Introduction
While Chile offers a great chance for MedTech and Biopharma companies to innovate while keeping costs in check, the path to success is fraught with challenges that require strategic navigation.
With a regulatory framework that supports swift approvals and a diverse patient population, Chile stands out as a strategic location for first-in-human studies.
Yet, navigating local regulations can be daunting for sponsors, often leading to delays and increased costs, especially when it comes to engaging patients effectively.
What strategies can sponsors adopt to truly harness Chile’s advantages for successful and cost-effective clinical trials?
Understand Early Phase Clinical Trials in Chile
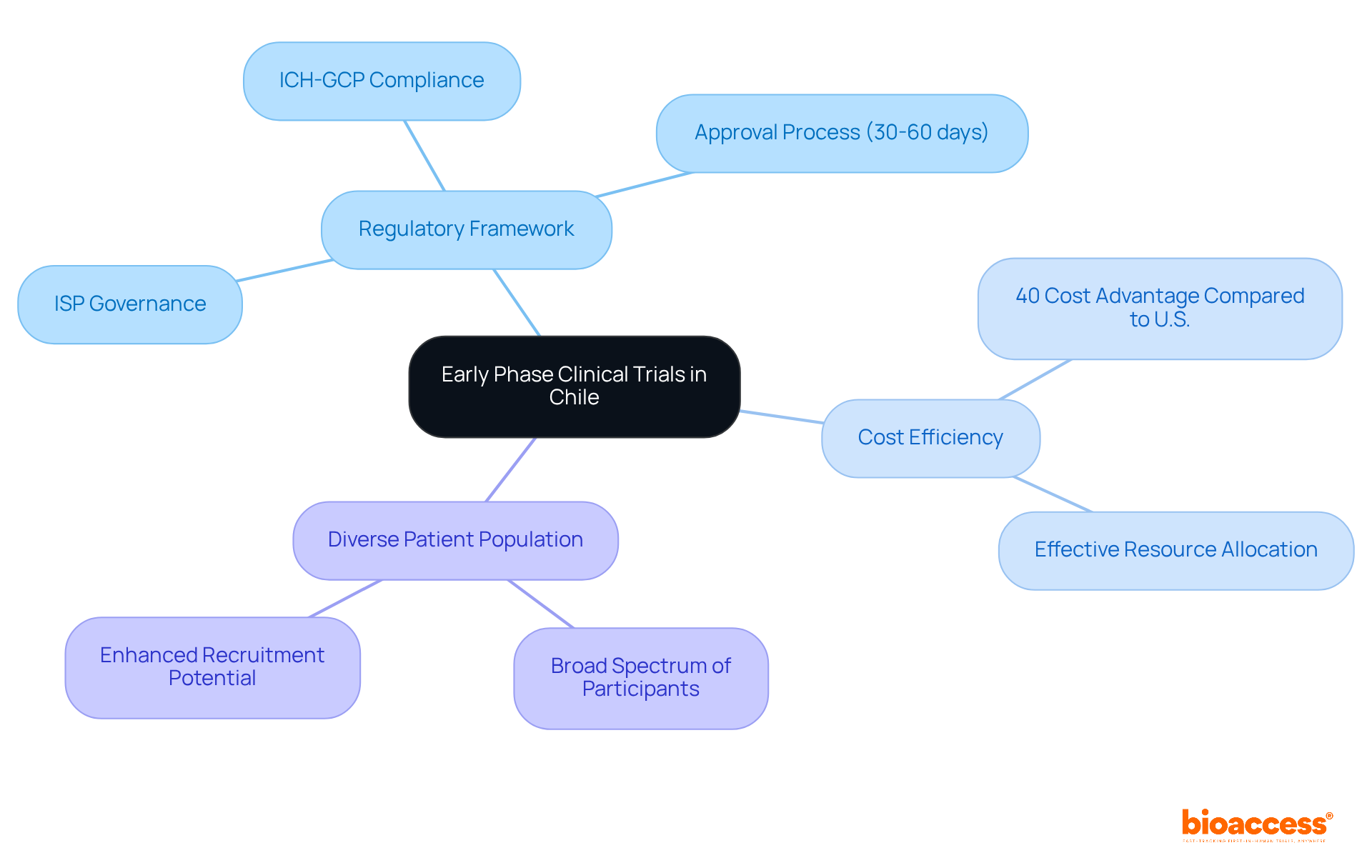
Initial phase clinical evaluations, particularly first-in-human (FIH) studies, are pivotal in the drug development process, shaping the future of innovative therapies. In this country, these studies are backed by a strong regulatory framework supervised by the Instituto de Salud Pública (ISP), which guarantees adherence to ICH-GCP standards. The approval process typically lasts between 30 to 60 days, with the ISP assessing applications within 30 business days. Conducting an affordable clinical trial in Chile offers a cost advantage of up to 40% compared to the U.S., positioning it as a strategic choice for MedTech and Biopharma startups.
Essential elements to consider are:
- Regulatory Framework: The ISP governs clinical trials, ensuring adherence to national and international guidelines. Familiarity with these regulations is essential for successful execution of the study.
- Cost Efficiency: The reduced operational expenses in the region enable sponsors to allocate resources more effectively, which is essential for startups functioning with constrained budgets. This cost advantage can significantly enhance the financial viability of affordable clinical trial Chile during early phase studies.
- Diverse Patient Population: The varied demographics of the country enhance recruitment potential, offering access to a broad spectrum of participants for research studies. This diversity accelerates enrollment and significantly enhances the representativeness of study results, a critical factor in clinical research.
By comprehending these components and utilizing insights from bioaccess®’s Global Trial Accelerators™, sponsors can enhance their readiness for the challenges and opportunities that early phase studies present in the region, ultimately driving their research towards success.
Navigate Regulatory Requirements for Clinical Trials
To successfully conduct clinical studies in Chile, sponsors must adeptly navigate a complex regulatory landscape established by the Instituto de Salud Pública (ISP) and the Agencia Nacional de Medicamentos (ANAMED). Here’s a step-by-step approach:
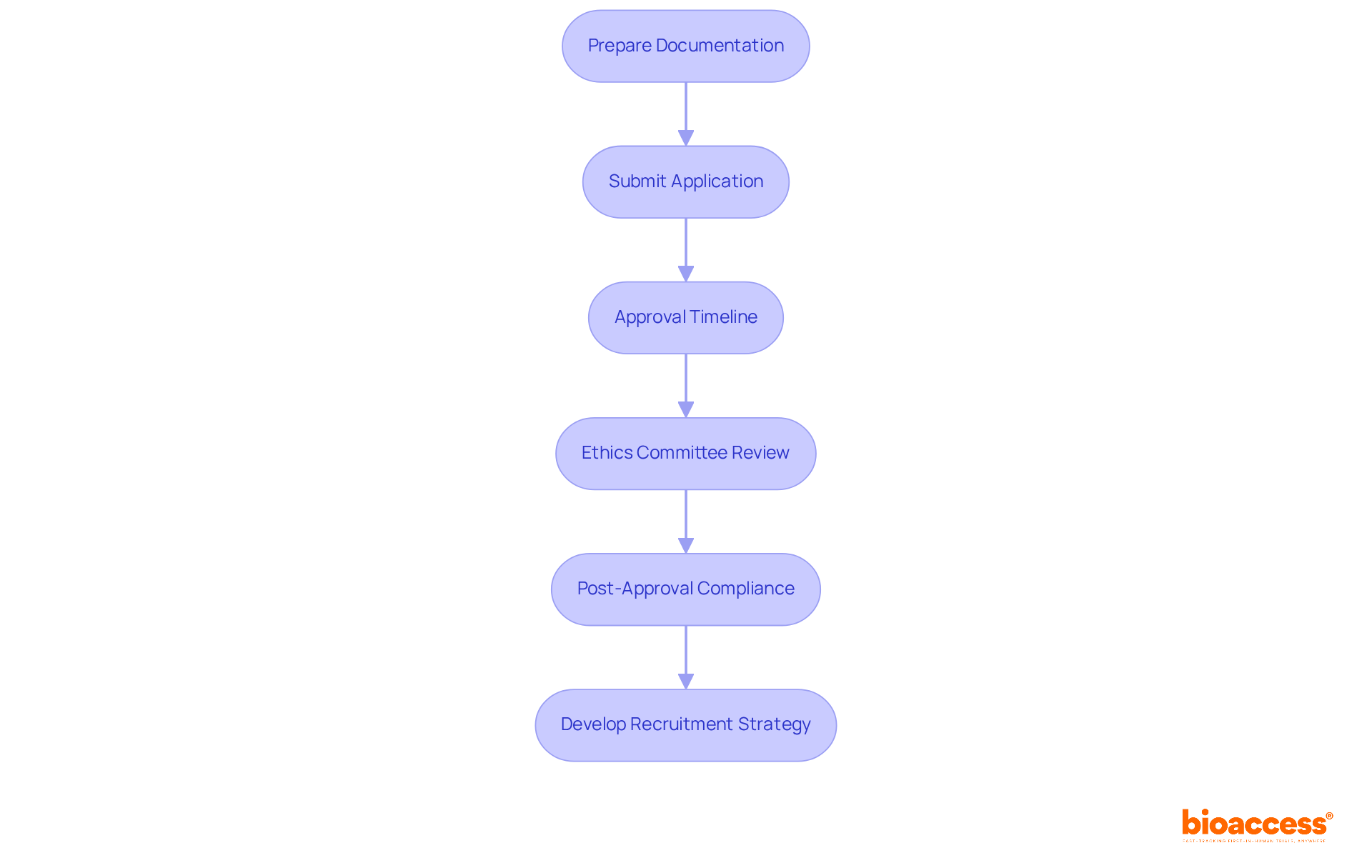
- Prepare Documentation: Gather all essential documents, including the research protocol, informed consent forms, and investigator brochures. Ensure that these documents adhere to ICH-GCP standards, which are vital for upholding the integrity and quality of the study.
- Submit Application: Submit your clinical study application to the ISP, including all compiled documents and any additional information requested by the regulatory body. This submission is crucial for kickstarting the approval process.
- Approval Timeline: The average duration for approval is approximately 30 business days, significantly faster than many other regions. The ISP has streamlined the research approval process, minimizing bureaucratic delays by over 30%. This represents a major advantage for conducting studies in Chile, enabling sponsors to commence trials more swiftly, particularly advantageous for First-in-Human (FIH) evaluations.
- Ethics Committee Review: Concurrently, submit your research for evaluation by an accredited ethics committee (EC). This step is vital for guaranteeing participant safety and ethical adherence, as only accredited ECs can approve research protocols. Compliance with the Declaration of Helsinki and local ethical standards is mandatory.
- Post-Approval Compliance: Once approved, you’ll need to keep up with regular reporting to the ISP and follow any conditions laid out during the approval process. Additionally, after receiving ISP approval, it’s essential to notify ANAMED to ensure all regulatory steps are completed.
- Develop Recruitment Strategy: A robust recruitment plan is essential for enrolling participants in research initiatives. Understanding the socio-political context in the region, including potential barriers to patient access, can enhance recruitment efforts and ensure a diverse participant pool. Leveraging bioaccess®’s expertise in early feasibility studies can further streamline this process, ensuring that sponsors can effectively engage with potential participants.
By adhering to these procedures and leveraging bioaccess®’s offerings, sponsors can optimize their regulatory submissions and improve the chances of a successful study launch in the region, positioning themselves for success in the dynamic Latin American market.
Implement Strategies for Successful Trial Execution
Navigating the complex regulatory landscape in Chile can be daunting for sponsors, but adopting an affordable clinical trial Chile strategy can pave the way for successful medical research implementation. Here are key strategies to consider:
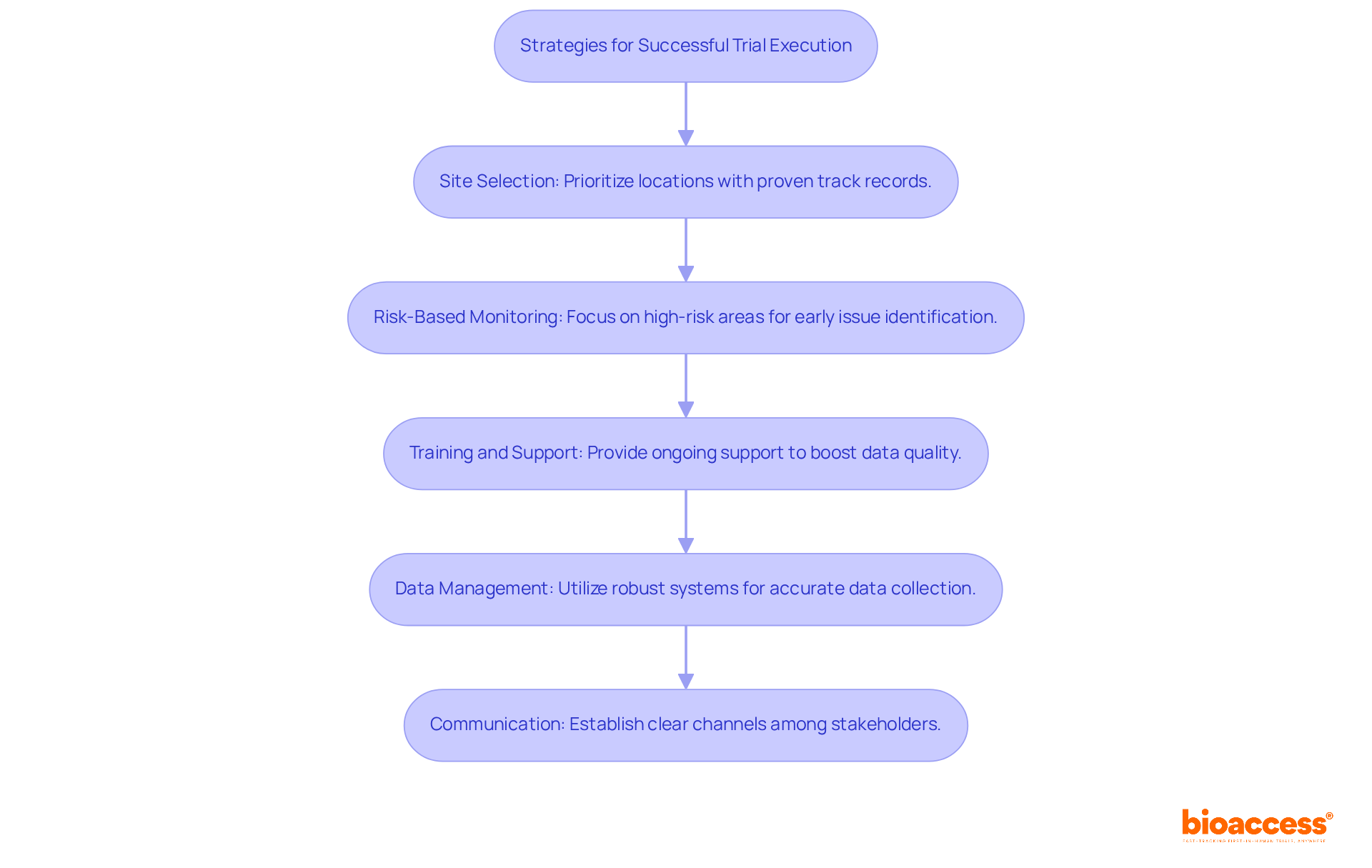
- Site Selection: Prioritize research locations with a proven track record in early phase studies. Assess their experience, infrastructure, and patient access to ensure they can meet the specific requirements of your study. Considering that over 96% of clinical studies in Chile are financed by external pharmaceutical companies, selecting sites with established relationships can enhance recruitment and retention for affordable clinical trial Chile. Utilizing bioaccess®’s pre-negotiated site agreements can result in substantial cost reductions of $25K per patient, improving the overall feasibility of your study.
- Risk-Based Monitoring: Implement a strategy focused on high-risk areas of the study by allocating resources effectively. This approach allows for early identification of potential issues, facilitating timely interventions and ensuring compliance with ICH-GCP standards. Reports suggest that effective risk management can greatly enhance research outcomes by addressing challenges proactively.
- Training and Support: Providing ongoing support during the study not only boosts data quality but also keeps participants safe, which is essential for preserving the integrity of the research and fulfilling regulatory expectations. Bioaccess® offers tailored training programs that align with local regulations, ensuring that your team is well-prepared.
- Data Management: Utilize robust data management systems to ensure accurate and timely data collection. This is crucial for preserving study integrity and enabling regulatory submissions, especially considering the average response time of 15 days from the Chilean Institute of Public Health for registration data. With bioaccess®, you can benefit from advanced data management solutions that streamline the process and improve efficiency.
- Communication: Establish clear communication channels among all stakeholders, including sponsors, investigators, and regulatory bodies. Regular updates and feedback loops can help address challenges promptly, ensuring that all parties are aligned and informed throughout the research process. Bioaccess® facilitates effective communication through its Global Trial Accelerators™, providing essential insights and market access strategies tailored for MedTech and Biopharma innovators in Latin America.
By embracing these strategies, sponsors not only enhance their study outcomes but also contribute to the advancement of affordable clinical trial Chile in healthcare.
Enhance Patient Recruitment and Retention Strategies
Despite the critical importance of patient enrollment and retention, many studies in Chile struggle to engage participants effectively in affordable clinical trial settings. Here are targeted strategies to enhance these processes:
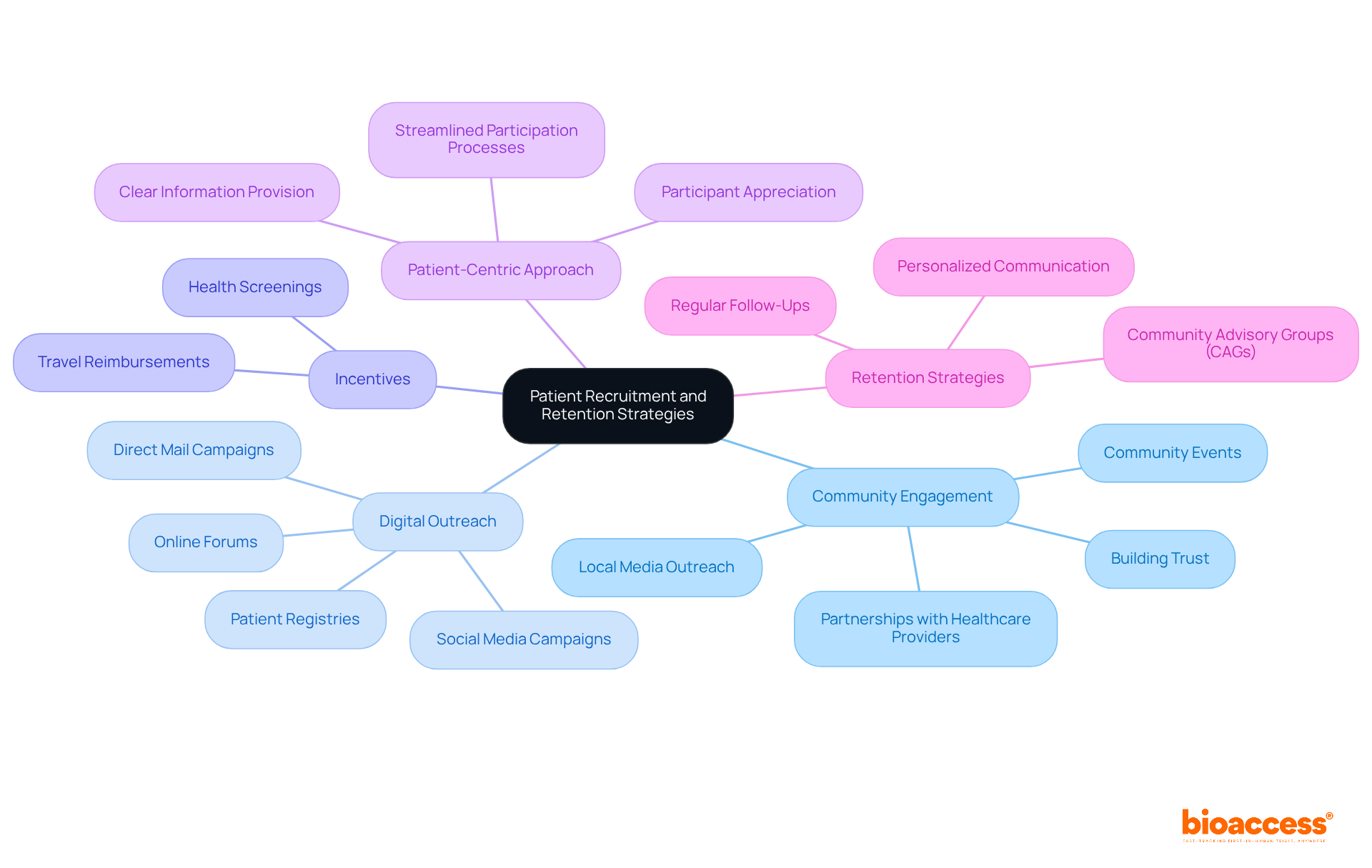
- Community Engagement: Actively engage with local communities to raise awareness about clinical trials. Utilize local media, community events, and partnerships with healthcare providers to effectively reach potential participants. This approach fosters trust and encourages participation, as evidenced by the strong doctor-patient relationships prevalent in the region.
- Digital Outreach: Leverage digital platforms for recruitment campaigns. Social media, online forums, and patient registries can efficiently identify and engage eligible candidates. Have you thought about using direct mail campaigns and targeted Facebook advertisements? One report indicates over 2.5 million impressions from Facebook ads alone, resulting in increased inquiries.
- Incentives: Have you thought about offering incentives for participation, like travel reimbursements or health screenings? These incentives can motivate individuals to enroll and remain committed to the study, addressing potential barriers to participation.
- Patient-Centric Approach: Design studies with the patient experience in mind. Streamline participation processes, offer clear information about the study, and ensure that participants feel appreciated and supported throughout their involvement. This approach can significantly enhance retention rates.
- Retention Strategies: Implement proactive retention strategies, such as regular follow-ups and personalized communication. Address participant concerns promptly and provide updates on study progress to keep them engaged. Community Advisory Groups (CAGs) can play a vital role in maintaining participant engagement by fostering a sense of community and support.
Improving these strategies is not just beneficial; it is essential for the integrity and success of affordable clinical trial Chile.
Conclusion
Conducting affordable clinical trials in Chile presents a unique opportunity for MedTech and Biopharma companies willing to tackle the complexities of early phase studies. Chile’s robust regulatory framework, cost efficiency, and diverse patient population provide strategic advantages that foster innovative research. Leveraging these factors is essential for achieving successful outcomes in clinical trials.
Key insights from this guide highlight the importance of:
- Navigating regulatory requirements effectively
- Implementing robust recruitment strategies
- Ensuring compliance with ICH-GCP standards
Sponsors must adopt a structured approach to documentation, approval processes, and participant engagement to streamline operations and mitigate risks. Furthermore, embracing community engagement and digital outreach can improve patient recruitment and retention, which are crucial for the integrity of clinical trials.
In conclusion, the landscape for early phase clinical trials in Chile is not only favorable but also ripe with potential for those willing to adopt a strategic approach. By taking advantage of the cost-effective solutions and regulatory efficiencies available, sponsors can position themselves for success in the competitive field of clinical research. To thrive in this competitive landscape, stakeholders must act decisively, embracing the opportunities that lie ahead.
Frequently Asked Questions
What are early phase clinical trials, specifically first-in-human (FIH) studies?
Early phase clinical trials, particularly FIH studies, are critical in the drug development process as they evaluate the safety and efficacy of new therapies in humans for the first time.
What regulatory body oversees clinical trials in Chile?
The Instituto de Salud Pública (ISP) is the regulatory authority that governs clinical trials in Chile, ensuring compliance with national and international standards, including ICH-GCP guidelines.
How long does the approval process for clinical trials take in Chile?
The approval process typically lasts between 30 to 60 days, with the ISP assessing applications within 30 business days.
What are the cost advantages of conducting clinical trials in Chile compared to the U.S.?
Conducting clinical trials in Chile can offer a cost advantage of up to 40% compared to the U.S., making it a strategic choice for MedTech and Biopharma startups.
Why is the regulatory framework important for clinical trials in Chile?
The regulatory framework established by the ISP is essential for ensuring that clinical trials adhere to both national and international guidelines, which is crucial for the successful execution of studies.
How does the diverse patient population in Chile benefit clinical trials?
The varied demographics in Chile enhance recruitment potential, allowing access to a broad spectrum of participants. This diversity accelerates enrollment and improves the representativeness of study results, which is vital for clinical research.
How can sponsors prepare for early phase studies in Chile?
Sponsors can enhance their readiness for early phase studies by understanding the regulatory framework, leveraging cost efficiency, and utilizing insights from bioaccess®’s Global Trial Accelerators™ to navigate challenges and opportunities in the region.
List of Sources
- Understand Early Phase Clinical Trials in Chile
- Trends in clinical trials performed in Chile (https://scielo.cl/article_plus.php?pid=S0034-98872021000100110&tlng=en&lng=es)
- Master Early Phase Clinical Trials in Chile: Key Strategies and Insights – cms.bioaccessla.com (https://cms.bioaccessla.com/blog/master-early-phase-clinical-trials-in-chile-key-strategies-and-insights)
- Clinical trials by start date Chile 1999-2024| Statista (https://statista.com/statistics/1560150/chile-number-new-clinical-trials?srsltid=AfmBOorZfdDWgLFN4scg4mpP4PN4GvW5AlH4oBlN3ZPu_u7GVhAZzokh)
- Navigate Regulatory Requirements for Clinical Trials
- Clinical trials by start date Chile 1999-2024| Statista (https://statista.com/statistics/1560150/chile-number-new-clinical-trials?srsltid=AfmBOoqAaIyhPOudE-3CnJ-2rG8iEkg32x2e8C9jKw8O98pAIF_Ci2N5)
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Master FDA Accepted Clinical Trials in Chile: A Step-by-Step Tutorial | bioaccess® (https://bioaccessla.com/blog/master-fda-accepted-clinical-trials-in-chile-a-step-by-step-tutorial)
- Clinical Trials in Latin America (https://languageconnections.com/clinical-trials-in-latin-america)
- How to Conduct First-in-Human Trials in Chile: A Step-by-Step Guide | bioaccess® (https://bioaccessla.com/blog/how-to-conduct-first-in-human-trials-in-chile-a-step-by-step-guide)
- Implement Strategies for Successful Trial Execution
- Trends in clinical trials performed in Chile (https://scielo.cl/article_plus.php?pid=S0034-98872021000100110&tlng=en&lng=es)
- Best Practices for Selecting Investigator Sites in Chile for Clinical Trials | bioaccess® (https://bioaccessla.com/blog/best-practices-for-selecting-investigator-sites-in-chile-for-clinical-trials)
- Clinical trials by start date Chile 1999-2024| Statista (https://statista.com/statistics/1560150/chile-number-new-clinical-trials?srsltid=AfmBOoo2lZFnkVetcBVZ7wGD75q2hgzL9HxCsd3fRjZ4_UyDRXJVOENx)
- Enhance Patient Recruitment and Retention Strategies
- Latin America: A Compelling Region To Conduct Your Clinical Trials (https://clinicalleader.com/doc/latin-america-a-compelling-region-to-conduct-your-clinical-trials-0001)
- Clinical trials by start date Chile 1999-2024| Statista (https://statista.com/statistics/1560150/chile-number-new-clinical-trials?srsltid=AfmBOopgWM_mePVImKyY_G0yLadVejeP-2EpQOKrCVwDv9_Hse-lRqX-)
- Community Informed Recruitment: A Promising Method to Enhance Clinical Trial Participation (https://jpsmjournal.com/article/S0885-3924(23)00398-6/fulltext)
- Community engagement strategies improve recruitment and enrollment in a pragmatic clinical trial – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC12444702)

Leave a Reply