Introduction
In the complex realm of clinical trials, the closure phase often gets overshadowed by the thrill of results and findings. Yet, the significance of a well-crafted trial closure documentation package is paramount; it lays the groundwork for regulatory compliance and ethical integrity. This article explores best practices to ensure that trial closure documentation fulfills essential requirements, spotlighting the critical components that protect data integrity and uphold participant rights.
What challenges do organizations encounter while navigating the intricacies of compliance? How can they effectively tackle these obstacles to build trust and transparency in clinical research?
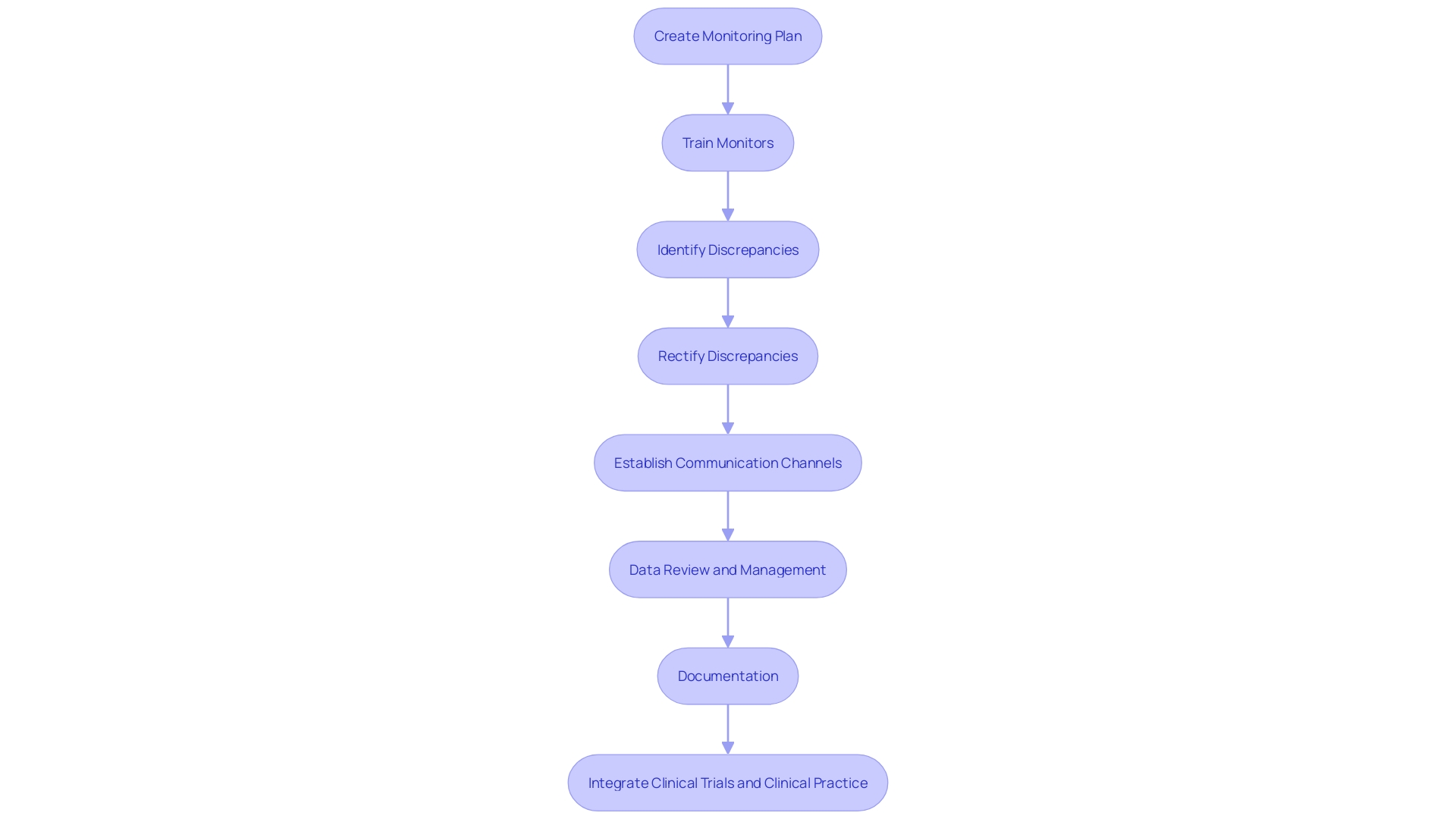
Identify Essential Components of Trial Closure Documentation
To ensure a comprehensive , it is essential to include the following components:
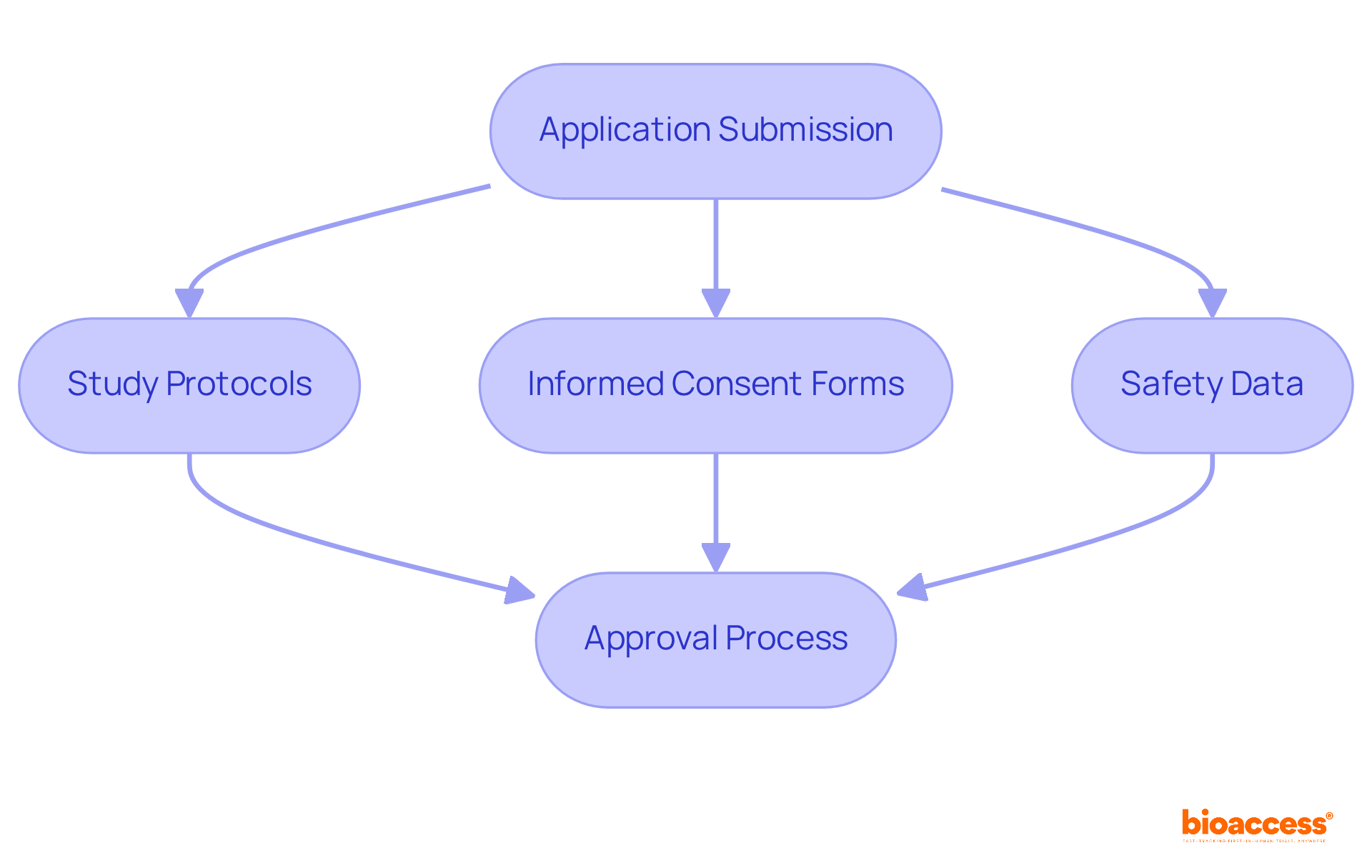
- : This document serves as the cornerstone of the closure process, summarizing the study’s objectives, methodology, results, and conclusions. It must be clear and concise, providing a complete overview of the examination to facilitate regulatory review and decision-making. With bioaccess’s expertise in project management, the final report is meticulously prepared and submitted in compliance with , such as the requirement for FDA IDE studies to be submitted to the IRB within six months of study completion or termination.
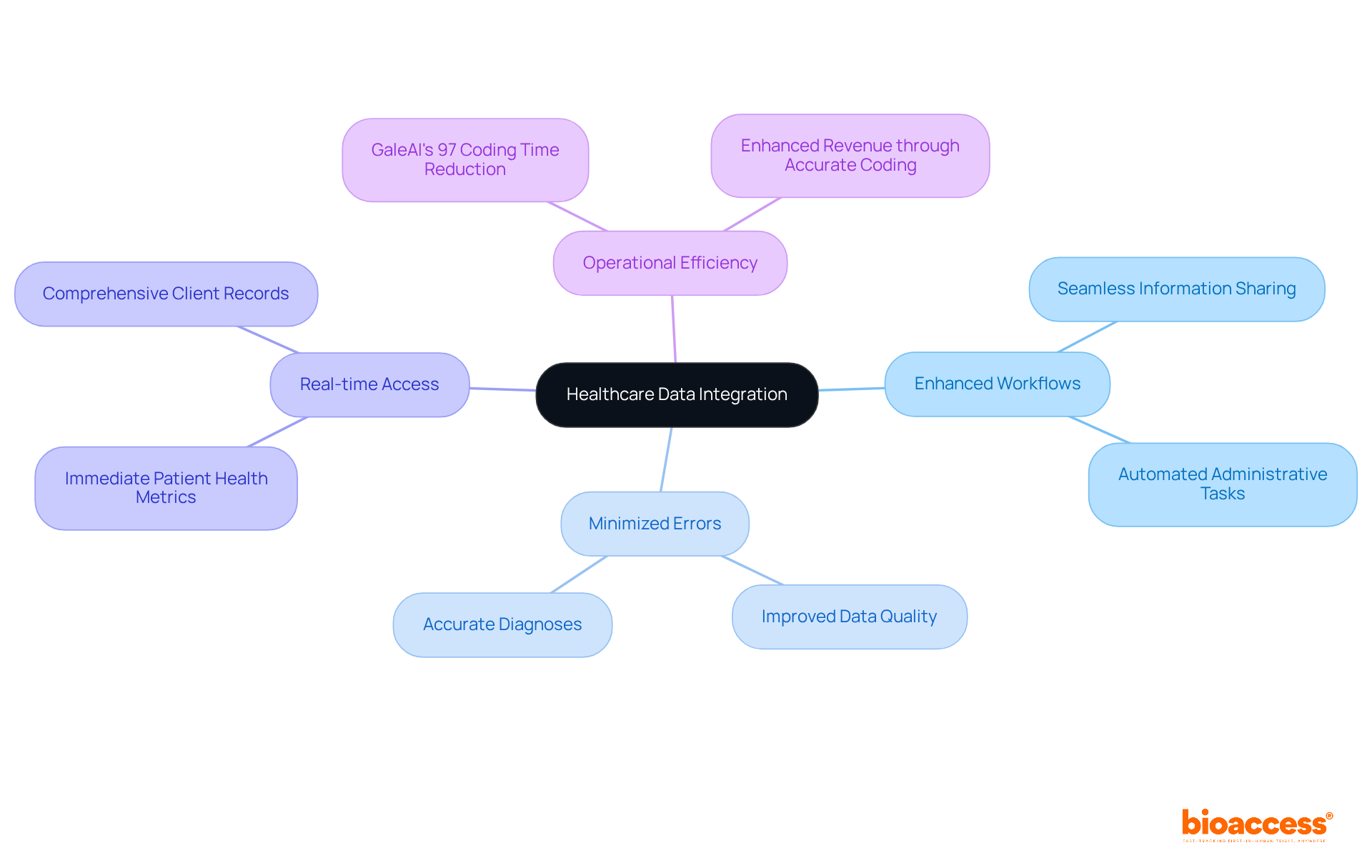
- : Include records that demonstrate the accuracy and completeness of the data collected during the trial. This may involve data validation reports and audit trails, which are crucial for maintaining data integrity and compliance with regulatory standards. Bioaccess’s services can assist in ensuring that all necessary records, including the Last Subject’s Last Visit (LSLV) details, are accurately maintained to declare study completion.
- : Ensure that all necessary approvals and compliance documents are included, such as Institutional Review Board (IRB) approvals, informed consent forms, and any correspondence with regulatory bodies. Bioaccess helps streamline this process, ensuring that all approvals are in place and that the FDA’s requirement for closure of any open IRB approvals is met, facilitating a smooth close-out process and future audits.
- : Document any follow-up procedures for participants post-trial, including safety monitoring and reporting of adverse events. This information is essential for ensuring participant safety and maintaining ethical standards in clinical research. Bioaccess supports the development of final safety reports that must be submitted to ethics committees/IRBs and regulatory authorities at study close-out.
- : Describe how and where the experimental records will be stored, ensuring that they are available for future reference and evaluations. Proper archiving is critical, as regulatory guidelines often require retention of documents for several years after study completion. Bioaccess can assist in creating a robust that adheres to ICH-GCP mandates for document retention.
- : Collect and include final financial disclosures from all investigators involved in the study. This transparency is necessary to uphold ethical standards and avoid conflicts of interest. Bioaccess ensures that all financial disclosures are collected and documented appropriately.
- : Utilize a checklist to confirm that all components have been addressed and completed before finalizing the documentation package. This structured approach helps prevent oversights and ensures compliance with regulatory expectations. Bioaccess’s extensive include a for nammd and ethics, ensuring the team remains aligned during the concluding phase of a clinical study. Additionally, multiple review cycles across different teams can significantly reduce errors and ensure consistency before submission.
Utilizing bioaccess’s extensive , such as feasibility assessments, site selection, compliance evaluations, and project oversight, can improve the efficiency of these elements, ensuring a thorough and compliant study completion process.
Ensure Compliance with NAMMD and Ethical Standards
To ensure compliance with NAMMD and uphold ethical standards during trial closure, implementing the following best practices is essential:
- Acquaint with : Gain a thorough understanding of the specific stipulations set by the National Agency for Medicines and Medical Devices (NAMMD) regarding trial completion records and reporting. to effectively navigate the trial closure documentation package for NAMMD and ethics regulations.
- Conduct a : Before finalizing documentation, perform an internal audit to confirm that all regulatory requirements have been met, focusing on the thoroughness and accuracy of the documentation. to support this process.
- : Submit all closure documents to the relevant ethical review board for approval, ensuring that the study adhered to ethical standards throughout its duration. Bioaccess can assist in facilitating this approval through its testing setup services.
- : Document the measures taken to protect participant rights, including informed consent procedures and any follow-up communications regarding study results. in its project management approach.
- : Ensure that all adverse events occurring during the study are reported in compliance with NAMMD regulations, detailing any follow-up actions taken. Bioaccess provides reporting services that ensure adherence to these regulatory standards.
- : Provide for all team members involved in concluding the study to ensure they are well-versed in compliance necessities and ethical considerations. Bioaccess offers assistance in training to enhance overall study management effectiveness.
Applying these practices not only improves adherence but also fosters trust and integrity in clinical research, ultimately aiding in the successful completion of studies.
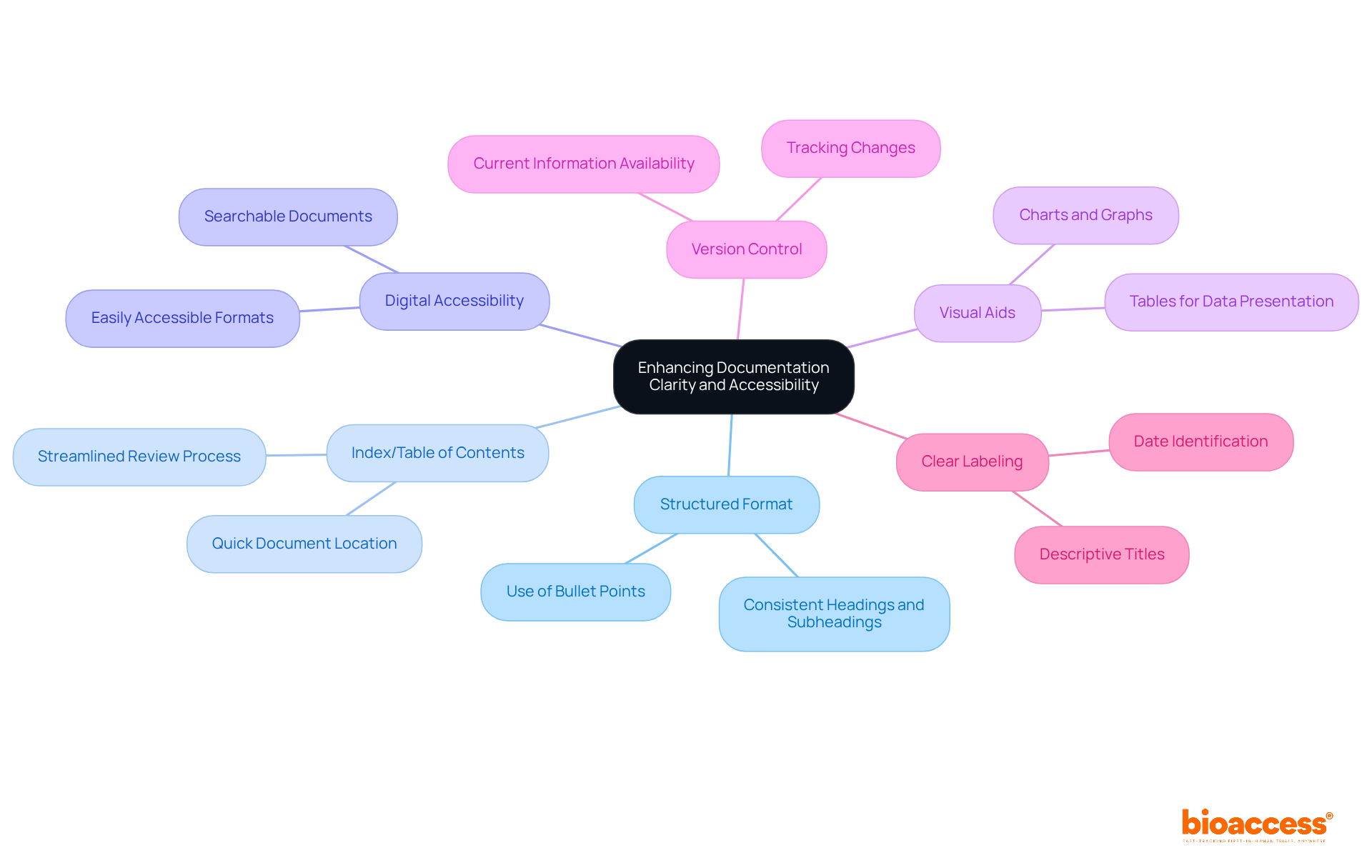
Organize and Present Documentation for Clarity and Accessibility
To enhance the clarity and accessibility of trial closure documentation, the following strategies should be implemented:
- Structured Format: Adopting a consistent format across all documents – utilizing headings, subheadings, and bullet points – significantly improves readability and comprehension. As W. Edwards Deming noted, ‘,’ highlighting the necessity for organized evidence in records.
- Establishing an index or table of contents in the for NAMMD and ethics enables stakeholders to swiftly locate specific documents or sections, streamlining the review process.
- : All documents should be available in and searchable, facilitating quick retrieval during audits and ensuring compliance with regulatory standards.
- Visual Aids: The inclusion of charts, graphs, and tables can effectively present data in a clear and concise manner, making complex information more digestible for stakeholders.
- Version Control: Implementing a is crucial for tracking changes and updates to documents, ensuring that the most current information is readily available and reducing the risk of errors.
- Clear Labeling: All documents must be clearly labeled with descriptive titles and dates to prevent confusion, allowing stakeholders to easily identify the content and its relevance.
The application of not only improves clarity but also aids in compliance and efficiency. Research shows that well-organized records can result in a 20% enhancement in quality, as illustrated in the case study ‘,’ which identified significant improvements in record quality after adopting structured formats. Additionally, common pitfalls such as inconsistent labeling or lack of can hinder the effectiveness of these strategies, so it is essential to address these issues proactively.
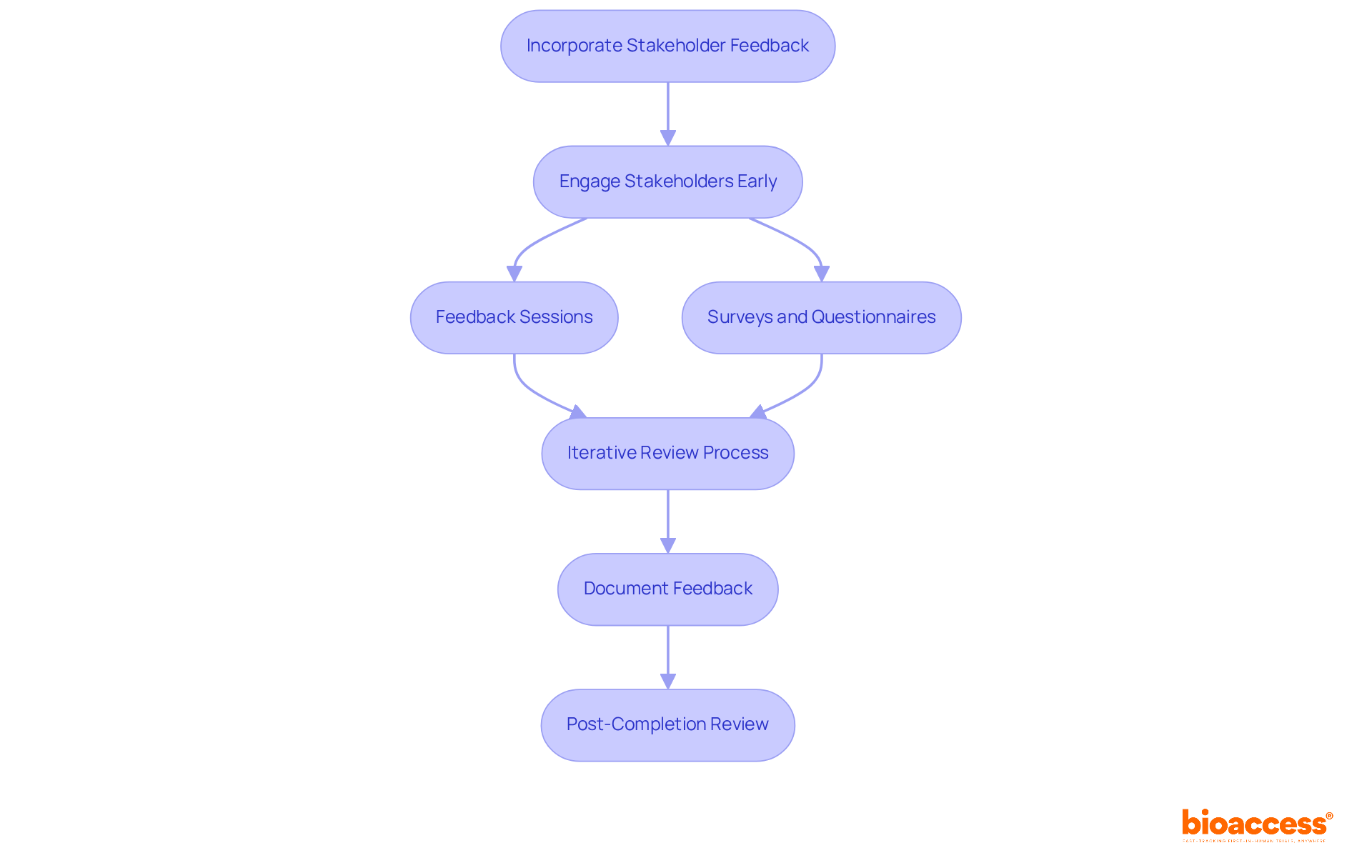
Incorporate Stakeholder Feedback for Comprehensive Documentation
To effectively incorporate into the for nammd and ethics, organizations must adopt strategic approaches that enhance the quality of their records and ensure compliance with regulatory standards.
- Engage Stakeholders Early: Involve key stakeholders-investigators, sponsors, and regulatory bodies-early in the record-keeping phase to gather their insights and expectations. Early engagement fosters a collaborative environment and aligns objectives, as emphasized in the , which clarifies investigator responsibilities and the importance of .
- Feedback Sessions: Organize where stakeholders can review draft documents and provide input on content, clarity, and completeness. These sessions not only improve the quality of records but also foster trust among participants, reinforcing the collaborative spirit essential in .
- Surveys and Questionnaires: Utilize surveys or questionnaires to collect structured feedback from stakeholders regarding their experiences and suggestions for improvement. This method allows for a broader range of insights and can highlight areas needing attention, ultimately driving better outcomes.
- : Implement an where records are revised based on . This guarantees that all issues are addressed prior to completion, resulting in a more robust and comprehensive set of materials that meet the needs of all parties involved.
- : Keep a record of all feedback received and how it was handled in the materials. This practice demonstrates responsiveness and accountability, strengthening stakeholder trust in the system. It is crucial to ensure that the new informed consent requirements introduced in the Draft Guidance are also documented appropriately.
- : Following the test conclusion, hold a review meeting with stakeholders to discuss the record-keeping process and collect lessons learned for upcoming tests. This reflective practice can significantly improve the quality of later record-keeping efforts, ensuring continuous improvement.
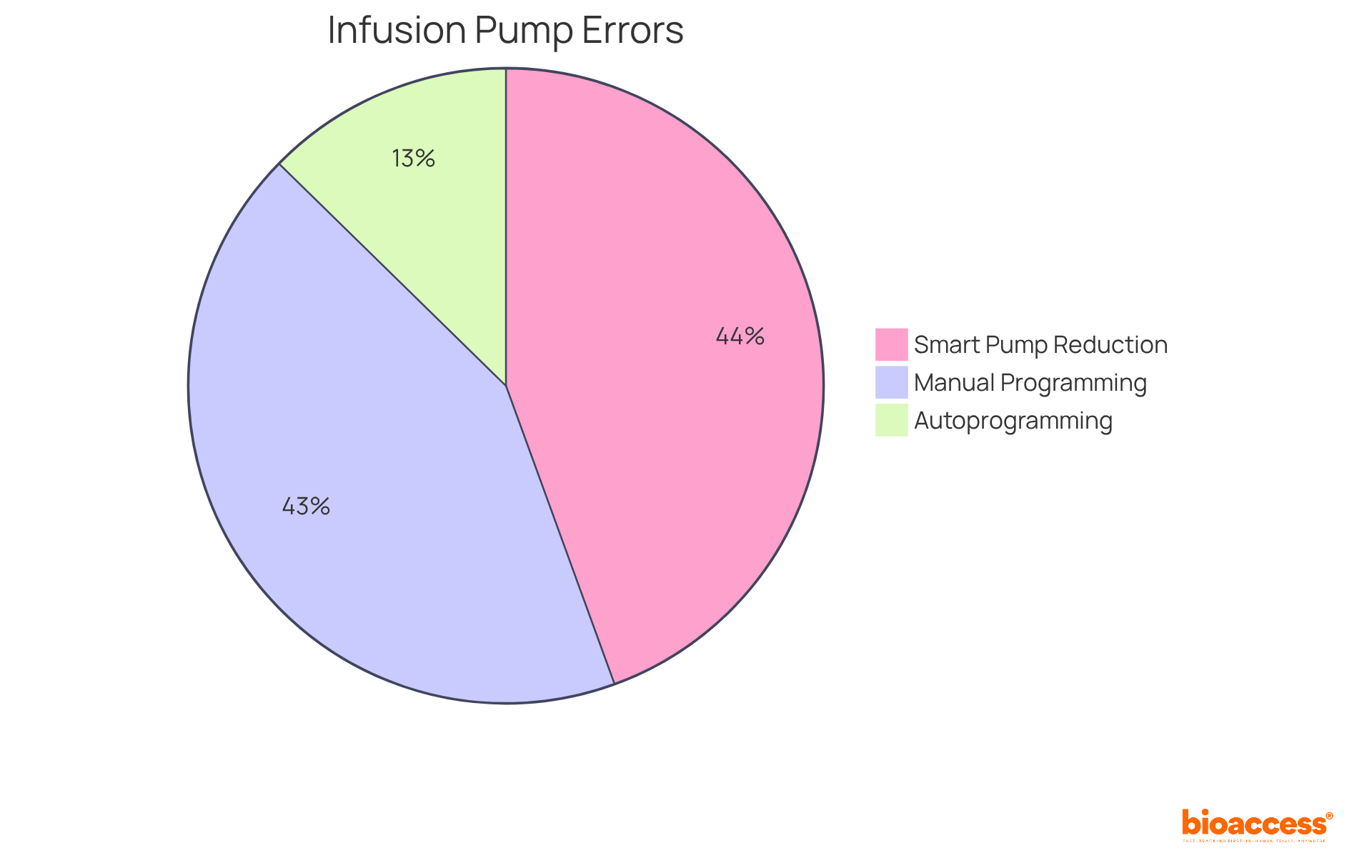
By adopting these strategies, organizations can enhance the quality of their for nammd and ethics, ensuring compliance with regulatory standards and meeting stakeholder expectations. For instance, utilizing PCA pumps with built-in capnography might avert over 60% of negative occurrences, underscoring the significance of and stakeholder involvement. As Brené Brown noted, the richness and depth of qualitative research underscore the value of in the documentation process.
Conclusion
Ensuring a thorough and compliant trial closure documentation package is essential for successful clinical research. This article underscores the critical components necessary for effective documentation, including:
- Final study report
- Data integrity records
- Regulatory compliance
- Participant follow-up information
- Archiving plan
By adhering to these best practices, organizations can streamline the closure process and uphold ethical standards, ultimately fostering trust and integrity in clinical research.
Key insights reveal the importance of:
- Understanding regulatory requirements
- Conducting compliance audits
- Engaging stakeholders throughout the documentation process
Implementing structured formats and incorporating feedback not only enhances the quality of records but also ensures that all ethical considerations are addressed. Utilizing tools such as checklists and version control allows organizations to mitigate the risk of oversights and maintain clarity in their documentation.
The significance of a comprehensive trial closure documentation package cannot be overstated. It serves as a foundation for regulatory review, protects participant rights, and promotes transparency in clinical research. Organizations are encouraged to adopt these best practices and actively seek stakeholder input to enhance their documentation processes, ultimately contributing to the success and integrity of future clinical trials.
Frequently Asked Questions
What is the purpose of the Final Study Report in trial closure documentation?
The Final Study Report summarizes the study’s objectives, methodology, results, and conclusions, serving as the cornerstone of the closure process. It must be clear and concise to facilitate regulatory review and decision-making.
Why is Data Integrity Documentation important in trial closure?
Data Integrity Documentation includes records that demonstrate the accuracy and completeness of the data collected during the trial, such as data validation reports and audit trails, which are crucial for maintaining compliance with regulatory standards.
What types of Regulatory Compliance Records should be included in trial closure documentation?
Regulatory Compliance Records should include approvals from Institutional Review Boards (IRBs), informed consent forms, and any correspondence with regulatory bodies to ensure that all necessary approvals are in place for a smooth close-out process.
What information is included in Participant Follow-Up Information?
Participant Follow-Up Information documents any follow-up procedures for participants post-trial, including safety monitoring and reporting of adverse events, which are essential for ensuring participant safety and maintaining ethical standards in clinical research.
What is the purpose of an Archiving Plan in trial closure documentation?
An Archiving Plan describes how and where the experimental records will be stored, ensuring they are available for future reference and evaluations, as regulatory guidelines often require retention of documents for several years after study completion.
Why are Financial Disclosure Forms necessary in trial closure documentation?
Financial Disclosure Forms are necessary to collect final financial disclosures from all investigators involved in the study, ensuring transparency to uphold ethical standards and avoid conflicts of interest.
How does a Study Closure Checklist assist in the trial closure process?
A Study Closure Checklist helps confirm that all components have been addressed and completed before finalizing the documentation package, preventing oversights and ensuring compliance with regulatory expectations.
How can bioaccess assist with trial closure documentation?
Bioaccess provides extensive clinical study management services, including the preparation of trial closure documentation packages, ensuring compliance with regulatory requirements, and improving the efficiency of the study completion process.
List of Sources
- Identify Essential Components of Trial Closure Documentation
- Clinical Study Report (CSR): The Definitive Guide for 2026 (https://clinion.com/insight/clinical-study-reports-csr-complete-guide)
- Study Close-Out for Clinical Trials: A GCP Checklist | IntuitionLabs (https://intuitionlabs.ai/articles/clinical-trial-site-close-out-checklist)
- 5 Aspects of Device Clinical Study Close-Out | MED Institute (https://medinstitute.com/blog/5-aspects-of-device-clinical-study-close-out)
- How to Write a Clinical Study Report (https://morulahealth.com/post/how-to-write-a-clinical-study-report)
- Ensure Compliance with NAMMD and Ethical Standards
- precisionformedicine.com (https://precisionformedicine.com/blog/case-study-effective-management-of-a-rescue-study)
- Study Close-Out for Clinical Trials: A GCP Checklist | IntuitionLabs (https://intuitionlabs.ai/articles/clinical-trial-site-close-out-checklist)
- Regulatory Compliance in Clinical Research | Novotech CRO (https://novotech-cro.com/faq/regulatory-compliance-clinical-research)
- Organize and Present Documentation for Clarity and Accessibility
- 70 Research Quotes to Inspire Your Work – Qualtrics (https://qualtrics.com/articles/strategy-research/research-quotes)
- Trial Exhibit Organization Tips for Maximum Courtroom Impact | Complete Legal (https://completelegal.us/trial-exhibit-organization-tips-for-maximum-courtroom-impact)
- Checking your browser – reCAPTCHA (https://pmc.ncbi.nlm.nih.gov/articles/PMC9135789)
- Incorporate Stakeholder Feedback for Comprehensive Documentation
- ppahs.org (https://ppahs.org/best-health-expert-quotes)
- Seven Opportunities for Stakeholder Engagement on FDA Draft Guidance Proposing Updates to Good Clinical Practice Guidelines (https://cov.com/en/news-and-insights/insights/2023/08/seven-opportunities-for-stakeholder-engagement-on-fda-draft-guidance-proposing-updates-to-good-clinical-practice-guidelines)
- Patient Engagement Quotes: For Every Purpose & Audience (https://nclusiv.co.uk/edi-consulting/f/patient-engagement-quotes-for-every-purpose-audience)
- 70 Research Quotes to Inspire Your Work – Qualtrics (https://qualtrics.com/articles/strategy-research/research-quotes)









 Flowchart: Data Management and Analysis in Clinical Trials](https://vectorly-files.s3.eu-central-1.amazonaws.com/22176642-2663-4edb-8623-4e19845b85a1.jpg)