Introduction
Researchers in the MedTech and Biopharma sectors face significant hurdles when navigating clinical trials in Chile. Understanding the specific FDA regulations is crucial. Local compliance requirements also play a key role in conducting successful studies that meet international standards. As researchers dive into FDA accepted clinical trials, they might ask themselves: how can they streamline the process while sticking to local and global regulations? This article provides a step-by-step tutorial to master the essentials of conducting FDA accepted clinical trials in Chile, equipping researchers with the knowledge to enhance their study execution and maximize their chances of success.
Explore FDA Regulations for Clinical Trials in Chile
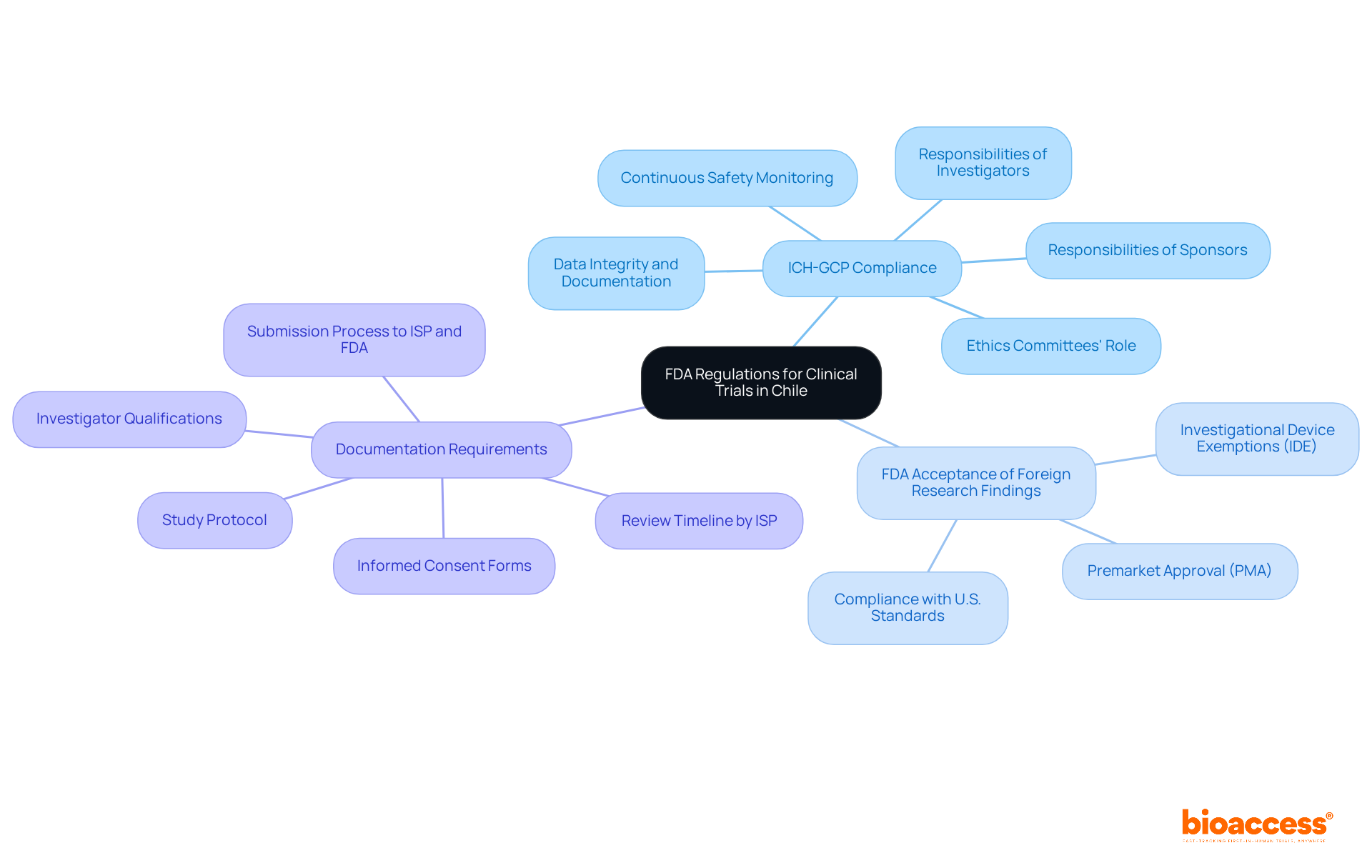
Navigating the regulatory landscape in Chile can be daunting for researchers, with numerous guidelines to follow. To successfully conduct an FDA accepted clinical trial in Chile, it is crucial to understand the specific regulations governing these studies. The FDA mandates compliance with Good Clinical Practice (GCP) guidelines, which protect the integrity of information and ensure participant safety. The Instituto de Salud Pública (ISP) plays a pivotal role in overseeing the regulatory framework in Chile, ensuring that researchers adhere to both local and FDA standards for information acceptance. Key regulations include:
- ICH-GCP Compliance: All trials must adhere to the International Council for Harmonisation (ICH) GCP guidelines, which delineate the responsibilities of sponsors, investigators, and ethics committees. Compliance with ICH GCP is essential, as it enhances the credibility of the research, opens doors to global research opportunities, and emphasizes the importance of continuous safety monitoring to identify and address adverse events promptly.
- FDA Acceptance of Foreign Research Findings: Under 21 CFR 812.28, the FDA acknowledges findings from foreign studies if they conform to U.S. standards. Consequently, studies conducted in Chile must meet the standards of an FDA accepted clinical trial in Chile to ensure that the data can be utilized for submissions such as Investigational Device Exemptions (IDE) or Premarket Approval (PMA).
- Documentation Requirements: Essential documents include the study protocol, informed consent forms, and investigator qualifications. These documents must be meticulously prepared and submitted to both the ISP and the FDA, ensuring that all aspects of the study are compliant with regulatory expectations. The ISP generally reviews applications within 30 business days, and it has reduced bureaucratic delays in research approval by over 30%, which enhances the efficiency of the regulatory process.
By mastering these regulations, researchers can ensure their studies not only comply but also stand out in the competitive field of clinical research.
Understand First-in-Human Trial Requirements in Chile
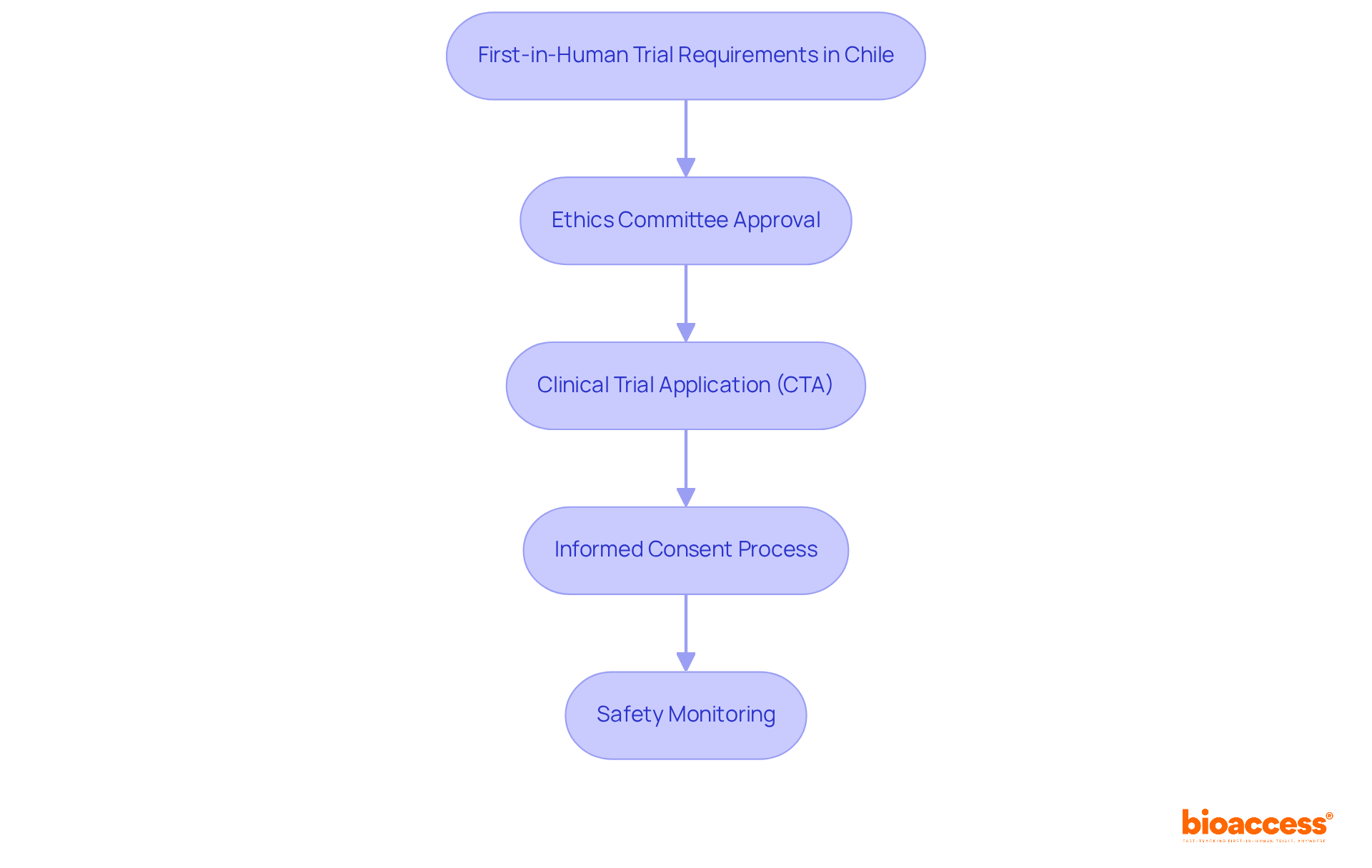
In Chile, the stringent requirements governing first-in-human studies present both challenges and opportunities for clinical research. Recognized as a frontrunner in the clinical research landscape, particularly in the MedTech, Biopharma, and Radiopharma sectors, Chile offers a pivotal phase for companies as they mark the initial testing of new therapies in human subjects. Key requirements include:
- Ethics Committee Approval: Prior to commencing a trial, obtaining approval from an accredited ethics committee is essential. This committee meticulously reviews the study protocol to confirm adherence to ethical standards and the protection of participant rights. Navigating the ethics committee approval can be a lengthy and complex process for many companies. With bioaccess®, companies can gain from expedited ethics approvals, usually within 4-8 weeks, facilitating the commencement of studies.
- Clinical Trial Application (CTA): A detailed CTA must be submitted to the Public Health Institute of Chile (ISP), encompassing comprehensive information about the study design, objectives, and methodologies. The average assessment duration for CTAs in Chile is around 30 business days, enabling timely commencement of studies. By leveraging bioaccess®’s expertise, companies can significantly streamline their approval processes, leading to faster study initiation.
- Informed Consent Process: Participants must receive comprehensive information regarding the study’s purpose, procedures, risks, and benefits. The informed consent form must be clear and concise. It should also be translated into Spanish to ensure all participants fully understand it.
- Safety Monitoring: It’s crucial to conduct continuous safety assessments throughout the study. This includes vigilant monitoring of adverse events and the management of any associated risks to ensure participant safety.
Chile’s strengths in key therapeutic areas such as oncology, autoimmune disorders, and vaccine development create significant opportunities for FIH research. Furthermore, the urbanized population supports fast recruitment and high protocol adherence, which are crucial for the success of these studies. By following these requirements and utilizing Chile’s effective regulatory structure, along with bioaccess®’s expertise in conducting early feasibility studies, researchers can optimize study execution and improve the credibility of their findings. Embracing these requirements with the support of bioaccess® can transform the landscape of clinical research in Chile, paving the way for groundbreaking therapies.
Implement Effective Strategies for Clinical Trial Execution
Navigating the complex landscape of clinical research in Chile can be daunting due to various operational and regulatory hurdles. Here are key strategies to consider:
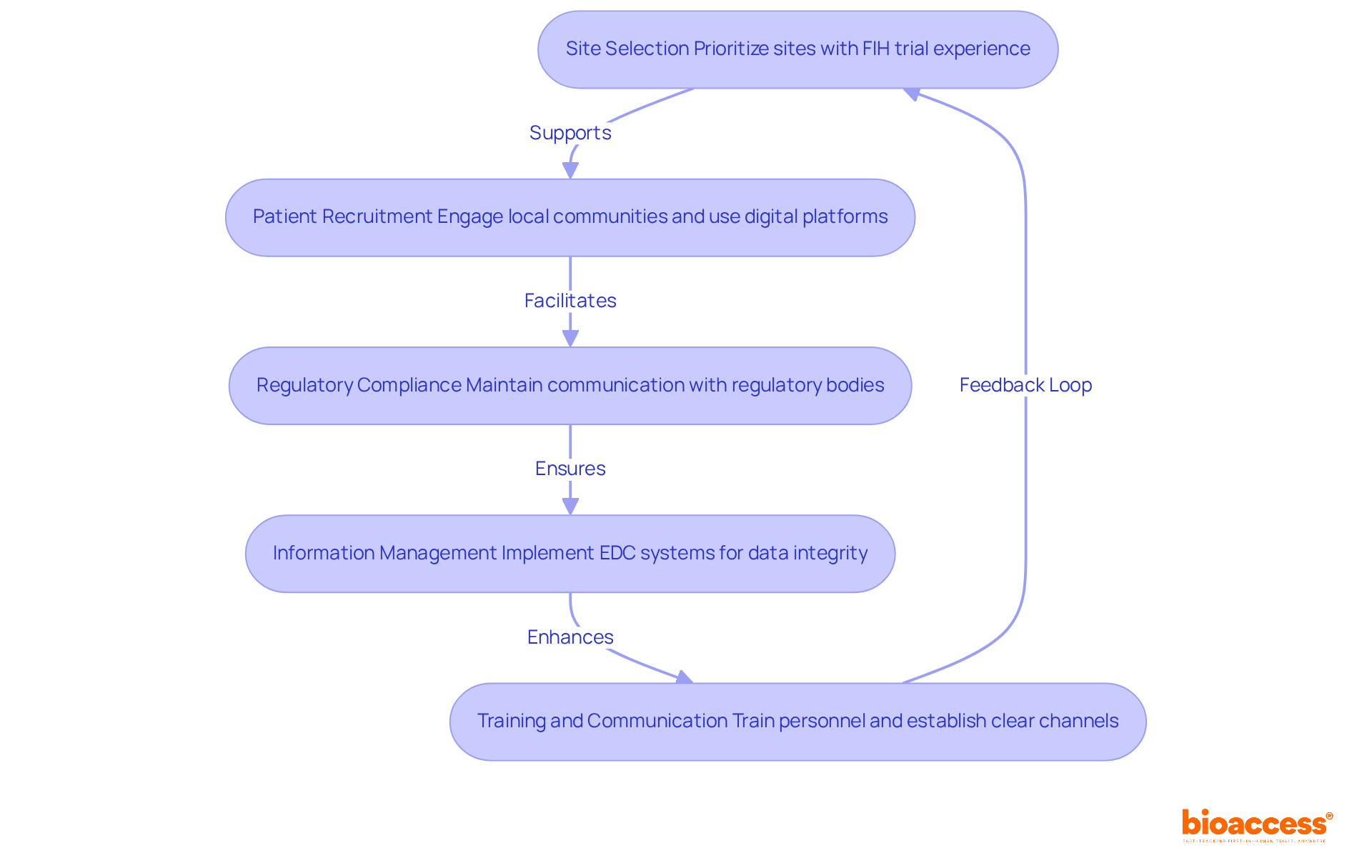
- Site Selection: Prioritize clinical sites with a proven history in conducting first-in-human (FIH) trials. Ensure that these sites are adequately staffed and equipped to meet the specific requirements of the study while adhering to ICH-GCP standards. Involving sites with prior experience in similar studies can greatly improve the chances of success.
- Patient Recruitment: Craft recruitment strategies that truly connect with local communities. Leverage community outreach initiatives and establish partnerships with local healthcare providers to bolster recruitment efforts. Additionally, utilizing digital platforms can effectively broaden the reach to potential participants, ensuring a diverse and representative sample.
- Regulatory Compliance: Maintain proactive communication with regulatory bodies such as the Instituto de Salud Pública (ISP) to guarantee that all submissions are timely and comprehensive. Regularly review compliance with local regulations and adapt protocols as necessary to align with evolving standards.
- Information Management: Implement robust information management systems to facilitate accurate and timely collection. Employing electronic data capture (EDC) systems can enhance data entry and monitoring processes, ensuring that data integrity is preserved throughout the study.
- Training and Communication: Provide thorough training for all personnel involved in the study to ensure they are well-versed in protocols and compliance requirements. Establish clear communication channels among sponsors, investigators, and regulatory bodies to foster collaboration and swiftly address any issues that may arise.
By implementing these strategies, researchers can enhance study efficiency, reduce costs, and boost the chances of success. This ultimately accelerates the path to regulatory approval and market entry.
Navigate Regulatory Submissions for FDA Acceptance
Navigating the regulatory submission process for an FDA accepted clinical trial in Chile is fraught with challenges that demand precision and expertise. Here’s how to effectively manage this process:
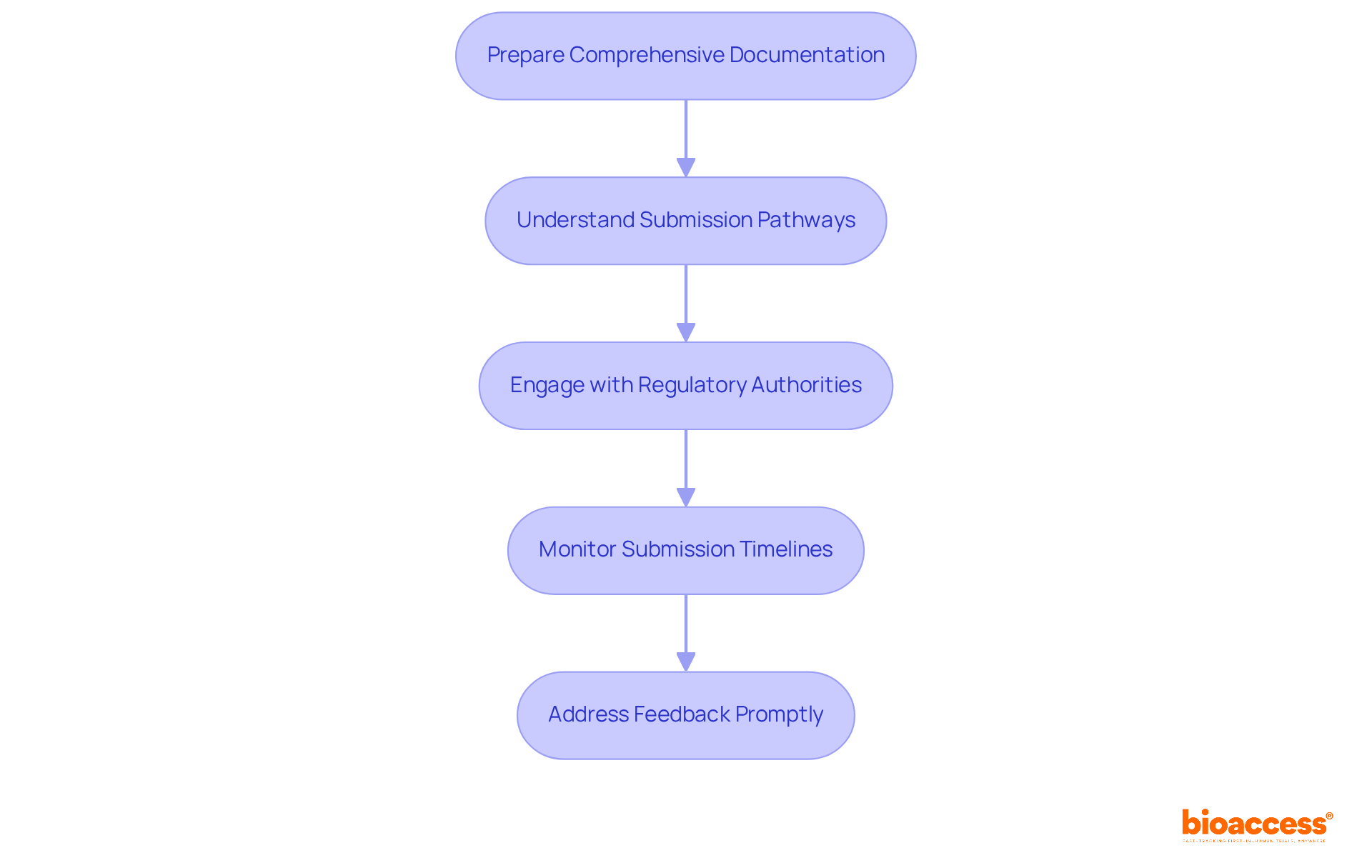
- Prepare Comprehensive Documentation: Ensure that all required documents are complete and accurate. This includes the study protocol, informed consent documents, investigator qualifications, and any additional information required for the FDA accepted clinical trial Chile, as well as from the ISP (Instituto de Salud Pública), INVIMA, ANVISA, and COFEPRIS. Adhering to eCTD standards can significantly enhance the organization of your submission, potentially reducing review times by up to 30%.
- Understand Submission Pathways: Familiarize yourself with the specific submission pathways for FDA acceptance. This may involve submitting a pre-investigational new drug application (pre-IND) or an Investigational Device Exemption (IDE) application, depending on the nature of the study. Engaging with the FDA early through pre-submission meetings clarifies expectations for the FDA accepted clinical trial in Chile. This step can significantly streamline the process.
- Engage with Regulatory Authorities: Maintain open lines of communication with both the ISP and the FDA accepted clinical trial Chile, alongside INVIMA, ANVISA, and COFEPRIS. Regular updates and proactive engagement can help address any potential concerns early in the process, ensuring a smoother approval pathway. With bioaccess®’s expertise, you can speed up ethics approvals, typically within 4-8 weeks, which is a substantial benefit in the competitive environment of early-stage studies.
- Monitor Submission Timelines: Keep track of submission timelines and follow up with regulatory bodies to ensure that your application is being processed. The typical approval duration for an FDA accepted clinical trial in Chile is about 30 days, which is considerably quicker than the six to nine months usually needed in other Latin American nations such as Brazil and Mexico. This velocity can be a strategic edge for early-stage studies.
- Address Feedback Promptly: Be prepared to respond to any queries or requests for additional information from regulatory authorities. Delays in the approval process can derail timelines and increase costs for researchers. Timely and thorough responses can significantly reduce these delays, enhancing the likelihood of a successful outcome.
By mastering these steps, researchers can not only expedite their trials but also elevate the standards of clinical research in their fields.
Conclusion
Successfully navigating the intricate landscape of FDA accepted clinical trials in Chile is not just a necessity; it’s a strategic imperative for researchers. By mastering essential regulations, including ICH-GCP compliance and documentation requirements, researchers can ensure their studies are compliant and positioned for success in a competitive field.
Key insights highlighted throughout the article emphasize the importance of thorough preparation, from obtaining ethics committee approval to implementing effective patient recruitment strategies. Engaging with regulatory authorities like the Instituto de Salud Pública (ISP) and leveraging the expertise of organizations such as bioaccess® can significantly streamline the approval process, ultimately leading to faster initiation of trials and more efficient pathways to market.
Adhering to these guidelines is essential for success in clinical research. As Chile emerges as a leading hub for clinical research in Latin America, the ability to navigate its regulatory environment effectively is crucial for researchers aiming to bring innovative therapies to market. Embracing these strategies not only enhances the credibility of clinical findings but also paves the way for groundbreaking advancements in MedTech and Biopharma sectors. Without a firm grasp of these regulations, researchers risk falling behind in a rapidly evolving MedTech and Biopharma landscape.
Frequently Asked Questions
What are the key regulations for conducting FDA accepted clinical trials in Chile?
Key regulations include adherence to Good Clinical Practice (GCP) guidelines as mandated by the FDA, compliance with International Council for Harmonisation (ICH) GCP standards, and the submission of essential documents such as study protocols and informed consent forms to both the Instituto de Salud Pública (ISP) and the FDA.
What is the role of the Instituto de Salud Pública (ISP) in clinical trials in Chile?
The ISP oversees the regulatory framework for clinical trials in Chile, ensuring that researchers comply with both local and FDA standards for information acceptance. It reviews applications typically within 30 business days and has improved the efficiency of the regulatory process by reducing bureaucratic delays.
What is ICH-GCP and why is it important for clinical trials in Chile?
ICH-GCP stands for International Council for Harmonisation Good Clinical Practice. It outlines the responsibilities of sponsors, investigators, and ethics committees. Compliance with ICH-GCP is crucial as it enhances research credibility, facilitates global research opportunities, and ensures continuous safety monitoring of participants.
How does the FDA accept findings from foreign studies, including those conducted in Chile?
Under 21 CFR 812.28, the FDA accepts findings from foreign studies if they conform to U.S. standards. Therefore, clinical trials conducted in Chile must meet the standards of an FDA accepted clinical trial to ensure that the data can be utilized for submissions like Investigational Device Exemptions (IDE) or Premarket Approval (PMA).
What documentation is required for conducting clinical trials in Chile?
Essential documents include the study protocol, informed consent forms, and investigator qualifications. These documents must be meticulously prepared and submitted to both the ISP and the FDA to ensure compliance with regulatory expectations.
How does the regulatory process for clinical trials in Chile benefit researchers?
The ISP’s efficient review process, typically completed within 30 business days, and its efforts to reduce bureaucratic delays by over 30% enhance the speed and efficiency of the regulatory process, making Chile an advantageous location for early-stage clinical trials.
Why is mastering FDA regulations important for researchers conducting trials in Chile?
Mastering these regulations ensures that studies comply with necessary standards, enhances the credibility of the research, and increases opportunities for global collaboration, ultimately allowing researchers to stand out in the competitive field of clinical research.
List of Sources
- Explore FDA Regulations for Clinical Trials in Chile
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- ICH GCP Compliant Clinical Trials: Ensuring Ethical and High-Quality Research (https://icbiocro.com/ich-gcp-compliant-clinical-trials-ensuring-ethical-and-high-quality-research)
- Comparison of the Latin America Regulation Landscape and International Reference Health Authorities to Hasten Drug Registration and Clinical Research Applications – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10579156)
- Understand First-in-Human Trial Requirements in Chile
- Navigate Clinical Trial Approval in Chile: 4 Essential Steps | bioaccess® (https://bioaccessla.com/blog/navigate-clinical-trial-approval-in-chile-4-essential-steps)
- Chile First-in-Human Clinical Trial Market Size, Share, Growth & Trends 2025–2033 (https://reedintelligence.com/market-analysis/chile-first-in-human-clinical-trial-market)
- Clinical trials by start date Chile 1999-2024| Statista (https://statista.com/statistics/1560150/chile-number-new-clinical-trials?srsltid=AfmBOorJIYqj6U6uuWMWEg6MxOshlXp1buJ3ojMu-vsM7IyRW4MIxinw)
- Master Phase 1 Clinical Trials in Chile: A Step-by-Step Guide | bioaccess® (https://bioaccessla.com/blog/master-phase-1-clinical-trials-in-chile-a-step-by-step-guide)
- Implement Effective Strategies for Clinical Trial Execution
- Clinical trials: Investment in Chile grew by 62% in three years (https://blog.investchile.gob.cl/clinical-trials-investment-in-chile)
- The Most Impactful Quotes From January | PharmExec (https://pharmexec.com/view/most-impactful-quotes-january)
- Chile Clinical Trial Supply & Logistics Market Size & Outlook, 2030 (https://grandviewresearch.com/horizon/outlook/clinical-trial-supply-logistics-market/chile)
- Clinical Trials Quotes (2 quotes) (https://goodreads.com/quotes/tag/clinical-trials)
- Navigate Regulatory Submissions for FDA Acceptance
- How Chile Is Shaping Medical Device Clinical Trials In Latin America (https://meddeviceonline.com/doc/how-chile-is-shaping-medical-device-clinical-trials-in-latin-america-0001)
- Drug Regulatory Agencies Guide 2026: FDA EMA WHO Approval (https://pharmuni.com/2026/04/03/drug-regulatory-agencies-complete-guide-year-regulatory-approval-compliance)
- Best Practices for Navigating FDA Submissions & approvals (https://biobostonconsulting.com/best-practices-for-navigating-fda-submissions-and-approvals)
- Navigate Clinical Trial Approval in Chile: 4 Essential Steps | bioaccess® (https://bioaccessla.com/blog/navigate-clinical-trial-approval-in-chile-4-essential-steps)

Leave a Reply