Introduction
Researchers face a daunting landscape when it comes to first-in-human (FIH) clinical trials in Chile, where both challenges and opportunities abound. The regulatory landscape, shaped by the Instituto de Salud Pública (ISP), along with a growing network of compliant sites, can feel overwhelming, but knowing the right steps can lead to successful trial execution.
What strategies can researchers employ to navigate the intricacies of compliance, patient recruitment, and data management, ensuring their studies reach their full potential and bring innovative therapies to market faster?
Understand the Regulatory Landscape for FIH Trials in Chile
Navigating the regulatory landscape for the FIH clinical trial Chile can be daunting, yet it is crucial for successful clinical research. Understanding the regulatory framework established by the Instituto de Salud Pública (ISP) is essential. Here are the key steps:
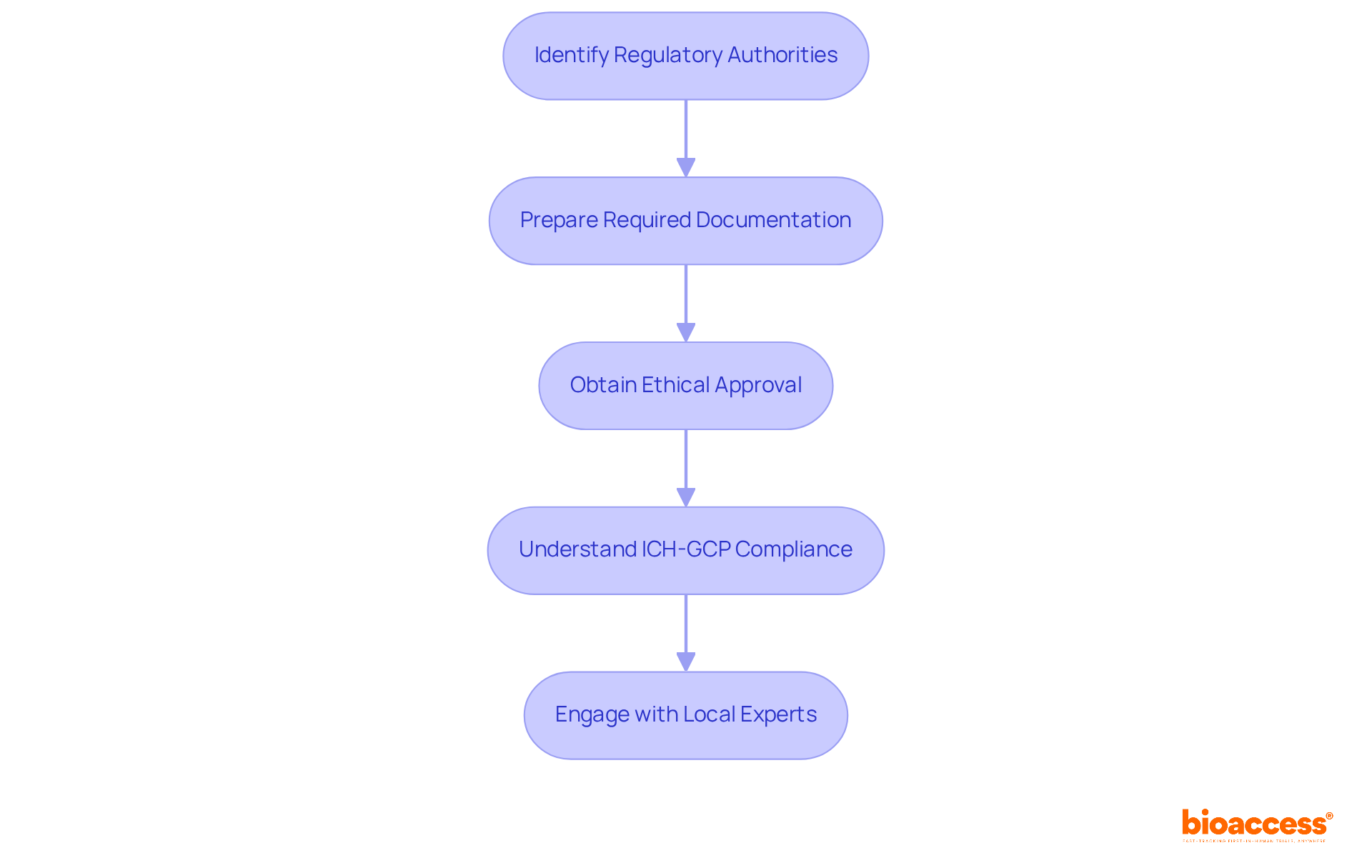
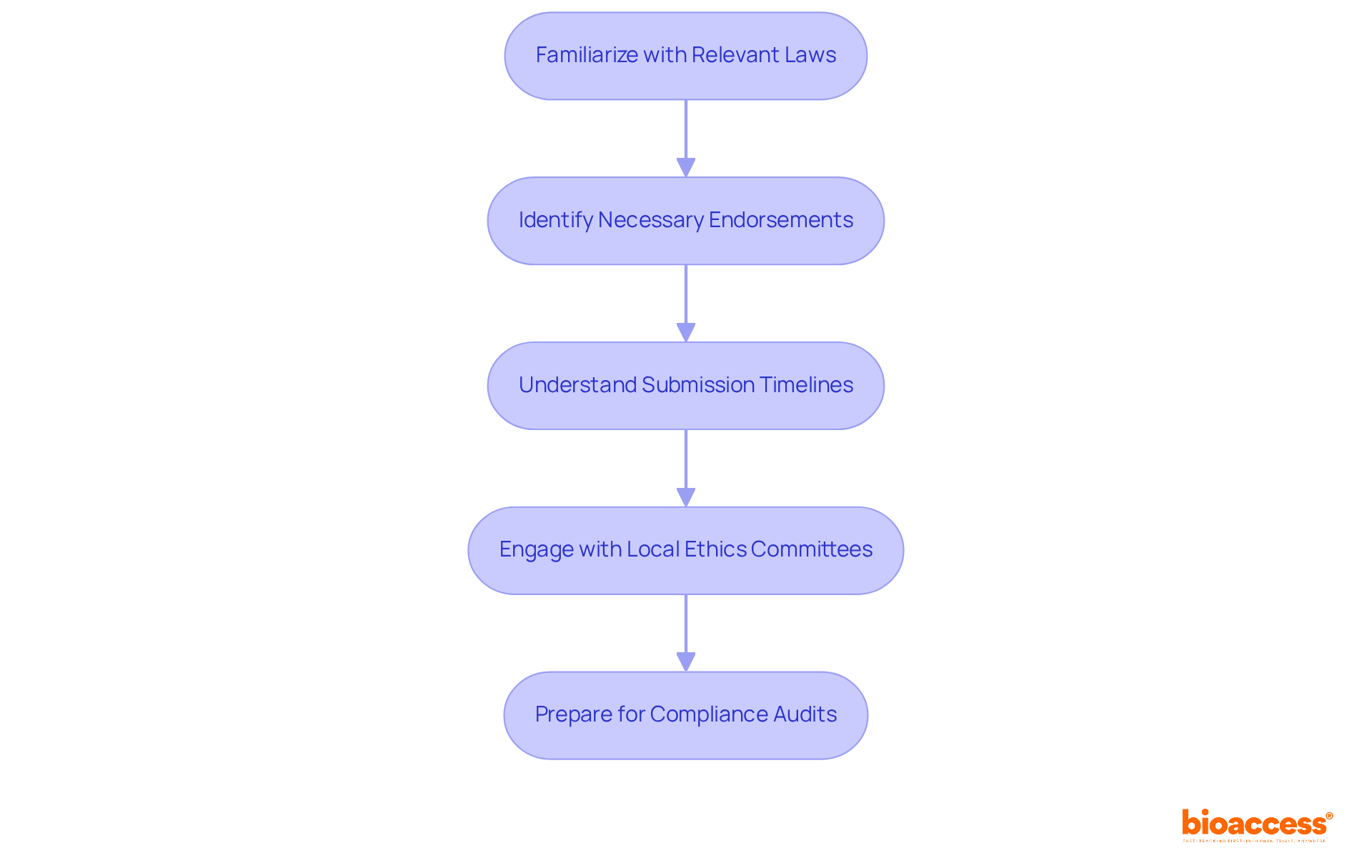
- Familiarize Yourself with Relevant Laws: Review Law No. 20,120, which governs human research in Chile. This law establishes the ethical and scientific standards necessary for clinical evaluations, ensuring participant safety and data integrity.
- Identify Necessary Endorsements: Acquire required endorsements from the ISP by presenting a detailed study protocol outlining the objectives, methodology, and ethical considerations of the investigation. This step is crucial for compliance and successful trial execution.
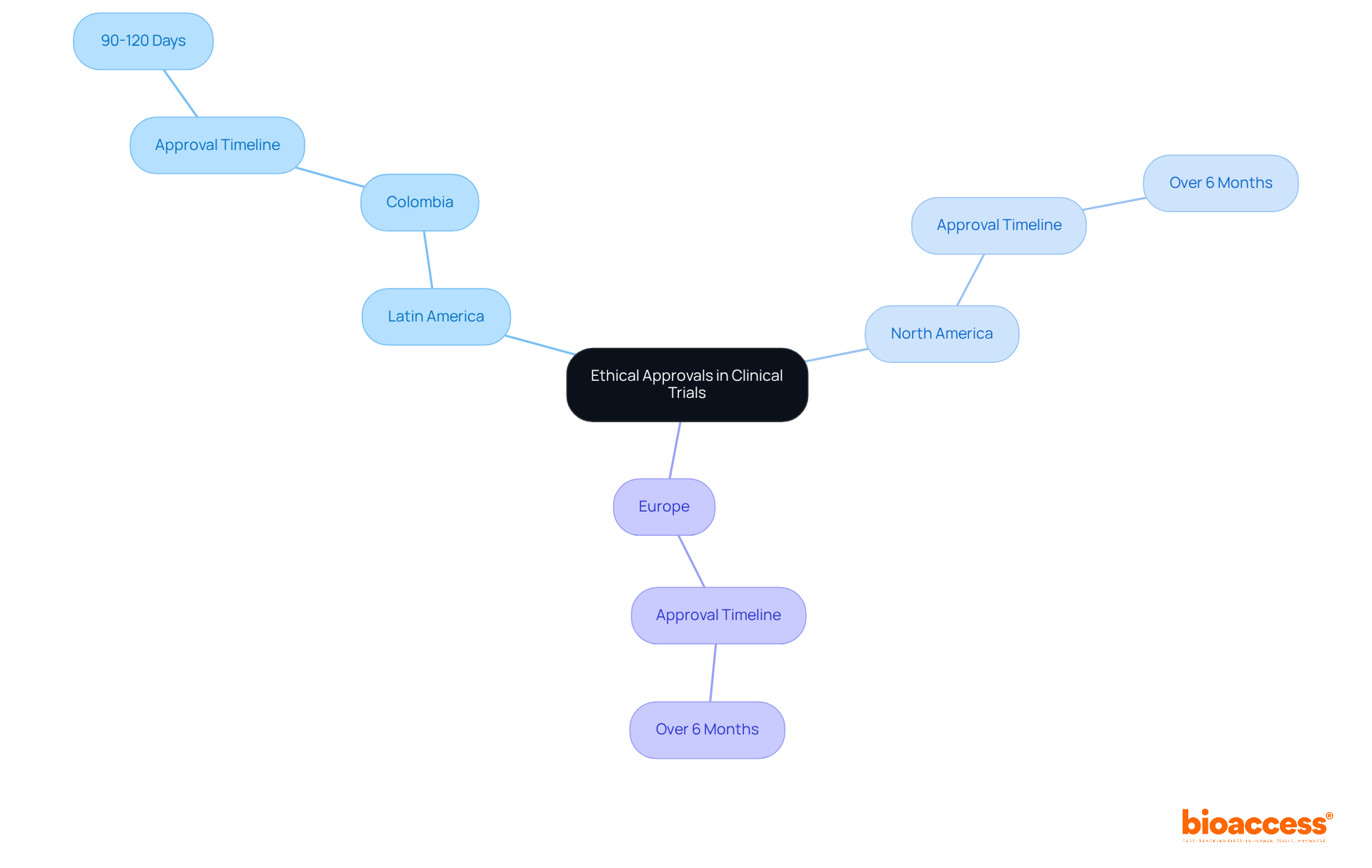
- Understand Submission Timelines: The authorization process typically spans 30 to 60 days. Preparing your documentation in advance can help mitigate delays and streamline the review process.
- Engage with Local Ethics Committees: Collaborate with accredited ethics committees to ensure your study adheres to ethical standards. This engagement is crucial for obtaining the necessary permissions and ensuring compliance throughout the study.
- Prepare for Compliance Audits: Be ready for potential audits by the ISP or ethics committees. Ensure that all study documentation is thorough and complies with ICH-GCP guidelines, which are critical for maintaining the integrity of your research.
By mastering these compliance aspects, you not only pave the way for your study’s success but also position yourself to capitalize on the unique opportunities within the FIH clinical trial Chile. Furthermore, consider subscribing to bioaccess®’s clinical insights service, which offers valuable updates on regulatory changes and market access strategies, enhancing your ability to leverage the region’s strategic advantages in speed and cost efficiency.
Select Sites and Recruit Patients Effectively
Navigating the complexities of the FIH clinical trial in Chile requires strategic site selection and effective participant recruitment. Follow these steps to ensure success:
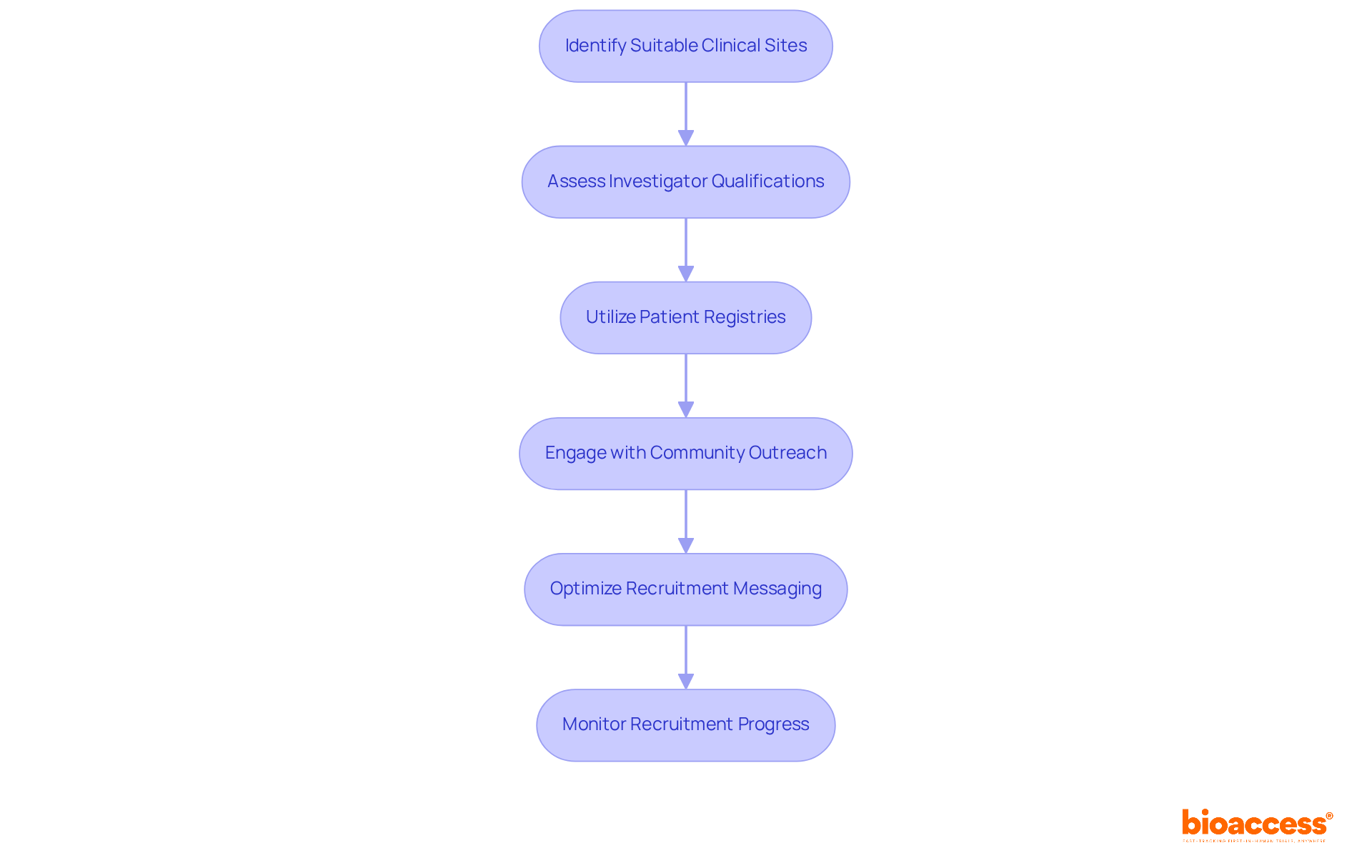
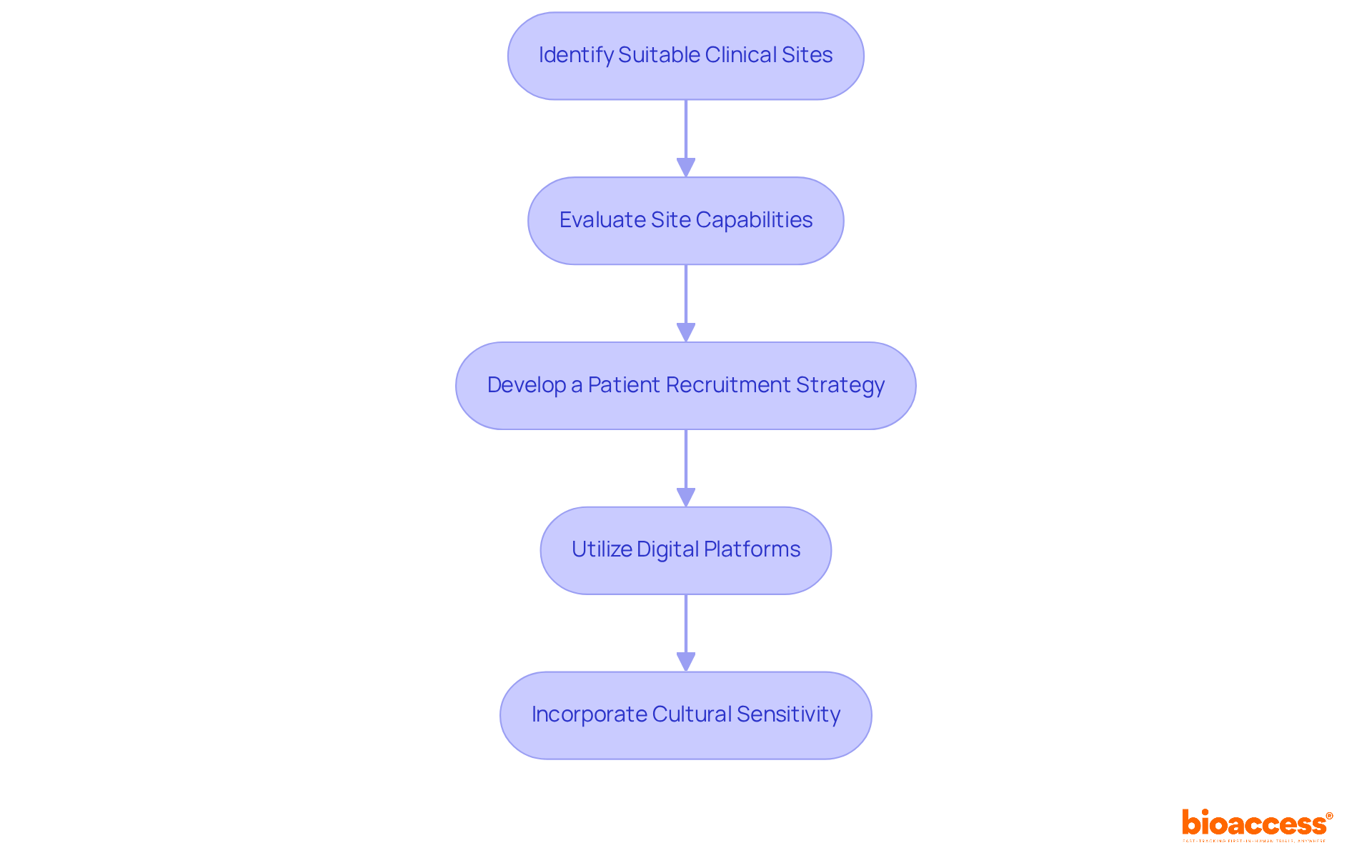
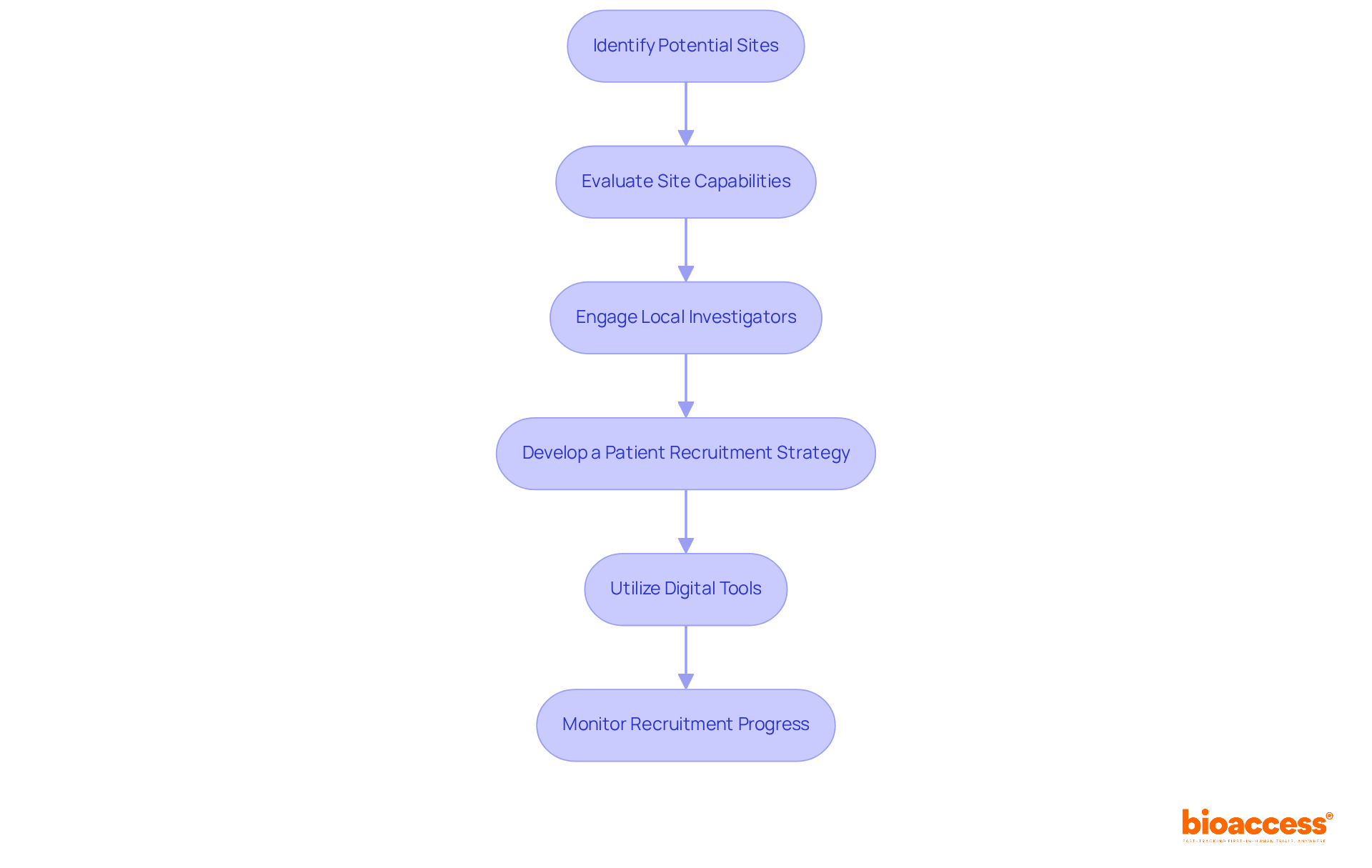
- Identify Potential Sites: Concentrate on clinical locations with demonstrated experience in FIH studies and a robust compliance record with ICH-GCP standards. Leverage bioaccess®’s extensive network of pre-qualified sites to streamline your selection process.
- Evaluate Site Capabilities: Assess each site’s infrastructure, ensuring access to necessary medical equipment and qualified staff. Confirm that they can meet the specific demands of your study, including surgical capabilities and procurement flexibility.
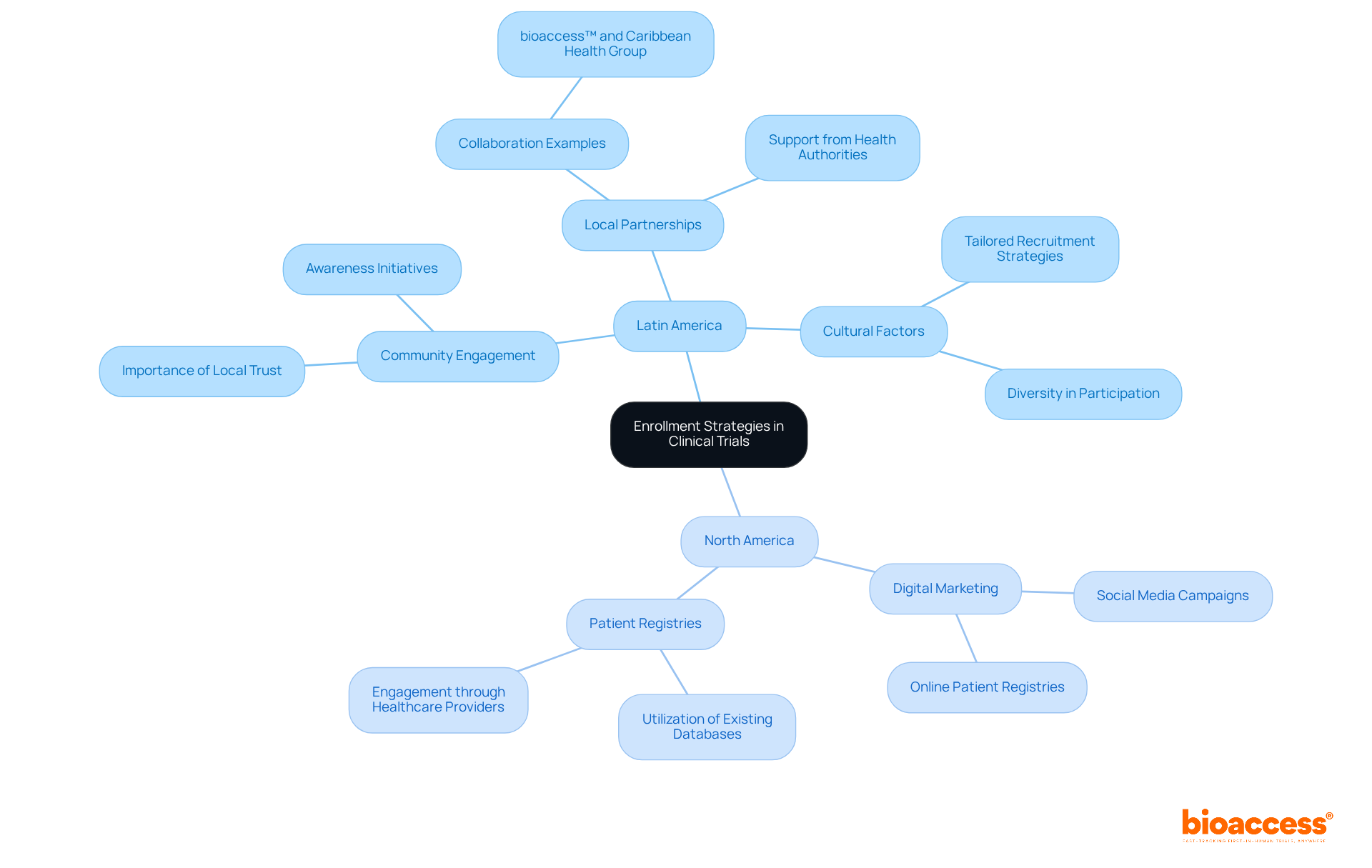
- Engage Local Investigators: Collaborate with local principal investigators who have established relationships within the patient community. Their insights and connections can significantly enhance recruitment efforts and build patient trust.
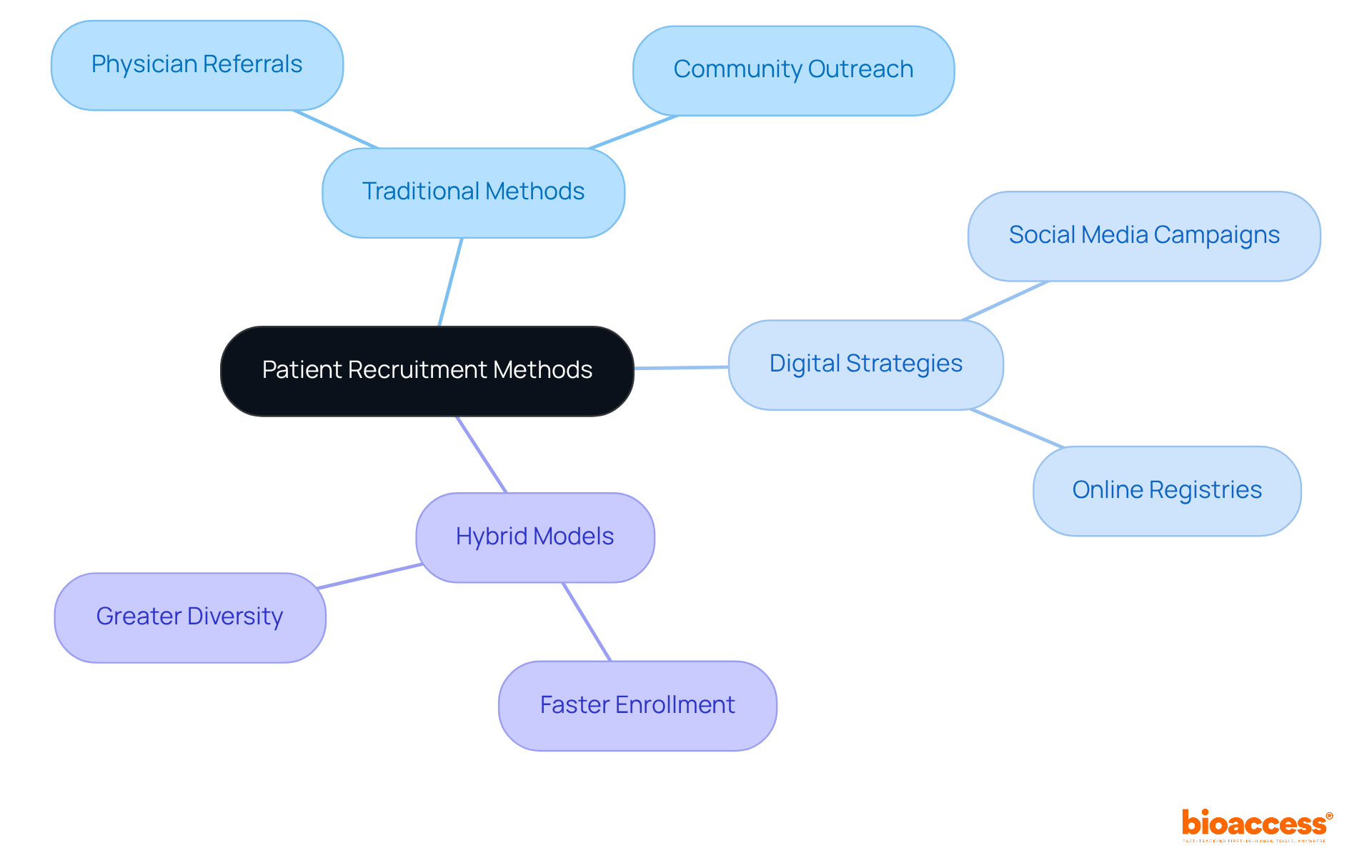
- Develop a Patient Recruitment Strategy: Create a comprehensive recruitment plan that includes outreach to local healthcare providers, community organizations, and online platforms. Tailor your messaging to resonate with potential participants, emphasizing the benefits of participation.
- Utilize Digital Tools: Leverage digital marketing and social media to broaden your reach. Consider using patient registries and databases to identify eligible candidates efficiently, enhancing your recruitment pool.
- Monitor Recruitment Progress: Regularly track recruitment metrics and adjust your strategies as necessary to ensure you meet enrollment targets within the planned timelines. This proactive strategy can assist in reducing delays frequently faced in clinical studies.
Implementing these strategies is crucial. Without them, the risk of delays and regulatory challenges in your FIH study could significantly increase.
Execute the Trial: Manage Timelines and Monitor Progress
Successfully executing a FIH clinical trial in Chile requires not just planning, but a strategic approach to overcome inherent challenges. Here’s how to effectively manage timelines and monitor progress:
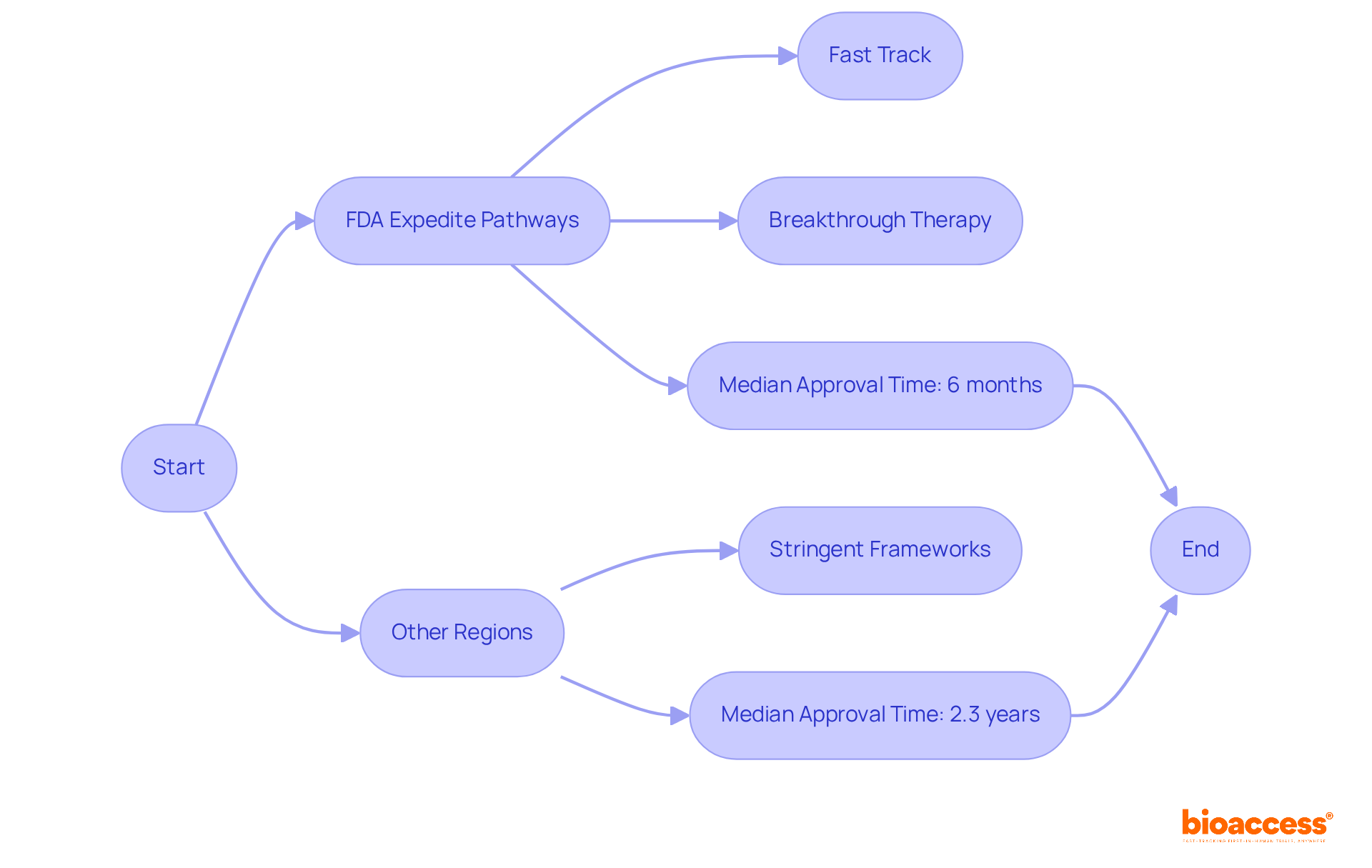
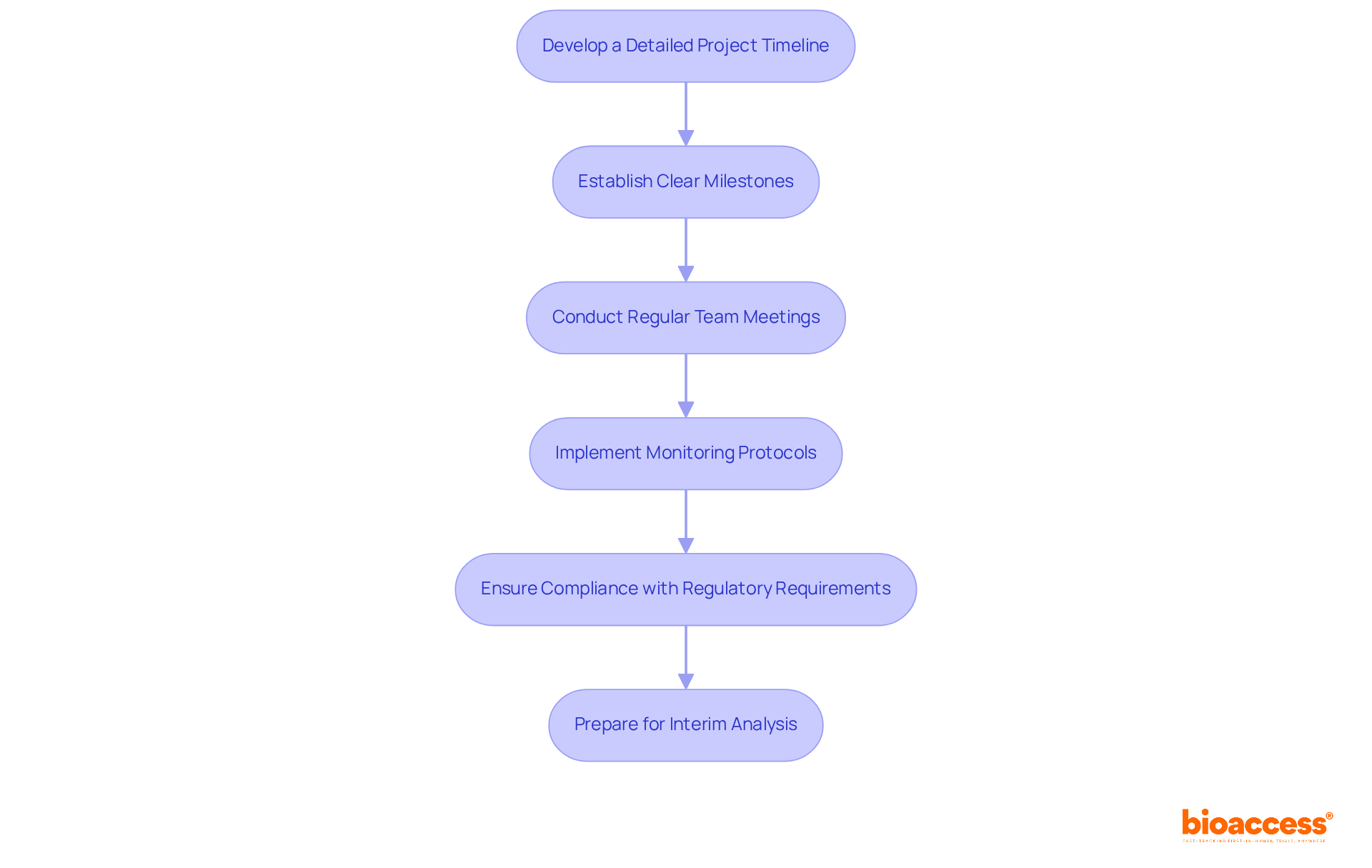
- Develop a Detailed Project Timeline: Create a comprehensive timeline that outlines all phases of the study, including site activation, patient enrollment, and data collection. Use project management tools to visualize progress. This ensures alignment with regulatory timelines set by authorities like ANVISA or COFEPRIS, which typically offer ethics approvals in just 4-8 weeks-far quicker than the 6+ months often required in the US and EU.
- Establish Clear Milestones: Set specific milestones for each phase of the study. This approach facilitates tracking progress and allows for early identification of potential delays, ensuring that the study adheres to the 12-month protocol-to-LPLV execution timeline. As indicated by a clinical study project manager, “Establishing clear milestones is vital for sustaining momentum and ensuring accountability within the team.”
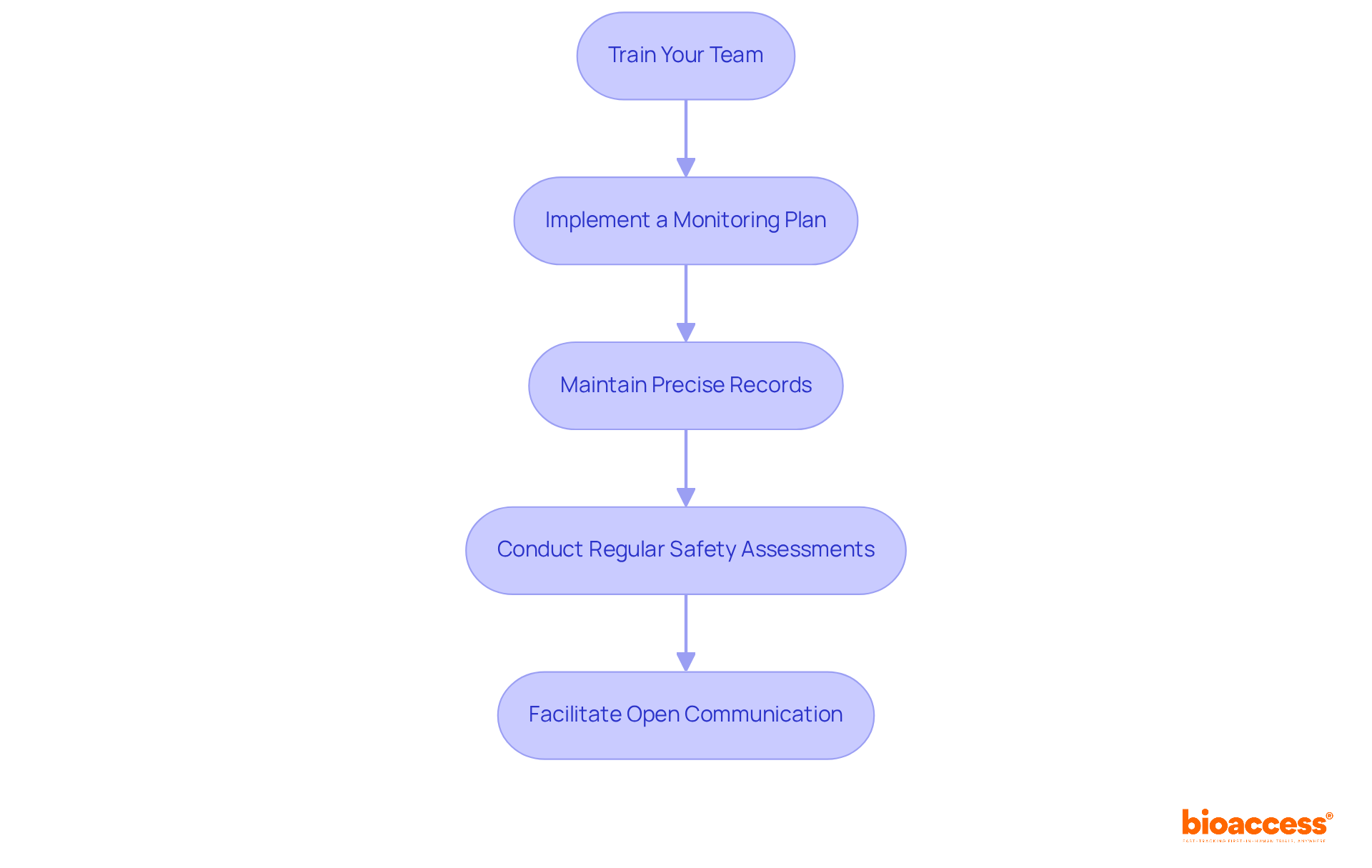
- Conduct Regular Team Meetings: Schedule frequent meetings with your clinical operations team to discuss progress, address challenges, and adjust timelines as necessary. Keeping the lines of communication open helps the team stay aligned and respond quickly to any issues that pop up.
- Implement Monitoring Protocols: Utilize real-time monitoring tools to track patient enrollment and information collection. This proactive approach enables quick identification of issues, ensuring that recruitment remains on target and compliant with ICH-GCP standards. For example, a diabetes study achieved a 95% retention rate by incorporating innovative monitoring methods, demonstrating the effectiveness of such tools.
- Ensure Compliance with Regulatory Requirements: Regularly review compliance with ICH-GCP guidelines and local regulations. Conduct internal audits to ensure that all experimental activities are documented and meet the stringent standards required for FDA-bridgeable data acceptance. Comprehending the specific needs of regulatory agencies such as ANVISA and COFEPRIS is crucial for successful execution of studies.
- Prepare for Interim Analysis: Plan for interim evaluations to assess the study’s progress. This enables well-informed choices about the continuation, alteration, or halt of the study based on the gathered data, enhancing resource allocation and efficiency.
By adhering to these steps, you can efficiently oversee your FIH clinical trial in Chile, ensuring that it stays on track and meets all compliance standards. Embracing these strategies not only enhances your study’s efficiency but also positions your innovative therapies for a faster market entry, ultimately benefiting patients in need.
Manage Data and Prepare for Regulatory Submissions
Navigating the complexities of compliance submissions is crucial for the success of your FIH clinical trial in Chile. Here’s how to ensure these processes are handled efficiently:
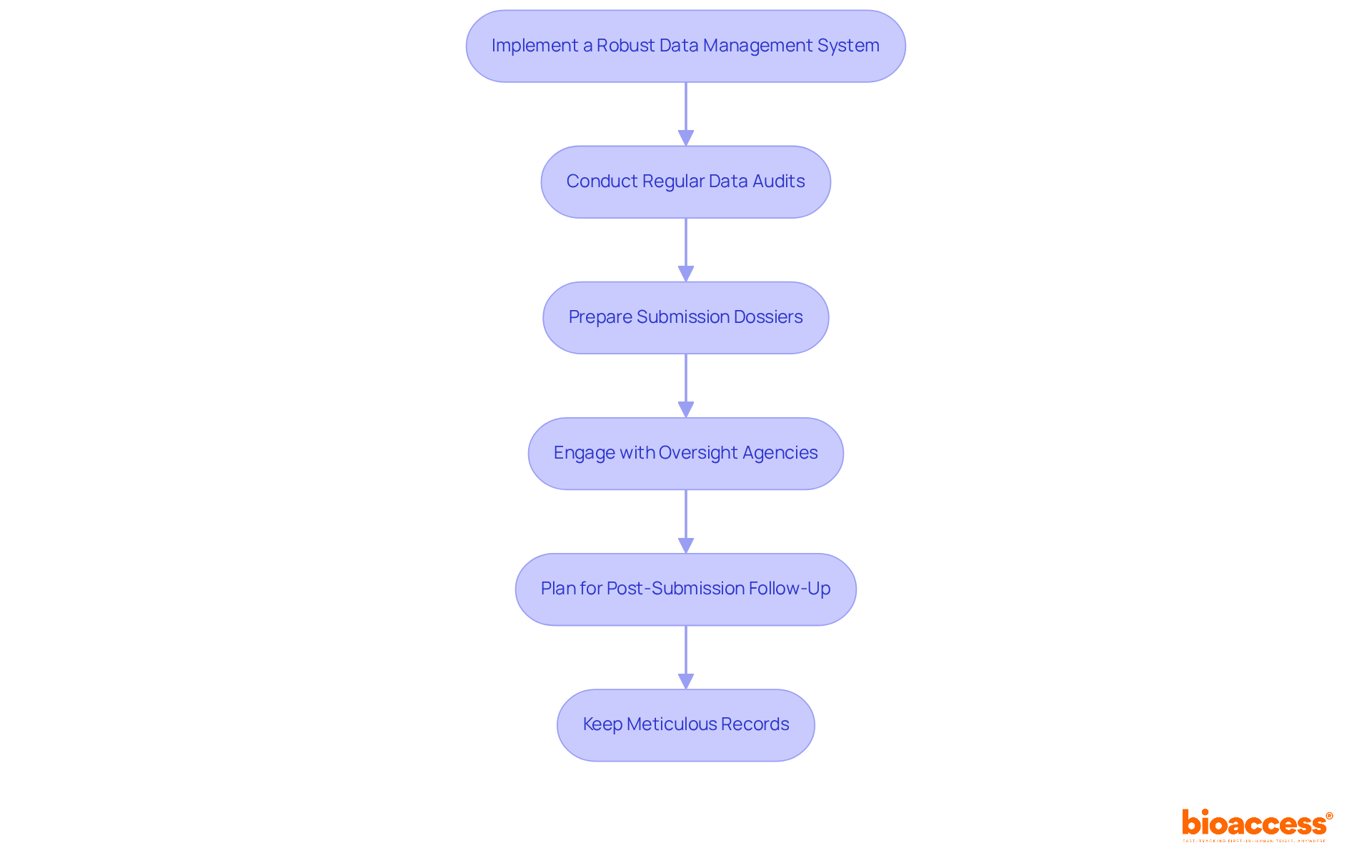
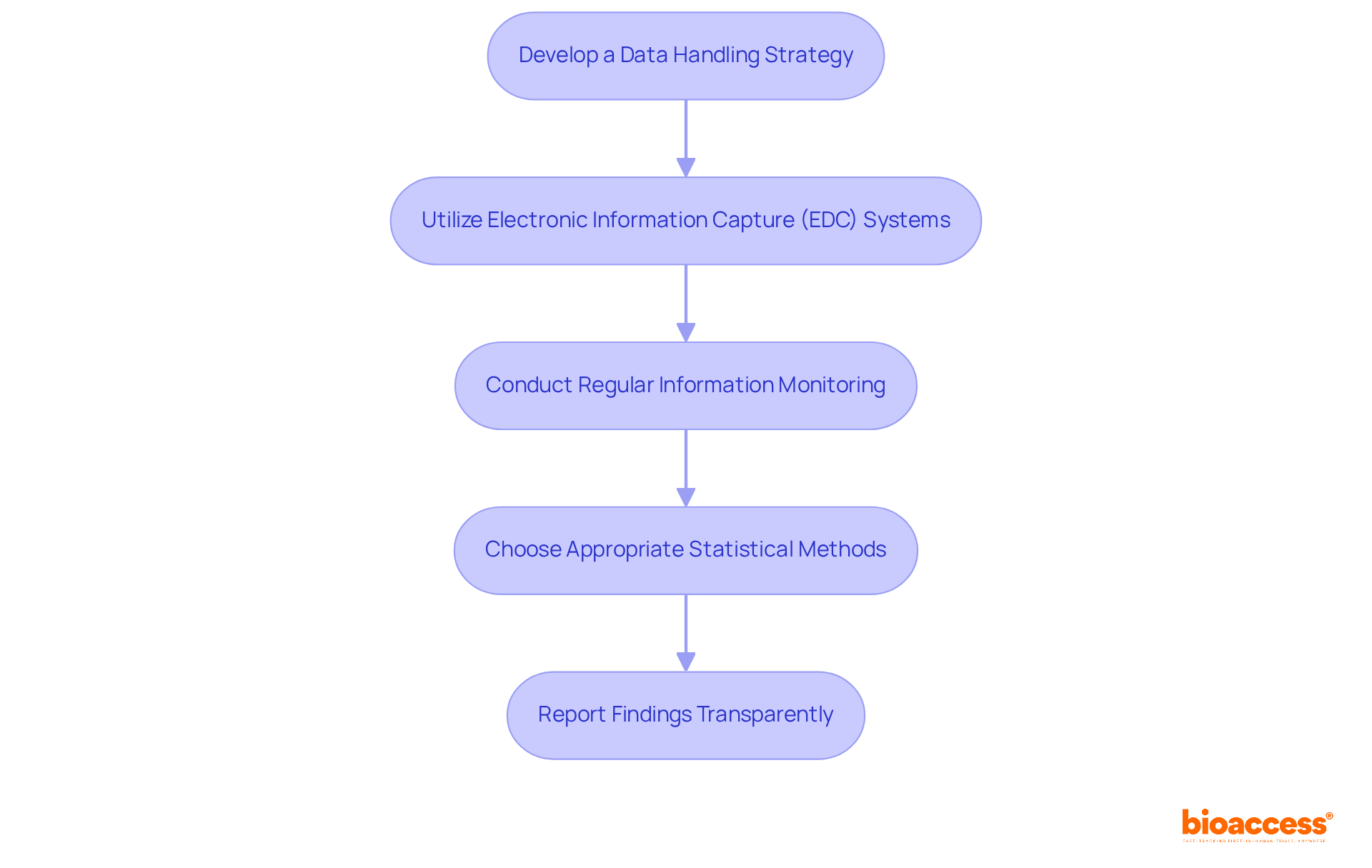
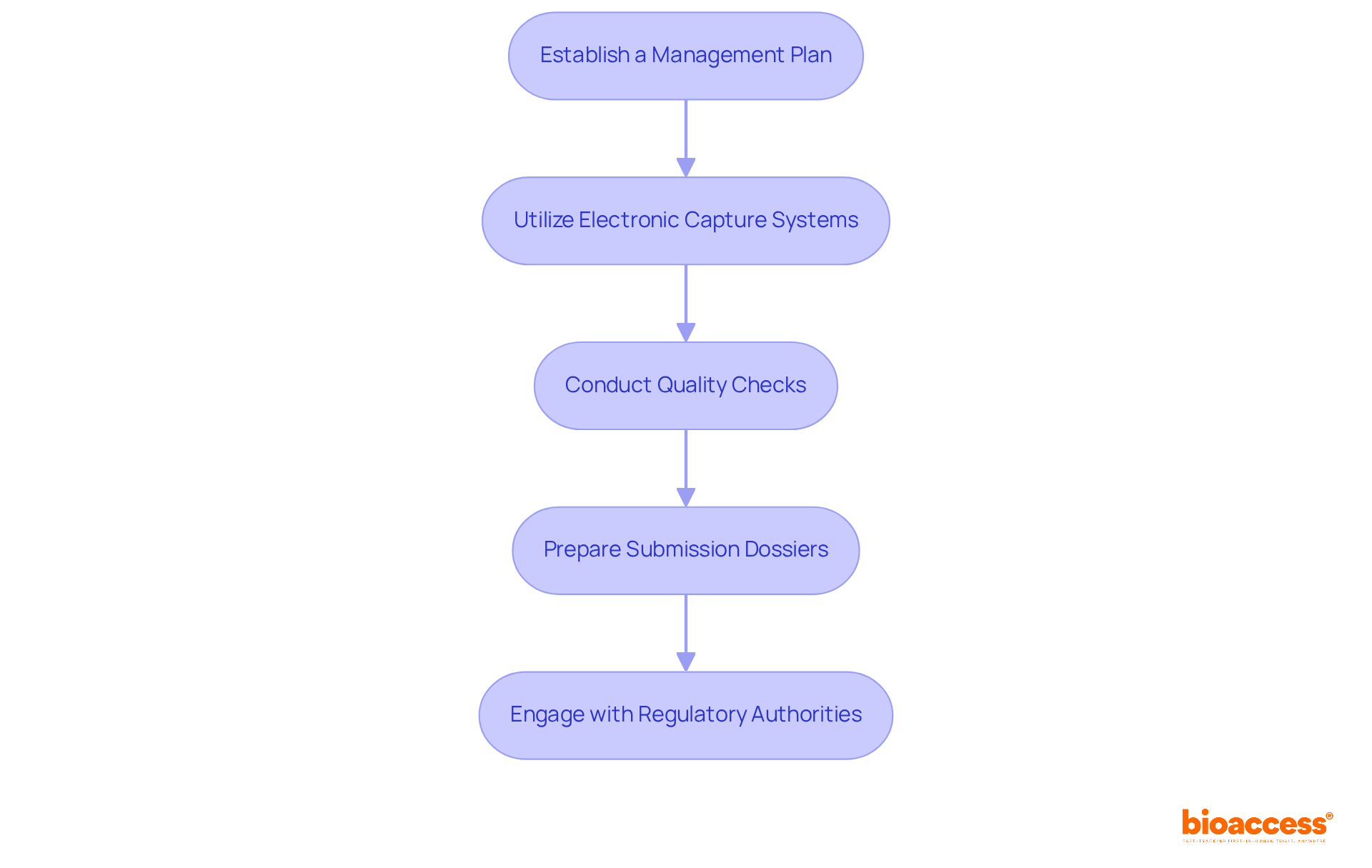
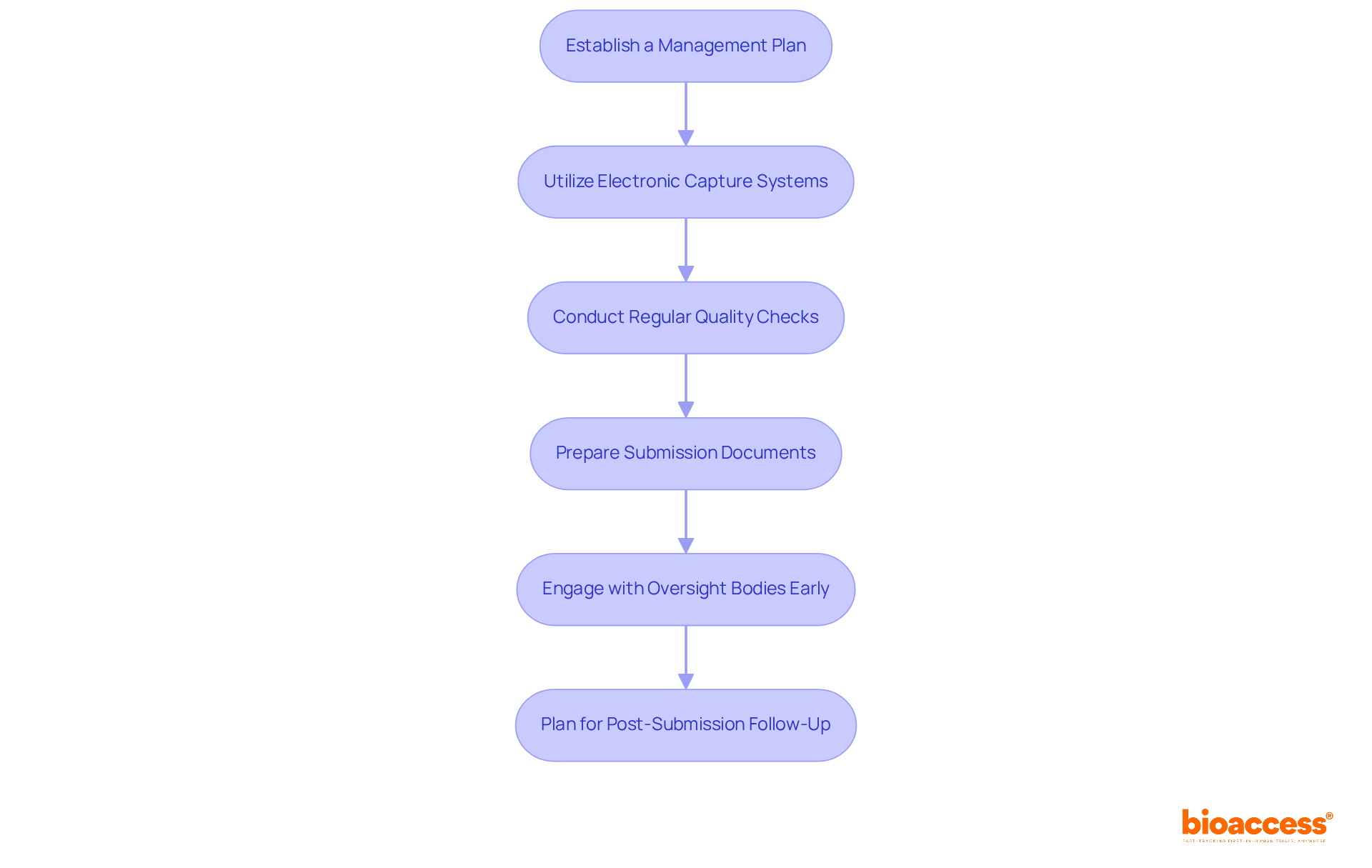
- Establish a Management Plan (MP): Develop a comprehensive MP that details how information will be collected, stored, and analyzed, ensuring compliance with ICH-GCP guidelines and local regulations such as those set by INVIMA in Colombia or ANVISA in Brazil. This plan should also outline specific timelines for regulatory approvals, which can be significantly faster in Latin America compared to other regions, leveraging insights from Global Trial Accelerators™.
- Utilize Electronic Capture Systems: Implement EDC systems to streamline information collection and minimize errors. These systems enhance information quality by permitting real-time input and validation, significantly reducing the risk of human error. As Carlos Verdú, a Clinical Data Manager, states, “EDC systems enable quicker information processing and enhance compliance with regulations by ensuring clean, complete datasets.”
- Conduct Regular Quality Checks: Perform routine assessments to ensure integrity and accuracy. Address any inconsistencies promptly to maintain high-quality information, which is essential for compliance submissions and overall trial success.
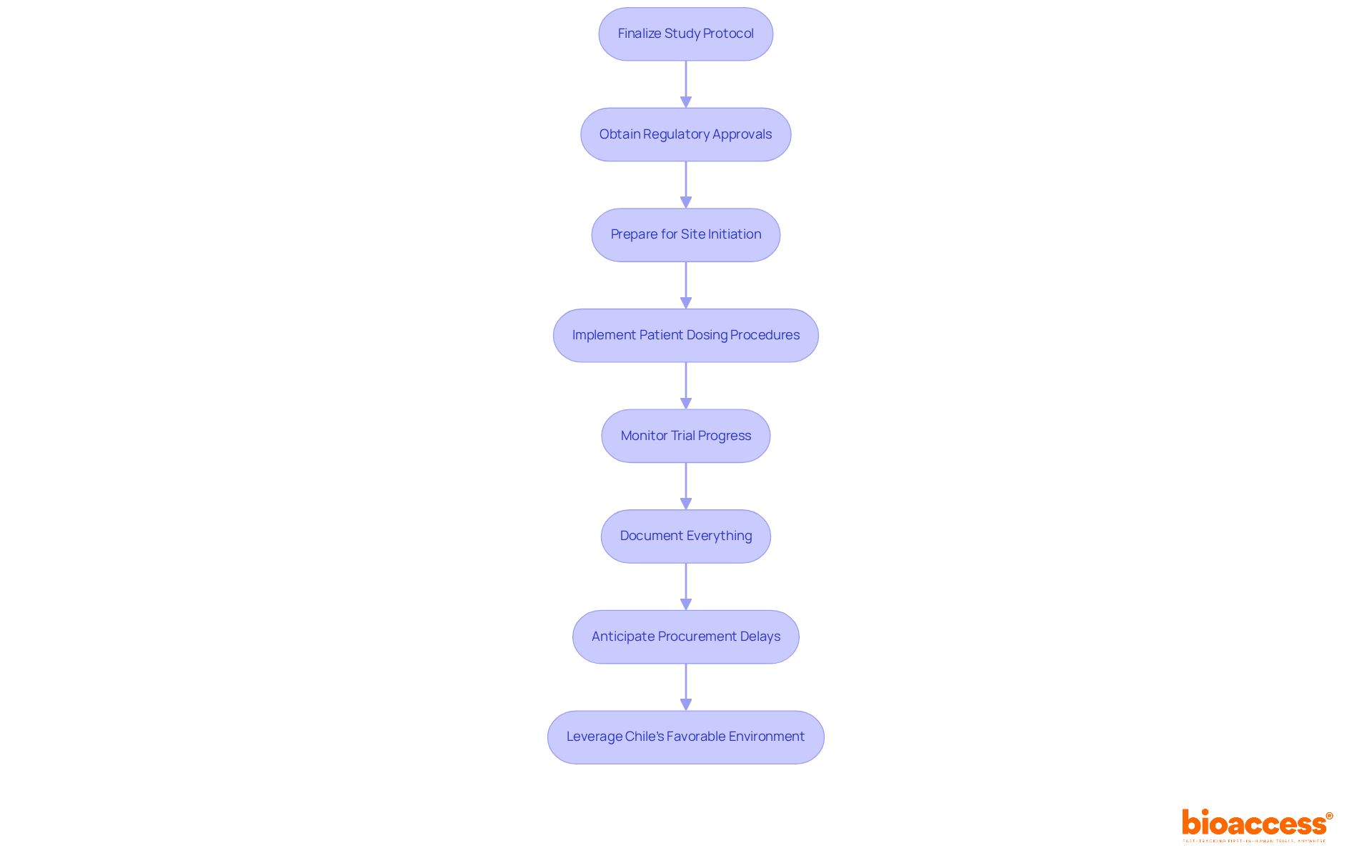
- Prepare Submission Documents: Compile all necessary documentation for submissions to authorities, including study protocols, informed consent forms, and data analysis reports. Ensure that these documents meet the requirements set by local governing bodies, such as the ISP in Chile, to facilitate a smoother review process.
- Engage with Oversight Bodies Early: Maintain open communication with oversight entities throughout the submission process. Early engagement can clarify questions and expedite the review process, which is critical in fast-paced environments like Latin America. Understanding the specific compliance requirements and authorization timelines can greatly improve your submission strategy, as emphasized by the Global Trial Accelerators™ insights.
- Plan for Post-Submission Follow-Up: After submission, be prepared to respond promptly to any inquiries from oversight authorities. Taking this proactive approach helps ensure a smoother approval process and keeps your clinical study timeline on track.
By effectively managing data and preparing for regulatory submissions, you enhance the likelihood of a successful outcome for the FIH clinical trial in Chile, leveraging the region’s strategic advantages in speed, cost efficiency, and patient recruitment, as emphasized by bioaccess®.
Conclusion
Navigating the first-in-human (FIH) clinical trial landscape in Chile presents unique challenges that demand a strategic approach to regulatory compliance and patient engagement. Successfully addressing these challenges requires a comprehensive understanding of regulatory requirements, effective site selection, and robust patient recruitment strategies. Mastering these elements significantly enhances researchers’ chances of conducting successful trials, capitalizing on Latin America’s advantages in speed, cost efficiency, and patient access.
This guide outlines key steps, highlighting the need to familiarize with local regulations like Law No. 20,120 and engage with ethics committees for compliance. Additionally, strategic site selection and innovative recruitment tactics play crucial roles in minimizing delays and optimizing enrollment. Implementing detailed project management practices and utilizing electronic data capture systems further streamline the trial process, ensuring adherence to ICH-GCP standards and enhancing data integrity.
The potential for successful FIH clinical trials in Chile is immense, driven by the region’s regulatory efficiency and access to diverse patient populations. The commitment to these strategies not only accelerates timelines but also positions researchers at the forefront of medical innovation, ultimately transforming patient care in the region.
Frequently Asked Questions
What is the primary law governing human research in Chile?
The primary law governing human research in Chile is Law No. 20,120, which establishes the ethical and scientific standards necessary for clinical evaluations, ensuring participant safety and data integrity.
What are the necessary endorsements required for conducting a FIH trial in Chile?
Necessary endorsements include acquiring approval from the Instituto de Salud Pública (ISP) by submitting a detailed study protocol that outlines the objectives, methodology, and ethical considerations of the investigation.
How long does the authorization process typically take for FIH trials in Chile?
The authorization process typically spans 30 to 60 days. Preparing documentation in advance can help mitigate delays and streamline the review process.
Why is it important to engage with local ethics committees when conducting clinical trials in Chile?
Engaging with accredited ethics committees is crucial to ensure that the study adheres to ethical standards, which is necessary for obtaining the required permissions and maintaining compliance throughout the study.
What should researchers be prepared for regarding compliance audits in Chile?
Researchers should be prepared for potential audits by the ISP or ethics committees and ensure that all study documentation is thorough and complies with ICH-GCP guidelines to maintain the integrity of the research.
How can subscribing to bioaccess®’s clinical insights service benefit researchers conducting FIH trials in Chile?
Subscribing to bioaccess®’s clinical insights service provides valuable updates on regulatory changes and market access strategies, enhancing researchers’ ability to leverage the region’s strategic advantages in speed and cost efficiency for their trials.
List of Sources
- Understand the Regulatory Landscape for FIH Trials in Chile
- Master Regulatory Compliance for Trials in Chi… | bioaccess® (https://bioaccessla.com/blog/master-regulatory-compliance-for-trials-in-chile-effectively)
- Country Report: Chile | PharmExec (https://pharmexec.com/view/country-report-chile)
- Select Sites and Recruit Patients Effectively
- First-in-Human Medical Device Trial in Chile | bioaccess® (https://bioaccessla.com/blog/first-in-human-medical-device-trial-chile)
- 2025 Trends In Patient Recruitment: From Disruption To Precision (https://clinicalleader.com/doc/trends-in-patient-recruitment-from-disruption-to-precision-0001)
- Clinical Trials Quotes (2 quotes) (https://goodreads.com/quotes/tag/clinical-trials)
- 10 Inspiring Patient Experience Quotes | Relias (https://relias.com/blog/patient-experience-quotes)
- Precision, Relevance and Prediction: The Future of Clinical Trial Recruitment (https://gcihealth.com/2026/01/22/precision-relevance-and-prediction-the-future-of-clinical-trial-recruitment)
- Execute the Trial: Manage Timelines and Monitor Progress
- 25+ useful clinical trial recruitment statistics for better results (https://antidote.me/blog/25-useful-clinical-trial-recruitment-statistics-for-better-results)
- 35 Best Project Management Quotes (https://projectmanager.com/blog/10-best-project-management-quotes)
- Enrollment and Retention: A Strategic Imperative for Clinical Trial Success – ConfidenceResearch (https://confidenceresearch.com/enrollment-and-retention-a-strategic-imperative-for-clinical-trial-success)
- The 62 Most Inspiring Project Management Quotes (https://inloox.com/company/blog/articles/the-62-most-inspiring-project-management-quotes)
- Manage Data and Prepare for Regulatory Submissions
- Paving the Path to Success Through Effective Clinical Trial Data Management (https://namsa.com/resources/blog/effective-clinical-trial-data-management)
- Electronic Data Capture (EDC) Systems Explained (https://ccrps.org/clinical-research-blog/electronic-data-capture-edc-systems)
- 8 key benefits of electronic data capture for clinical trials | Viedoc (https://viedoc.com/blog/key-benefits-electronic-data-capture-clinical-trials)
- The Ultimate Guide to Electronic Data Capture for Clinical Trials (https://quanticate.com/blog/electronic-data-capture-systems-in-clinical-trials)
- Data Management Plans: Implications for Automated Analyses | Data Science Journal (https://datascience.codata.org/articles/dsj-2023-002)