Introduction
The Case Report Form (CRF) stands as the cornerstone of clinical research, diligently gathering vital data from study participants while upholding regulatory compliance. Grasping CRF abbreviations transcends mere terminology; it is essential for fostering effective communication and averting costly mistakes in medical studies. Given the intricate nature of these abbreviations and their implications, how can researchers guarantee both clarity and efficiency in their documentation practices?
Define CRF and Its Importance in Clinical Research
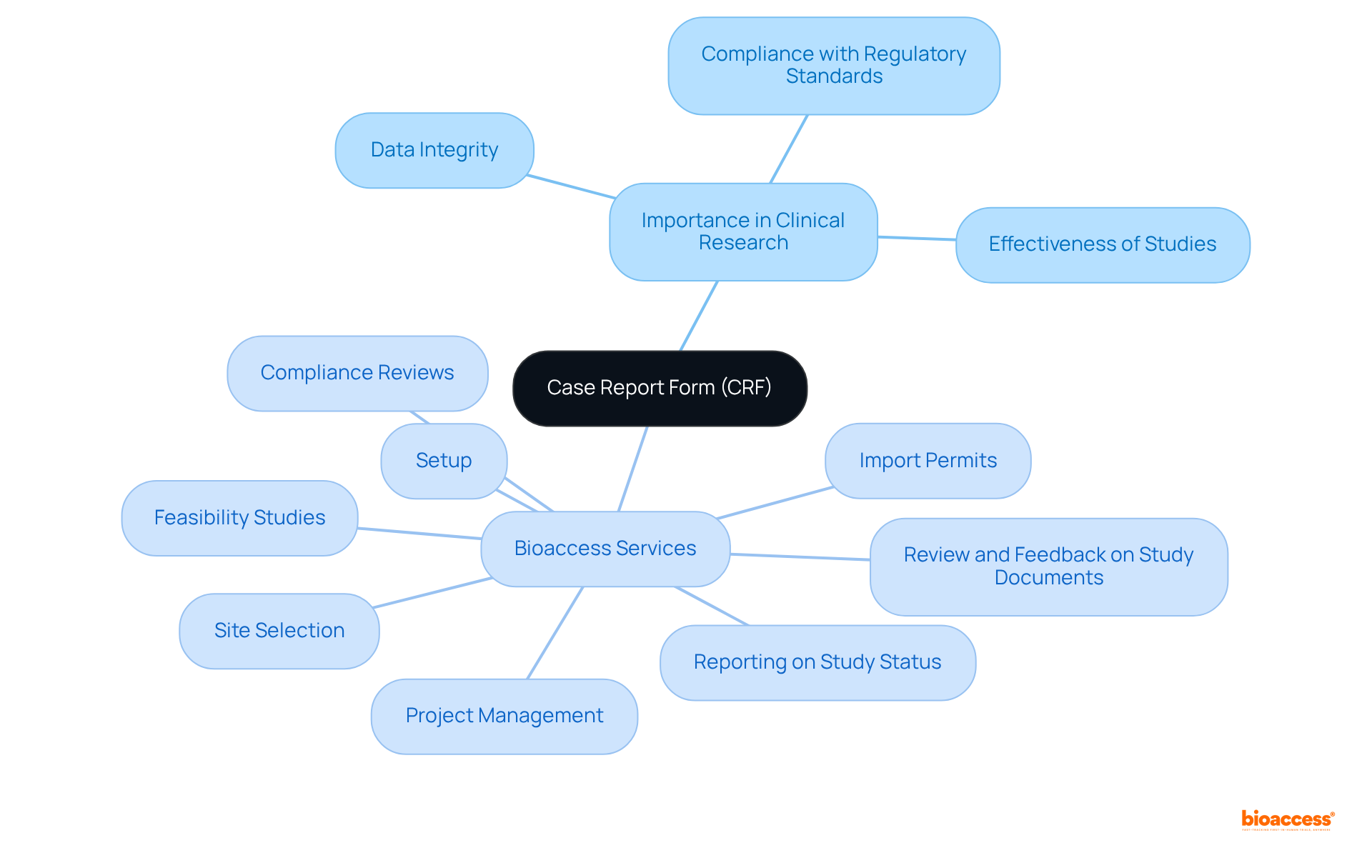
The Case Report Form (CRF), known in by the , is a vital document designed to collect data from study participants, ensuring it encompasses all protocol-required information. The CRFs, often referred to by the , are instrumental in maintaining data integrity and compliance with regulatory standards, thereby facilitating the evaluation of safety and effectiveness in clinical studies. They may be paper-based or electronic (eCRF), and their design must align with the research protocol to guarantee accurate data collection. A well-structured significantly impacts both the quality of data collected and the overall effectiveness of the study.
In the realm of , bioaccess offers the following services:
- Feasibility studies
- Site selection
- Compliance reviews
- Setup
- Import permits
- Project management
- Reporting on study status
- Review and feedback on study documents
These services are essential for the effective implementation of CRFs and the success of research initiatives.
List Key CRF Abbreviations and Their Meanings
-
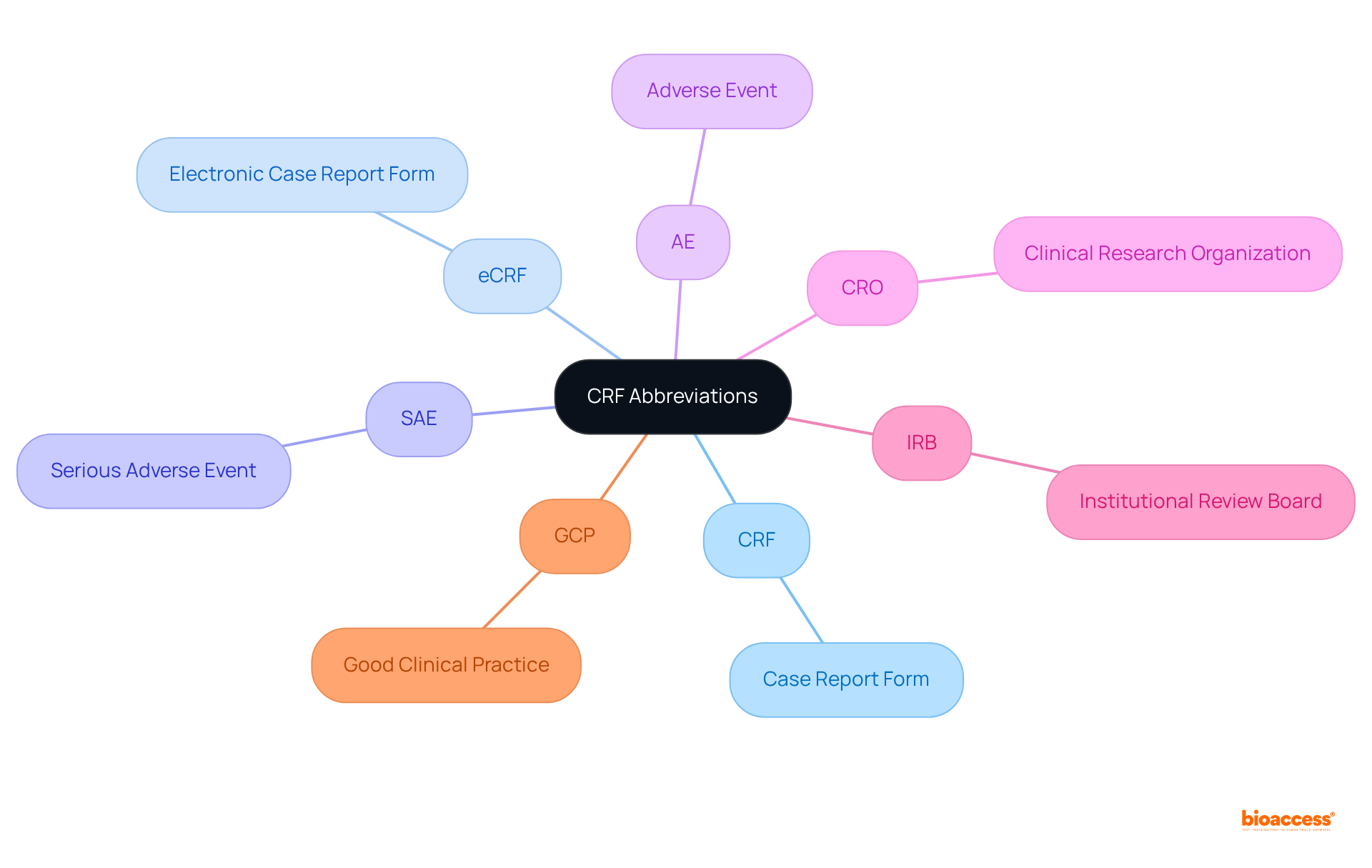
Compile a list of common CRF abbreviations, including:
- CRF:
- eCRF:
- SAE:
- AE: Adverse Event
- CRO:
- IRB:
- GCP:
-
Provide concise definitions for each abbreviation to clarify their meanings and importance in medical research. Understanding these terms is essential for within the field.
-
It is crucial for readers to become acquainted with these terms, as doing so will significantly enhance their communication and documentation abilities in research studies, ultimately contributing to the success of clinical research initiatives.
Apply CRF Abbreviations in Documentation and Communication
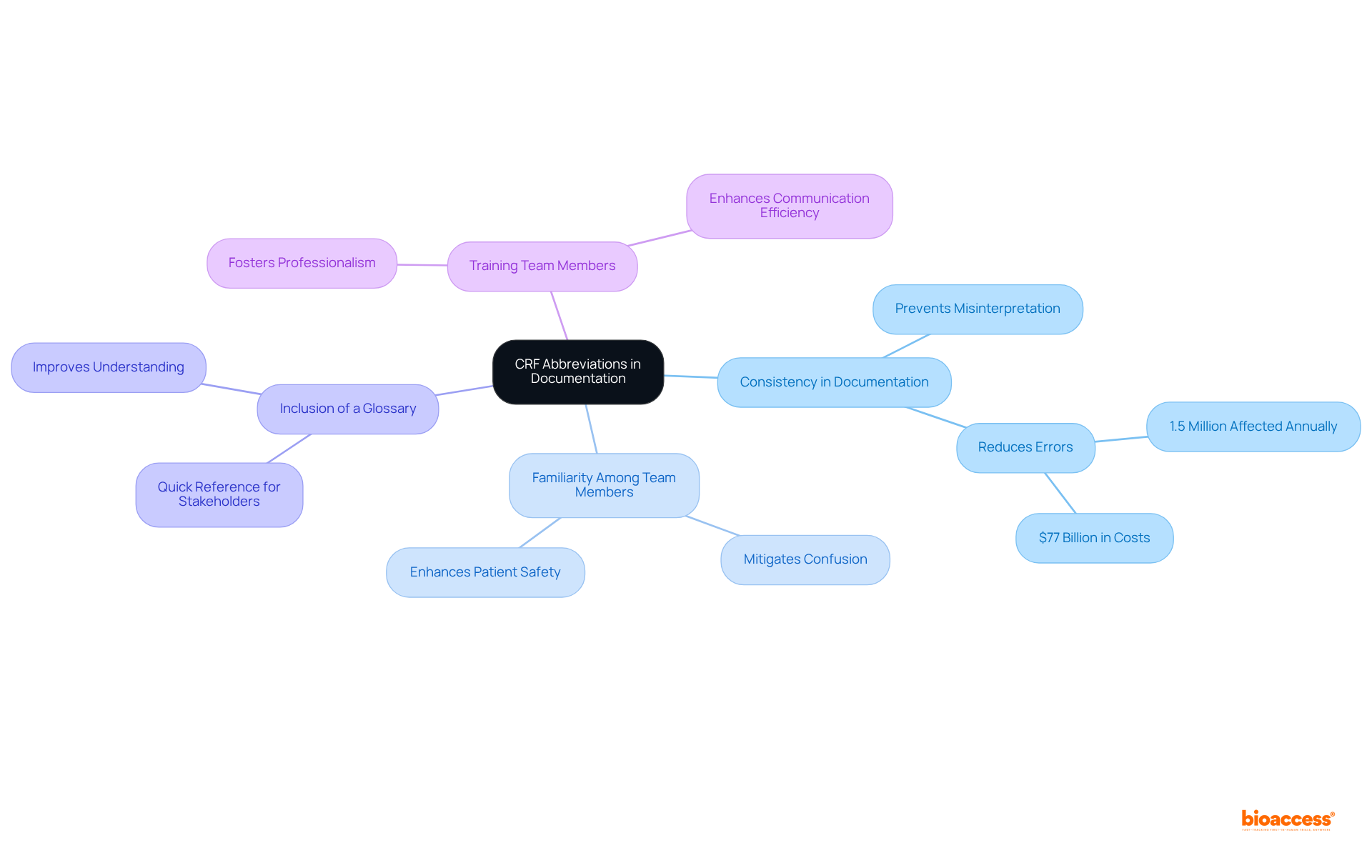
- The consistent use of the across all documentation—such as study protocols, informed consent forms, and reports—is crucial for maintaining clarity and preventing misinterpretation. Miscommunication in medical studies can lead to , with and resulting in approximately $77 billion annually in morbidity and mortality.
- Familiarity with the among all team members is essential to , which can have dire consequences for and the overall success of research trials.
- Including a in study documents acts as a quick reference for all stakeholders, enhancing understanding and reducing the risk of miscommunication. Expert opinions underscore that clear communication is vital in medical research to avert errors and ensure .
- Training team members on the importance of , such as the , not only fosters professionalism but also enhances communication efficiency, ultimately contributing to the success of initiatives and .
Designing Effective CRFs
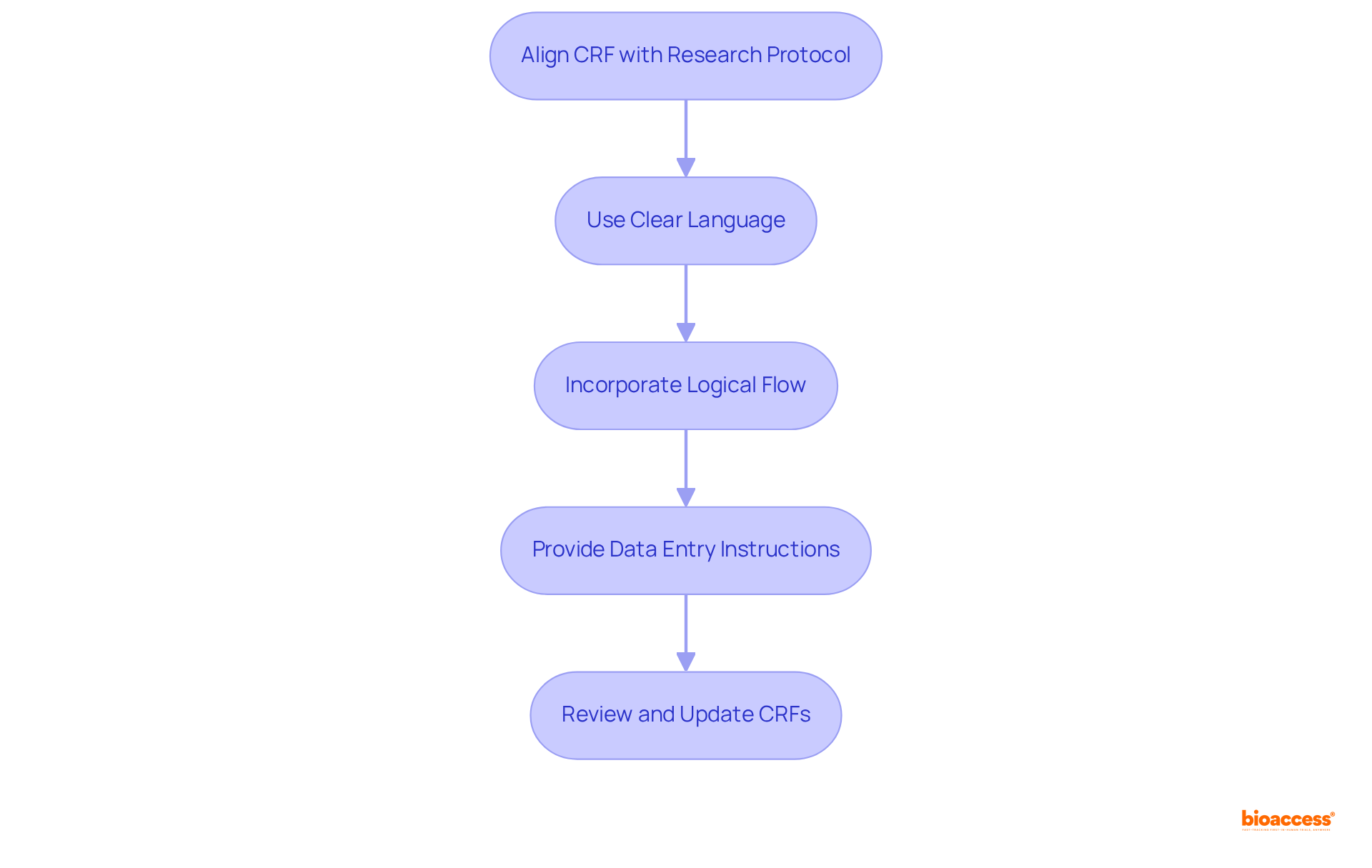
- Ensure alignment of the with the , capturing all necessary data points without redundancy to uphold the integrity of the study.
- Utilize in questions to eliminate ambiguity, thereby facilitating accurate responses from participants and enhancing .
- Incorporate a logical flow and grouping of related questions to significantly improve the usability of the CRF, making it intuitive for users.
- Provide and handling to guide users in completing the CRF accurately and efficiently.
- Regularly based on user feedback and regulatory changes, ensuring their effectiveness and relevance in the dynamic landscape of .
Implementing CRF Training for Staff
To effectively implement CRF abbreviation medical training for staff, it is essential to develop a comprehensive program that emphasizes the importance of the , its design, and . This program should integrate insights from our extensive service capabilities, including:
- Site selection
- Setup
- Reporting
to illustrate how these elements influence CRF effectiveness. must be conducted to keep staff updated on and any changes in regulations or protocols, ensuring compliance with country requirements, including those set by INVIMA. Utilizing case studies and real-world examples will demonstrate the impact of appropriate CRF usage on study results, particularly in the context of setup and project management. Furthermore, encouraging feedback from staff on the training program will facilitate continuous improvement, enhancing the effectiveness and relevance of our .
Conclusion
The significance of the Case Report Form (CRF) in clinical research is paramount. As a foundational tool for data collection, the CRF ensures the integrity and compliance of research protocols, ultimately influencing the success of clinical trials. It is essential for researchers to understand and effectively utilize CRF abbreviations, as this fosters clearer communication and enhances the quality of documentation throughout the study process.
Key insights from this article underscore the necessity of familiarizing all team members with common CRF abbreviations, such as eCRF and SAE, to minimize miscommunication and potential errors that could jeopardize patient safety. The design of CRFs must align with research protocols, employing clear language and a logical flow of questions to optimize data collection. Regular training and updates for staff on CRF usage are crucial for maintaining the relevance and effectiveness of these forms in the ever-evolving landscape of clinical research.
In conclusion, the role of CRFs and their associated abbreviations extends beyond mere documentation; they are vital for ensuring the accuracy, safety, and success of clinical trials. By prioritizing effective communication and training within research teams, stakeholders can significantly enhance the quality of clinical research outcomes. Embracing these best practices will not only improve individual studies but also contribute to the broader field of medical research, ultimately benefiting patient care and safety.
Frequently Asked Questions
What is a Case Report Form (CRF) and why is it important in clinical research?
A Case Report Form (CRF) is a vital document used to collect data from study participants in clinical research. It ensures that all protocol-required information is included, maintaining data integrity and compliance with regulatory standards. This facilitates the evaluation of safety and effectiveness in clinical studies.
What types of CRFs are there?
There are two main types of CRFs: paper-based CRFs and electronic CRFs (eCRF). Both types are designed to align with the research protocol to ensure accurate data collection.
How does the design of a CRF affect a clinical study?
A well-structured CRF significantly impacts the quality of data collected and the overall effectiveness of the study. It is essential for ensuring that all necessary information is gathered accurately.
What services does bioaccess provide in relation to CRFs?
Bioaccess offers several services essential for the effective implementation of CRFs, including feasibility studies, site selection, compliance reviews, setup, import permits, project management, reporting on study status, and review and feedback on study documents.
What are some common CRF abbreviations and their meanings?
Common CRF abbreviations include: – CRF: Case Report Form – eCRF: Electronic Case Report Form – SAE: Serious Adverse Event – AE: Adverse Event – CRO: Clinical Research Organization – IRB: Institutional Review Board – GCP: Good Clinical Practice
Why is it important to understand CRF abbreviations in medical research?
Understanding CRF abbreviations is crucial for effective communication within the field of medical research. Familiarity with these terms enhances communication and documentation abilities in research studies, contributing to the success of clinical research initiatives.
List of Sources
- Apply CRF Abbreviations in Documentation and Communication
- Medication Errors | AMCP.org (https://amcp.org/concepts-managed-care-pharmacy/medication-errors)

Leave a Reply