Introduction
The landscape of clinical trials is rapidly evolving, propelled by the urgent need for innovative solutions in Medtech and Biopharma. As these industries endeavor to introduce groundbreaking therapies to the market, the significance of robust clinical trial supply services has never been more pronounced. This article explores ten essential services that not only streamline the research process but also tackle the unique challenges encountered by Medtech innovators.
What strategies can companies implement to navigate the complexities of clinical trials while ensuring efficiency and compliance? The answers are found within the diverse range of services that can transform the landscape of clinical research.
bioaccess: Accelerated Clinical Trial Supply Services for Medtech and Biopharma
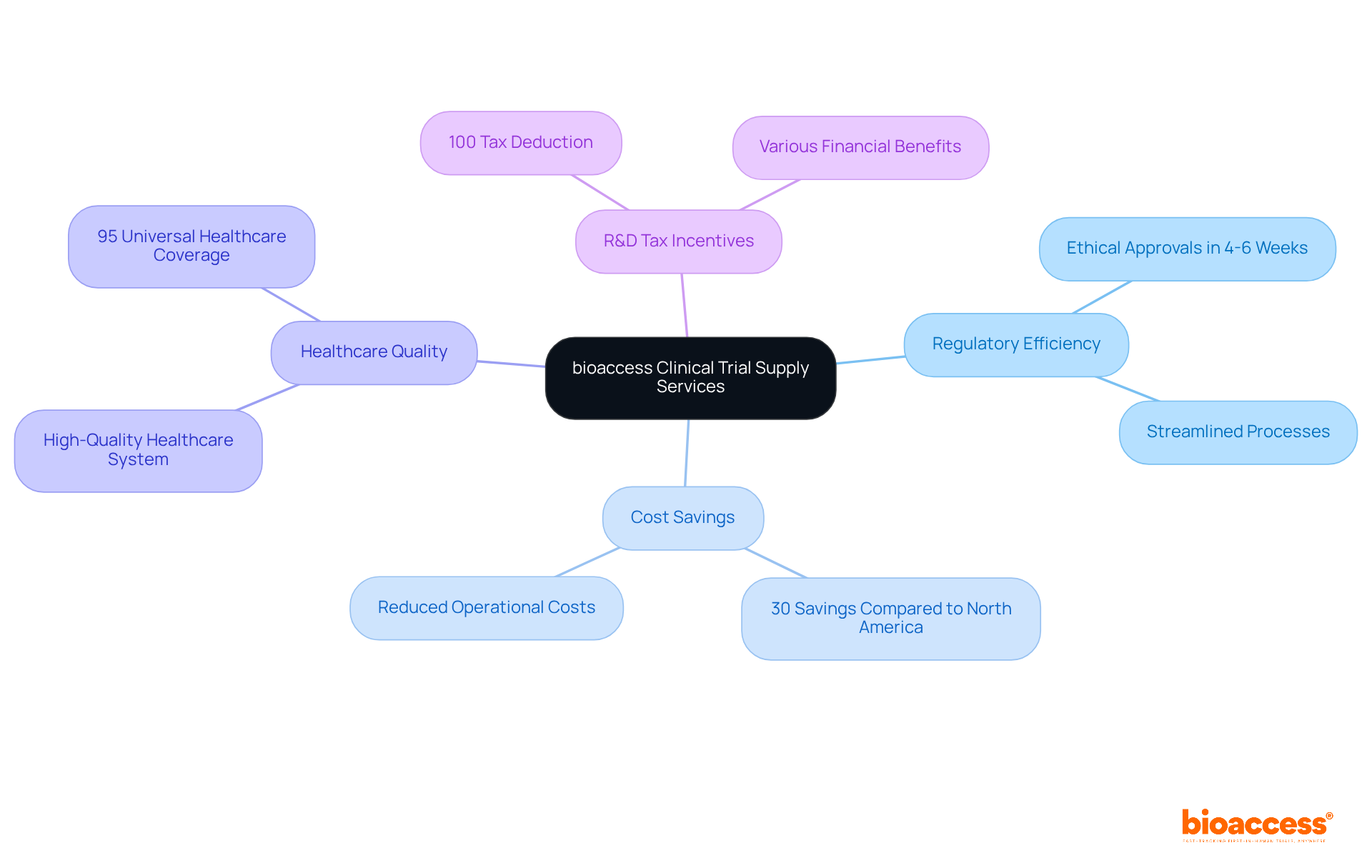
bioaccess® delivers expedited research supply services that leverage Colombia’s competitive advantages, including , enabling ethical approvals in just 4-6 weeks. This rapid transition is essential for Medtech and Biopharma firms seeking to and bring innovations to market more swiftly. With cost savings exceeding 30% compared to North America and Western Europe, bioaccess® empowers clients to sustain momentum in their research and development efforts. Furthermore, Colombia boasts a , ranked among the best globally, alongside a population of over 50 million with 95% universal healthcare coverage, facilitating .
In addition, the , which feature a 100% tax deduction and various financial benefits, enhance the attractiveness of conducting studies in the region. By optimizing the supply chain and ensuring the timely delivery of investigational products, bioaccess® offers that help clients navigate the complexities of clinical studies within Colombia’s regulatory framework. This support is particularly beneficial for operations in Latin America, Eastern Europe, and Australia, reinforcing bioaccess®’s commitment to addressing the challenges faced by the Medtech landscape and positioning itself as a vital partner in for clinical research.
Cencora: Comprehensive Drug Research and Clinical Development Support
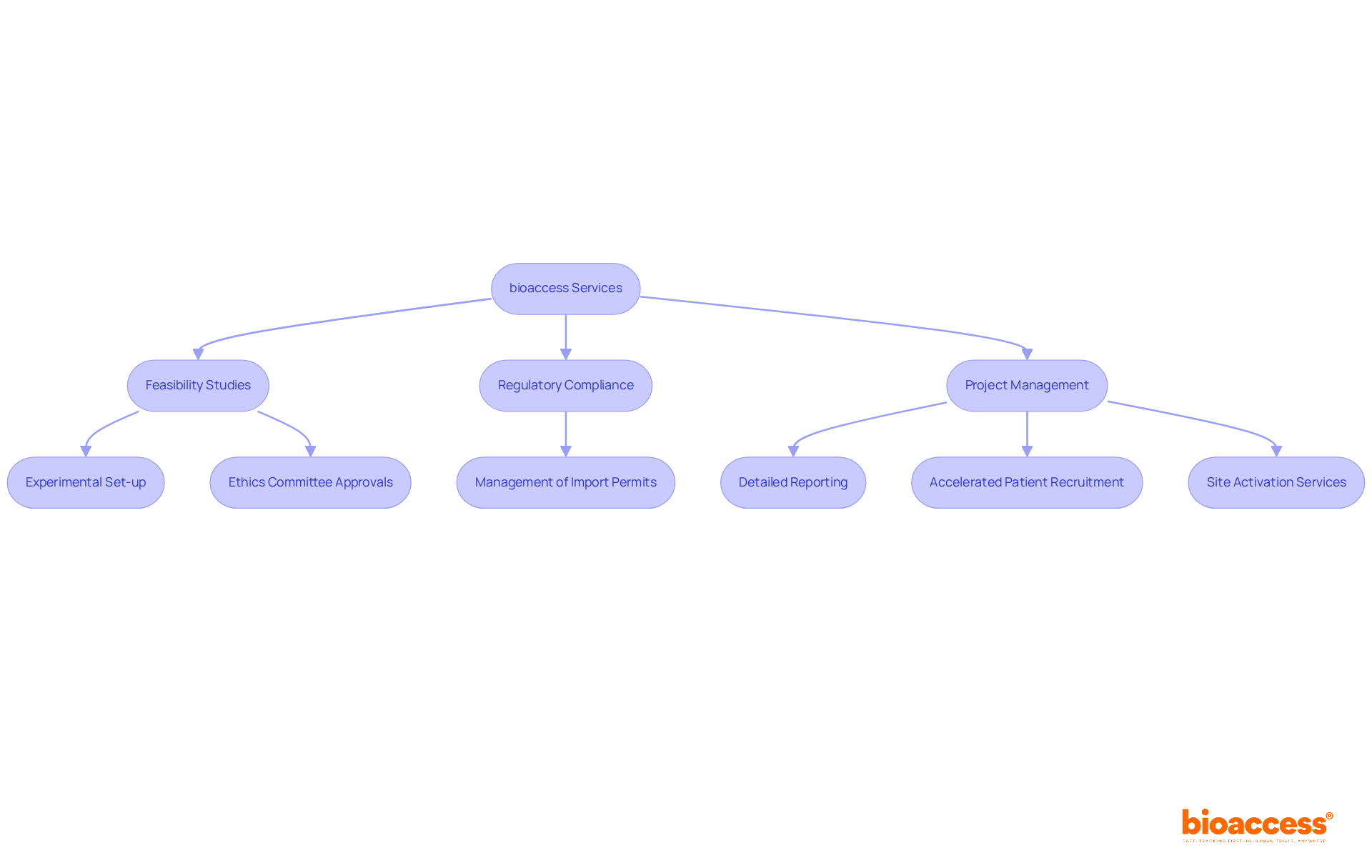
bioaccess offers an extensive range of services designed to effectively. Their expertise includes:
- Selection of
- Ensuring compliance with
This encompasses:
- Experimental set-up
- Start-up approvals from ethics committees and health ministries
- Management of
- Detailed reporting on study status, inventory, and adverse events
By providing robust , bioaccess guarantees that all facets of the research process are meticulously managed.
In the face of challenges such as regulatory hurdles, competition, recruitment issues, and financial constraints, bioaccess distinguishes itself by offering accelerated and site activation services. Their partnership with Caribbean Health Group aims to establish Barranquilla as a premier location for medical studies in Latin America, a goal supported by Colombia’s Minister of Health. This strategic partnership not only enhances the research landscape in the region but also addresses the critical need for effective recruitment strategies, particularly for underrepresented groups.
Furthermore, bioaccess’s collaboration with GlobalCare Clinical Trials has yielded significant success, achieving over a 50% reduction in recruitment time while maintaining a 95% retention rate. This underscores their commitment to improving research outcomes and ensuring that technology and pharmaceutical firms can navigate the complexities of studies efficiently, ultimately accelerating the path to commercialization.
Catalent: Tailored Direct-to-Patient Clinical Supply Services
bioaccess® excels in delivering comprehensive tailored for Medtech and Biopharma startups. Leveraging its extensive network across Latin America, bioaccess® accelerates through meticulous feasibility assessments, , , meticulous study preparation, and streamlined import permits. This innovative approach not only enhances but also addresses , ensuring that studies are conducted seamlessly and efficiently.
By focusing on project management and detailed reporting, bioaccess® empowers Medtech companies to refine their research processes and elevate participant engagement. The impact of these medical studies extends beyond the experiments themselves; they contribute to local economies through job creation, , and improved healthcare outcomes, fostering international collaboration in the process.
 and study efficiency. Each box shows a step in the clinical study management process. Follow the arrows to see how each step leads into the next, ultimately enhancing patient recruitment and study efficiency.](https://images.tely.ai/telyai/cmiyxnfq-each-box-shows-a-step-in-the-clinical-study-management-process-follow-the-arrows-to-see-how-each-step-leads-into-the-next-ultimately-enhancing-patient-recruitment-and-study-efficiency.webp)
Marken: Expert Clinical Trial Supply Chain Management
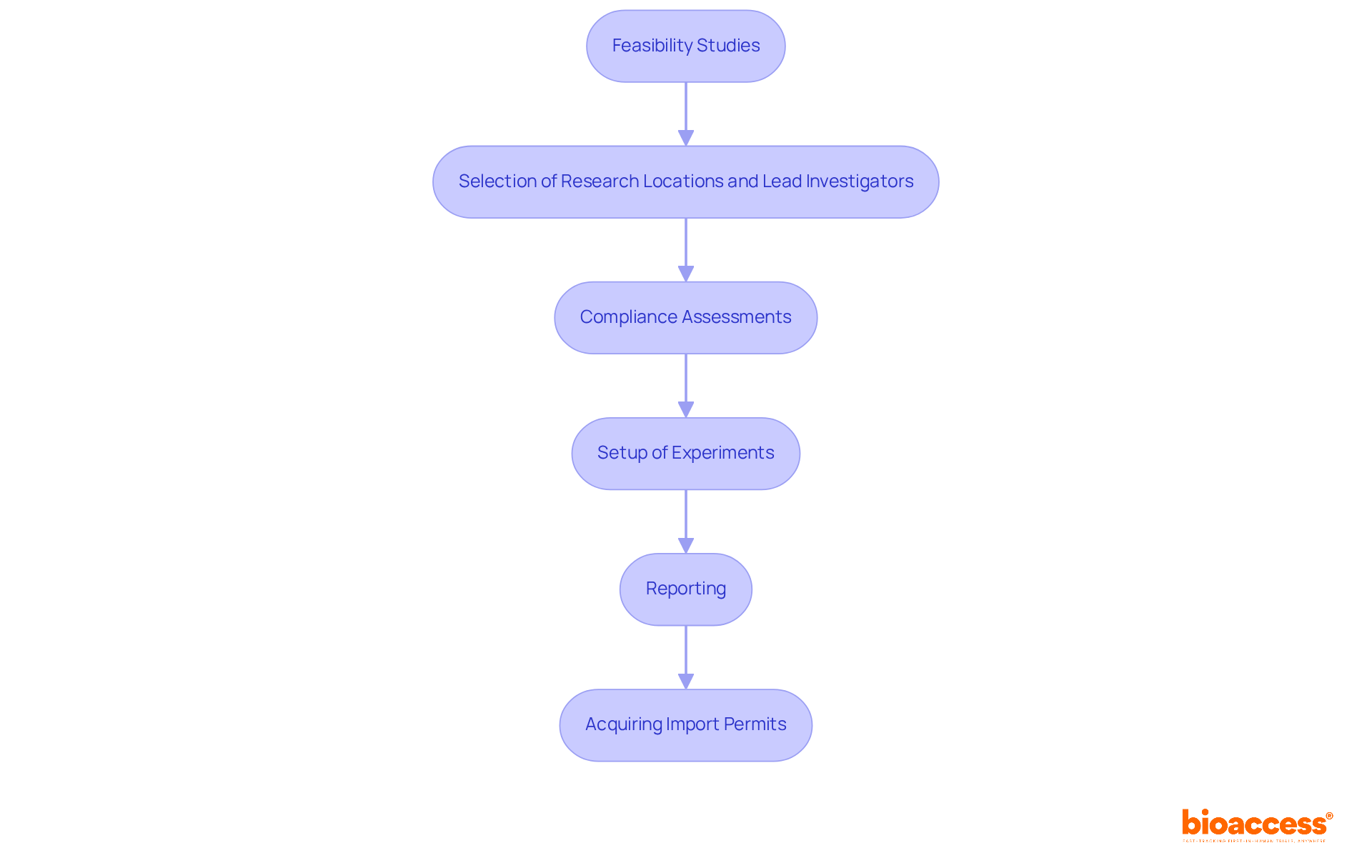
Bioaccess excels in , effectively managing the supply chain for research studies and delivering investigational products with accuracy and dependability. Their extensive service capabilities encompass:
- The selection of research locations and lead investigators
- Compliance assessments of study documents
- The setup of experiments
- Reporting
- Acquiring necessary import permits
This comprehensive method is essential for guaranteeing that research studies are carried out effectively and in accordance with regulatory standards.
The are particularly crucial for . They employ advanced that ensure compliance with stringent regulatory standards. This capability is essential, as even slight variations in temperature can undermine the integrity of research materials, potentially influencing study results. The medical research resources market is expected to expand from $3.9 billion in 2023 to $6.3 billion by 2028, emphasizing the rising need for strong logistics solutions. As Priyanka Bhendale, team lead at Markets and Markets, observes, ‘Logistics and distribution are essential elements of in the clinical research materials market.’
By leveraging and IoT devices, Bioaccess ensures continuous monitoring of environmental conditions, enhancing visibility throughout the supply chain. This allows Medtech companies to proactively address potential disruptions. Successful instances of their involve collaborations that have efficiently handled the delivery of sensitive biologics and investigational drugs, ensuring they reach research locations in optimal condition. This strategic approach not only reduces risks but also strengthens the integrity of research studies, ultimately aiding the progress of innovative therapies.
To maximize the effectiveness of , Medtech companies should consider implementing that provide real-time data on environmental conditions during transit.
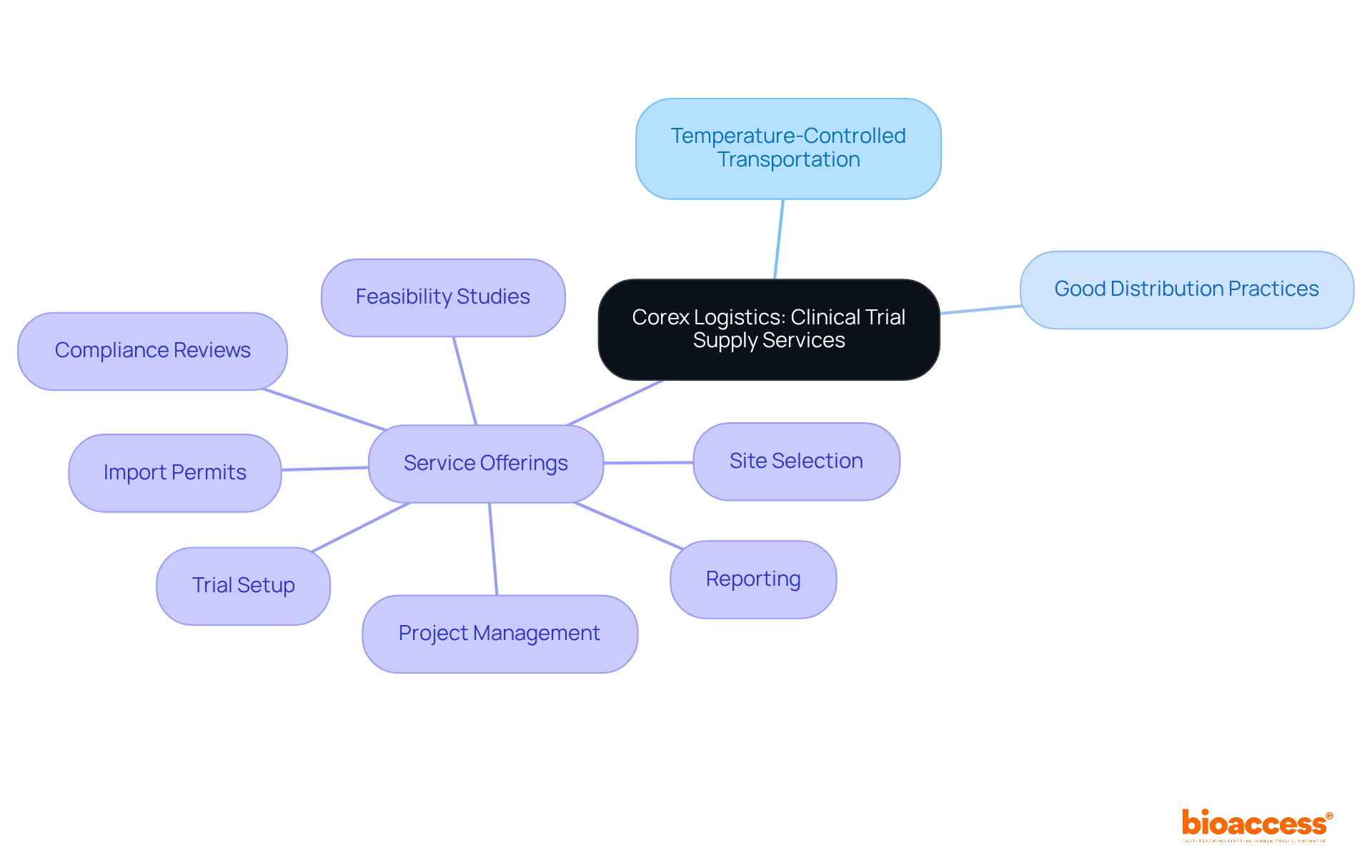
Corex Logistics: Reliable Clinical Trial Supply Services
Corex Logistics is recognized for its reliable in clinical research, emphasizing temperature-controlled transportation and strict adherence to Good Distribution Practices (GDP). Their extensive network and expertise in managing ensure that research materials are delivered efficiently and safely.
Coupled with Bioaccess’s comprehensive , which encompass:
- Import permits
- Reporting
the critical role of logistics in clinical trials is further highlighted. Medtech companies can depend on Corex to manage their , enabling them to concentrate on their core research activities.
As the landscape of research studies evolves, the demand for specialized , such as those offered by Corex and Bioaccess, continues to rise, underscoring the vital role of and thorough study services in achieving successful outcomes. Furthermore, the growing complexity of medical studies necessitates specialized logistics solutions, making Corex’s commitment to excellence increasingly essential.
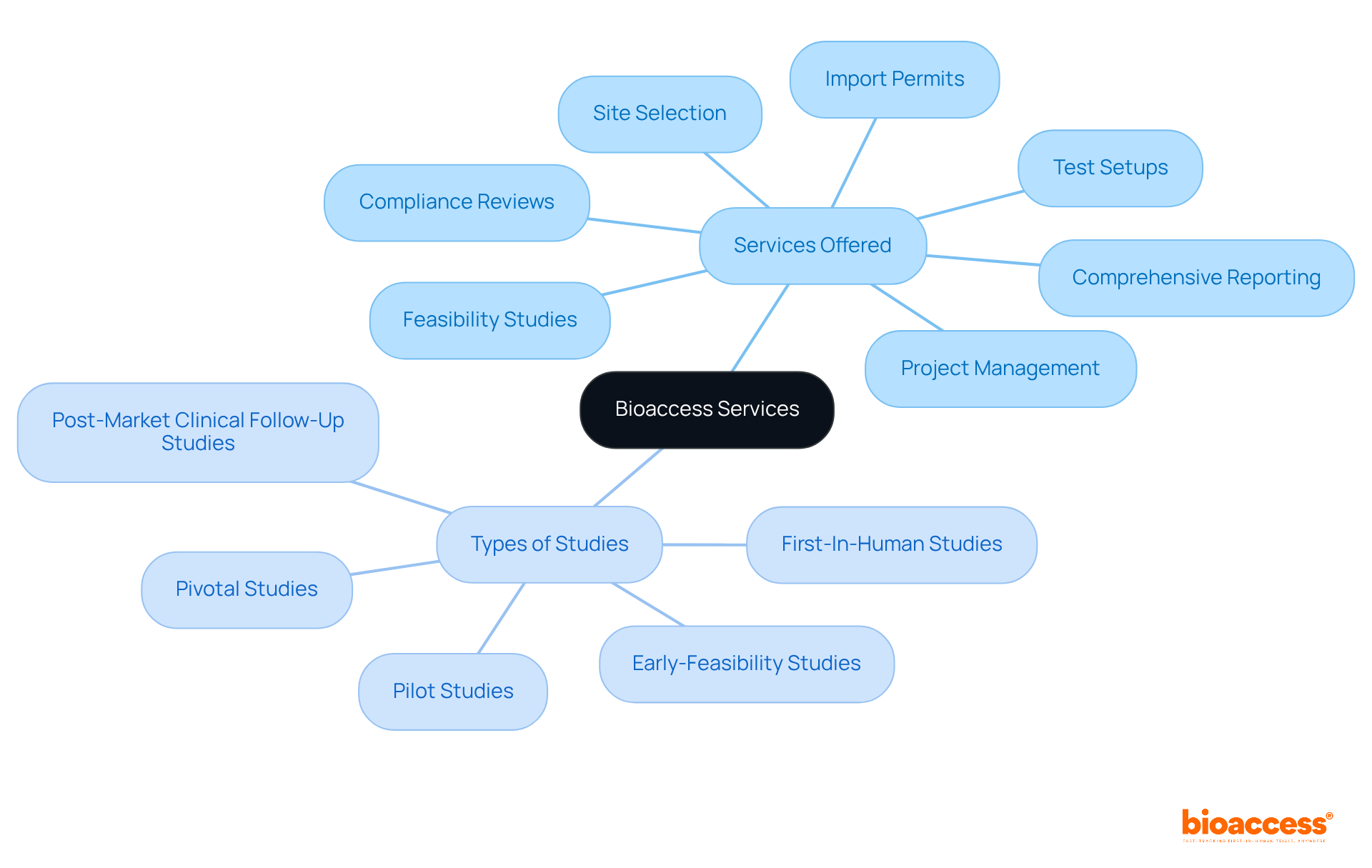
Proventa International: Solutions for Clinical Trial Supply Chain Challenges
bioaccess® delivers innovative solutions to common logistics challenges, such as visibility and communication issues, tailored specifically for Medtech, Biopharma, and Radiopharma startups. Their strategic approach leverages technology and data analytics to enhance efficiency, ensuring that materials are delivered promptly and in compliance with regulatory standards.
By partnering with bioaccess®, companies can access a comprehensive suite of services, including:
- Study design
This collaboration facilitates faster site activation and compliance, particularly for in Colombia and extends to regions such as Latin America, Eastern Europe, and Australia.
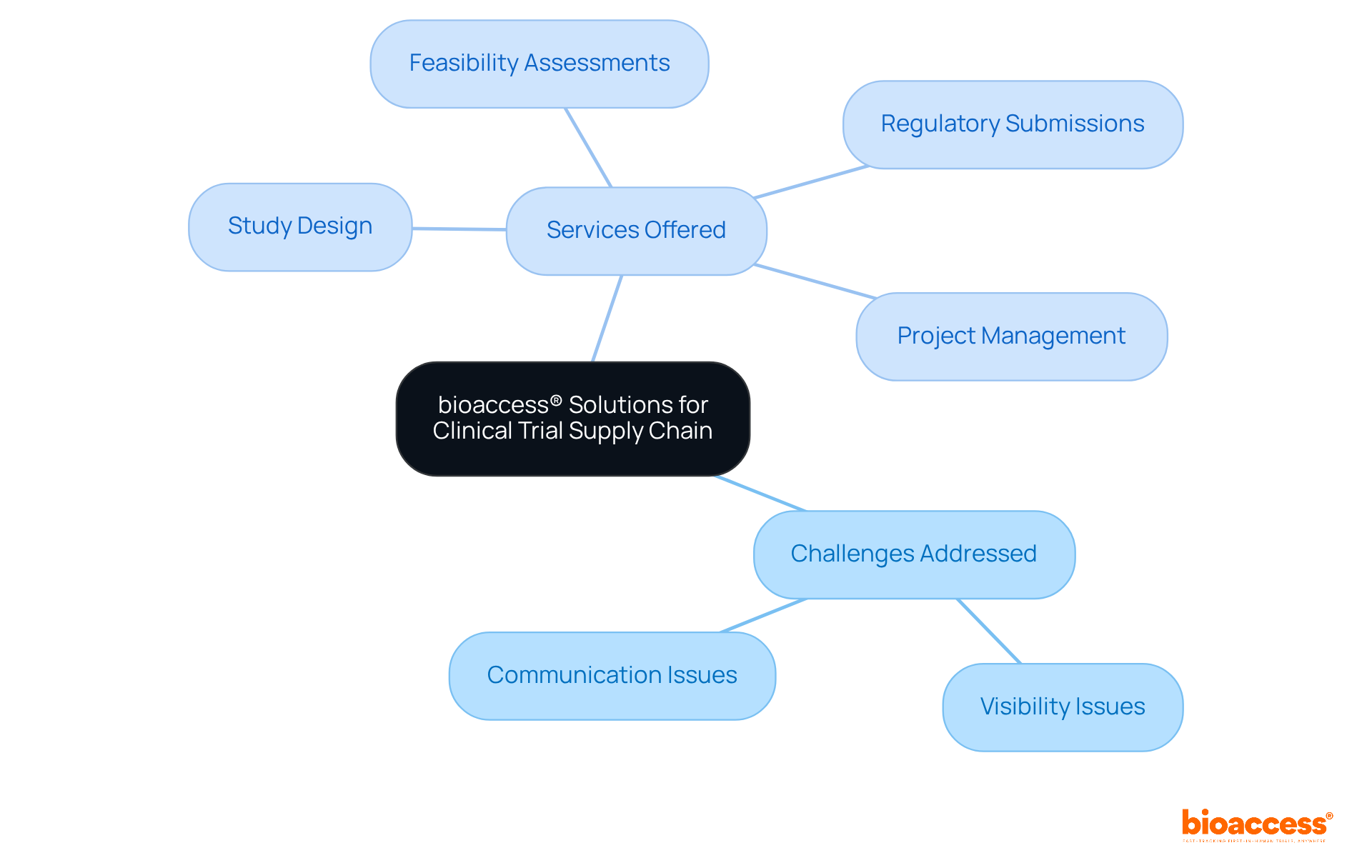
Experic Services: Secure Clinical Storage and Logistics Solutions
Experic Services excels in providing secure medical storage and logistics solutions, featuring temperature-controlled environments and stringent . These facilities are meticulously designed to meet the rigorous standards of , ensuring that are stored and handled with the utmost care.
Maintaining a robust chain-of-custody is essential, as it directly impacts the integrity of evidence materials. In fact, studies indicate that can reduce discrepancies in case data by up to 30%.
As William A. Foster aptly stated, ‘Quality is never an accident; it is always the result of high intention, sincere effort, intelligent direction, and skillful execution.’
By leveraging Experic’s , can ensure the throughout the research process, ultimately leading to more reliable outcomes.
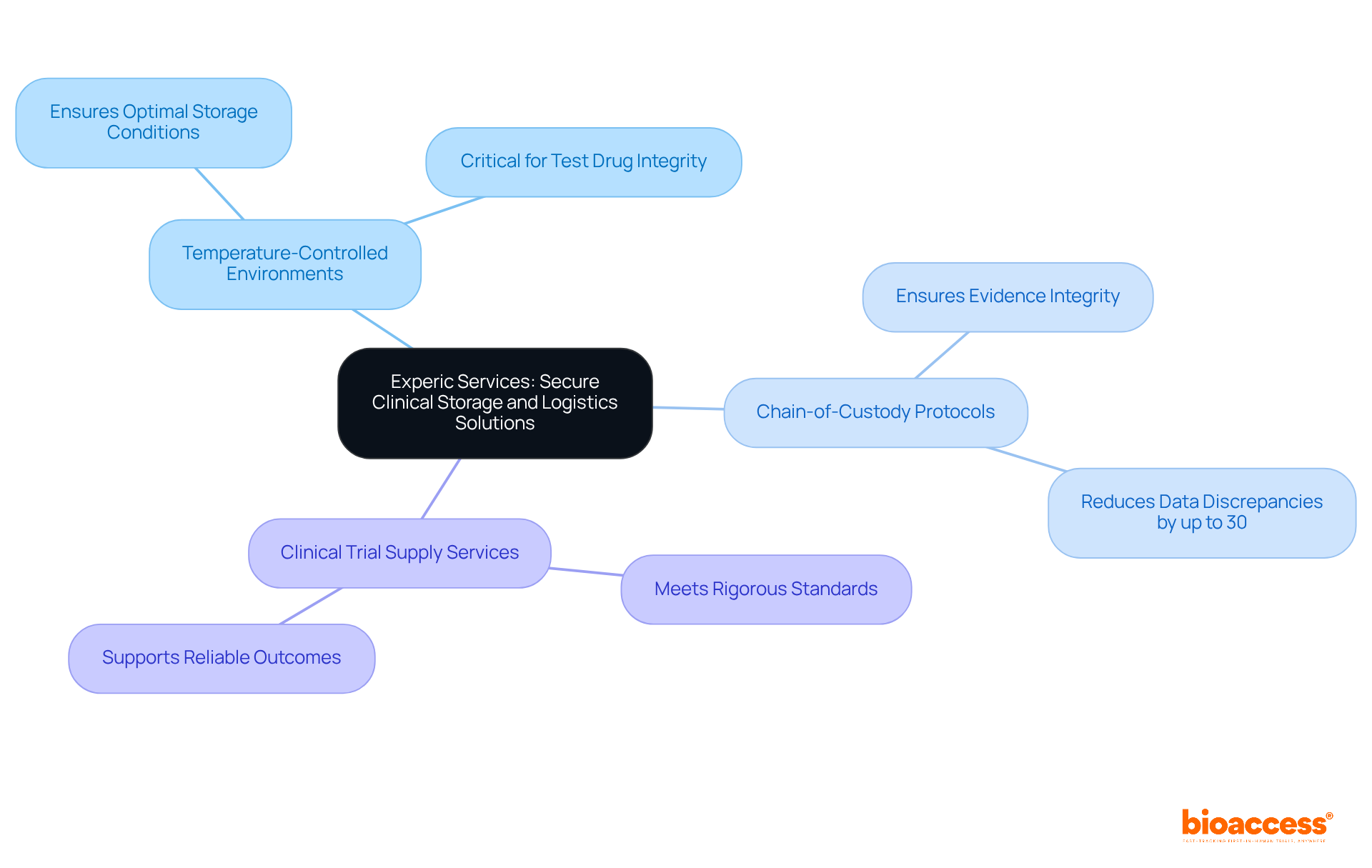
Icon PLC: Comprehensive Clinical Supplies Management
Efficient management of medical resources is essential for aiming to optimize and . Comprehensive encompass:
- Feasibility studies
- Site selection
- Compliance reviews
- Trial setup
- Import permits
- Project management
- Reporting
Each is critical for successful . and just-in-time inventory strategies exemplify effective management of temperature-sensitive products, ensuring resources are available when needed while minimizing waste.
Current trends indicate a shift towards . AI-driven forecasting and IoT-enabled tracking systems are enhancing chain transparency and efficiency. These innovations not only mitigate logistical risks but also improve compliance with regulatory requirements, ultimately accelerating the research process.
Research directors emphasize the significance of robust inventory management, asserting that ” are crucial in managing the intricacies of research studies.” As the research supply market evolves, with , staying abreast of these trends will empower medical technology firms to refine their operations and focus on delivering innovative therapies to patients. Furthermore, the successful execution of research studies can profoundly impact local economies, fostering job creation, economic development, and healthcare improvements, thereby promoting international collaboration within the medical technology sector.
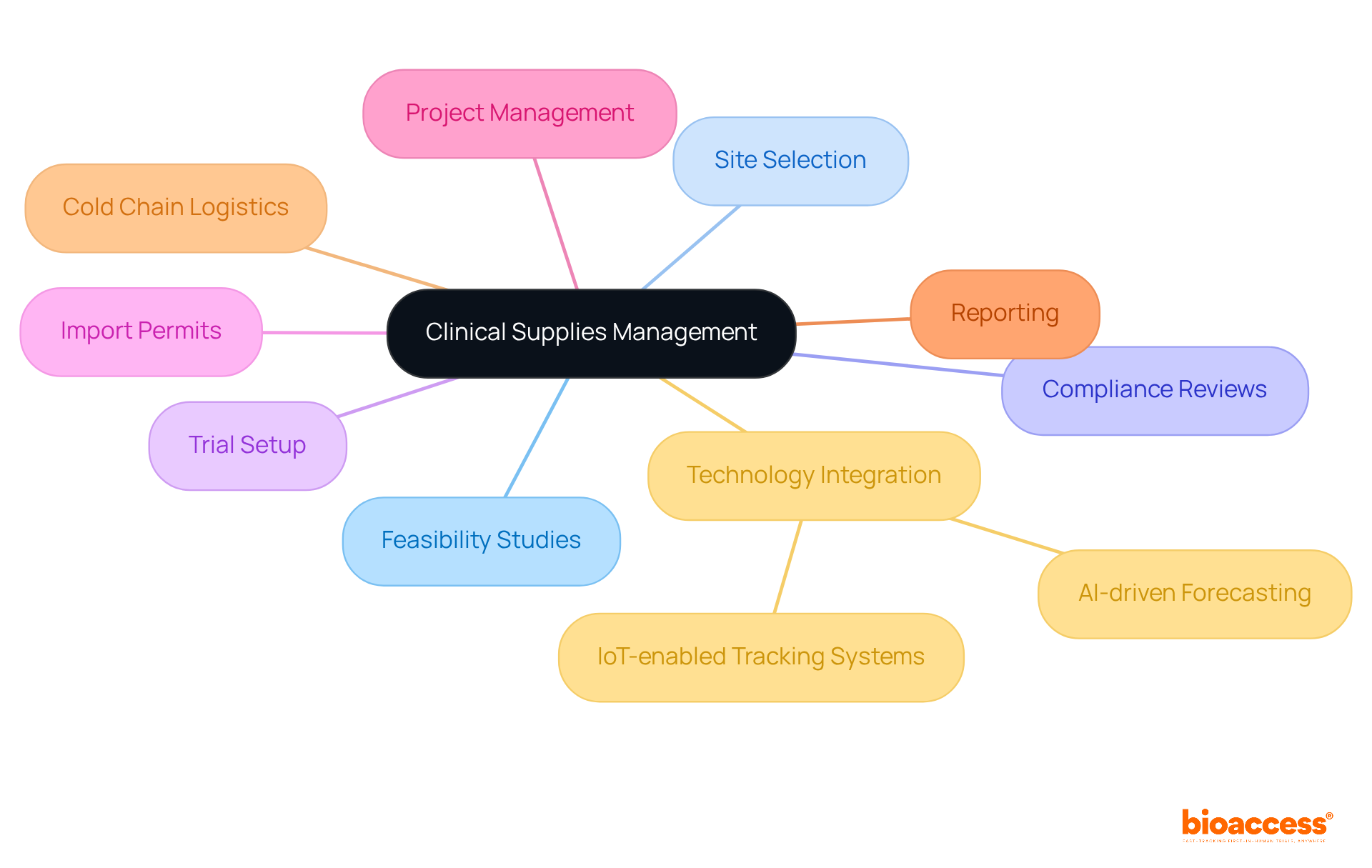
McKesson: Simplified Clinical Trial Logistics and Research Services
Bioaccess plays a crucial role in optimizing , focusing on the thorough management of studies. With over 20 years of experience in medical technology, bioaccess adeptly navigates the complexities of research studies, enabling businesses to .
By leveraging bioaccess’s services, benefit from:
- Expert feasibility studies
- Site selection
- Compliance reviews
- Test setups
- Import permits
- Project management
Bioaccess specializes in managing:
Current trends indicate a growing emphasis on , positioning bioaccess at the forefront of expedited medical device research services in Latin America. This proactive approach not only accelerates the testing process but also elevates the overall quality of research outcomes. Medtech innovators are encouraged to collaborate with bioaccess to adeptly navigate the evolving landscape of medical studies and achieve successful results.
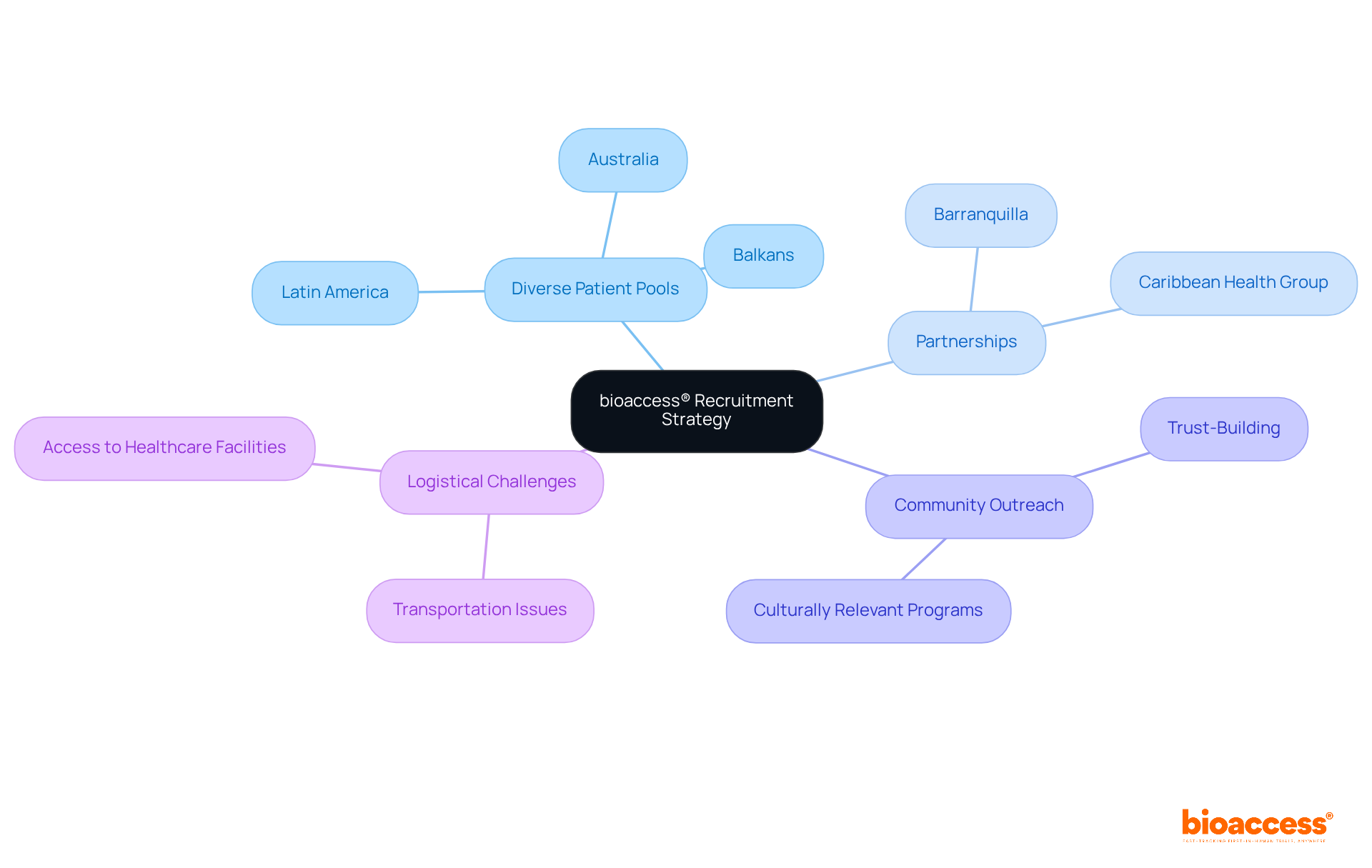
bioaccess: Leveraging Diverse Patient Pools for Faster Recruitment
bioaccess® strategically employs from Latin America, the Balkans, and Australia to optimize recruitment for research studies. This approach not only increases participant diversity but also greatly . By engaging with varied demographics, bioaccess® enables Medtech companies to and achieve objectives more efficiently. The is crucial, as it yields comprehensive insights into treatment efficacy across different groups, ultimately fostering innovations applicable to a broader patient base. This commitment to diversity not only enriches the research environment but also aligns with industry leaders’ demands for inclusive practices that enhance the reliability and applicability of study results.
Moreover, bioaccess®’s partnership with Caribbean Health Group aims to establish Barranquilla as a premier location for , endorsed by the approval of Colombia’s Minister of Health. This collaboration is expected to improve , leading to quicker recruitment and more effective data delivery. To further enhance participant diversity, bioaccess® underscores the significance of and trust-building within these populations, as emphasized by industry experts like Singh, who advocate for . Additionally, addressing , such as transportation and access to healthcare facilities, is vital for ensuring that various groups can participate in research trials. By implementing actionable strategies—such as providing incentives for participation and collaborating with local organizations—bioaccess® strives to cultivate a more inclusive environment that benefits all stakeholders in the clinical research process.
Conclusion
The landscape of clinical trial supply services is pivotal for Medtech innovators striving to enhance research efficiency and accelerate market entry. By leveraging the strengths of various service providers, including bioaccess, Cencora, and others, companies can navigate the complexities of clinical trials with greater ease. These services not only facilitate regulatory compliance but also optimize logistics and patient recruitment, ultimately ensuring that innovative medical technologies reach those who need them most.
Key insights from the article highlight the importance of tailored solutions, such as temperature-controlled logistics and diverse patient recruitment strategies, which significantly impact the success of clinical studies. The collaboration between organizations like bioaccess and Caribbean Health Group exemplifies how strategic partnerships can improve recruitment processes and data quality, benefiting both the research community and the populations they serve.
As the Medtech sector continues to evolve, embracing these essential clinical trial supply services will be crucial for organizations aiming to stay competitive. By prioritizing innovation, efficiency, and inclusivity in clinical trials, Medtech companies can not only enhance their operational effectiveness but also contribute positively to global healthcare outcomes. Engaging with these services today will pave the way for tomorrow’s breakthroughs in medical technology.
Frequently Asked Questions
What services does bioaccess® provide for Medtech and Biopharma firms?
bioaccess® offers expedited research supply services, including regulatory efficiency for ethical approvals, clinical trial supply services, and comprehensive project management to enhance medical device evaluations.
How quickly can bioaccess® facilitate ethical approvals in Colombia?
bioaccess® can enable ethical approvals in just 4-6 weeks, which is crucial for firms looking to expedite their research processes.
What are the cost benefits of using bioaccess® compared to North America and Western Europe?
Clients can achieve cost savings exceeding 30% when conducting studies with bioaccess® compared to costs in North America and Western Europe.
What advantages does Colombia offer for clinical trials?
Colombia has a high-quality healthcare system, a population of over 50 million with 95% universal healthcare coverage, and R&D tax incentives, including a 100% tax deduction, making it an attractive location for clinical trials.
How does bioaccess® assist in patient recruitment for clinical trials?
bioaccess® accelerates patient recruitment and site activation services, addressing challenges in recruitment, particularly for underrepresented groups, and improving research outcomes.
What role does bioaccess® play in managing regulatory requirements?
bioaccess® ensures compliance with regulatory requirements, including managing start-up approvals from ethics committees, health ministries, and import permits for investigational devices.
What partnerships does bioaccess® engage in to enhance clinical research?
bioaccess® partners with Caribbean Health Group to establish Barranquilla as a premier location for medical studies and collaborates with GlobalCare Clinical Trials to reduce recruitment time significantly while maintaining high retention rates.
How does bioaccess® contribute to local economies through its research studies?
The medical studies conducted by bioaccess® contribute to local economies through job creation, economic growth, and improved healthcare outcomes, fostering international collaboration.
List of Sources
- bioaccess: Accelerated Clinical Trial Supply Services for Medtech and Biopharma
- bioaccessla.com (https://bioaccessla.com/blog/10-key-features-of-clinical-trial-software-for-medtech-success)
- bioaccess® | MedTech, Biopharma & Radiopharma CRO in Latin America (https://bioaccessla.com/br/blog/7-ways-bioaccess-accelerates-clinical-research-for-medtech)
- Latin America’s Landscape For Medtech Clinical Trials (https://clinicalleader.com/doc/latin-america-s-landscape-for-medtech-clinical-trials-0001)
- Latin America: A Compelling Region To Conduct Your Clinical Trials (https://clinicalleader.com/doc/latin-america-a-compelling-region-to-conduct-your-clinical-trials-0001)
- statista.com (https://statista.com/statistics/1013599/latin-america-clinical-trials)
- Cencora: Comprehensive Drug Research and Clinical Development Support
- agingresearch.org (https://agingresearch.org/blog/new-york-times-article-on-clinical-trials-features-quote-from-the-alliance-for-aging-research)
- symogen.com (https://symogen.com/case_studies.html)
- Catalent: Tailored Direct-to-Patient Clinical Supply Services
- Enrollment and Retention: A Strategic Imperative for Clinical Trial Success – ConfidenceResearch (https://confidenceresearch.com/enrollment-and-retention-a-strategic-imperative-for-clinical-trial-success)
- 25+ useful clinical trial recruitment statistics for better results (https://antidote.me/blog/25-useful-clinical-trial-recruitment-statistics-for-better-results)
- Improving Participant Recruitment in Clinical Trials: Comparative Analysis of Innovative Digital Platforms (https://jmir.org/2024/1/e60504)
- Mastering Patient Recruitment in Clinical Trials (https://clinicalleader.com/topic/patient-recruitment-and-enrollment)
- antidote.me (https://antidote.me/blog/what-clinical-trial-statistics-tell-us-about-the-state-of-research-today)
- Marken: Expert Clinical Trial Supply Chain Management
- drug-dev.com (https://drug-dev.com/supply-chain-solutions-from-lab-to-life-strategies-for-unwavering-resilience-in-the-clinical-supply-chain)
- clinicalleader.com (https://clinicalleader.com/doc/clinical-trial-supply-market-outlook-developments-technology-trends-0001)
- verifiedmarketresearch.com (https://verifiedmarketresearch.com/product/clinical-trial-supply-management-market)
- Corex Logistics: Reliable Clinical Trial Supply Services
- grandviewresearch.com (https://grandviewresearch.com/industry-analysis/clinical-trial-supplies-market)
- Case Study | GreenTech Solutions Group, LLC. (https://greentechsolutionsgroup.com/case_studies)
- Experic Services: Secure Clinical Storage and Logistics Solutions
- 5 Important Things for Life Sciences Companies to Consider | MasterControl (https://mastercontrol.com/gxp-lifeline/quality-inspiration)
- Quality Control Quotes Emphasizing Quality Over Quantity (https://vnvis.com/quality-control-quotes-emphasizing-quality-over-quantity)
- 50 Quotes About Quality That Sum Up Its Importance – jonble (https://jonble.com/blog/quotes-about-quality)
- denyo.co.jp (https://denyo.co.jp/english/case_studies/denyo-generators-provide-a-secure-work-environment-for-storage-operations-of-test-drugs-requiring-24-hour-temperature-control)
- Icon PLC: Comprehensive Clinical Supplies Management
- agingresearch.org (https://agingresearch.org/blog/new-york-times-article-on-clinical-trials-features-quote-from-the-alliance-for-aging-research)
- grandviewresearch.com (https://grandviewresearch.com/industry-analysis/clinical-trial-supplies-market)
- qualtrics.com (https://qualtrics.com/blog/research-quotes)
- McKesson: Simplified Clinical Trial Logistics and Research Services
- viima.com (https://viima.com/blog/innovation-quotes)
- precedenceresearch.com (https://precedenceresearch.com/clinical-trial-supply-and-logistics-market)
- AI’s Influence on Healthcare: Key Quotes from 2000 to 2025 (https://askfeather.com/resources/ai-impact-healthcare-quotes-2000-2025)
- 30 Quotes About the Future of Healthcare: Expert Takes (https://deliberatedirections.com/quotes-future-of-healthcare)
- acumenresearchandconsulting.com (https://acumenresearchandconsulting.com/press-releases/clinical-trial-supply-and-logistics-market)
- bioaccess: Leveraging Diverse Patient Pools for Faster Recruitment
- Diversity in Clinical Trials: A Scientific Imperative for 2025 – Clinical Trial Listing Database & Insights | CenterWatch (https://wcgclinical.com/insights/diversity-in-clinical-trials-a-scientific-imperative-for-2025)
- A statistical overview of diversity in clinical trials (https://antidote.me/blog/statistical-overview-of-diversity-in-clinical-trials)
- Embracing Diversity: The Imperative for Inclusive Clinical Trials | Harvard Medical School Professional, Corporate, and Continuing Education (https://learn.hms.harvard.edu/insights/all-insights/embracing-diversity-imperative-inclusive-clinical-trials)
- lazarex.org (https://lazarex.org/state-of-diversity-in-cancer-clinical-trials-2024)
- Improved clinical trial race/ethnicity reporting and updated inclusion profile, 2017–2022: A New Jersey snapshot – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10801241)

Leave a Reply