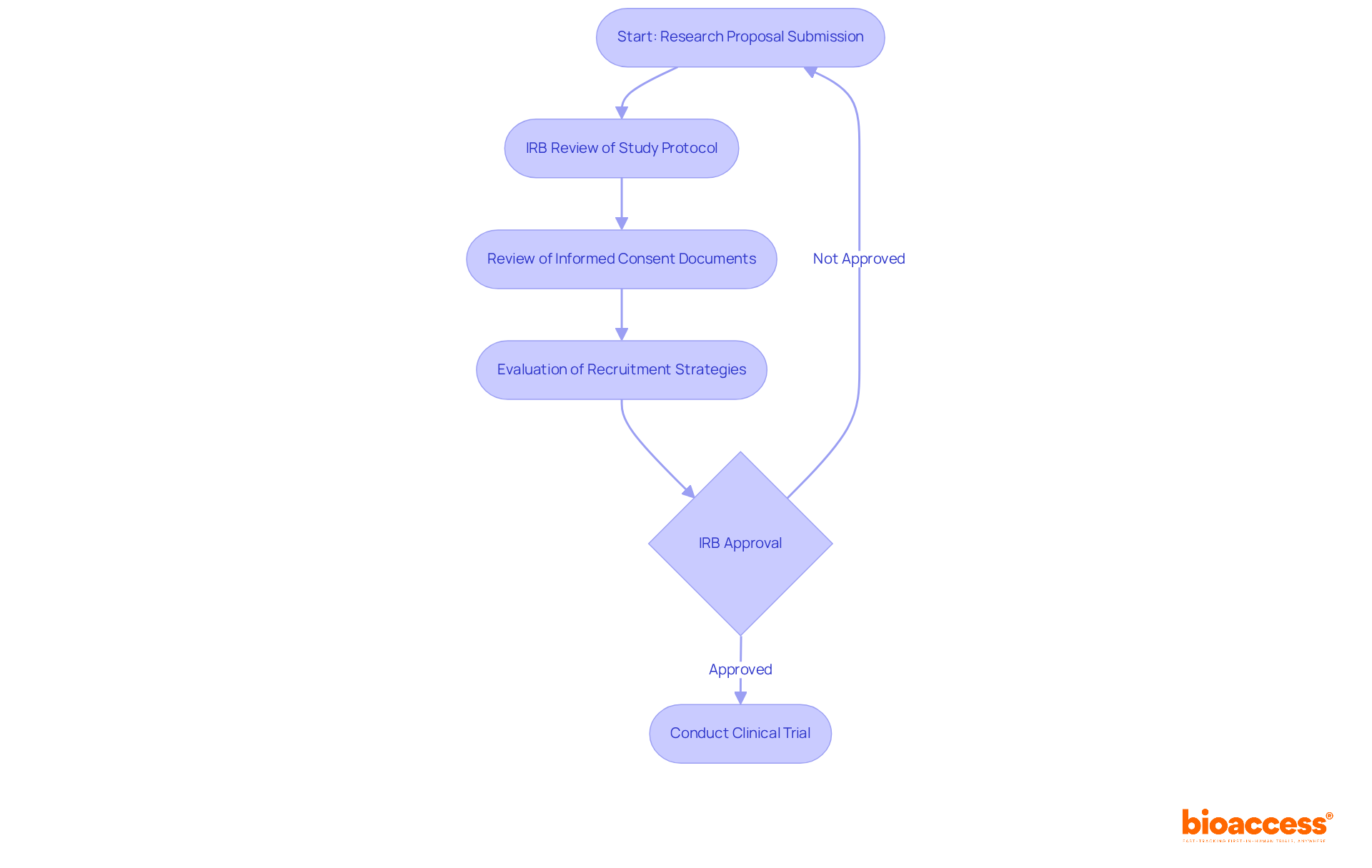
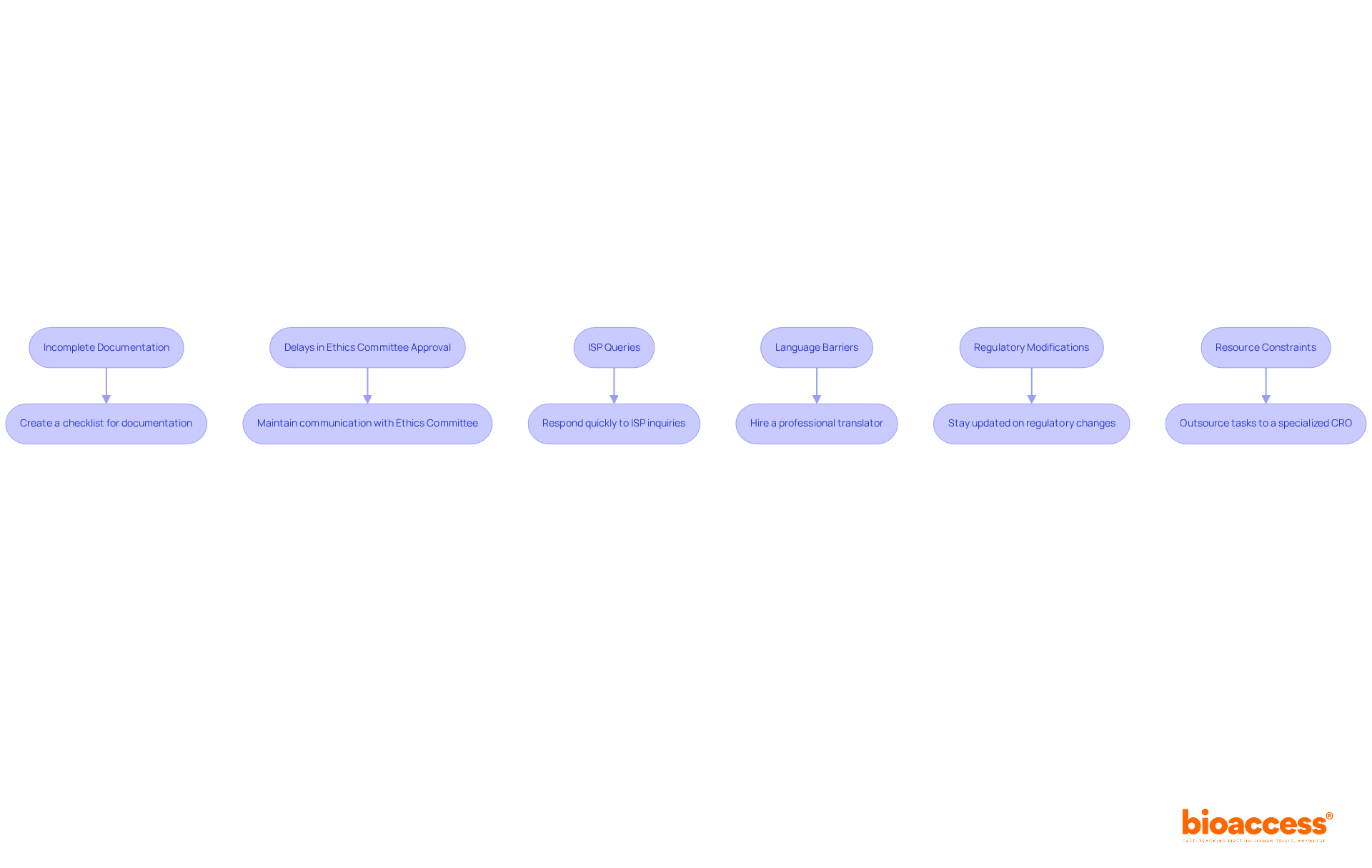
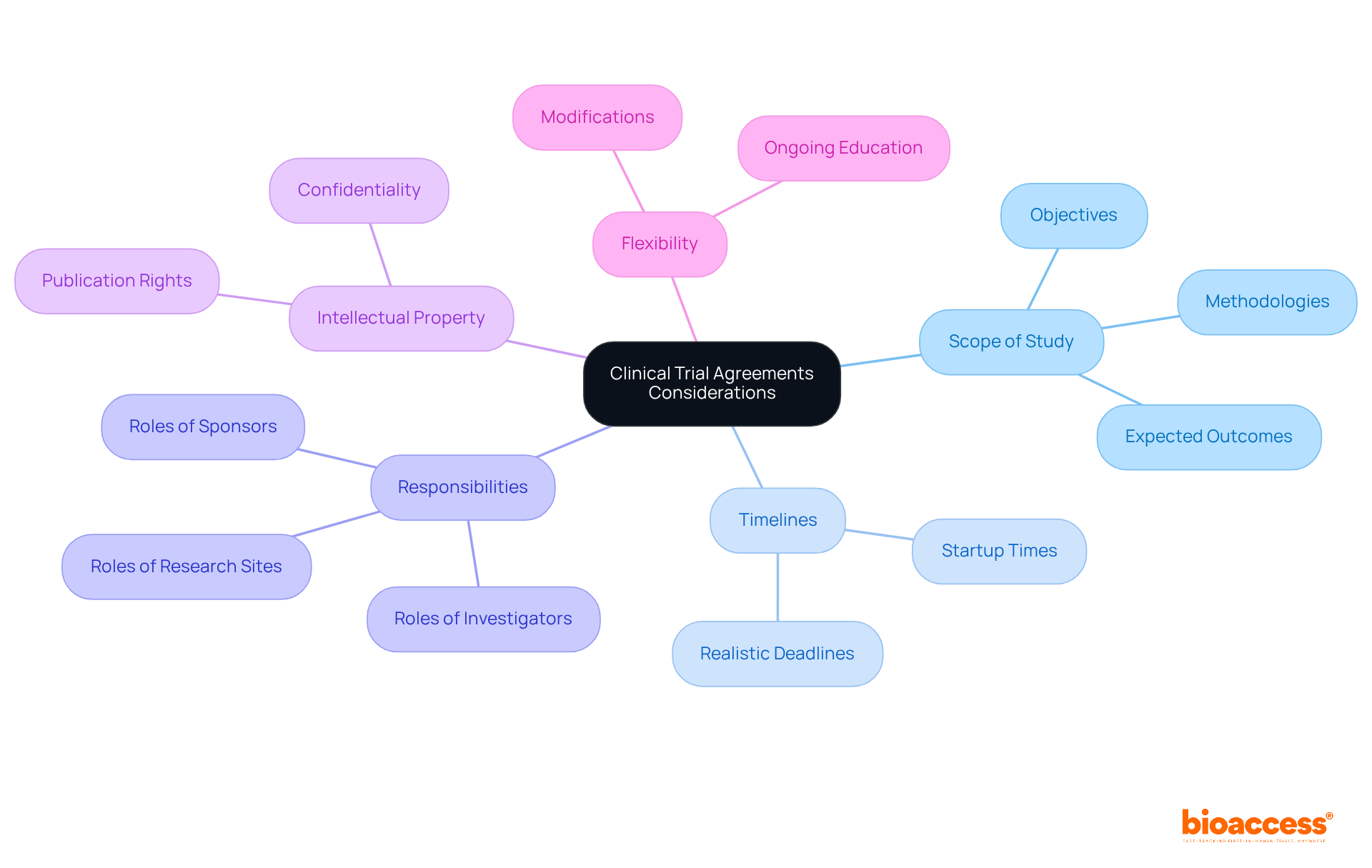
University of Wisconsin Important Considerations for Clinical Trial Agreements
Creating effective (CTCs) is essential for fostering successful partnerships in . is paramount; this involves specifying objectives, methodologies, and expected outcomes to align all parties involved. Establishing clear timelines is equally critical, as studies show that longer startup times correlate with slower overall enrollment rates. can significantly enhance efficiency.
Outlining the responsibilities of each party is crucial for ensuring accountability and compliance. This includes detailing the , investigators, and research sites, making sure everyone understands their obligations. Additionally, within the contract safeguards the interests of all stakeholders involved.
Flexibility within CTAs is vital, allowing for modifications that reflect the dynamic nature of medical research. As the landscape evolves, ongoing education and awareness of best practices can enhance the effectiveness of these agreements. By proactively addressing these factors, researchers can cultivate a collaborative environment that not only protects their interests but also promotes scientific advancement and accelerates the delivery of innovative therapies to market.
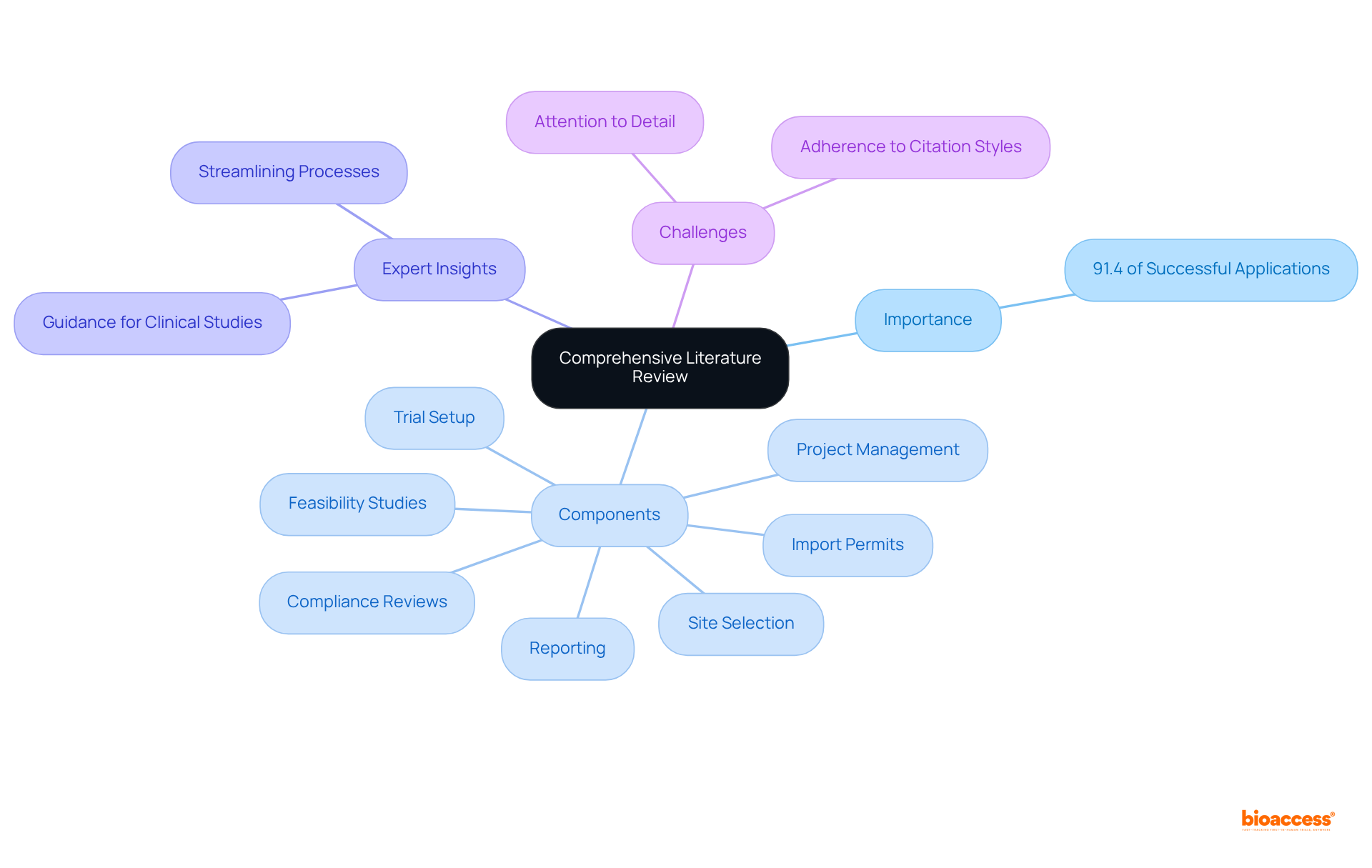
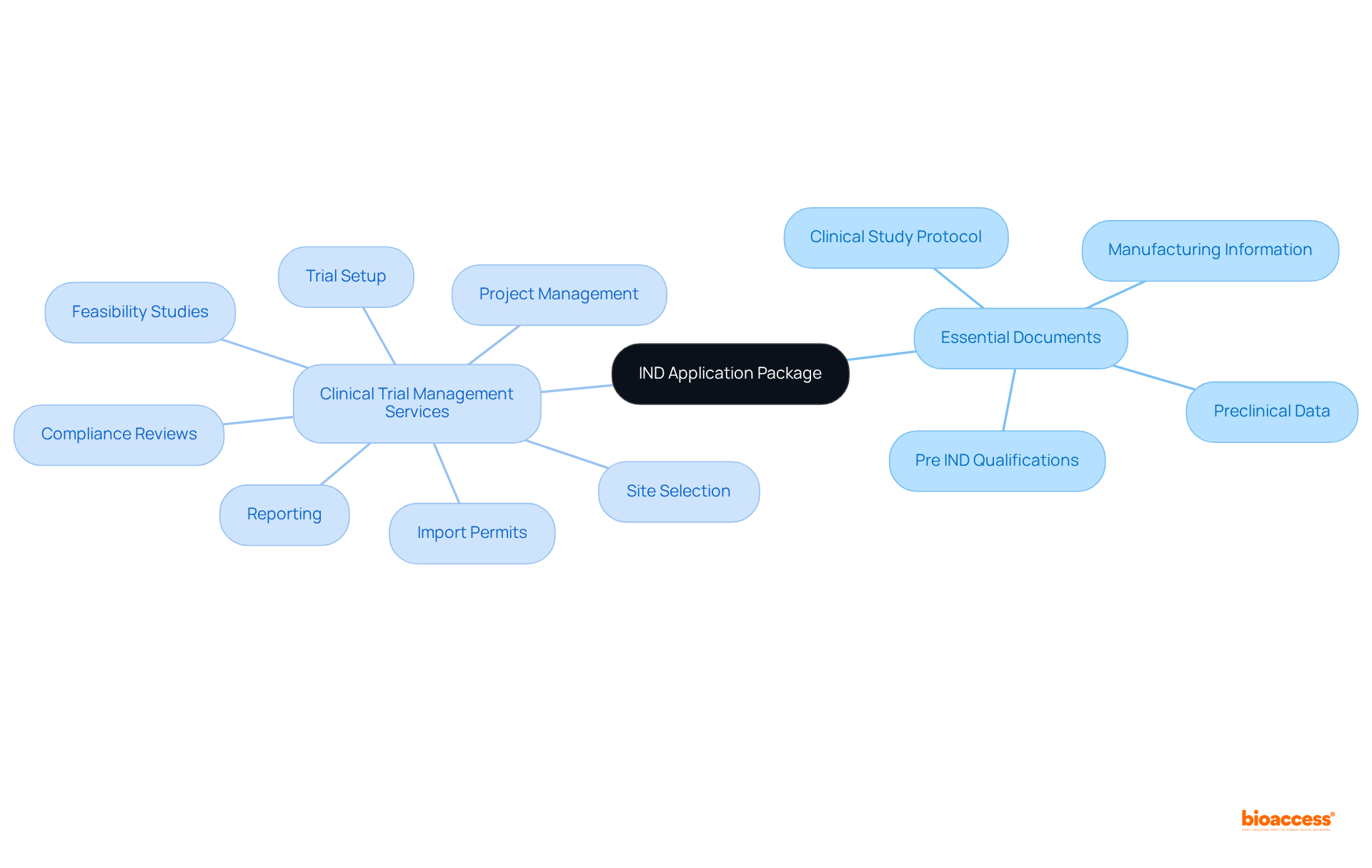
In this context, bioaccess™ plays a pivotal role in managing , offering that include:
- Feasibility assessments
- Site selection
- Compliance reviews
- Setup
- Import permits
- Project management
- Reporting
Their partnership with firms such as Welwaze Medical Inc. for the Celbrea® launch in Colombia exemplifies their commitment to enabling successful research and improving healthcare outcomes in local economies.
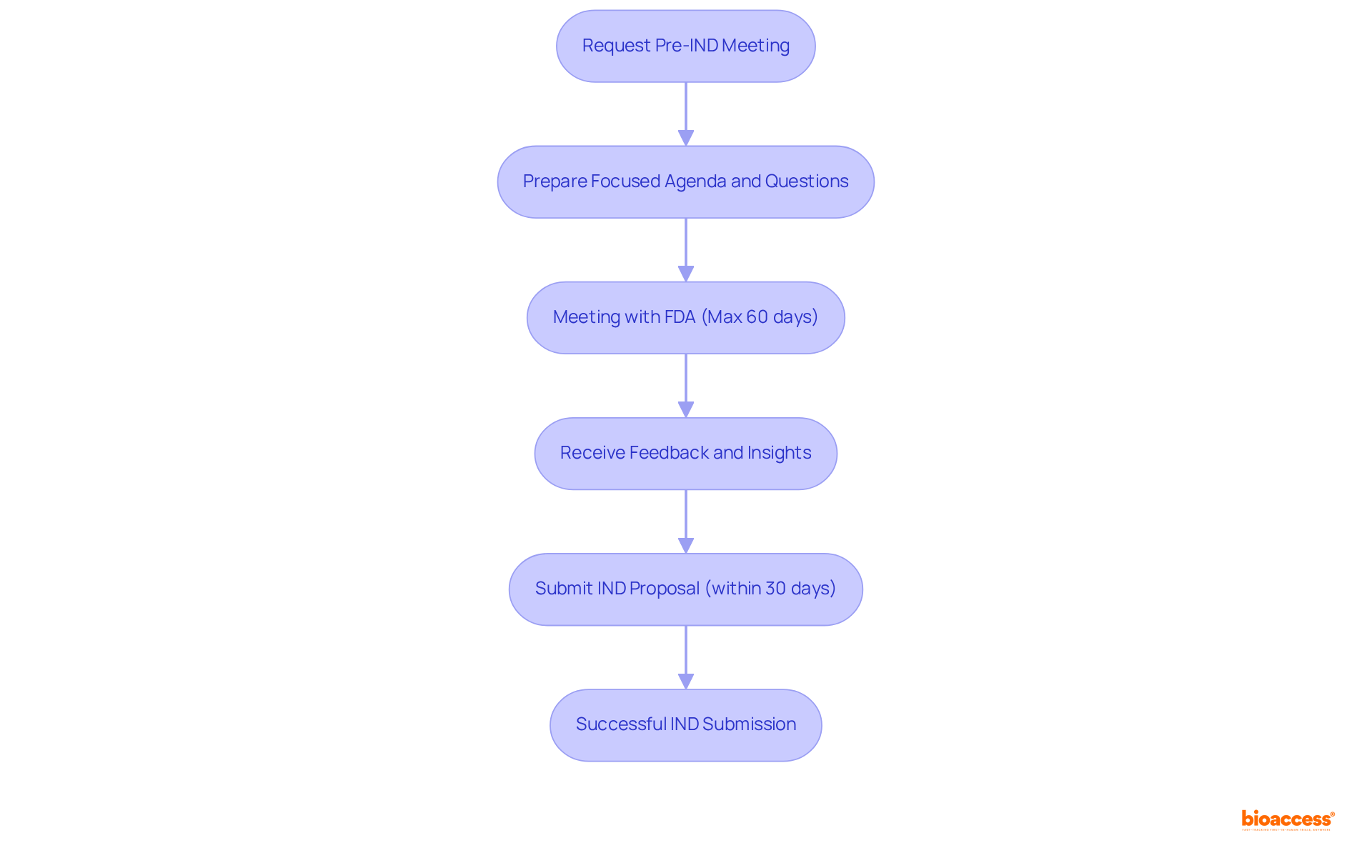
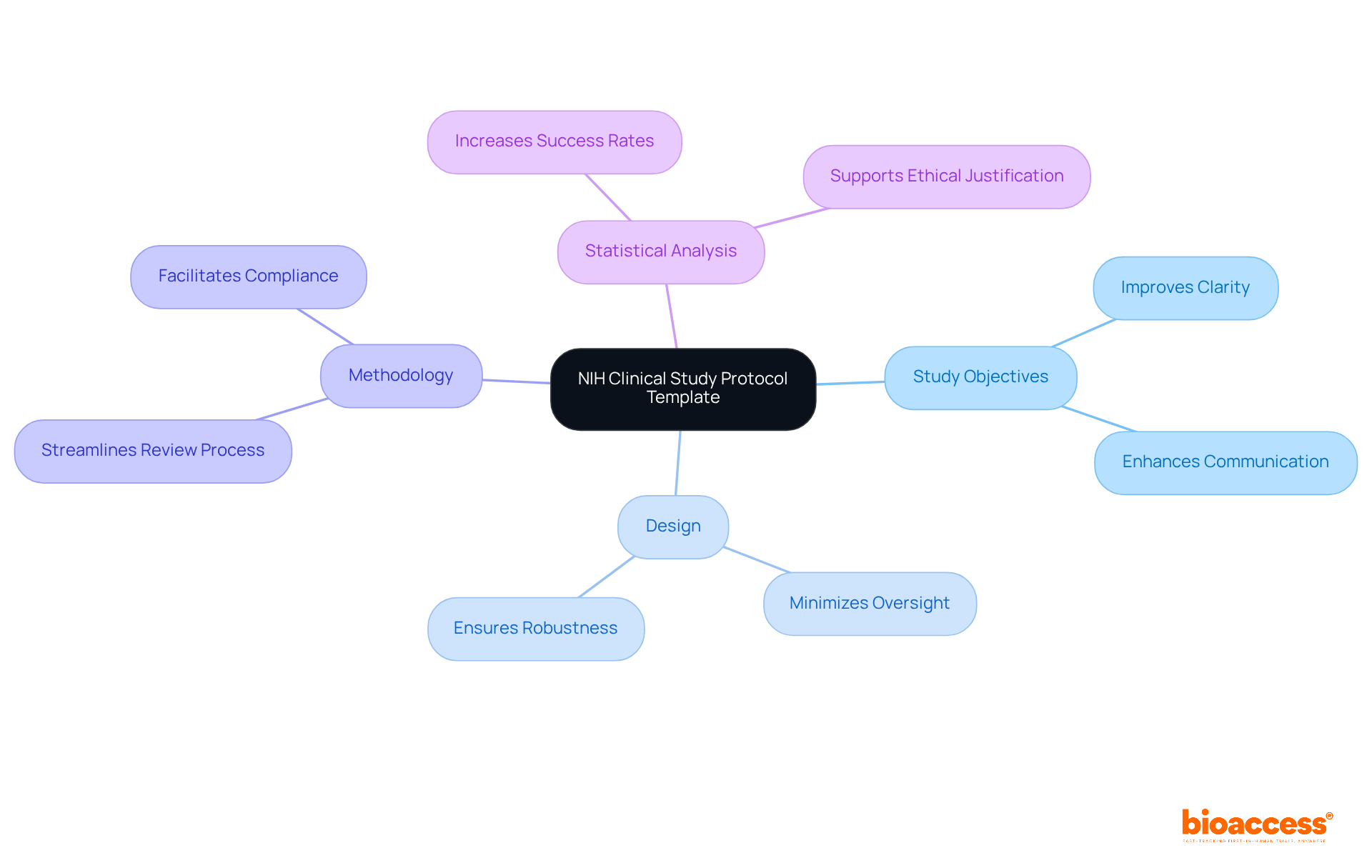
NIH Clinical Trial Protocol Template
The serves as a vital resource for researchers developing comprehensive protocols for NIH-funded studies. This structured template includes essential sections such as , ensuring that all critical components are thoroughly addressed. By following this format, researchers can significantly improve the clarity and rigor of their protocols, streamlining the review process and increasing the chances of successful funding and regulatory approval.
. They provide a and minimizes the risk of oversight. Experts in the field emphasize that a well-defined protocol is crucial for the integrity and efficacy of medical research, leading to more reliable outcomes and advancements in medical science.
Moreover, with bioaccess®’s capabilities, researchers can enroll 50% faster than traditional methods, achieving savings of $25K per patient with . This efficiency, combined with bioaccess®’s extensive services, including feasibility studies, site selection, compliance evaluations, experiment setup, import permits, project management, and reporting, empowers researchers to effectively address and .
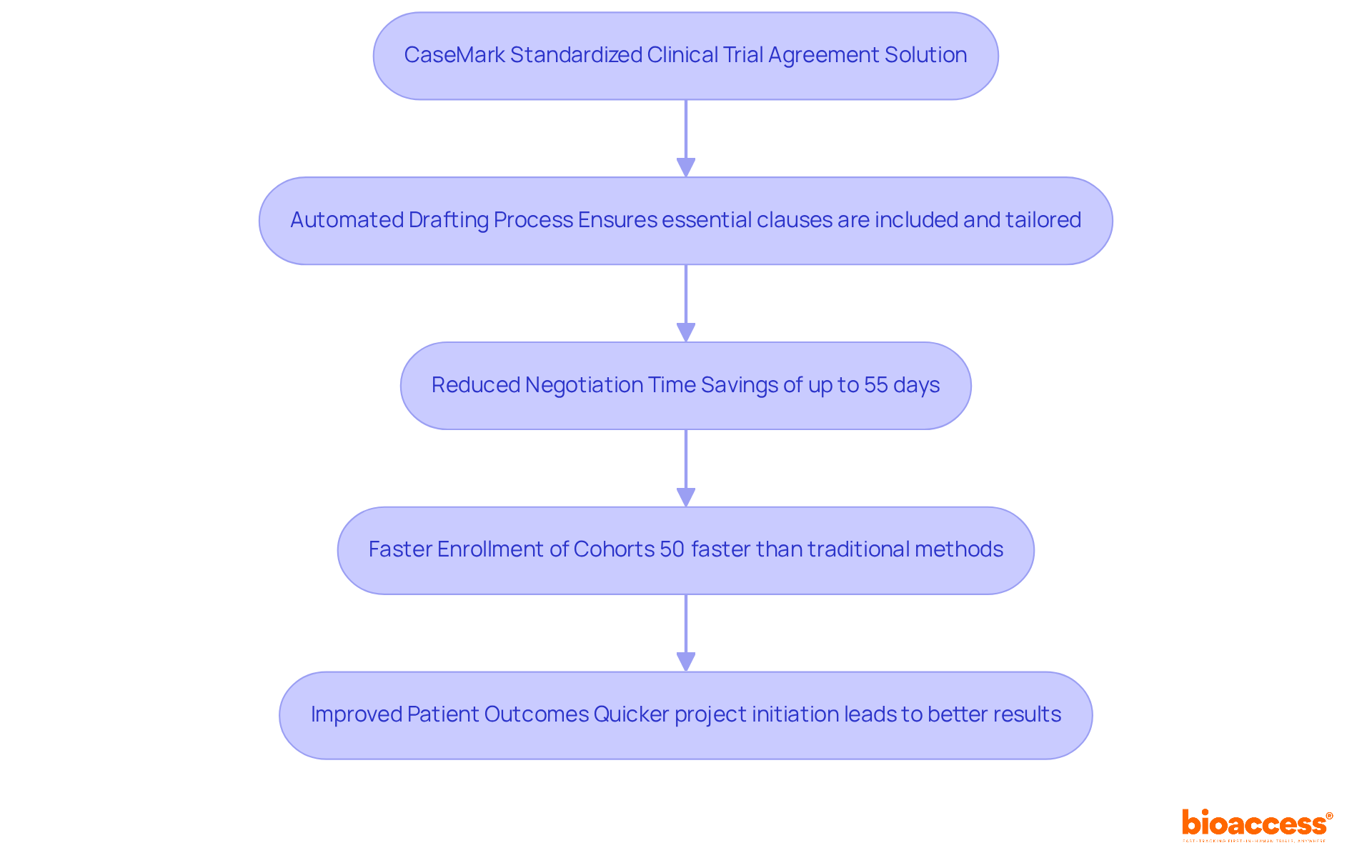
CaseMark Standardized Clinical Trial Agreement Solution
The CaseMark Solution stands as a pivotal tool in the realm of , offering a robust framework for drafting agreements that meet regulatory standards. This solution harnesses advanced technology to automate the drafting process, ensuring that all essential clauses are not only included but also tailored to the specific needs of each case. Organizations that adopt CaseMark’s solution can significantly cut down on – reports suggest savings of up to 55 days compared to traditional methods. This newfound efficiency allows research teams to focus on advancing their initiatives, leading to quicker project initiation and ultimately .
Moreover, by leveraging bioaccess®’s capabilities, organizations can than their Western counterparts, translating to an impressive $25K savings per patient with – no rework, no delays. Given that the average negotiation time for industry-sponsored agreements exceeds 100 days, the CaseMark solution, combined with bioaccess’s extensive , effectively addresses a critical demand in the research landscape. This collaboration not only streamlines processes but also of .
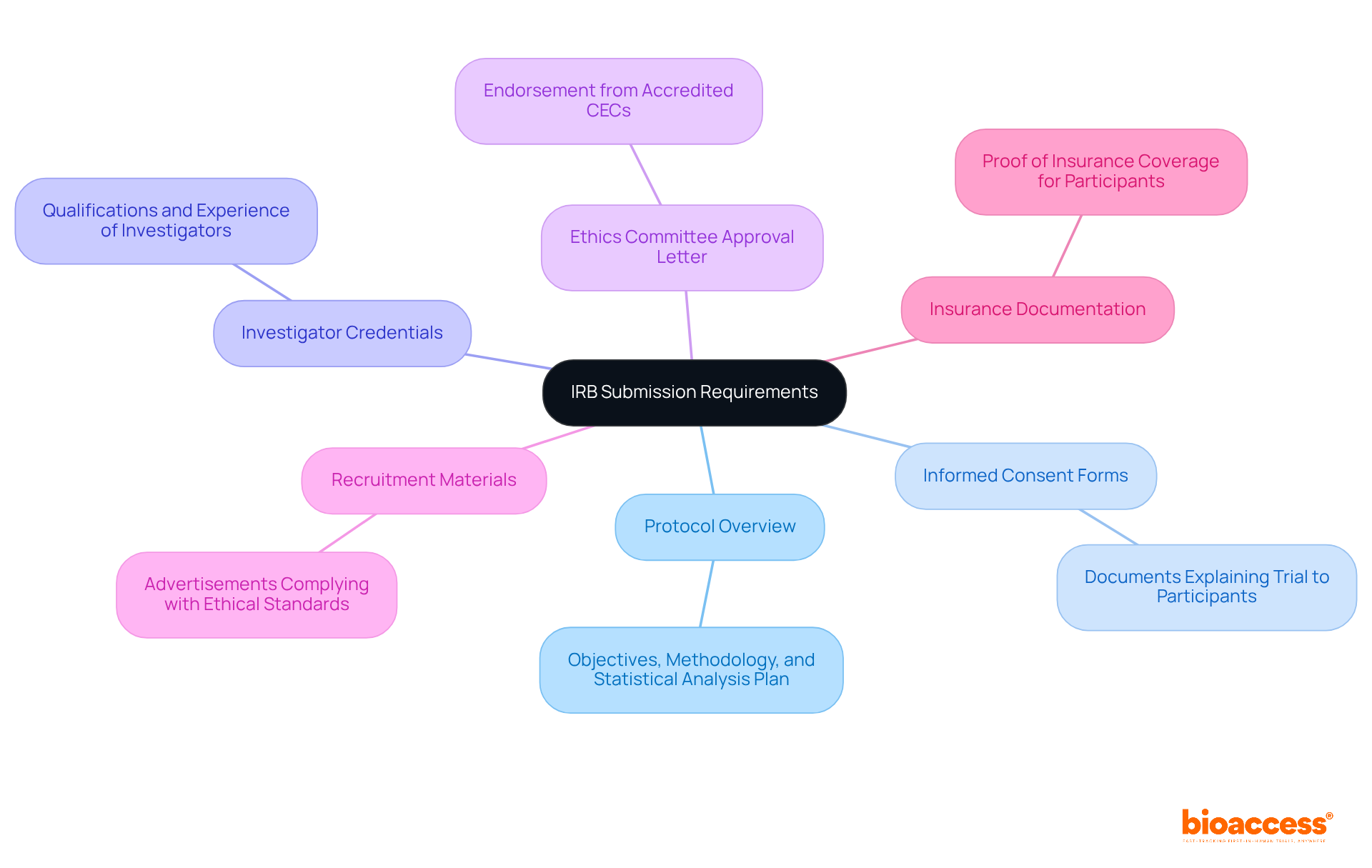
Clinical Trial Specific Agreement (CTSA) Template
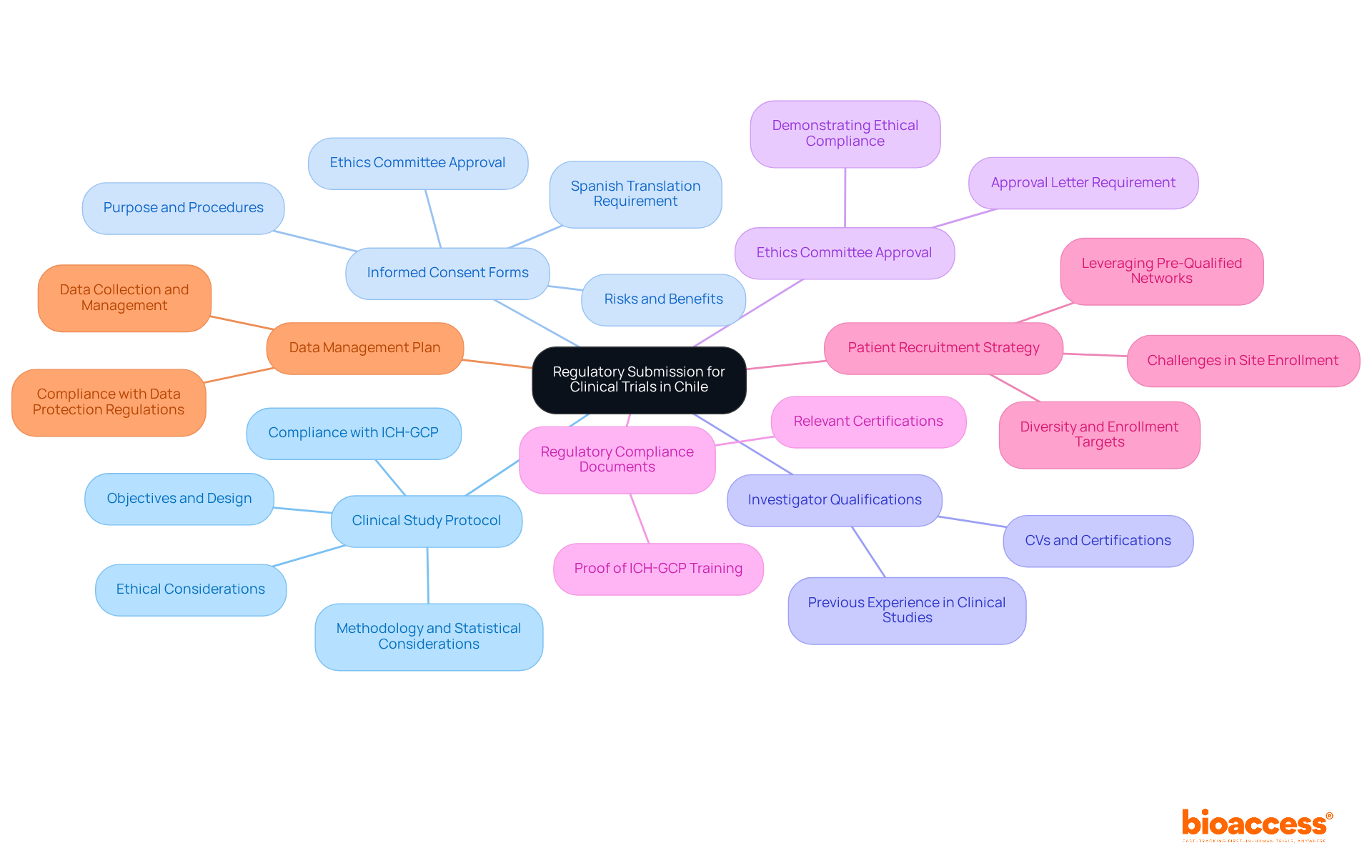
The (CTSA) Template is expertly designed to capture the unique elements of individual , ensuring that all relevant aspects are comprehensively addressed. This framework includes dedicated sections for study objectives, participant eligibility criteria, and specific . By leveraging the CTSA structure, researchers can craft agreements that are finely tuned to the specific needs of their studies. This level of customization not only streamlines negotiations but also establishes clearer expectations among all stakeholders, significantly enhancing the likelihood of .
Moreover, the inherent adaptability of the allows for modifications that reflect the evolving landscape of research, making it an invaluable asset for study sponsors and sites alike. With bioaccess®’s capabilities, researchers can achieve 50% faster and realize savings of $25K per patient. This is made possible by utilizing to expedite approval processes and tackle recruitment challenges in early-phase studies. How can your organization benefit from such efficiencies in ?
Confidentiality and IP Protection in Clinical Trial Agreements
Confidentiality and are essential components of , designed to and proprietary data. These contracts must include explicit provisions that delineate the responsibilities of each party regarding the management of and the ownership of any resulting IP. By establishing strong protections, researchers can foster trust among stakeholders while adhering to , ultimately .
Recent legal frameworks underscore the importance of these protections, as they not only secure the interests of sponsors but also cultivate a that encourages innovation. Establishing clear is paramount; these clauses should define the duration of confidentiality obligations and the specific circumstances under which information may be disclosed. This clarity is instrumental in and in research.
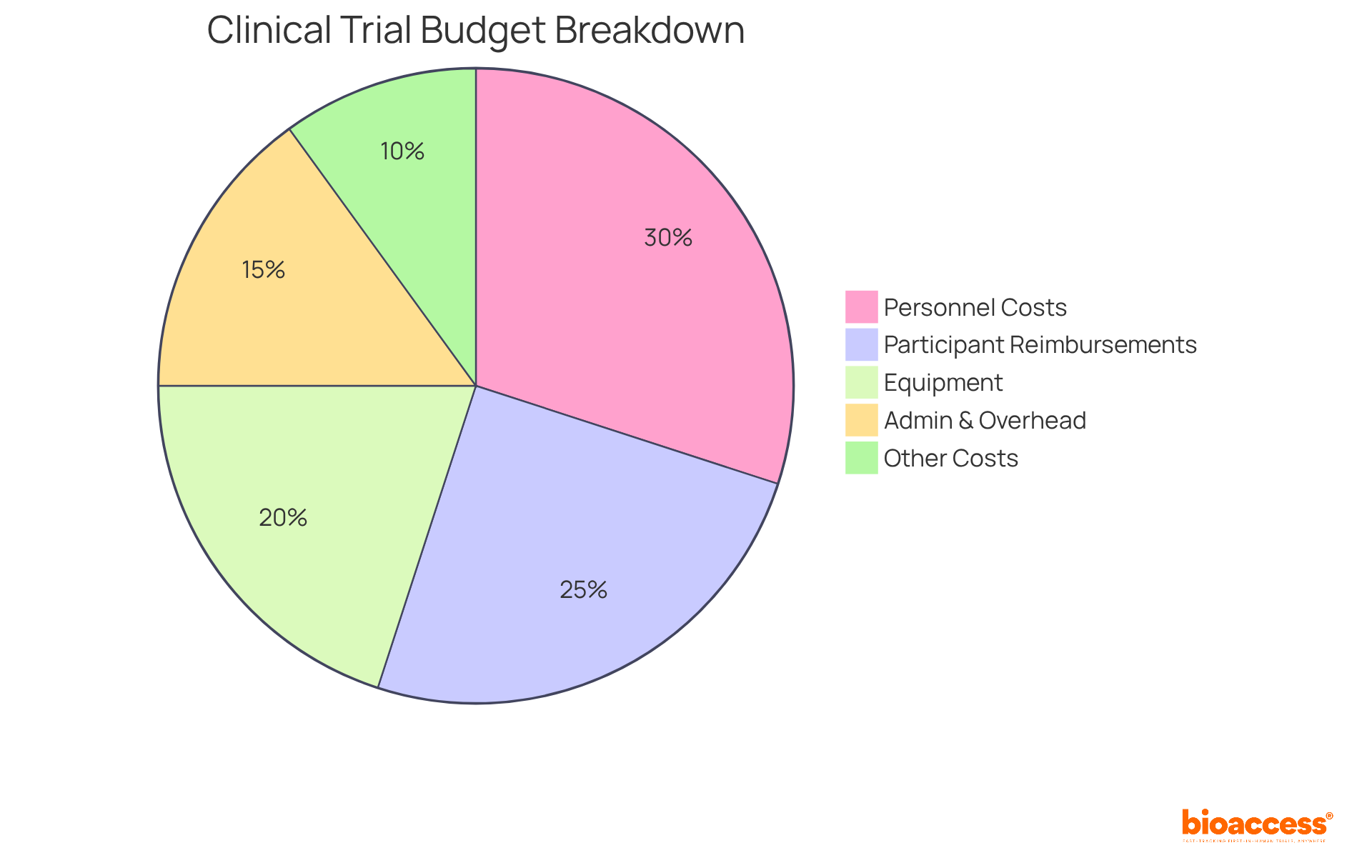
Budget Templates for Clinical Trial Agreements
are crucial for ensuring that all monetary aspects of a study are meticulously organized and documented. These formats typically include line items for:
- Personnel costs
- Equipment
- Participant reimbursements
- Administrative and overhead expenses
- Other essential costs
By adopting a , researchers can enhance , paving the way for and ensuring that studies remain within financial constraints.
The impact of on research success cannot be overstated. A well-structured budget framework aids in making data-informed decisions by clearly outlining each cost element, allowing for the examination of trade-offs before the study begins. This proactive approach is vital, especially considering that approximately 57% of clinical study protocols undergo at least one significant amendment, often leading to unexpected expenses. By implementing a that includes a contingency fund for unforeseen costs and protocol changes, organizations can mitigate the risk of underfunding and unexpected financial hurdles during the study.
Moreover, utilizing streamlines financial management, enabling the swift creation of initial budgets and facilitating real-time expense tracking. This efficiency is essential in an environment where timely responses to funding opportunities can significantly impact a study’s success. As the Trialytix team aptly puts it, “it’s time to say goodbye to spreadsheet chaos and hello to scalable, confident budgeting.” Ultimately, incorporating a not only strengthens the financial health of research projects but also builds trust among stakeholders, creating a pathway for successful outcomes.
Frequently Asked Questions About Clinical Trial Agreements
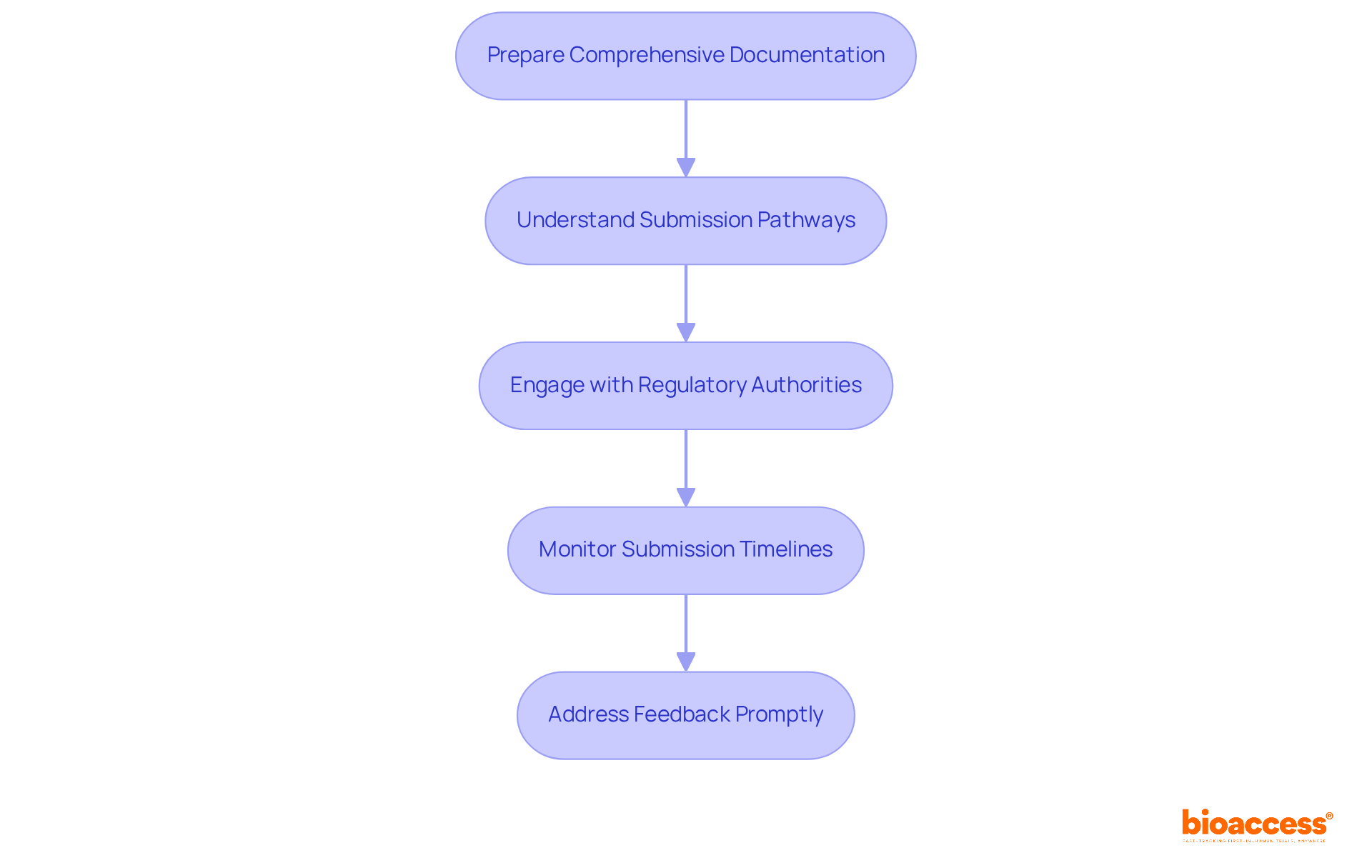
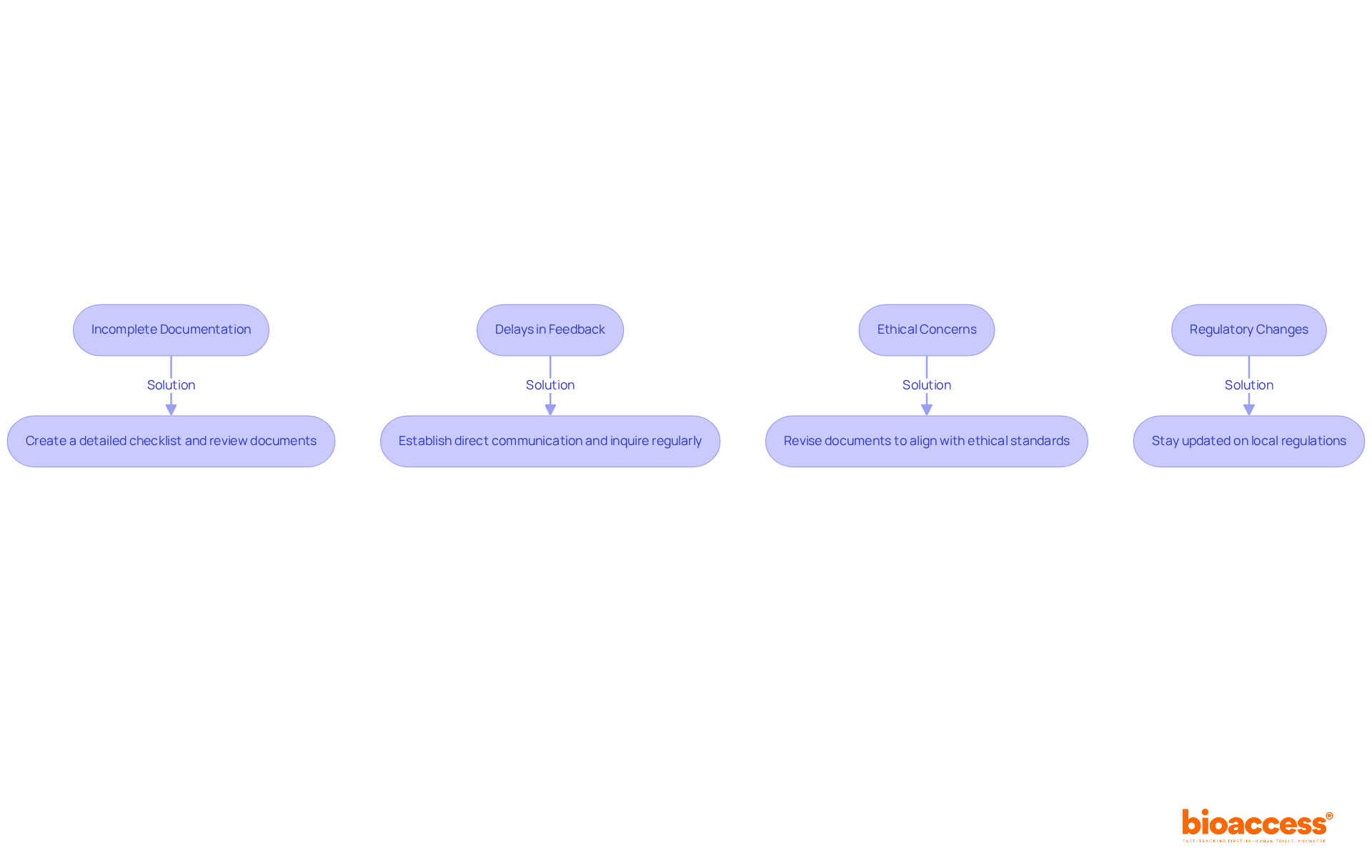
Commonly asked questions (FAQs) about research contracts address that arise during the negotiation and execution of these contracts. Understanding these elements is vital for stakeholders to manage the intricacies of effectively.
- Roles and Responsibilities: Each party’s obligations should be clearly defined to avoid misunderstandings. This includes specifying who is responsible for , data management, and .
- Timelines for Execution: Establishing clear of agreements can help prevent delays in the commencement of proceedings. Stakeholders should agree on deadlines for each phase of the process.
- Confidentiality Clauses: These clauses safeguard sensitive information exchanged during the proceedings. It is vital to understand the scope of confidentiality and the duration for which it applies to ensure compliance and protect proprietary data.
- Common Concerns: Negotiations often involve discussions about funding, liability, and publication rights. Addressing these concerns early can lead to smoother negotiations and foster a collaborative environment.
- Successful Resolution: Effective communication and a willingness to compromise are essential for addressing shared issues in research negotiations. Establishing a clear framework for conflict resolution can also facilitate a more productive negotiation process.
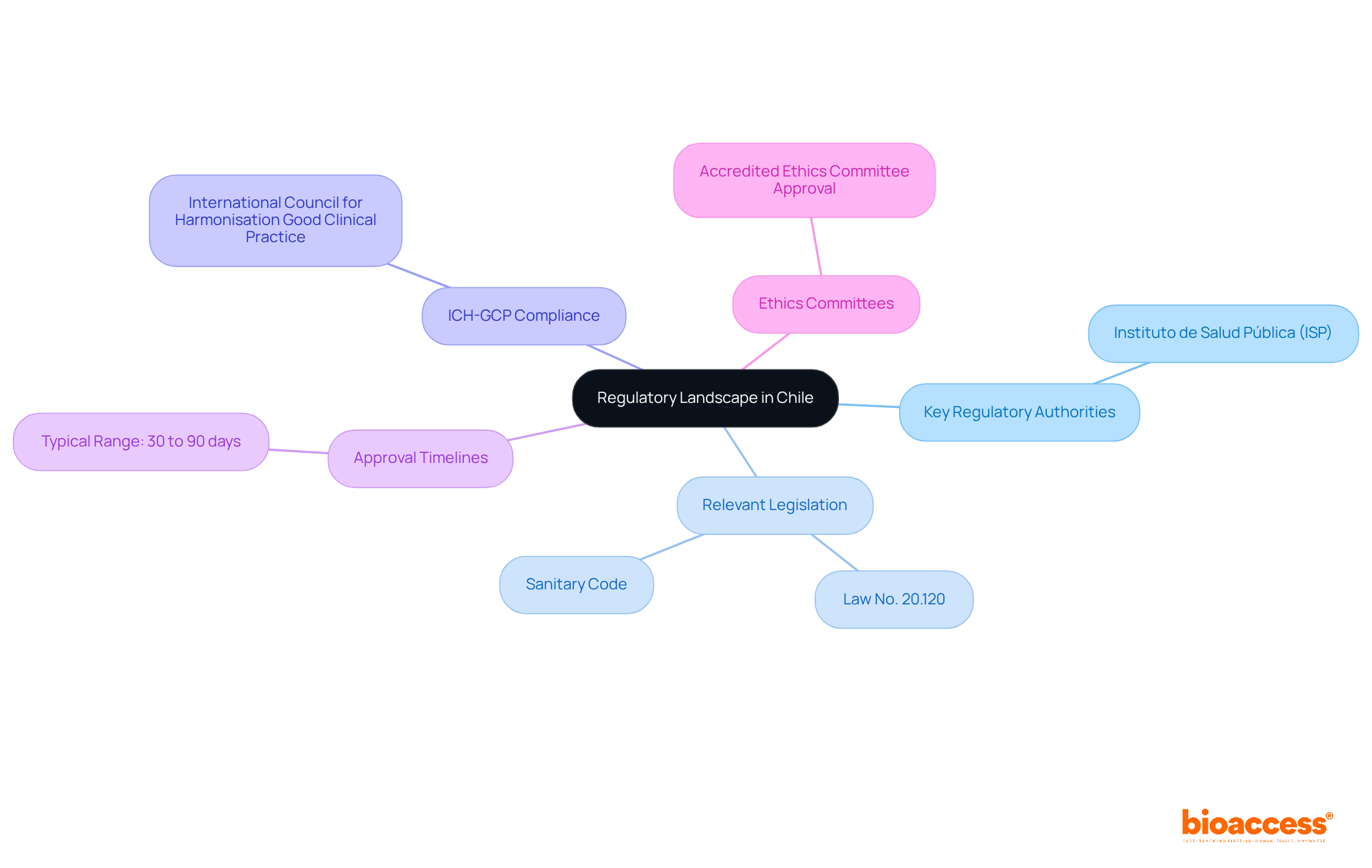
At bioaccess®, we focus on extensive research management services customized for in Latin America. Our expertise spans Early-Feasibility Studies, , Pilot Studies, Pivotal Studies, and Post-Market Clinical Follow-Up Studies. Understanding the subtleties of , such as , is essential for ensuring adherence and achieving positive results in these studies. With our dedicated team, including experts like Katherine Ruiz in and in vitro diagnostics, we ensure that all aspects of are meticulously managed to facilitate efficient execution and adherence to regulatory standards.
Conclusion
In the realm of clinical research, trial agreement templates for device and drug trials stand out as a crucial strategy for enhancing efficiency and compliance. These templates streamline the negotiation process and ensure that all parties involved are aligned in their roles and responsibilities. This alignment ultimately leads to faster patient access to innovative therapies. By adopting these structured agreements, researchers can navigate the complexities of clinical trials with greater agility and clarity.
Throughout this discussion, essential templates have been highlighted, including:
- bioaccess® Clinical Trial Agreement
- Ontario Universal Agreement for Clinical Trials
- Northwestern University Clinical Trial Agreement Template
Each of these resources addresses critical aspects of clinical research, such as budget management, confidentiality, and regulatory compliance. The emphasis on collaboration and adaptability within these agreements underscores their importance in fostering successful partnerships and expediting the research process.
As the landscape of clinical trials continues to evolve, embracing these trial agreement templates is vital for stakeholders aiming to overcome common challenges in research. By prioritizing effective communication, clear timelines, and robust protections for intellectual property, organizations can enhance the integrity of their studies and contribute to the advancement of medical science. The call to action is clear: leveraging these essential tools will empower researchers to achieve impactful outcomes and drive innovation in the healthcare sector.
Frequently Asked Questions
What is the bioaccess® Clinical Trial Agreement Template?
The bioaccess® Clinical Trial Agreement Template is a resource for Medtech innovators that enhances efficiency in clinical research by providing trial agreement templates for device and drug trials, ensuring compliance with local regulations and streamlining the approval process.
How does the bioaccess® template improve the clinical trial process?
It delineates roles, responsibilities, and timelines, which fosters clarity and operational efficiency in research studies, leading to a more efficient negotiation process and quicker study initiation, ultimately enabling faster patient access to innovative medical devices.
What advantages does the Ontario Universal Agreement for Clinical Studies (UACT) provide?
The UACT streamlines the initiation of clinical studies in Ontario by reducing the time and resources needed for contract negotiations, allowing studies to start in as little as 45 days and transforming wait times from months to weeks.
What key provisions are included in the UACT?
The UACT includes vital provisions regarding liability, confidentiality, and intellectual property rights, making it essential for both sponsors and research institutions.
How has the UACT impacted patient enrollment and cost savings?
The UACT has led to a 50% faster enrollment rate for treatment-naive cardiology or neurology cohorts compared to traditional Western sites, resulting in substantial cost savings of $25K per patient.
What is the purpose of the Northwestern University Clinical Trial Agreement (CTA) Template?
The Northwestern University CTA Template serves as a vital document for conducting clinical studies within the university, outlining the responsibilities of both the sponsor and the institution while ensuring compliance with federal regulations and institutional policies.
What are the key components of the Northwestern University CTA Template?
Key components include budgetary considerations, publication rights, and data management protocols, which help researchers align their studies with established best practices and institutional guidelines.
How does leveraging bioaccess® enhance study efficiency in clinical trials?
Utilizing bioaccess®’s capabilities allows for a 50% faster enrollment of treatment-naive cardiology or neurology groups, leading to significant savings of $25K per patient with FDA-ready data that eliminates rework and delays.
Why is collaboration important in the Medtech landscape?
Collaboration is essential as it enables researchers to navigate challenges more effectively and drive impactful results, particularly when utilizing innovative solutions like the CTA Template and bioaccess®.
List of Sources
- bioaccess® Clinical Trial Agreement Template for Medtech Innovations
- 9 Essential Components of a Clinical Trial Agreement (https://clinicaltrialsarena.com/news/9-essential-components-of-a-clinical-trial-agreement-5885280-2)
- Clinical Trial Case Studies | Innovative Trials (https://innovativetrials.com/case-studies)
- linkedin.com (https://linkedin.com/posts/juliomartinezclark_mexico-streamlines-clinical-trial-approvals-activity-7313213647210565633-cbML)
- Latin America’s Landscape For Medtech Clinical Trials (https://clinicalleader.com/doc/latin-america-s-landscape-for-medtech-clinical-trials-0001)
- 6 Important Quotes from Medtech Leaders (https://mddionline.com/business/6-important-quotes-from-medtech-leaders)
- Ontario Universal Agreement for Clinical Trials (UACT)
- Streamlining paperwork, saving lives – Hamilton Health Sciences (https://hamiltonhealthsciences.ca/share/faster-access-to-trials)
- Universal Agreement for Clinical Trials (UACT) – Clinical Trials Ontario (https://ctontario.ca/universal-agreement-for-clinical-trials)
- Ontario’s Universal Agreement for Clinical Trials: New Partners, New Version – Clinical Trials Ontario (https://ctontario.ca/uact-new-partners-new-version)
- Northwestern University Clinical Trial Agreement (CTA) Template
- Northwestern University Case Study: Centralized Tracking As A Shared Service – Form.io (https://form.io/case-studies/northwestern-university-case-study-centralized-tracking-as-a-shared-service)
- Clinical Trial Agreements: Sponsored Research – Northwestern University (https://sponsoredresearch.northwestern.edu/agreements/cta)
- Joint statement on public disclosure of results from clinical trials (https://who.int/news/item/18-05-2017-joint-statement-on-registration)
- Global Healthcare Case Studies & Partnerships (https://northwesternglobalservices.com/case-studies.html)
- University of Wisconsin Important Considerations for Clinical Trial Agreements
- Clinical Trial Delays: Key Challenges from Phase I to III | IntuitionLabs (https://intuitionlabs.ai/articles/clinical-trial-delays-phase-i-iii)
- 9 Essential Components of a Clinical Trial Agreement (https://clinicaltrialsarena.com/news/9-essential-components-of-a-clinical-trial-agreement-5885280-2)
- Clinical Trial Agreements | Practical Law (https://content.next.westlaw.com/practical-law/document/I61c72451888011e598dc8b09b4f043e0/Clinical-Trial-Agreements?viewType=FullText&transitionType=Default&contextData=(sc.Default))
- bioaccessla.com (https://bioaccessla.com/blog/10-essential-ct-as-in-clinical-research-every-director-should-know)
- NIH Clinical Trial Protocol Template
- Notices of NIH Policy Changes | Grants & Funding (https://grants.nih.gov/policy-and-compliance/notice-of-policy-changes)
- Statistics and clinical trials: It’s all about the design (https://onlinelibrary.wiley.com/doi/10.1111/test.12325)
- Implementation of New Initiatives and Policies | Grants & Funding (https://grants.nih.gov/policy-and-compliance/implementation-of-new-initiatives-and-policies)
- Quotes About Medical Research (https://changingthepresent.org/pages/quotes-about-medical-research)
- CaseMark Standardized Clinical Trial Agreement Solution
- Quantitative assessment of the impact of standard agreement templates on multisite clinical trial start up time – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10565190)
- Clinical Trial Specific Agreement (CTSA) Template
- A Guide to Clinical Trial Agreements | ICON news and blogs (https://careers.iconplc.com/blogs/2025-4/a-guide-to-clinical-trial-agreements)
- COVID-19 and Clinical Trial Agreements: Advice for Sponsors for Now and the Future | Leibowitz Law | Life Sciences & Tech Lawyers (https://leibowitzlawteam.com/covid-19-clinical-trial-agreements-advice-sponsors-now-future)
- Case Studies in Translational Science: Identifying Best Practices to Accelerate the Translational Process | CWRU Newsroom | Case Western Reserve University (https://case.edu/news/case-studies-translational-science-identifying-best-practices-accelerate-translational-process)
- grants.nih.gov (https://grants.nih.gov/policy-and-compliance/policy-topics/clinical-trials/case-studies)
- Confidentiality and IP Protection in Clinical Trial Agreements
- USTR Releases 2025 Special 301 Report on Intellectual Property Protection and Enforcement (https://ustr.gov/about/policy-offices/press-office/press-releases/2025/april/ustr-releases-2025-special-301-report-intellectual-property-protection-and-enforcement)
- TOP 25 INTELLECTUAL PROPERTY QUOTES (of 84) | A-Z Quotes (https://azquotes.com/quotes/topics/intellectual-property.html)
- New Guidance, Clinical Trials, Intellectual Property Protection | JD Supra (https://jdsupra.com/topics/new-guidance/clinical-trials/intellectual-property-protection)
- The Business of Clinical Trials, Part 1: Negotiating Confidentiality, (https://mddionline.com/business/the-business-of-clinical-trials-part-1-negotiating-confidentiality-ip-and-publications)
- Budget Templates for Clinical Trial Agreements
- Trialytix – Free Budget Template (https://discover.trialytix.io/form-free-budget-template)
- Frequently Asked Questions About Clinical Trial Agreements
- Statistical Issues and Recommendations for Clinical Trials Conducted During the COVID-19 Pandemic (https://tandfonline.com/doi/full/10.1080/19466315.2020.1779122)
- Frequently Asked Questions | ClinicalTrials.gov (https://clinicaltrials.gov/policy/faq)
- Streamlining Clinical Trial Agreement Negotiations: Avoiding Common Pitfalls for Faster Signatures | WCG (https://wcgclinical.com/insights/streamlining-clinical-trial-agreement-negotiations-avoiding-common-pitfalls-for-faster-signatures)