Introduction
For contract research organizations (CROs) in Chile, the regulatory landscape of medical device trials is both a formidable challenge and a unique opportunity. With a robust regulatory framework governed by the Instituto de Salud Pública and specific laws like Law N° 20.120, understanding these guidelines is crucial for ensuring compliance and expediting the approval process.
Consider this: CROs that engage local experts see a 30% increase in patient recruitment efficiency. How can MedTech startups leverage this advantage to not only meet regulatory demands but also enhance patient recruitment and trial efficiency?
Let’s explore some best practices that can help CROs navigate these trials effectively and position themselves for success in this competitive arena.
Understand Regulatory Frameworks for Medical Device Trials in Chile
Navigating the regulatory landscape for medical device trial CRO Chile can be a complex endeavor, yet it is essential for successful clinical research. In Chile, the Instituto de Salud Pública (ISP) and the Sanitary Code oversee the regulatory framework for studies related to the medical device trial CRO Chile. They outline the criteria for clinical research involving these devices. Key regulations, particularly in the context of medical device trial CRO Chile, include:
- Law N° 20.120, which addresses human research and genome studies.
- Decree N° 114 (2010) from the Ministry of Health (MINSAL), mandating adherence to Good Clinical Practice (GCP) standards, specifically ICH-GCP.
The approval timelines for medical device assessments typically range from 30 to 90 days, depending on the classification of the device. Grasping these regulations is vital. It ensures compliance and speeds up the approval process. Sponsors must prepare comprehensive documentation, including pre-market authorization applications, to facilitate a smooth review process. Familiarity with these frameworks not only supports compliance but also boosts the credibility of the study data, which is crucial for subsequent regulatory submissions in other jurisdictions, such as the FDA or EMA. Without this understanding, sponsors risk delays and complications in their studies.
Chile’s regulatory efficiency and cost-effectiveness make it an attractive spot for medical device trial CRO Chile. This allows sponsors to navigate the complexities of first-in-human research with ease. By utilizing the insights and strategies offered by bioaccess® through their Global Trial Accelerators™, Medtech startups can enhance their approach to clinical research in Chile, ensuring they fulfill regulatory requirements while capitalizing on the region’s advantageous conditions for patient recruitment and study execution. By leveraging bioaccess®’s expertise, sponsors can not only meet regulatory demands but also position themselves for success in the competitive Medtech landscape.

Select the Right CRO for Efficient Trial Execution
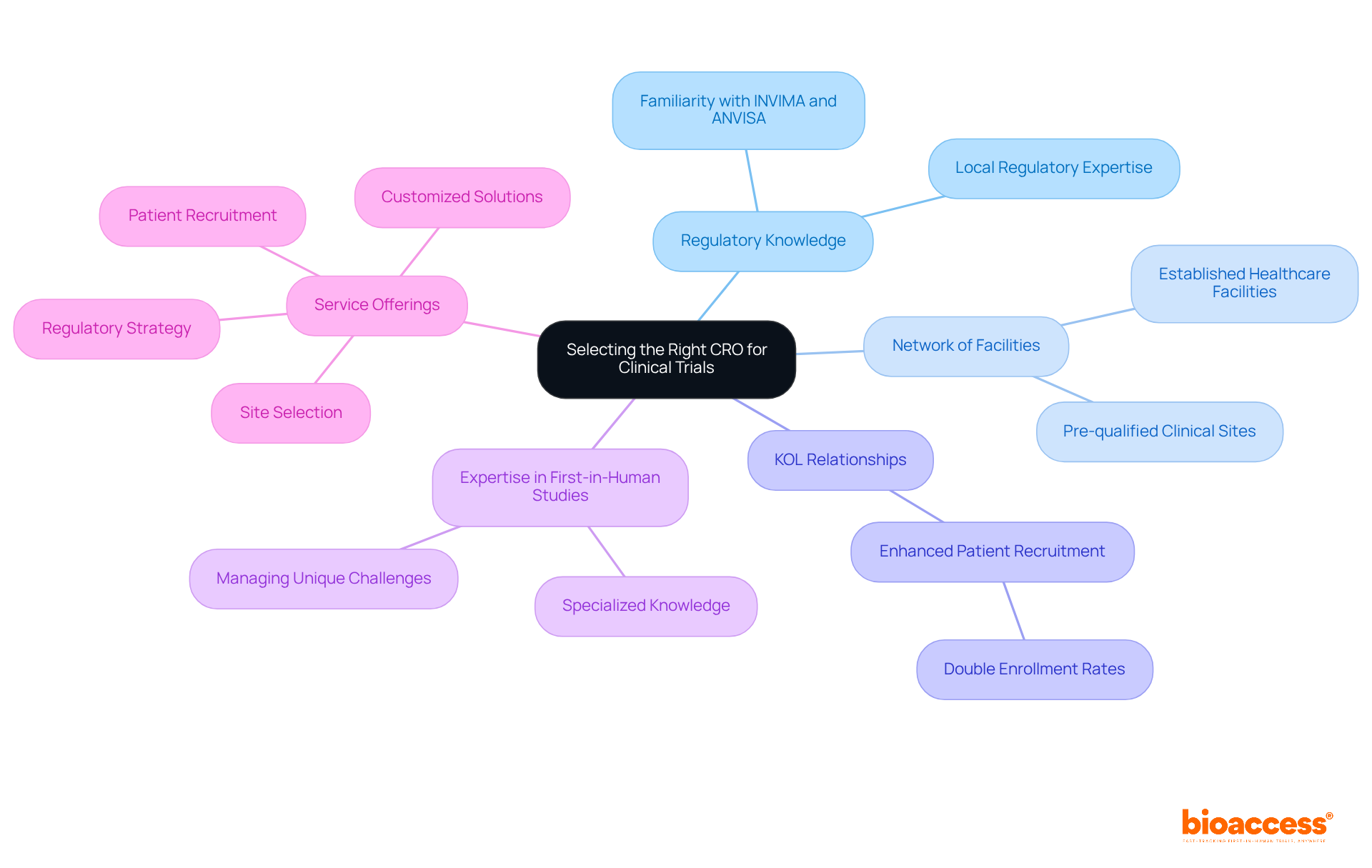
Selecting the right medical device trial CRO in Chile is not just important; it’s essential for success in a highly regulated environment. Evaluating a CRO’s familiarity with INVIMA and ANVISA regulations, along with their track record of successful studies in the region, is crucial. A CRO with a well-established network of healthcare facilities and pre-qualified clinical sites can significantly enhance patient enrollment and streamline data collection processes. Utilizing established Key Opinion Leader (KOL) relationships can double enrollment rates, significantly enhancing the efficiency of clinical trials.
Expertise in managing first-in-human studies is vital, as these investigations present unique challenges that require specialized knowledge. Look for a CRO that offers extensive services, including regulatory strategy, site selection, and patient recruitment, to ensure a seamless study process. bioaccess® stands out in this field, providing customized solutions that directly address the unique challenges faced by MedTech and Biopharma startups. Their Innovation Runway accelerates the development process, ensuring quicker activation timelines-typically within 30 to 90 days-and lower expenses, positioning them as a strategic ally for MedTech startups facing the challenges of early-phase studies in Chile.
Plus, having local regulatory expertise can really speed up the approval process, as the average timeline for clinical study approvals in Chile can be prolonged without the right knowledge. In fact, 70% of startups attempting first-in-human assessments in Chile’s public hospital system encounter delays due to tender blockades, which can result in procurement setbacks of 2 to 6 months for investigational devices not on existing tender lists. CROs like bioaccess® that comprehend the complexities of the tender system and have built connections with local hospitals can reduce these delays, ensuring that medical device trial CRO Chile studies proceed efficiently. By prioritizing these factors, MedTech startups can improve their chances of success in the competitive environment of clinical assessments in Chile. By making informed choices about CRO partnerships, MedTech startups can navigate the complexities of clinical trials and position themselves for success in the Chilean market.
Implement Effective Patient Recruitment Strategies
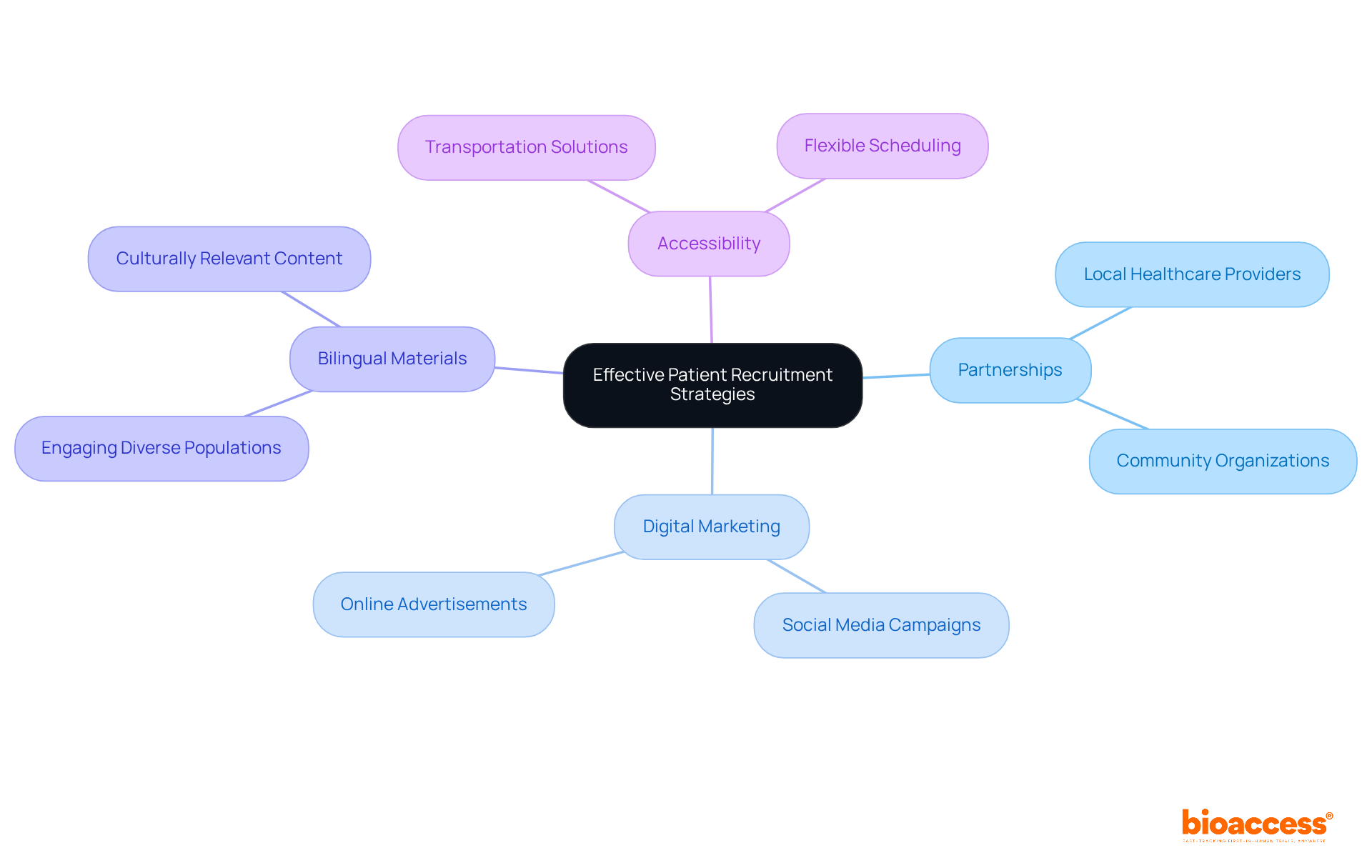
Effective patient engagement strategies are essential for the success of medical device trial CRO Chile. Regional healthcare networks and community outreach programs significantly enhance awareness about the trial and its benefits. Targeted digital marketing strategies, including social media campaigns and online advertisements, are crucial for effectively reaching potential participants. Bilingual outreach materials engage diverse patient populations and promote inclusivity.
Collaboration with regional physicians and healthcare providers enables referrals and fosters trust within the community, which is essential for successful hiring. For instance, bioaccess® has successfully employed these strategies, achieving rapid patient enrollment and significantly reducing the time to first patient in. This is especially crucial in the context of first-in-human (FIH) trials, where timely patient enrollment can expedite the overall trial timeline.
To enhance recruitment efforts, consider the following actionable steps:
- Establish partnerships with local healthcare providers to facilitate referrals.
- Implement targeted digital marketing campaigns to raise awareness.
- Create bilingual materials for hiring to engage diverse populations.
- Address barriers to participation, such as transportation and scheduling, to improve accessibility.
Many trials conducted by medical device trial CRO Chile struggle with participant engagement, leading to significant delays. Utilizing these strategies can alleviate challenges and enhance the process. Furthermore, grasping the regulatory environment, encompassing adherence to ICH-GCP standards and local authorities like ANVISA, is crucial for ensuring that participant sourcing efforts align with regulatory obligations and timelines. Importantly, bioaccess® can streamline 4-8 week ethics approvals and produce FDA/EMA-ready research data, further aiding the recruitment process. By focusing on these strategies, clinical studies can not only enhance recruitment but also ensure timely and successful outcomes.
Leverage Local Expertise for Enhanced Trial Outcomes
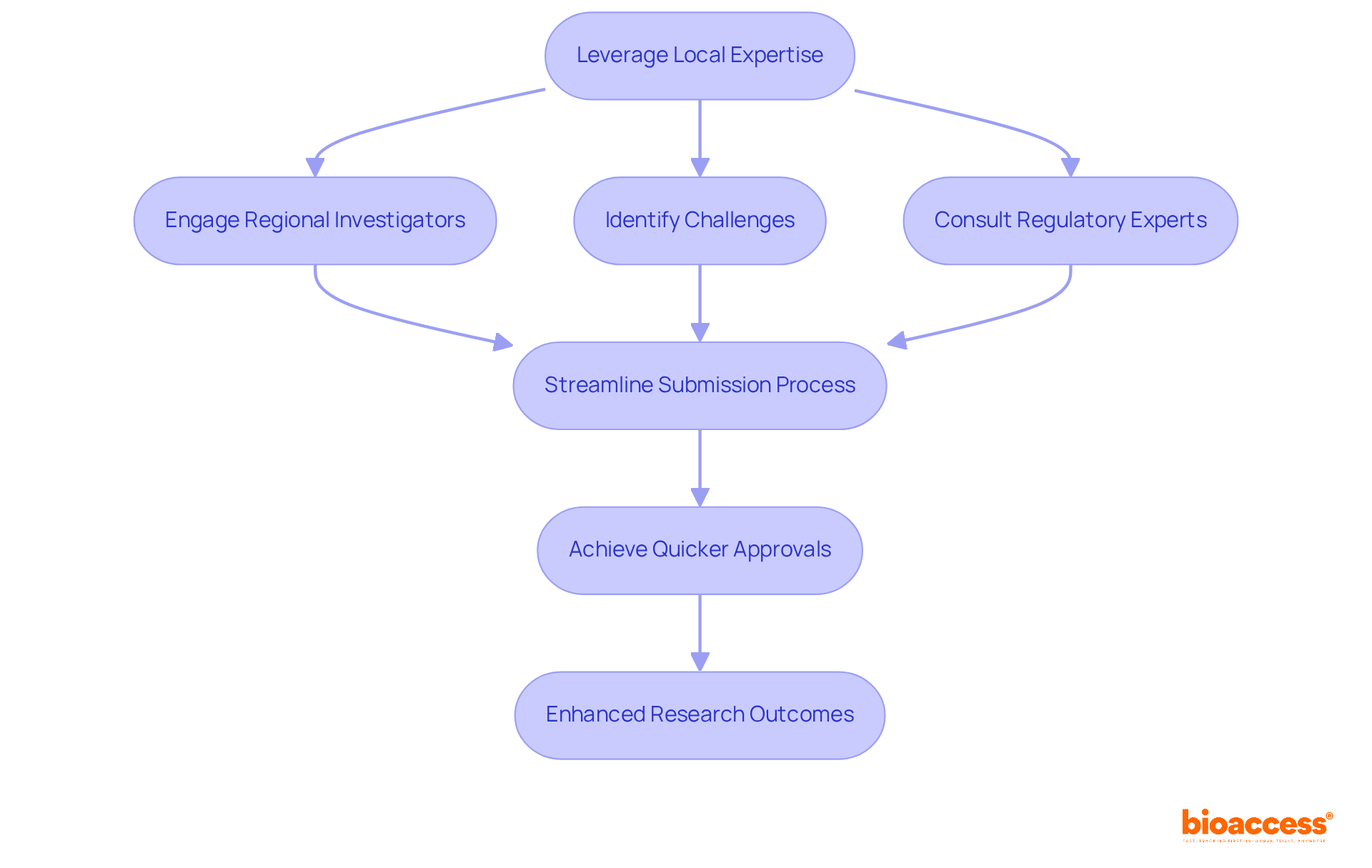
In the competitive landscape of clinical research, leveraging regional expertise in Chile is not just beneficial; it’s essential for success. Utilizing regional investigators who possess deep knowledge of the healthcare system and patient demographics provides invaluable insights into study design and execution. These regional experts are pivotal in pinpointing challenges in patient recruitment, regulatory compliance, and cultural nuances that can hinder trial success. Engaging regional regulatory consultants is crucial for streamlining the submission process, ensuring compliance with national guidelines established by the Instituto de Salud Pública (ISP) and accredited Ethics Committees (ECs).
For instance, bioaccess® has established partnerships with regional healthcare institutions and regulatory bodies, enabling efficient navigation of the regulatory landscape and achieving quicker approvals, typically within 30 business days. By incorporating regional expertise into study planning and execution, sponsors can enhance their strategies, lower expenses, and improve the overall quality of research data. This collaborative approach accelerates the timeline for initial human studies and aligns research initiatives with regional health priorities, ultimately resulting in more favorable research outcomes. Furthermore, with 52 percent of global clinical trials taking place outside the U.S., leveraging local expertise in Chile presents a strategic advantage for early-stage clinical trials.
Conclusion
While Chile offers a promising landscape for medical device trials, navigating its complexities can be daunting. Understanding the regulatory framework is crucial, as is selecting the right contract research organization (CRO). By leveraging local expertise and implementing effective patient recruitment strategies, MedTech startups can significantly enhance their chances of success.
Key insights include:
- The importance of adhering to local regulations established by the Instituto de Salud Pública (ISP)
- The necessity of selecting a CRO with a proven track record in the region
However, many MedTech startups struggle to find the right path amidst these complexities. The ability to effectively engage with patients through targeted outreach and utilize established networks can streamline the trial process, reduce timelines, and ultimately lead to more efficient outcomes. Furthermore, recognizing the regulatory nuances and approval timelines can greatly benefit early-stage clinical trials.
In conclusion, Chile’s strategic advantages, including regulatory efficiency and cost-effectiveness, make it an ideal location for early-stage clinical trials in the MedTech sector. By focusing on these best practices and collaborating with local experts, sponsors can not only navigate the complexities of clinical trials but also drive innovation and improve patient outcomes in the competitive landscape of medical device research. Ultimately, those who embrace these strategies will not only thrive in clinical trials but also contribute to groundbreaking advancements in patient care.
Frequently Asked Questions
What regulatory authorities oversee medical device trials in Chile?
In Chile, the Instituto de Salud Pública (ISP) and the Sanitary Code oversee the regulatory framework for studies related to medical device trials.
What are the key regulations for medical device trials in Chile?
Key regulations include Law N° 20.120, which addresses human research and genome studies, and Decree N° 114 (2010) from the Ministry of Health (MINSAL), which mandates adherence to Good Clinical Practice (GCP) standards, specifically ICH-GCP.
What is the typical approval timeline for medical device assessments in Chile?
The approval timelines for medical device assessments in Chile typically range from 30 to 90 days, depending on the classification of the device.
Why is understanding the regulatory framework important for sponsors conducting medical device trials in Chile?
Understanding the regulatory framework is vital for ensuring compliance, speeding up the approval process, and enhancing the credibility of study data for subsequent regulatory submissions in other jurisdictions, such as the FDA or EMA.
What documentation must sponsors prepare for medical device trials in Chile?
Sponsors must prepare comprehensive documentation, including pre-market authorization applications, to facilitate a smooth review process.
How does Chile’s regulatory environment benefit sponsors of medical device trials?
Chile’s regulatory efficiency and cost-effectiveness make it an attractive location for medical device trials, allowing sponsors to navigate the complexities of first-in-human research more easily.
How can bioaccess® support sponsors in navigating clinical research in Chile?
Bioaccess® offers insights and strategies through their Global Trial Accelerators™, helping Medtech startups enhance their approach to clinical research, ensuring they fulfill regulatory requirements while optimizing conditions for patient recruitment and study execution.
List of Sources
- Understand Regulatory Frameworks for Medical Device Trials in Chile
- First-in-Human Medical Device Trial in Chile | bioaccess® (https://bioaccessla.com/blog/first-in-human-medical-device-trial-chile)
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Drug & Medical Device Litigation Laws and Regulations Report 2026 Chile (https://iclg.com/practice-areas/drug-and-medical-device-litigation/chile)
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- Select the Right CRO for Efficient Trial Execution
- First-in-Human Medical Device Trial in Chile | bioaccess® (https://bioaccessla.com/blog/first-in-human-medical-device-trial-chile)
- Key Factors in CRO Selection | Applied Clinical Trials Online (https://appliedclinicaltrialsonline.com/view/key-factors-cro-selection-0)
- Should You Conduct Your Medical Device Clinical Trial In Latin America? – Journal for Clinical Studies (https://journalforclinicalstudies.com/conduct-medical-device-clinical-trial-latin-america)
- Latin America’s Landscape For Medtech Clinical Trials (https://clinicalleader.com/doc/latin-america-s-landscape-for-medtech-clinical-trials-0001)
- Selecting the Ideal CRO: 4 Key Factors for Successful Clinical Research Partnerships (https://clinixir.com/blog/selecting-the-ideal-cro-4-key-factors-for-successful-clinical-research-partnerships)
- Implement Effective Patient Recruitment Strategies
- Patient Recruitment Strategies for Clinical Trials (https://freyrsolutions.com/blog/patient-recruitment-strategies-for-clinical-trials)
- Enrollment in Clinical Trials: Statistics and Patient Recruitment Strategies | Power (https://withpower.com/guides/enrollment-in-clinical-trials-statistics-and-patient-recruitment-strategies)
- Clinical studies: the challenge of patient recruitment (https://servier.com/en/newsroom/clinical-studies-patient-recruitment)
- Patient Recruitment Strategies for Clinical Trial Success (https://totaldiversity.com/clinical-study-recruitment)
- Leverage Local Expertise for Enhanced Trial Outcomes
- Clinical trial success depends on strong partnerships – here’s why – Siron Clinical (https://sironclinical.com/clinical-trial-success-depends-on-strong-partnerships-heres-why)
- 6 Important Quotes from Medtech Leaders (https://mddionline.com/business/6-important-quotes-from-medtech-leaders)
- Latin America: A Compelling Region To Conduct Your Clinical Trials (https://clinicalleader.com/doc/latin-america-a-compelling-region-to-conduct-your-clinical-trials-0001)
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- 6 Benefits of Clinical Research Collaboration and Partnerships (https://vccrn.org/benefits-clinical-research-collaboration-and-partnerships)

Leave a Reply