Introduction
In a sea of choices, how do MedTech and Biopharma companies in Brazil find the right Contract Research Organization (CRO) to ensure their clinical trials succeed? This guide will help you identify and partner with a CRO that meets your specific clinical trial needs, ensuring compliance with local regulations and optimizing patient recruitment strategies. Choosing poorly could jeopardize your trial’s success and compliance, so how do you navigate the complexities of CRO selection?
Identify Your Clinical Trial Needs and Objectives
Before selecting a Contract Research Organization (CRO), it’s essential to pinpoint your clinical study needs and objectives to ensure a successful partnership. Here’s how to do it:
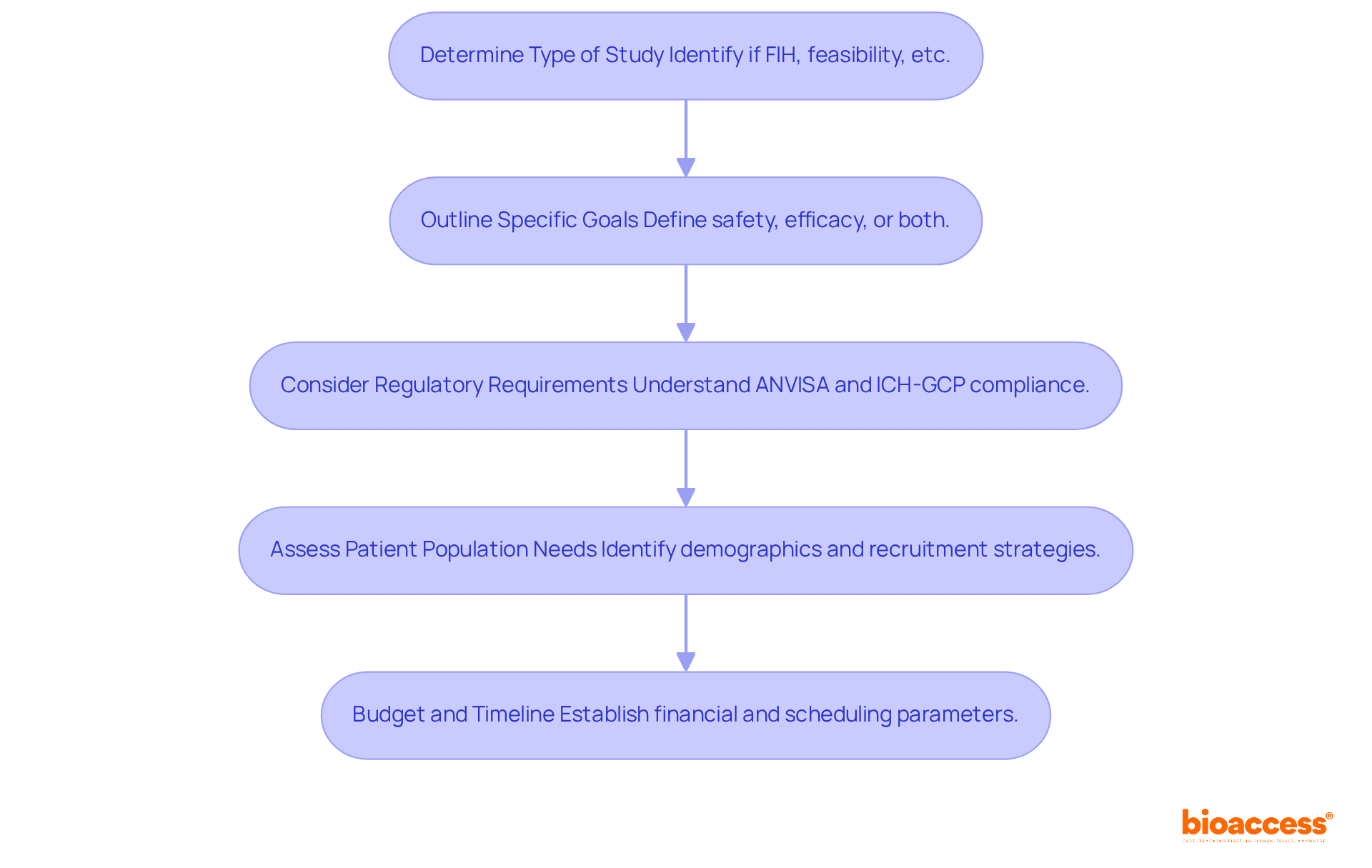
- Determine the Type of Study: Identify whether your study is a first-in-human (FIH) investigation, a feasibility assessment, or another category. This distinction will greatly impact the expertise needed from the CRO, especially in navigating the complexities of early-phase studies.
- Outline Specific Goals: Clearly define what you aim to achieve with the trial. Are you seeking safety data, efficacy data, or both? This clarity is vital for choosing a CRO that specializes in your focus area. Remember, around 40% to 50% of drug failures happen due to insufficient clinical efficacy.
- Consider the regulatory requirements in Brazil, particularly when collaborating with a contract research organization Brazil, including compliance with ANVISA and ICH-GCP guidelines. Acquaintance with these regulations will assist you in recognizing a contract research organization Brazil that is knowledgeable about local compliance, which is essential for successful study execution. Regulatory compliance is not merely a formality; it is a vital element that can greatly influence study outcomes. Engaging with bioaccess can provide you with essential insights into regulatory updates and market access strategies tailored for Latin America.
- Assess Patient Population Needs: Determine the demographics of the patient population you wish to study. This is crucial for CROs that have access to treatment-naive cohorts, which can expedite recruitment. In Latin America, patient recruitment can be significantly faster, with studies enrolling participants 50% quicker than in the US. By leveraging the expertise of bioaccess, you can enhance your recruitment strategies and capitalize on the region’s advantages.
- Budget and Timeline: By understanding these parameters, you can find a CRO that fits your budget and timeline. Typically, the cost of an FIH study ranges from USD 1.5 million to USD 5 million. Bioaccess can aid in optimizing your budget and schedule by offering insights into cost-effective strategies and efficient study management.
By clearly defining your requirements and goals, you establish a strong base for the following steps in choosing the appropriate CRO, ultimately improving the chances of study success and effective resource utilization. With bioaccess by your side, you can confidently navigate the complexities of clinical trials in Latin America, ensuring your study’s success and resource efficiency.
Assess the CRO’s Experience and Expertise in Brazil
Choosing the right contract research organization Brazil is crucial for the success of your clinical research. Once you have determined your research needs, the next step is to evaluate the experience and expertise of potential contract research organization Brazil. Follow these steps:
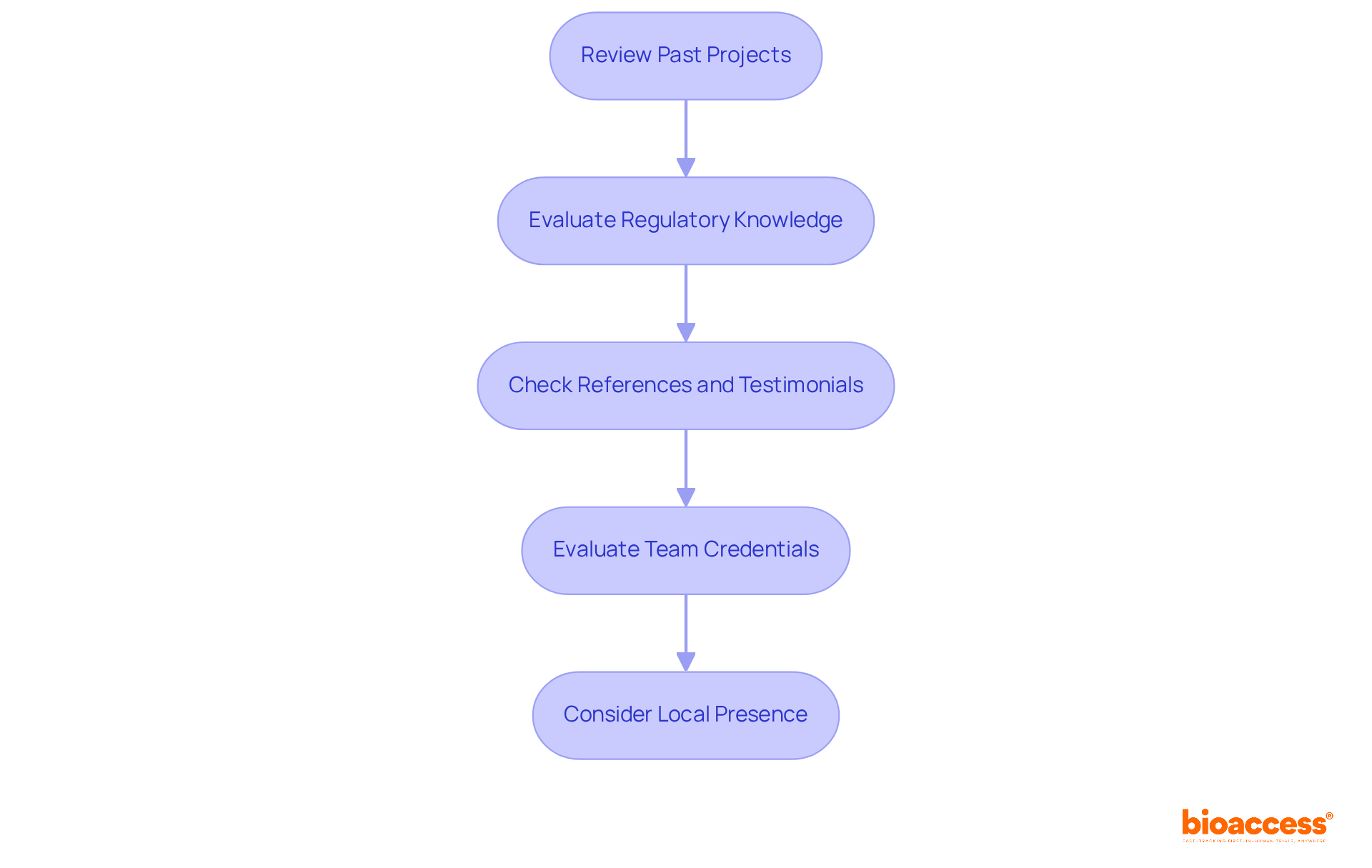
- Review Past Projects: Look for CROs that have successfully managed studies similar to yours. Concentrate on their portfolio for first-in-human (FIH) studies in your therapeutic area. This experience is essential for navigating the complexities of early-stage clinical research. bioaccess® has accelerated over 50 MedTech, Biopharma, and Radiopharma companies from concept to first-in-human studies. This success highlights their capability in navigating early-stage clinical research.
- Evaluate Regulatory Knowledge: Ensure the contract research organization Brazil has a strong understanding of Brazilian regulations, particularly those set by ANVISA. Familiarity with local compliance requirements, including ICH-GCP standards and specific approval timelines, is essential for smooth trial execution and timely approvals. Recent regulatory improvements have streamlined the approval processes, significantly reducing initiation timelines. bioaccess®’s extensive understanding of these regulations enables them to navigate the landscape effectively, accelerating the development process.
- Check References and Testimonials: Reach out to previous clients or read testimonials to gauge the CRO’s reliability and performance. Understanding their operational efficiency and responsiveness can assist you in evaluating their ability to meet your study’s demands. Founders and medical leaders have commended bioaccess® for their dedication to closing the divide between medical innovation and research potential in Latin America, emphasizing their success in navigating regulatory challenges.
- Evaluate Team Credentials: Examine the qualifications of the team overseeing your study. Seek certifications in research related to health and pertinent experience in your particular area, as a skilled team can greatly influence study outcomes. bioaccess® was established by physicians with extensive clinical expertise, ensuring that their team is well-prepared to manage the complexities of FIH studies.
- Consider Local Presence: Having a contract research organization Brazil with a local presence means they can navigate the regulatory landscape more smoothly and may have established relationships with local ethics committees and investigators. This local expertise can accelerate patient recruitment and improve study efficiency. bioaccess®’s established connections in Latin America enable quicker patient recruitment and smoother operational processes.
Have you thoroughly evaluated the CRO’s experience and knowledge, especially in FIH studies? Doing so ensures they are well-equipped to meet your research needs. With Brazil’s research trials market valued at around USD 1.58 billion in 2025 and expected to increase to USD 2.55 billion by 2035, choosing the appropriate CRO like bioaccess® is essential for capitalizing on the strategic benefits of speed, cost efficiency, and compliance pathways in this dynamic environment. In a rapidly evolving market, the right CRO can be the difference between success and stagnation in your clinical research efforts.
Compare Services, Costs, and Timelines of Potential CROs
Choosing the right CRO can feel overwhelming, given the myriad of options and the stakes involved in clinical research. After assessing the experience and expertise of potential CROs, the next step is to compare their services, costs, and timelines effectively. Here’s how to approach this:
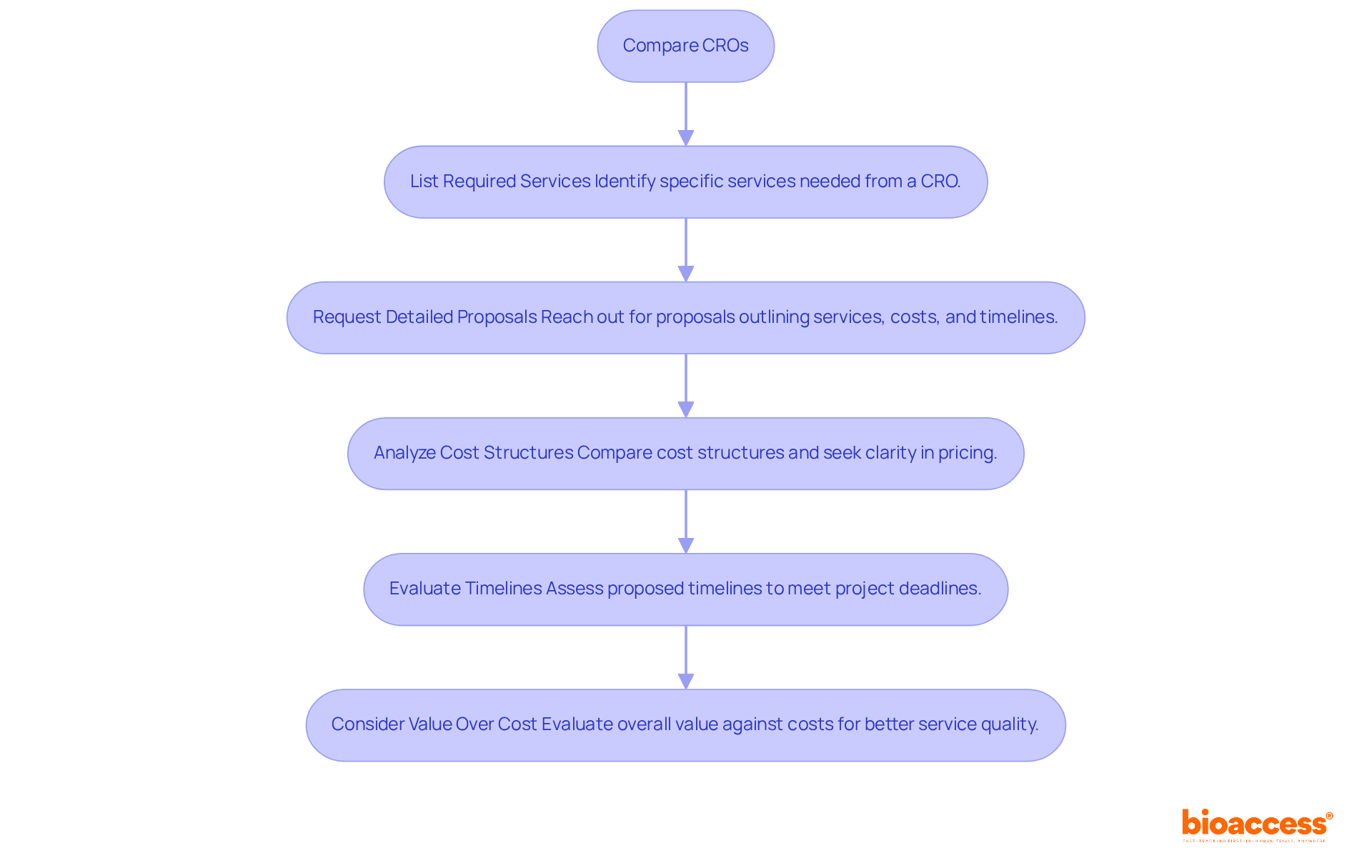
- List Required Services: Clearly identify the specific services you need from a CRO, such as patient recruitment, data management, regulatory submissions, and monitoring. Being clear about what you need makes it easier to communicate with potential CROs.
- Request Detailed Proposals: Reach out to shortlisted CROs and request detailed proposals that outline their services, associated costs, and timelines. Detailed proposals are crucial as they provide insight into each CRO’s capabilities and how they align with your project goals. As Patricio Ledesma, Head of Clinical Operations at Sofpromed CRO, emphasizes, “A comprehensive proposal is essential for understanding the nuances of each CRO’s offerings and ensuring alignment with your clinical objectives.”
- Analyze Cost Structures: Compare the cost structures of different CROs. Seek clarity in pricing and remain vigilant about any possible concealed charges that may emerge during the evaluation. Understanding the full financial picture is essential for budgeting. The contract research organization Brazil market in the pharmaceutical sector is projected to reach USD 2,531.9 million by 2033, indicating a competitive landscape that can influence pricing strategies.
- Evaluate Timelines: Assess the proposed timelines for each CRO. Ensure they can meet your project deadlines, especially if you are working under tight schedules. In Brazil, the contract research organization Brazil often benefits from streamlined regulatory processes governed by ANVISA, which can significantly reduce approval times, sometimes to as little as 4-8 weeks for ethics approval.
- Consider Value Over Cost: While cost is a critical factor, consider the overall value each CRO brings to the table. A somewhat increased expense may be warranted by enhanced service quality, quicker timelines, or access to treatment-naive patient groups, which can improve the study’s success.
Navigating the complexities of CRO services and costs can be daunting, especially when each offers unique strengths and weaknesses. By thoroughly assessing services, expenses, and schedules, you can make an informed choice that aligns with your research objectives. Ultimately, the right CRO can be the difference between a successful study and a missed opportunity in the competitive landscape of clinical research.
Establish Communication and Collaboration with Your CRO
In the high-stakes world of clinical research, effective communication with your contract research organization Brazil, like bioaccess, is not just beneficial – it’s essential for success, especially in first-in-human studies in Latin America. Here are steps to establish a strong partnership:
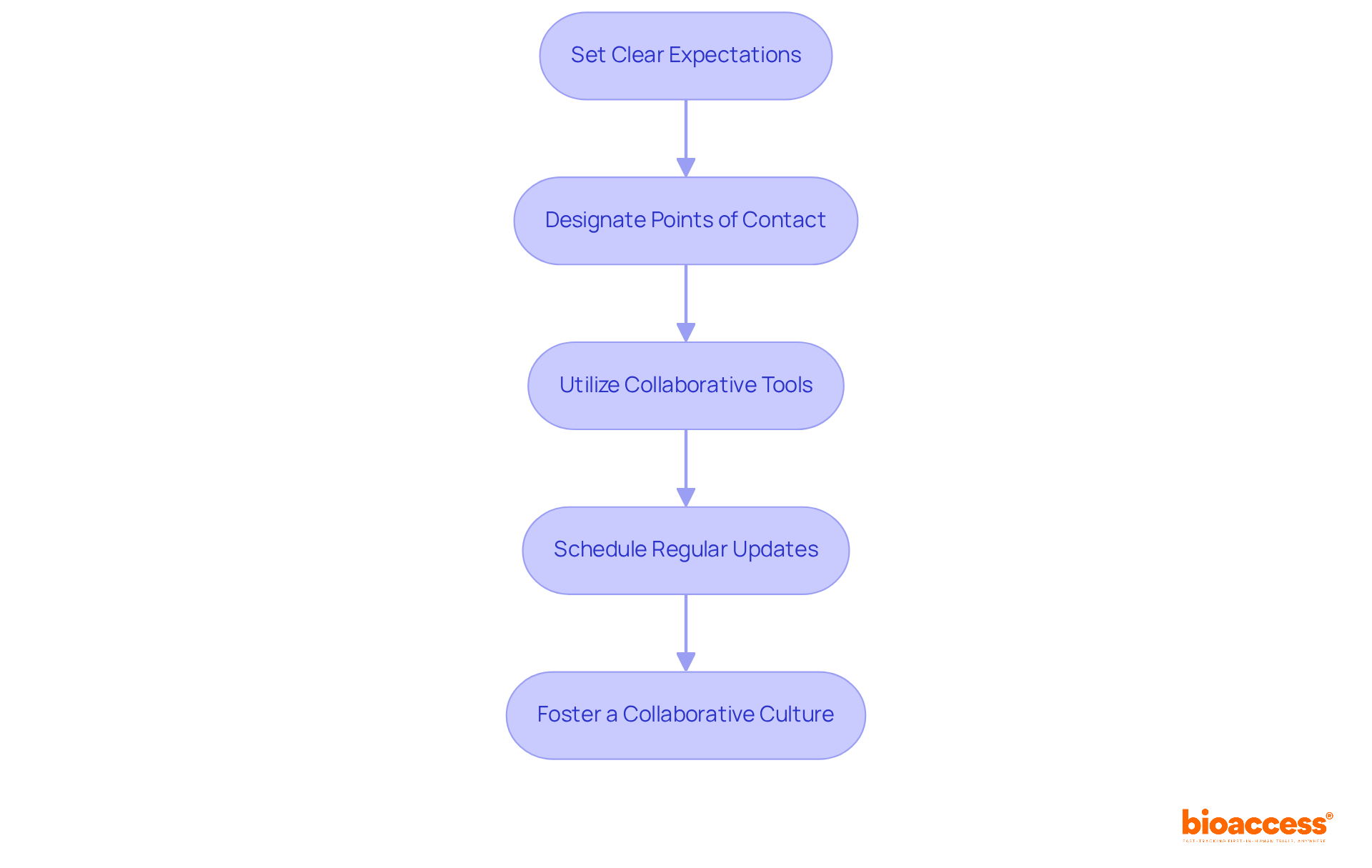
- Set Clear Expectations: Establishing clear expectations is vital; with drug development costs soaring, inefficiencies can lead to significant financial losses. Clearly communicate your expectations regarding timelines, deliverables, and communication frequency from the outset. In Latin America, utilizing a contract research organization Brazil can help speed up Phase I studies with 4-8 week ethics approvals, significantly reducing time to market.
- Designate Points of Contact: Establish primary points of contact on both sides to streamline communication. This ensures efficient information flow and minimizes the risk of miscommunication, which is particularly important in navigating the regulatory requirements set by authorities like INVIMA, ANVISA, and COFEPRIS.
- Utilize Collaborative Tools: Using collaborative tools can ease frustrations about technology integration, keeping all stakeholders informed about study progress. Leverage technology to facilitate communication through project management tools, shared documents, and regular video calls.
- Schedule Regular Updates: Plan consistent meetings to discuss progress, address challenges, and adjust timelines as necessary. This proactive approach assists in early detection of potential issues, improving overall management of the study. Bioaccess’s Global Trial Accelerators™ service can provide valuable insights during these discussions.
- Foster a Collaborative Culture: Encourage a culture of collaboration where team members feel comfortable sharing ideas and feedback. This openness can lead to innovative solutions and a more efficient process for trials. As one expert noted, “Treat us as partners and treat us with respect,” emphasizing the importance of mutual respect in these relationships.
By prioritizing collaboration, you position your clinical trials for success, ensuring timely results that can significantly impact patient care and innovation. In Latin America, where a contract research organization Brazil can efficiently navigate compliance pathways and expedite patient recruitment, these practices become even more critical for achieving timely and cost-effective results. Highlighting successful collaborations, such as those facilitated by bioaccess that have led to accelerated patient recruitment and streamlined regulatory approvals, can further illustrate the benefits of effective partnerships.
Conclusion
Navigating the complexities of clinical trials in Brazil demands a strategic choice of Contract Research Organization (CRO), especially for first-in-human studies. A strategic approach, grounded in a clear understanding of clinical needs and objectives, lays the groundwork for successful collaboration. Identifying the study type, outlining clear goals, and understanding regulatory requirements are essential steps for researchers to select a CRO that meets their specific needs.
Key insights discussed include:
- The importance of evaluating a CRO’s experience and expertise, particularly in navigating local regulations and patient recruitment strategies.
- It’s crucial to compare services, costs, and timelines among potential CROs to make an informed decision.
- Establishing effective communication and collaboration practices can significantly enhance the efficiency and success of clinical trials in Brazil.
In conclusion, leveraging Brazil’s advantages – such as expedited patient recruitment and streamlined regulatory processes – can lead to successful clinical research outcomes. By following the outlined steps and engaging with a specialized CRO like bioaccess, researchers can navigate the complexities of early-stage trials with confidence, ensuring that their studies are not only compliant but also positioned for success in a competitive landscape. Choosing the right CRO not only enhances trial success but also positions researchers to lead in innovation and patient care across Latin America.
Frequently Asked Questions
What should I identify before selecting a Contract Research Organization (CRO)?
Before selecting a CRO, you should identify your clinical study needs and objectives, including the type of study, specific goals, regulatory requirements, patient population needs, budget, and timeline.
How do I determine the type of study for my clinical trial?
You should identify whether your study is a first-in-human (FIH) investigation, a feasibility assessment, or another category, as this distinction impacts the expertise needed from the CRO.
Why is it important to outline specific goals for the trial?
Clearly defining your goals, such as seeking safety data, efficacy data, or both, is vital for choosing a CRO that specializes in your focus area, as around 40% to 50% of drug failures occur due to insufficient clinical efficacy.
What regulatory requirements should I consider when working with a CRO in Brazil?
You should consider compliance with ANVISA and ICH-GCP guidelines, as familiarity with these regulations is essential for recognizing a CRO knowledgeable about local compliance, which influences study outcomes.
How can understanding patient population needs impact my study?
Determining the demographics of your desired patient population is crucial, as CROs with access to treatment-naive cohorts can expedite recruitment, especially in Latin America where studies can enroll participants 50% faster than in the US.
What are the typical costs and timelines for a first-in-human (FIH) study?
The cost of an FIH study typically ranges from USD 1.5 million to USD 5 million. Understanding your budget and timeline helps in finding a CRO that fits your needs.
How can bioaccess assist in optimizing my clinical trial process?
Bioaccess can provide insights into regulatory updates, market access strategies, cost-effective strategies, and efficient study management, helping you navigate the complexities of clinical trials in Latin America effectively.
List of Sources
- Identify Your Clinical Trial Needs and Objectives
- 90% of drugs fail clinical trials (https://asbmb.org/asbmb-today/opinions/031222/90-of-drugs-fail-clinical-trials)
- First-in-Human Clinical Trial Market Size, Growth, Demand 2030 (https://reedintelligence.com/market-analysis/first-in-human-clinical-trial-market)
- Why do 90% of clinical trials fail? (https://synapse.patsnap.com/blog/why-do-90-of-clinical-trials-fail)
- Clinical Trial Success Rates: How Many Drugs Make It to Market? (Latest Approval Stats) (https://patentpc.com/blog/clinical-trial-success-rates-how-many-drugs-make-it-to-market-latest-approval-stats)
- Assess the CRO’s Experience and Expertise in Brazil
- Brazil Clinical Trials Market Size & Future Growth 2035 (https://expertmarketresearch.com/reports/brazil-clinical-trials-market)
- Unlocking Brazil’s Clinical Trial Opportunity: A Strategic Roadmap for Healthcare Leaders (https://lek.com/insights/life-sciences-pharma/unlocking-brazils-clinical-trial-opportunity-strategic-roadmap)
- CLINICAL TRIALS SCENARIO IN BRAZIL- STUDY AND SPONSOR PROFILES (https://ispor.org/heor-resources/presentations-database/presentation/ispor-19th-annual-european-congress/clinical-trials-scenario-in-brazil-study-and-sponsor-profiles)
- Brazil Clinical Trials Market Size, Share | Analysis Report [2032] (https://fortunebusinessinsights.com/brazil-clinical-trials-market-112310)
- Compare Services, Costs, and Timelines of Potential CROs
- Brazil Pharmaceutical CRO Market Size & Outlook, 2033 (https://grandviewresearch.com/horizon/outlook/pharmaceutical-cro-market/brazil)
- Brazil In Vivo CRO Market Size & Outlook, 2030 (https://grandviewresearch.com/horizon/outlook/in-vivo-cro-market/brazil)
- Pharmaceutical CRO Market Size & Share Analysis Report 2032 (https://snsinsider.com/reports/pharmaceutical-cro-market-5797)
- Brazil Clinical Trials Market Size, Share | Analysis Report [2032] (https://fortunebusinessinsights.com/brazil-clinical-trials-market-112310)
- The Ultimate Guide to Clinical Trial Costs in 2025 (https://sofpromed.com/ultimate-guide-clinical-trial-costs)
- Establish Communication and Collaboration with Your CRO
- Clinical Trial Success Rates: How Many Drugs Make It to Market? (Latest Approval Stats) (https://patentpc.com/blog/clinical-trial-success-rates-how-many-drugs-make-it-to-market-latest-approval-stats)
- Back to (Communication) Basics: Reducing Site Burden and Establishing a Sponsor/CRO-of-Choice Relationship with Investigative Sites (https://globalforum.diaglobal.org/issue/june-2024/back-to-communication-basics-reducing-site-burden-and-establishing-a-sponsor-cro-of-choice-relationship-with-investigative-sites)
- Clinical Trial Success Rates: What Makes Some Companies Stand Out? | Ozmosi (https://ozmosi.com/clinical-trial-success-rates)

Leave a Reply