Introduction
The landscape of clinical research for medical devices in Latin America is rapidly evolving, shaped by a blend of local regulations and international standards. Good Clinical Practice (GCP) stands as a cornerstone for ensuring ethical and scientific integrity in clinical studies, safeguarding participant rights while enhancing the credibility of research data. However, organizations navigating this complex regulatory terrain often face significant challenges that can impede their progress. What strategies can be employed to overcome these hurdles and ensure successful outcomes in medical device trials within this dynamic environment?
As we delve deeper into the Medtech landscape, it becomes clear that understanding these challenges is crucial. The role of organizations like Bioaccess is pivotal in addressing these key issues, providing insights and solutions that can streamline the research process. Collaboration and strategic planning are essential in this ever-changing field, as they can lead to more effective trials and better patient outcomes.
In conclusion, the importance of collaboration cannot be overstated. By working together and leveraging expertise, stakeholders can navigate the complexities of clinical research more effectively. The next steps involve not only recognizing these challenges but also actively seeking partnerships that foster innovation and success in medical device trials.
Define Good Clinical Practice for Medical Devices in Latin America
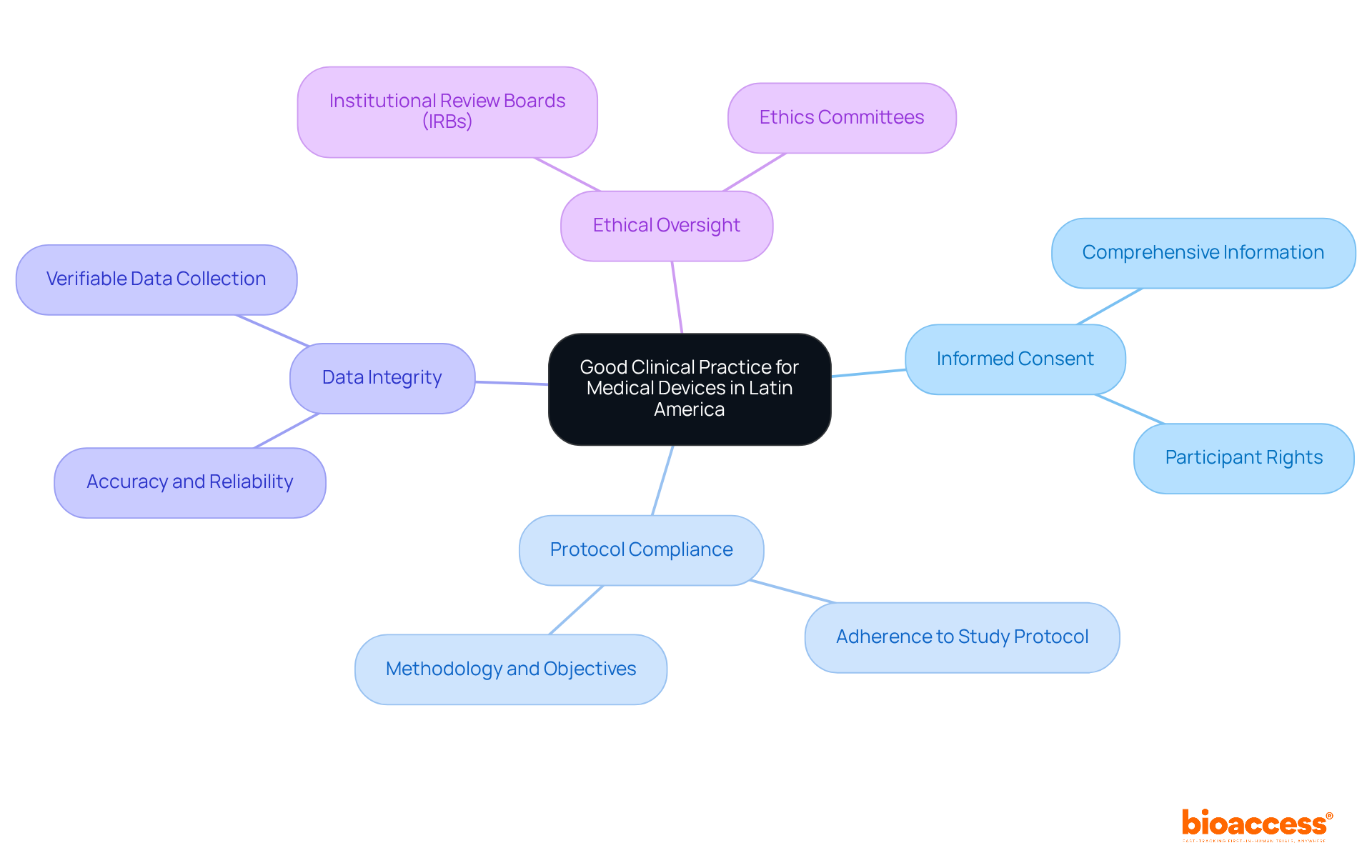
Medical device serves as a framework of internationally recognized ethical and scientific quality standards that govern the design, conduct, performance, monitoring, auditing, recording, analysis, and reporting of . These practices are essential for protecting the rights, safety, and well-being of participants while ensuring the credibility of the data collected. In Latin America, GCP is influenced by local regulations and international guidelines, particularly those from the International Conference on Harmonization (ICH). Key components of GCP include:
- : Participants must receive comprehensive information regarding the trial’s purpose, procedures, risks, and benefits before consenting to participate.
- Protocol Compliance: Trials must strictly adhere to the approved study protocol, which outlines the methodology and objectives.
- : All data collected must be accurate, reliable, and verifiable, ensuring that the results can be trusted.
- : Institutional Review Boards (IRBs) or Ethics Committees are responsible for reviewing and approving studies to ensure that are upheld.
Recent updates to reflect a commitment to enhancing the ethical framework of . For instance, approximately 85% of global sponsors rated data quality from Chilean studies as ‘excellent’ or ‘very good,’ underscoring the region’s dedication to high . Additionally, Colombia’s oversight agency, INVIMA, is recognized for its rigorous supervision, comparable to that of the FDA, which bolsters the credibility of research findings.
Understanding these principles is vital for organizations aiming to conduct studies in Latin America, as they form the foundation of compliance and ethical research practices. The region’s evolving oversight landscape, characterized by a push towards digitalization and streamlined procedures, further facilitates the implementation of GCP, making Latin America an increasingly attractive destination for research. bioaccess® stands at the forefront of supporting these research initiatives, offering such as feasibility assessments, study setup, project oversight, and reporting, ensuring innovation and compliance excellence in the Latin American Medtech landscape.
Navigate the Regulatory Landscape for Medical Device Trials
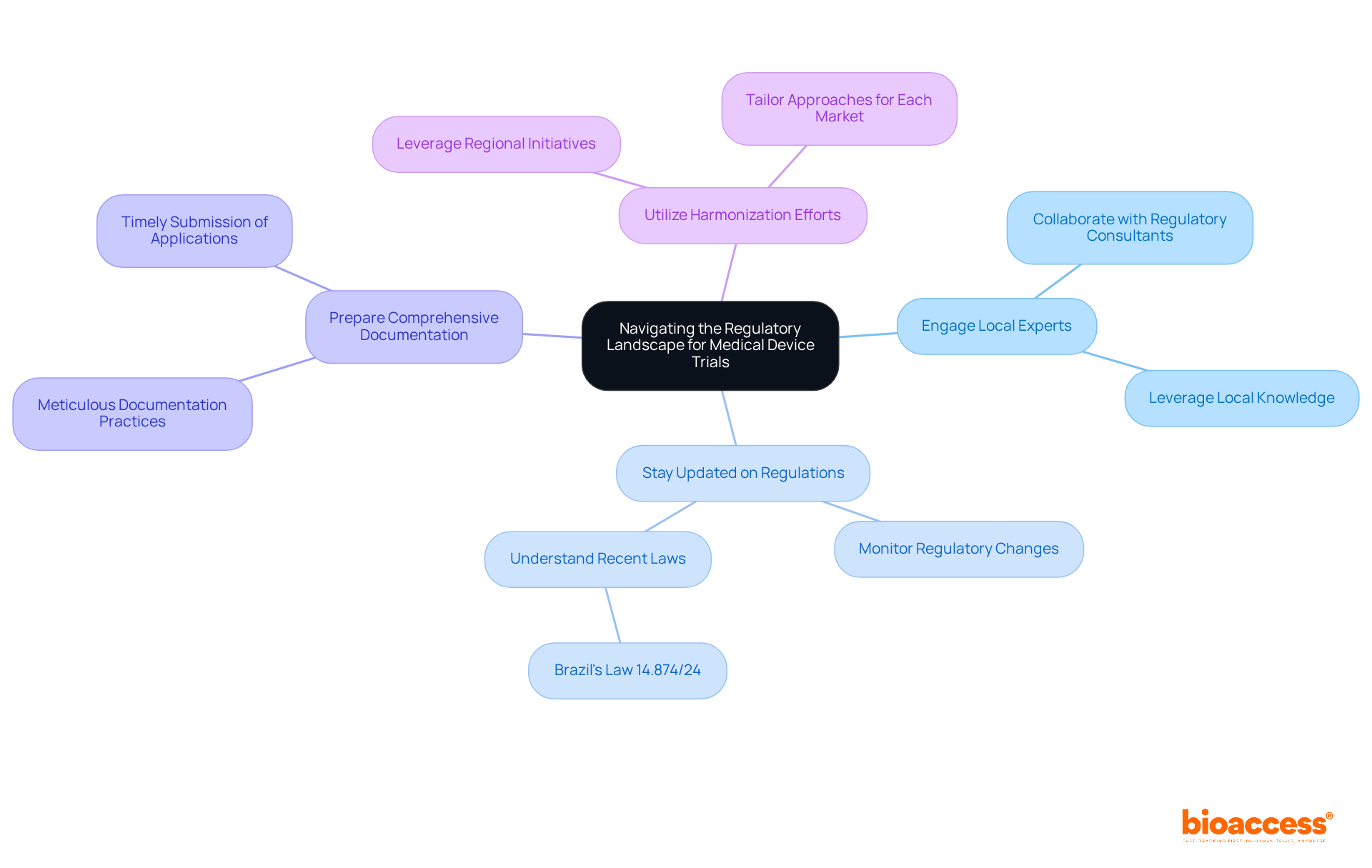
Navigating the compliance landscape for medical device is crucial for successful clinical research. Understanding the diverse rules that vary by country is essential. Each nation has its own – each with unique stipulations for . In Colombia, for instance, obtaining study approval involves securing consent from your site’s institutional review board (IRB) or ethics committee (EC), followed by approval from INVIMA and an import permit from the Ministry of Industry and Commerce (MinCIT) to ship investigational devices.
To effectively navigate this complex landscape, consider these key strategies:
- Engage Local Experts: Collaborating with local into specific requirements and expedite the approval process. Their expertise is crucial in addressing the complexities of compliance, which can vary significantly across jurisdictions.
- Stay Updated on Regulations: Regulatory frameworks are continually evolving. Staying informed about changes, such as , which simplifies approval processes, can prevent compliance issues and enhance operational efficiency.
- Prepare Comprehensive Documentation: Meticulously preparing all necessary documentation, including , is essential to avoid delays. In Colombia, the review process typically takes 90-120 days, making thorough documentation critical for timely approvals. Furthermore, studies performed under strict guidelines produce more trustworthy data for compliance submissions.
- Utilize Harmonization Efforts: Leverage regional harmonization initiatives aimed at aligning regulations across countries to simplify the approval process. While compliance requirements in Mercosur nations are largely harmonized, variations still exist, necessitating a tailored approach for each market.
By employing these strategies, organizations can significantly enhance their prospects of successfully maneuvering through the compliance environment and executing studies effectively. This ultimately accelerates their market entry in the sector. Bioaccess® stands ready to assist, offering accelerated backed by over 20 years of expertise. We help Medtech, Biopharma, and Radiopharma startups , including Early-Feasibility Studies, , Pilot Studies, Pivotal Studies, and Post-Market Clinical Follow-Up Studies.
Implement Strategies to Overcome Clinical Trial Challenges
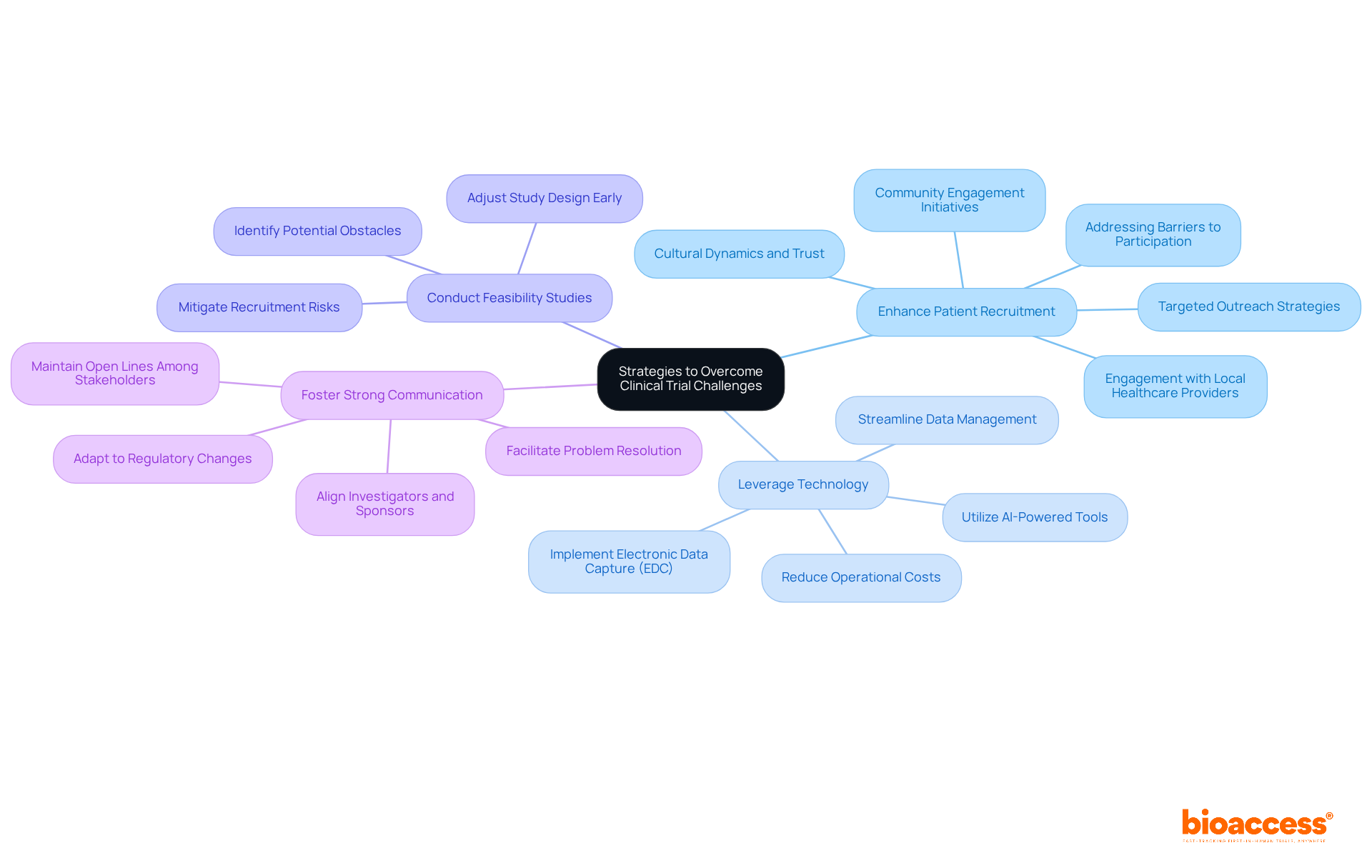
Clinical studies for medical devices face significant challenges, particularly in , , and data management. To effectively navigate these hurdles, organizations must adopt strategic approaches that enhance their chances of success.
- : Targeted outreach strategies are essential. Engaging with communities and collaborating with local healthcare providers can attract suitable participants. Notably, in Latin America, is remarkably efficient; studies from 2022 show that . This achievement stems from the region’s diverse population and cultural dynamics, which foster trust in physician-patient relationships.
- Leverage Technology: Implementing electronic data capture (EDC) systems is crucial for streamlining data collection and management. This technology minimizes errors and boosts efficiency, especially in a landscape where . Furthermore, AI-powered tools can effectively identify eligible patients and manage logistics, enhancing overall study performance.
- : Prior to initiating a test, conducting feasibility evaluations is vital. This proactive approach helps and allows for necessary adjustments to the study design. By addressing participant recruitment and risks early on, organizations can mitigate common causes of delays in studies, which is essential for ensuring .
- : Maintaining open lines of communication among all stakeholders-including investigators, sponsors, and oversight entities-is essential. This ensures alignment and facilitates swift problem resolution, particularly in a rapidly evolving regulatory landscape, such as in .
By proactively implementing these strategies, organizations can not only mitigate risks but also enhance the likelihood of successful outcomes, ultimately advancing the progress of .
Ensure Ethical Standards and Patient Safety in Trials
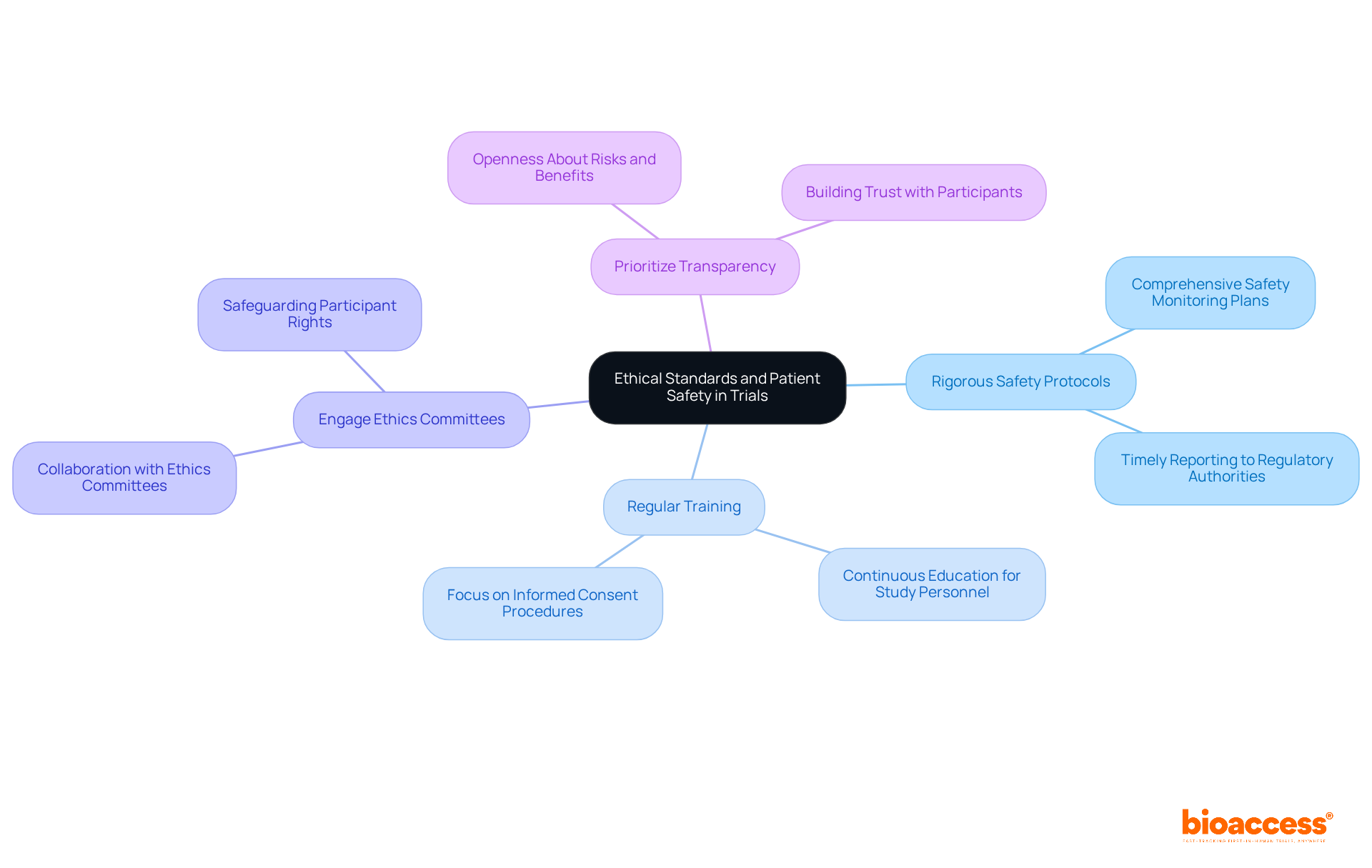
Maintaining is not just a responsibility; it’s a fundamental duty of all researchers. To uphold these standards effectively, consider the following key practices:
- Implement Rigorous Safety Protocols: Establish that track adverse events and ensure timely reporting to regulatory authorities. This proactive approach is essential for maintaining participant safety and adhering to moral guidelines.
- Conduct Regular Training: Offer continuous education for all study personnel on moral considerations, , and . Research indicates that a significant portion of study personnel in Latin America are educated on the , underscoring the importance of these ethical standards in daily operations.
- Engage : Collaborate closely with to ensure that every aspect of the study is ethically sound and that participant rights are safeguarded. This partnership is crucial for navigating the complexities of medical research in the region.
- : Maintain openness with participants regarding the risks and benefits of the study. This fosters trust and encourages informed decision-making, particularly in an area where past moral failures have shaped public perception of clinical studies.
By prioritizing ethical standards and patient safety, organizations can significantly enhance the credibility of their trials. This commitment not only advances medical knowledge but also benefits both participants and the broader healthcare community.
Conclusion
Good Clinical Practice (GCP) for medical devices in Latin America is a vital framework that upholds the ethical and scientific integrity of clinical studies. By adhering to these internationally recognized standards, researchers not only safeguard the rights and safety of participants but also bolster the credibility of the data collected. The dynamic landscape of GCP in the region, shaped by local regulations and international guidelines, highlights the necessity of compliance and ethical research practices for successful medical device trials.
This article underscores several key components of GCP, such as:
- Informed consent
- Protocol compliance
- Data integrity
- Ethical oversight
It stresses the importance of grasping the diverse regulatory environments across Latin American countries, including Brazil, Mexico, and Colombia. By collaborating with local experts, staying informed about regulations, preparing thorough documentation, and leveraging harmonization efforts, organizations can adeptly navigate the complexities of compliance. Furthermore, implementing strategic approaches to address challenges in patient recruitment, data management, and ethical standards is crucial for advancing clinical research in the region.
Ultimately, a steadfast commitment to high ethical standards and patient safety not only enhances the credibility of clinical trials but also cultivates trust within the healthcare community. As the landscape for medical device GCP continues to evolve, organizations are urged to adopt these best practices and strategies, paving the way for innovative research and improved patient outcomes in Latin America. By prioritizing compliance and ethical considerations, stakeholders can significantly contribute to the advancement of medical knowledge and the overall enhancement of healthcare in the region.
Frequently Asked Questions
What is Good Clinical Practice (GCP) for medical devices in Latin America?
Good Clinical Practice for medical devices in Latin America is a framework of internationally recognized ethical and scientific quality standards governing the design, conduct, performance, monitoring, auditing, recording, analysis, and reporting of clinical studies.
Why are GCP practices important?
GCP practices are essential for protecting the rights, safety, and well-being of participants while ensuring the credibility of the data collected during clinical studies.
What influences GCP in Latin America?
GCP in Latin America is influenced by local regulations and international guidelines, particularly those from the International Conference on Harmonization (ICH).
What are the key components of GCP?
The key components of GCP include Informed Consent, Protocol Compliance, Data Integrity, and Ethical Oversight.
What does Informed Consent entail?
Informed Consent requires that participants receive comprehensive information about the trial’s purpose, procedures, risks, and benefits before agreeing to participate.
What is Protocol Compliance?
Protocol Compliance refers to the necessity for trials to strictly adhere to the approved study protocol, which outlines the methodology and objectives of the study.
How is Data Integrity maintained in GCP?
Data Integrity is maintained by ensuring that all collected data is accurate, reliable, and verifiable, which allows the results to be trusted.
What role do Institutional Review Boards (IRBs) or Ethics Committees play in GCP?
IRBs or Ethics Committees are responsible for reviewing and approving studies to ensure that ethical standards are upheld.
How have recent updates to GCP regulations in Latin America improved clinical studies?
Recent updates reflect a commitment to enhancing the ethical framework of clinical studies, with a significant percentage of global sponsors rating data quality from Chilean studies as ‘excellent’ or ‘very good.’
What is INVIMA and its significance in Colombia?
INVIMA is Colombia’s oversight agency, recognized for its rigorous supervision, comparable to that of the FDA, which enhances the credibility of research findings.
Why is understanding GCP principles important for organizations conducting studies in Latin America?
Understanding GCP principles is vital for organizations as they form the foundation of compliance and ethical research practices in the region.
How is the oversight landscape in Latin America evolving?
The oversight landscape is evolving with a push towards digitalization and streamlined procedures, facilitating the implementation of GCP and making Latin America an attractive destination for research.
What services does bioaccess® provide to support research initiatives in Latin America?
bioaccess® offers comprehensive services such as feasibility assessments, study setup, project oversight, and reporting, ensuring innovation and compliance excellence in the Latin American Medtech landscape.
List of Sources
- Define Good Clinical Practice for Medical Devices in Latin America
- meddeviceonline.com (https://meddeviceonline.com/doc/how-chile-is-shaping-medical-device-clinical-trials-in-latin-america-0001)
- Latin America’s Landscape For Medtech Clinical Trials (https://clinicalleader.com/doc/latin-america-s-landscape-for-medtech-clinical-trials-0001)
- bioaccessla.com (https://bioaccessla.com/blog/understanding-regulatory-requirements-for-latin-america-trials-a-comprehensive-overview-of-regulatory-requirements-for-latin-america-trials)
- Latin America: A Compelling Region To Conduct Your Clinical Trials (https://clinicalleader.com/doc/latin-america-a-compelling-region-to-conduct-your-clinical-trials-0001)
- Checking your browser – reCAPTCHA (https://pmc.ncbi.nlm.nih.gov/articles/PMC9468347)
- Navigate the Regulatory Landscape for Medical Device Trials
- Latin American countries’ regulatory approval processes | MDRC (https://mdrc-consulting.com/south-america-regulatory-processes-en)
- bioaccessla.com (https://bioaccessla.com/blog/understanding-regulatory-requirements-for-latin-america-trials-a-comprehensive-overview-of-regulatory-requirements-for-latin-america-trials)
- bioaccessla.com (https://bioaccessla.com/blog/latin-america-medical-device-regulations-compared-key-insights-and-challenges)
- Should You Conduct Your Medical Device Clinical Trial In Latin America? – Journal for Clinical Studies (https://journalforclinicalstudies.com/conduct-medical-device-clinical-trial-latin-america)
- Implement Strategies to Overcome Clinical Trial Challenges
- bioaccess® | MedTech, Biopharma & Radiopharma CRO in Latin America (https://bioaccessla.com/blog/the-latin-american-advantage-why-clinical-trial-recruitment-and-retention-outpace-the-us-and-europe)
- 7 quotes from 2023 to guide you on the medtech market (https://tiinatyni.com/blogi/7-quotes-from-2023-to-guide-you-on-the-medtech-market)
- Clinical Trials Patient Recruitment in Latin America | H Clinical (https://hclinical.com/patient-recruitment)
- Clinical studies: the challenge of patient recruitment (https://servier.com/en/newsroom/clinical-studies-patient-recruitment)
- 30 Quotes About the Future of Healthcare: Expert Takes (https://deliberatedirections.com/quotes-future-of-healthcare)
- Ensure Ethical Standards and Patient Safety in Trials
- Ethical Violations in Clinical Research: Lessons from the Past, Challenges for the Future – ACRP (https://acrpnet.org/2025/06/16/ethical-violations-in-clinical-research-lessons-from-the-past-challenges-for-the-future)
- Prevalence of adverse events in the hospitals of five Latin American countries: results of the ‘Iberoamerican Study of Adverse Events’ (IBEAS). | PSNet (https://psnet.ahrq.gov/issue/prevalence-adverse-events-hospitals-five-latin-american-countries-results-iberoamerican-study)
- Exploring Ethics in Clinical Research | Harvard Medical School Professional, Corporate, and Continuing Education (https://learn.hms.harvard.edu/insights/all-insights/exploring-ethics-clinical-research)
- Latin America: A Compelling Region To Conduct Your Clinical Trials (https://clinicalleader.com/doc/latin-america-a-compelling-region-to-conduct-your-clinical-trials-0001)
- The importance of ethical considerations in research and clinical trials | Penn LPS Online (https://lpsonline.sas.upenn.edu/features/importance-ethical-considerations-research-and-clinical-trials)

Leave a Reply