Introduction
In the dynamic landscape of clinical trials, Chile emerges as a prime location for early feasibility studies and first-in-human trials, offering a streamlined regulatory environment that can significantly accelerate research timelines. Understanding the intricacies of the Chilean clinical trial landscape allows companies to capitalize on rapid approval processes. They can also benefit from cost efficiencies and diverse patient recruitment opportunities. Yet, the complexity of local regulations can hinder progress.
So, how can sponsors strike the right balance between leveraging Chile’s advantages and ensuring compliance?
Understand the Clinical Trial Landscape in Chile
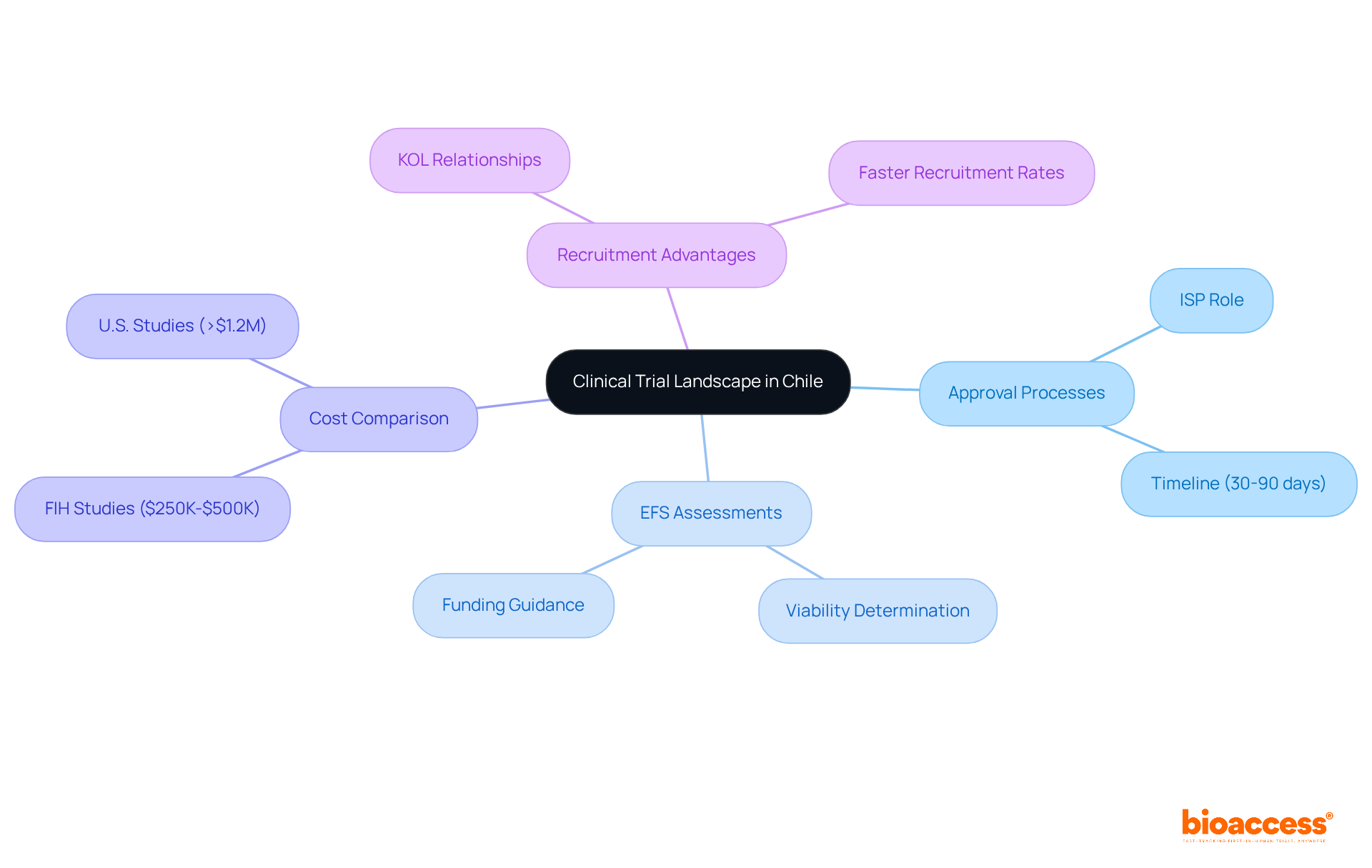
In the competitive landscape of clinical trials, the efs clinical trial chile emerges as a strategic choice for early feasibility studies and first-in-human trials. The country benefits from a streamlined oversight environment, with approval processes typically ranging from 30 to 90 days – significantly faster than many other regions. The Instituto de Salud Pública (ISP) plays a crucial role in overseeing this efficient governance framework, essential for MedTech and Biopharma companies aiming to expedite their clinical research.
Have you considered how EFS assessments can determine the viability of medical devices in small participant groups? These assessments provide vital information that guides further development and submission processes, making them indispensable for startups seeking funding and market access. The diverse patient demographic in the region supports rapid recruitment, often achieving rates that are double those of conventional approaches, thanks to established connections with key opinion leaders (KOLs).
By blending U.S. regulatory standards with local practices, Chile offers a unique path that reduces costs and timelines compared to trials in the U.S. or Europe. For instance, the overall program expense for FIH studies in the region ranges from $250K to $500K, while comparable studies in the U.S. can exceed $1.2 million. This cost-effectiveness, along with the ability to secure ethical approvals in as few as 15 days, positions the country as an attractive option for initial-stage research studies.
Understanding the local medical study landscape, including compliance requirements and execution benefits, is crucial for navigating the complexities of EFS clinical trial Chile and FIH research. By leveraging the strengths of the Chilean system, companies can accelerate their development processes and enhance their chances of achieving successful outcomes. With bioaccess®’s tailored solutions, startups can maximize their investment in R&D and expedite their path to market. Failing to leverage Chile’s clinical trial advantages could hinder your progress in medical innovation.
Identify Regulatory Requirements and Key Agencies
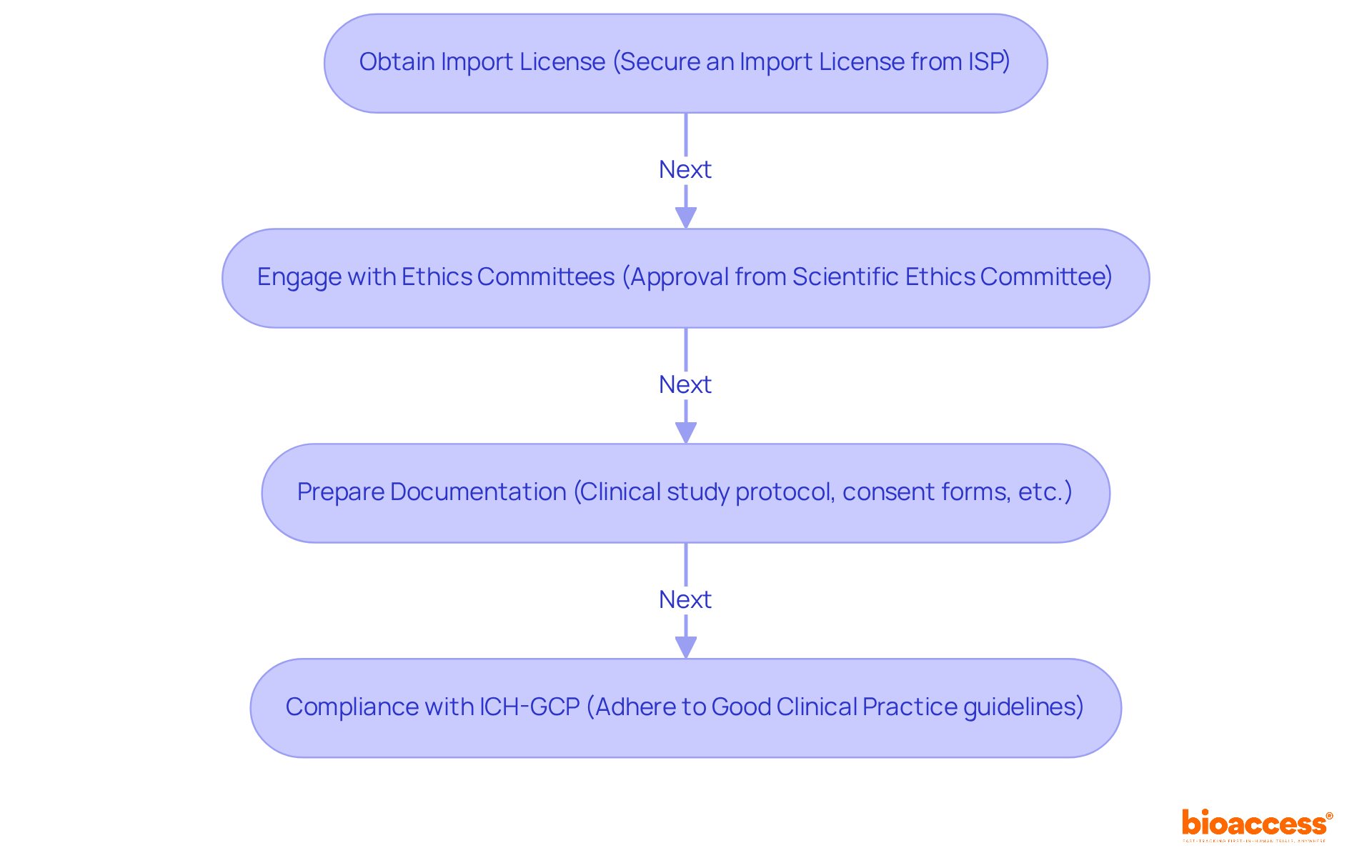
Successfully conducting medical research in Chile requires a thorough understanding of the regulatory landscape, which can be challenging for many sponsors. To effectively carry out medical research, it is crucial to navigate the regulatory environment established by key organizations, mainly the Instituto de Salud Pública (ISP). This agency plays a crucial role in approving medical studies and ensuring they meet national health regulations. Here are the critical steps to follow:
- Obtain an Import License: Sponsors must first secure an Import License from the ISP, allowing the importation of medical devices or pharmaceuticals intended for use in medical settings.
- Engage with Ethics Committees: Approval from an accredited Scientific Ethics Committee (CEC) is mandatory. This committee assesses the study protocol to ensure ethical standards are maintained, typically within a timeframe of 4 to 8 weeks.
- Prepare Documentation: Essential documents include the clinical study protocol, informed consent forms, and any relevant data that supports the study’s feasibility and safety.
- Compliance with ICH-GCP: All studies must adhere to the International Council for Harmonisation’s Good Clinical Practice (ICH-GCP) guidelines, which protect the rights, safety, and well-being of participants.
Understanding these compliance standards is crucial. Effectively interacting with the relevant agencies ensures a seamless research process in the region. Navigating the regulatory landscape can be daunting for sponsors, often leading to delays and confusion. The ISP’s streamlined approval process, which has decreased bureaucratic delays by over 30%, enhances the research environment, making this location an appealing choice for early feasibility studies. By leveraging bioaccess®’s expertise, sponsors can streamline their research efforts and avoid common pitfalls. As Julio G. Martinez-Clark observes, navigating the regulatory framework in the region is essential for success, especially in the evolving environment of medical research. Embracing collaboration with experts like bioaccess® can be the key to overcoming these challenges and achieving research success.
Prepare and Submit Required Documentation
In Chile, the preparation and submission of documentation are not just procedural steps; they are pivotal to the success of clinical research approvals. Here are the key steps to ensure compliance and facilitate a smooth submission:
-
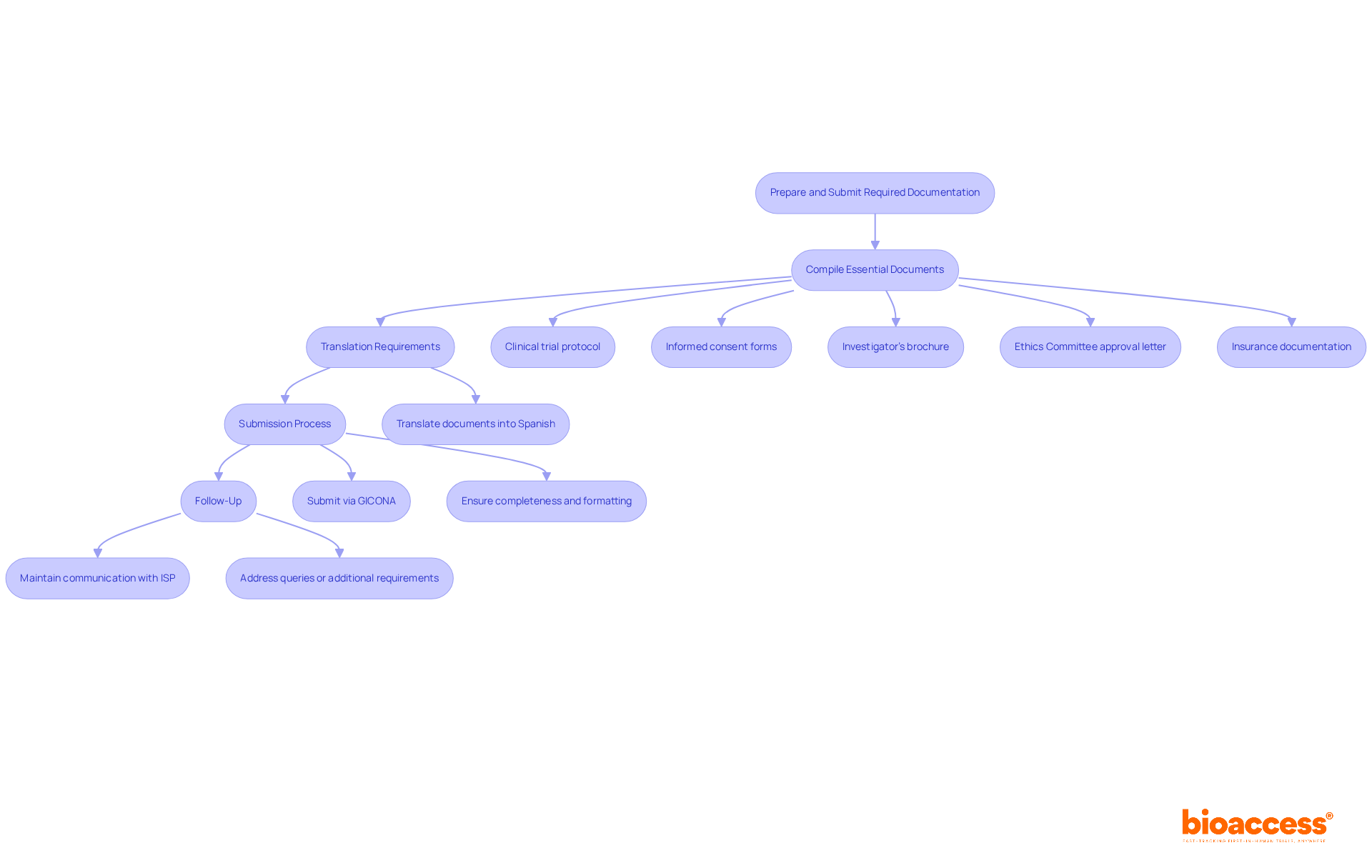
Compile Essential Documents: The following documents are typically required:
- Clinical trial protocol detailing the study design, objectives, and methodology.
- Informed consent forms that comply with local regulations.
- Investigator’s brochure containing information about the investigational product.
- Ethics Committee approval letter.
- Insurance documentation covering trial participants.
-
Translation Requirements: All documents must be translated into Spanish, as this is the official language of the country. Accurate and culturally appropriate translations are crucial to avoid misunderstandings during the review process. This requirement is underscored by the necessity for all documentation to be submitted in a format that meets the Instituto de Salud Pública (ISP) standards.
-
Submission Process: Submit the compiled documentation to the ISP through their online platform, GICONA. Ensure that all documents are complete and formatted according to ISP guidelines to minimize the risk of delays. Typically, the ISP reviews applications in about 30 business days, but if your submission isn’t complete, it could take anywhere from 30 to 60 days. Significantly, the average approval duration for research studies in the region can exceed six months, making comprehensive preparation crucial.
-
Follow-Up: After submission, maintain communication with the ISP to address any queries or additional requirements they may have. This proactive approach can help expedite the review process. Engaging with the ISP can also clarify any specific documentation needs or adjustments that may be required, enhancing the likelihood of a timely approval. Additionally, completing site agreements and investigator training typically takes 2-3 weeks, and obtaining an import permit through Chilean customs can take 2-4 weeks, which are important considerations for planning your timelines.
With meticulous preparation and submission of the necessary documentation, sponsors can significantly enhance their chances of securing prompt regulatory approval for their studies in this region, utilizing the area’s effective regulatory framework and dedication to ethical research. Moreover, with bioaccess®’s proficiency in managing these processes, sponsors can gain from expedited timelines and economical solutions, transforming the regulatory landscape into a strategic advantage for their studies in Latin America.
Navigate the Review Process and Address Challenges
Navigating the review process for the efs clinical trial in Chile can be fraught with challenges that demand strategic foresight and meticulous planning. Here are strategies to effectively manage the review process and address potential obstacles:
-
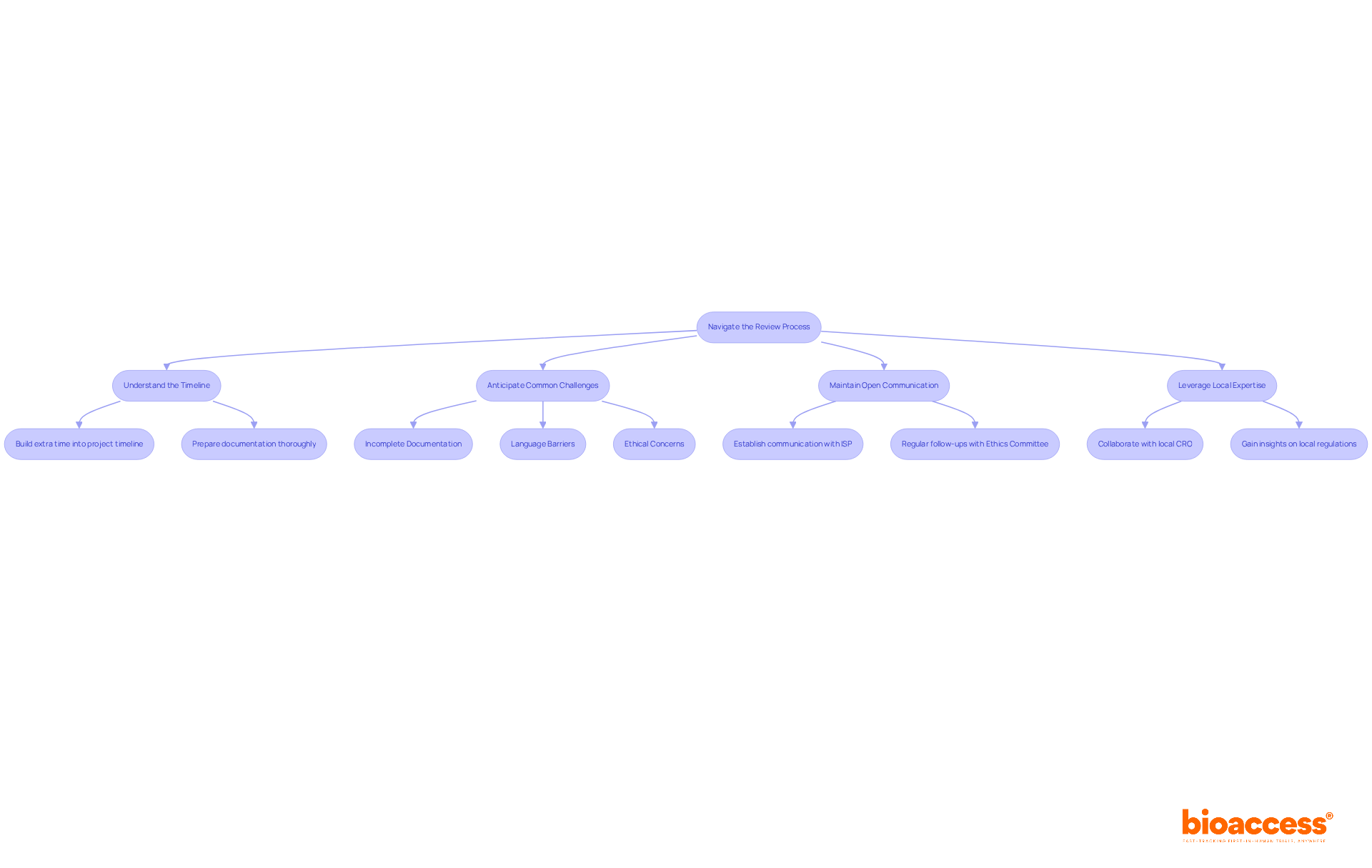
Understand the Timeline: The review process typically takes about 30 days from the date of submission. However, delays can significantly impact your project timeline if documentation is not thoroughly prepared. To mitigate this risk, build extra time into your project timeline to accommodate potential delays.
-
Anticipate Common Challenges: What are the common issues you might face? Here are a few to consider:
- Incomplete Documentation: Ensure all required documents are submitted in full to avoid unnecessary delays.
- Language Barriers: Accurate and culturally relevant translations are crucial for effective communication with oversight bodies.
- Ethical Concerns: Address any ethical considerations raised by the Ethics Committee promptly to maintain compliance and trust.
-
Maintain Open Communication: Establish a line of communication with the Instituto de Salud Pública (ISP) and the Ethics Committee. Regular follow-ups can help clarify any questions they may have and demonstrate your commitment to compliance.
-
Leverage Local Expertise: Collaborating with a local CRO like bioaccess® can greatly improve your comprehension of the Chilean compliance environment. Their expertise in navigating the complexities of local regulations and ethical standards can provide valuable insights and support throughout the review process.
By proactively tackling these challenges and maintaining clear communication with oversight bodies, sponsors can navigate the review process more efficiently. Failure to navigate these challenges can lead to prolonged timelines and increased costs. By embracing these strategies, you not only enhance compliance but also position your efs clinical trial in Chile for timely regulatory approval, which is a critical factor in the competitive landscape of clinical research.
Conclusion
The evolving landscape of EFS clinical trials in Chile offers MedTech and Biopharma companies a strategic advantage. The efficient approval processes and cost-effective strategies in this region expedite research timelines. They also enhance the potential for successful outcomes in early-stage studies.
Key insights from this guide highlight the importance of:
- Understanding the local regulatory landscape
- Engaging with key agencies like the Instituto de Salud Pública (ISP)
- Meticulously preparing the necessary documentation
Navigating compliance can be daunting, but local expertise can ease this burden. By adhering to compliance requirements such as ICH-GCP, sponsors can significantly reduce the risks associated with clinical trial delays and challenges.
With the increasing demand for innovation, the time to act is now. Companies should take proactive steps to harness these benefits, ensuring they are well-equipped to navigate the regulatory pathways and capitalize on the rapid patient recruitment capabilities. Embracing the strategic advantages of the Chilean clinical trial landscape will not only facilitate timely approvals but also pave the way for groundbreaking advancements in medical research. Seizing these opportunities now will not only enhance research outcomes but also position companies at the forefront of medical innovation.
Frequently Asked Questions
What is the significance of Chile in the clinical trial landscape?
Chile is recognized as a strategic choice for early feasibility studies and first-in-human trials due to its streamlined oversight environment, with approval processes typically ranging from 30 to 90 days, which is significantly faster than many other regions.
Who oversees the clinical trial processes in Chile?
The Instituto de Salud Pública (ISP) plays a crucial role in overseeing the governance framework for clinical trials in Chile, ensuring efficient regulatory processes for MedTech and Biopharma companies.
How do EFS assessments benefit medical device startups in Chile?
EFS assessments provide vital information that determines the viability of medical devices in small participant groups, guiding further development and submission processes, which is essential for startups seeking funding and market access.
What advantages does Chile offer for patient recruitment in clinical trials?
The diverse patient demographic in Chile supports rapid recruitment, often achieving rates that are double those of conventional approaches, aided by established connections with key opinion leaders (KOLs).
How does the cost of clinical trials in Chile compare to those in the U.S.?
The overall program expense for first-in-human studies in Chile ranges from $250K to $500K, while comparable studies in the U.S. can exceed $1.2 million, making Chile a cost-effective option.
What is the timeline for securing ethical approvals in Chile?
Ethical approvals in Chile can be secured in as few as 15 days, which enhances the speed of initiating clinical trials.
Why is understanding the local medical study landscape important for conducting trials in Chile?
Understanding the local landscape, including compliance requirements and execution benefits, is crucial for navigating the complexities of early feasibility studies (EFS) and first-in-human (FIH) research, ultimately enhancing the chances of successful outcomes.
How can companies leverage Chile’s clinical trial advantages?
By leveraging the strengths of the Chilean system, companies can accelerate their development processes and maximize their investment in R&D, expediting their path to market.
What could happen if companies fail to utilize Chile’s clinical trial benefits?
Failing to leverage the advantages of Chile’s clinical trial landscape could hinder a company’s progress in medical innovation, potentially delaying their development and market entry.
List of Sources
- Understand the Clinical Trial Landscape in Chile
- First-in-Human Medical Device Trial in Chile | bioaccess® (https://bioaccessla.com/blog/first-in-human-medical-device-trial-chile)
- Early Feasibility Studies in Latin America (https://greenlight.guru/blog/early-feasibility-studies-in-latin-america)
- Chile First-in-Human Clinical Trial Market Size, Share, Growth & Trends 2025–2033 (https://reedintelligence.com/market-analysis/chile-first-in-human-clinical-trial-market)
- Latin America the ‘Hidden Gem’ for First-in-Human Medical Device Trials (https://clinicalresearchnewsonline.com/news/2025/08/14/latin-america-the-hidden-gem-for-first-in-human-medical-device-trials)
- Identify Regulatory Requirements and Key Agencies
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Chile (ISP – Instituto de Salud Pública) – PharmaRegulatory.in – India’s Regulatory Knowledge Hub (https://pharmaregulatory.in/country-specific-regulatory-affairs/chile-isp-instituto-de-salud-publica)
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- Chile Clinical Trial Regulations | ISP Guide | bioaccess® (https://bioaccessla.com/regulatory-guide/chile)
- Prepare and Submit Required Documentation
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Chile Clinical Trial Regulations | ISP Guide | bioaccess® (https://bioaccessla.com/regulatory-guide/chile)
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- Clinical Trial Requirements In Latin America A Complete Pre Clinical Document Checklist For Sponsors | bioaccess® (https://bioaccessla.com/blog/clinical-trial-requirements-in-latin-america-a-complete-pre-clinical-document-checklist-for-sponsors)
- Chile (ISP – Instituto de Salud Pública) – PharmaRegulatory.in – India’s Regulatory Knowledge Hub (https://pharmaregulatory.in/country-specific-regulatory-affairs/chile-isp-instituto-de-salud-publica)
- Navigate the Review Process and Address Challenges
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)

Leave a Reply