Introduction
In the fast-evolving world of clinical trials, understanding ICH-GCP guidelines is crucial for success, especially in Latin America. This article delves into the essential principles of ICH-GCP compliance for clinical trials in Chile, offering actionable insights that can significantly enhance research quality and expedite approval processes. Yet, sponsors often wonder: how can they navigate local regulations while ensuring ethical conduct and participant safety?
Navigating the intricate web of local regulations can be daunting for sponsors. Failure to effectively navigate these complexities can lead to delays and compromised study integrity.
Understand ICH-GCP Principles for Compliance
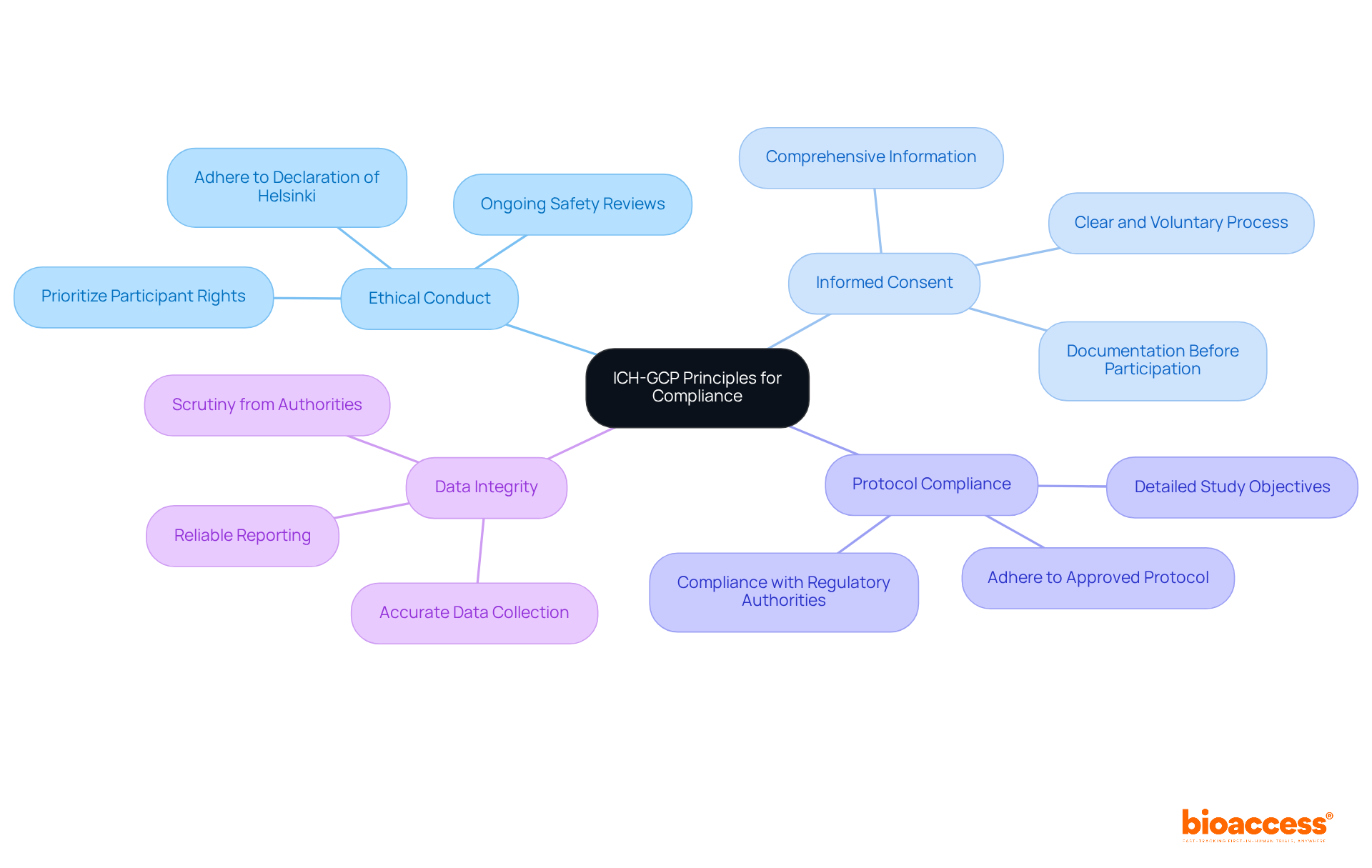
The ICH-GCP clinical trial Chile guidelines are not merely recommendations; they are crucial for ensuring the integrity and success of clinical studies in Latin America where bioaccess® operates. Key principles include:
- Ethical Conduct: Trials must adhere to ethical principles that prioritize the rights, safety, and well-being of participants, ensuring that their interests are placed above scientific objectives, as outlined in the Declaration of Helsinki.
- Informed Consent: Participants must receive comprehensive information regarding the study’s purpose, procedures, risks, and benefits, allowing them to make informed decisions about their participation. This process must be clear, voluntary, and documented before participation.
- Protocol Compliance: Strict adherence to the approved protocol is essential. This document details the study’s objectives, design, methodology, and statistical considerations, ensuring that all trial activities align with compliance expectations from authorities such as ANVISA and COFEPRIS.
- Data Integrity: The collection and reporting of accurate and reliable data are paramount. This ensures that findings are credible and can endure scrutiny from authorities, which is vital for achieving timely approvals from organizations such as ANVISA and COFEPRIS.
Here are some actionable steps for clinical trial sponsors to effectively apply these principles:
- Engage with Regulatory Authorities Early: Establish communication with ANVISA and COFEPRIS to understand specific requirements and timelines for approval, which can range from 30 to 90 days in Latin America.
- Develop Comprehensive Protocols: Ensure that study protocols are meticulously crafted to meet both ICH-GCP standards and local compliance requirements, facilitating smoother approval processes.
- Implement Robust Data Management Systems: Utilize technology to ensure data integrity and compliance with standards, enhancing the credibility of study findings.
Mastering these principles enhances compliance and significantly improves research quality. By prioritizing compliance with ICH-GCP guidelines in the ich-gcp clinical trial chile, sponsors can enhance their research quality and secure a competitive edge in the market. This is especially beneficial in Latin America, where bioaccess® utilizes streamlined approval processes to attain authorizations in as little as 30-90 days, thereby assisting startups in their pursuit of funding and strategic acquisitions. Moreover, organizations recognized for compliant studies enhance their reputations quickly, reinforcing the significance of adhering to these guidelines.
Navigate Chile’s Regulatory Landscape for Clinical Trials
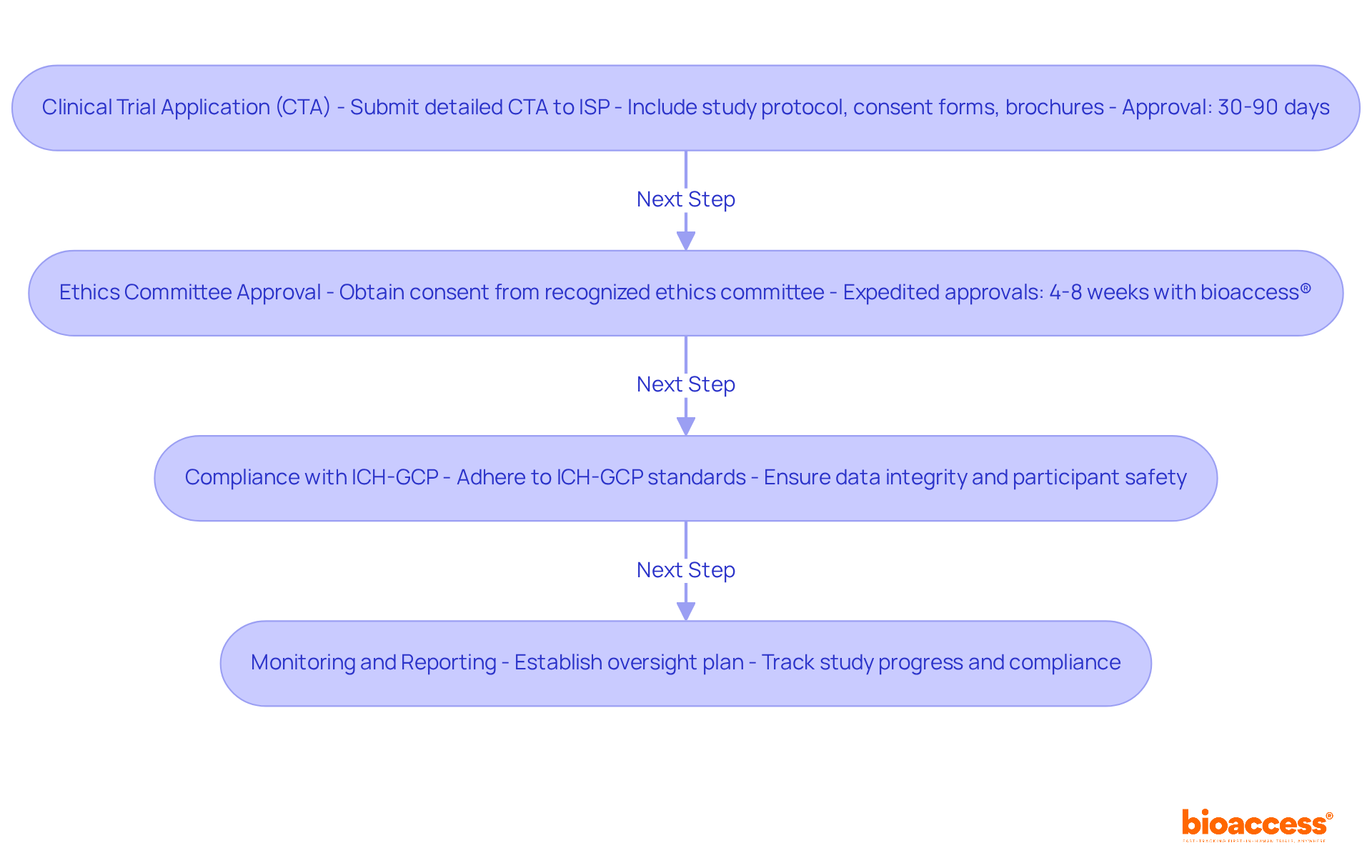
Navigating the regulatory landscape for ich-gcp clinical trial chile is crucial for successful clinical research. Conducting an ich-gcp clinical trial in Chile requires a thorough understanding of the regulatory framework supervised by the Instituto de Salud Pública (ISP). The following steps outline the Clinical Trial Application (CTA) process:
- Clinical Trial Application (CTA): Submit a detailed CTA to the ISP, which must include the study protocol, informed consent forms, and investigator brochures. The approval process is efficient, typically taking between 30 to 90 days.
- Ethics Committee Approval: Obtaining consent from a recognized ethics committee is essential for all clinical studies in Chile. This step is crucial for ensuring that the study meets ethical standards and protects participant welfare. With bioaccess®, sponsors of the ich-gcp clinical trial chile can benefit from expedited ethics approvals, often attained within 4-8 weeks, considerably decreasing time to study initiation.
- Compliance with ICH-GCP clinical trial Chile: All study activities must adhere to ICH-GCP standards, which are vital for maintaining data integrity and ensuring participant safety throughout the process. Ethical considerations are paramount, as they align research with local health needs and prioritize participant welfare.
- Monitoring and Reporting: Establish a comprehensive oversight plan to track study progress and ensure continuous adherence to compliance requirements during the research.
Organizations often struggle with delays due to incomplete documentation or misunderstandings of compliance requirements. These challenges can significantly hinder the progress of clinical studies, impacting timelines and outcomes. Keeping the lines of communication open with regulatory agencies and getting advice from local experts can help sponsors tackle these challenges head-on. Furthermore, utilizing bioaccess®’s expertise in early feasibility studies throughout Colombia, Brazil, and Mexico can further enhance the efficiency of study execution.
By carefully adhering to these steps and using the resources available through bioaccess®, sponsors can simplify the approval process, minimize possible delays, and improve the feasibility of their ich-gcp clinical trial chile. This structured approach not only leads to quicker approvals but also showcases the positive economic impact of medical research in the region, fostering job creation and healthcare advancements.
Implement Effective Patient Recruitment Strategies
To enhance patient recruitment for clinical trials in Chile, it’s essential to adopt strategic approaches that resonate with local communities:
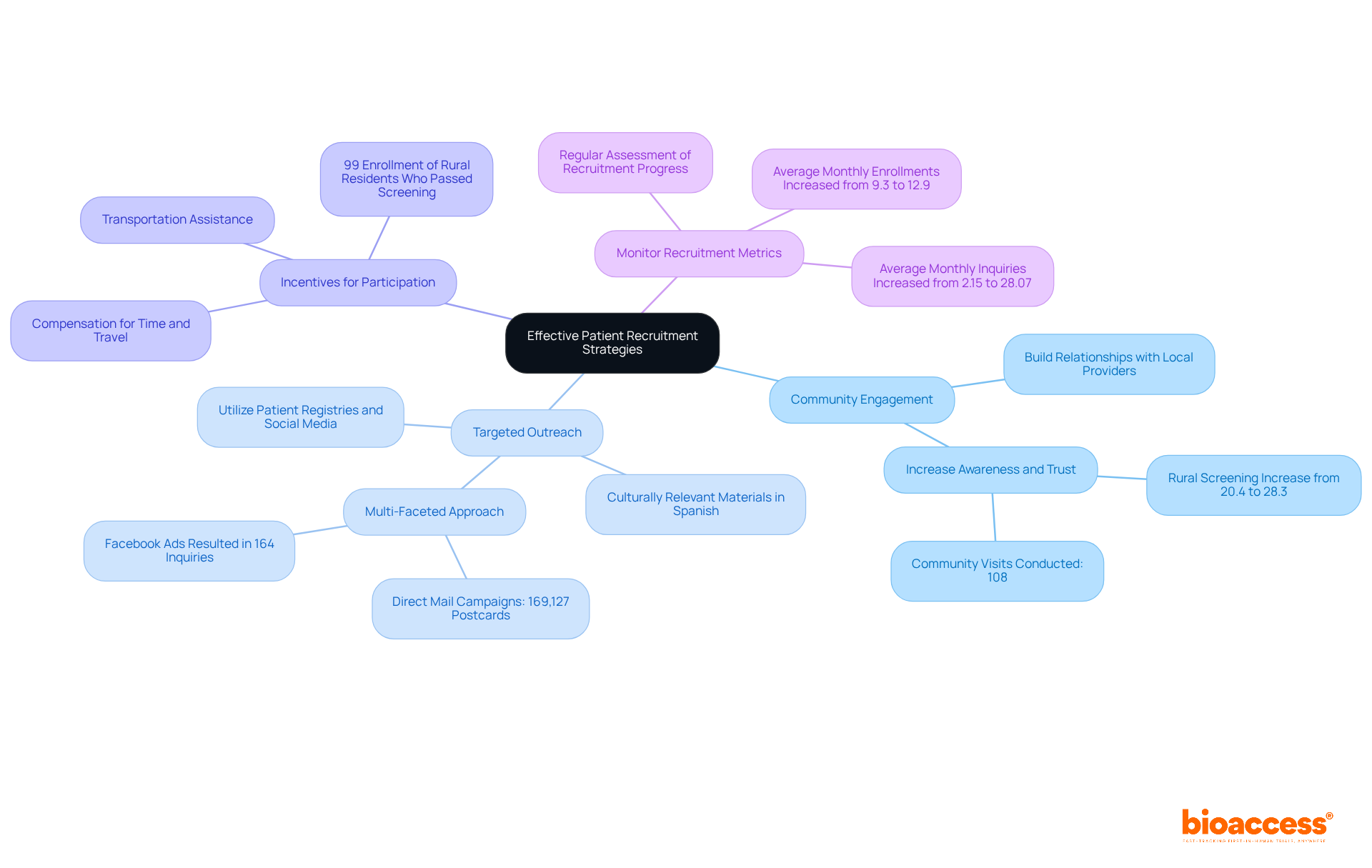
- Community Engagement: Establish strong relationships with local healthcare providers and community organizations. This method not only enhances awareness about the study but also fosters trust, which is essential for participation. For instance, community engagement strategies have shown to increase the proportion of rural residents screened for studies from 20.4% to 28.3%. As highlighted by our clients, effective community engagement has been crucial in building trust and participation in clinical studies.
- Targeted Outreach: Leverage patient registries and social media platforms to reach potential participants effectively. Ensure that outreach materials are culturally relevant and available in Spanish to resonate with the local population. A multi-faceted approach combining direct mail, social media ads, and local media outreach has proven effective, resulting in significant increases in inquiries and enrollment. Clients have shared that when outreach strategies are tailored, participant interest significantly increases.
- Incentives for Participation: Offer incentives such as transportation assistance or compensation for time and travel. This can help mitigate barriers, particularly for rural populations who may face travel challenges. For instance, collaborations with local clinics can lessen travel distances for participants, facilitating their involvement in the study. Notably, 99% of rural residents who passed screening enrolled in the study, showcasing the success of these partnerships. Our clients have emphasized the importance of addressing logistical challenges to facilitate participation.
- Monitor Recruitment Metrics: Regularly assess recruitment progress and adjust strategies as needed to address challenges or delays. Tracking metrics such as the average monthly inquiries, which increased from 2.15 to 28.07 after implementing community engagement strategies, can provide valuable insights into the effectiveness of your recruitment efforts. Our team at bioaccess® highlights the significance of data-driven decision-making to enhance recruitment strategies and ensure timely completion of studies.
Implementing these strategies not only enhances participation but also accelerates the overall research timeline. Involving local healthcare providers and employing customized outreach strategies are vital for navigating the intricacies of ich-gcp clinical trial chile, where a conducive compliance environment and diverse patient population offer considerable benefits. With bioaccess®’s expertise in expediting research studies and navigating regulatory hurdles, sponsors can confidently pursue their research objectives in Latin America.
Ensure Robust Data Management Practices
In the fast-evolving landscape of clinical trials, effective information management is not just beneficial; it’s essential for success. Consider these key practices to elevate your information management strategy:
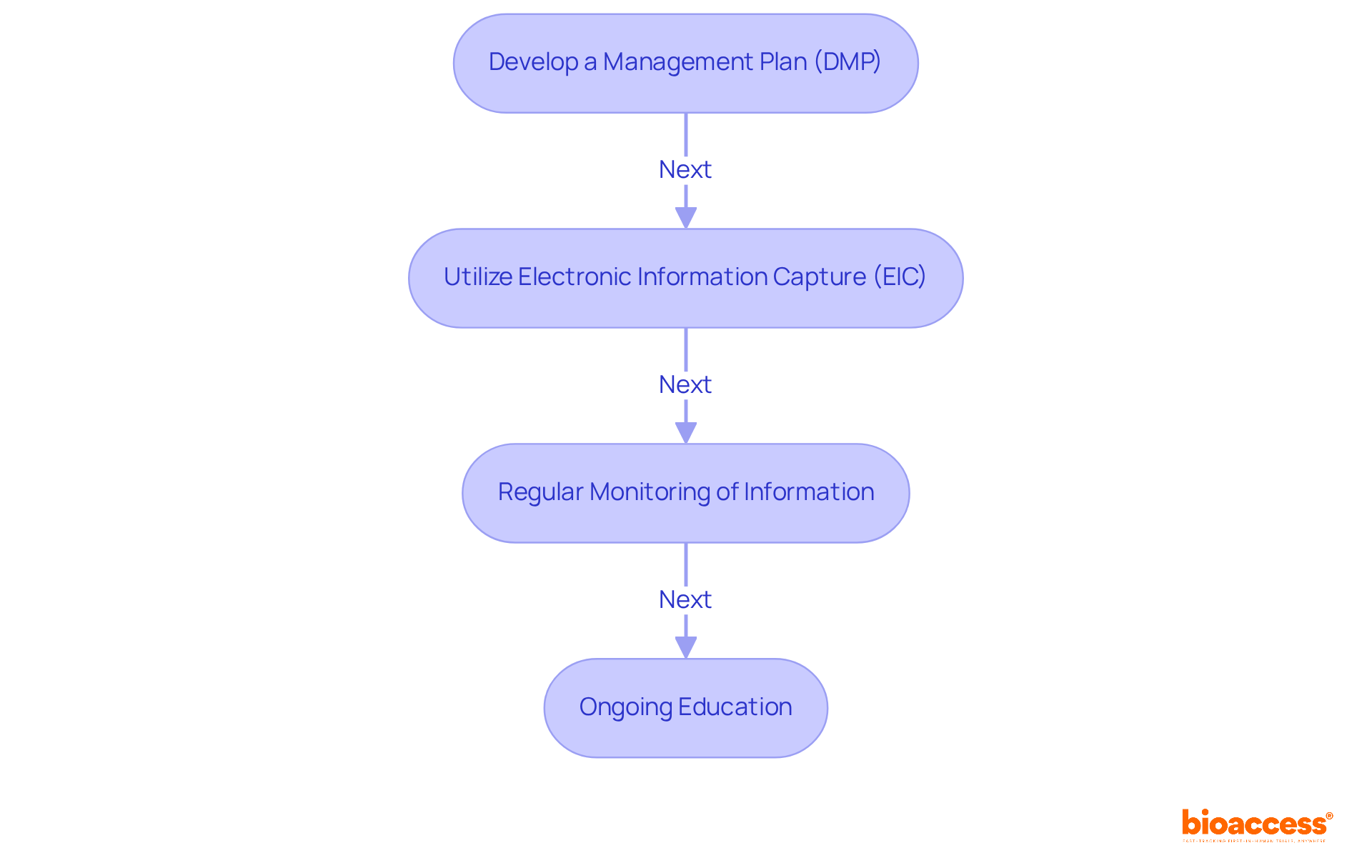
- Develop a Management Plan (DMP): A comprehensive DMP is vital, detailing collection methods, storage solutions, and analysis protocols. This ensures compliance with ICH-GCP standards and facilitates regulatory submissions to authorities like ANVISA and COFEPRIS.
- Utilize Electronic Information Capture (EIC): Implementing EIC systems streamlines collection processes, significantly reduces errors, and enhances integrity. This technology enables real-time information entry, which is essential for maintaining high-quality datasets.
- Regular Monitoring of Information: Conducting periodic audits and supervision is essential to identify discrepancies and ensure accuracy and completeness. This proactive strategy aids in reducing risks linked to integrity concerns, which have been noted in around 32% of early-stage studies, according to recent analyses.
- Ongoing Education: Ensuring that team members are well-versed in the latest regulations and best practices fosters a culture of quality and compliance. Continuous education on information management practices and compliance requirements is essential.
By embracing these practices, sponsors not only safeguard their studies but also pave the way for groundbreaking advancements in patient care. As Ramkumar Kali noted, ‘Clinical Data Management is the backbone of successful clinical trials, ensuring data integrity, regulatory compliance, and patient safety.
Conclusion
Navigating the complexities of ICH-GCP compliance is not just a regulatory requirement; it’s a strategic imperative for clinical trial success in Chile. By adhering to these guidelines, sponsors empower themselves to enhance research quality while effectively navigating regulatory complexities, ensuring timely approvals and positively impacting public health.
The article emphasized several key principles of ICH-GCP compliance, including the importance of:
- Ethical conduct
- Informed consent
- Strict protocol adherence
It also highlighted actionable steps for clinical trial sponsors, such as:
- Early engagement with regulatory authorities like ANVISA and COFEPRIS
- Developing comprehensive protocols
- Implementing robust data management systems
These practices are vital for overcoming common challenges and expediting the clinical trial process in Chile.
Ultimately, prioritizing ICH-GCP compliance empowers sponsors to lead the way in ethical research, driving innovation and better health outcomes across Latin America. By prioritizing these best practices, sponsors can enhance patient recruitment, streamline approval processes, and ultimately contribute to the growth of the clinical research landscape.
Frequently Asked Questions
What are the key principles of ICH-GCP for clinical trials in Latin America?
The key principles include ethical conduct prioritizing participant rights and well-being, informed consent with comprehensive information provided to participants, strict protocol compliance ensuring alignment with study objectives, and data integrity for accurate and reliable reporting.
How does ethical conduct influence clinical trials?
Ethical conduct ensures that the rights, safety, and well-being of participants are prioritized above scientific objectives, adhering to guidelines such as the Declaration of Helsinki.
What is the importance of informed consent in clinical trials?
Informed consent is crucial as it allows participants to receive detailed information about the study’s purpose, procedures, risks, and benefits, enabling them to make voluntary and informed decisions about their participation.
Why is protocol compliance essential in clinical trials?
Protocol compliance is essential to ensure that all trial activities align with the approved study’s objectives, design, and methodology, meeting compliance expectations from regulatory authorities like ANVISA and COFEPRIS.
What role does data integrity play in clinical trials?
Data integrity is vital for collecting and reporting accurate and reliable data, which ensures that study findings are credible and can withstand scrutiny from regulatory authorities, facilitating timely approvals.
What steps can clinical trial sponsors take to apply ICH-GCP principles effectively?
Sponsors should engage with regulatory authorities early, develop comprehensive protocols that meet ICH-GCP and local requirements, and implement robust data management systems to ensure compliance and data integrity.
How can engaging with regulatory authorities benefit clinical trial sponsors?
Early engagement with authorities like ANVISA and COFEPRIS helps sponsors understand specific requirements and timelines for approval, which can range from 30 to 90 days, facilitating smoother approval processes.
What advantages does Latin America offer for early-stage clinical trials?
Latin America provides strategic advantages such as streamlined approval processes that can lead to authorizations in as little as 30-90 days, cost efficiency, and effective patient recruitment, which are beneficial for startups seeking funding and strategic acquisitions.
How does compliance with ICH-GCP guidelines impact research quality?
Mastering ICH-GCP principles enhances compliance, significantly improves research quality, and helps organizations build a strong reputation quickly by being recognized for conducting compliant studies.
List of Sources
- Understand ICH-GCP Principles for Compliance
- Ensuring Quality Clinical Trials with ICH-GCP Compliance (https://statswork.com/insights/articles/statistical-programming-biostatistics/ich-gcp-compliance-clinical-trials)
- Discover the 11 Principles of ICH GCP E6(R3) | Medicover MICS (https://medicover-mics.com/the-11-principles-of-ich-good-clinical-practice)
- ICH-GCP Principles for Clinical Researchers | Anila Vs posted on the topic | LinkedIn (https://linkedin.com/posts/anila-vs-3420bb2a9_13-principles-of-ich-gcp-every-clinical-activity-7420399483886133248-gDZu)
- Navigate Chile’s Regulatory Landscape for Clinical Trials
- Navigate Clinical Trial Approval in Chile: 4 Essential Steps | bioaccess® (https://bioaccessla.com/blog/navigate-clinical-trial-approval-in-chile-4-essential-steps)
- Best Practices for Clinical Trial Outsourcing in Chile | bioaccess® (https://bioaccessla.com/blog/best-practices-for-clinical-trial-outsourcing-in-chile)
- How the Scientific Ethical Committees work in Chile (https://linkedin.com/pulse/how-scientific-ethical-committees-work-chile-bopalcro-sigce)
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- Implement Effective Patient Recruitment Strategies
- Master Early Phase Clinical Trials in Chile: Key Strategies and Insights | bioaccess® (https://bioaccessla.com/blog/master-early-phase-clinical-trials-in-chile-key-strategies-and-insights)
- Patient Engagement Quotes: For Every Purpose & Audience (https://nclusiv.co.uk/edi-consulting/f/patient-engagement-quotes-for-every-purpose-audience)
- Community engagement strategies improve recruitment and enrollment in a pragmatic clinical trial – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC12444702)
- Conducting First-in-Human Clinical Trials in Chile: A Step-by-Step Guide | bioaccess® (https://bioaccessla.com/blog/conducting-first-in-human-clinical-trials-in-chile-a-step-by-step-guide)
- Ensure Robust Data Management Practices
- Best Practices in Clinical Data Management for High-Quality Outcomes – Vita Global Sciences (https://vitaglobalsciences.com/blog/best-practices-in-clinical-data-management)
- Best Practices for Ensuring Data Quality in Clinical Trials (https://cytel.com/perspectives/best-practices-for-ensuring-data-quality-in-clinical-trials)
- 5 Best Practices for Clinical Data Management | ACL Digital (https://acldigital.com/blogs/5-best-practices-effective-clinical-data-management-enhancing-data-quality-compliance)
- Detecting Fraud in Clinical Trials Using Statistical Data Monitoring | Applied Clinical Trials Online (https://appliedclinicaltrialsonline.com/view/detecting-fraud-in-clinical-trials-using-statistical-data-monitoring)
- Data Management in Clinical Research: Best Practices | Datavant (https://datavant.com/blog/clinical-data-management)

Leave a Reply