Introduction
While Chile offers a promising landscape for first-in-human clinical trials, the path is fraught with challenges that demand careful navigation.
With its streamlined regulatory processes and robust healthcare infrastructure, Chile stands out as a strategic location for early-stage clinical research.
However, navigating these complexities can hinder progress and delay trial timelines.
By understanding these challenges, researchers can turn potential obstacles into strategic advantages.
Understand Regulatory Requirements for FIH Trials in Chile
To successfully conduct a first-in-human clinical trial in Chile, understanding the compliance framework is not just beneficial – it’s essential. Here are the essential steps to follow:
- Identify Regulatory Authorities: The Instituto de Salud Pública (ISP) is the main regulatory organization supervising clinical studies in Chile. Familiarize yourself with their guidelines and requirements to ensure compliance.
- Review Relevant Legislation: Key laws, including Law No. 20.120, govern human research and the Sanitary Code, outlining the ethical and procedural standards necessary for conducting studies.
- Prepare Required Documentation: Gather key documents like the clinical study protocol, informed consent forms, and qualifications of your investigators. Ensure all documents comply with ICH-GCP standards to facilitate approval.
- Submit to Ethics Committees: Obtain approval from an accredited Scientific Ethics Committee (CEC) before submitting to the ISP. This step is vital for ensuring ethical compliance and safeguarding patient welfare.
- Submit Application to ISP: After securing CEC approval, submit your application to the ISP. The review process typically takes about 30 days. Ensure your submission includes all required documentation to prevent delays.
- Understand FDA Acceptance: If you plan to submit data to the FDA, ensure your study design aligns with their requirements for acceptance of foreign clinical data under 21 CFR 812.28.
Navigating the compliance framework can be daunting, especially for first-time researchers involved in the first-in-human clinical trial in Chile. By following these steps, you not only ensure compliance but also enhance the likelihood of a successful study outcome. The ISP’s dedication to regulatory speed and efficiency, along with the country’s robust healthcare infrastructure, establishes it as a strategic advantage for early-stage clinical research.
Select and Prepare Clinical Trial Sites in Chile
Selecting the right clinical research locations is a critical factor that can determine the success of your first-in-human clinical trial in Chile. Here’s a structured approach to ensure effective site selection and preparation:
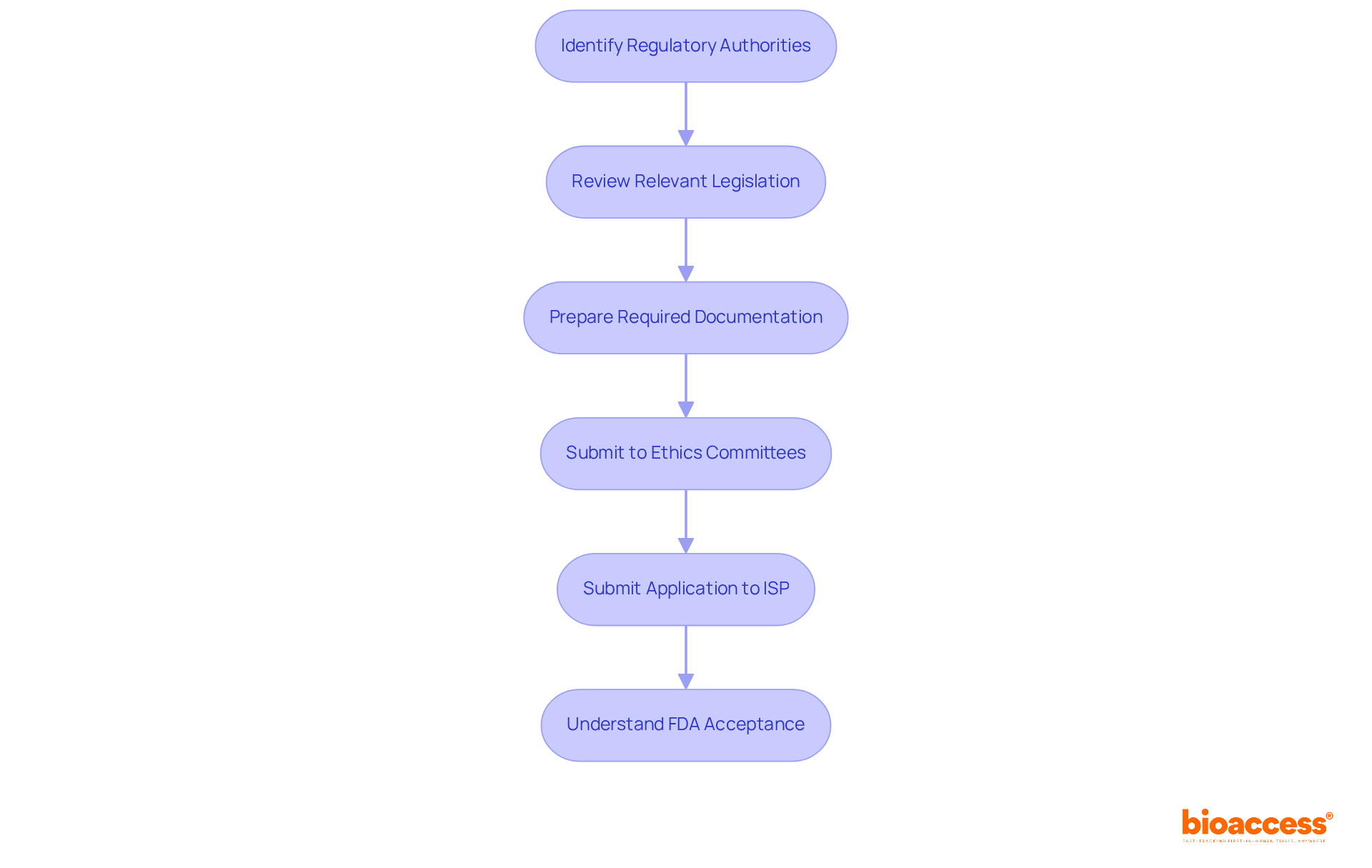
- Identify Potential Locations: Start by recognizing clinical locations with a demonstrated history in FIH studies. Look for facilities that have successfully conducted similar studies and show a track record of compliance and quality outcomes.
- Evaluate Location Capabilities: Assess each location’s capabilities, focusing on their infrastructure, staff qualifications, and patient access. Ensure that the location can meet the specific needs of your study protocol, including essential medical equipment and qualified personnel.
- Conduct Feasibility Studies: Perform comprehensive feasibility studies to assess the location’s capability to recruit the necessary number of participants within your desired timeline. Understanding local demographics can be challenging, yet it is essential for effective recruitment, especially in a context where Chile has streamlined its clinical study approval processes, reducing bureaucratic delays by over 30% (Instituto de Salud Pública, 2026). Early feasibility studies can further reduce risks in medical device development, allowing for a more informed approach to location selection.
- Prepare Location for Testing Execution: After selecting the locations, ready them for testing execution by training staff on the protocol and ensuring adherence to ICH-GCP guidelines. This preparation includes setting up necessary equipment and resources to facilitate smooth operations.
- Establish Communication Channels: Create clear communication pathways between the sponsor, staff at the location, and regulatory bodies such as INVIMA. Regular updates and meetings are essential for addressing any issues that may arise during the study, fostering collaboration and commitment.
- Monitor Site Readiness: Prior to trial initiation, conduct site visits to confirm that all preparations are complete and that the site is fully equipped to start enrolling participants. This proactive approach helps reduce delays and ensures that you meet industry standards.
As Julio G. Martinez-Clark, CEO of bioaccess® regulatory, highlights, “In the dynamic landscape of clinical research, navigating the regulatory framework in this region is crucial for success.” By prioritizing strategic site selection and preparation, you position your study as a first-in-human clinical trial in Chile for optimal success in the competitive landscape of clinical research. Moreover, consider the pediatric population, which makes up 30% of the residents in Chile, as an important patient group that could be approached for studies with customized recruitment strategies.
Implement Effective Patient Recruitment Strategies
Recruiting patients for first-in-human studies is not just a task; it’s a critical challenge that can determine the success of groundbreaking medical advancements. Effective patient recruitment is essential for the success of your first-in-human (FIH) study. Here are strategies to enhance recruitment efforts, particularly when leveraging bioaccess®’s Innovation Runway:
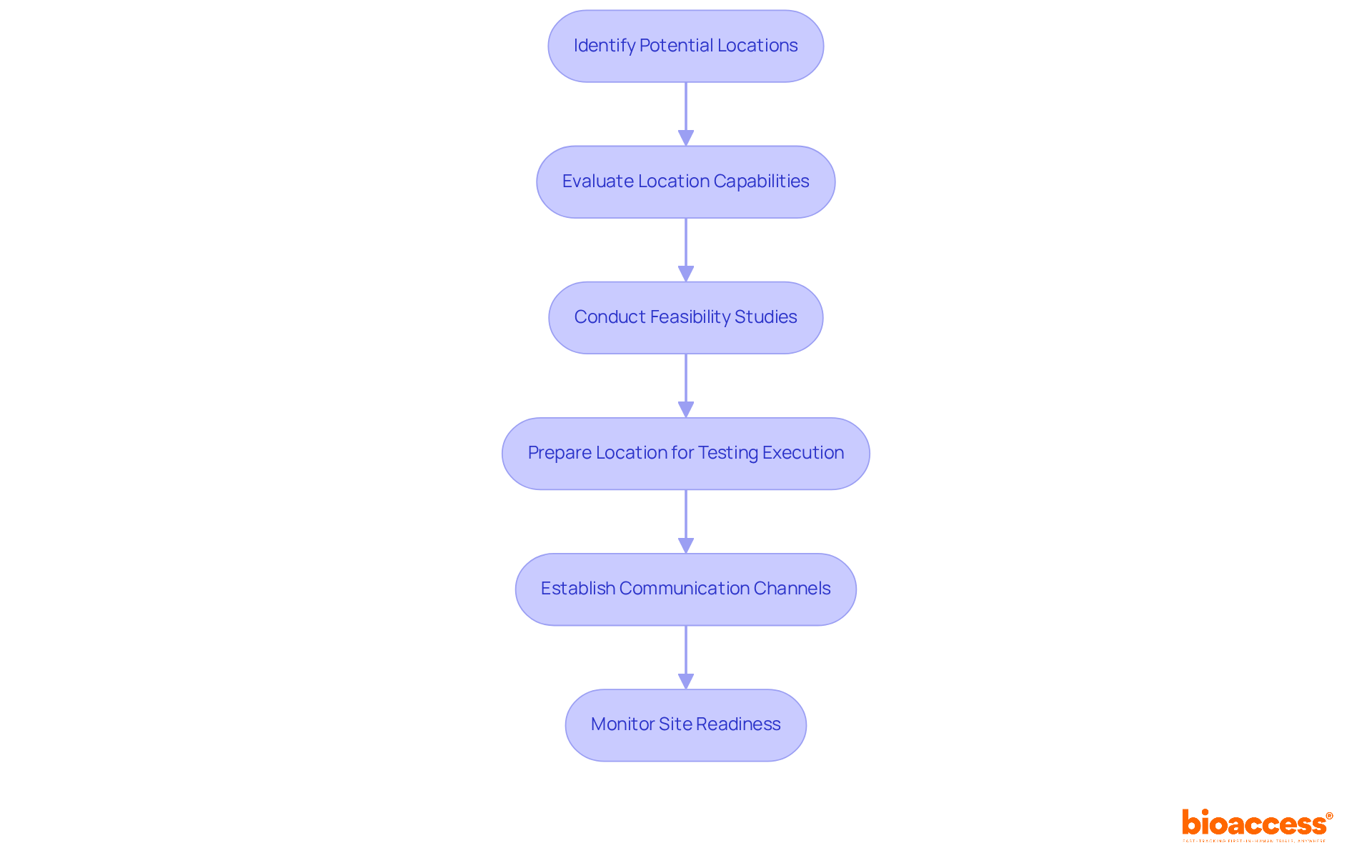
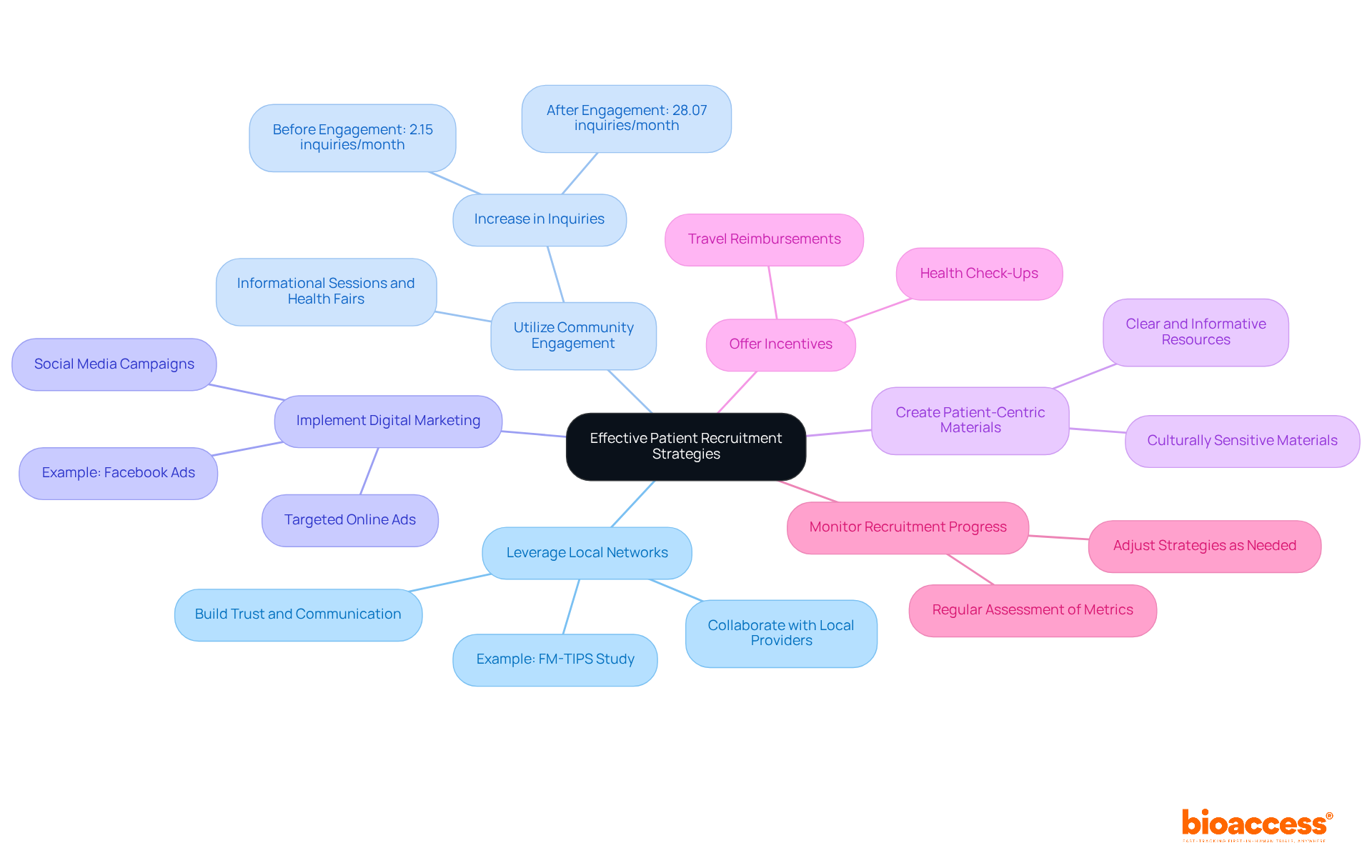
- Leverage Local Networks: Collaborate with local healthcare providers and institutions to identify potential participants. Building relationships with physicians can facilitate referrals, ensuring that you tap into established patient bases. For instance, the FM-TIPS study demonstrated that engaging local providers significantly increased referrals and highlighted the necessity of building trust and communication between researchers and community healthcare professionals.
- Utilize Community Engagement: By engaging with the community through informational sessions, health fairs, and local events, you not only raise awareness about the study but also build trust and interest among potential participants. Notably, community engagement efforts have shown that average monthly inquiries across clinics increased from 2.15 inquiries per month prior to engagement to 28.07 inquiries per month after implementing these strategies.
- Implement Digital Marketing: Use digital marketing strategies, including social media campaigns and targeted online ads, to reach a broader audience. Emphasize the study’s purpose and potential advantages to generate interest. Successful campaigns have demonstrated significant increases in inquiries and engagement, showcasing the power of online outreach.
- Create Patient-Centric Materials: Develop clear and informative resources that describe the study process, potential risks, and benefits. Ensure that these materials are culturally sensitive and available in Spanish to resonate with the local population. This approach aligns with the need for effective communication in diverse communities.
- Offer Incentives: Consider providing incentives for participation, such as travel reimbursements or health check-ups. This can inspire individuals to sign up and stay involved throughout the study, addressing potential obstacles to participation. However, it is essential to ensure that compensation is appropriate to avoid perceptions of coercion.
- Monitor Recruitment Progress: Regularly assess recruitment metrics to identify any challenges or delays. Adjust strategies as needed to ensure that enrollment targets are met on time. Continuous monitoring allows for timely interventions, enhancing overall recruitment efficiency.
Without effective recruitment strategies, the potential benefits of innovative treatments may never reach those who need them most. By embracing these strategies, you not only enhance recruitment but also pave the way for innovations, such as those arising from the first-in-human clinical trial in Chile, that could transform patient care in Chile and beyond. Additionally, understanding the regulatory landscape, including the streamlined assessment process outlined in Law 14.874/24, can further capitalize on the region’s unique advantages in speed, cost efficiency, and compliance pathways.
Monitor Trial Progress and Manage Data Effectively
In the high-stakes environment of the first-in-human clinical trial in Chile, effective monitoring and data management are not just beneficial – they’re essential for success. To ensure effective oversight, consider these critical steps:
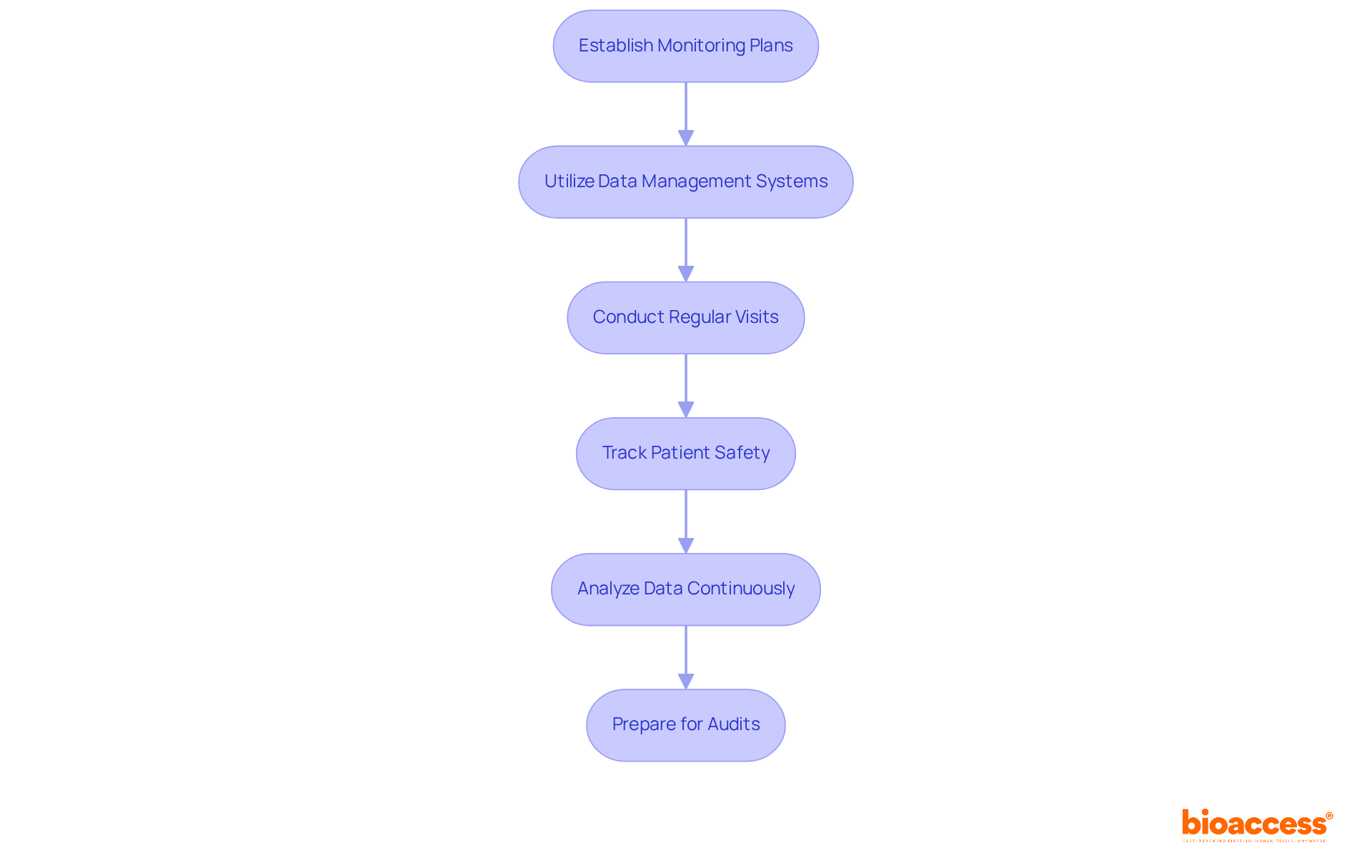
- Establish Monitoring Plans: Create a detailed monitoring plan that outlines the frequency and methods for overseeing activities, patient safety, and data integrity, aligning with ICH-GCP guidelines.
- Utilize Data Management Systems: Implement a robust clinical data management system (CDMS) that streamlines data collection, storage, and analysis while ensuring adherence to standards set by authorities such as ANVISA and COFEPRIS.
- Conduct Regular Visits: Schedule frequent visits to assess adherence to the trial protocol and compliance requirements. These visits should also provide support to site staff and address any emerging issues promptly.
- Track Patient Safety: Vigilantly monitor patient safety by reviewing adverse event reports and ensuring that all incidents are documented and reported to the relevant oversight bodies, such as INVIMA.
- Analyze Data Continuously: Engage in ongoing data analysis to identify trends and potential issues. This proactive approach enables timely interventions and necessary adjustments to the study protocol.
- Prepare for Audits: Organize all documentation for easy access during audits by regulatory authorities. This involves maintaining precise records of study activities, patient consent forms, and data entries to ensure compliance and transparency.
Neglecting these steps could jeopardize not only the trial’s integrity but also patient safety, making adherence to these guidelines imperative.
Conclusion
Navigating first-in-human clinical trials in Chile presents unique challenges that demand a strategic approach. Successfully navigating these trials requires a solid grasp of regulatory frameworks. It also demands strategic site selection, effective patient recruitment, and diligent monitoring of trial progress. By following the outlined step-by-step guide, researchers can streamline their approach, ensuring compliance and maximizing the potential for successful outcomes in this vital phase of clinical research.
Key insights from this guide emphasize the importance of engaging with regulatory authorities like the Instituto de Salud Pública (ISP) and adhering to ethical standards set by Scientific Ethics Committees. Additionally, selecting appropriate clinical trial sites and employing innovative recruitment strategies are crucial for attracting participants and maintaining momentum throughout the study. Navigating the complex regulatory landscape and ensuring effective patient recruitment can be daunting for researchers. The proactive management of trial data and patient safety monitoring further reinforces the integrity and reliability of the research process.
Embracing the advantages of conducting first-in-human trials in Chile – such as expedited approval timelines, robust healthcare infrastructure, and a diverse patient population – positions researchers to make significant strides in medical innovation. When stakeholders apply these best practices, they truly help create a thriving clinical research environment that enhances patient care in Chile and sets a model for successful trials across Latin America.
Frequently Asked Questions
What is the main regulatory authority for clinical trials in Chile?
The main regulatory authority supervising clinical studies in Chile is the Instituto de Salud Pública (ISP).
What key legislation governs human research in Chile?
Key legislation includes Law No. 20.120 and the Sanitary Code, which outline the ethical and procedural standards necessary for conducting studies.
What documentation is required to conduct a first-in-human clinical trial in Chile?
Required documentation includes the clinical study protocol, informed consent forms, and qualifications of investigators, all of which must comply with ICH-GCP standards.
What is the process for obtaining ethical approval for a clinical trial in Chile?
Researchers must obtain approval from an accredited Scientific Ethics Committee (CEC) before submitting their application to the ISP. This step is crucial for ensuring ethical compliance and patient welfare.
How long does the ISP take to review applications for clinical trials?
The review process typically takes about 30 days once the application is submitted to the ISP.
What should be considered if planning to submit data to the FDA?
If planning to submit data to the FDA, ensure that the study design aligns with their requirements for acceptance of foreign clinical data under 21 CFR 812.28.
Why is understanding the compliance framework essential for first-in-human trials in Chile?
Understanding the compliance framework is essential to ensure regulatory adherence, enhance the likelihood of a successful study outcome, and navigate the complexities of conducting clinical trials in Chile effectively.
What advantages does Chile offer for early-stage clinical research?
Chile offers advantages such as regulatory speed and efficiency, a robust healthcare infrastructure, and cost-effective patient recruitment, making it a strategic location for early-stage clinical trials.
List of Sources
- Understand Regulatory Requirements for FIH Trials in Chile
- Chile Clinical Trial Regulations | ISP Guide | bioaccess® (https://bioaccessla.com/regulatory-guide/chile)
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- First-in-Human Medical Device Trial in Chile | bioaccess® (https://bioaccessla.com/blog/first-in-human-medical-device-trial-chile)
- Chile (ISP – Instituto de Salud Pública) – PharmaRegulatory.in – India’s Regulatory Knowledge Hub (https://pharmaregulatory.in/country-specific-regulatory-affairs/chile-isp-instituto-de-salud-publica)
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Select and Prepare Clinical Trial Sites in Chile
- Clinical Trial Site Selection Latin America Ke… | bioaccess® (https://bioaccessla.com/blog/clinical-trial-site-selection-latin-america-key-strategies)
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Chile First-in-Human Clinical Trial Market Size, Share, Growth & Trends 2025–2033 (https://reedintelligence.com/market-analysis/chile-first-in-human-clinical-trial-market)
- Conducting Site Selection: The Key Factors and Strategies in Site Selection for Clinical Trials – NoyMed CRO (https://noymed.com/~noymedco/conducting-site-selection-the-key-factors-and-strategies-in-site-selection-for-clinical-trials)
- Clinical Trial Site Selection: Key Challenges & Solutions for Phase 3 Success (https://precisionformedicine.com/blog/clinical-trial-site-selection-key-challenges-solutions-for-phase-3-success)
- Implement Effective Patient Recruitment Strategies
- Community engagement is key to clinical trial recruitment and diversity (https://statnews.com/2019/08/23/clinical-trial-recruitment-diversity-community-engagement)
- Improving Recruitment Through Community Engagement (https://clinicalleader.com/doc/improving-recruitment-through-community-engagement-0001)
- Community engagement strategies improve recruitment and enrollment in a pragmatic clinical trial – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC12444702)
- Latin America: A Compelling Region To Conduct Your Clinical Trials (https://clinicalleader.com/doc/latin-america-a-compelling-region-to-conduct-your-clinical-trials-0001)
- 10 Inspiring Patient Experience Quotes | Relias (https://relias.com/blog/patient-experience-quotes)
- Monitor Trial Progress and Manage Data Effectively
- US FDA to Monitor Clinical Trial Data in Real Time in Pilot Program Aimed at Speeding Approvals (https://usnews.com/news/us/articles/2026-04-28/us-fda-to-monitor-clinical-trial-data-in-real-time-in-pilot-program-aimed-at-speeding-approvals)
- Quotes Related to Data and Data Governance (https://blog.idatainc.com/quotes-related-to-data-and-data-governance)
- FDA Moves to Real-Time Clinical Trial Patient Monitoring, Faster Drug Review – Drugs.com MedNews (https://drugs.com/news/fda-moves-real-time-clinical-trial-patient-monitoring-faster-review-129839.html)
- 19 Inspirational Quotes About Data | The Pipeline | ZoomInfo (https://pipeline.zoominfo.com/operations/19-inspirational-quotes-about-data)
- Importance of Clinical Data Management in Clinical Trials (https://sofpromed.com/importance-of-clinical-data-management-in-clinical-trials)

Leave a Reply