Introduction
While Chile offers a promising landscape for medical device clinical trials, the path to success is fraught with challenges that sponsors must navigate carefully.
With a regulatory framework that encourages swift approvals and a robust healthcare system, Chile stands out as a strategic hub for early-stage clinical trials.
However, navigating these complexities can often feel overwhelming for sponsors. Without a clear strategy, sponsors risk delays and setbacks in their trials.
This guide will help you master the clinical trial process in Chile, giving you the insights you need to thrive in this competitive environment.
Understand the Regulatory Landscape for Medical Device Trials in Chile
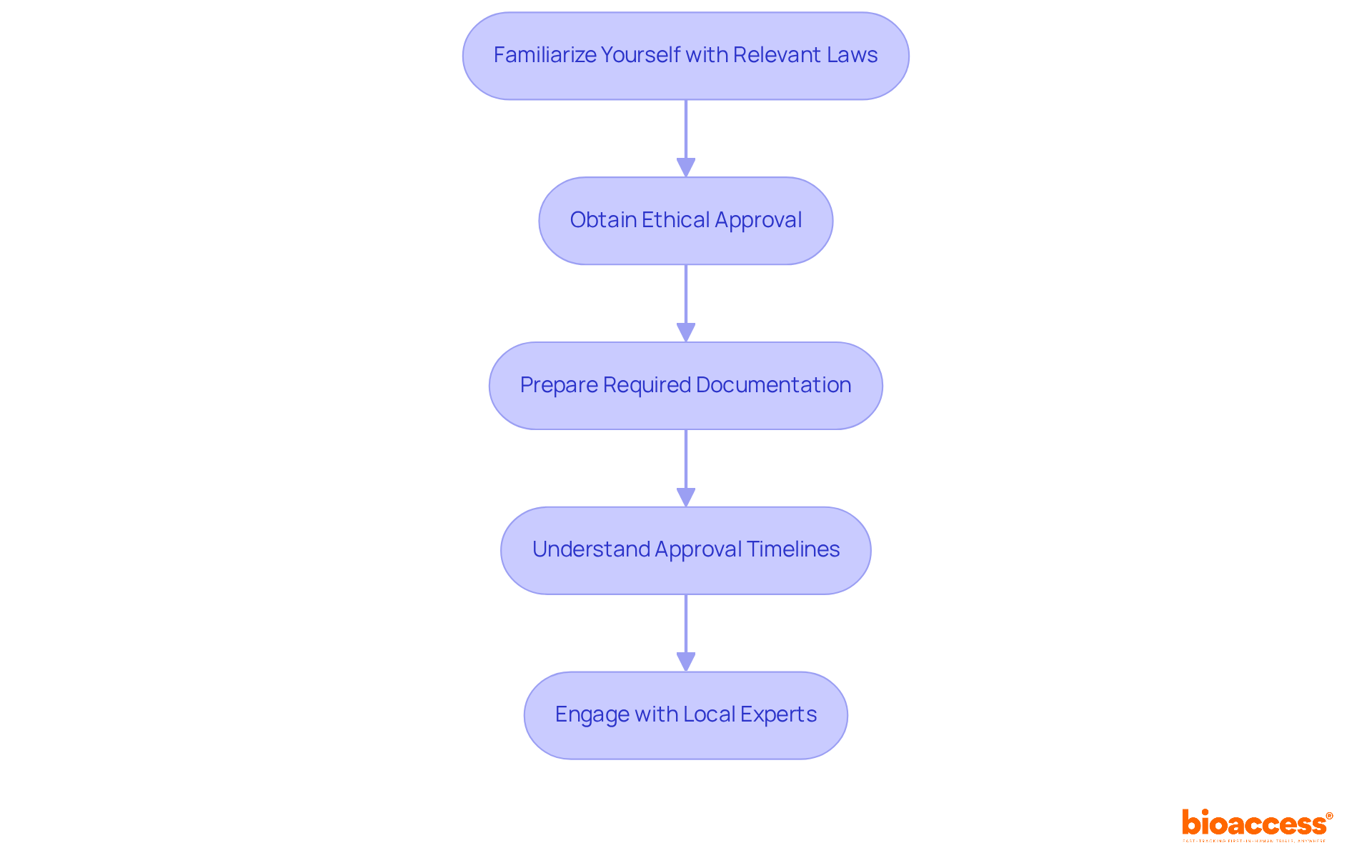
Understanding the regulatory landscape for the medical device clinical trial Chile is crucial for successful study execution. Navigating the medical device clinical trial Chile necessitates a comprehensive grasp of the regulatory framework, primarily overseen by the Instituto de Salud Pública (ISP). Here are the essential steps to ensure compliance:
- Familiarize Yourself with Relevant Laws: Review the Medical Devices Law and associated regulations, particularly Law N° 20.120, which outlines the requirements for human research.
- Obtain Ethical Approval: Have you secured endorsement from an accredited Ethics Committee (EC) before commencing any study? While this process is voluntary, it is critical for ensuring compliance with ethical standards.
- Prepare Required Documentation: Compile all necessary documentation, including the clinical research protocol, informed consent forms, and any prior approvals. Ensure that all documents adhere to ISP standards to facilitate a smooth review process for the medical device clinical trial in Chile.
- Understand approval timelines for medical device clinical trial Chile: Typically, securing authorization for medical device studies in Chile takes around 30 days, but this can vary based on study complexity and the completeness of the submitted documentation.
- Engage with Local Experts: Have you considered collaborating with local compliance consultants or specialized CROs like bioaccess®? Their expertise can help you navigate the approval landscape and expedite the authorization process.
By adhering to these steps, sponsors can utilize Chile’s advantageous regulatory landscape, marked by efficient approval procedures and a strong healthcare system, to expedite their research studies. Embracing these steps not only streamlines your research process but also positions you for success in a competitive market.
Prepare for Your Trial: Site Selection and Patient Recruitment Strategies
Strategic site selection and effective patient recruitment are critical for the success of your medical device clinical trial in Chile. Follow these steps:
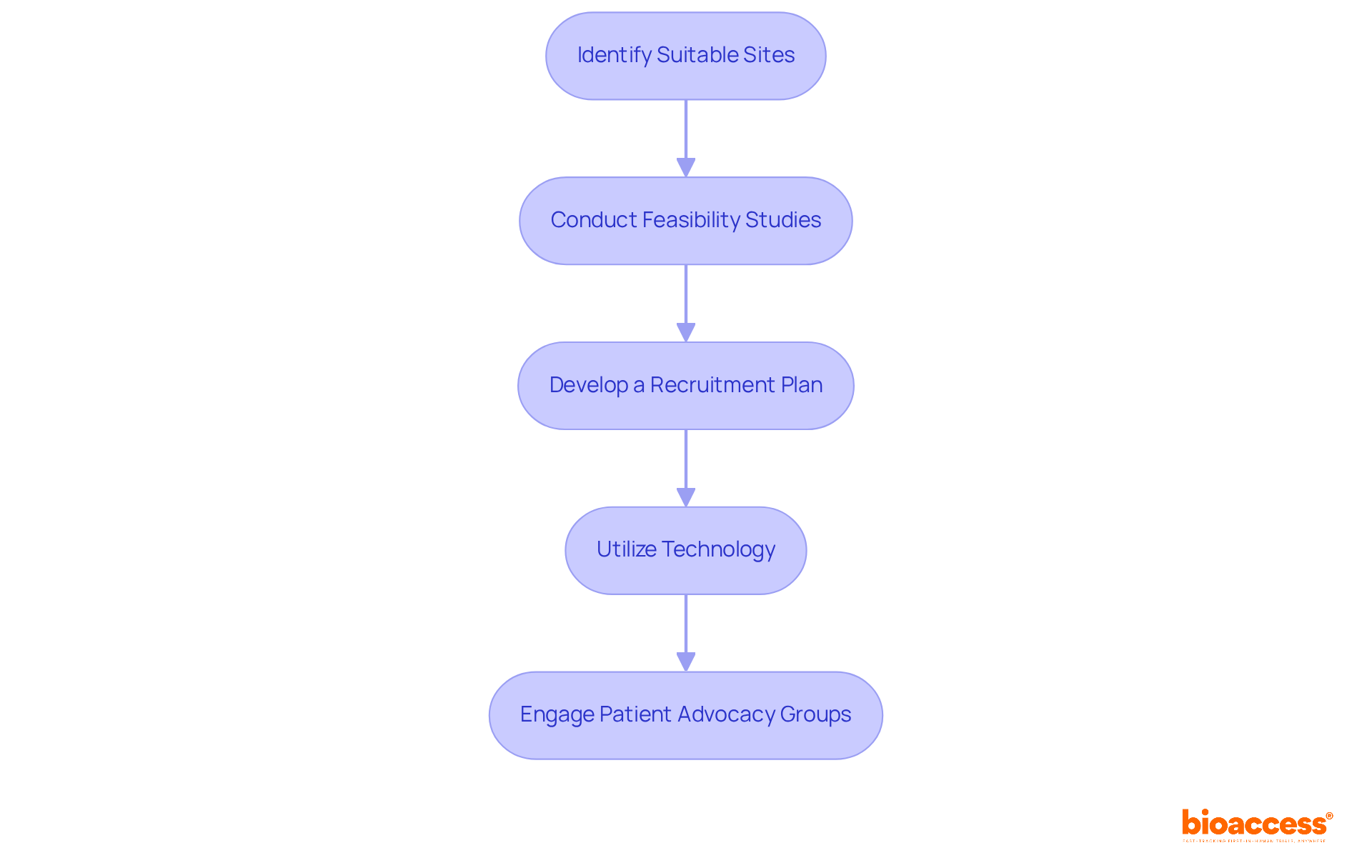
- Identify Suitable Sites: Assess potential clinical research locations based on their experience with medical device studies, patient demographics, and infrastructure. Leverage bioaccess®’s network of over 50 pre-qualified sites to streamline this process. This ensures compliance with ICH-GCP standards and local regulations from authorities like the Instituto de Salud Pública (ISP) and the Agencia Nacional de Medicamentos (ANAMED).
- Conduct Feasibility Studies: Assess the availability of eligible patients and the site’s capacity to meet enrollment goals. This can involve reviewing historical data on patient recruitment and retention, which is crucial for understanding site performance and optimizing recruitment strategies. You’ll find that Bioaccess® offers valuable insights into market access strategies that can really help inform your feasibility studies.
- Develop a Recruitment Plan: Create a comprehensive recruitment strategy that includes digital marketing, community outreach, and partnerships with local healthcare providers. Customize your messaging to connect with the intended patient group, highlighting the advantages of involvement in the medical device clinical trial Chile. How can you engage with local communities to enhance trust and participation rates?
- Utilize Technology: Leverage digital platforms and social media to reach potential participants effectively. Consider using telemedicine for initial consultations to broaden your reach and facilitate engagement with patients who may have logistical barriers to in-person visits. Recent studies indicate that digital recruitment strategies can significantly enhance patient enrollment by targeting specific demographics, a strategy supported by bioaccess®’s insights.
- Engage Patient Advocacy Groups: Collaborate with patient advocacy organizations to enhance recruitment efforts and build trust within the community. These groups can provide valuable insights into patient needs and preferences, helping to refine your recruitment messaging and strategies. Bioaccess® encourages partnerships with these organizations to further strengthen recruitment efforts.
By prioritizing these tailored strategies, you can significantly enhance your chances of successful patient recruitment in Chile.
Execute the Trial: Operational Processes and Compliance Monitoring
Executing a medical device clinical trial in Chile involves unique operational challenges that require meticulous attention to compliance. Here are several key processes to consider:
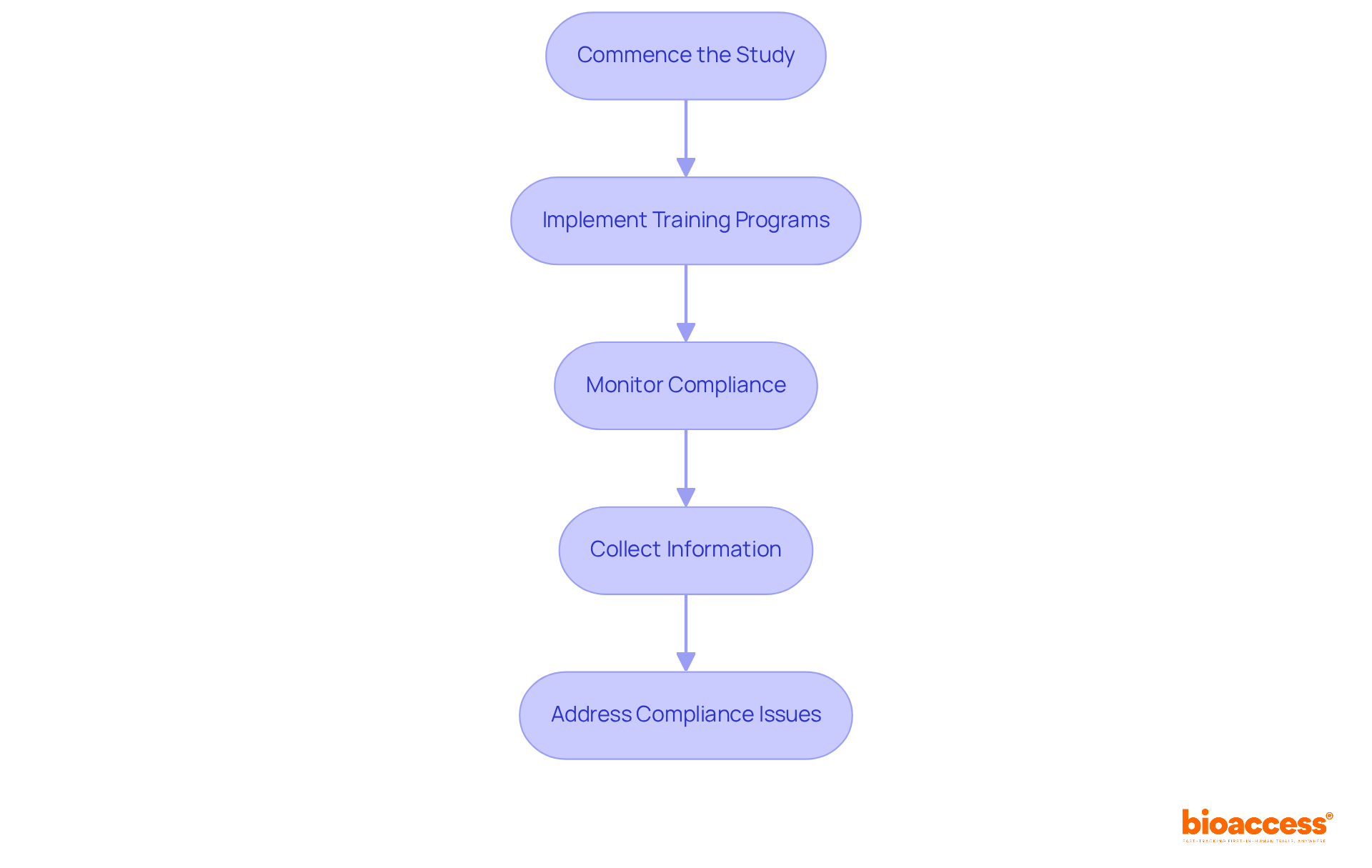
- Commence the Study: After obtaining all essential approvals from oversight bodies such as ANVISA and COFEPRIS, kick off the study with a meeting that includes all stakeholders, such as site personnel and researchers. This meeting is crucial for aligning expectations and clarifying roles.
- Implement Training Programs: Ensure that all site personnel are thoroughly trained on the study protocol, ICH-GCP guidelines, and specific regulatory requirements. When training programs are effective, they not only boost compliance but also significantly lower the chances of deviations. Tailor training to the study specifics, utilizing interactive modules to engage staff effectively.
- Monitor Compliance: Establish a robust compliance oversight plan that includes regular site visits, information verification, and audits. In 2026, following ICH-GCP standards is essential, as statistics show that non-compliance can cause significant delays in trial approval and inflate costs. Regular audits help identify potential compliance gaps early, allowing for timely corrective actions.
- Collect Information: Utilize electronic information capture (EIC) systems to streamline collection and management. These systems enable real-time information entry, improving precision and reducing mistakes. The transition to EDC has been nearly universal in clinical research, providing immediate data availability for review and reducing the time required for data processing.
- Address Compliance Issues: Proactively identify and resolve any compliance concerns that arise during the study. This may involve implementing corrective actions, providing additional training, or making necessary protocol amendments. Ongoing oversight and effective communication with site personnel are crucial to uphold compliance and guarantee participant safety throughout the study.
By adhering to these guidelines, research teams can effectively navigate the complexities of medical device clinical trial Chile. By prioritizing compliance, research teams not only enhance trial integrity but also pave the way for successful outcomes in the competitive Medtech landscape.
Manage Data and Submit for Regulatory Approval After the Trial
Completing a clinical trial is just the beginning; the path to regulatory approval is where the real challenges lie. After finishing the trial, effective data management and regulatory submission preparation are critical steps:
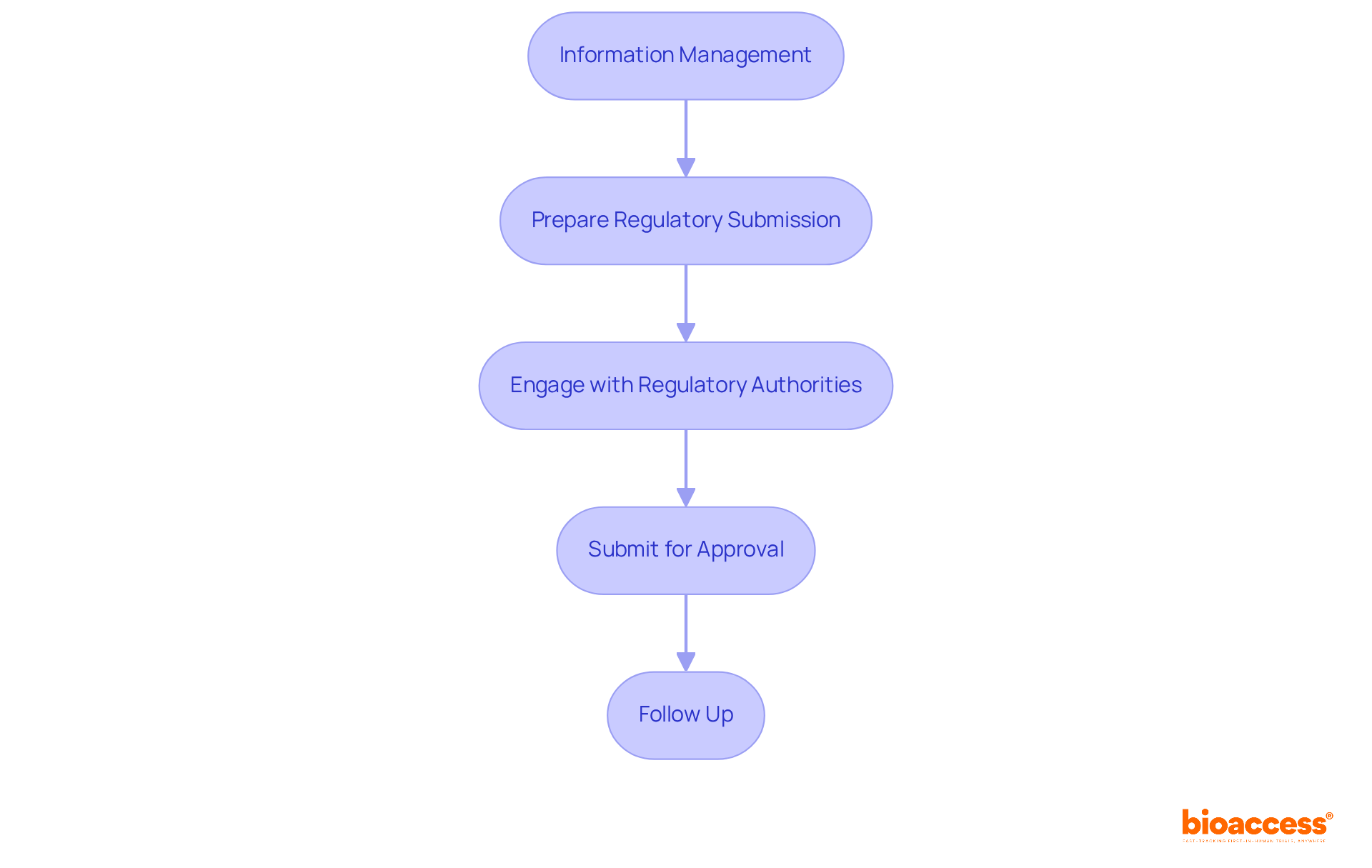
- Information Management: Accurately compile and analyze all trial results. Use statistical software to perform essential analyses and create detailed reports that meet compliance standards.
- Prepare Regulatory Submission: Assemble all necessary documentation for regulatory submission, including clinical study reports, safety information, and efficacy results. Make sure your submission meets the Instituto de Salud Pública (ISP) requirements, which usually means following ICH-GCP standards.
- Engage with Regulatory Authorities: Maintain proactive communication with the ISP throughout the submission process. This engagement can facilitate the resolution of any questions or concerns that may arise, ensuring a smoother review process.
- Submit for Approval: Submit the compiled documentation to the ISP for review. Typically, you can expect an approval timeline of around 30 business days, but keep in mind that this can change depending on how complex your submission is and how complete your data looks.
- Follow Up: After submission, actively follow up with the ISP to monitor the status of your application. Be prepared to address any additional requests for information promptly, which can help expedite the approval process. Your diligence in managing data and engaging with authorities can make all the difference in achieving a successful outcome.
Conclusion
The medical device clinical trial landscape in Chile presents both challenges and unparalleled opportunities for sponsors seeking to accelerate their research. Understanding the essential steps – from regulatory compliance to patient recruitment – empowers stakeholders to position their studies for success in a competitive environment.
Key insights include:
- The necessity of familiarizing oneself with the regulatory landscape governed by the Instituto de Salud Pública (ISP)
- Securing ethical approvals
- Engaging with local experts to streamline the approval process
- Strategic site selection
- Innovative patient recruitment strategies
These are critical for achieving enrollment goals, while robust compliance monitoring ensures trial integrity throughout execution.
In conclusion, by not seizing these strategic advantages, sponsors risk falling behind in the rapidly evolving MedTech landscape. Leveraging Chile’s efficient regulatory pathways, cost-effectiveness, and patient access can significantly accelerate the journey from concept to market, ultimately contributing to advancements in the MedTech field.
Frequently Asked Questions
What is the primary regulatory authority overseeing medical device clinical trials in Chile?
The primary regulatory authority overseeing medical device clinical trials in Chile is the Instituto de Salud Pública (ISP).
What key law should be reviewed when planning a medical device trial in Chile?
It is essential to review the Medical Devices Law and associated regulations, particularly Law N° 20.120, which outlines the requirements for human research.
Is ethical approval mandatory for medical device clinical trials in Chile?
While obtaining ethical approval from an accredited Ethics Committee (EC) is voluntary, it is critical for ensuring compliance with ethical standards before commencing any study.
What documentation is required for a medical device clinical trial in Chile?
Required documentation includes the clinical research protocol, informed consent forms, and any prior approvals, all of which must adhere to ISP standards for a smooth review process.
What is the typical timeline for securing authorization for medical device studies in Chile?
The typical timeline for securing authorization for medical device studies in Chile is around 30 days, although this may vary based on the study’s complexity and the completeness of the submitted documentation.
How can local experts assist in the medical device trial process in Chile?
Collaborating with local compliance consultants or specialized Contract Research Organizations (CROs) like bioaccess® can help navigate the approval landscape and expedite the authorization process.
What advantages does Chile offer for conducting early-stage clinical trials?
Chile offers an advantageous regulatory landscape characterized by efficient approval procedures and a strong healthcare system, which can expedite research studies and enhance competitiveness in the market.
List of Sources
- Understand the Regulatory Landscape for Medical Device Trials in Chile
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- Chile (ISP – Instituto de Salud Pública) – PharmaRegulatory.in – India’s Regulatory Knowledge Hub (https://pharmaregulatory.in/country-specific-regulatory-affairs/chile-isp-instituto-de-salud-publica)
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Latin America the ‘Hidden Gem’ for First-in-Human Medical Device Trials (https://clinicalresearchnewsonline.com/news/2025/08/14/latin-america-the-hidden-gem-for-first-in-human-medical-device-trials)
- Prepare for Your Trial: Site Selection and Patient Recruitment Strategies
- Evolving Trends in Latin American Participation in Global Oncology Clinical Trials: A Decade of Phase III Activity (2013-2022) | JCO Global Oncology (https://ascopubs.org/doi/10.1200/GO-25-00411)
- Clinical Trial Site Selection: Key Challenges & Solutions for Phase 3 Success (https://precisionformedicine.com/blog/clinical-trial-site-selection-key-challenges-solutions-for-phase-3-success)
- Patient Recruitment and Retention Considerations for Clinical Trials in Latin America (https://clinicaltrialsarena.com/comment/patient-recruitment-retention-considerations)
- Enhancing site selection strategies in clinical trial recruitment using real-world data modeling – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10927105)
- Clinical studies: the challenge of patient recruitment (https://servier.com/en/newsroom/clinical-studies-patient-recruitment)
- Execute the Trial: Operational Processes and Compliance Monitoring
- Medical Device Clinical Trials: An Overview [+Types] (https://greenlight.guru/blog/medical-device-clinical-trials)
- Complete Guide to EDC Systems in Clinical Trials (2026) (https://triticon.com/blogs/usa/complete-guide-edc-systems-2026)
- Importance of Compliance in Clinical Trials (https://primerevivalresearch.com/blogs/importance-of-compliance-in-clinical-trials)
- Site Training for Clinical Trial success | ICON plc (https://iconplc.com/insights/blog/2024/03/25/site-training-clinical-trial-success)
- FDA Guidance on Statistical Methods for Clinical Trials – ECA Academy (https://gmp-compliance.org/gmp-news/fda-guidance-on-statistical-methods-for-clinical-trials)
- Manage Data and Submit for Regulatory Approval After the Trial
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- 2026 Clinical Data Trend Report | Veeva (https://veeva.com/2026-clinical-data-trend-report)
- Clinical Data Management: stakes & regulations (https://efor-group.com/en/clinical-data-management-art-of-managing-clinical-trial-data)
- FDA Guidance on Statistical Methods for Clinical Trials – ECA Academy (https://gmp-compliance.org/gmp-news/fda-guidance-on-statistical-methods-for-clinical-trials)
- 19 Inspirational Quotes About Data | The Pipeline | ZoomInfo (https://pipeline.zoominfo.com/operations/19-inspirational-quotes-about-data)

Leave a Reply