Introduction
While Chile offers a promising landscape for first-in-human trials, the path to success is fraught with challenges that require careful navigation for sponsors. With a regulatory landscape governed by the Instituto de Salud Pública (ISP) and a fast-track approval process, Chile stands out as a strategic location for early-stage clinical research. However, navigating these complexities can be daunting for sponsors, leading to uncertainty in their trial strategies.
What are the essential steps to ensure a successful trial while maximizing the advantages offered by this vibrant market? Understanding these complexities is crucial for sponsors aiming to capitalize on Chile’s unique advantages in clinical research.
Understand Regulatory Requirements for First-in-Human Trials in Chile
To understand how to conduct first-in-human trial Chile successfully, a thorough understanding of the compliance framework is non-negotiable. The Instituto de Salud Pública (ISP) serves as the primary regulatory body overseeing clinical studies. Let’s explore the essential steps to ensure compliance:
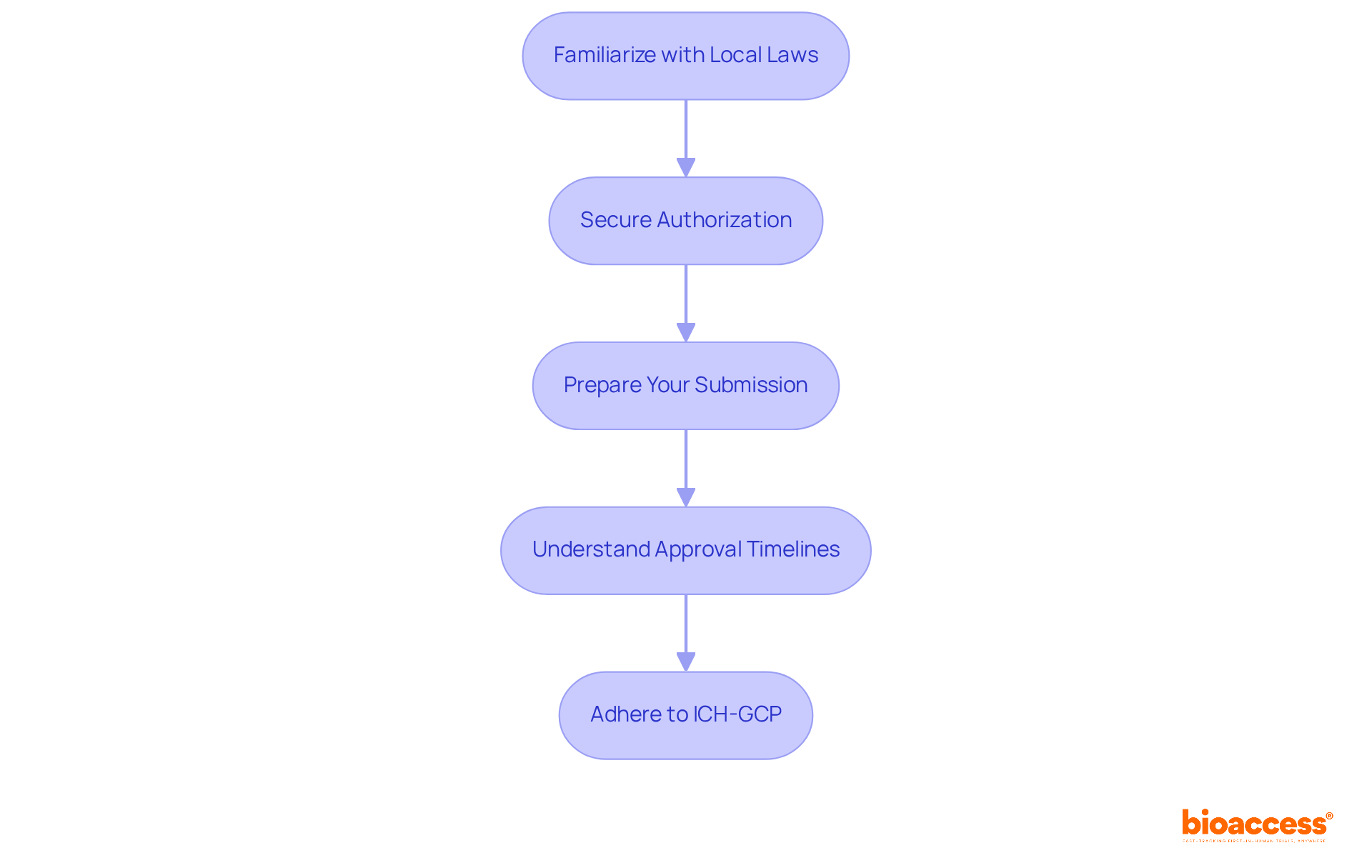
- Familiarize Yourself with Local Laws: Review Technical Norm Nº 57, which outlines the regulations for conducting clinical studies in Chile, detailing requirements for ethical approvals, informed consent, and safety monitoring.
- Secure Authorization: Obtain approval from an accredited Scientific Ethics Committee (CEC) before commencing a study. This process typically takes 30 to 60 days. Ensure that all documentation is in Spanish and adheres to local ethical standards.
- Prepare Your Submission: Compile all necessary documents, including the clinical study protocol, informed consent forms, and investigator qualifications. Thorough submissions are critical to avoid delays.
- Understand Approval Timelines: The average duration for authorization in Chile is roughly 30 days, significantly faster than in many other areas. This accelerated process provides a strategic benefit for understanding how to conduct first-in-human trial Chile.
- Adherence to ICH-GCP: Ensure that all research activities align with International Council for Harmonisation Good Clinical Practice (ICH-GCP) guidelines, crucial for preserving study integrity and ensuring acceptance by international oversight bodies like the FDA.
Navigating the complex regulatory landscape can be daunting for sponsors. However, by mastering these regulations, sponsors can expedite their research timelines and enhance study integrity. Mastering these compliance steps not only facilitates smoother study initiation but also positions sponsors for success in the competitive clinical research landscape.
Gather Essential Preclinical Data and Conduct Safety Assessments
Before learning how to conduct first-in-human trial Chile, it is crucial to understand that the stakes are high and thorough preparation is non-negotiable. Here’s a structured approach to ensure readiness:
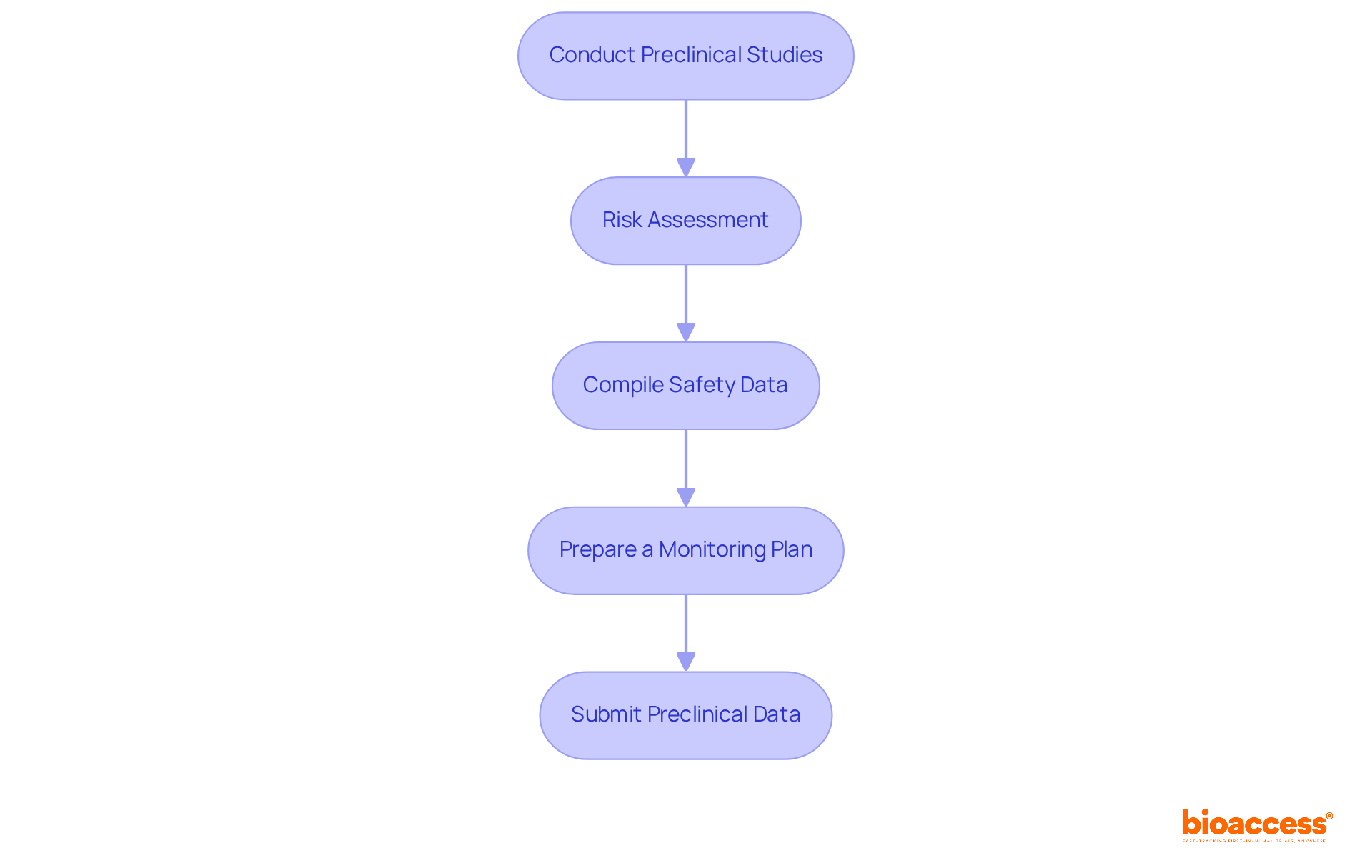
- Conduct Preclinical Studies: Execute both in vitro and in vivo studies to evaluate the safety and efficacy of the investigational product. This encompasses pharmacokinetic (PK) and pharmacodynamic (PD) evaluations to comprehend the drug’s behavior in biological systems, which is essential for establishing a robust foundation for human studies.
- Risk Assessment: Identify potential risks related to the investigational product by analyzing historical information from similar compounds and conducting toxicity studies. This process helps determine safe dosing levels and informs the overall risk profile of the product.
- Compile Safety Data: Document all findings from preclinical studies, including any adverse effects and observed toxicities. This thorough data regarding risk management is essential for regulatory submissions to the Instituto de Salud Pública (ISP) and for informing the study design.
- Prepare a Monitoring Plan: Develop a comprehensive monitoring plan that outlines how adverse events will be tracked and managed during the study. This plan must comply with ICH-GCP guidelines and receive approval from the ethics committee, ensuring that participant safety is prioritized throughout the trial.
- Submit Preclinical Data: Include all preclinical findings in your submission to the ISP. This information is crucial for demonstrating that the investigational product is reasonably safe for initial testing in humans, facilitating a smoother approval process.
By prioritizing these steps, sponsors not only enhance participant safety but also learn how to conduct first-in-human trial Chile for a more efficient regulatory journey. Leveraging the fast-track regulatory pathways available in Chile can further expedite the process, allowing for quicker access to critical clinical data.
Design and Execute the First-in-Human Trial Effectively
Understanding how to conduct first-in-human trial Chile involves navigating the complexities of a FIH study, which requires not just planning but also a strategic approach to leverage the unique advantages of Latin America. Here are the essential steps:
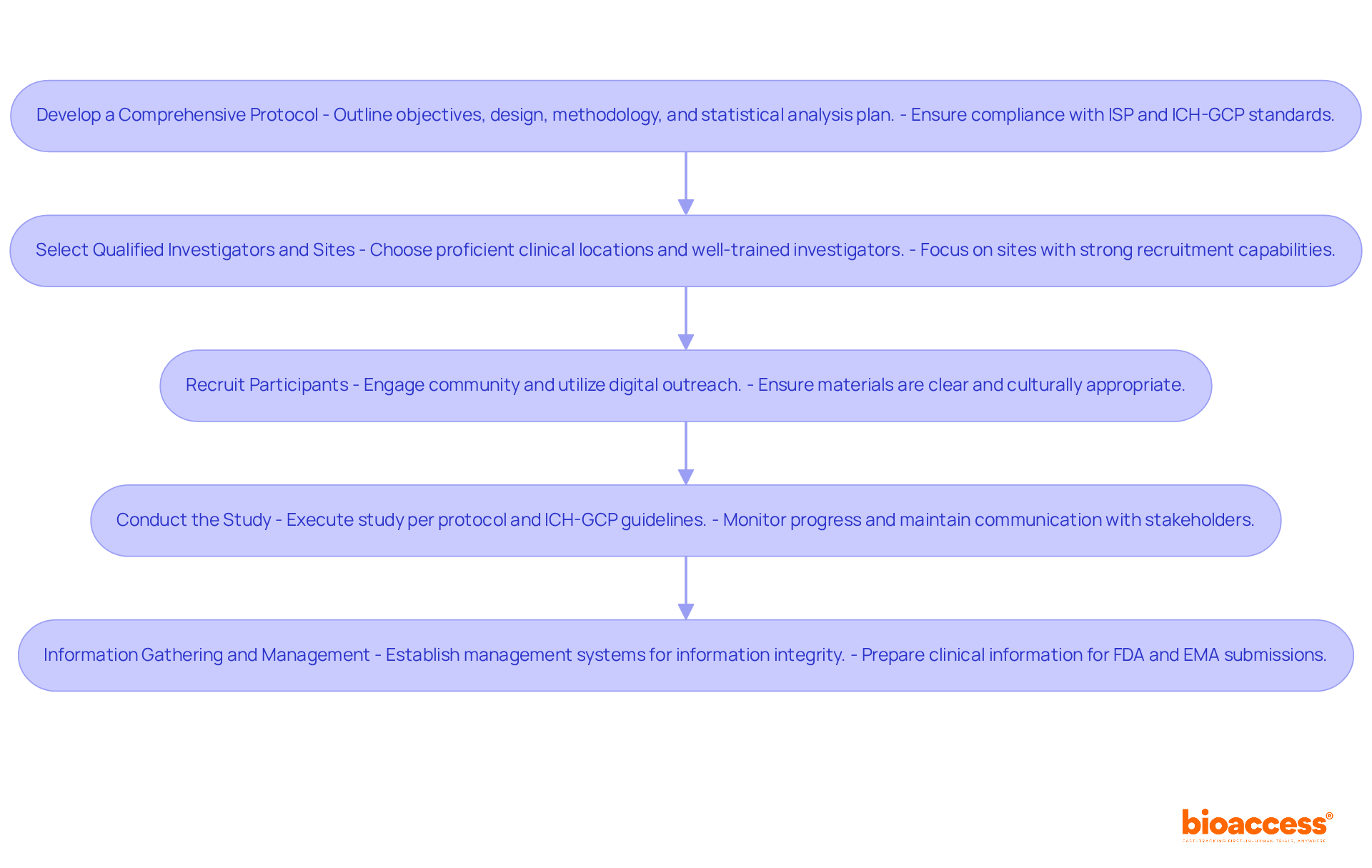
- Develop a Comprehensive Protocol: Craft a detailed clinical study protocol that clearly outlines the objectives, design, methodology, and statistical analysis plan. Ensure alignment with compliance requirements set by authorities such as the Chilean Public Health Institute (ISP) and adherence to ICH-GCP standards.
- Select Qualified Investigators and Sites: Choose clinical locations proficient in FIH studies. Investigators should be well-trained and familiar with both the protocol and regulatory requirements. This selection is crucial, as 11% of research sites fail to enroll a single patient, highlighting the importance of choosing sites with proven recruitment capabilities. With bioaccess®, you can identify sites that have strong recruitment records, ensuring a higher likelihood of participant enrollment.
- Recruit Participants: Consider using patient recruitment strategies that engage the community and utilize digital outreach effectively. Since 73% of patients prefer to learn about clinical studies from their healthcare providers, ensure that recruitment materials are clear, culturally appropriate, and accessible. Utilizing technology to reduce site visits can also enhance participation, particularly for patients with mobility challenges. With bioaccess®, you can benefit from tailored recruitment strategies that align with local practices and preferences.
- Conduct the Study: Execute the study in strict accordance with the approved protocol, ensuring compliance with ICH-GCP guidelines. Regular monitoring of study progress and maintaining open communication with all stakeholders is essential for addressing any issues that arise during the research. The expedited ethics approvals in Chile, typically taking 4 to 8 weeks, enable faster commencement of studies compared to the 6+ months often experienced in the US and EU. This speed not only accelerates your timeline but also contributes to significant cost savings, including $25K per patient with pre-negotiated site contracts and 30% lower overall trial costs.
- Information Gathering and Management: Establish robust management systems to ensure information integrity and adherence to compliance standards. Consistently assess information for precision and thoroughness, as efficient management is essential for fulfilling submission criteria. By utilizing bioaccess®’s expertise, you can ensure that your clinical information is not only compliant but also prepared for FDA and EMA submissions, improving your chances of successful market access.
By embracing these strategies, sponsors can not only enhance their study outcomes but also position themselves for success in a competitive landscape.
Implement Monitoring and Risk Mitigation Strategies During Trials
In first-in-human (FIH) investigations, ensuring participant safety and study integrity is paramount, yet it often faces significant challenges. To address these, applying effective monitoring and risk mitigation strategies is vital. Here’s a structured approach:
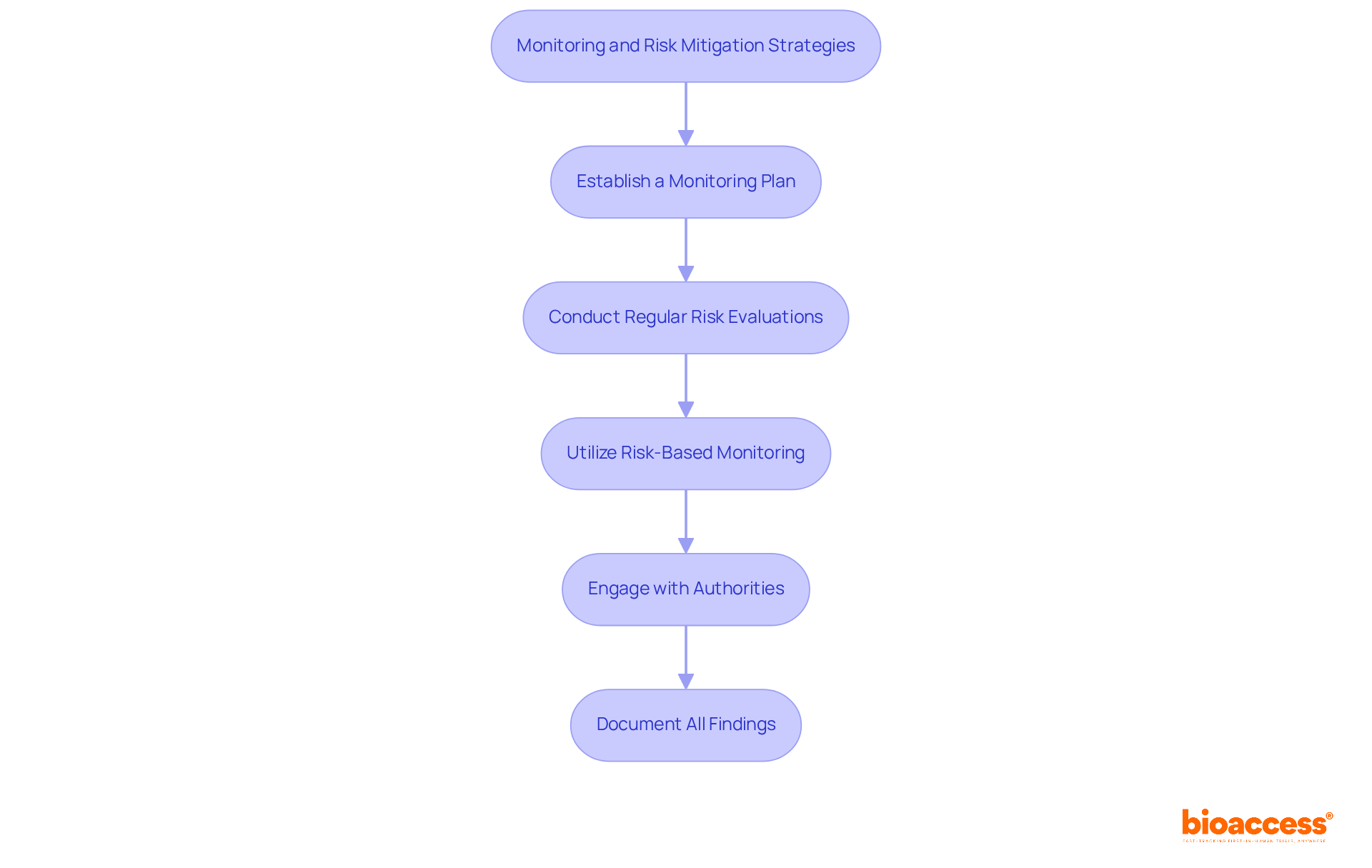
- Establish a Monitoring Plan: Create a comprehensive monitoring strategy that outlines how study activities will be supervised. This includes data collection techniques, reporting protocols for risks, and compliance with the study protocol. The FDA strongly advises setting up a Data Monitoring Committee (DMC) for studies involving serious risks to ensure participant well-being.
- Conduct Regular Risk Evaluations: Perform ongoing assessments throughout the study. This includes monitoring adverse events. Additionally, conducting interim analyses is essential to assess the safety and efficacy of the investigational product, in line with ICH-GCP guidelines. Ensure that findings are documented comprehensively to meet regulatory submission requirements and uphold study integrity.
- Utilize Risk-Based Monitoring: Implement a risk-based monitoring strategy to concentrate resources on high-risk areas of the trial. This approach optimizes monitoring efficiency and enhances participant safety, allowing for timely interventions when necessary. Continuous monitoring of risks throughout the clinical development process is essential, with timely access to accurate data for informed decision-making.
- Engage with Authorities: Maintain proactive communication with the Instituto de Salud Pública (ISP) and other relevant oversight bodies throughout the trial. This engagement is essential for quickly addressing emerging issues and ensuring adherence to local legal requirements, which can accelerate approval timelines.
- Document All Findings: Keeping detailed records is vital for compliance submissions and for upholding the integrity of the study. Furthermore, be mindful of the possible dangers linked to inadequate staffing at research locations, which can jeopardize participant safety and study integrity.
Have you considered how adopting these monitoring and risk mitigation strategies could enhance participant safety in your trials? By implementing these strategies, sponsors not only protect participants but also pave the way for smoother regulatory processes and successful trials.
Conclusion
The complexities of first-in-human (FIH) trials in Chile can be daunting, yet they offer a unique opportunity for sponsors willing to navigate them. Understanding the critical steps – from regulatory compliance to effective trial execution – is vital for ensuring participant safety and study integrity. Navigating these critical steps can be daunting for sponsors. From regulatory compliance to effective trial execution, each step is vital for ensuring participant safety and study integrity. By mastering these processes, sponsors can position themselves advantageously in the competitive landscape of clinical research.
The article highlights several key components necessary for successful FIH trials in Chile. It emphasizes the importance of:
- Familiarizing oneself with local regulations
- Securing necessary ethical approvals
- Conducting thorough preclinical studies to establish a solid foundation for human testing
- Strategically selecting qualified investigators and sites
- Implementing effective participant recruitment strategies
These components play a vital role in enhancing study outcomes. What advantages do FIH trials in Chile offer? Expedited approval processes and cost efficiencies are just the beginning. It’s crucial to implement robust monitoring and risk mitigation strategies throughout the trial to keep participants safe and ensure regulatory compliance.
In conclusion, the insights provided underscore the significant advantages of conducting FIH trials in Chile. By embracing these best practices and focusing on regulatory adherence, sponsors can not only enhance their chances of successful trials but also contribute to advancing medical innovations in a region poised for growth. By leveraging local expertise and fast-track pathways, sponsors can transform challenges into opportunities for groundbreaking clinical research, ultimately benefiting both the sponsors and the communities they serve.
Frequently Asked Questions
What is the primary regulatory body overseeing clinical studies in Chile?
The primary regulatory body overseeing clinical studies in Chile is the Instituto de Salud Pública (ISP).
What is Technical Norm Nº 57?
Technical Norm Nº 57 outlines the regulations for conducting clinical studies in Chile, detailing requirements for ethical approvals, informed consent, and safety monitoring.
How do I secure authorization for a first-in-human trial in Chile?
To secure authorization, you must obtain approval from an accredited Scientific Ethics Committee (CEC) before commencing the study. This process typically takes 30 to 60 days.
What language must all documentation be in for submissions in Chile?
All documentation must be in Spanish and adhere to local ethical standards.
What documents are necessary for submission when conducting a clinical study in Chile?
Necessary documents include the clinical study protocol, informed consent forms, and investigator qualifications. Thorough submissions are critical to avoid delays.
What is the average duration for authorization of clinical studies in Chile?
The average duration for authorization in Chile is roughly 30 days, which is significantly faster than in many other regions.
What guidelines must research activities align with in Chile?
Research activities must align with International Council for Harmonisation Good Clinical Practice (ICH-GCP) guidelines to preserve study integrity and ensure acceptance by international oversight bodies like the FDA.
How can understanding the regulatory landscape benefit sponsors conducting first-in-human trials in Chile?
By mastering the regulatory requirements, sponsors can expedite their research timelines and enhance study integrity, which positions them for success in the competitive clinical research landscape.
List of Sources
- Understand Regulatory Requirements for First-in-Human Trials in Chile
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Master The Clinical Trial Approval Process In … | bioaccess® (https://bioaccessla.com/blog/master-the-clinical-trial-approval-process-in-chile)
- Clinical Trials in Latin America (https://languageconnections.com/clinical-trials-in-latin-america)
- Gather Essential Preclinical Data and Conduct Safety Assessments
- Statistical Considerations for Premarketing Risk Assessment (https://fda.gov/drugs/news-events-human-drugs/statistical-considerations-premarketing-risk-assessment-05162024)
- What is Preclinical Research and Why is it so Important? (https://als.net/news/what-is-preclinical-research)
- Assessing Safety in Clinical Trials | Applied Clinical Trials Online (https://appliedclinicaltrialsonline.com/view/assessing-safety-clinical-trials)
- Design and Execute the First-in-Human Trial Effectively
- 25+ useful clinical trial recruitment statistics for better results (https://antidote.me/blog/25-useful-clinical-trial-recruitment-statistics-for-better-results)
- FDA Issues Guidance on Modernizing Statistical Methods for Clinical Trials (https://fda.gov/news-events/press-announcements/fda-issues-guidance-modernizing-statistical-methods-clinical-trials)
- Enrollment in Clinical Trials: Statistics and Patient Recruitment Strategies | Power (https://withpower.com/guides/enrollment-in-clinical-trials-statistics-and-patient-recruitment-strategies)
- Recruiting Clinical Trial Participants: How to Balance Data and Trust | Applied Clinical Trials Online (https://appliedclinicaltrialsonline.com/view/recruiting-clinical-trial-participants-how-to-balance-data-and-trust)
- Design and Conduct Considerations for First‐in‐Human Trials – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC6342261)
- Implement Monitoring and Risk Mitigation Strategies During Trials
- Data and Safety Monitoring Board Best Practices in Clinical Trials (https://cureus.com/articles/432271-data-and-safety-monitoring-board-best-practices-in-clinical-trials)
- Reducing Risk Through Mitigation Strategies | Applied Clinical Trials Online (https://appliedclinicaltrialsonline.com/view/reducing-risk-through-mitigation-strategies)
- Strategies for Risk Mitigation to Prevent Deviations in Clinical Trials – Imperial Clinical Research Services Blog (https://imperialcrs.com/blog/business-insights/strategies-for-risk-mitigation-to-prevent-deviations-in-clinical-trials)
- risk-management-in-clinical-trials-pms-comprehensive-guide (https://ccrps.org/clinical-research-blog/risk-management-in-clinical-trials-pms-comprehensive-guide)

Leave a Reply