Introduction
The pharmaceutical landscape is undergoing a rapid transformation, driven by the urgent need for innovative drug formulations that address the complexities of modern healthcare. As clinical research becomes increasingly competitive, navigating regulatory hurdles, leveraging cutting-edge technology, and adopting patient-centric approaches is paramount.
This article explores ten effective strategies designed to enhance drug formulation processes, empowering researchers to streamline development while simultaneously improving patient outcomes.
How can organizations effectively balance the urgency of market demands with the meticulous nature of drug formulation to ensure both efficacy and safety?
bioaccess®: Accelerate Clinical Research with Fast Ethical Approvals
bioaccess® strategically leverages its locations in Latin America, the Balkans, and Australia to secure . This expedited process is essential for innovators who are eager to accelerate their market entry. By adeptly navigating the , bioaccess® minimizes delays, empowering clients to focus on their core research activities.
Rapid not only by up to 50% but also significantly shorten the overall —an imperative in a competitive industry where delays in can cost sponsors between $600,000 and $8 million per day. Companies that have effectively harnessed this speed include those engaged in , which have been shown to enhance and drive innovation.
For example, Sparta Biomedical reaped in Colombia, underscoring the advantages of collaboration with bioaccess®. As the MedTech industry has recorded at least 4% revenue growth annually over the past eight years, the ability to obtain swift regulatory approvals will continue to be a critical differentiator for success in clinical research.
Leverage Human Expertise and Technology for Drug Formulation
Successful hinges on the collaboration of skilled experts and cutting-edge technology. Investigators, well-versed in the intricacies of scientific development, are pivotal in creating . The integration of advanced technologies, particularly , significantly enhances the development process. These technologies facilitate rapid iterations and improvements driven by , optimizing pharmaceutical design and progression.
is profound. For example, AI systems can analyze extensive datasets to predict physicochemical properties, thereby enhancing solubility and stability while minimizing the risk of costly trial failures. This capability enables researchers to focus on innovative solutions, . Moreover, AI-driven platforms have demonstrated the ability to , underscoring their potential to transform traditional practices.
Formulation scientists are increasingly recognizing the and the development of medicine. Many assert that AI not only accelerates the identification of effective compounds but also enhances the accuracy of s, ultimately leading to . AI can aid in decision-making and facilitate logical medication design, further boosting efficiencies in the development process. As AI continues to evolve, its influence on processes is expected to expand, fostering additional efficiencies and innovations within the pharmaceutical sector.
Partner with CDMOs for Streamlined Drug Development
Collaborating with the . These organizations provide , encompassing everything from development to manufacturing. By harnessing their , companies can effectively and . This empowers innovators to concentrate on their while .
Understand Regulatory Requirements to Navigate Formulation Challenges
Navigating necessitates a comprehensive understanding of the evolving requirements set by regulatory bodies. For companies aiming to ensure compliance and mitigate development challenges, staying informed about the is crucial. The FDA’s recent initiatives, such as the introduction of a and the PRISM Project designed to , underscore the importance of adaptability in . Organizations that proactively adjust their production processes in response to these guidelines can significantly enhance their chances of securing smoother approvals and achieving quicker market entry.
Statistics reveal that approximately 34% of new molecular entities authorized by the FDA in 2022 were classified as , illustrating the growing complexity of compliance in development. This trend compels companies to not only grasp regulatory expectations but also to innovate in their strategic approaches to meet the demands of personalized medicine. By leveraging insights from regulatory affairs specialists, organizations can adeptly and position themselves for success within the competitive landscape of . Furthermore, with the global pharmaceutical market projected to reach USD 2.8 trillion by 2035, effectively managing these regulatory hurdles is more critical than ever. bioaccess® offers that empower clients to capitalize on the vast potential of Latin America’s healthcare market, further exemplifying how companies can successfully navigate these complexities.
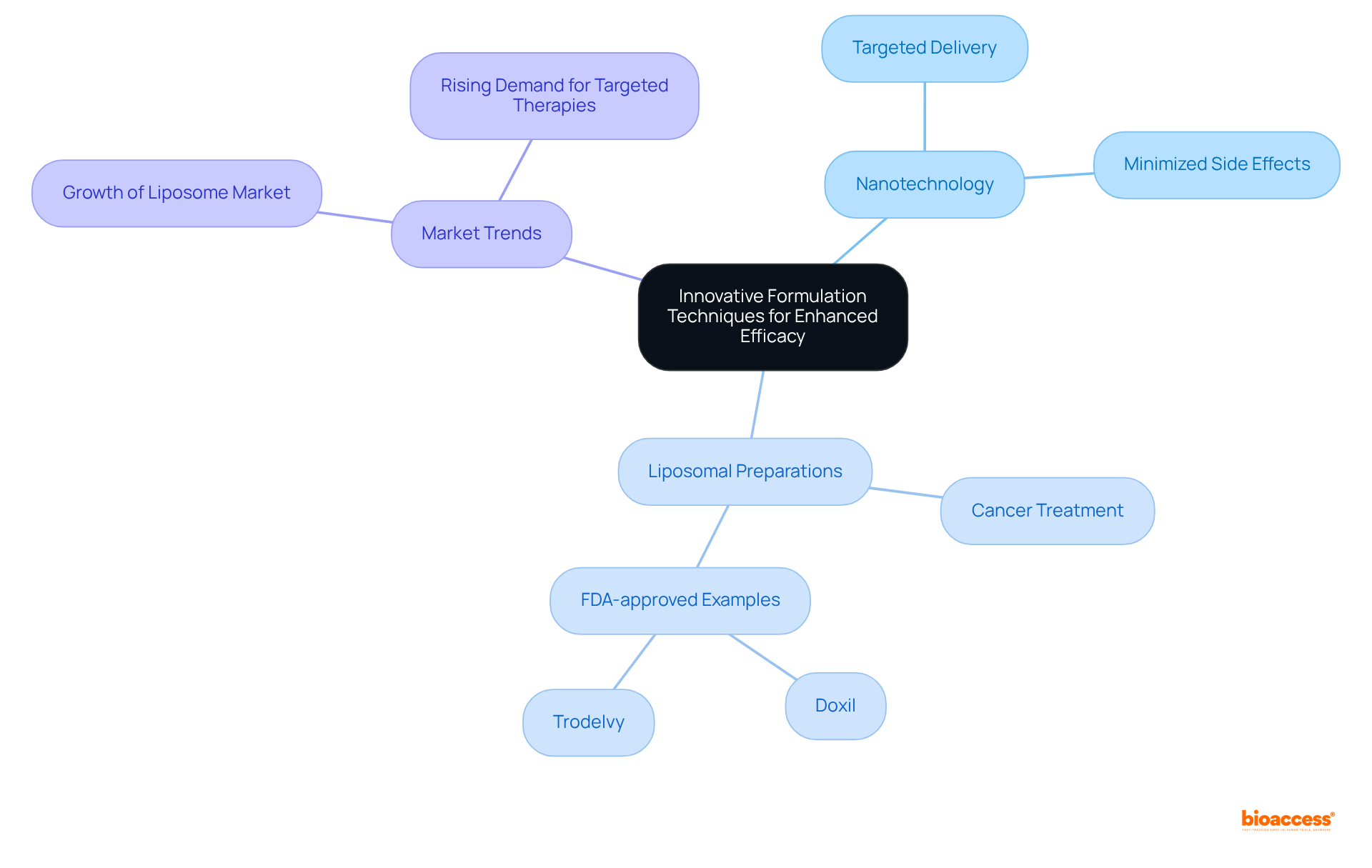
Implement Innovative Formulation Techniques for Enhanced Efficacy
Integrating creative preparation methods is essential for enhancing the effectiveness of medicine. Techniques such as nanotechnology and have shown significant improvements in . For instance, lipid-based mixtures can enhance the solubility and uptake of poorly water-soluble medications, addressing a common challenge in pharmaceutical advancement. Notably, approximately 40% of medications in development are poorly water-soluble, making these innovative techniques crucial. The lipid-based preparation of cyclosporine (Neoral) improved bioavailability by 30-50% and reduced interpatient variability by 50-70%.
Recent advancements in nanotechnology have further revolutionized drug formulation. Nanoparticles and nanocarriers enable , maximizing therapeutic efficacy while minimizing adverse effects. The integration of these technologies into is gaining traction, facilitating tailored treatments that align with individual patient needs.
Current trends indicate an increasing reliance on liposomal formulations, particularly in cancer treatment, where they and mitigate side effects. The cancer therapy sector leads the , driven by the rising incidence of cancer and the demand for targeted treatments. The in 2023 was estimated at USD 5.46 billion, underscoring its significance. Notable examples include FDA-approved like Doxil and Trodelvy, which have demonstrated enhanced effectiveness in treating various cancers.
By remaining at the forefront of scientific development, companies can create more efficient therapies that meet the evolving needs of individuals, ultimately resulting in improved health outcomes.
Adopt Patient-Centric Approaches in Drug Formulation
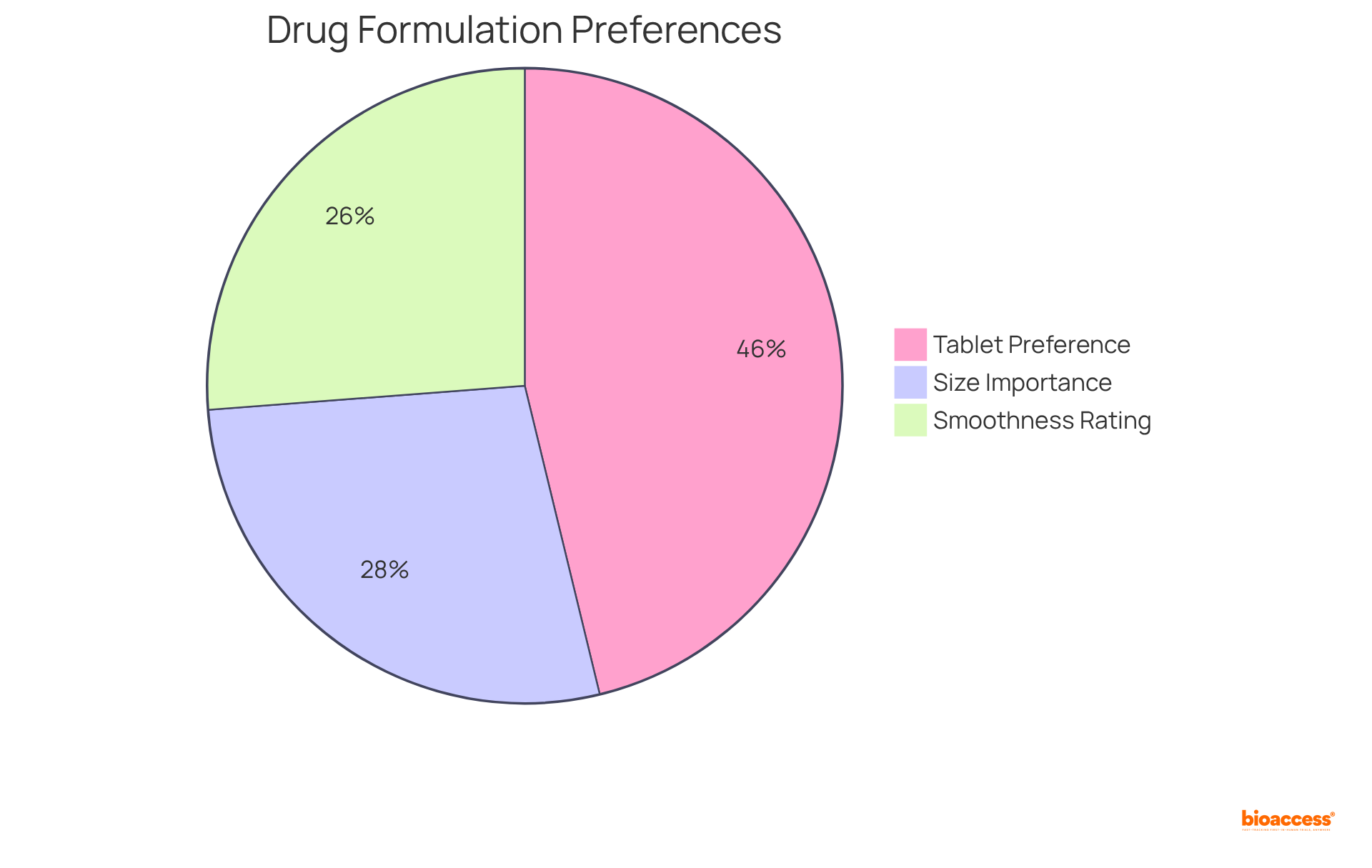
Integrating a user-focused strategy in is essential for aligning therapies with individual needs and preferences. This approach actively considers factors such as , , administration route, and potential side effects during the development process. Engaging individuals in discussions about their experiences and preferences leads to the creation of more suitable and .
Research indicates that:
- 67% of individuals favor over alternatives, underscoring the significance of in adherence.
- 40% of individuals have identified for the acceptability of s in children, with 96% agreeing that the ideal size ranges from 4 to 9 mm.
- 38% of individuals rated smoothness as a significant characteristic for the ‘s swallowability.
Healthcare professionals recognize that greatly influence , with many advocating for to enhance adherence rates. As Brett Hauber observed, while existing studies have concentrated on appearance and swallowability, the importance of must not be overlooked. By prioritizing consumer feedback, medication creators can improve treatment outcomes and foster a more collaborative healthcare environment.
Utilize Diverse Patient Populations for Comprehensive Insights
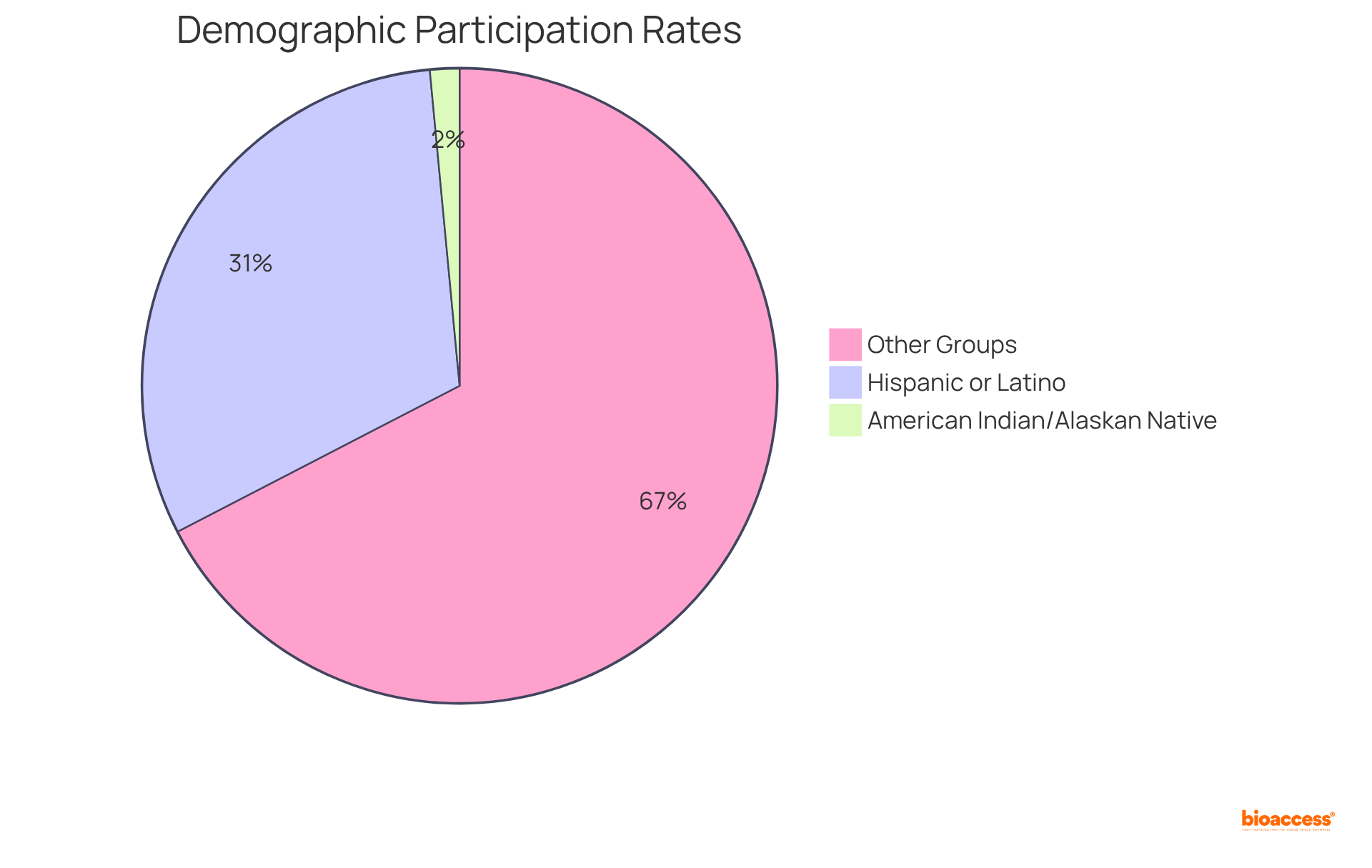
Including varied groups in clinical studies is crucial for obtaining a thorough understanding of how different demographics react to drug formulations. Studies have shown that variations in drug efficacy and safety can significantly differ across populations, underscoring the need for .
For instance, recent analyses indicate that minority groups often experience disparities in , with Hispanic or Latino individuals participating at rates such as 31.1% in specific trials like Inpefa. Additionally, the participation of American Indian or Alaskan Native individuals has historically been under 1% to 2%.
By conducting research in , researchers can ensure that their findings are pertinent to a wider array of individuals, ultimately enhancing the applicability of their results. This method not only promotes inclusivity but also , as it enables the identification of and potential side effects that may not be clear in more homogeneous study groups.
As highlighted by Bibbins-Domingo K., a is likely to accelerate scientific progress and lead to more for all patients. Moreover, with clinical trial failure rates varying between 80% and 90%, the importance of becomes even more essential in enhancing pharmaceutical creation results.
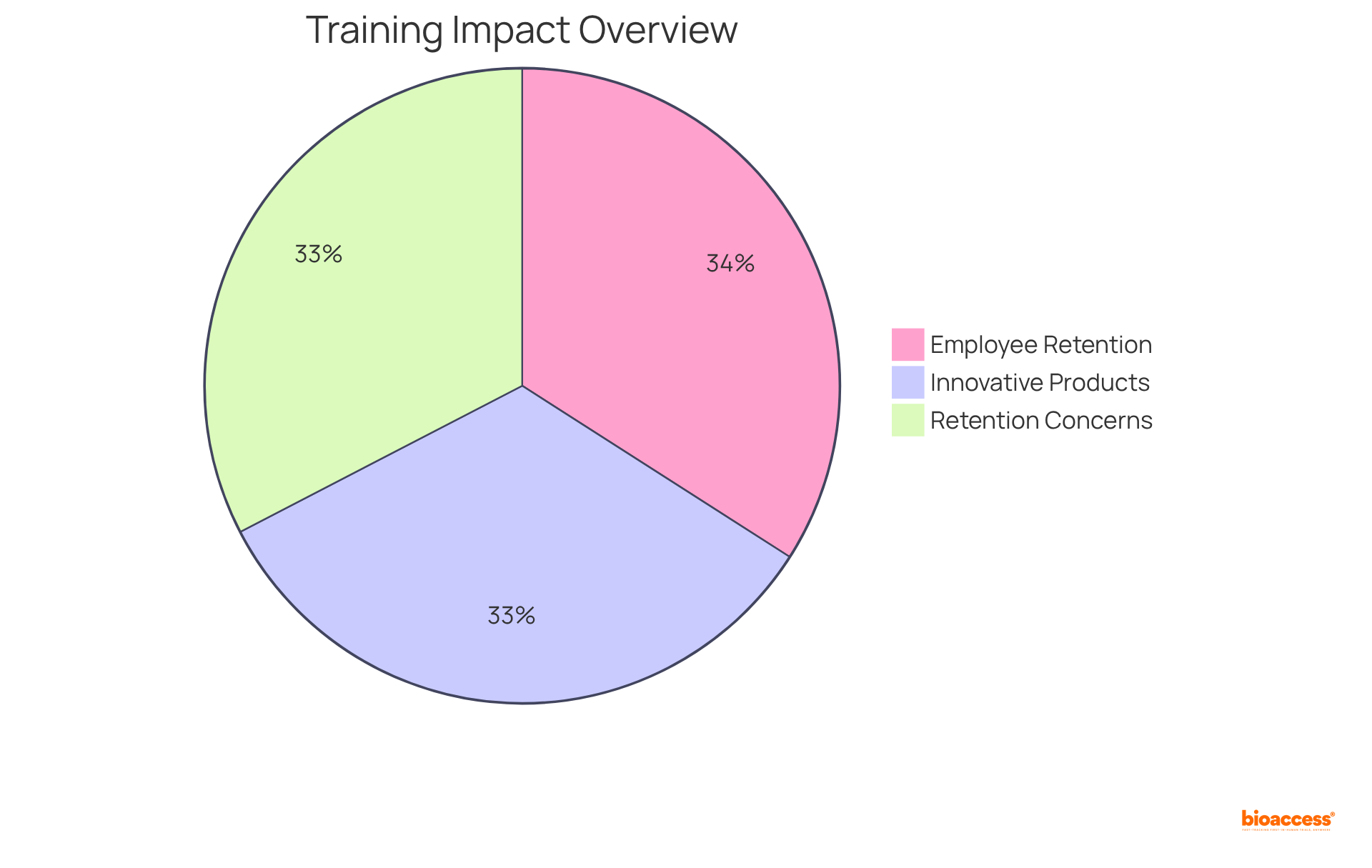
Invest in Continuous Training for Research Teams
is essential for upholding . Regular training sessions that focus on the , , and equip team members with the critical knowledge and skills needed to excel. This commitment to not only enhances the quality of medication creation but also fosters a culture of excellence and innovation within the organization.
Notably, industry leaders acknowledge that organizations prioritizing are:
- 92% more likely to create innovative products and processes.
- 94% of employees would remain with a company longer if it invested in their career development.
Given that 90% of companies express concern about employee retention, the becomes paramount for maintaining a competitive edge.
Leverage Data Analytics to Optimize Drug Formulation
Utilizing is crucial for . By analyzing data from earlier studies, researchers can uncover trends and correlations that inform . not only helps in recognizing potential challenges but also enables , thereby improving the overall strategy.
For instance, , such as the ADA-DT model achieving an R² score of 0.9738 for drug solubility prediction, illustrate the significant improvements in accuracy that can be realized. Moreover, the methodical elimination of less significant features via Recursive Feature Elimination (RFE) can enhance model performance, resulting in more informed .
Data scientists, like Dr. Ashwin Kuchekar, emphasize that integrating with traditional methods can lead to better , ultimately reducing the time and costs associated with bringing new treatments to market.
To implement effectively, directors of should consider investing in to utilize these advanced analytical tools, ensuring they can leverage in their strategy development.
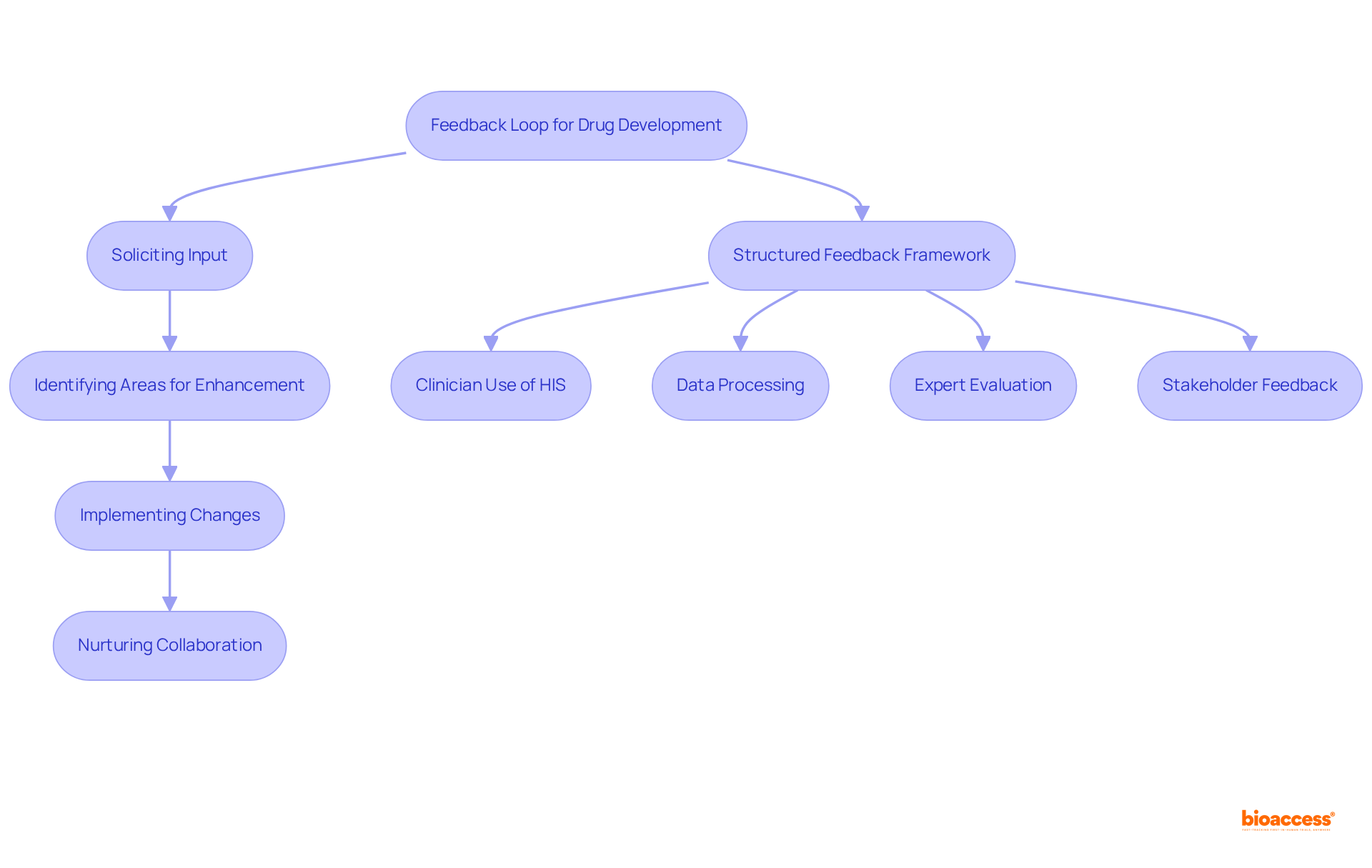
Establish Feedback Loops for Continuous Improvement in Formulation
Creating is essential for fostering continuous improvement. By actively soliciting input from team members, stakeholders, and clients, organizations can identify areas for enhancement and implement effective changes. This iterative approach not only improves but also nurtures a that is conducive to innovation.
Notably, bioaccess® has shown that leveraging diverse patient pools and regulatory speed can significantly , achieving results 50% faster than traditional markets. Furthermore, industry leaders assert that and enhancing drug development outcomes.
A , which encompasses:
- Clinician use of hospital information systems (HIS)
- Data processing
- Expert evaluation
ensures that insights are systematically integrated into drug formulation strategies. Additionally, maintaining is vital for utilizing in scientific contexts, ultimately leading to safer and more effective pharmaceutical products.
It is also imperative to recognize the limitations in analyzing free text not mapped to ATC codes, as this presents challenges within the feedback loop framework.
Conclusion
Effective drug formulation in clinical research stands as a multifaceted endeavor, necessitating a strategic approach to navigate the complexities of the pharmaceutical landscape. By implementing various strategies—such as expedited ethical approvals, leveraging human expertise and technology, and fostering partnerships with CDMOs—companies can significantly enhance their drug development processes. The focus on patient-centric approaches and the integration of diverse populations enriches the understanding of drug efficacy and safety, ensuring that therapies are tailored to meet the needs of all individuals.
Key insights from the article underscore the importance of:
- Regulatory compliance
- Innovative formulation techniques
- Utilization of data analytics
Understanding regulatory requirements is essential for mitigating challenges, while innovative techniques like nanotechnology and liposomal preparations play a crucial role in enhancing drug efficacy. Additionally, the power of data analytics in optimizing drug formulation processes is paramount, as it empowers researchers to make informed decisions based on real-time insights.
Ultimately, continuous investment in training research teams and establishing feedback loops fosters a culture of improvement and innovation. By prioritizing these strategies, organizations can accelerate the development of effective medications and contribute to a more inclusive and responsive healthcare system. Embracing these practices is not merely a pathway to success in clinical research; it represents a commitment to improving patient outcomes and advancing the future of medicine.
Frequently Asked Questions
What is bioaccess® and how does it benefit clinical research?
bioaccess® is a company that leverages its locations in Latin America, the Balkans, and Australia to secure ethical approvals for clinical research in an impressive 4-6 weeks. This expedited process helps Medtech, Biopharma, and Radiopharma innovators accelerate their market entry by minimizing delays and allowing clients to focus on their core research activities.
How do rapid ethical approvals impact clinical trials?
Rapid ethical approvals can boost enrollment speeds by up to 50% and significantly shorten the overall time to market. This is crucial in a competitive industry where delays in clinical trials can cost sponsors between $600,000 and $8 million per day.
Can you provide an example of a company that benefited from bioaccess®?
Sparta Biomedical experienced substantial benefits during its initial human study in Colombia by collaborating with bioaccess®, highlighting the advantages of their expedited approval process.
What role does AI and machine learning play in drug formulation?
AI and machine learning enhance the medication development process by facilitating rapid iterations and improvements through real-time data analysis. They help predict physicochemical properties, improve solubility and stability, and reduce the risk of costly trial failures.
How much can AI reduce medication development costs?
AI-driven platforms can reduce medication development costs by as much as 25%, transforming traditional practices in the pharmaceutical sector.
What is the role of formulation scientists in drug development?
Formulation scientists play a critical role in drug formulation and the development of medicine by integrating advanced technologies like AI, which aids in decision-making and enhances the accuracy of drug formulations, ultimately leading to improved patient outcomes.
What are the benefits of partnering with CDMOs in drug development?
Partnering with Contract Development and Manufacturing Organizations (CDMOs) streamlines the pharmaceutical creation process by providing comprehensive services that encompass development and manufacturing. This collaboration helps reduce time-to-market and ensures compliance with regulatory standards, allowing companies to focus on their core competencies.
List of Sources
- bioaccess®: Accelerate Clinical Research with Fast Ethical Approvals
- antidote.me (https://antidote.me/blog/what-clinical-trial-statistics-tell-us-about-the-state-of-research-today)
- bioaccessla.com (https://bioaccessla.com/blog/9-benefits-of-investigator-initiated-studies-for-clinical-research)
- Factors influencing the time to ethics and governance approvals for clinical trials: a retrospective cross-sectional survey – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10693024)
- Paul-Ehrlich-Institut – Information on procedures and timelines for submission of clinical trials (https://pei.de/EN/regulation/clinical-trials/procedures-timelines/procedures-timelines-node.html)
- bioaccessla.com (https://bioaccessla.com/es/blog/7-ways-croe-accelerates-clinical-research-for-medtech-startups)
- Leverage Human Expertise and Technology for Drug Formulation
- AI in Drug Discovery: How AI Is Accelerating Pharma Research (Key Stats) (https://patentpc.com/blog/ai-in-drug-discovery-how-ai-is-accelerating-pharma-research-key-stats)
- fjps.springeropen.com (https://fjps.springeropen.com/articles/10.1186/s43094-024-00625-1)
- pharma-stats.com (https://pharma-stats.com/pharmaceutical-statistics)
- Accelerating Drug Development with AI in the U.S. Pharmaceutical Industry | IntuitionLabs (https://intuitionlabs.ai/articles/accelerating-drug-development-ai-pharma)
- sciencedirect.com (https://sciencedirect.com/science/article/pii/S266732582400205X)
- Partner with CDMOs for Streamlined Drug Development
- fortunebusinessinsights.com (https://fortunebusinessinsights.com/contract-development-and-manufacturing-organization-cdmo-outsourcing-market-102502)
- cervicornconsulting.com (https://cervicornconsulting.com/contract-development-and-manufacturing-organization-market)
- expertmarketresearch.com (https://expertmarketresearch.com/reports/contract-development-and-manufacturing-organization-cdmo-market?srsltid=AfmBOoowFgSZYideM8_ZjSF6WYL_Kt3Qw68mnNXWqr7EEc1-kx1aG33R)
- statifacts.com (https://statifacts.com/outlook/pharmaceutical-cdmo-for-formulations-market)
- globenewswire.com (https://globenewswire.com/news-release/2025/05/06/3075216/0/en/Pharmaceutical-CDMO-Market-Set-to-Surpass-315-08-Billion-by-2034-Driven-by-7-24-CAGR.html)
- Understand Regulatory Requirements to Navigate Formulation Challenges
- futuremarketinsights.com (https://futuremarketinsights.com/reports/drug-formulation-market)
- Regulatory Trends in Pharma Manufacturing: Key Changes to Watch in 2025 (https://news-medical.net/life-sciences/Regulatory-Trends-in-Pharma-Manufacturing-Key-Changes-to-Watch-in-2025.aspx)
- Five Regulatory Trends to Harness in 2025 (https://contractpharma.com/exclusives/five-regulatory-trends-to-harness-in-2025)
- thebusinessresearchcompany.com (https://thebusinessresearchcompany.com/report/drug-formulation-global-market-report)
- 2025 Safety and Regulatory Compliance Trends and Predictions for Pharma and Biotech (https://iqvia.com/library/white-papers/2025-safety-and-regulatory-compliance-trends-and-predictions-for-pharma-and-biotech)
- Implement Innovative Formulation Techniques for Enhanced Efficacy
- fastercapital.com (https://fastercapital.com/content/Drug-formulation-Innovative-Drug-Formulation-Techniques–A-Game-Changer-for-Startups.html)
- openaccessjournals.com (https://openaccessjournals.com/articles/advancements-in-drug-formulation-innovations-shaping-the-future-of-medicine-16658.html)
- datahorizzonresearch.com (https://datahorizzonresearch.com/liposome-drug-delivery-market-4269)
- finance.yahoo.com (https://finance.yahoo.com/news/lipid-nanoparticles-market-industry-trends-100000788.html)
- databridgemarketresearch.com (https://databridgemarketresearch.com/reports/global-liposome-drug-delivery-market?srsltid=AfmBOoqh55E2x4NJPo6A_kJdx9zQxE-r0ioX2__6RNrm2EZsXzkvTmkh)
- Adopt Patient-Centric Approaches in Drug Formulation
- researchgate.net (https://researchgate.net/publication/379268499_Patients’_Preference_for_Pharmaceutical_Dosage_Forms_Does_It_Affect_Medication_Adherence_A_Cross-Sectional_Study_in_Community_Pharmacies)
- dovepress.com (https://dovepress.com/patient-acceptability-and-preferences-for-solid-oral-dosage-form-drug–peer-reviewed-fulltext-article-PPA)
- mdpi.com (https://mdpi.com/1660-4601/18/9/4916)
- The impact of patients’ preference for pharmaceutical dosage forms on medication discontinuation among patients attending Red Cross pharmacies in Northwest Ethiopia – Scientific Reports (https://nature.com/articles/s41598-024-76113-6)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC6266025)
- Utilize Diverse Patient Populations for Comprehensive Insights
- clinicaltrialsarena.com (https://clinicaltrialsarena.com/news/fda-report-highlights-need-to-improve-diversity-in-clinical-trials)
- statnews.com (https://statnews.com/2022/06/23/needed-clearer-explanation-importance-diversity-in-clinical-trials)
- Key Trends in Demographic Diversity in Clinical Trials – Improving Representation in Clinical Trials and Research – NCBI Bookshelf (https://ncbi.nlm.nih.gov/books/NBK584392)
- Embracing Diversity: The Imperative for Inclusive Clinical Trials | Harvard Medical School Professional, Corporate, and Continuing Education (https://learn.hms.harvard.edu/insights/all-insights/embracing-diversity-imperative-inclusive-clinical-trials)
- Rebooting the Statistic That 5% of Eligible Patients Participate in Clinical Trials | Applied Clinical Trials Online (https://appliedclinicaltrialsonline.com/view/rebooting-the-statistic-that-5-of-eligible-patients-participate-in-clinical-trials)
- Invest in Continuous Training for Research Teams
- Data Management in Clinical Research: Best Practices | Datavant (https://datavant.com/clinical-research/clinical-data-management)
- 2026 Training Industry Statistics: Data, Trends & Predictions | Research.com (https://research.com/careers/training-industry-statistics)
- Top-Notch 10 Statistics That Highlight The Significance Of Continuous Training (https://trainingindustry.com/articles/performance-management/top-notch-10-statistics-that-highlight-the-significance-of-continuous-training)
- vouchfor.com (https://vouchfor.com/blog/continuous-learning-quotes)
- Leverage Data Analytics to Optimize Drug Formulation
- mdpi.com (https://mdpi.com/2227-9717/11/7/2096)
- sciencedirect.com (https://sciencedirect.com/science/article/abs/pii/S2452414X25000202)
- nature.com (https://nature.com/articles/s41598-025-05535-7)
- pharmafocuseurope.com (https://pharmafocuseurope.com/information-technology/pharmaceutical-industry-data-science)
- intechopen.com (https://intechopen.com/chapters/78091)
- Establish Feedback Loops for Continuous Improvement in Formulation
- researchgate.net (https://researchgate.net/publication/363274213_Pharmaceutical_Feedback_Loop_-_A_Concept_to_Improve_Prescription_Safety_and_Data_Quality)
- drugpatentwatch.com (https://drugpatentwatch.com/blog/pharma-starts-a-feedback-loop?srsltid=AfmBOoo03fT82r_HsECEvooa4DbprQPWLSTfFKnQdlDuBPbbGYmcdyMS)
- advisoryexcellence.com (https://advisoryexcellence.com/the-impact-of-stakeholder-feedback-on-project-outcomes)
- 26 powerful quotes about feedback (https://netigate.net/articles/surveys/quotes-about-feedback)
- 49 Quotes on the Power of Feedback (https://linkedin.com/pulse/49-quotes-power-feedback-judy-romano-mba)

Leave a Reply