Introduction
Understanding the IRB approval process is crucial for researchers aiming to navigate Brazil’s complex clinical trial landscape. Researchers can gain invaluable insights into the structured steps necessary for securing approval from the National Commission for Ethics in Research (CONEP) and local ethics committees, which are vital for advancing innovative therapies.
However, as the regulatory environment evolves, these challenges can delay research and hinder the development of new therapies. What strategies can researchers use to tackle these obstacles and make the most of Brazil’s growing clinical research opportunities?
By addressing these issues, researchers can unlock the full potential of Brazil’s clinical research landscape.
Understand the IRB Approval Process in Brazil
In Brazil, navigating the irb approval brazil process is crucial for the success of clinical trials involving human subjects. The National Commission for Ethics in Research (CONEP) serves as the primary regulatory authority, tasked with assessing the moral dimensions of research proposals. Before submitting an application to CONEP, researchers must adhere to the guidelines established by the Brazilian Health Surveillance Agency (ANVISA), which plays a vital role in the registration and regularization of medical devices.
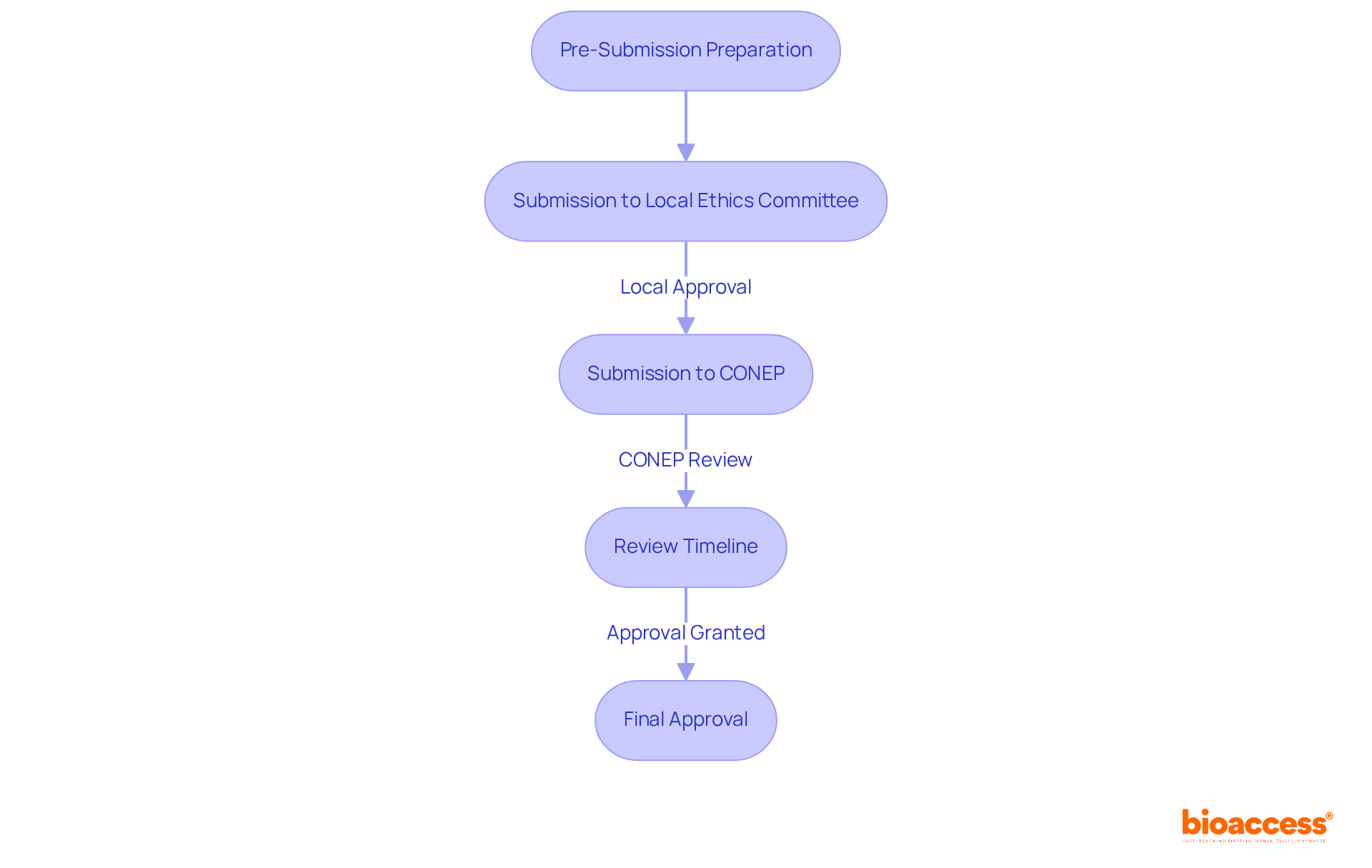
Understanding the irb approval brazil process is essential for researchers aiming to conduct clinical trials. It typically involves several key steps:
- Pre-Submission Preparation: Familiarize yourself with the moral guidelines and requirements established by CONEP and ANVISA. This includes understanding essential documentation, such as the study protocol and informed consent forms, along with considerations specific to your study.
- Submission to Local Ethics Committee: Prior consent from a local ethics committee (CEP) is mandatory before submitting to CONEP. This committee reviews your study’s ethical implications and provides essential feedback, ensuring that all ethical standards are met. Notably, the review period for local ethics committee opinions is now capped at 30 days, significantly reducing previous timelines.
- Submission to CONEP: After obtaining local approval, submit your application to CONEP for further review related to irb approval brazil. This submission must include all relevant documentation, including the study protocol, informed consent forms, and any additional materials required by CONEP.
- Review Timeline: CONEP’s evaluation can span from 30 to 90 days, depending on study complexity, which can be a significant hurdle for researchers. Recent changes, particularly the Executive Order #12,651/2025, have streamlined this approach, enhancing Brazil’s competitiveness in global clinical research by simplifying regulatory pathways and improving participant protections.
Mastering this process not only ensures compliance but also speeds up your clinical trial timeline, a crucial advantage in today’s competitive landscape. By leveraging bioaccess®’s Global Trial Accelerators™ services, researchers can navigate Brazil’s favorable regulatory environment more effectively, achieving faster approvals and access to a diverse patient population. This ultimately accelerates the path to market for innovative therapies. With the projected doubling of clinical studies in Brazil, now is the time for MedTech, Biopharma, and Radiopharma startups to seize this opportunity and lead in clinical research innovation.
Gather Required Documentation for IRB Submission
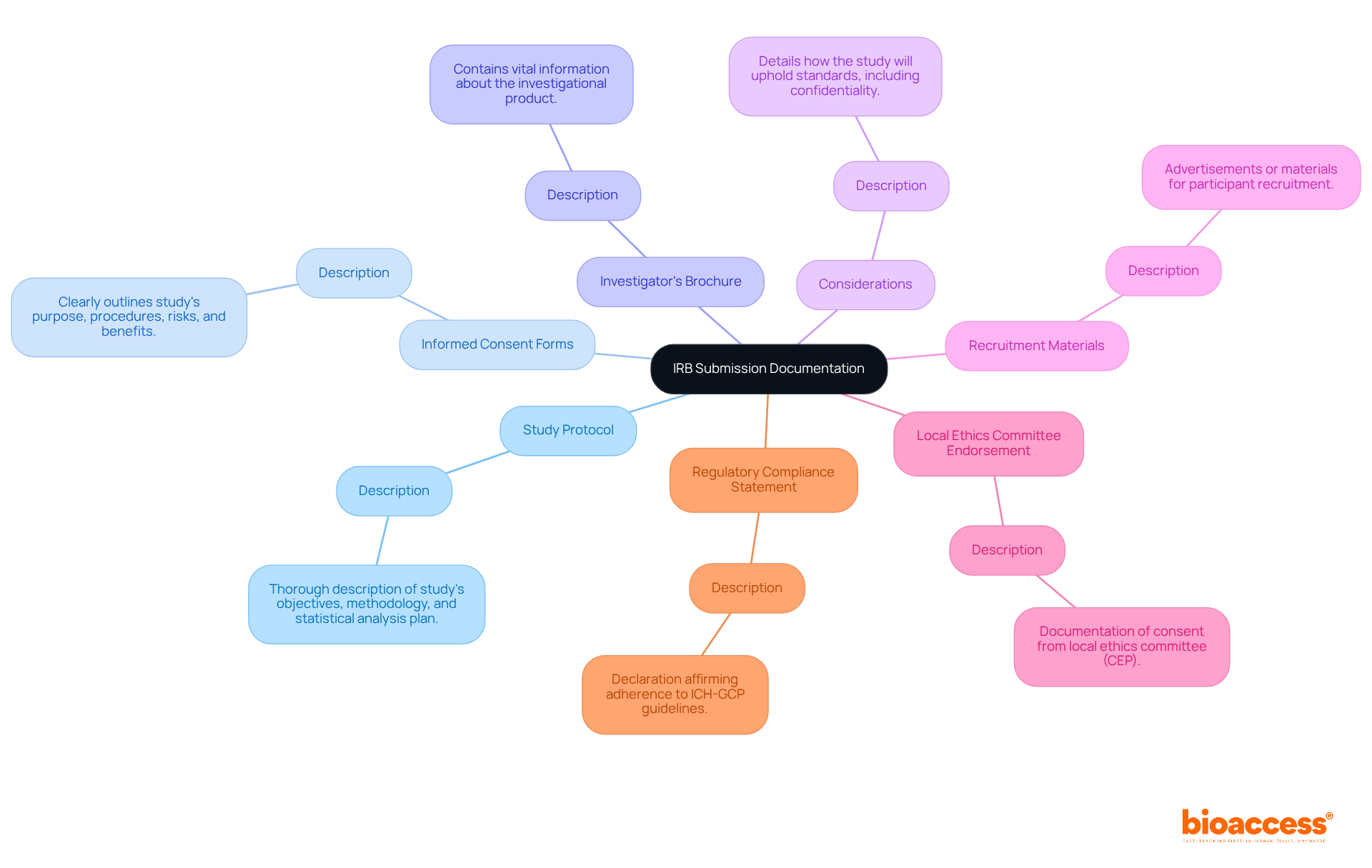
Submitting your IRB approval in Brazil can feel overwhelming, but with the right preparation, you can navigate the IRB approval Brazil process with confidence. To ensure compliance with regulatory standards, it’s crucial to gather a comprehensive collection of documents. Here’s a checklist of essential documents required for the IRB application process:
- Study Protocol: A thorough description of the study’s objectives, methodology, and statistical analysis plan, ensuring clarity and precision.
- Informed Consent Forms: Well-structured consent forms that clearly outline the study’s purpose, procedures, risks, and benefits, ensuring participants are fully informed before enrollment.
- Investigator’s Brochure: A document containing vital information about the investigational product, including preclinical and clinical data that supports its use in the trial.
- Considerations: A statement detailing how the study will uphold standards, including measures for participant confidentiality and data protection.
- Recruitment Materials: Any advertisements or materials intended for participant recruitment, ensuring compliance with ethical guidelines and local regulations.
- Local Ethics Committee Endorsement: Documentation of consent from the local ethics committee (Comitê de Ética em Pesquisas, CEP) prior to submission to the National Ethics Committee (CONEP).
- Regulatory Compliance Statement: A declaration affirming that the study will adhere to ICH-GCP guidelines and any other applicable regulations.
By meticulously preparing these documents, you can significantly reduce the risk of delays and ensure compliance with regulatory standards, especially in relation to irb approval Brazil. Ultimately, thorough preparation not only streamlines your submission but also enhances the integrity of your research project.
Submit Your IRB Application and Navigate the Review Process
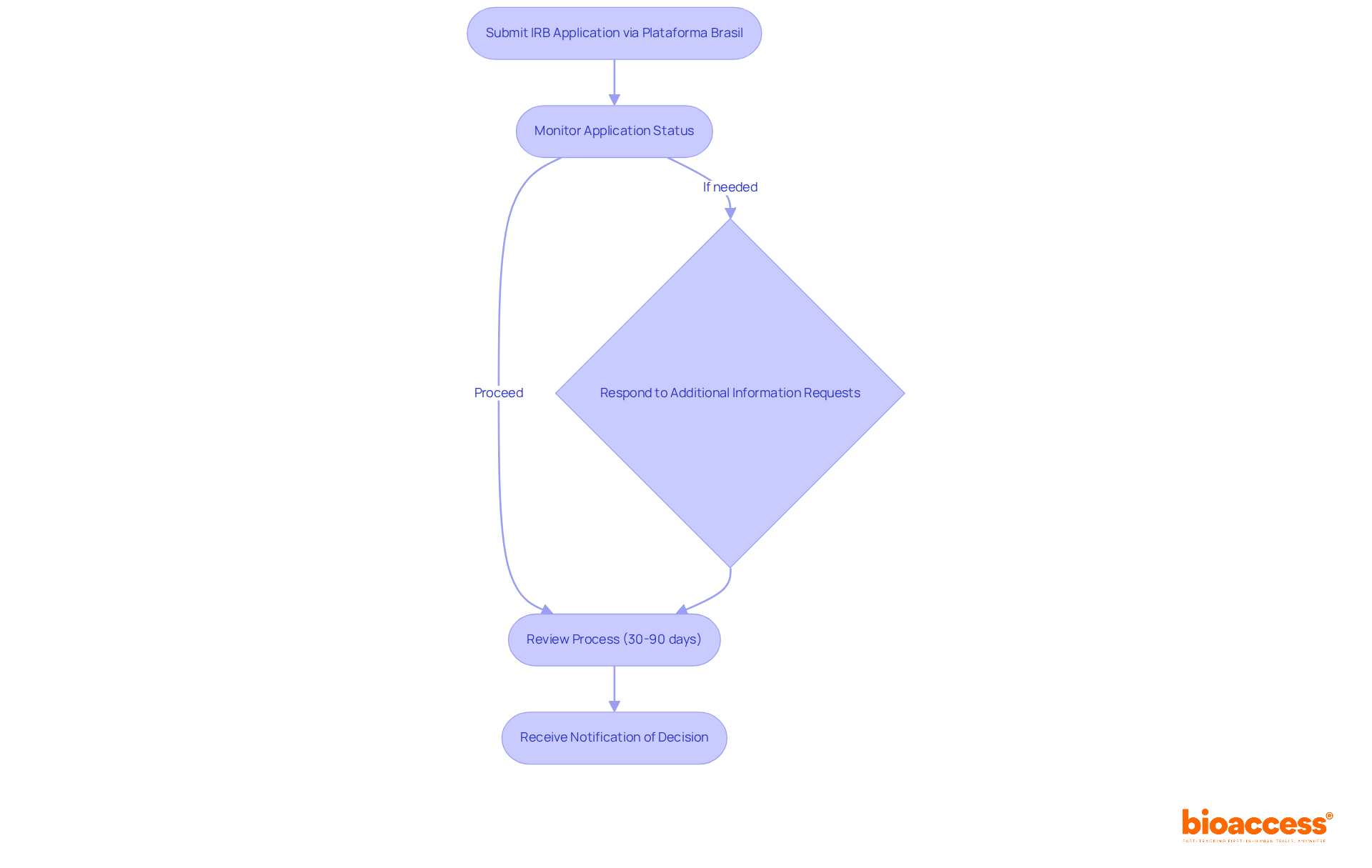
Navigating the IRB approval Brazil process is crucial for the success of your clinical research projects. Start by submitting your application for IRB approval Brazil through Plataforma Brasil, the designated online system for managing research ethics submissions in Brazil. Once you submit your application, how will you keep track of its progress? Actively monitor the status via Plataforma Brasil to stay informed.
When the IRB or CONEP requests additional information, your swift and thorough response is essential to avoid jeopardizing your project timeline. Prepare for a review process that can take anywhere from 30 to 90 days – timeliness is key to your project’s success. Once the review is complete, you’ll receive a notification about the decision or any conditions that need addressing – act promptly to keep your project on track.
By tackling the IRB application undertaking with thoroughness and care for detail, you can greatly improve your likelihood of obtaining timely IRB approval Brazil for your clinical trials.
Troubleshoot Common Challenges in the IRB Approval Process
Navigating the IRB approval Brazil process presents unique challenges that can significantly impact clinical research timelines. Here are some common issues and how to address them:
-
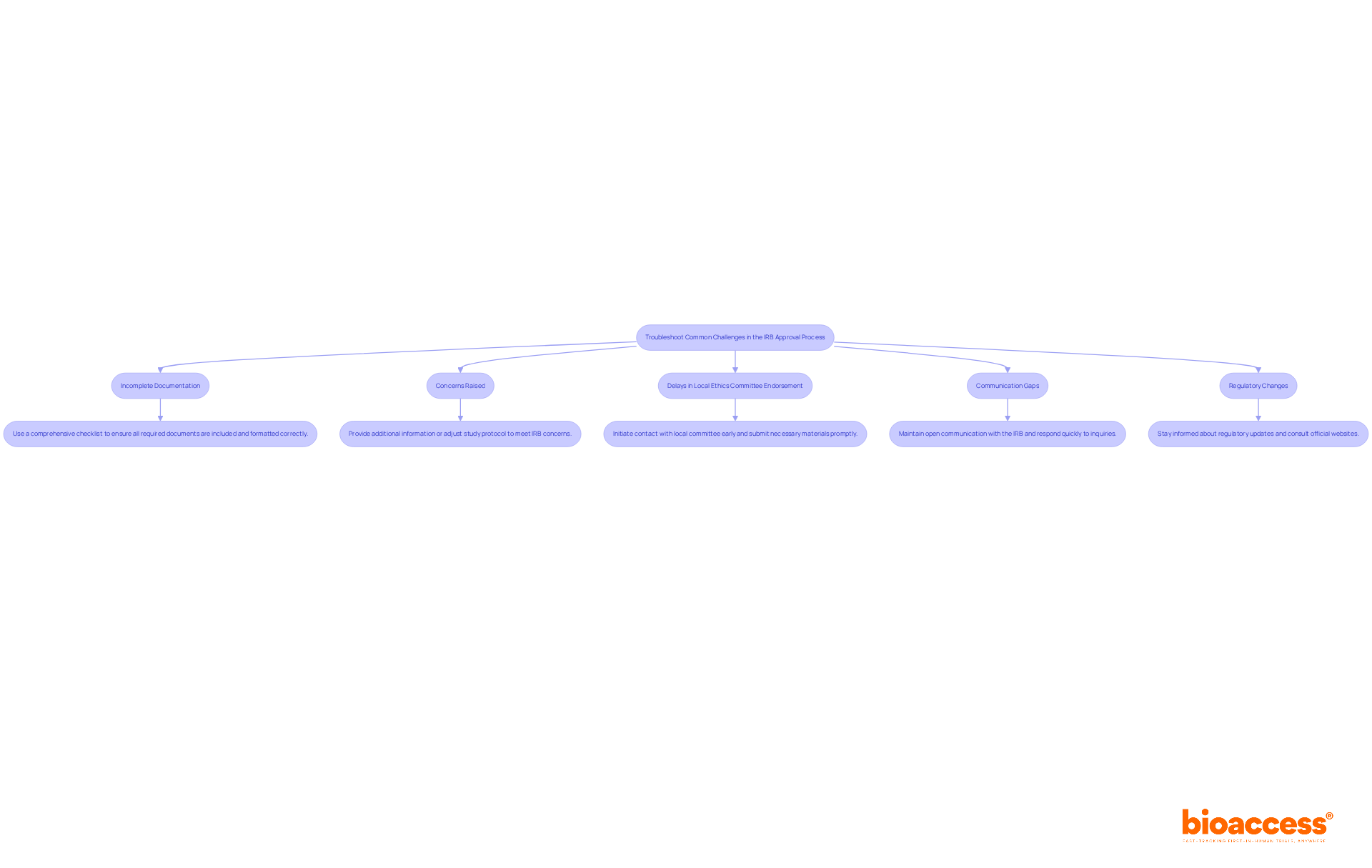
Incomplete Documentation: One of the most frequent reasons for delays is incomplete submissions. Recent findings indicate that a significant percentage of IRB approvals are delayed due to missing or inadequate documentation.
- Solution: Utilize a comprehensive checklist to ensure all required documents, including consent forms and recruitment materials, are included and formatted correctly before submission. Conduct a thorough review to confirm completeness.
-
Concerns Raised: The IRB may highlight issues regarding participant safety or informed consent, which are critical in the context of first-in-human trials.
- Solution: Consider providing additional information or adjusting your study protocol to meet these concerns head-on. Engage with the IRB early to clarify their expectations and ensure that your protocol aligns with ethical standards.
-
Delays in Local Ethics Committee Endorsement: Delays in obtaining local ethics committee consent can hinder progress, especially in regions with limited trial availability.
- Solution: Initiate contact with the local committee early on and ensure that all necessary materials are submitted promptly to avoid unnecessary delays.
-
Communication Gaps: Miscommunication with the IRB can result in misunderstandings and delays in the authorization timeline.
- Solution: Maintain open lines of communication with the IRB and respond to any inquiries as quickly as possible. Regular updates can help clarify any potential issues before they escalate.
-
Regulatory Changes: Modifications in rules or guidelines from authorities such as ANVISA or CONEP can influence the endorsement timeline.
- Solution: Stay informed about any regulatory updates that may affect your submission. Regularly consult the official websites of these regulatory bodies to ensure compliance with the latest requirements.
Addressing these challenges not only streamlines the IRB approval Brazil process but also accelerates the introduction of groundbreaking therapies to the Latin American market.
Conclusion
The IRB approval process in Brazil is a critical hurdle that can determine the success of clinical trials for MedTech, Biopharma, and Radiopharmaceutical companies. Grasping the intricacies of this process ensures compliance with regulatory standards. It also speeds up the timeline for bringing innovative therapies to market. By following the outlined steps – from pre-submission preparation to tackling common challenges – researchers can greatly improve their chances of securing timely approvals.
The article highlights several key components necessary for securing IRB approval in Brazil. Researchers must prepare comprehensive documentation, including:
- Study protocols
- Informed consent forms
- Endorsements from local ethics committees
Make it a priority to keep an eye on the review process and respond quickly to any requests for more information. Furthermore, many researchers face hurdles like incomplete documentation and communication gaps that can stall their progress, but addressing these challenges can prevent delays and streamline the approval process.
Ultimately, this favorable environment allows researchers to capitalize on opportunities that can lead to groundbreaking advancements. By leveraging the favorable regulatory environment and the strategic advantages of conducting early-stage trials in Latin America, companies can not only foster innovation but also improve patient outcomes. Engaging with specialized services like bioaccess®’s Global Trial Accelerators™ can facilitate this journey, ensuring that groundbreaking therapies reach those in need more efficiently. By embracing these strategies, researchers can not only navigate the complexities of IRB approval but also position themselves at the forefront of innovation in clinical research.
Frequently Asked Questions
What is the role of CONEP in the IRB approval process in Brazil?
The National Commission for Ethics in Research (CONEP) is the primary regulatory authority in Brazil responsible for assessing the moral dimensions of research proposals involving human subjects.
What guidelines must researchers follow before submitting an application to CONEP?
Researchers must adhere to the guidelines established by the Brazilian Health Surveillance Agency (ANVISA), which include understanding essential documentation such as the study protocol and informed consent forms.
What is the first step in the IRB approval process in Brazil?
The first step is Pre-Submission Preparation, where researchers familiarize themselves with the moral guidelines and requirements established by CONEP and ANVISA.
Is prior consent from a local ethics committee required before submitting to CONEP?
Yes, prior consent from a local ethics committee (CEP) is mandatory before submitting an application to CONEP.
What is the review timeline for local ethics committee opinions?
The review period for local ethics committee opinions is capped at 30 days, which has significantly reduced previous timelines.
What happens after obtaining local approval in the IRB approval process?
After obtaining local approval, researchers must submit their application to CONEP, including all relevant documentation such as the study protocol and informed consent forms.
How long does CONEP’s evaluation typically take?
CONEP’s evaluation can take between 30 to 90 days, depending on the complexity of the study.
What recent changes have been made to streamline the IRB approval process in Brazil?
Recent changes, particularly the Executive Order #12,651/2025, have streamlined the approval process, enhancing Brazil’s competitiveness in global clinical research.
Why is mastering the IRB approval process important for researchers?
Mastering the process ensures compliance and speeds up the clinical trial timeline, providing a crucial advantage in the competitive landscape of clinical research.
How can researchers leverage services like bioaccess®’s Global Trial Accelerators™?
Researchers can use these services to navigate Brazil’s favorable regulatory environment more effectively, achieving faster approvals and access to a diverse patient population.
List of Sources
- Understand the IRB Approval Process in Brazil
- New regulations for clinical research in Brazil | Licks Attorneys (https://lickslegal.com/post/new-regulations-for-clinical-research-in-brazil)
- Brazil Cuts Clinical Trial Approval Timeline to 90 Days | bioaccess®: Fast-Tracking First-in-Human Trials, Anywhere posted on the topic | LinkedIn (https://linkedin.com/posts/bioaccess_clinicaltrials-brazil-anvisa-activity-7447976698958106627-98MW)
- Gather Required Documentation for IRB Submission
- New rules on research with human subjects in Brazil – Cescon Barrieu – Centro de Inteligência Jurídica (https://cesconbarrieu.com.br/en/new-rules-on-research-with-human-subjects-in-brazil)
- Clinical Trial Regulatory Process – Brazil | Credevo Articles (https://credevo.com/articles/2019/03/14/clinical-trial-regulatory-process-brazil)
- Submit Your IRB Application and Navigate the Review Process
- Clinical Trial Requirements In Latin America A Complete Pre Clinical Document Checklist For Sponsors | bioaccess® (https://bioaccessla.com/blog/clinical-trial-requirements-in-latin-america-a-complete-pre-clinical-document-checklist-for-sponsors)
- Instructions for submitting Research Protocols – CEP Pecege (https://cep.pecege.com/en/instructions-for-submitting)
- Troubleshoot Common Challenges in the IRB Approval Process
- Top 10 Common Problems to Avoid with IRB Protocol Submissions | Ohio University (https://ohio.edu/research/compliance/human-subjects/top10)
- IRB: Common Application Problems | Office of Research (https://sjsu.edu/research/research-compliance/irb/irb-help/common-problems.php)
- Current scenario and future perspectives of clinical research in Brazil: a national survey – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10898894)
- Top Reasons for IRB Submission Delays | Office of the Vice President for Research | University of Connecticut (https://ovpr.uchc.edu/top-reasons-for-irb-submission-delays)

Leave a Reply