Introduction
As Brazil positions itself as a pivotal player in first-in-human trials, the stakes for clinical research organizations have never been higher. This country leverages its diverse demographics and advanced healthcare infrastructure to provide a rich pool of treatment-naive patients, essential for generating high-quality clinical data. Additionally, Brazil boasts significant cost efficiencies and expedited regulatory processes that can reduce timelines by up to 65%. However, navigating the evolving landscape of regulations and competitive pressures poses significant challenges for clinical research organizations in Brazil. To thrive in this dynamic environment, organizations must develop strategic approaches that leverage Brazil’s unique advantages.
Explore Brazil’s Strategic Advantages for First-in-Human Trials
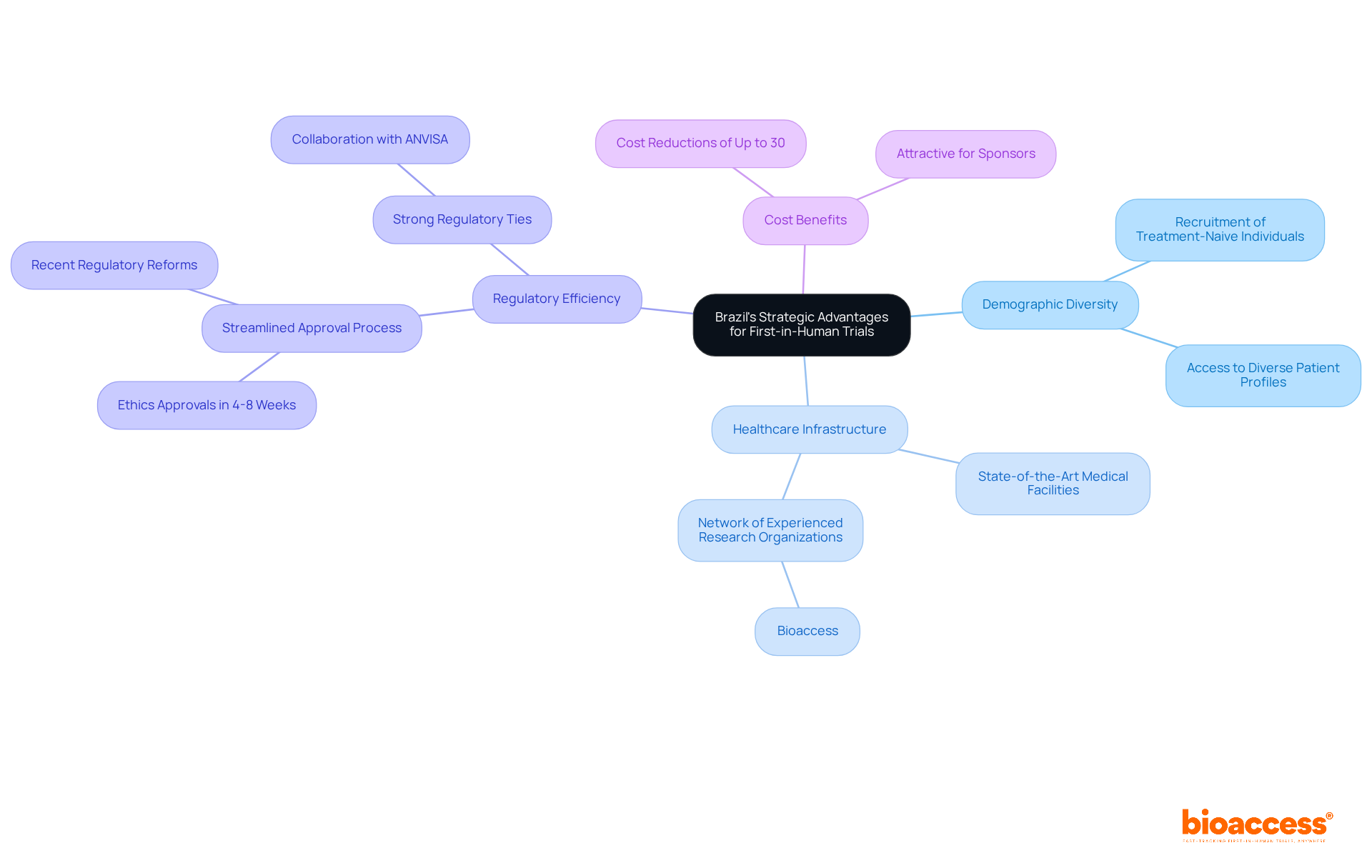
This nation stands out as a premier destination for CRO Brazil first-in-human clinical studies, driven by its unique demographic and healthcare advantages. The country’s demographic diversity facilitates the recruitment of treatment-naive individuals, a vital component for ensuring the integrity of FIH studies. With over 210 million residents, the urban areas of this nation offer access to a diverse array of profiles, improving the generalizability of research outcomes.
Furthermore, the healthcare system in the country is well-equipped with state-of-the-art medical facilities and a network of seasoned research organizations like Bioaccess, which specializes in CRO Brazil first-in-human studies. Bioaccess® leverages its strong regulatory ties with ANVISA and INVIMA to drive efficient patient recruitment and retention, essential elements for the success of early-stage studies. Recent regulatory reforms, such as Law 14.874/2024, have streamlined the approval process. Now, Bioaccess® can secure ethics approvals in just 4-8 weeks, a stark contrast to the typical 6-12 months in the US/EU. This efficiency, along with the cost reductions of up to 30% compared to US/EU studies, makes this nation an increasingly appealing choice for sponsors seeking to accelerate their clinical development timelines.
With these advancements, sponsors can not only expedite their timelines but also enhance the quality of their clinical research outcomes.
Leverage Cost Efficiency and Speed in Brazilian CROs
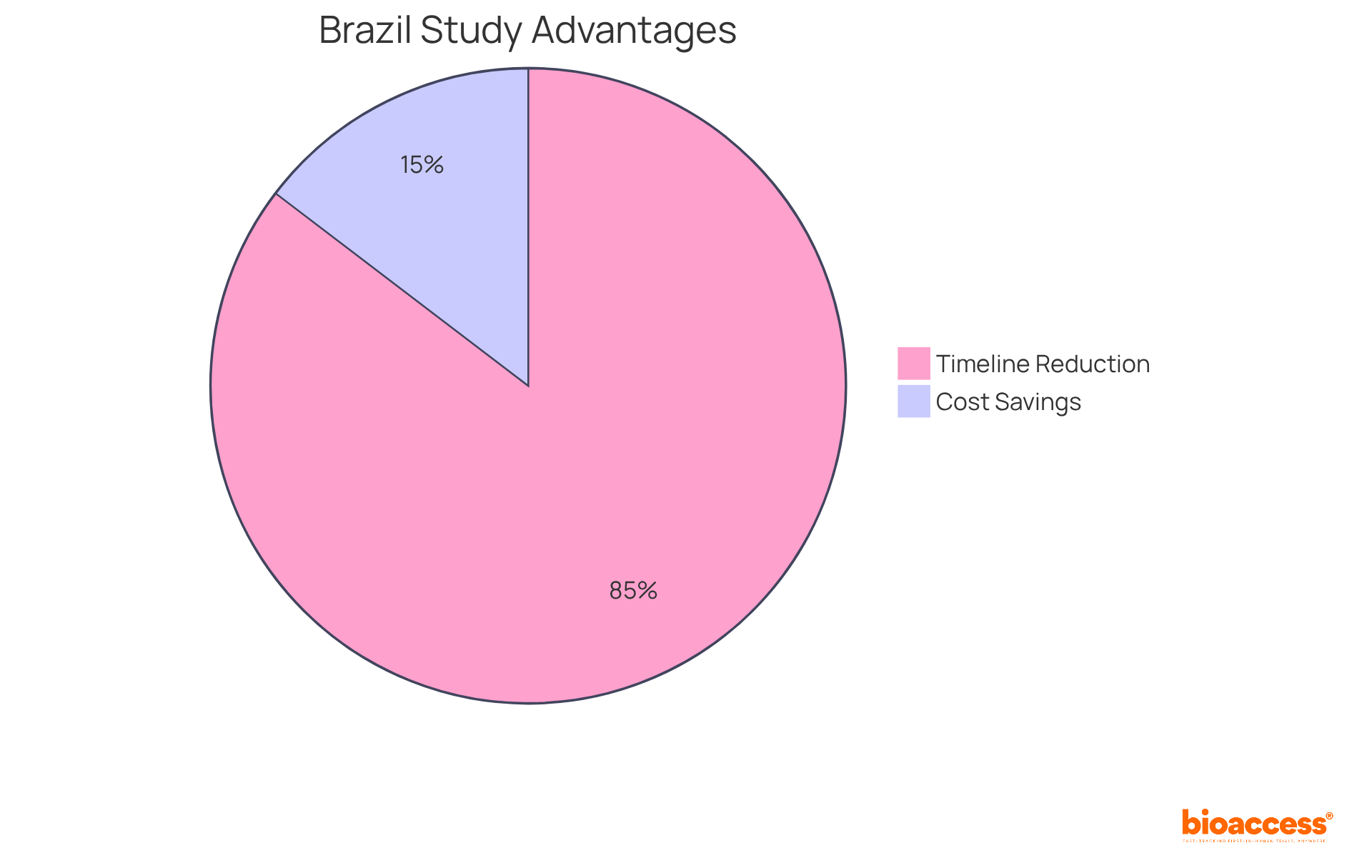
Conducting first-in-human studies in Brazil presents a compelling alternative to the high costs and lengthy timelines of conventional markets like the U.S. and EU. Clinical studies in this country generally cost 25-35% less than comparable research in these areas, primarily due to reduced site fees and operational costs. For instance, the typical expense per patient in Brazilian medical device studies varies from $15,000 to $35,000, whereas in the U.S., this can rise to between $40,000 and $75,000.
Moreover, Brazil has significantly reduced approval timelines, making it more attractive for clinical studies. Recent legislative modifications, including Law #14.874/2024, have cut the average approval duration for studies from 265 days to around 90 days. This faster timeline is vital for startups looking to generate data quickly, attract investors, and bring their products to market.
How can sponsors leverage insights and market access strategies from bioaccess’s Global Trial Accelerators™ to optimize resources and navigate the regulatory landscape effectively? By doing so, they can ensure compliance with ANVISA regulations and ICH-GCP standards. This positions Brazil as an increasingly appealing location for CRO Brazil first-in-human studies, especially for MedTech, Biopharma, and Radiopharmaceutical firms aiming to expedite their development processes.
With these advantages, Brazil is poised to become a key player in the global MedTech landscape, inviting innovators to seize the opportunity for growth and collaboration.
Navigate the Regulatory Landscape for Swift Approvals
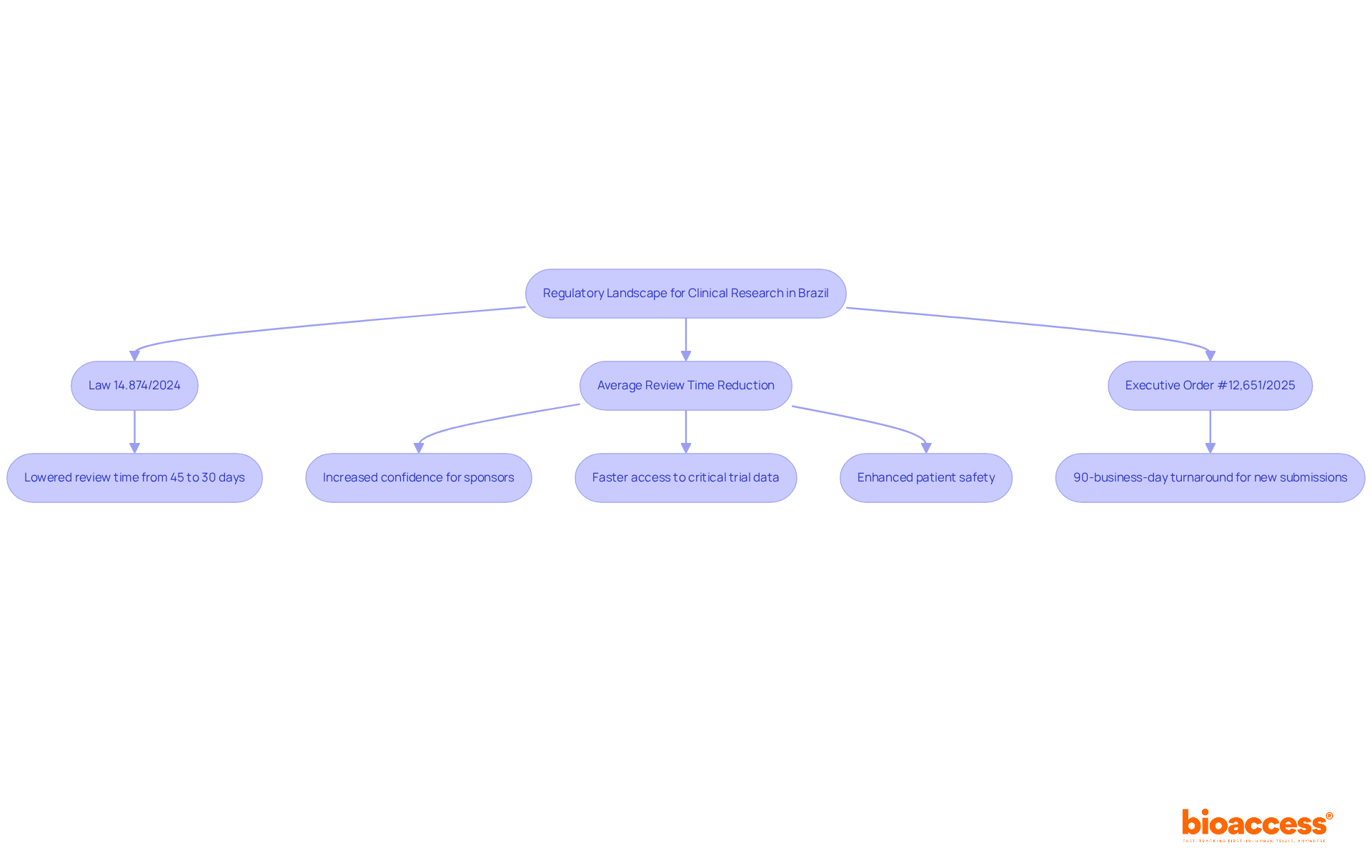
The evolving regulatory landscape in Brazil presents both challenges and opportunities for clinical research sponsors. The regulatory framework for clinical studies in the country is supervised by ANVISA (Agência Nacional de Vigilância Sanitária), which has implemented significant reforms to improve the approval process. The introduction of Law 14.874/2024 aligns Brazil’s regulations with international Good Clinical Practice (GCP) standards, thus creating a more predictable and efficient pathway for CRO Brazil first-in-human studies.
Under these new regulations, the average review time for ethical approval has been lowered from 45 days to around 30 days, allowing sponsors to commence studies more swiftly. Additionally, submitting research study applications alongside other regulatory filings boosts operational efficiency. Navigating the complexities of regulatory approval can often be daunting for sponsors, but this regulatory clarity safeguards patient safety and empowers them to plan their studies with increased confidence, knowing that the approval process is both transparent and expedited.
The recent Executive Order #12,651/2025 commits to a 90-business-day turnaround for responses to new study submissions, further enhancing this landscape. This initiative seeks to greatly enhance the number of medical studies performed in Brazil, with forecasts indicating that the count of these studies could double in the upcoming years. By effectively navigating this evolving regulatory environment, CROs can ensure compliance with ICH-GCP standards while minimizing delays, ultimately leading to faster access to critical trial data. With bioaccess®’s Global Trial Accelerators™, sponsors can utilize these benefits to improve their market access strategies and achieve successful results in their studies. By leveraging these regulatory advancements, sponsors can not only expedite their studies but also enhance their competitive edge in the market.
Access Treatment-Naive Patient Cohorts for Enhanced Outcomes
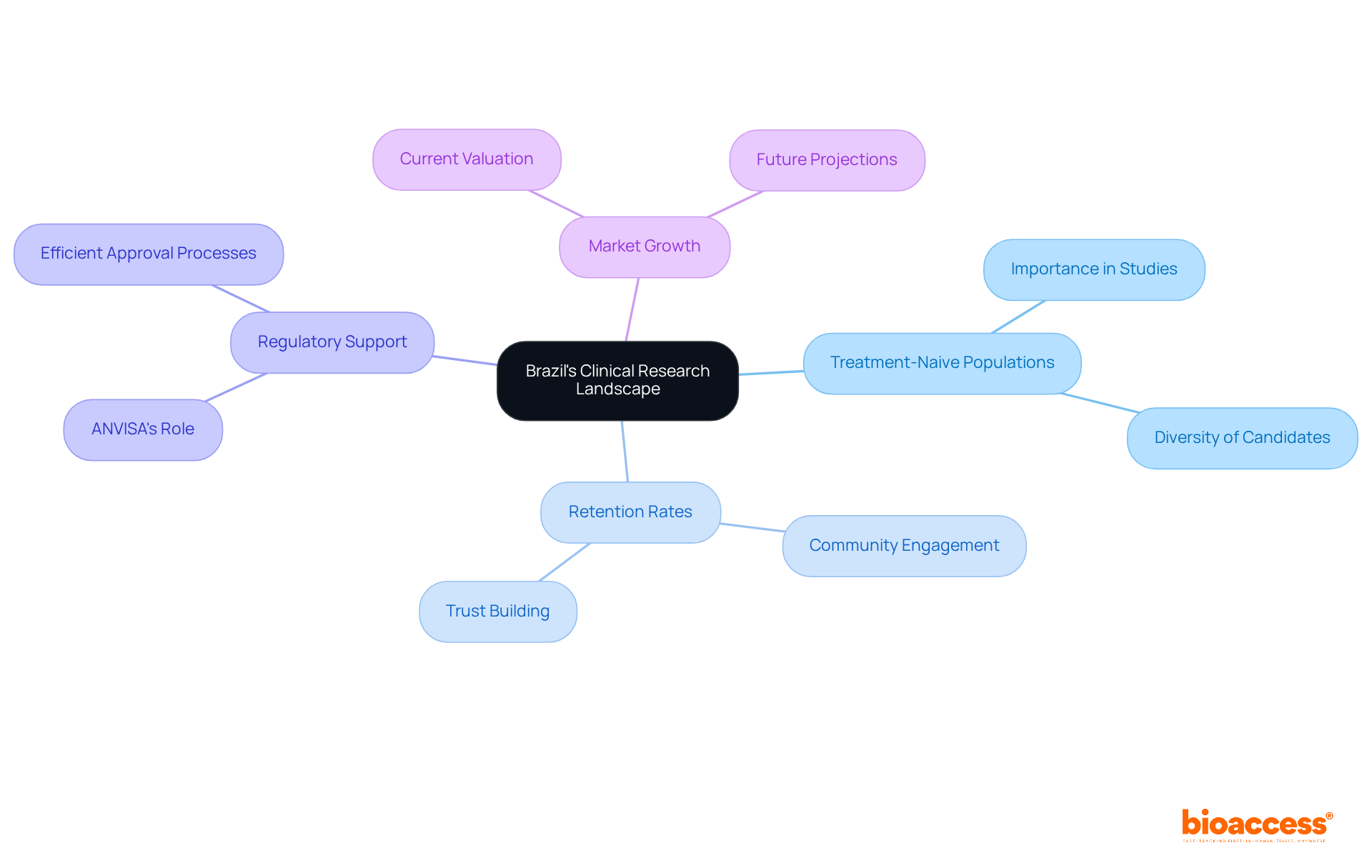
Brazil’s unique position as a hub for clinical research is exemplified by its role in CRO Brazil first-in-human studies, stemming from its access to treatment-naive populations, which is a critical factor. These individuals, who have not previously received treatment for their conditions, are essential for evaluating the safety and efficacy of new therapies. Brazil’s diverse population, with its rich mix of ethnicities and health profiles, provides a vital pool of treatment-naive candidates essential for high-quality research data.
Recruiting individuals who have not undergone treatment significantly enhances the credibility of study results and contributes to impressive retention rates. Brazilian clinical studies often achieve retention rates exceeding 95%, a figure that surpasses many other regions. This impressive retention stems from how CROs in Brazil engage with communities and educate patients, building trust and commitment. By leveraging these treatment-naive groups, sponsors of CRO Brazil first-in-human studies can ensure that their studies generate robust and reliable data, ultimately accelerating regulatory approvals and enabling faster market entry.
Moreover, the Brazilian regulatory landscape, governed by authorities such as ANVISA, supports efficient approval processes, further enhancing the attractiveness of conducting trials in this region. This regulatory framework not only accelerates approvals but also elevates the quality of research in Brazil. The Innovation Runway by bioaccess® has utilized this environment, allowing MedTech, Biopharma, and Radiopharma startups to achieve milestones 40% faster. The Brazil clinical trials market was valued at USD 571.3 million in 2025 and is projected to reach USD 1,033.6 million by 2033, reflecting the growing demand for clinical research in this vibrant market. As the demand for clinical research grows, Brazil’s landscape offers unparalleled opportunities for innovation and collaboration.
Conclusion
Why is Brazil becoming the go-to destination for first-in-human (FIH) trials? Brazil has emerged as a leading choice, offering a unique blend of demographic diversity, cost efficiency, and an evolving regulatory environment that benefits sponsors in the MedTech and Biopharma sectors. Contract research organizations (CROs) can tap into Brazil’s rich patient population and streamlined processes. This allows them to conduct high-quality clinical studies more effectively than in traditional markets like the U.S. and EU.
The advantages of conducting FIH trials in Brazil are compelling. Access to a treatment-naive patient population, significantly reduced costs – up to 30% lower than in the U.S. and EU – and expedited approval timelines are just a few. Recent legislative reforms have allowed for ethics approvals in as little as 4-8 weeks. With Brazil’s strong healthcare infrastructure and the know-how of organizations like Bioaccess®, sponsors can easily navigate the regulatory landscape, enhancing the overall quality and reliability of clinical research outcomes.
As demand for clinical trials rises, have you considered how Brazil could be your strategic hub for innovation and collaboration in early-stage research? By choosing Brazil for FIH studies, sponsors not only benefit from faster timelines and lower costs but also position themselves to capitalize on the rich opportunities this vibrant market offers. Ignoring Brazil’s potential could mean missing out on groundbreaking advancements in clinical research.
Frequently Asked Questions
Why is Brazil considered a premier destination for first-in-human (FIH) clinical trials?
Brazil’s demographic diversity and well-equipped healthcare system make it an attractive location for FIH clinical trials. The country has over 210 million residents, allowing for the recruitment of treatment-naive individuals, which is essential for the integrity of FIH studies.
What advantages does Brazil offer in terms of patient recruitment for clinical studies?
The urban areas of Brazil provide access to a diverse array of profiles, improving the generalizability of research outcomes. This demographic diversity facilitates efficient recruitment of treatment-naive individuals, which is crucial for FIH trials.
How does the healthcare system in Brazil support early-stage clinical trials?
Brazil’s healthcare system is equipped with state-of-the-art medical facilities and a network of experienced research organizations like Bioaccess, which specializes in FIH studies. These organizations leverage strong regulatory ties with ANVISA and INVIMA to enhance patient recruitment and retention.
What recent regulatory reforms have impacted the approval process for clinical trials in Brazil?
Recent reforms, particularly Law 14.874/2024, have streamlined the approval process, allowing organizations like Bioaccess to secure ethics approvals in just 4-8 weeks, compared to the typical 6-12 months in the US and EU.
How does the cost of conducting clinical trials in Brazil compare to the US and EU?
Conducting clinical trials in Brazil can result in cost reductions of up to 30% compared to studies in the US and EU, making it a more appealing choice for sponsors looking to accelerate their clinical development timelines.
What benefits do sponsors gain by conducting FIH studies in Brazil?
Sponsors can expedite their clinical development timelines, enhance the quality of research outcomes, and benefit from efficient patient recruitment and retention due to the country’s regulatory advantages and demographic diversity.
List of Sources
- Explore Brazil’s Strategic Advantages for First-in-Human Trials
- Brazil Clinical Trials Market Size & Future Growth 2035 (https://expertmarketresearch.com/reports/brazil-clinical-trials-market)
- Brazil Clinical Trials Market Size & Outlook, 2033 (https://grandviewresearch.com/horizon/outlook/clinical-trials-market/brazil)
- Brazil Clinical Trials Market Size, Share | Analysis Report [2032] (https://fortunebusinessinsights.com/brazil-clinical-trials-market-112310)
- Leverage Cost Efficiency and Speed in Brazilian CROs
- Brazil Clinical Trials Market Size & Outlook, 2033 (https://grandviewresearch.com/horizon/outlook/clinical-trials-market/brazil)
- Unlocking Brazil’s Clinical Trial Opportunity: A Strategic Roadmap for Healthcare Leaders (https://lek.com/insights/life-sciences-pharma/unlocking-brazils-clinical-trial-opportunity-strategic-roadmap)
- Brazil Clinical Trials Market Size, Share | Analysis Report [2032] (https://fortunebusinessinsights.com/brazil-clinical-trials-market-112310)
- Navigate the Regulatory Landscape for Swift Approvals
- New regulations for clinical research in Brazil | Licks Attorneys (https://lickslegal.com/post/new-regulations-for-clinical-research-in-brazil)
- REBEC (https://ensaiosclinicos.gov.br/news/566)
- How Brazil’s New Law Is Redefining The Country’s Clinical Research (https://clinicalleader.com/doc/how-brazil-s-new-law-is-redefining-the-country-s-clinical-research-0001)
- Access Treatment-Naive Patient Cohorts for Enhanced Outcomes
- Unlocking Brazil’s Clinical Trial Opportunity: A Strategic Roadmap for Healthcare Leaders (https://lek.com/insights/life-sciences-pharma/unlocking-brazils-clinical-trial-opportunity-strategic-roadmap)
- Clinical outcomes of dolutegravir treatment in people living with HIV in Brazil: protocol for the CODE cohort – BMC Infectious Diseases (https://link.springer.com/article/10.1186/s12879-025-11700-0)
- Brazil Clinical Trials Market Size & Outlook, 2033 (https://grandviewresearch.com/horizon/outlook/clinical-trials-market/brazil)
- Brazil Clinical Trials Market Size, Share | Analysis Report [2032] (https://fortunebusinessinsights.com/brazil-clinical-trials-market-112310)

Leave a Reply