Introduction
Navigating the complexities of ethics committee approval is a critical step for any clinical trial in Belize. Here, safeguarding participant rights and adhering to regulatory standards are paramount. This guide delves into the essential components of the approval process, offering insights into required documentation and best practices that can streamline applications.
However, the road to securing ethics approval is often fraught with challenges. How can researchers effectively overcome these hurdles to ensure their trials not only comply with local regulations but also achieve successful outcomes?
Understand the Importance of Ethics Committee Approval
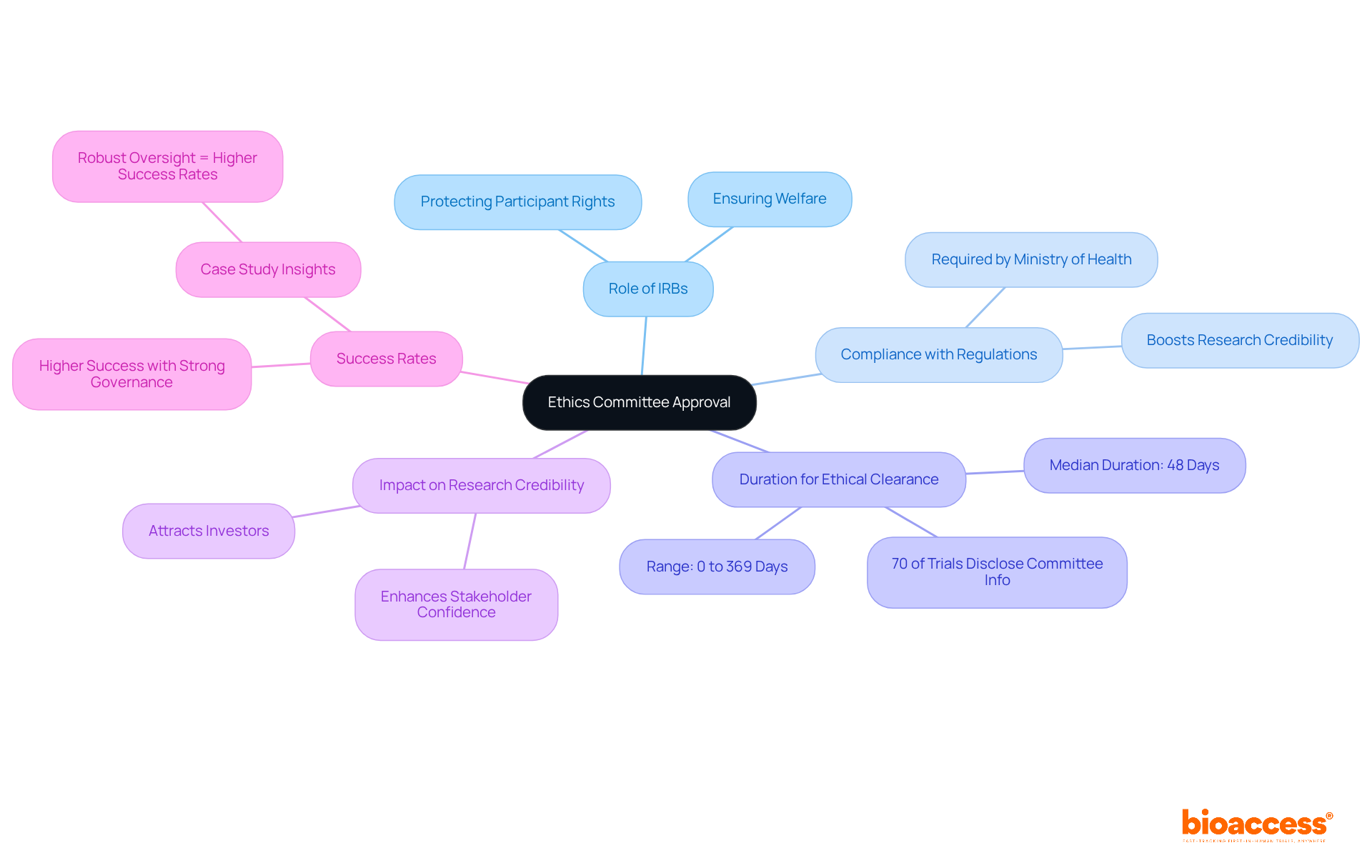
Ethics committee approval is essential for a Belize clinical trial, as Institutional Review Boards (IRBs) play a vital role in protecting participant rights and welfare. Required by the Ministry of Health, all clinical trials involving human subjects must secure ethics committee approval before they can commence. This process not only ensures compliance with local regulations but also significantly boosts the credibility of the research, making it more appealing to potential investors and stakeholders.
Research indicates that the median duration for ethical clearance is 48 days, with a range from 0 to 369 days, and 70% of trials disclose committee information. Notably, trials with strong governance oversight tend to achieve higher success rates, underscoring the critical importance of these endorsements in the clinical research landscape. A case study revealed that trials with robust oversight had a success rate significantly higher than those without, highlighting the essential role of ethical review in achieving research objectives.
By leveraging insights from bioaccess’s Global Trial Accelerators™, medtech startups can navigate these complexities more effectively. This ensures they meet regulatory requirements while enhancing their market access strategies. Collaboration and strategic partnerships are key to overcoming challenges in clinical research, paving the way for successful outcomes.
Gather Required Documentation for Submission
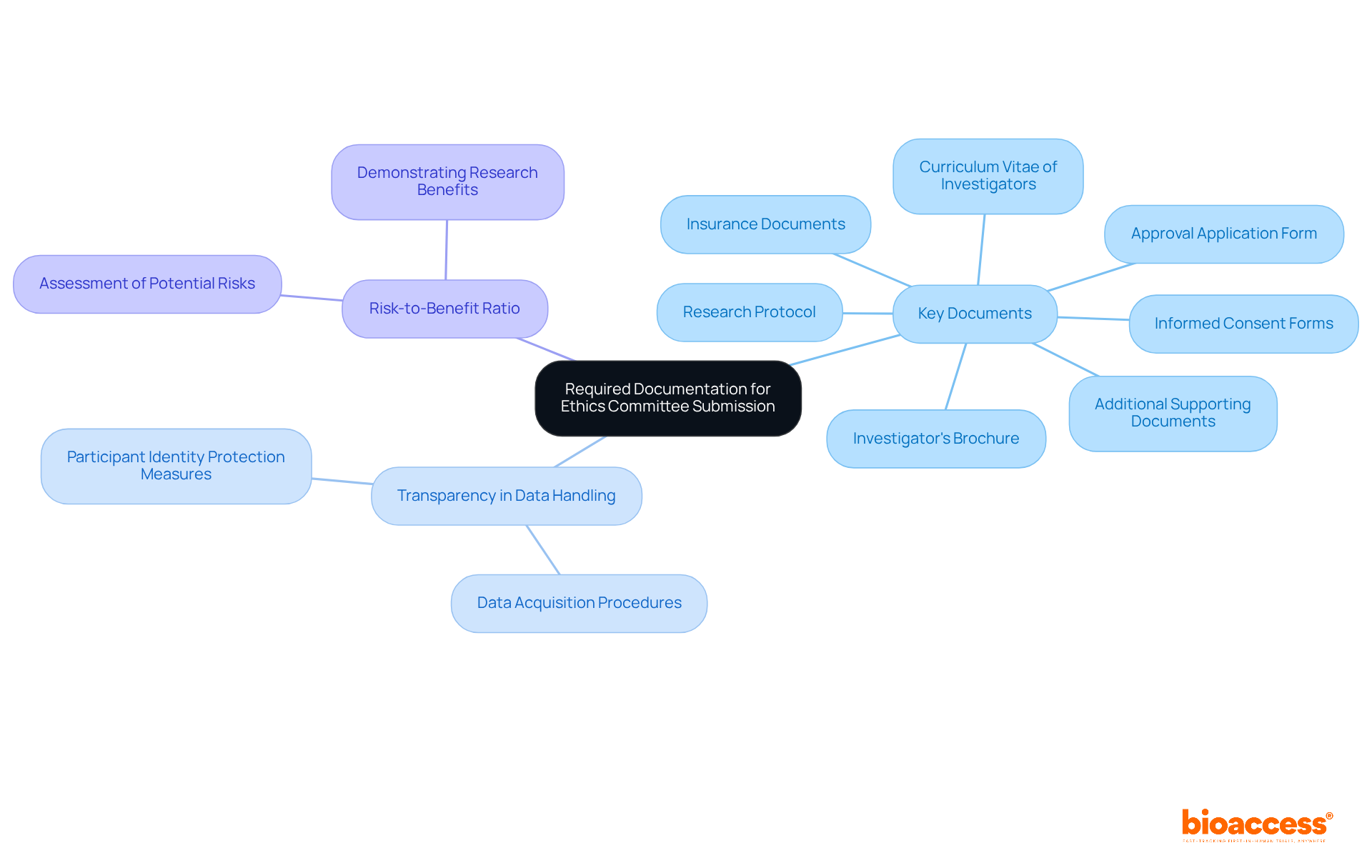
To successfully submit your application for the ethics committee approval Belize clinical trial, it is essential to gather several key documents. These typically include:
- Research Protocol: A comprehensive description of the study, detailing objectives, methodology, and participant criteria.
- Informed Consent Forms: Documents that clearly explain the study to participants and secure their consent to participate. As highlighted, “Ultimately, one of the key criteria in the ethical authorization application process is ensuring that participants are well-informed about the research procedures.”
- Investigator’s Brochure: Information regarding the investigational product, including safety and efficacy data.
- Curriculum Vitae of Investigators: Evidence of the qualifications and experience of the research team.
- Approval Application Form: A specific document supplied by the review board that outlines the study details.
- Insurance Documents: Proof of insurance coverage for participants in the event of trial-related injuries.
- Any Additional Supporting Documents: This may include letters of support from institutions or prior ethics endorsements, if applicable.
In addition to gathering these documents, ensuring transparency in data handling is crucial. Researchers should clearly outline how data will be acquired or transferred from participants to the researcher, along with the measures taken to protect participant identities. This transparency is vital for obtaining ethical consent.
Furthermore, addressing the risk-to-benefit ratio of the study is essential. Researchers must demonstrate that the potential benefits of the research outweigh any risks to participants. This consideration not only increases the chance of ethics committee approval for the Belize clinical trial but also demonstrates a commitment to ethical research standards.
Ensure that all documents are complete and formatted according to the group’s guidelines to facilitate a smooth evaluation. Following these best practices not only increases the chances of acceptance but also reflects a dedication to ethical research standards.
Submit Your Application to the Ethics Committee
To initiate the ethics committee approval Belize clinical trial process, it’s essential to follow a structured approach for presenting your application. Here are the key steps to ensure a smooth submission:
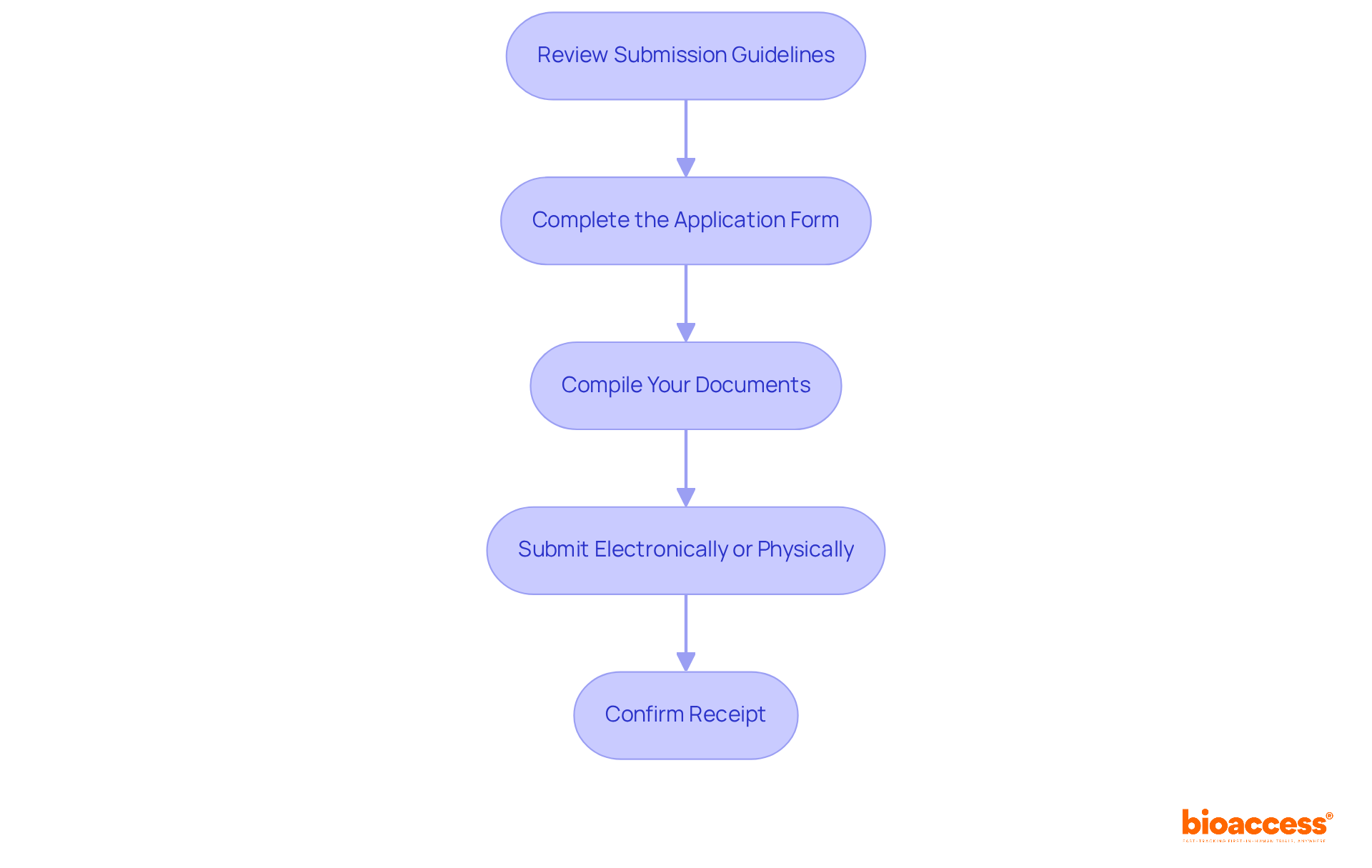
- Review Submission Guidelines: Start by visiting the committee’s official website or contacting them directly to understand their specific submission requirements and deadlines. Familiarizing yourself with these details will help you meet any unique criteria.
- Complete the Application Form: Fill out the ethics approval application form accurately, ensuring that every section is thoroughly completed. Incomplete forms can lead to delays in the review process.
- Compile Your Documents: Organize all necessary documents in the order specified by the group. Each document should be clearly labeled and easy to read, facilitating a smoother review. Be mindful that applications often encounter issues due to insufficient information or inconsistencies, making thoroughness crucial.
- Submit Electronically or Physically: Depending on the group’s preferences, submit your application either electronically through their online portal or in person at their office. If submitting physically, ensure you have multiple copies ready if required.
- Confirm Receipt: After submission, it’s vital to confirm with the committee that your application has been received. Additionally, inquire about the anticipated review schedule for the ethics committee approval Belize clinical trial, which typically spans from 1 to 3 months, with governance endorsements averaging around 28 days following the pandemic.
By following these steps, you can enhance the efficiency of your application submission and increase the likelihood of a timely review.
Navigate Common Challenges in the Approval Process
Navigating the ethics validation procedure can be challenging, yet understanding common issues and strategies can significantly ease the process. Here are some key considerations:
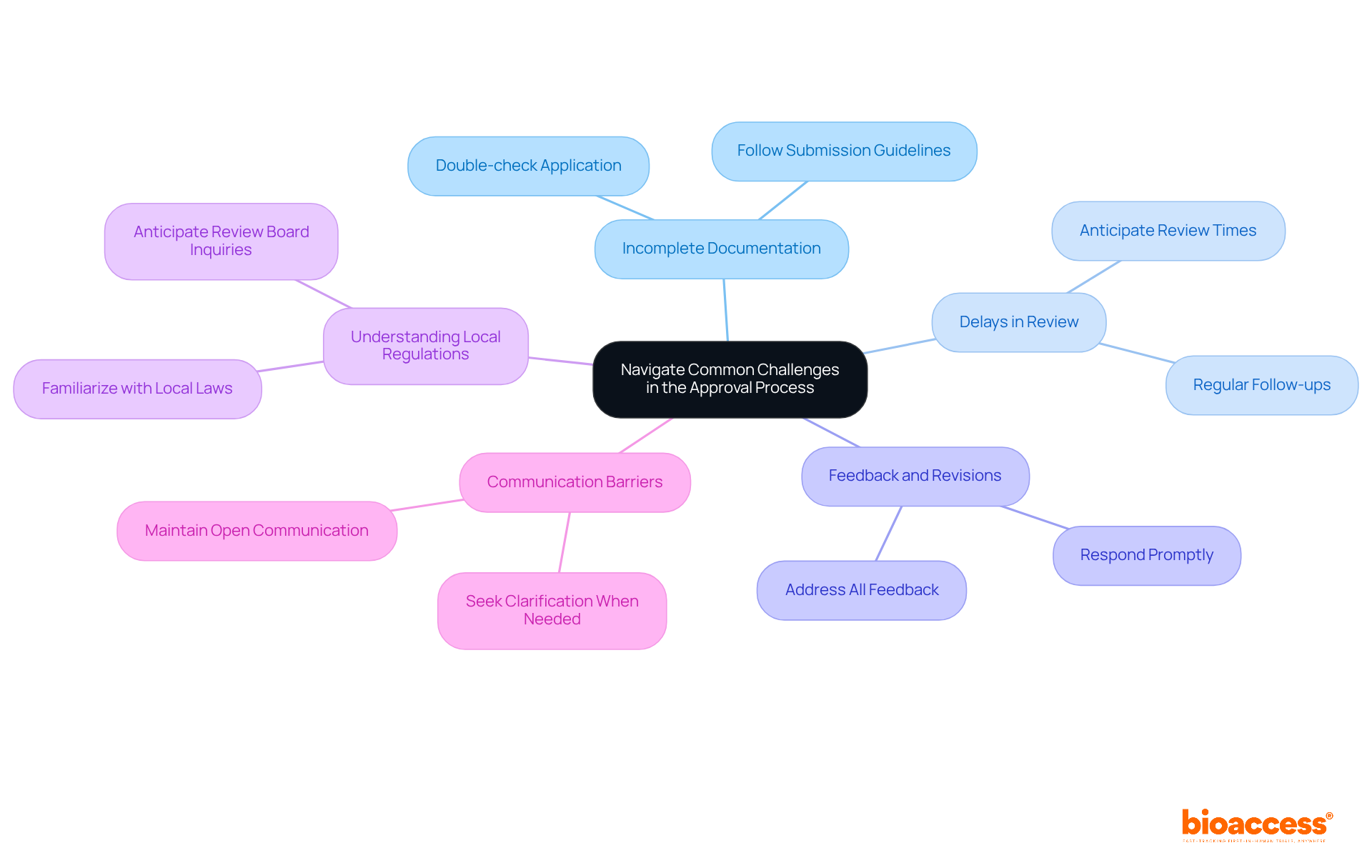
- Incomplete Documentation: It’s crucial to submit all required documents in full. Many delays arise when investigators overlook submission guidelines, leading to setbacks. To avoid this, double-check your application against the checklist to ensure nothing is missed.
- Delays in Review: Anticipate potential delays in the review process. The average time to secure IRB approval can vary dramatically, with some protocols taking up to 396 days. Regularly follow up with the ethics group to check on your application’s status; proactive communication can help mitigate extended waiting times.
- Feedback and Revisions: When the review group requests modifications, respond promptly and thoroughly. Delays often stem from reviewers not providing timely feedback or missing meetings. Addressing all feedback not only shows your commitment to ethical standards but also helps expedite the review process.
- Understanding Local Regulations: Familiarize yourself with Belize’s specific regulations regarding clinical trials. This knowledge can help you anticipate inquiries or issues from the review board. Grasping the local context is essential, as variations in IRB functioning can impact the timeline for protocol approval.
- Communication Barriers: Keep communication lines open with the ethics committee. If you face challenges, don’t hesitate to reach out for clarification or assistance. Effective communication can resolve misunderstandings and facilitate a smoother review process.
By being proactive and prepared, you can effectively navigate these challenges and advance your clinical trial.
Conclusion
Securing ethics committee approval is a crucial step in conducting clinical trials in Belize. It not only safeguards participant rights but also enhances the integrity of research. This process, mandated by the Ministry of Health, ensures compliance with local regulations and elevates the study’s credibility, making it more appealing to potential investors and stakeholders. Recognizing this significance is the first step toward successful clinical research.
The article outlines essential components for navigating the ethics approval process, including necessary documentation, structured submission steps, and strategies for overcoming common challenges. Key documents such as the research protocol, informed consent forms, and investigator’s brochures are vital for a successful application. Furthermore, adhering to submission guidelines and maintaining open communication with the ethics committee can significantly influence the approval timeline and the overall success of the trial.
Ultimately, the journey to ethics committee approval transcends mere regulatory compliance; it fosters trust and ensures the welfare of participants. By diligently preparing and proactively addressing potential challenges, researchers can pave the way for ethical and impactful clinical trials in Belize. Embracing these practices not only enhances the likelihood of approval but also contributes to the advancement of medical research and innovation in the region.
Frequently Asked Questions
Why is ethics committee approval important for clinical trials in Belize?
Ethics committee approval is essential for protecting participant rights and welfare, ensuring compliance with local regulations, and enhancing the credibility of the research.
What is required for a clinical trial involving human subjects in Belize?
All clinical trials involving human subjects must secure ethics committee approval from the Ministry of Health before they can commence.
How long does the ethical clearance process typically take?
The median duration for ethical clearance is 48 days, with a range from 0 to 369 days.
What percentage of clinical trials disclose their ethics committee information?
70% of trials disclose committee information.
How does governance oversight affect the success rates of clinical trials?
Trials with strong governance oversight tend to achieve higher success rates compared to those without, highlighting the importance of ethical review.
What insights can medtech startups leverage to navigate the complexities of clinical trials?
Medtech startups can leverage insights from bioaccess’s Global Trial Accelerators™ to meet regulatory requirements and enhance their market access strategies.
What role do collaboration and strategic partnerships play in clinical research?
Collaboration and strategic partnerships are key to overcoming challenges in clinical research and paving the way for successful outcomes.
List of Sources
- Understand the Importance of Ethics Committee Approval
- Clinical Trial Success Rates: How Many Drugs Make It to Market? (Latest Approval Stats) (https://patentpc.com/blog/clinical-trial-success-rates-how-many-drugs-make-it-to-market-latest-approval-stats)
- Factors influencing the time to ethics and governance approvals for clinical trials: a retrospective cross-sectional survey – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10693024)
- Investigation of ethics approval as part of a research integrity assessment of randomised controlled trials in COVID-19 evidence syntheses: a meta-epidemiological study – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC11934354)
- pubmed.ncbi.nlm.nih.gov (https://pubmed.ncbi.nlm.nih.gov/29394327)
- Gather Required Documentation for Submission
- Comparison of requirements of research ethics committees in 11 European countries for a non-invasive interventional study (https://bmj.com/content/328/7432/140)
- Obtaining Ethical Approval for Academic Research — Indeemo (https://indeemo.com/blog/guide-to-ethics-approval)
- Obtaining Ethics Approval, Data Management, and Budget Formulation (https://radiologykey.com/obtaining-ethics-approval-data-management-and-budget-formulation)
- Ethics Quotes and Morality Quotes Related to Decision Making (https://decision-making-solutions.com/ethics_quotes.html)
- Submit Your Application to the Ethics Committee
- Ultimate Guide to ISO 14155:2020 for Medical Devices (https://greenlight.guru/blog/iso-14155)
- Global comparison of research ethical review protocols: insights from an international research collaborative – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC11975204)
- How to write a successful ethics application (https://researchwhisperer.org/2019/10/29/ethics-application)
- Overall median (range) times for site activation, ethics approval and… (https://researchgate.net/figure/Overall-median-range-times-for-site-activation-ethics-approval-and-governance_fig1_376166321)
- Navigate Common Challenges in the Approval Process
- An examination of timelines in the expedited ethics review process at the University of KwaZulu-Natal, biomedical research ethics committee. (https://researchspace.ukzn.ac.za/items/3aacd327-1d51-478d-9b2c-57b9fefaa14b)
- Delays and diversity in the practice of local research ethics committees – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC1377056)
- Reducing the Time for IRB Reviews: A Case Study (https://srainternational.org/blogs/martha-jack/2017/09/15/reducing-the-time-for-irb-reviews)
- Understanding constraints and enablers of turnaround time for ethics review: The case of institutional review boards in Tanzania – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC8530844)
- Challenges in institutional ethical review process and approval for international multicenter clinical studies in lower and middle-income countries: the case of PARITY study – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC11577162)

Leave a Reply