Introduction
Understanding the Importance of Clinical Trials in Healthcare Innovation
Clinical trials play a crucial role in driving healthcare innovation, particularly in the field of medical devices. However, the landscape of clinical trials is evolving rapidly, with increasing complexities and a surge in data volume. From logistical challenges to regulatory nuances, this article explores the intricacies of modern clinical trials and their impact on patient experience and global coordination.
It also delves into the expanding data landscape and the need for sophisticated data management methods. Additionally, regulatory oversight and compliance are highlighted as crucial aspects of medical device trials. By understanding the importance of clinical trials and their various facets, stakeholders can contribute to the advancement of medical treatments and enhance patient care.
Understanding the Importance of Clinical Trials in Healthcare Innovation
The landscape of is evolving rapidly, with a surge in the volume of data and an increasingly complex global environment. For instance, a patient with a rare condition in rural Pennsylvania may find hope in a clinical trial for a novel . However, the trial’s location in Turkey presents significant , such as securing visas, navigating foreign documentation, and coordinating travel.
Such scenarios underscore the intricacies of modern , which extend far beyond data collection to encompass and global coordination. Furthermore, the in has expanded monumentally. A Phase 3 trial now produces an average of 3.6 million data points, a threefold increase from a decade ago.
This aligns with the broader trend in healthcare where medical data is doubling every 70 days, compared to every 50 years in 1950. This explosion of information necessitates sophisticated methods to manage, analyze, and leverage data to enhance clinical trial processes and outcomes. remains a cornerstone of trials.
In the United States, the FDA’s classification system categorizes devices from low-risk class one to high-risk class three based on potential risks and the level of regulatory control required. High-risk class three devices, such as life-sustaining implantable pacemakers, undergo stringent reviews and have lengthy approval processes. Understanding these regulatory nuances is crucial for stakeholders, as it informs the pathway a will take before it can reach those who need it most.
Marketing Strategies for Clinical Trials: Lessons from Business
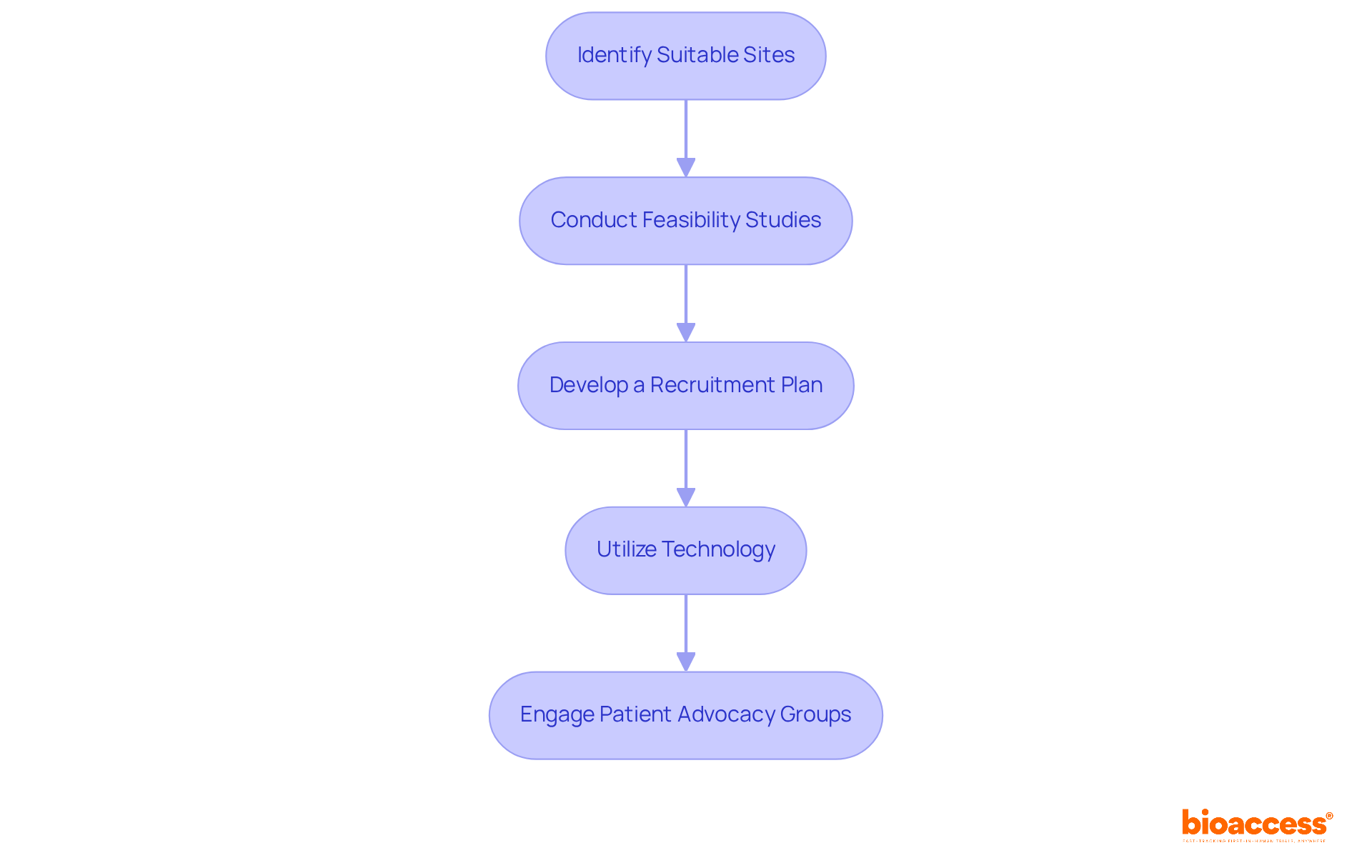
face the challenge of not only developing safe, effective medical products but also effectively recruiting and retaining trial participants. A prime example is a patient from rural Pennsylvania with an ultra-rare disease, who must navigate the complexities of international travel to join a trial in Turkey. This patient’s dilemma underscores the importance of a strategic marketing approach that goes beyond traditional and digital advertising to address potential participants’ concerns and logistical hurdles.
To enhance , marketing strategies should incorporate clear communication about the practical aspects of trial participation, such as in a foreign language. Additionally, it is imperative to highlight and the provided by . These courses, which cater to healthcare professionals and those involved in drug development, emphasize the safety concerns inherent in medical product development.
As one healthcare business development leader with over three decades of experience notes, successful medical companies—and by extension, clinical trials—rely on . These can include acquisitions, IPOs, partnerships, and strategic alliances, which not only contribute to the success of the companies but also impact the clinical trials they oversee. By aligning marketing strategies with these business objectives and addressing the specific needs of the target populations, can significantly improve recruitment outcomes and drive the success of their trials.
The Role of Marketing in Clinical Trials: A Case Study
are increasingly recognizing the power of to enhance the success of their studies, particularly when it comes to . A compelling case study can be drawn from the innovative approach of using (RCTs) in healthcare delivery.
This method, as implemented by , bypasses the traditional, lengthy process of recruiting participants for medical experiments. Instead, it rapidly tests and refines , adapting based on immediate results.
The effectiveness of such marketing strategies becomes even more evident when considering the challenges faced by potential participants. For instance, a patient in rural Pennsylvania with an ultra-rare disease may have to navigate complex logistics to join a trial in Turkey, dealing with language barriers and travel coordination.
Marketing efforts that address these concerns and provide clear guidance can be critical in encouraging participation. Moreover, the contrast between consumer and medical marketing is highlighted by the nuances of promoting health-related services. As noted by a senior vice president and executive creative director, the healthcare sector requires an understanding of these subtle yet significant differences. Marketing medical devices and therapies involves not only reaching the target audience but also educating and guiding them through the complexities of . By analyzing such case studies, can better comprehend and apply sophisticated marketing tactics to attract and retain participants, ultimately contributing to the success of their .
Key Takeaways from a Scientific Article on Clinical Trials
Exploring the intricacies of medical device clinical trials, it is crucial to address the challenges and complexities faced by patients and researchers alike. Take, for example, the plight of a patient from rural Pennsylvania, grappling with an ultra-rare disease and presented with the chance to join a clinical trial in Turkey.
The logistical hurdles, such as visa procurement, navigating foreign paperwork, and coordinating travel, become as significant as the trial itself. The scenario underscores the necessity for to consider patient accessibility and support in their protocols.
is another pivotal aspect, with the and the EMA in Europe ensuring meet stringent safety standards. The FDA’s classification of devices into three risk-based categories, with class three devices like pacemakers undergoing the most rigorous scrutiny, highlights the complexity of bringing to market.
These devices, crucial to life support, represent a mere 10% of FDA-regulated devices but face lengthy approval processes. This information is vital for to navigate the effectively. Moreover, is a key component of medical device trials. Details such as device type, manufacturer, brand name, and lot number, along with the nature of the problem – be it a defect or malfunction – and the context of use are critical for ensuring ongoing patient safety and improving device reliability. must meticulously track and analyze such data to maintain regulatory compliance and uphold the highest standards of patient care.
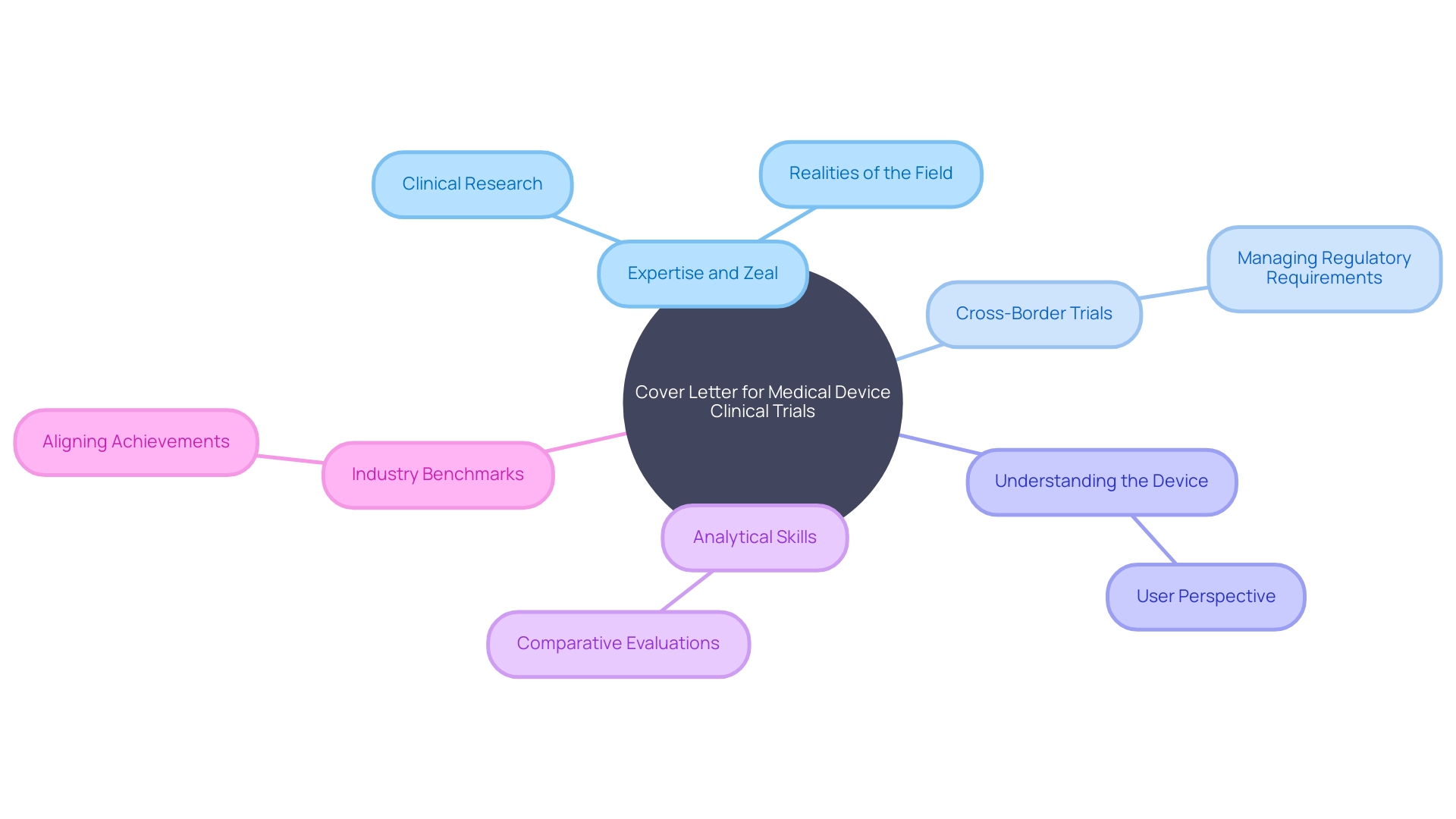
Crafting a Killer Cover Letter for Clinical Trial Professionals
Creating an impactful cover letter for positions in involves more than just stating your interest in the role. It requires a narrative that connects your expertise and zeal for with the intricate realities of the field.
Imagine a scenario where a patient with a rare disease in rural Pennsylvania has the chance to join a clinical trial in Turkey. The logistical challenges they face underscore the complexities you must navigate as a clinical trial professional.
Your cover letter should articulate how you can support such situations, showcasing your ability to handle , manage , and empathize with patients’ circumstances. In your cover letter, it’s imperative to demonstrate a deep understanding of the you’ll be working with.
This means delving into the needs of all users, from clinicians to patients, and comprehending the device’s instructions, warnings, and cautions. Highlight your proactive efforts in researching competitive devices, illustrating your analytical skills through comparative evaluations you’ve constructed. Furthermore, align your achievements with the industry’s benchmarks for success, such as participation in successful or integration projects between software and hardware, to show your potential value to the company. By intertwining your qualifications with real-world applications, your cover letter will not only capture the attention of hiring managers but also convey your readiness to contribute to the advancement of medical treatments.
Healthcare Content Marketing Strategies for Clinical Trials
stands as an indispensable strategy for aiming to enlighten and captivate their audience, particularly in the realm of . Crafting educational blog posts, dynamic videos, interactive webinars, and eye-catching infographics are pivotal in disseminating information about , which range from simple implements like tongue depressors to complex machinery aiding in medical testing.
Such content not only educates on the device’s purpose—diagnosis, monitoring, or treatment of health conditions—but also illustrates the life-enhancing benefits these devices provide. For instance, consider the profound impact of informative content on a patient in rural Pennsylvania, grappling with a rare disease and contemplating participation in an overseas .
Clear, accessible content could navigate them through the complexities of international travel logistics, visas, and language barriers, thereby easing their journey. This underlines the power of content in fostering trust and credibility, essential for enhancing awareness and trial enrollment.
Moreover, can also highlight the for medical device companies, such as acquisitions, IPOs, or strategic alliances, which serve as markers of success in the industry. As noted by industry leaders, the shared experiences and insights within the content can significantly influence the perceptions of healthcare professionals and patients alike, ultimately advancing medical practices and . Supporting this, thematic intelligence reports suggest that informed decision-making is crucial for businesses, urging the provision of sample pages to help stakeholders understand the value of full reports. In essence, is more than just promotion—it’s a conduit for connection, education, and empowerment in the medical device sector.
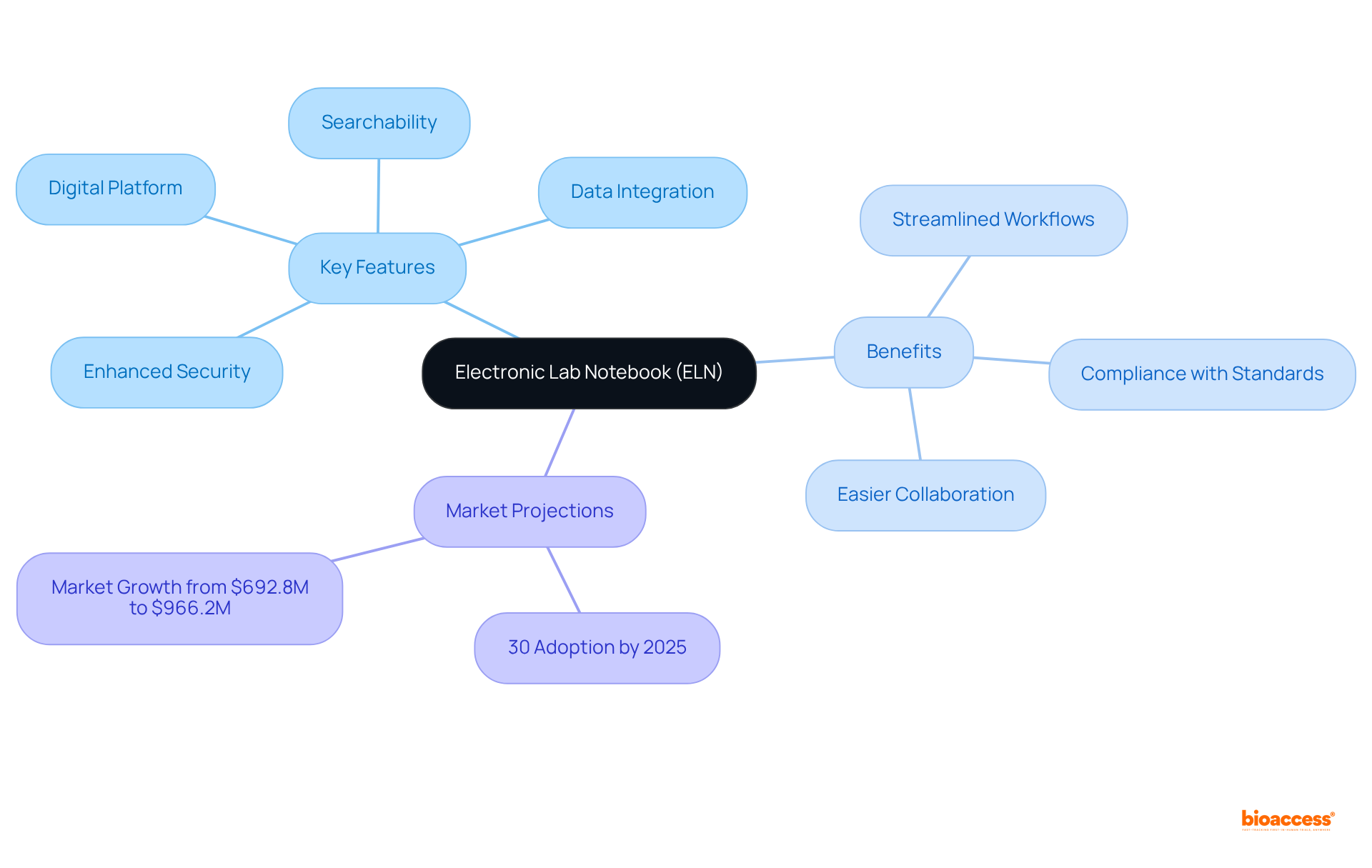
Challenges in Healthcare Content Marketing for Clinical Trials
Navigating the realm of presents with distinct challenges, ranging from adhering to stringent regulatory requirements to effectively reaching and engaging a diverse audience. The complexities of the demand not only technical accuracy in writing but also a nuanced approach to selling services. For instance, Cognitive FX leverages its expertise in treating concussion patients to construct content that resonates with readers’ needs while simultaneously promoting their services, illustrating the delicate balance between educating and selling.
Moreover, the intricacies of SEO keyword strategies cannot be understated. They must align with the specific inquiries of the target audience, reflecting their stage in the healthcare journey, and indicating a readiness to engage with the services offered. This level of precision is often a formidable task for and product companies, as evidenced by the experiences of our healthcare clients.
Additionally, the industry faces challenges in creating a universal understanding of elements, prompting the need to clarify the distinctions between various forms of content such as white papers, e-books, and campaigns. To underscore the importance of precise and compassionate , consider the patient from rural Pennsylvania navigating the logistical hurdles of . Their plight emphasizes the need for content that not only informs but also assists in the complex decision-making process.
Statistics further demonstrate the shifting landscape of healthcare, with telehealth services surging in importance due to the pandemic. This rise in virtual healthcare services offers a new avenue for , emphasizing the need to inform and support patients remotely. The adoption of telehealth showcases the potential for to adapt and thrive amidst changing healthcare dynamics and patient needs.
 Challenges in the Healthcare Industry The Mind Map of Content Marketing Challenges in the Healthcare Industry](https://vectorly-files.s3.eu-central-1.amazonaws.com/c3aad015-c086-47c2-9245-35c85d1ea4fc.jpg)
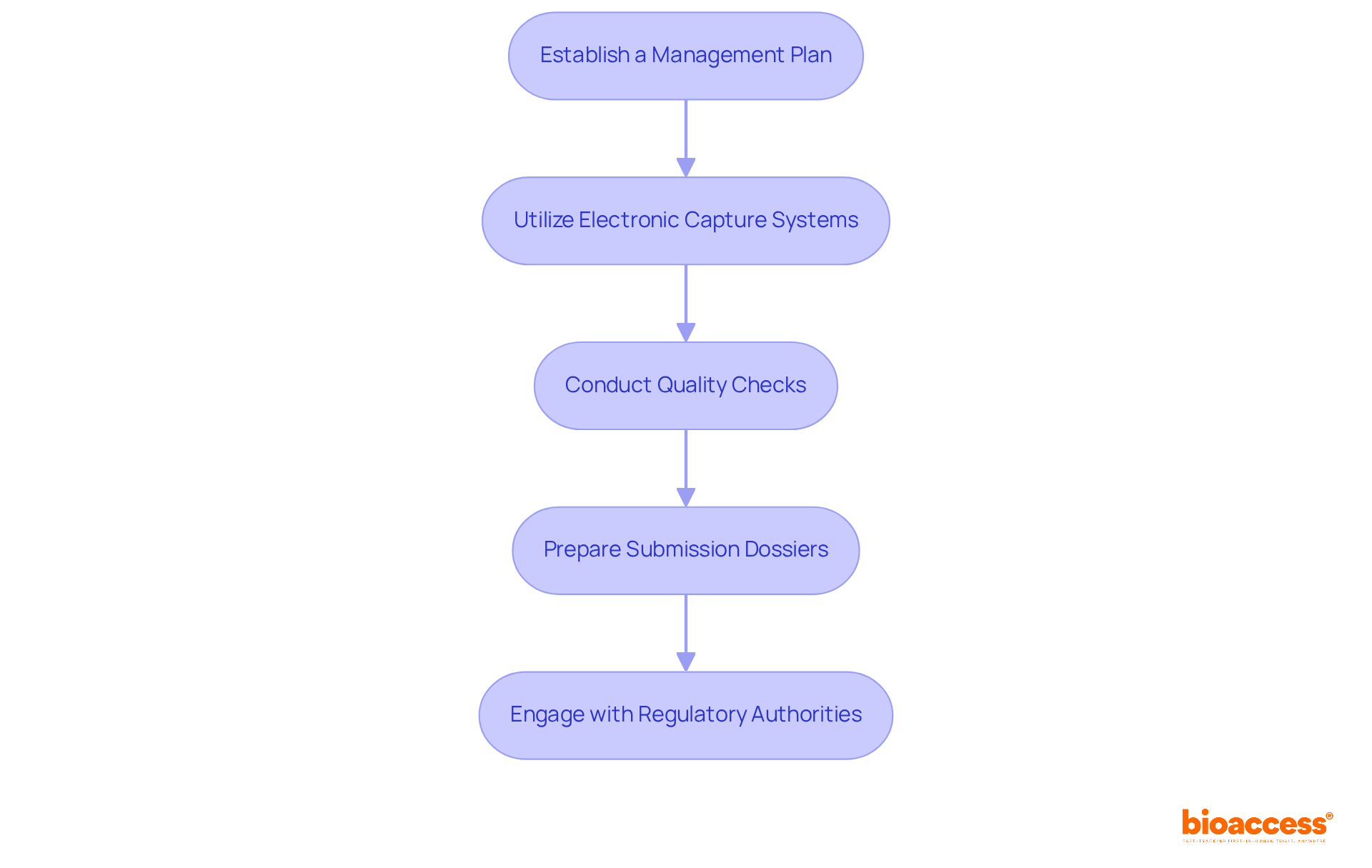
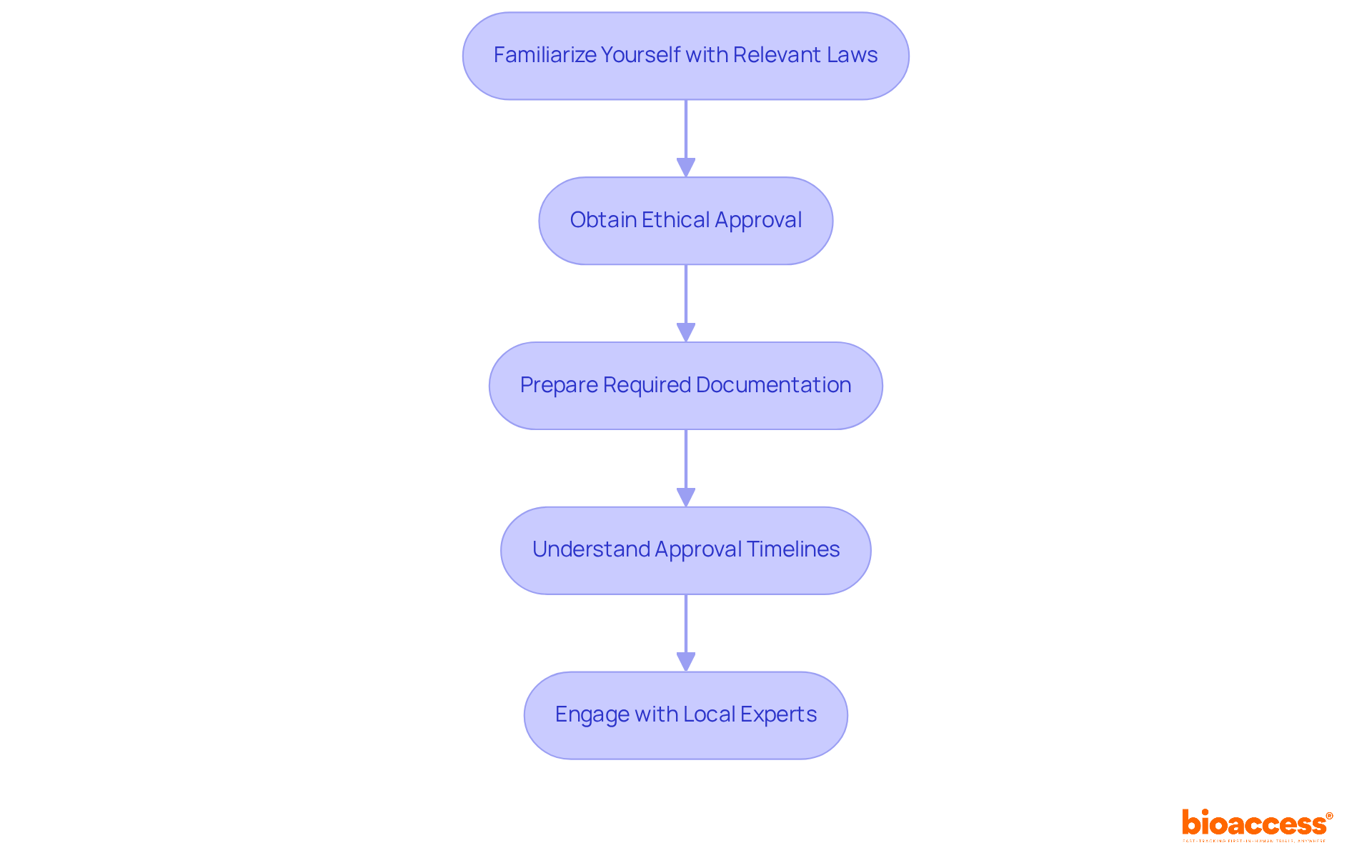
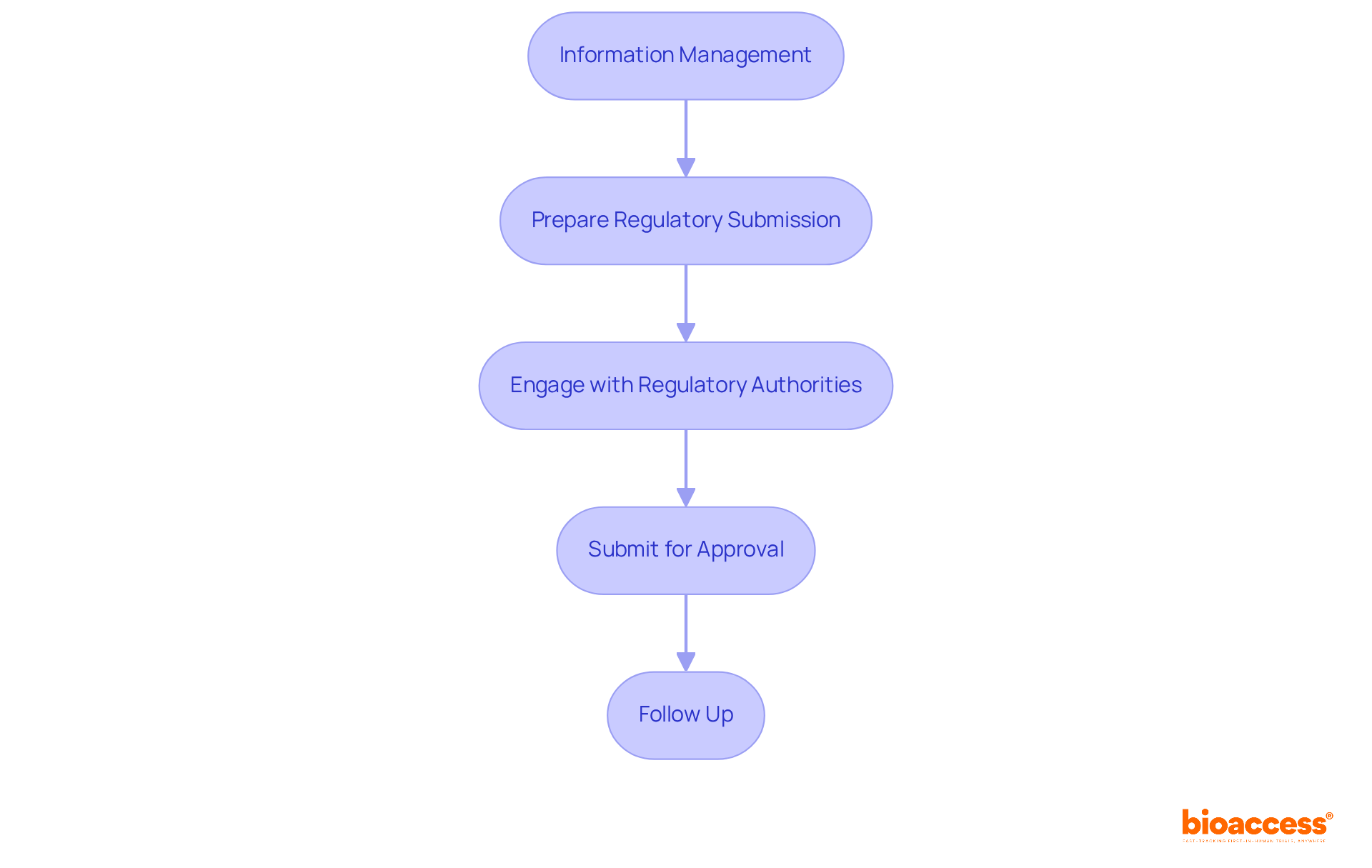
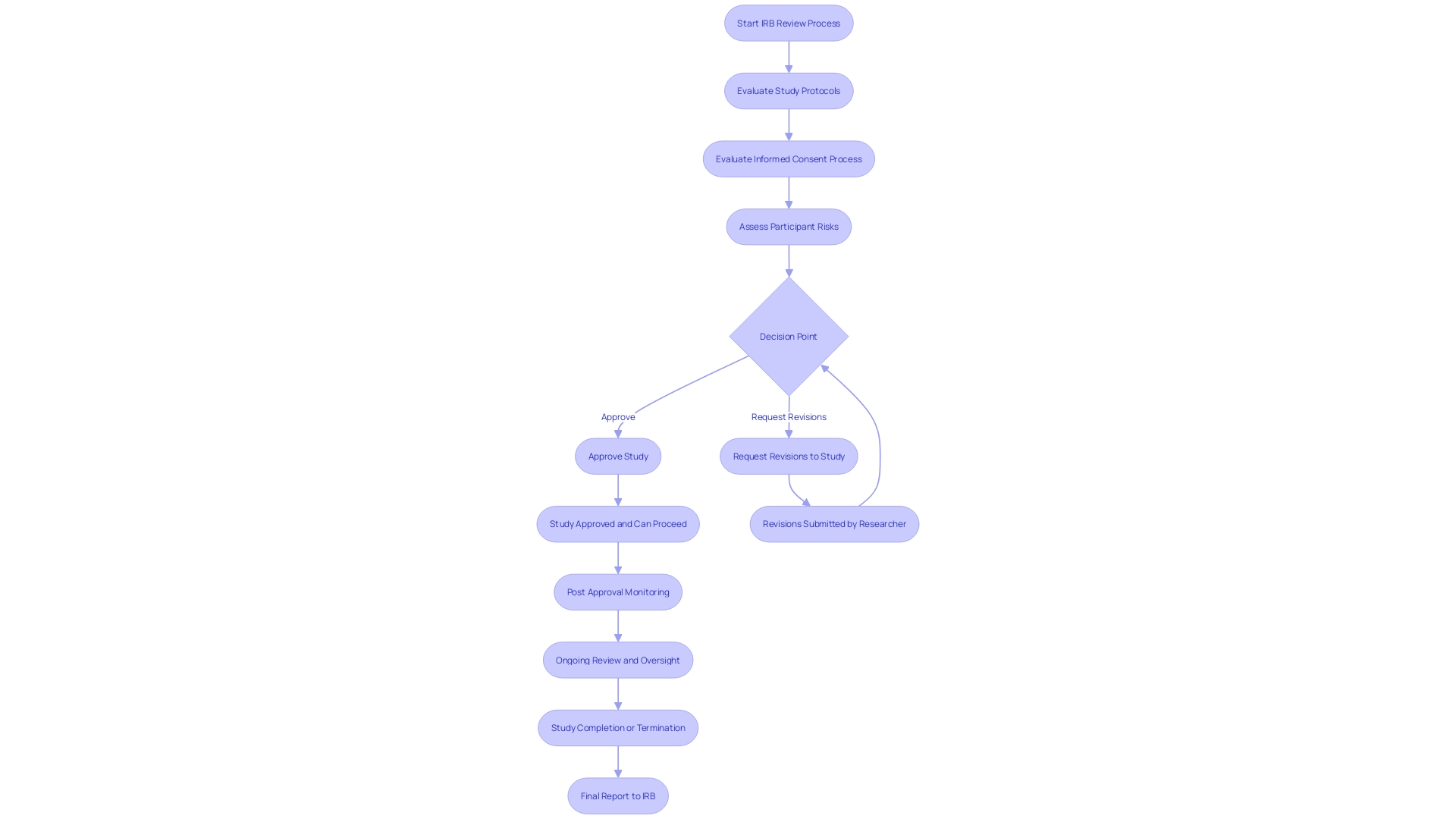
Best Practices for Medical Device Clinical Trials: Regulations and Compliance
Adherence to is a cornerstone of conducting medical device , which are paramount for and ensuring the reliability of trial data. Clinical trial companies are tasked with navigating a complex , often spearheaded by the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) among other global entities.
These agencies are dedicated to the public health by ensuring medical devices’ safety, efficacy, and security. As new medical device technologies emerge, the regulatory framework continues to evolve, necessitating companies to stay abreast of the latest guidelines to mitigate risks and avoid delays in bringing products to market.
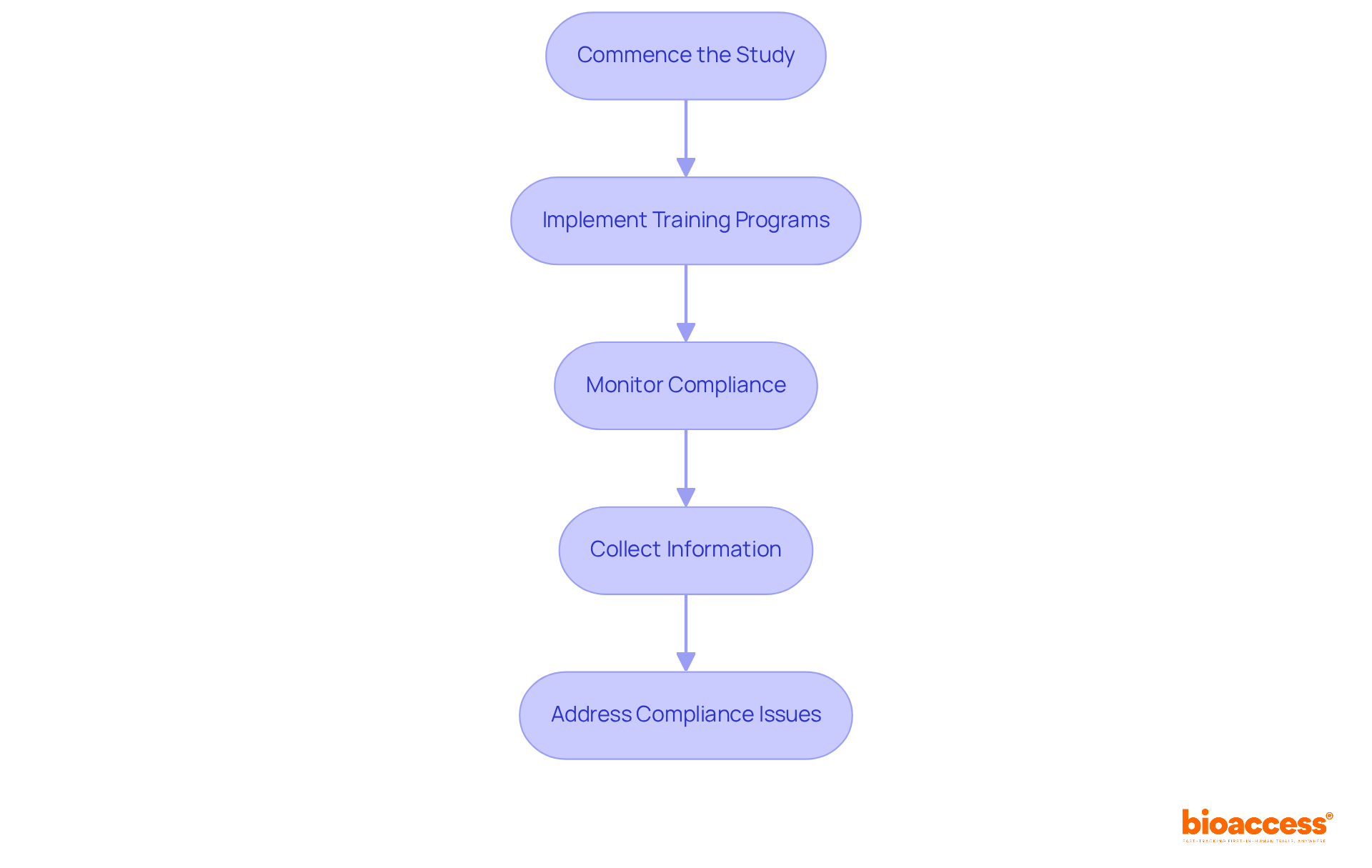
To maintain compliance, it’s essential for clinical trial companies to procure all necessary clearances from regulatory bodies, strictly adhere to ethical standards, and implement comprehensive data management systems. Conducting regular audits is also pivotal to ensure ongoing adherence to regulatory requirements.
In light of the rapid advancements in the medical device industry, regulatory authorities regularly update their guidelines to reflect scientific progress and reinforce patient safety. In this dynamic landscape, clinical trial companies bear the responsibility of managing increasing regulatory complexities, especially as devices become more digitalized and sophisticated. The importance of in determining the safety and efficacy of new treatments cannot be overstated. With providing crucial data that influences patient care and outcomes, it is imperative that these studies are . From initial safety assessments in phase one trials to efficacy evaluations in phase two, each stage of must meet stringent standards, underscoring the critical role of .
Conclusion
Clinical trials are vital for healthcare innovation, especially in the realm of medical devices. The landscape of these trials is rapidly evolving, with increasing complexities and a surge in data volume.
Modern trials encompass not only data collection but also patient experience and global coordination, presenting logistical challenges that must be overcome. Managing the expanding data landscape requires sophisticated methods to enhance trial processes and outcomes.
Regulatory oversight and compliance are crucial, with understanding classification systems being vital for stakeholders. Postmarket reporting of adverse events plays a key role in ensuring ongoing patient safety.
Effective marketing strategies are essential for successful trials, going beyond traditional advertising to address participants’ concerns and logistical hurdles. Clear communication about practical aspects is imperative, along with content marketing to educate and engage the audience effectively. Overall, stakeholders must understand the intricacies of clinical trials to contribute to medical advancements and improve patient care. Adherence to regulatory standards, sophisticated data management methods, and strategic marketing approaches are crucial for driving successful trials. By prioritizing quality, integrity, and compliance throughout the process, stakeholders can make significant contributions to healthcare innovation and improve patient outcomes.
Frequently Asked Questions
What is the current trend in the volume of data for medical device clinical trials?
The current trend shows a surge in the volume of data, with a Phase 3 trial now producing an average of 3.6 million data points, which is three times more than a decade ago.
How often does medical data double in size, and how does this compare to the past?
Medical data is currently doubling every 70 days, a significant increase when compared to every 50 years in 1950.
What are the logistical challenges patients might face when participating in clinical trials abroad?
Patients might face challenges like securing visas, navigating foreign documentation, and coordinating travel.
How does the FDA classify medical devices, and what does each class represent?
The FDA classifies medical devices into three risk-based categories: low-risk class one, medium-risk class two, and high-risk class three, with class three devices undergoing the most stringent reviews.
What kind of medical device falls under the FDA’s high-risk class three category?
Life-sustaining implantable pacemakers are an example of high-risk class three devices.
Why is regulatory oversight important in medical device trials?
Regulatory oversight is crucial to ensure medical devices meet stringent safety standards and to inform the pathway a medical device will take before reaching patients.
How do clinical trial companies improve patient recruitment?
Companies improve recruitment by incorporating clear communication about practical aspects of participation, highlighting safety measures, and providing educational opportunities through clinical investigator training courses.
What role does marketing play in clinical trials?
Marketing plays a critical role in addressing potential participants’ concerns, streamlining their decision-making process, and enhancing trial success by effectively reaching and educating target audiences.
What is the innovative approach used by Dr. Leora Horwitz’s team in clinical trials?
Dr. Leora Horwitz’s team uses rapid randomized controlled trials (RCTs) to quickly test and refine healthcare delivery improvements.
What should a cover letter for a clinical trial professional demonstrate?
A cover letter should connect the applicant’s expertise and passion for clinical research with the complexities of the field, including the ability to handle cross-border trials, manage regulatory requirements, and empathize with patients’ circumstances.
What is the purpose of content marketing in clinical trials?
Content marketing aims to educate and captivate the audience about medical devices, highlighting their purpose and benefits, and fostering trust and credibility to enhance awareness and trial enrollment.
What are some challenges faced in healthcare content marketing for clinical trials?
Challenges include adhering to regulatory requirements, reaching and engaging diverse audiences, maintaining technical accuracy, and balancing educational content with promotional goals.
What are the best practices for ensuring compliance in medical device clinical trials?
Best practices include obtaining all necessary regulatory clearances, adhering to ethical standards, implementing data management systems, conducting regular audits, and staying updated with the latest guidelines.
Why is compliance critical in medical research?
Compliance is critical to ensure the safety and efficacy of new treatments, maintain quality and integrity in trials, and influence patient care and outcomes.
List of Sources
- Understanding the Importance of Clinical Trials in Healthcare Innovation
- globaldata.com (https://www.globaldata.com:443/store/report/medical-devices-industry-m-and-a-deals-by-theme-quarterly-analysis/)
- medicaldevice-network.com (https://www.medicaldevice-network.com/sponsored/reducing-medical-device-approval-times-in-2023/)
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- medtechintelligence.com (https://medtechintelligence.com/column/the-power-of-ai-to-enhance-clinical-trials/)
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- medicaldevice-network.com (https://www.medicaldevice-network.com/sponsored/reducing-medical-device-approval-times-in-2023/)
- Marketing Strategies for Clinical Trials: Lessons from Business
- starfishmedical.com (https://starfishmedical.com/blog/18-business-factors-that-determine-successful-medical-device-exits/)
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- starfishmedical.com (https://starfishmedical.com/blog/medical-device-phase-one-product-development-pro-tips/)
- fda.gov (https://www.fda.gov/drugs/news-events-human-drugs/fda-clinical-investigator-training-course-citc-2023-12062023)
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- clinicaltrialsarena.com (https://www.clinicaltrialsarena.com/sponsored/why-patient-centricity-is-the-answer-to-the-clinical-trial-enrolment-gap/)
- The Role of Marketing in Clinical Trials: A Case Study
- theatlantic.com (https://www.theatlantic.com/health/archive/2023/08/preventive-medicine-screenings-a-b-testing/675058/)
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- pharmalive.com (https://www.pharmalive.com/overcoming-the-highly-nuanced-challenges-of-medical-marketing-to-drive-meaningful-patient-connection/)
- Key Takeaways from a Scientific Article on Clinical Trials
- fda.gov (https://www.fda.gov/medical-devices/human-factors-and-medical-devices/human-factors-postmarket-information-device-surveillance-and-reporting-processes)
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- medicaldevice-network.com (https://www.medicaldevice-network.com/sponsored/reducing-medical-device-approval-times-in-2023/)
- Crafting a Killer Cover Letter for Clinical Trial Professionals
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- medicaldevice-network.com (https://www.medicaldevice-network.com/buyers-guide/medical-devices-development/)
- medtechintelligence.com (https://medtechintelligence.com/feature_article/510k-submission-planning/)
- starfishmedical.com (https://starfishmedical.com/blog/starting-medical-device-projects-on-the-right-foot/)
- starfishmedical.com (https://starfishmedical.com/blog/successful-medical-device-project-managers-skills/)
- Healthcare Content Marketing Strategies for Clinical Trials
- infomeddnews.com (https://infomeddnews.com/about-medical-device-news-magazine-2024/)
- starfishmedical.com (https://starfishmedical.com/blog/18-business-factors-that-determine-successful-medical-device-exits/)
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- growandconvert.com (https://www.growandconvert.com/content-marketing/healthcare-content-marketing/)
- pmlive.com (https://pmlive.com/intelligence/pharmas-content-conundrum/)
- globaldata.com (https://www.globaldata.com:443/store/report/medical-devices-industry-m-and-a-deals-by-theme-quarterly-analysis/)
- Challenges in Healthcare Content Marketing for Clinical Trials
- growandconvert.com (https://www.growandconvert.com/content-marketing/healthcare-content-marketing/)
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- smartinsights.com (https://www.smartinsights.com/digital-marketing-strategy/healthcare-marketing-strategy-trends/)
- contentmarketinginstitute.com (https://contentmarketinginstitute.com/articles/b2b-content-marketing-trends-research/)
- growandconvert.com (https://www.growandconvert.com/content-marketing/healthcare-content-marketing/)
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- Best Practices for Medical Device Clinical Trials: Regulations and Compliance
- fda.gov (https://www.fda.gov/news-events/press-announcements/fda-roundup-september-8-2023)
- fda.gov (https://www.fda.gov/news-events/press-announcements/fda-roundup-november-21-2023)
- medicaldevice-network.com (https://www.medicaldevice-network.com/sponsored/analysis-navigating-an-evolving-medical-device-regulatory-landscape/)
- scientia.global (https://www.scientia.global/mr-anthony-keyes-understanding-and-improving-clinical-trial-compliance/)



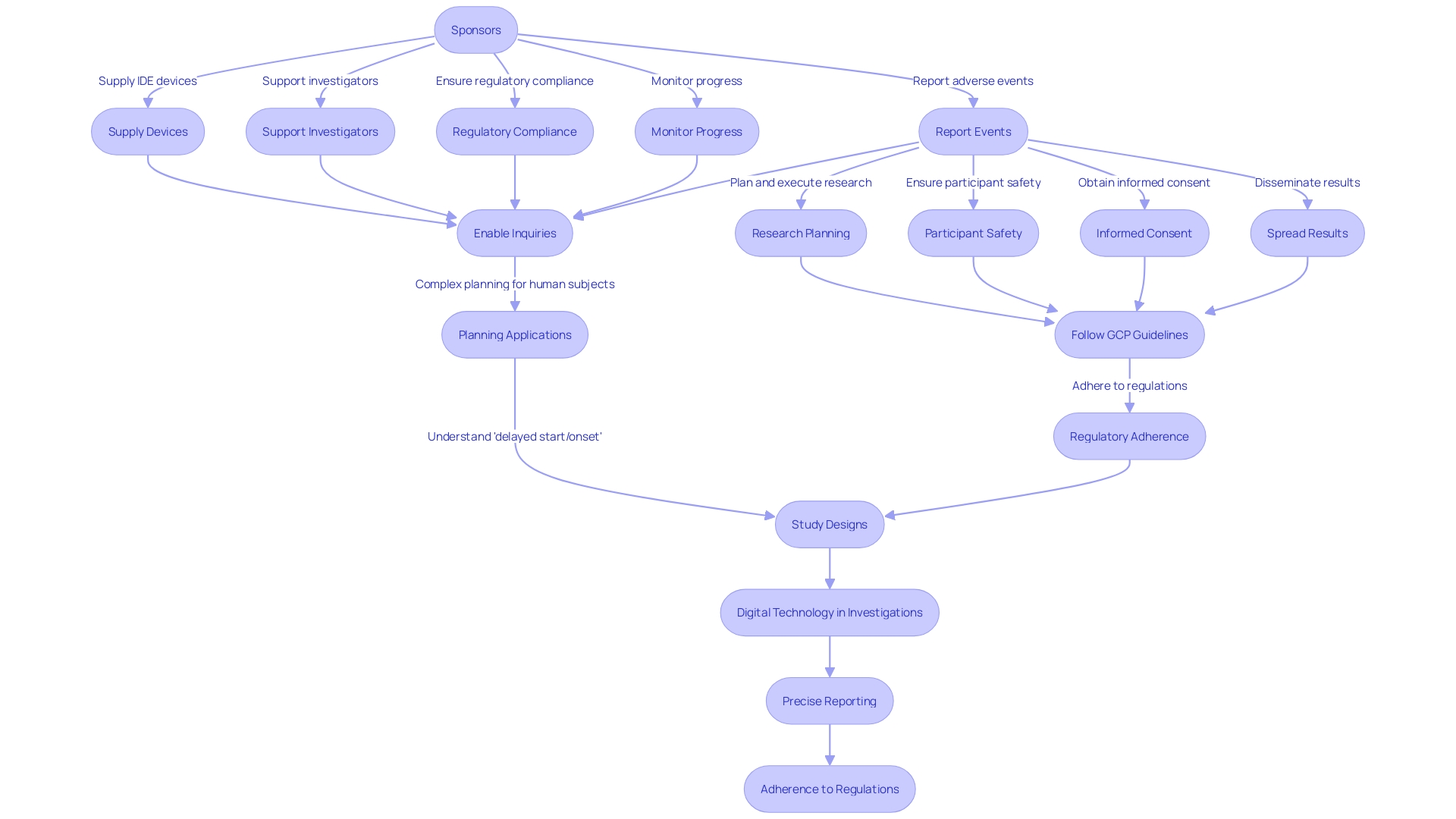
 for Clinical Investigations Flowchart: Good Clinical Practice (GCP) Guidelines for Clinical Investigations](https://tely.blob.core.windows.net/telyai/flowchart-good-clinical-practice-gcp-guidelines-for-clinical-investigations.jpg)