Introduction
While conducting a first-in-human study in Chile presents exciting opportunities, it also comes with significant challenges that researchers must navigate.
Let’s explore the essential steps you need to take to navigate the intricate regulatory requirements and ethical considerations, ensuring a streamlined process from approval to execution.
Navigating these complexities can be daunting for researchers. Without effective management, trials may face significant setbacks.
So, how can you tackle these challenges to ensure successful outcomes in your trials?
Understand Regulatory Requirements and Ethical Considerations
Before embarking on a first-in-human study in Chile, it is crucial to understand the intricate compliance landscape for success. Follow these steps to ensure compliance and streamline your process:
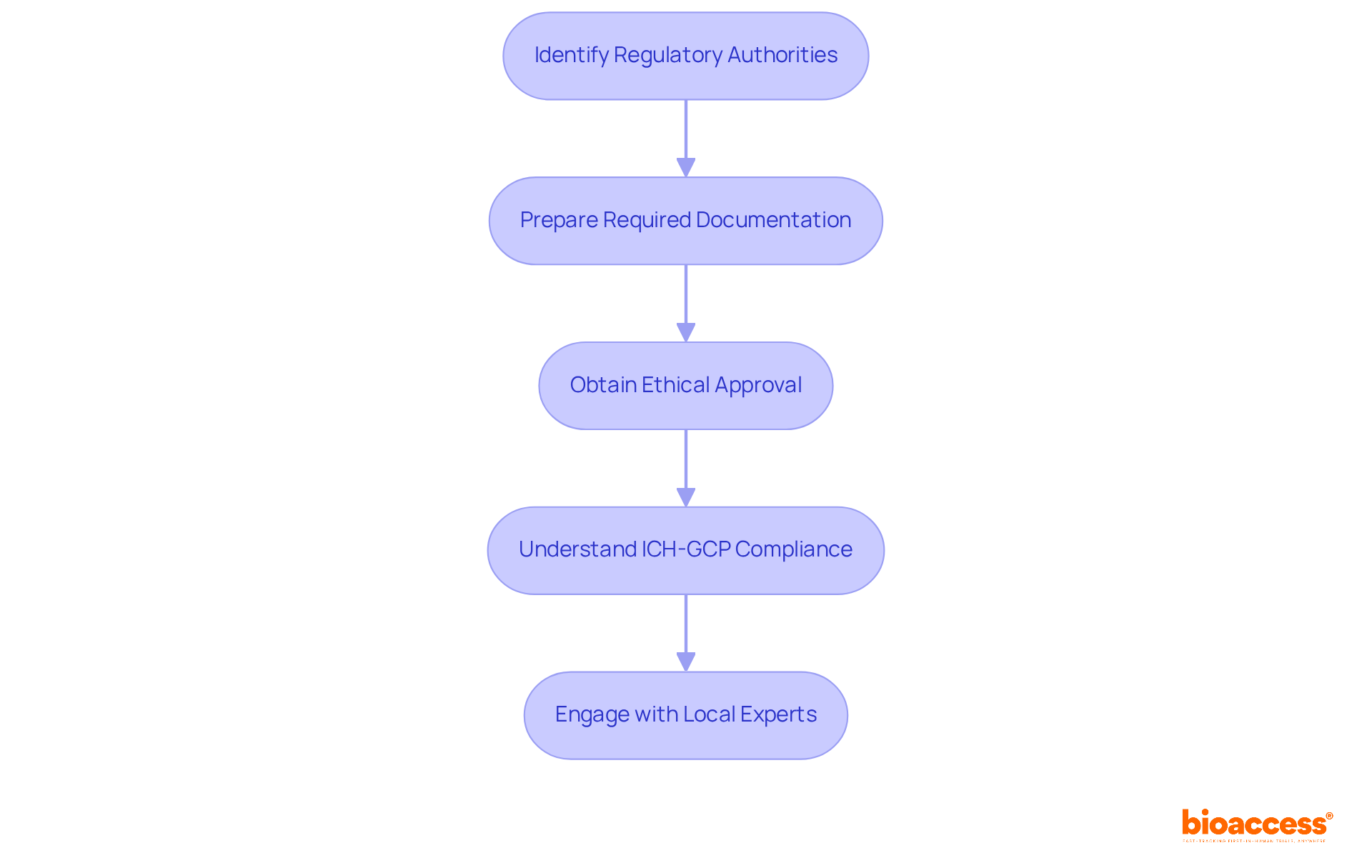
- Identify Regulatory Authorities: The Instituto de Salud Pública (ISP) is the primary regulatory organization supervising clinical studies in Chile. Familiarize yourself with their guidelines and requirements to ensure adherence.
- Prepare Required Documentation: Compile all necessary documents, including the clinical study protocol, informed consent forms, and investigator brochures. Ensure these documents are prepared in Spanish to comply with local regulations.
- Obtain Ethical Approval: Submit your study protocol to an accredited Scientific Ethics Committee (CEC) for review. This process typically takes 30-60 days. Ensure your informed consent process is robust and complies with Law 20.120, which mandates participant insurance.
- Understand ICH-GCP Compliance: Adhere to the International Council for Harmonisation’s Good Clinical Practice (ICH-GCP) guidelines, which are essential for ensuring the ethical and scientific quality of trials.
- Engage with Local Experts: Collaborate with local regulatory consultants or specialized CROs like bioaccess® to navigate the complexities of the approval process effectively. Their expertise can expedite your submission and help avoid common pitfalls.
Navigating the compliance landscape in Chile can be daunting for many researchers. By prioritizing compliance and ethical standards, you not only safeguard your study but also enhance its credibility in the eyes of stakeholders.
Select Clinical Sites and Recruit Patients Effectively
Recruiting the right patients at the right clinical sites is crucial for the success of your first-in-human study in Chile. Here’s how to approach this:
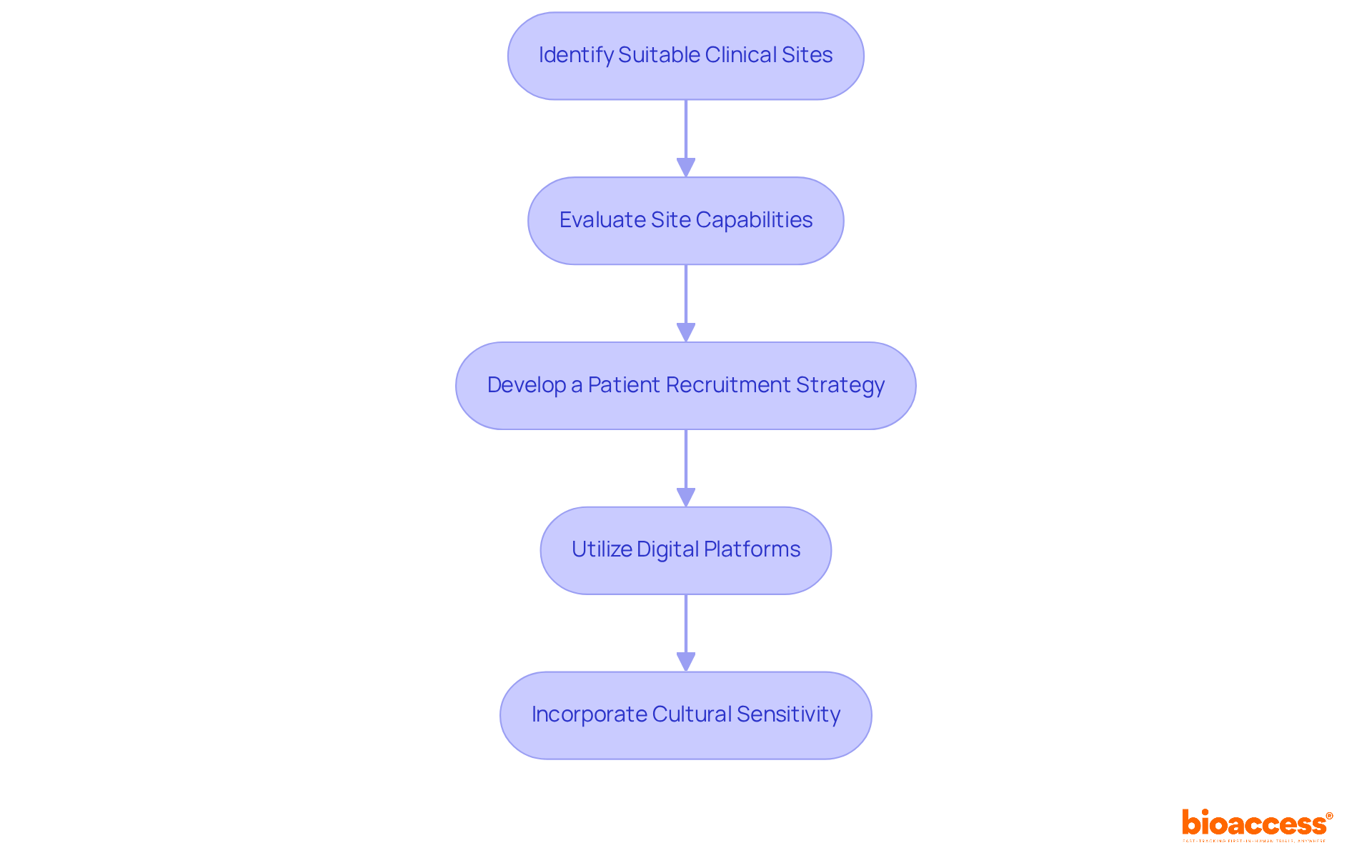
- Identify Suitable Clinical Sites: Focus on locations with proven experience in FIH studies and a strong track record of patient recruitment. Utilize bioaccess®’s extensive network of pre-qualified clinical research locations to streamline this process, ensuring you select sites that adhere to standards established by the Public Health Institute (ISP).
- Evaluate Site Capabilities: Assess each site’s infrastructure, including access to necessary medical equipment and the expertise of the staff. Confirm that they comply with ICH-GCP standards and have relevant experience with the specific type of intervention being tested, which is crucial for maintaining data integrity and regulatory compliance.
- Develop a Patient Recruitment Strategy: Create a targeted recruitment plan that includes outreach to local healthcare providers, community organizations, and patient advocacy groups. Tailor your messaging to resonate with the local population, emphasizing the benefits of participation and addressing common concerns.
- Utilize Digital Platforms: Leverage social media and online patient registries to reach potential participants effectively. Consider utilizing platforms that specifically serve the Chilean demographic to enhance engagement and increase visibility among potential candidates for participation.
- Incorporate Cultural Sensitivity: Understand the cultural nuances of the Chilean population. Engage local community leaders to establish trust and promote involvement in the study. This approach not only fosters goodwill but also enhances recruitment efforts by making potential participants feel valued and understood.
Recruiting patients for the first-in-human study in Chile can be daunting, especially in a diverse landscape. By prioritizing these strategies, you not only enhance recruitment but also pave the way for successful trial outcomes and timely approvals.
Execute the Study with Rigorous Protocol Adherence
Conducting a first-in-human study in Chile presents unique challenges that demand unwavering adherence to established protocols. To ensure compliance and maintain data integrity, follow these essential steps:
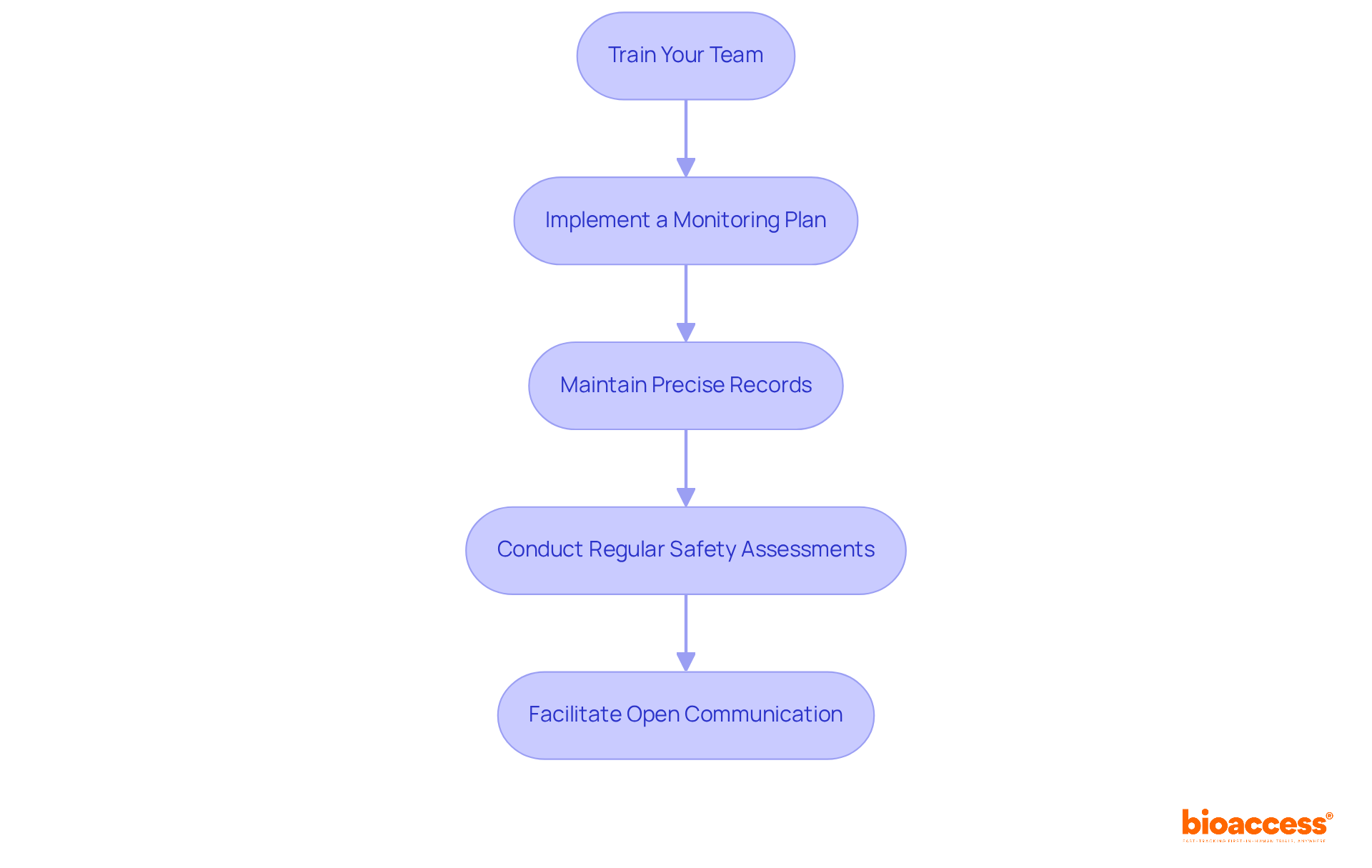
- Train Your Team: Implement comprehensive training sessions for all study personnel, focusing on the protocol, ICH-GCP guidelines, and ethical considerations. Every team member needs to grasp their specific roles and responsibilities to create a compliant environment.
- Implement a Monitoring Plan: Create a comprehensive monitoring strategy that incorporates regular site visits, information verification, and compliance checks. This plan should be designed to ensure strict adherence to the protocol and facilitate timely identification of any deviations.
- Maintain Precise Records: Ensure meticulous upkeep of all study-related documents, including source information and case report forms (CRFs). Precise record maintenance is vital for regulatory inspections and audits, enhancing the integrity of your research findings.
- Conduct Regular Safety Assessments: Continuously monitor participant safety throughout the trial. Establish a Data Safety Monitoring Board (DSMB) to review safety data and provide recommendations as necessary, ensuring participant welfare is prioritized.
- Facilitate Open Communication: Create an environment that encourages open dialogue among team members, investigators, and oversight bodies. Regular updates on study progress and any emerging issues are essential for maintaining transparency and trust.
By adhering to these practices, you enhance the reliability of your results. This also ensures compliance with the requirements set by the Instituto de Salud Pública (ISP) and COFEPRIS, which typically has approval timelines of 30 to 60 days. This organized method not only protects participant safety but also facilitates the journey to obtaining approval, ultimately bolstering the success of your clinical trial.
Manage Data and Prepare for Regulatory Submissions
Navigating the regulatory landscape for your first-in-human study in Chile can be a complex endeavor, but effective management and preparation are key to ensuring the success of the first-in-human study in Chile. Here’s how to navigate this process:
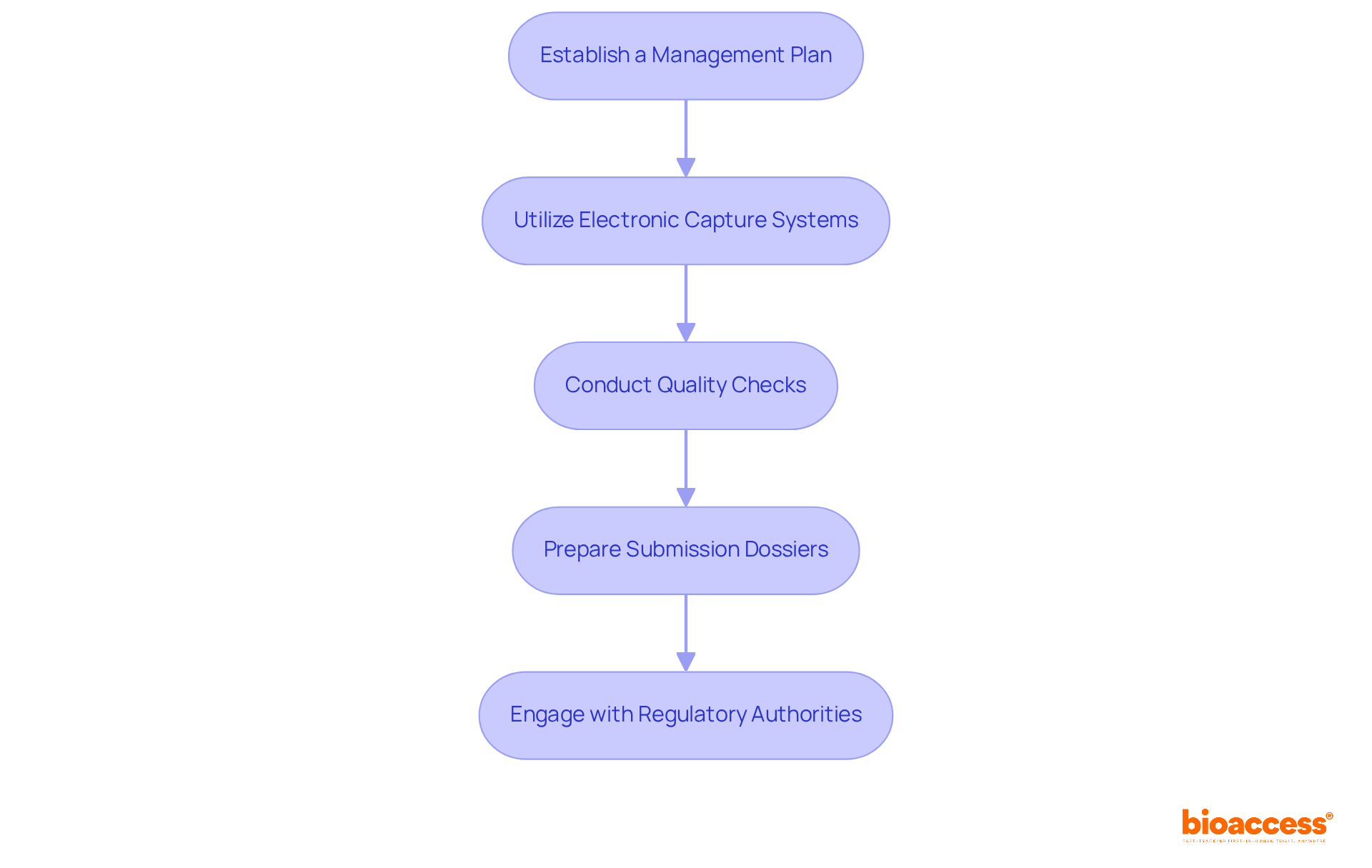
- Establish a Management Plan: Develop a comprehensive management strategy that outlines procedures for collection, storage, and analysis. It’s essential to comply with ICH-GCP standards and the local regulations from the Instituto de Salud Pública (ISP).
- Utilize Electronic Capture Systems: Implement EDC systems to streamline information collection and minimize errors. These systems must be user-friendly and comply with local regulations. They should also facilitate real-time access to information and validation. According to industry insights, EDC systems enhance accuracy and enable real-time access to clinical trial information, which is crucial for timely decision-making.
- Conduct Quality Checks: Regularly perform quality assessments to identify and rectify discrepancies. This includes verifying source information against case report forms (CRFs) and ensuring that all entries are precise and thorough. Measures to ensure the integrity of information may include implementing access controls and maintaining audit trails of modifications.
- Prepare Submission Dossiers: Gather all essential documentation for submissions, including clinical study reports, safety information, and informed consent forms. Ensure that these documents are translated into Spanish and meet ISP requirements for clarity and completeness.
- Engage with Regulatory Authorities: Maintain open lines of communication with the ISP throughout the submission process. Be prepared to respond promptly to any queries or requests for additional information, which can expedite the approval timeline. As pointed out by Patti Smith, a well-selected EDC system enhances information collection and aids in adherence, ultimately safeguarding the integrity of your clinical research information.
By following these guidelines, you can ensure efficient information management and thorough regulatory submissions, paving the way for successful study outcomes. The electronic data capture market is projected to reach USD 5.73 billion by 2034, highlighting the increasing relevance of EDC systems in clinical trials.
Conclusion
Navigating the regulatory landscape for first-in-human studies in Chile presents significant challenges that demand careful attention and ethical commitment. This guide outlines essential steps to navigate the complexities of conducting clinical trials, highlighting the critical roles of compliance, patient recruitment, and strict protocol adherence. By following these structured processes, researchers can uphold the integrity and success of their clinical trials.
Key insights from this article include:
- The necessity of engaging with local regulatory authorities like the Instituto de Salud Pública (ISP).
- Preparing comprehensive documentation in Spanish.
- Implementing a robust patient recruitment strategy that respects cultural nuances.
- Effective data management and open communication with regulatory bodies are crucial for securing timely approvals and ensuring participant safety.
Embracing the unique advantages that Chile offers for early-stage clinical trials-such as efficient regulatory pathways and a diverse patient population-can enhance the likelihood of successful study outcomes. By prioritizing compliance and ethical considerations, researchers not only safeguard their studies but also contribute to the advancement of medical science in Latin America. Ultimately, the success of first-in-human studies hinges on a proactive approach to compliance and ethical standards, shaping the future of medical research in Latin America.
Frequently Asked Questions
What is the primary regulatory authority for clinical studies in Chile?
The primary regulatory authority overseeing clinical studies in Chile is the Instituto de Salud Pública (ISP).
What documentation is required for a first-in-human study in Chile?
Required documentation includes the clinical study protocol, informed consent forms, and investigator brochures, all of which must be prepared in Spanish to comply with local regulations.
How do I obtain ethical approval for my study in Chile?
You must submit your study protocol to an accredited Scientific Ethics Committee (CEC) for review, which typically takes 30-60 days. Additionally, ensure that your informed consent process complies with Law 20.120, which mandates participant insurance.
What guidelines should be followed to ensure compliance with ethical standards in clinical trials?
Adherence to the International Council for Harmonisation’s Good Clinical Practice (ICH-GCP) guidelines is essential for ensuring the ethical and scientific quality of trials.
How can engaging local experts benefit my clinical study in Chile?
Collaborating with local regulatory consultants or specialized Contract Research Organizations (CROs) like bioaccess® can help navigate the complexities of the approval process, expedite submissions, and avoid common pitfalls.
Why is it important to prioritize compliance and ethical standards in clinical studies?
Prioritizing compliance and ethical standards safeguards your study and enhances its credibility in the eyes of stakeholders, which is crucial for the success of first-in-human studies.
List of Sources
- Understand Regulatory Requirements and Ethical Considerations
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- Chile (ISP – Instituto de Salud Pública) – PharmaRegulatory.in – India’s Regulatory Knowledge Hub (https://pharmaregulatory.in/country-specific-regulatory-affairs/chile-isp-instituto-de-salud-publica)
- First-in-Human Trial Participants: Not a Vulnerable Population, but Vulnerable Nonetheless – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC2692671)
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Select Clinical Sites and Recruit Patients Effectively
- Clinical Trial Site Selection Latin America Ke… | bioaccess® (https://bioaccessla.com/blog/clinical-trial-site-selection-latin-america-key-strategies)
- Patient Engagement Quotes: For Every Purpose & Audience (https://nclusiv.co.uk/edi-consulting/f/patient-engagement-quotes-for-every-purpose-audience)
- 10 Inspiring Patient Experience Quotes | Relias (https://relias.com/blog/patient-experience-quotes)
- Enrollment in Clinical Trials: Statistics and Patient Recruitment Strategies | Power (https://withpower.com/guides/enrollment-in-clinical-trials-statistics-and-patient-recruitment-strategies)
- Execute the Study with Rigorous Protocol Adherence
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Importance of Compliance in Clinical Trials (https://primerevivalresearch.com/blogs/importance-of-compliance-in-clinical-trials)
- Patient Engagement Quotes: For Every Purpose & Audience (https://nclusiv.co.uk/edi-consulting/f/patient-engagement-quotes-for-every-purpose-audience)
- Manage Data and Prepare for Regulatory Submissions
- 2026 Clinical Data Trend Report | Veeva (https://veeva.com/2026-clinical-data-trend-report)
- A guide to creating a clinical trial data management plan | Clinical Trials Hub (https://clinicaltrialshub.htq.org.au/blog/a-guide-to-creating-a-clinical-trial-data-management-plan)
- Electronic Data Capture Market Size, Share | Growth [2034] (https://fortunebusinessinsights.com/electronic-data-capture-market-115364)
- Electronic Data Capture (EDC) Systems Explained (https://ccrps.org/clinical-research-blog/electronic-data-capture-edc-systems)
- EDC Clinical Trials: Is Your EDC System Fit for Purpose? (https://namsa.com/resources/blog/edc-clinical-trials)

Leave a Reply