Introduction
The intricate regulatory landscape for medical device studies in Chile poses significant challenges for researchers aiming for success. Navigating these challenges requires not just knowledge but strategic planning. This guide will walk you through the essential steps you need to take for successful clinical research, from understanding local regulations to implementing effective patient recruitment strategies.
With compliance complexities and the pressing need for timely approvals, how can researchers ensure their studies meet regulatory demands while also achieving impactful outcomes in a competitive market? Delays can hinder progress and impact the success of clinical trials in a competitive landscape.
Understand the Regulatory Framework for Medical Device Studies in Chile
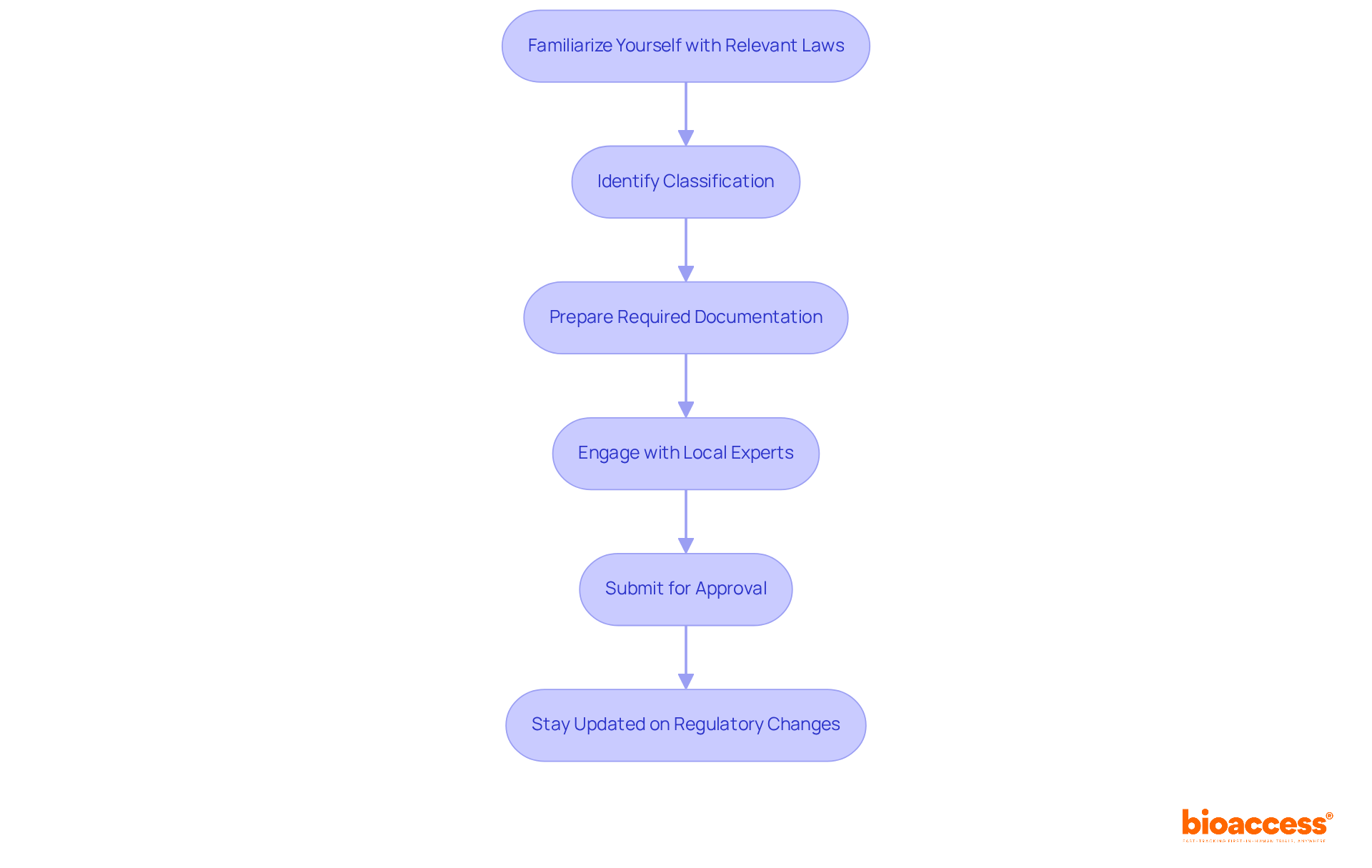
Navigating the compliance framework for a medical device study in Chile can be daunting, yet it is crucial for success in clinical research. To effectively carry out the medical device study in Chile, understanding the regulatory landscape is essential. The Instituto de Salud Pública (ISP) serves as the main regulatory body, overseeing the approval and monitoring of medical instruments. Let’s explore the key steps to successfully navigate this framework:
- Familiarize Yourself with Relevant Laws: Review the Medical Devices Law and associated regulations that dictate the requirements for clinical trials in Chile.
- Identify Classification: Determine the category of your medical apparatus (Class I, II, or III). This classification affects the regulatory pathway and approval timelines. Class I devices typically require less documentation and have faster approval times, while Class III devices may take longer due to more stringent requirements.
- Prepare Required Documentation: Gather necessary documents, including clinical trial protocols, informed consent forms, and evidence of compliance with Good Clinical Practice (GCP) standards. All documentation must be translated into Spanish for submission to the ISP and relevant ethics committees.
- Engage with Local Experts: Collaborating with local regulatory consultants or specialized CROs like bioaccess® can streamline the submission process and ensure adherence to local regulations, enhancing the likelihood of a successful outcome.
- Submit for Approval: Once your documentation is complete, submit it to the ISP for review. Approval timelines can vary, but expect approximately 30 days for Class I devices and 2-3 months for Class II and III devices.
- Stay Updated on Regulatory Changes: Regularly check for updates from the ISP, as regulations can evolve, impacting your project’s compliance requirements. Proactive communication with oversight agencies can further streamline the evaluation process.
Understanding these steps not only enhances your project’s success but also strategically positions you within the evolving landscape of the medical device study Chile in Latin America.
Conduct Early Feasibility Studies to Validate Your Medical Device
Navigating the complexities of early feasibility assessments (EFA) is crucial for the success of medical devices in today’s competitive landscape. Here’s how to effectively conduct EFS in Chile, leveraging the advantages of bioaccess®’s expertise in navigating the regulatory landscape:
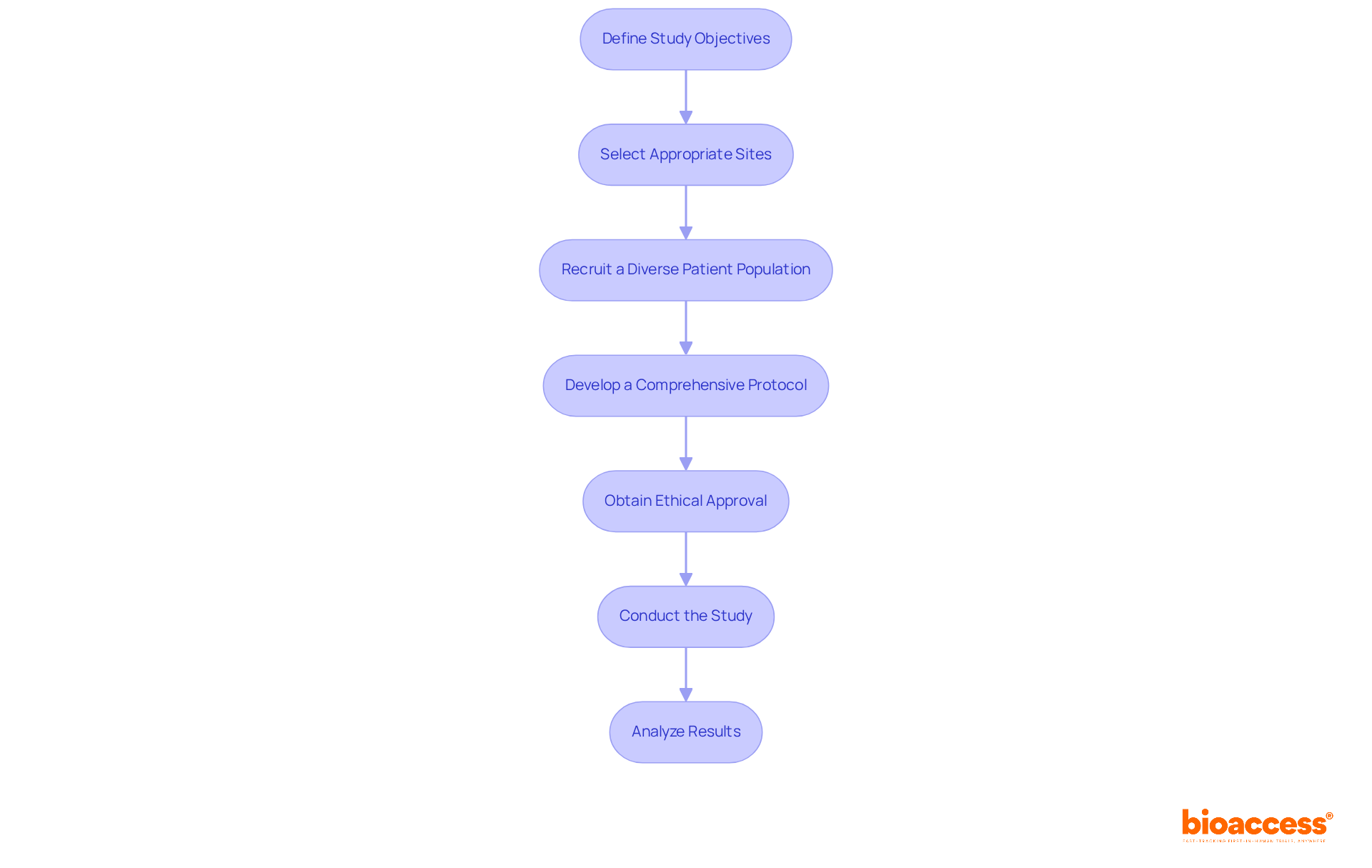
- Define Study Objectives: Clearly outline the objectives of your EFS, focusing on key questions such as safety, usability, and preliminary efficacy. This clarity helps shape your research design and ensures your regulatory submissions are on point.
- Select Appropriate Sites: Choose clinical locations knowledgeable in early feasibility assessments. bioaccess® can assist in identifying pre-qualified sites that align with your research needs, ensuring compliance with local regulations and ICH-GCP standards.
- Recruit a Diverse Patient Population: Ensure your trial includes a representative sample of the target population to gather relevant data. How can you enhance recruitment efforts? Leverage community outreach and partnerships with local healthcare providers to address the common challenge of patient enrollment.
- Develop a Comprehensive Protocol: Create a detailed research protocol that includes methodology, endpoints, and statistical analysis plans. Ensure adherence to ICH-GCP guidelines and local requirements established by INVIMA, which oversees clinical trials in Colombia, as well as similar organizations involved in the medical device study in Chile.
- Obtain Ethical Approval: Submit your research protocol to an ethics committee for review and approval. This step is essential for ensuring participant safety and ethical compliance, which is a critical aspect of the regulatory process. In Latin America, this process can take as little as 4-8 weeks, significantly faster than in the US or EU.
- Conduct the Study: Execute the EFS while closely monitoring data collection and participant feedback. Real-time data will guide necessary adjustments to the apparatus or study design, enhancing the study’s relevance and effectiveness. This method can result in $25K/patient savings with pre-negotiated site contracts and 30% lower overall study costs.
- Analyze Results: After completing the EFS, examine the data to determine if the instrument meets the predefined objectives. Use this information to refine your clinical trial strategy, ensuring that you are well-prepared for subsequent pivotal studies.
Without a robust EFS strategy, your device may struggle to gain traction in the market, ultimately impacting patient outcomes and your bottom line.
Navigate the Medical Device Registration Process in Chile
Navigating the medical device study Chile registration process can be complex, but understanding the key steps can streamline your path to market entry. Successfully registering your medical device involves several critical actions:
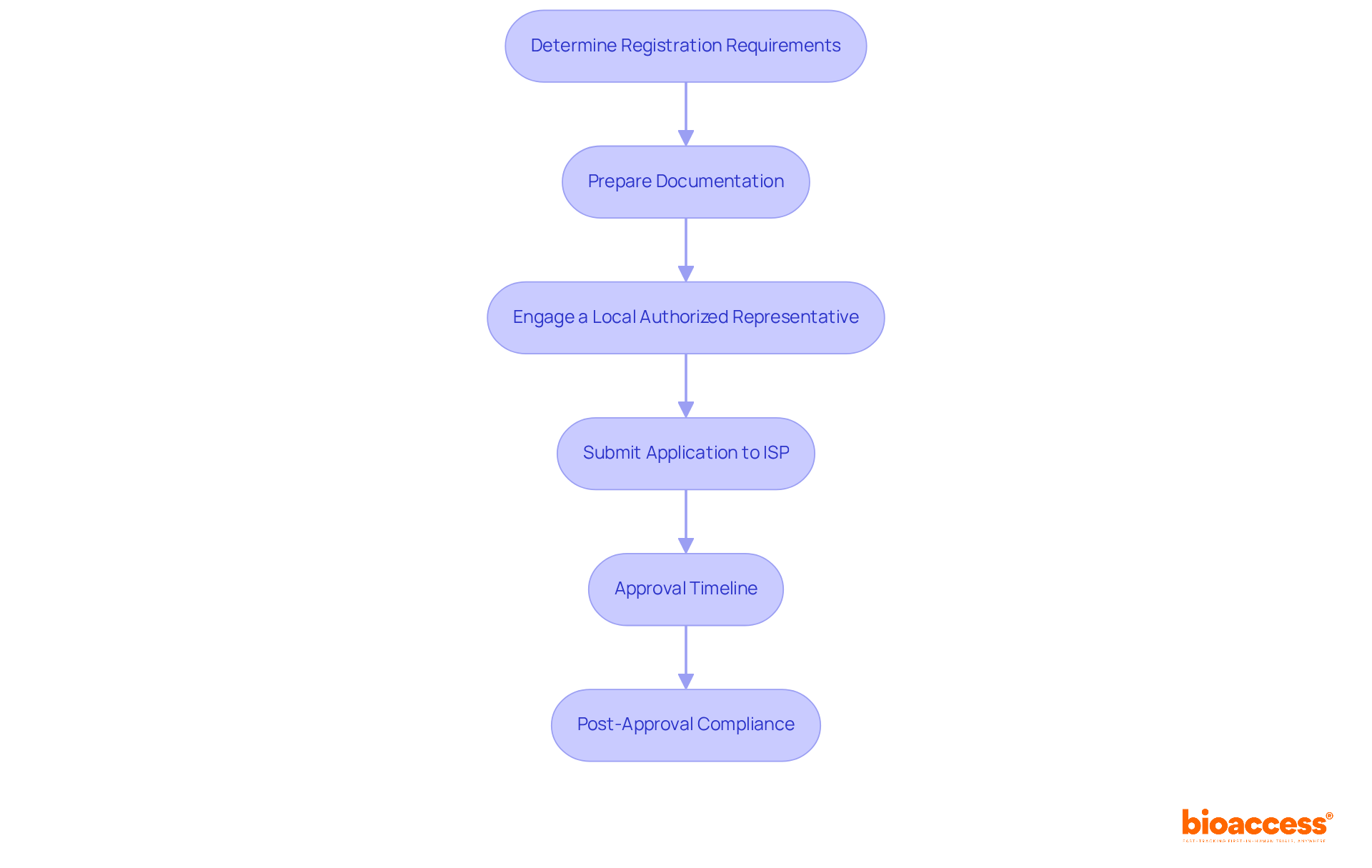
- Determine Registration Requirements: Identify the specific criteria for your classification of the apparatus. Class I products may have fewer requirements compared to Class II and III items, which necessitate more extensive documentation.
- Prepare Documentation: Gather essential documents to ensure a smooth registration process, such as:
- Technical files
- Quality Management System (QMS) certificates
- Clinical data supporting safety and efficacy
- Labels and instructions for use
- Engage a Local Authorized Representative: If you are not based in Chile, appoint a local representative to facilitate the registration process and communicate with the ISP.
- Submit Application to ISP: Ensure your application is thorough and complete to avoid delays in the registration process with the ISP. Submit your registration application along with the required documentation.
- Approval Timeline: Expect a review period of 2-3 months for Class II devices and longer for Class III devices. Monitor the status of your application and be prepared to respond to any queries from the ISP. Furthermore, utilizing bioaccess®’s rapid patient recruitment capabilities and access to over 50 pre-qualified clinical study sites throughout Latin America can significantly accelerate your medical device study Chile timelines.
- Post-Approval Compliance: After receiving approval, it is crucial to maintain compliance with local regulations, including rigorous post-market surveillance and timely reporting of any adverse events. Familiarize yourself with the requirements set forth by regulatory authorities such as ANVISA, INVIMA, and COFEPRIS to ensure a smooth market entry and sustained compliance.
Implement Effective Patient Recruitment Strategies
The success of clinical research hinges on recruiting the right patients, yet many studies face significant challenges in this area. Here are effective strategies to enhance patient recruitment in Chile:
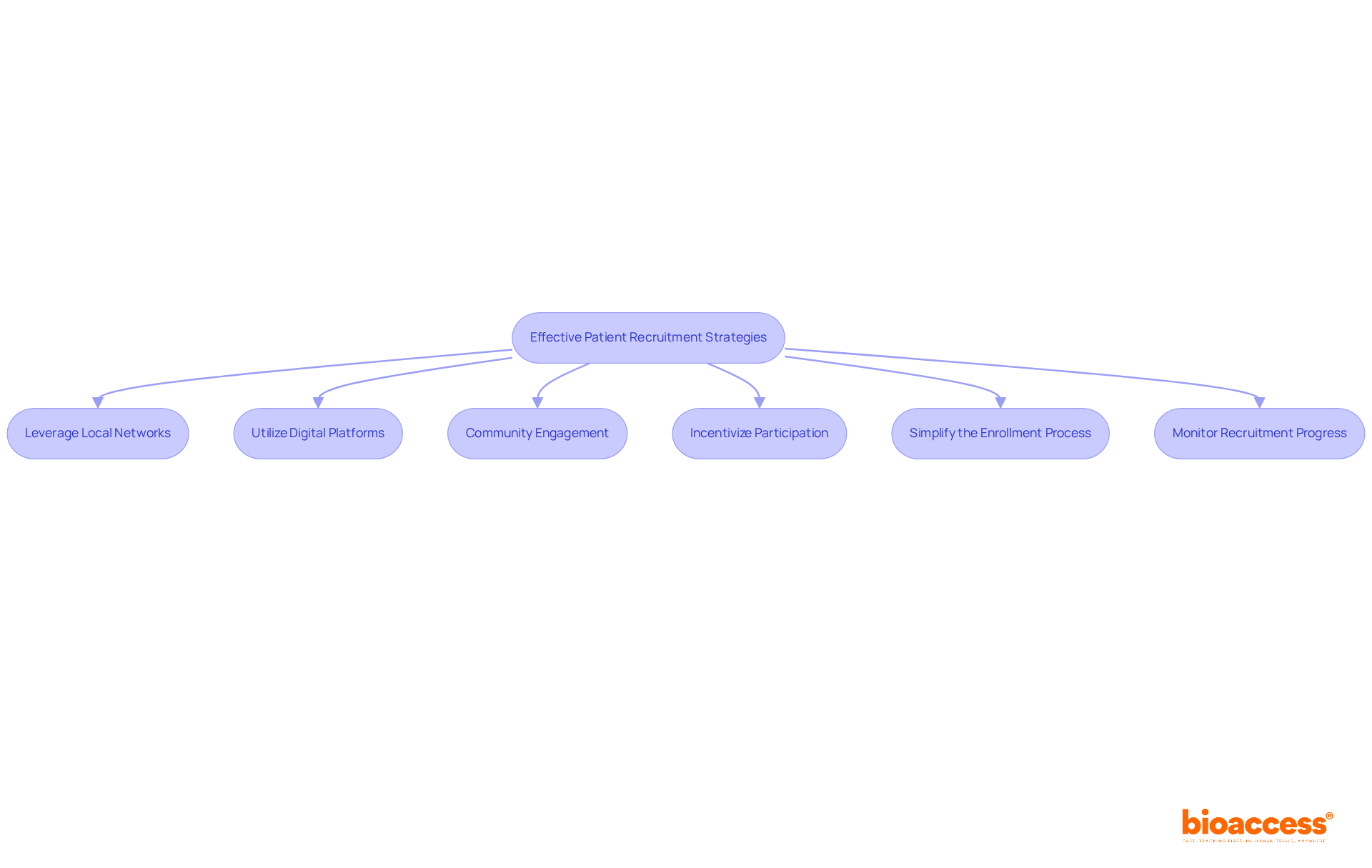
- Leverage Local Networks: Collaborate with local healthcare providers and institutions to identify potential participants. Building relationships with physicians can facilitate referrals, as they are trusted sources for patient recommendations.
- Utilize Digital Platforms: Implement online recruitment strategies, including social media campaigns and dedicated trial websites, to reach a broader audience. Highlight the benefits of participation, such as access to cutting-edge treatments and contributing to medical advancements, to attract interest.
- Community Engagement: Organize informational sessions in community centers to educate potential participants about the project and its significance. Address any concerns they may have regarding participation, emphasizing the safety measures and ethical standards in place.
- Incentivize Participation: Consider offering incentives for participation, such as travel reimbursements or health screenings, to encourage enrollment. This approach can significantly enhance participation rates, especially among underrepresented populations.
- Simplify the Enrollment Process: Streamline the enrollment process to reduce barriers for potential participants. Ensure that the consent process is clear and accessible, utilizing plain language to explain the study’s purpose and procedures.
- Monitor Recruitment Progress: Regularly assess recruitment metrics to identify challenges and adjust strategies as needed. Utilizing data analytics can optimize outreach efforts and improve enrollment rates, ensuring that recruitment goals are met efficiently.
Many studies struggle to meet their recruitment goals, leading to delays and increased costs. By concentrating on localized recruitment approaches, as highlighted by the VP of Clinical Research Optimization and Site Engagement, the medical device study Chile can improve patient recruitment, ultimately resulting in more successful outcomes and quicker approvals. Ensuring compliance with local regulations not only streamlines recruitment but also enhances the credibility of the study, paving the way for successful outcomes.
Ensure Rigorous Clinical Monitoring and Data Management
To ensure the success of your clinical trial in Chile, it’s essential to implement robust monitoring and data management practices, leveraging the strategic advantages of conducting First-in-Human (FIH) trials in Latin America:
- Establish a Monitoring Plan: Develop a comprehensive monitoring plan that outlines the frequency and methods of monitoring activities. Conduct site visits at least once a month. This ensures active surveillance and compliance with regulatory standards.
- Train Clinical Staff: Ensure that all clinical staff are thoroughly trained in Good Clinical Practice (GCP) and the specific protocols of the study. This training is crucial for maintaining compliance and data integrity, as well as safeguarding participant safety throughout the trial.
- Utilize Data Management Systems: Implement electronic data capture (EDC) systems to streamline data collection and management. These systems must adhere to compliance requirements for data security and integrity, facilitating accurate and efficient data handling.
- Conduct Regular Audits: Schedule regular audits of clinical sites to ensure adherence to protocols and compliance standards. In the context of the medical device study in Chile, maintaining an average audit frequency of once per month is recommended. This allows you to promptly address any discrepancies and uphold compliance. Documentation after each site visit, including findings, noncompliance issues, and corrective actions, is essential for maintaining compliance and data integrity.
- Engage with Regulatory Authorities: How can you ensure effective communication with regulatory authorities? Maintain open communication with the Instituto de Salud Pública (ISP), ANVISA, INVIMA, and COFEPRIS throughout the study. This engagement ensures that all monitoring and data management practices align with their expectations and regulatory frameworks, particularly in navigating the requirements set forth by these authorities for rapid market access.
- Conduct ongoing data analysis to identify trends and issues early in the study. This proactive approach allows you to make timely changes to the protocol when necessary, improving the overall quality and dependability of the outcomes. Incorporating a risk-based monitoring approach can further improve efficiency and focus on high-risk areas, ensuring compliance with regulatory requirements. By leveraging the fast ethics approvals and early feasibility studies available in Latin America, you can significantly accelerate your clinical trial timelines.
By prioritizing these practices, you not only enhance compliance but also pave the way for successful trial outcomes and market entry.
Conclusion
Navigating the complexities of conducting medical device studies in Chile is a challenge that can determine the success of clinical outcomes. Understanding the regulatory framework and engaging with local experts are crucial. By implementing effective patient recruitment and monitoring strategies, researchers can navigate the complexities of the Chilean landscape more efficiently. This structured approach helps ensure that medical devices not only meet regulatory requirements but also align with market needs, ultimately enhancing patient safety and outcomes.
Key steps discussed in this guide include:
- Familiarizing oneself with the local laws
- Classifying devices appropriately
- Preparing the necessary documentation for submission to the Instituto de Salud Pública (ISP)
The importance of early feasibility studies cannot be overstated; they validate the device’s efficacy and safety while also optimizing recruitment strategies to engage the right patient population. Furthermore, maintaining rigorous clinical monitoring and data management practices is crucial in ensuring compliance with ICH-GCP standards and fostering a transparent relationship with regulatory authorities.
Have you considered how leveraging the strategic advantages of conducting early-stage clinical trials in Latin America, like faster approval timelines and cost efficiencies, could enhance your medical device studies? By prioritizing thorough preparation and proactive engagement with local regulatory bodies, researchers can not only streamline their processes but also contribute to the advancement of medical technology in the region. By embracing these best practices, researchers not only enhance their chances of success but also play a crucial role in advancing medical technology that benefits patients.
Frequently Asked Questions
What is the main regulatory body for medical device studies in Chile?
The main regulatory body overseeing medical device studies in Chile is the Instituto de Salud Pública (ISP).
What are the key steps to navigate the regulatory framework for medical device studies in Chile?
The key steps include familiarizing yourself with relevant laws, identifying the classification of your medical device, preparing required documentation, engaging with local experts, submitting for approval, and staying updated on regulatory changes.
How do I determine the classification of my medical device in Chile?
You must classify your medical device as Class I, II, or III, which will affect the regulatory pathway and approval timelines. Class I devices generally require less documentation and have faster approval times, while Class III devices face more stringent requirements and longer timelines.
What documentation is required for submitting a medical device study in Chile?
Required documentation includes clinical trial protocols, informed consent forms, and evidence of compliance with Good Clinical Practice (GCP) standards, all of which must be translated into Spanish for submission.
How can local experts assist in the medical device study process in Chile?
Local regulatory consultants or specialized Contract Research Organizations (CROs) like bioaccess® can help streamline the submission process and ensure adherence to local regulations, improving the chances of a successful outcome.
What are the expected approval timelines for medical device studies in Chile?
Approval timelines vary based on the device classification: approximately 30 days for Class I devices and 2-3 months for Class II and III devices.
Why are early feasibility studies (EFS) important for medical devices?
Early feasibility studies are crucial for validating safety, usability, and preliminary efficacy of medical devices, which can significantly impact market traction and patient outcomes.
What should I focus on when defining the objectives of an early feasibility study?
Clearly outline the objectives focusing on key questions such as safety, usability, and preliminary efficacy to shape your research design and regulatory submissions effectively.
How can I ensure effective patient recruitment for my study?
Recruit a diverse patient population by leveraging community outreach and forming partnerships with local healthcare providers to address recruitment challenges.
What is involved in obtaining ethical approval for a medical device study in Chile?
You must submit your research protocol to an ethics committee for review and approval, which is essential for ensuring participant safety and ethical compliance. This process can take 4-8 weeks in Latin America.
What is the significance of analyzing results after conducting an early feasibility study?
Analyzing results helps determine if the medical device meets predefined objectives, allowing you to refine your clinical trial strategy and prepare for subsequent pivotal studies.
List of Sources
- Understand the Regulatory Framework for Medical Device Studies in Chile
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- Chile (ISP – Instituto de Salud Pública) – PharmaRegulatory.in – India’s Regulatory Knowledge Hub (https://pharmaregulatory.in/country-specific-regulatory-affairs/chile-isp-instituto-de-salud-publica)
- Conduct Early Feasibility Studies to Validate Your Medical Device
- Early feasibility studies on devices: “doing it sooner” to avoid trial failure | Meditrial (https://meditrial.net/2022/09/early-feasibility-studies-on-devices-doing-it-sooner-to-avoid-trial-failure)
- Early Feasibility Studies in Latin America (https://greenlight.guru/blog/early-feasibility-studies-in-latin-america)
- Early Feasibility Studies: Top 6 Considerations | MED Institute (https://medinstitute.com/blog/early-feasibility-studies-top-6-considerations)
- Stakeholder Perspectives on Early Feasibility Studies for Digital Health Technologies in the European Union: Qualitative Interview Study (https://jmir.org/2025/1/e77982)
- Implement Effective Patient Recruitment Strategies
- 10 Inspiring Patient Experience Quotes | Relias (https://relias.com/blog/patient-experience-quotes)
- Patient Recruitment Strategies for Clinical Trial Success (https://totaldiversity.com/clinical-study-recruitment)
- Clinical Trials Patient Recruitment in Latin America | H Clinical (https://hclinical.com/patient-recruitment)
- Patient Recruitment for Clinical Trials: Strategies That Actually Work (https://kapsuletech.com/blog/patient-recruitment-clinical-trials)
- Patient Engagement Quotes: For Every Purpose & Audience (https://nclusiv.co.uk/edi-consulting/f/patient-engagement-quotes-for-every-purpose-audience)
- Ensure Rigorous Clinical Monitoring and Data Management
- Guide to Clinical Research Monitoring | CCRPS (https://ccrps.org/clinical-research-blog/clinical-research-monitoring-a-guide-to-clinical-monitoring)
- The Importance of Statistical Monitoring in ICH E6 (R2) Addendum MakroCare (https://makrocare.com/blog/the-importance-of-statistical-monitoring-in-ich-e6-r2-addendum)
- 19 Inspirational Quotes About Data | The Pipeline | ZoomInfo (https://pipeline.zoominfo.com/operations/19-inspirational-quotes-about-data)
- Data Management in Clinical Research: Best Practices | Datavant (https://datavant.com/blog/clinical-data-management)

Leave a Reply