Introduction
Design control processes play a crucial role in medical device development, ensuring that products meet quality standards, regulatory requirements, and deliver safe and effective results. These processes involve a systematic approach to managing the design and development life cycle, mitigating risks, and upholding the highest quality. Real-world applications of design controls, such as technologies developed for critical situations like in Ukraine, demonstrate their importance.
Incorporating design controls early in the process can also influence cost-effectiveness and market competitiveness. Quality assurance in MedTech extends across the entire product lifecycle, emphasizing the importance of design control and process validation. The versatility of electronic height gages and the relevance of design controls in Agile methodologies are also noteworthy.
The pragmatic approach to engineering and quality assurance advocated by experts underscores the significance of design controls in medical device production. Overall, design control processes are essential for successful, compliant, and beneficial medical device manufacturing.
Understanding Design Control Processes
are crucial in , providing a framework to ensure that products not only meet quality standards but also comply with , and ultimately, deliver safe and effective results. A methodical approach to managing the , from conception to final production, these processes aim to mitigate risks and uphold the highest quality.
Practical applications of control systems are demonstrated by and deployed in critical situations, such as in Ukraine. One such innovation can adapt to any ventilator type and has been evaluated for field use by prestigious bodies like the US Army Aeromedical Research Laboratory.
Integrating controls early can also impact the cost-effectiveness and market competitiveness of a medical device. By taking into account the ultimate costs from the beginning, manufacturers can guarantee affordability and accessibility, which are crucial for widespread adoption. For instance, adopting a platform approach to oligonucleotide process development, as approved by the US FDA, can expedite process development, enabling quicker delivery of drugs to patients without sacrificing quality.
Ensuring high standards in MedTech is crucial, encompassing the entire lifecycle of the offering, including control of the development and validation of the procedure. It’s not just about compliance; it’s about delivering items that enhance patient outcomes. The significance of is emphasized by the modifications in regulatory documents for drug-device combinations, which outline the lifecycle management of these products based on real-world experiences.
In addition, the adaptability of electronic height gages supported by Mahr emphasizes the precision and efficiency that can be achieved through strong control measures. The incorporation of such tools in the planning stage can result in high-quality outcomes in .
The importance of control measures in the context of Agile methodologies is also worth mentioning. Inquiries regarding utilizing documentation from Application Lifecycle Management (ALM) tools to meet regulatory control requirements are answered affirmatively; with proper understanding and application, Agile can be compatible with regulatory compliance.
Kevin Becker’s quotes highlight a pragmatic approach to engineering and quality assurance, advocating for action over paralysis by analysis in development. This perspective is crucial in the ever-changing realm of medical equipment creation and emphasizes the significance of controlling the development process for prosperous, compliant, and advantageous medical tool manufacturing.
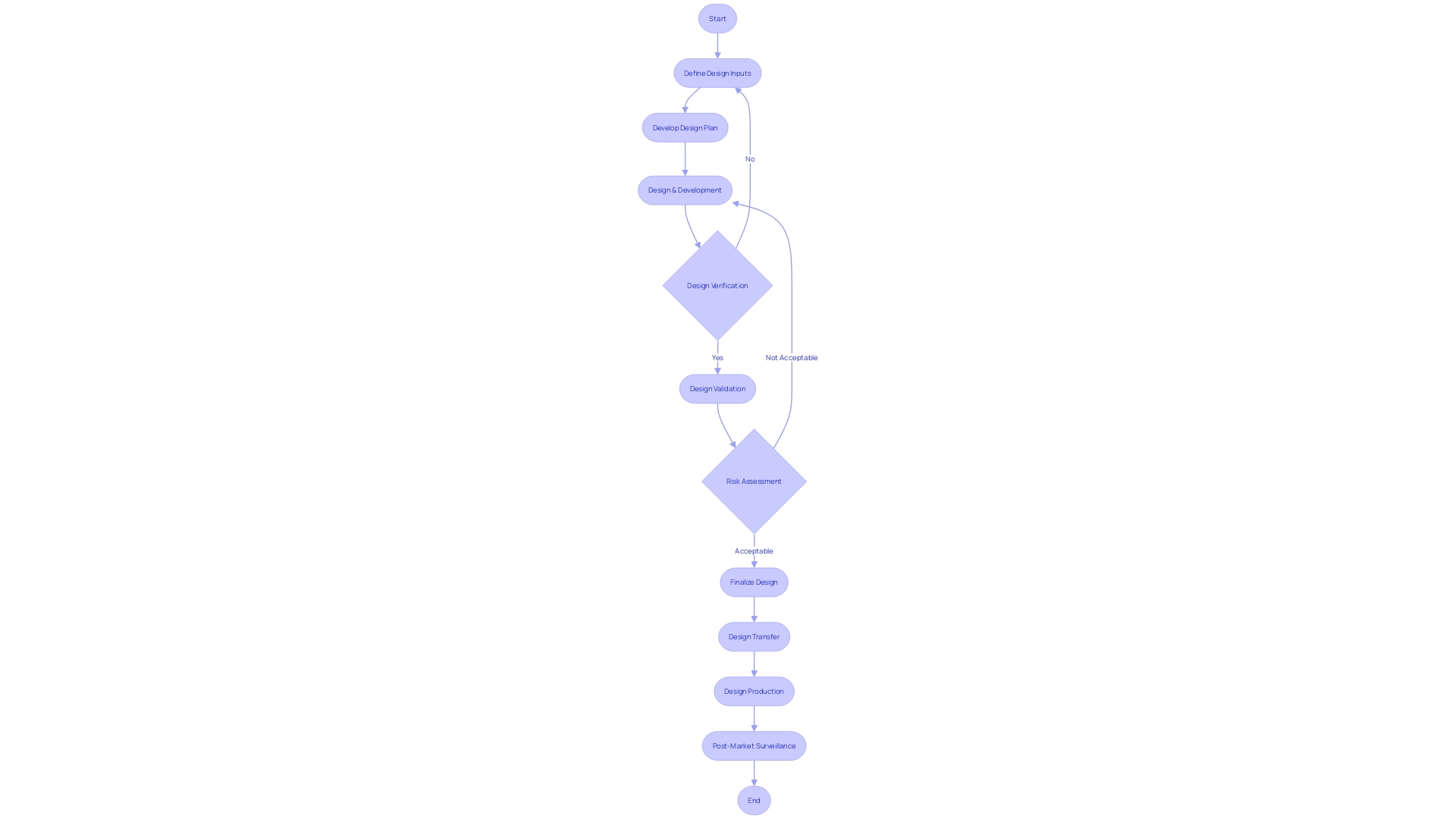
Key Components of Design Controls
development and manufacturing are regulated by comprehensive control processes that shape the foundation for creating products that meet user requirements and . These processes encompass a structured framework that includes identification of inputs, formulation of outputs, and a series of evaluations through reviews, verification, and validation activities. Furthermore, the transition from creation to production, referred to as transfer of the plan, is carefully controlled to guarantee uniformity and excellence. Following alterations to the structure are meticulously managed to uphold the integrity and adherence of the apparatus.
To ensure resilience in the face of the evolving healthcare landscape, adaptability within these components is paramount. This flexibility is demonstrated in the ongoing evaluation and incorporation of , as highlighted by industry professionals, to tackle the risk of inaccuracies in tools such as In Vitro Diagnostics (IVDs). The Global Podcast, powered by Greenlight Guru, underscores the importance of understanding the specific data and performance characteristics that regulatory bodies such as the FDA require for IVDs.
The industry is also witnessing a surge in novel regulatory considerations, particularly in the realm of integrated drug-device combinations and companion diagnostics. These regulations require comprehensive guidance encompassing the lifecycle management of these combinations, labeling requirements for co-packaged products, and consultations for products with ancillary medicinal substances. These changing regulatory environments emphasize the crucial importance of control measures in guaranteeing that medical products are not just efficient but also secure and in accordance with existing and upcoming standards.
In the context of cardiovascular diseases (CVD), the importance of strong control measures becomes even more evident. Progress in CVD management and treatment has been facilitated by meticulous control processes that guarantee the creation of groundbreaking diagnostics and therapeutic tools, which are revolutionizing patient outcomes. This progression is a testament to the significance of maintaining stringent s that align with the dynamic nature of healthcare technology and regulatory expectations.
Design Control Process Steps
Design control is an essential element of the development of devices, crucial for ensuring that items are safe and effective. This multifaceted process requires manufacturers to systematically evaluate their product from conception to market launch. As per the insights from UL Solutions, Michigan’s thriving medical technology sector underscores the importance of these controls in maintaining a competitive edge in the industry.
The begins with a , a cornerstone of any sound regulatory strategy. This involves meticulous research into FDA databases to determine the product code, potential submission pathways, and exemptions. Such assessments also guide interactions with the FDA, such as determining the need for Q-submissions and the relevant inquiries to make.
As highlighted by Medtronic’s Interstim system’s approval in 1997 and recent advances in Crohn’s disease and ulcerative colitis treatments, success in this space requires a clear understanding of regulatory requirements. Manufacturers must integrate effectively, setting clear objectives and realistic goals to align efforts. A phased implementation plan, building on the success of previous stages, ensures a solid foundation for future processes.
Furthermore, the field requires resilience and adaptability, characteristics that prosperous entrepreneurs in the exhibit. This resilience is crucial considering the challenges such as supply chain issues post-Brexit, impacting manufacturers of healthcare equipment and professionals alike, as stated in recent manifestos calling for the establishment of a National Stakeholder Forum.
, such as those for integral drug-device combinations and co-packaged products, provide procedural advice based on the collective experience and actual cases, ensuring products are effectively labeled and managed throughout their lifecycle. These extensive guidelines also encompass consultation procedures for instruments with accompanying therapeutic substances and companion diagnostics.
In the healthcare instrument sector, understanding is crucial. Staying informed through resources like the Global Medical Device Podcast can provide valuable insights into FDA expectations for IVDs, emphasizing the importance of accurate in regulatory submissions. This information not only informs manufacturers but also equips them to navigate the complexities of bringing innovative healthcare solutions to market.
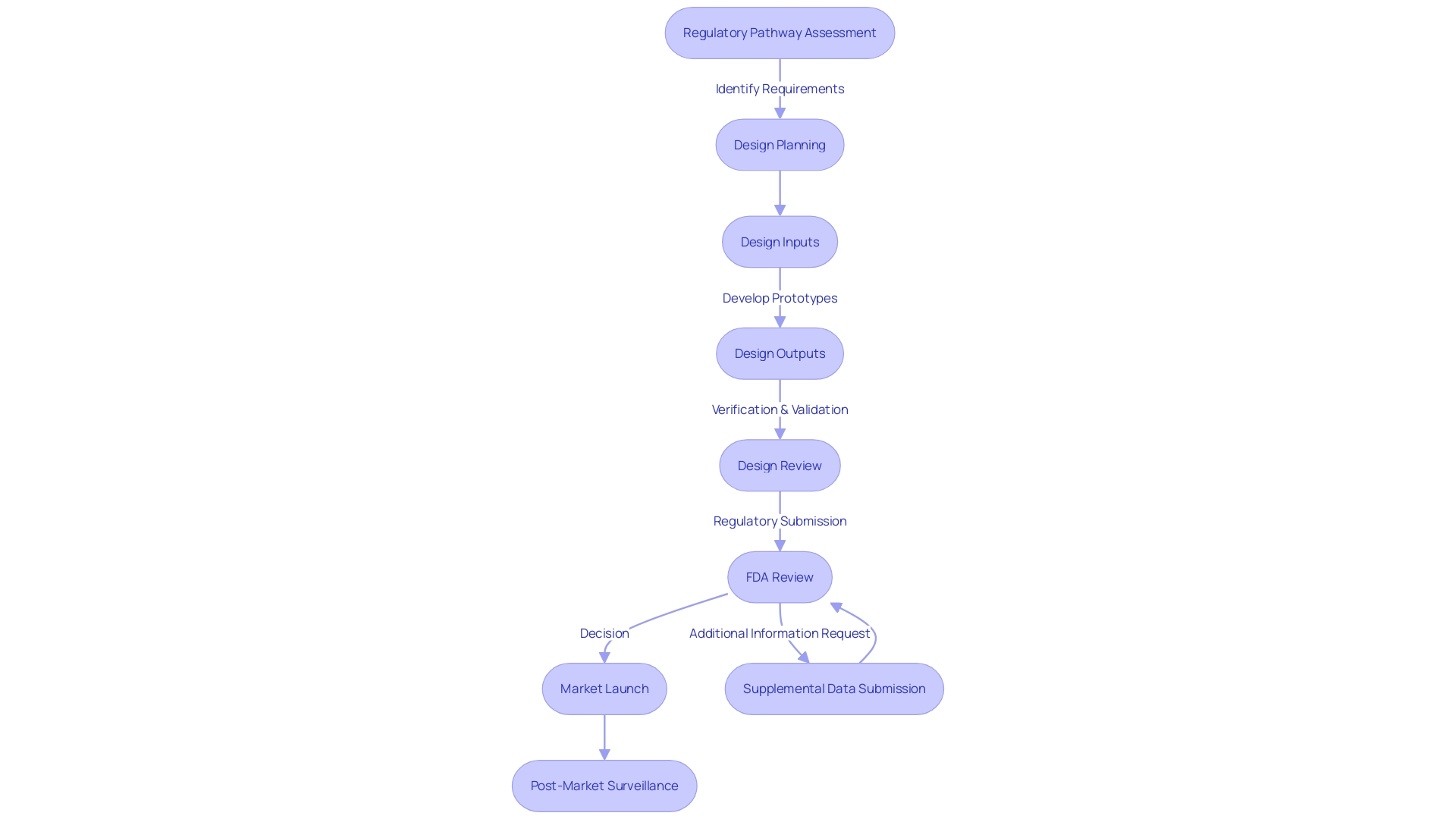
Design and Development Planning
As we begin the control process for , it’s crucial to comprehend that planning for development and creation is more than just the groundwork; it’s the blueprint for success. The process commences with a robust definition of the project’s scope, where clear objectives for design and development are set. A tactical plan is then formulated, outlining the necessary resources, delineating timelines, and specifying deliverables to achieve the set goals.
It’s crucial to recognize, as highlighted by Perry Parendo in his analysis of product development, that the focus on compliance and must be balanced with a keen awareness of project risk. Truly, it’s the synchronization of methodologies—whether waterfall, agile, or set-based design—that customizes the approach to the distinct requirements of . The wisdom shared by seasoned entrepreneurs in the emphasizes qualities such as resilience and adaptability as vital in navigating the intricate journey of bringing a medical instrument to the market.
Furthermore, the growing incorporation of wireless technologies in medical instruments calls for compliance with developing technical standards and regulatory prerequisites. National regulations for radio equipment, for example, must be meticulously complied with to ensure . Keeping abreast of market trends, such as the predicted growth of the corneal implant market to nearly $600 million by 2033, provides a backdrop for strategic planning.
In the field of , the accurate categorization of products and the choice of the suitable FDA registration pathway are fundamental. As industry veterans like Bijan Elahi articulate, the grasp of and the clarity it brings to the development process are non-negotiable. The convergence of application lifecycle management and control reflects a shared ethos of continual improvement and quality assurance.
In the end, the cost of healthcare equipment is affected by a multitude of elements, such as the intricacy of treatments and competitive environments. Insights obtained from market research shed light on the customer’s viewpoint, unveiling preferences and satisfaction levels with current products, which are crucial in informing excellent product development. So, as we explore the complexities of control measures, it is this versatile comprehension that will direct us toward developing healthcare instruments that are not only in accordance but also align with market requirements and patient care superiority.
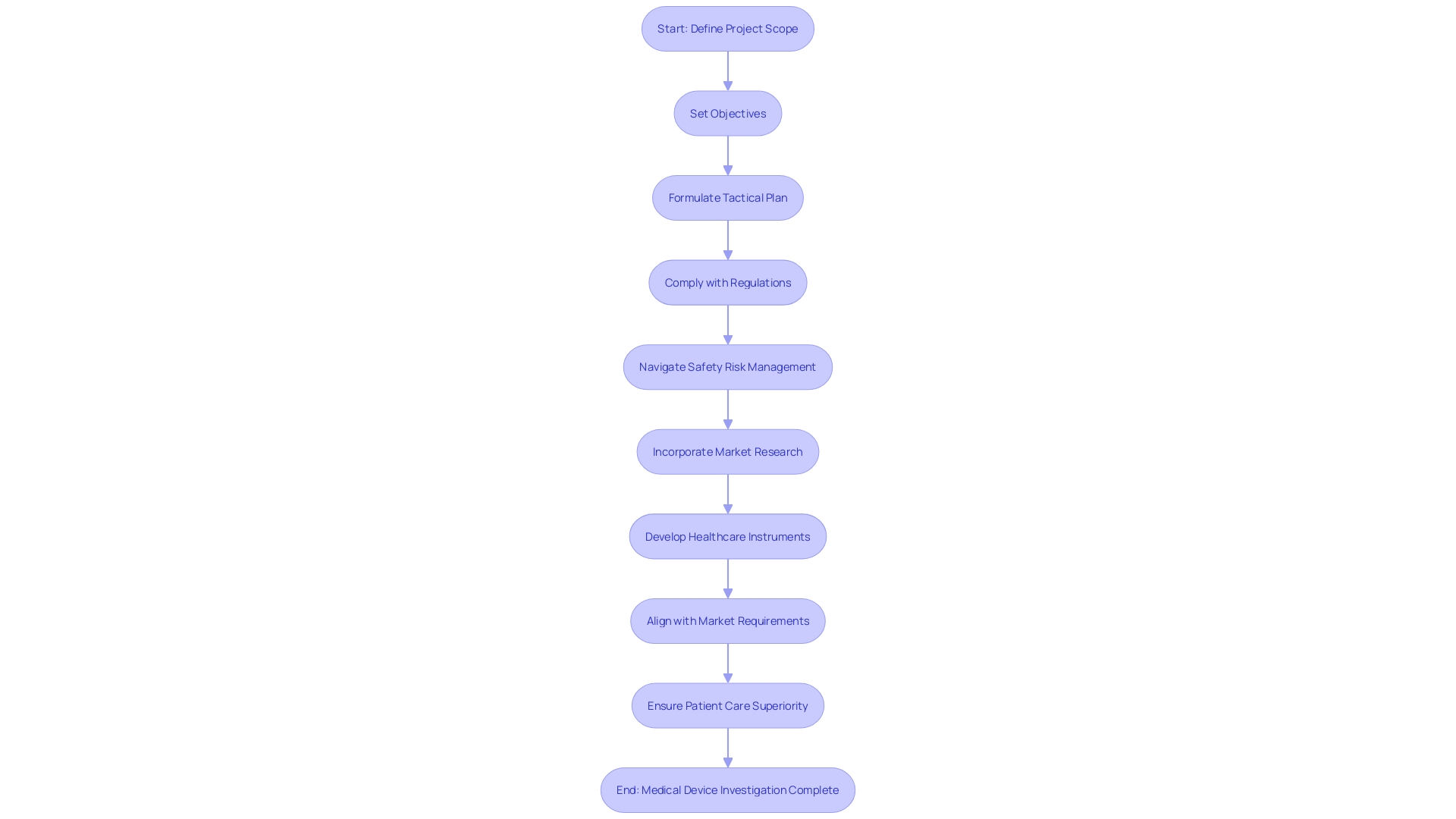
The creation of starts with a crucial stage: establishing the inputs for the blueprint. This procedure involves a systematic assessment of user requirements and specifications, which are based on a comprehensive comprehension of the intended usage of the equipment, the qualities of the user, and the necessary performance features. These inputs serve as the foundation for all subsequent control activities, directing the development process towards a successful result.
Historically, companies like Philips have demonstrated the significance of , which is rooted in a deep understanding of what customers genuinely need. This approach has been integral to their growth and continued success, fostering a culture of partnership and collaboration with clinical partners to drive innovation. Likewise, when creating healthcare instruments, integrating input from those involved at the beginning guarantees that the end outcome is in sync with the real requirements of the .
Furthermore, the recent changes in regulatory guidance for healthcare instruments, such as drug-device combinations and companion diagnostics, emphasize the significance of taking into account the complete lifespan of a product from the beginning. The complexity of regulations emphasizes the necessity for thorough that account for all aspects of an apparatus’s use, from integration with other systems to post-market upgrades and enhancements.
Furthermore, the market success of a medical product is not only determined by its technological innovation but also by its cost-effectiveness and accessibility. Designers must consider the ultimate costs from the start, which can impact the affordability, competitive positioning, and adoption rate of the object. Making sustainable choices not only supports the environment but also adds to the long-term sustainability of the item.
Embracing a philosophy that anticipates regular product changes is crucial, particularly for items that contain software or need to interface with other systems. Quality system procedures should be designed to accommodate updates and ensure continuous alignment with user needs. This approach to not only ensures but also and enhances the overall value of devices in the .
Design Outputs: Detailed Specifications and Documentation
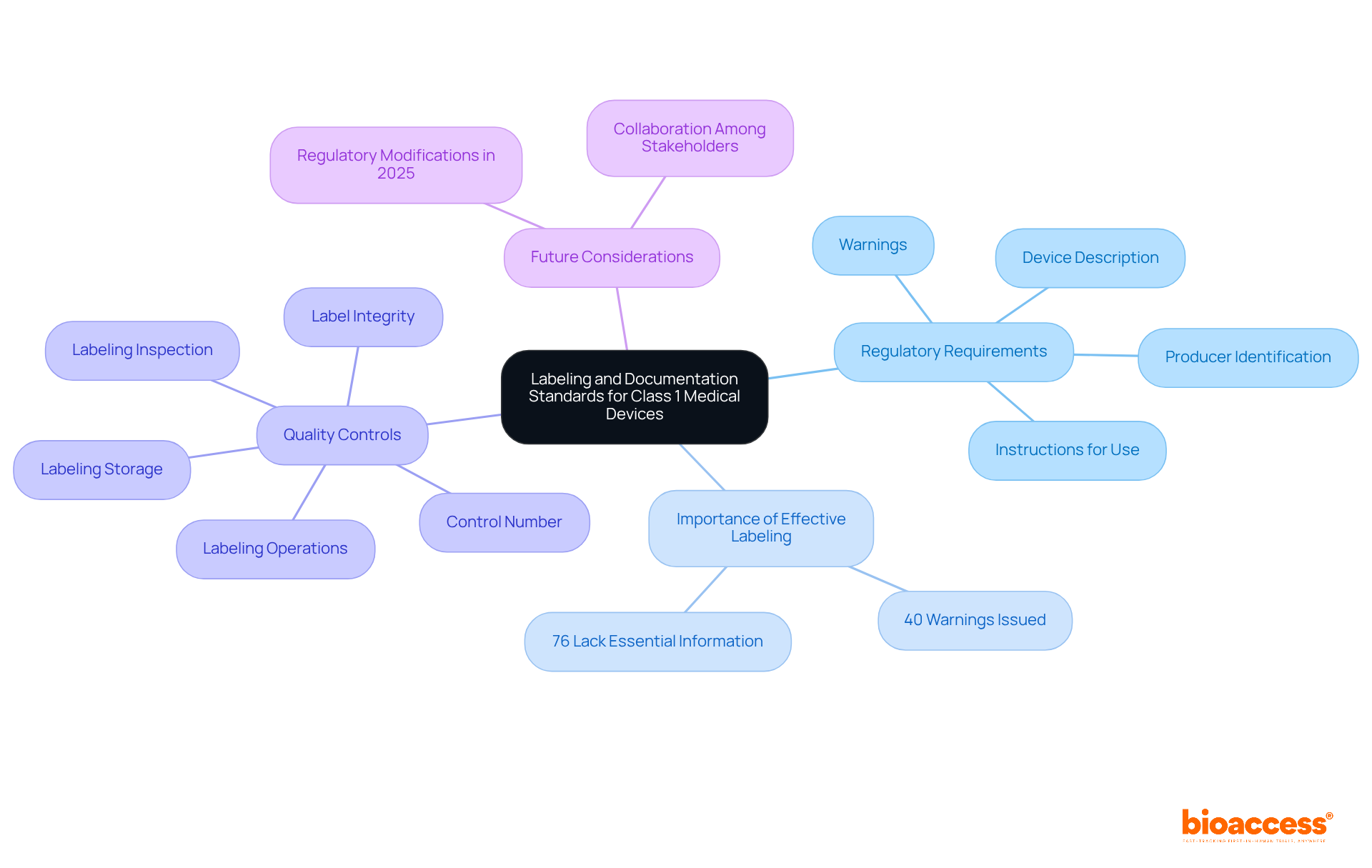
constitute the foundation of medical equipment development, including a comprehensive set of specifications and documentation that precisely outline the physical and functional characteristics of the product. This critical phase articulates the product’s attributes through comprehensive , precise schematics, robust software code, and specific packaging and labeling requirements. These outputs are crucial for not only guiding the manufacturing process but also for ensuring the equipment adheres to all necessary and performs reliably in .
In the field of healthcare equipment investigations, where compliance with strict regulations is an undeniable necessity, design outcomes serve as a concrete plan that connects the abstract design with the tangible outcome. They provide teams with an ultimate reference that enables a methodical approach to – crucial for preserving uniformity, dependability, and adherence throughout the lifecycle of the item, from beginning to post-market monitoring. As such, they are essential in the of drug-device combinations, co-packaged products, and companion diagnostics, ensuring every aspect of the device is examined and validated against the highest standards of device quality and safety.
Furthermore, in the rapidly changing area of , where the incorporation of advanced systems like artificial intelligence (AI) is becoming more and more common, the outputs must also include the intricacy of software components. As AI expands the capabilities of traditional automation by managing complex, non-fixed inputs and outputs, the documentation must reflect this intricacy, ensuring that every component is accounted for and potential vulnerabilities are preemptively addressed. Therefore, the outputs not only tell the tale of the intended functionality of the apparatus but also establish the foundation for a secure and efficient deployment within the healthcare ecosystem.
Design Reviews: Systematic Assessments and Compliance Checks
Control processes are crucial in the development and manufacturing of , ensuring that items meet both and the intended inputs. These processes encompass a series of rigorous evaluations, engaging cross-functional teams to meticulously examine each for potential issues and alignment with industry best practices.
By utilizing industry examples, we comprehend that the objective is to attain a shared language and set of expectations among all stakeholders—from the FDA to end-users—which simplifies proactive troubleshooting and improves performance reliability. This is akin to obtaining certifications like UL, which, contrary to popular belief, is not exclusive to large corporations but is also attainable by smaller organizations dedicated to quality and safety.
The recent regulatory revisions, informed by firsthand experiences and case studies, offer comprehensive guidance on managing lifecycles of items, particularly for drug-device combinations and co-packaged medical items. This includes detailed protocols for labelling, consultations for apparatus with ancillary medicinal substances, and companion diagnostics. These insights into are crucial for maintaining compliance throughout a product’s lifecycle.
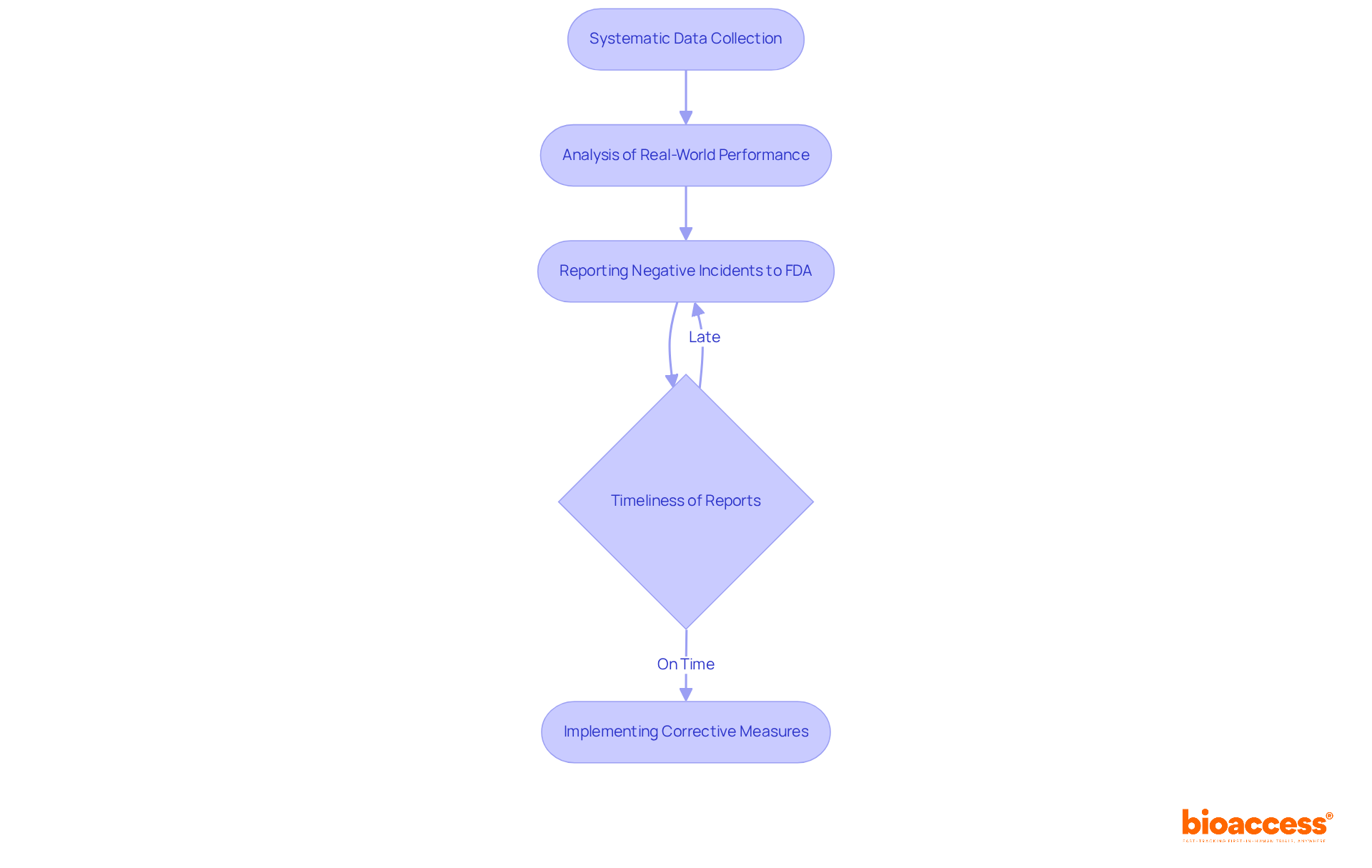
Data indicates that only a limited amount of AI/ML-enabled healthcare equipment have obtained approval, emphasizing the significance of comprehensive to evaluate the effectiveness and safety of these technologies. These trials must consider diverse patient demographics to ensure external validity and inform regulatory bodies, healthcare professionals, and the public accurately.
The healthcare device industry’s heartbeat is its commitment to innovation while navigating project and patient health risks. With a variety of design methodologies—from waterfall to agile—companies like Ketryx leverage to software development. This not only promotes a culture of rigor but also ensures improved documentation, risk management, and ultimately a compliant product that aligns with the high-quality standards required in the healthcare field.
In the context of global patent filings and grants, companies like SomaLogic lead in Healthtech and nanomedicine innovations, with a significant portion of their patents granted in Canada. This shows the significance of intellectual property in advancing technology and ensuring these advancements are protected and acknowledged within the sector.
Design Verification: Testing and Analysis to Confirm Design Outputs
is a critical step in ensuring medical instruments meet the necessary design outputs and function as intended. It includes a variety of tests, such as performance, safety, and reliability testing to confirm that the item complies with all specified requirements. For example, in the realm of Electromagnetic Compatibility (EMC), objects are exposed to thorough emissions (EMI) and immunity (EMS) testing. These tests ensure that products operate normally without causing or being affected by , a crucial factor as electronic products are frequently used in close proximity to each other. Moreover, the aspect of ” is often overlooked but holds immense value, particularly in software components of a device. It involves considering testability throughout the process, leading to improved engineering effectiveness, additional testing capabilities, and ultimately, time and cost savings during validation tasks.
The significance of comprehensive verification is further emphasized by the rapid technological advancements in industries like telecommunications, where EMC tests have become a global industry requirement. Additionally, embracing modern statistical approaches such as has been beneficial for healthcare companies in regulatory submissions, offering flexibility and valuable insights for decision-making in complex .
In the end, the process of confirming the correctness is not only about complying with but also about guaranteeing the of equipment used in healthcare, which is crucial for the well-being of patients and achieving success in the market. It is a testament to the industry’s commitment to excellence and continuous improvement in the face of evolving technological landscapes and regulatory frameworks.
Design Validation: Ensuring Devices Meet User Needs and Intended Use
At the heart of development for healthcare instruments, is the crucial process that verifies a tool meets the requirements of users and is suitable for its intended purpose. This procedure examines the performance of the equipment in its actual service setting, guaranteeing that it functions properly under anticipated circumstances. It is a comprehensive approach, often encompassing to ascertain therapeutic effectiveness, user studies to verify ease of use and acceptance, and real-world testing to validate consistent performance outside of controlled environments. For instance, in a recent deployment, a new medical technology initially created for the US Marine Corps was subjected to rigorous real-world testing, including field use by the US Army Aeromedical Research Laboratory and the Air Force Medical Evaluation Support Activity, to confirm its adaptability and operation with various ventilator types.
The significance of integrating validation of the blueprint throughout the complete life cycle of the equipment is emphasized by the belief that ‘ anticipate regular changes in the merchandise.’ This is particularly pertinent for devices containing software where upgrades are anticipated, or for devices that must interface with other evolving systems. A strategic approach to means keeping the user at the center of the design process. Drafting a is crucial, prompting developers to continually ask who the users are, what the intended uses of the product are, and in which environments it will be deployed.
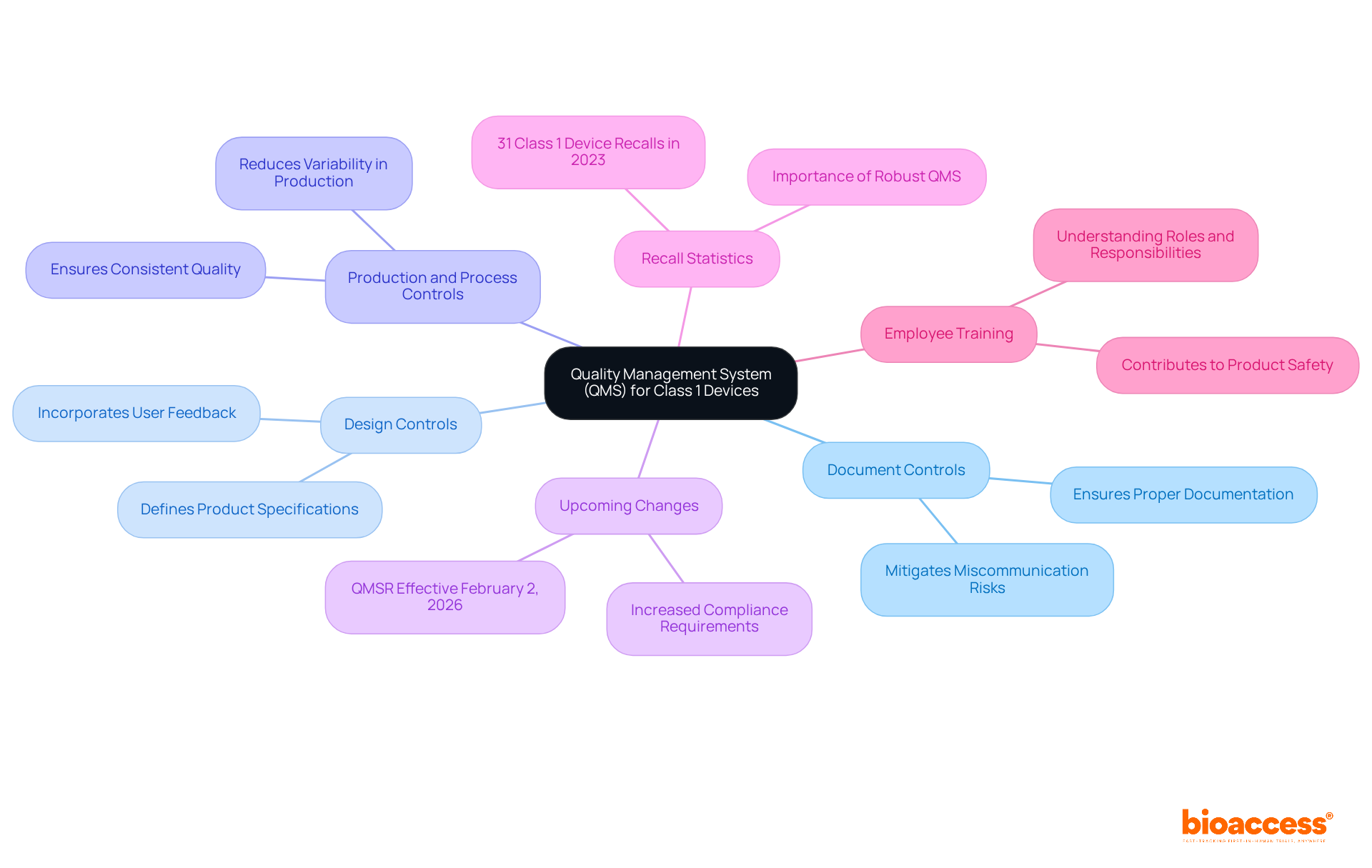
in medical product development does not solely focus on meeting regulatory requirements; it’s about ensuring that products consistently deliver safe and effective outcomes. This multifaceted approach includes , ensuring manufacturing consistency, and , addressing potential risks in the product’s structure. The stakes of are high, as any shortfall could lead to catastrophic outcomes. In the MedTech sector, companies encounter the difficulties of strict regulations, fast technological advancements, intricate global supply chains, and fierce market competition, all of which emphasize the necessity for thorough validation to uphold throughout the product’s lifespan.
Design Transfer: Transitioning from Development to Production
is more than just a handoff from the development team to manufacturing; it’s a crucial stage in that ensures the device can be produced with the same rigor and precision as it was designed. This procedure includes carefully moving all the documentation, specifications, and manufacturing processes. It’s a phase where the focus shifts from the ‘shiny’ aspects of creation — the sleek engineering and attractive aesthetics that make an item stand out — to the less glamorous, yet vitally important ”. This idea centers on into the process from the beginning, guaranteeing that every element is reachable and testable, which is not just vital for but also for the effectiveness of the engineering team and the performance of the end product.
Recent advancements in medical technology underscore the importance of this phase. As an example, the launch of the Vscan Air SL by GE Healthcare is a testament to successful , where the equipment not only meets the expectations but also delivers clear cardiac and vascular images in a clinical setting. Such innovations highlight the smooth transition from concept to clinic, which is the aim of transfer. It’s a strategic process that requires clear objectives, realistic goals, and a phased implementation plan to ensure every aspect of the product’s development is ready for production and meets the standards that patients and healthcare providers expect.
Design Changes: Managing and Documenting Post-Production Modifications
Changes to a ‘s blueprint after manufacturing are a crucial part of its . These can stem from various sources, including user feedback, evolving , and technological advancements. It is crucial that such changes are managed with a meticulous approach to uphold the safety and efficacy of the equipment. As emphasized by the most recent , the incorporation of healthcare instruments with medicines, such as pre-filled syringes, requires a thorough comprehension of both pharmaceutical and equipment frameworks. The underscores the importance of maintaining detailed documentation for integral drug-device combinations and their lifecycle. In the context of wireless , compliance with national regulatory wireless requirements is non-negotiable, given their dual nature as both medical and radio instruments. These scenarios illustrate the continuous requirement for strong control processes that anticipate regular updates, especially in the realm of capital equipment where upgrades are frequent. By engaging in meticulous preparation and following the seven crucial components of control over the creation process, which encompass recorded outcomes, developers can guarantee that their products not only satisfy initial design prerequisites but also adjust effectively to subsequent enhancements or alterations in regulations.
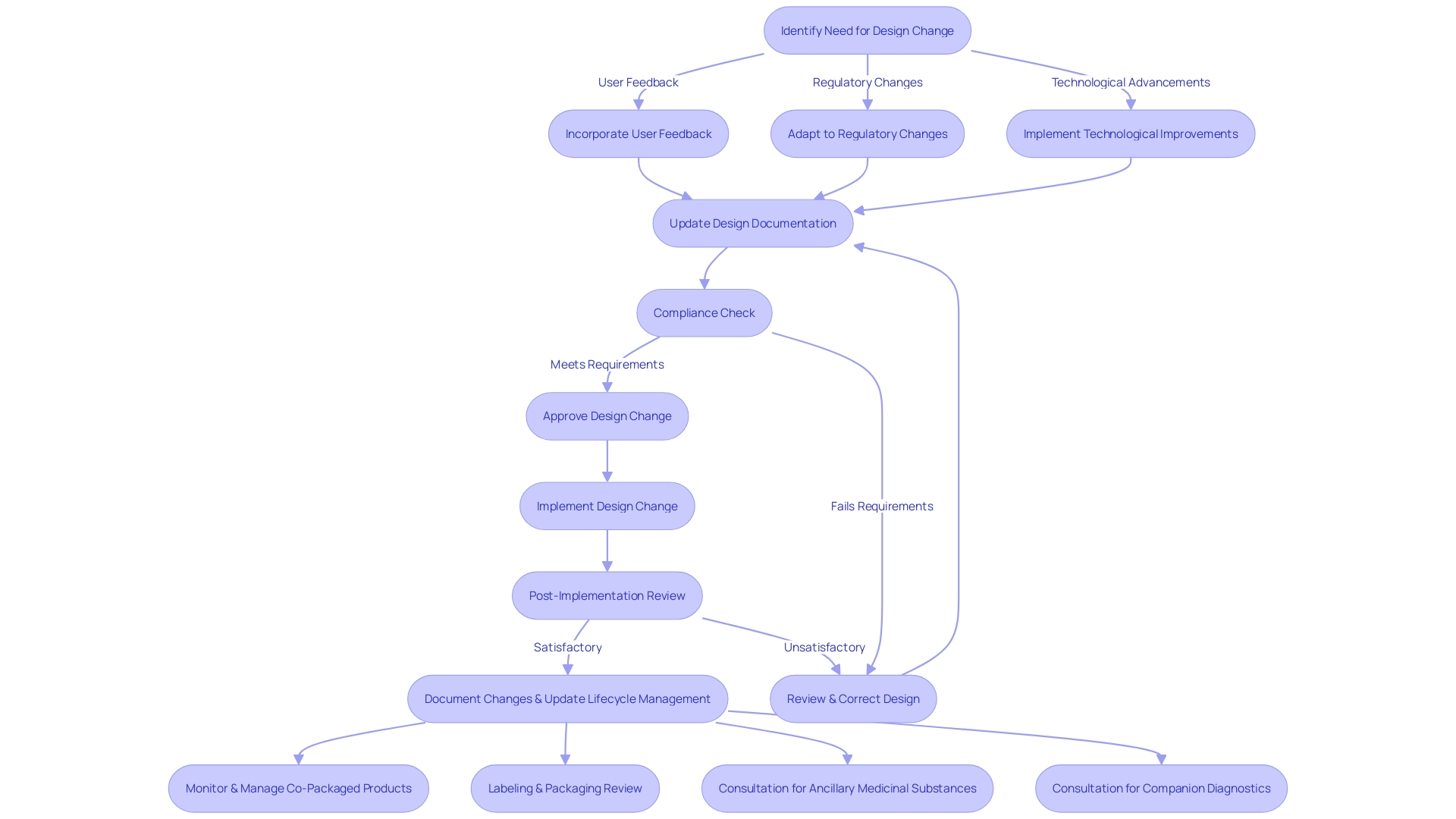
Integrating Risk Management into Design Controls
The combination of and control measures is a pivotal strategy in the , particularly as it pertains to mitigating potential risks throughout the product’s lifespan. A thorough strategy for necessitates manufacturers to proactively recognize, evaluate, and handle risks linked to the product’s structure, production, and operational utilization. This integrated process is not only about safeguarding patient safety but also about adhering to the . As an example, the FDA’s focus on integrating into every stage of the software lifecycle highlights the crucial importance of . This implies that from the , testing, monitoring, and maintenance, manufacturers must demonstrate through meticulous documentation how risks are being managed, responding to the complexity of modern threats, and ensuring the product’s security throughout its total . As healthcare equipment become more and more interconnected and dependent on software, the capability to anticipate and navigate potential cyber threats through systematic becomes indispensable. The sector’s shift towards all-encompassing hazard management frameworks demonstrates a wider dedication to the safety, efficiency, and security of healthcare equipment in a time where both automation and AI play substantial roles in equipment functionality and patient care.
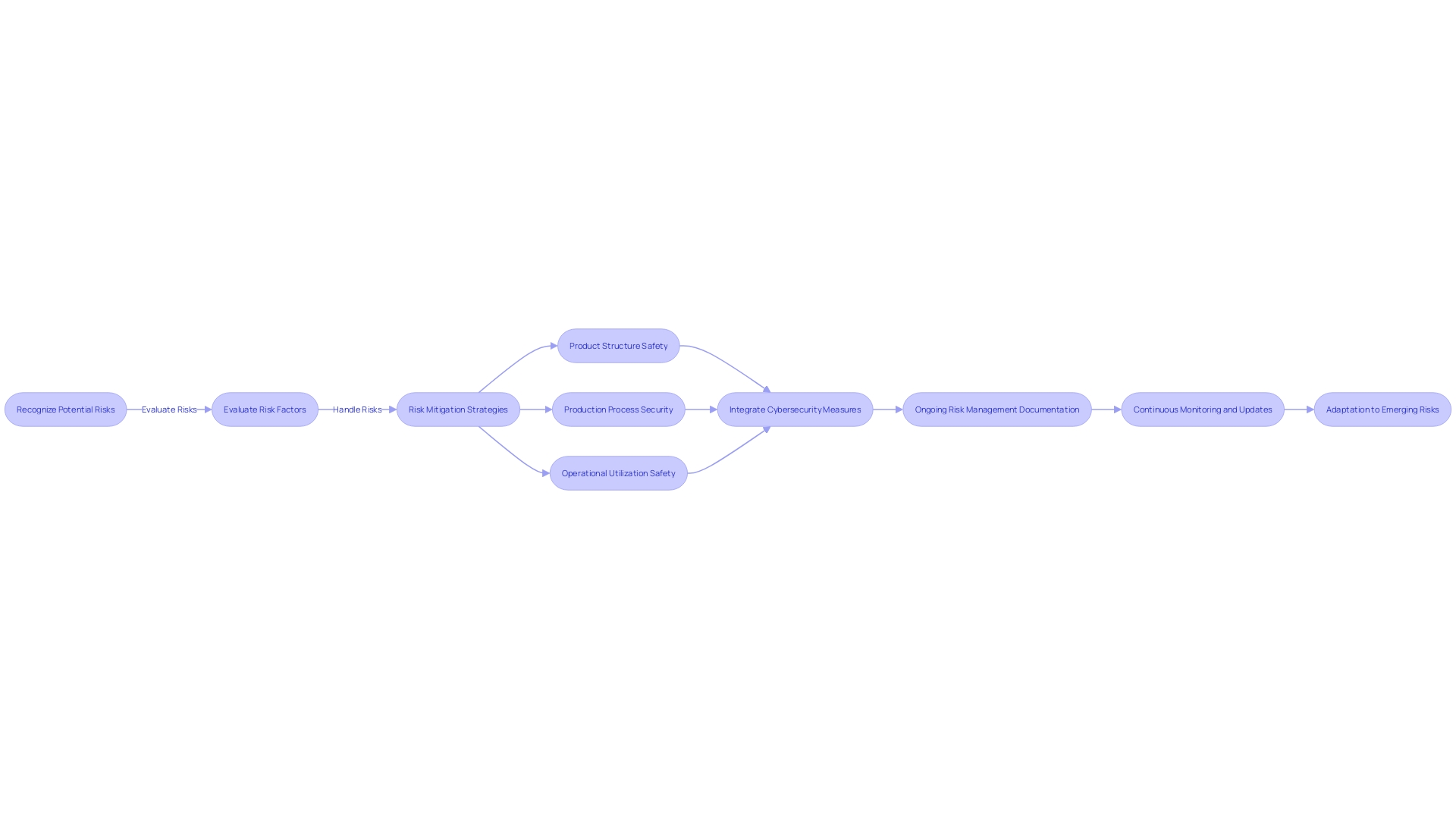
Compliance with FDA 21 CFR 820 and ISO 13485:2016
Ensuring that medical equipment meets strict safety and effectiveness standards is a fundamental aspect of , with requirements such as the FDA’s 21 CFR 820 and ISO 13485:2016 at the forefront. These standards require thorough controls over the structure, covering risk management, documentation, traceability, and a strong . A crucial update by the FDA has further emphasized the integration of cybersecurity into the entirety of a product’s lifecycle, highlighting the need for secure software practices from initial development to post-market maintenance. This includes not just the ‘shiny’ aspects of an object, such as advanced technical features or aesthetic appeal, but also the foundational elements like , which although less glamorous, are vital for regulatory approval.
Furthermore, the notion of is becoming more and more significant in a world where electronic devices are omnipresent. EMC refers to the capability of a piece of equipment to operate properly in its electromagnetic surroundings without emitting levels of electromagnetic interference that could impact other devices. Regulatory bodies, recognizing the growing complexity of telecommunication products and their susceptibility to electromagnetic disturbance, require thorough EMC testing to prevent interference issues.
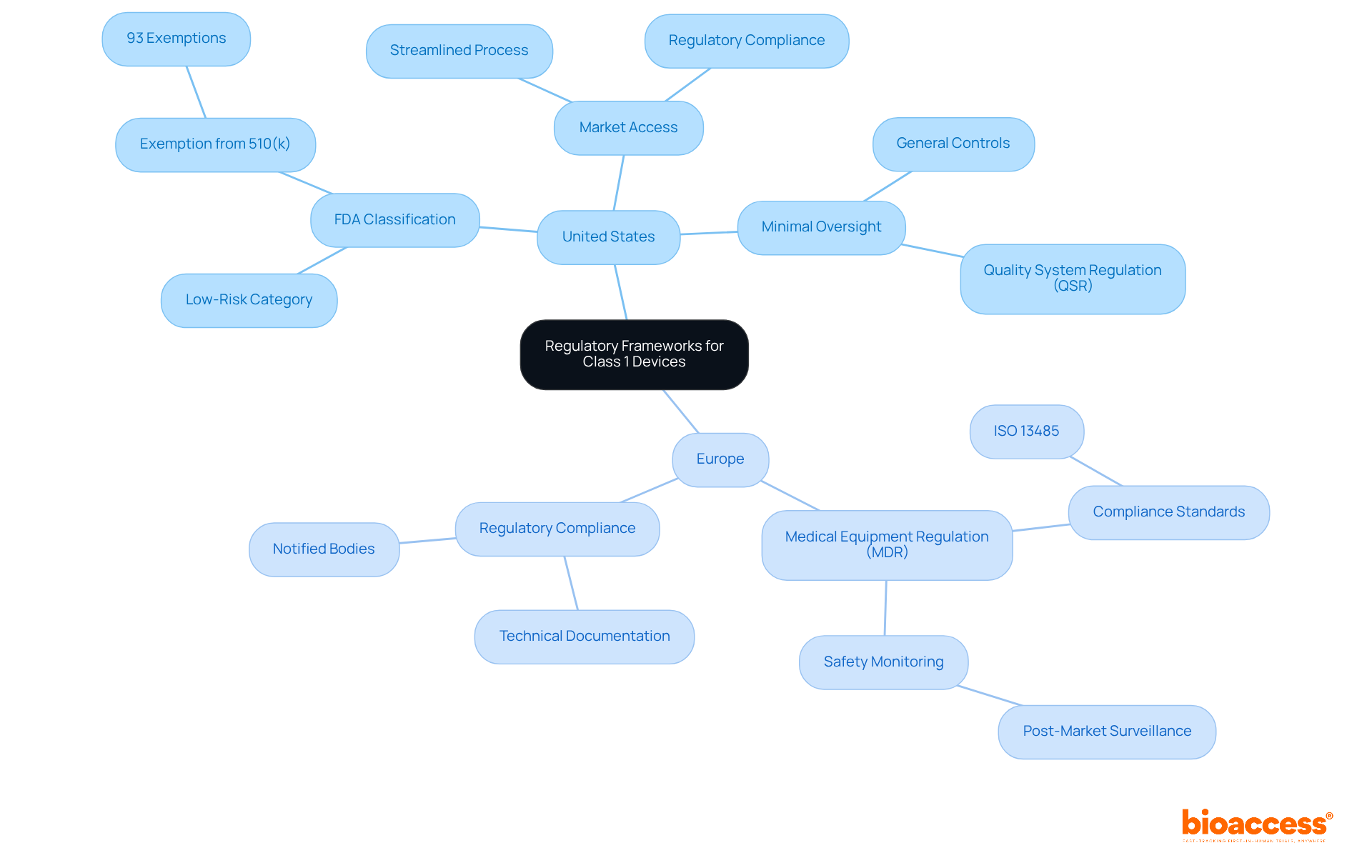
In the realm of , every step from design to market is thoroughly examined. The into three categories based on patient risk, with each category requiring a specific regulatory pathway—Premarket Notification (510(k)), Premarket Approval (PMA), or De Novo process—to legally market an item in the U.S. Understanding the significance of establishment registration and item listing with the FDA is crucial for professionals navigating the approval or clearance process.
This comprehensive approach to control and lifecycle management ensures that not only meet current regulatory standards but are also prepared for future challenges, safeguarding both public health and a manufacturer’s ability to innovate and compete in the global market.
Best Practices for Implementing Design Controls
For , the implementation of controls is not just a regulatory requirement; it is a strategic approach to ensure product quality and safety. To optimize this process, it is essential to create a encompassing various disciplines. This team should work cohesively, with clearly delineated roles and responsibilities, to foster a culture of accountability and transparency. Thorough documentation is essential, recording every decision and activity within the to uphold a strong and traceable development history.
Engaging in open communication with all stakeholders, including regulatory authorities and end-users, is crucial. This conversation ensures that the control process remains aligned with the and regulatory expectations, as highlighted by the Association for the Advancement of Medical Instrumentation (AAMI), which sets the consensus standards for the field.
Undertaking at every phase of the development process is essential. As Bijan Elahi, an expert with extensive experience in safety for medical devices, suggests, this is imperative to minimize potential hazards associated with the use of the device. His work emphasizes that is an ongoing process that demands continuous education and vigilance.
Furthermore, the efficiency of controls for the creation process must be constantly monitored and assessed. This proactive approach is similar to how companies such as Xbox, in their dedication to environmental responsibility, have seamlessly incorporated energy efficiency enhancements into their offerings, without compromising the user experience. Likewise, manufacturers of healthcare equipment can incorporate control measures in a way that boosts the advancement of goods without hindering creativity.
Staying abreast of the latest is also crucial. For instance, the FDA’s recent final rule on direct-to-consumer prescription drug advertisements underscores the importance of clear, conspicuous, and neutral presentation of information. This principle is equally applicable to the medical device sector, where clarity and compliance in communication can significantly impact the success and safety of the medical device.
As the industry evolves, so too must the approach to control of creation. The dynamic nature of regulations and the introduction of novel product combinations, such as drug-device integrals, necessitate an adaptive and informed strategy. It is through such diligence and dedication to best practices that manufacturers can achieve excellence in design control processes, ensuring the delivery of safe, effective, and high-quality medical devices.
Conclusion
In conclusion, design control processes are essential in medical device development, ensuring products meet quality standards, comply with regulations, and deliver safe and effective results. These processes involve a systematic approach to managing the design and development life cycle, mitigating risks, and upholding the highest quality.
Real-world applications of design controls, like technologies developed for critical situations, demonstrate their importance. Incorporating design controls early can also influence cost-effectiveness and market competitiveness. Quality assurance in MedTech extends across the entire product lifecycle, emphasizing the significance of design control and process validation.
The versatility of electronic height gages and the relevance of design controls in Agile methodologies are noteworthy. The integration of electronic height gages in the design phase can lead to high-quality outcomes in medical device manufacturing. Design controls can also be compatible with Agile methodologies when used properly, meeting regulatory compliance.
Experts advocate for a pragmatic approach to engineering and quality assurance, highlighting the significance of design controls in medical device production. Design control processes are crucial for successful, compliant, and beneficial medical device manufacturing.
To ensure success, it is important to have a cross-functional team, rigorous documentation, and open communication with stakeholders. Risk assessments should be conducted at each stage, and the effectiveness of design controls should be continually monitored. Staying informed about the latest regulatory requirements and adapting to the evolving industry landscape are also key.
By implementing best practices, manufacturers can achieve excellence in design control processes, delivering safe, effective, and high-quality medical devices. The commitment to design controls is essential for regulatory compliance, patient safety, and competitiveness in the market.
Implement best practices in design control processes for safe, effective, and high-quality medical devices.
Frequently Asked Questions
What are design control processes?
Design control processes are systematic frameworks used in medical device development to ensure that products meet quality standards and comply with regulatory requirements, ultimately ensuring their safety and effectiveness.
Why are design control processes important?
These processes help manage the entire development life cycle of a medical device, mitigating risks and maintaining high quality throughout the stages of conception, production, and post-market monitoring.
How do design control processes affect cost and market competitiveness?
Integrating controls early in the development process can lead to better cost management and affordability, making medical devices more accessible and competitive in the market.
What role does regulatory compliance play in design control?
Regulatory compliance is crucial; design control processes ensure adherence to regulations like the FDA’s standards, which are necessary for market approval and maintaining product safety.
What is the significance of quality assurance in MedTech?
Quality assurance ensures that medical devices not only comply with regulations but also enhance patient outcomes, thus highlighting the importance of continuous monitoring and control throughout the device’s lifecycle.
How do Agile methodologies fit into design control processes?
Agile methodologies can be compatible with regulatory requirements when implemented with a clear understanding of documentation and controls, allowing for flexibility in development while ensuring compliance.
What are the key components of design control processes?
Key components include regulatory pathway assessments, risk management strategies, detailed documentation, and continuous evaluation of product performance against user needs and regulatory standards.
How are design verification and validation conducted?
Design verification involves testing to ensure that the product meets specified requirements, while validation assesses whether the device functions effectively in real-world conditions and meets the needs of users.
What challenges do manufacturers face in maintaining design controls?
Challenges include evolving regulations, technological advancements, and the need for continuous adaptation of processes to ensure both safety and compliance in a competitive market.
What is the importance of risk management in the design control process?
Risk management is vital for identifying and mitigating potential hazards throughout the product’s lifecycle, ensuring not just compliance but also the safety and reliability of medical devices.
How do manufacturers ensure effective communication and collaboration in design controls?
By forming cross-functional teams with clearly defined roles, engaging stakeholders, and maintaining thorough documentation, manufacturers can foster a culture of accountability and transparency in the design control process.
What is the future outlook for design control processes in medical technology?
As regulations evolve and new technologies emerge, manufacturers must adopt adaptive strategies to ensure that their design control processes remain effective, compliant, and aligned with user needs and market trends.
List of Sources
- Understanding Design Control Processes
- newsweek.com (https://newsweek.com/quality-assurance-medtech-safeguarding-excellence-innovation-1861147)
- medtechintelligence.com (https://medtechintelligence.com/news_article/medical-devices-new-guidance-for-industry-and-notified-bodies)
- greenlight.guru (https://greenlight.guru/blog/alm-and-design-controls)
- greenlight.guru (https://greenlight.guru/blog/quality-myths-lessons-learned-part-ii)
- medicaldevice-network.com (https://medicaldevice-network.com/news/hksh-medical-proton-therapy-hong-kong)
- medicaldevice-network.com (https://medicaldevice-network.com/news/philips-launch-ai-imaging-and-reporting-tools-to-support-prostate-cancer-care)
- linkedin.com (https://linkedin.com/posts/amdr_medical-device-reprocessing-design-tips-from-activity-7123639561527693312-1jf9)
- qualitymag.com (https://qualitymag.com)
- medicaldevice-network.com (https://medicaldevice-network.com/news/thornhill-anaesthesia-production-boost)
- pubs.acs.org (https://pubs.acs.org/doi/10.1021/acs.oprd.3c00303)
- greenlight.guru (https://greenlight.guru/blog/ai-automation-risk-medtech)
- med-technews.com (https://med-technews.com/medtech-insights/digital-in-healthcare-insights/tapping-into-new-potential-realising-the-value-of-data-in-he)
- Key Components of Design Controls
- medicaldevice-network.com (https://medicaldevice-network.com/sponsored/digital-deals-in-life-sciences-podcast-corporate-culture)
- digikey.com (https://digikey.com/en/blog/smarter-motion-control-for-smart-manufacturing?article_name=smarter_motion_control_fo&utm_campaign=blog&utm_content=digikey&utm_medium=social&utm_source=digikey)
- medtechintelligence.com (https://medtechintelligence.com/feature_article/ul-certification-a-launchpad-for-product-development-excellence)
- starfishmedical.com (https://starfishmedical.com/blog/medical-device-entrepreneur-traits)
- greenlight.guru (https://greenlight.guru/blog/considerations-for-ivd)
- medicaldevice-network.com (https://medicaldevice-network.com/sponsored/the-innovations-and-trends-offering-hope-in-the-cardiovascular-market)
- hbiostat.org (https://hbiostat.org/proj/covid19/statdesign#fundamental-elements-of-outcome-assessments)
- medtechintelligence.com (https://medtechintelligence.com/news_article/medical-devices-new-guidance-for-industry-and-notified-bodies)
- Design Control Process Steps
- medicaldevice-network.com (https://medicaldevice-network.com/news/bhta-manifesto-calls-for-greater-collaboration-from-uk-government)
- greenlight.guru (https://greenlight.guru/blog/considerations-for-ivd)
- starfishmedical.com (https://starfishmedical.com/blog/medical-device-entrepreneur-traits)
- medtechintelligence.com (https://medtechintelligence.com/news_article/medical-devices-new-guidance-for-industry-and-notified-bodies)
- medicaldevice-network.com (https://medicaldevice-network.com/news/ul-medical-testing-us)
- nejm.org (https://nejm.org/doi/full/10.1056/NEJMimc2306268)
- medicaldevice-network.com (https://medicaldevice-network.com/news/boomerang-hits-milestone-in-pilot-study-of-neurostimulator-implant-for-ibd)
- medicaldevice-network.com (https://medicaldevice-network.com/news/artivion-presents-double-win-at-aats-for-aortic-devices)
- raps.org (https://raps.org/News-and-Articles/News-Articles/2023/10/regulatory-pathway-assessment-interview?utm_campaign=online_u_pathways_series&utm_medium=social)
- medtechintelligence.com (https://medtechintelligence.com/ate/harnessing-the-potential-of-digital-quality-transforming-manufacturing-outcomes-for-long-term-success)
- appsilon.com (https://appsilon.com/r-vs-sas-pharma-life-sciences?utm_source=social&utm_medium=twitter&utm_campaign=sas-r)
- insights.axtria.com (https://insights.axtria.com/industry-primers/data-governance)
- Design and Development Planning
- greenlight.guru (https://greenlight.guru/blog/project-management-in-medtech)
- medtechintelligence.com (https://medtechintelligence.com/feature_article/designing-compliant-wireless-medical-devices)
- starfishmedical.com (https://starfishmedical.com/blog/medical-device-entrepreneur-traits)
- medicaldevice-network.com (https://medicaldevice-network.com/news/pantheon-meets-with-fda-to-discuss-pma-submission-for-corneal-implants)
- medicaldevice-network.com (https://medicaldevice-network.com/news/telus-health-medical-alert-system)
- rimsys.io (https://rimsys.io/blog/fda-listed-cleared-approved-granted)
- medtechsafety.com (https://medtechsafety.com)
- medicaldevice-network.com (https://medicaldevice-network.com/data-insights/medical-devices-reef-micro-pricing)
- researchgate.net (https://researchgate.net/publication/222289497_Predevelopment_Activities_Determine_New_Product_Success)
- linkedin.com (https://linkedin.com/posts/ajosephprofile_fda-medicaldevices-designcontrols-activity-7168268183764717569-MPAa?utm_source=share&utm_medium=member_desktop&lipi=urn:li:page:d_flagship3_pulse_read%3BDwZ7TdkYSeyNcVSmwyMcHA%3D%3D)
- greenlight.guru (https://greenlight.guru/blog/alm-and-design-controls)
- starfishmedical.com (https://starfishmedical.com/blog/medical-device-commercialization-vision)
- medtechintelligence.com (https://medtechintelligence.com/news_article/medical-devices-new-guidance-for-industry-and-notified-bodies)
- Design Inputs: Identifying User Needs and Product Requirements
- philips.com (https://philips.com/a-w/about/innovation.html)
- linkedin.com (https://linkedin.com/posts/ajosephprofile_fda-medicaldevices-designcontrols-activity-7168268183764717569-MPAa?utm_source=share&utm_medium=member_desktop&lipi=urn:li:page:d_flagship3_pulse_read%3BDwZ7TdkYSeyNcVSmwyMcHA%3D%3D)
- linkedin.com (https://linkedin.com/posts/amdr_medical-device-reprocessing-design-tips-from-activity-7123639561527693312-1jf9)
- cdc.gov (https://cdc.gov/mmwr/volumes/73/su/su7303a1.htm?s_cid=su7303a1_w)
- medtechintelligence.com (https://medtechintelligence.com/news_article/medical-devices-new-guidance-for-industry-and-notified-bodies)
- Design Outputs: Detailed Specifications and Documentation
- cdc.gov (https://cdc.gov/mmwr/volumes/73/su/su7303a1.htm?s_cid=su7303a1_w)
- medtechintelligence.com (https://medtechintelligence.com/feature_article/510k-submission-planning)
- starfishmedical.com (https://starfishmedical.com/blog/sbom-analysis-and-value)
- starfishmedical.com (https://starfishmedical.com/blog/medical-device-entrepreneur-traits)
- greenlight.guru (https://greenlight.guru/blog/ai-automation-risk-medtech)
- medtechintelligence.com (https://medtechintelligence.com/news_article/medical-devices-new-guidance-for-industry-and-notified-bodies)
- newsweek.com (https://newsweek.com/quality-assurance-medtech-safeguarding-excellence-innovation-1861147)
- Design Reviews: Systematic Assessments and Compliance Checks
- greenlight.guru (https://greenlight.guru/blog/project-management-in-medtech)
- fda.gov (https://fda.gov/drugs/information-consumers-and-patients-drugs/overview-our-role-regulating-and-approving-drugs-video-series)
- greenlight.guru (https://greenlight.guru/blog/considerations-for-ivd)
- medtechintelligence.com (https://medtechintelligence.com/news_article/medical-devices-new-guidance-for-industry-and-notified-bodies)
- medtechintelligence.com (https://medtechintelligence.com/feature_article/ul-certification-a-launchpad-for-product-development-excellence)
- ketryx.com (https://ketryx.com/blog/announcing-ketryxs-ul-certification)
- fda.gov (https://fda.gov/drugs/regulatory-science-action/adaptive-trial-design-testing-bioequivalence-generics-highly-variable-drugs)
- ai.nejm.org (https://ai.nejm.org/doi/full/10.1056/AIpc2300038)
- medicaldevice-network.com (https://medicaldevice-network.com/uncategorized/somalogic-patent-activity)
- Design Verification: Testing and Analysis to Confirm Design Outputs
- starfishmedical.com (https://starfishmedical.com/blog/medical-device-commercialization-vision)
- rimsys.io (https://rimsys.io/blog/fda-listed-cleared-approved-granted)
- medtechsafety.com (https://medtechsafety.com)
- tuv.com (https://tuv.com/india/en/lp/tec.html)
- starfishmedical.com (https://starfishmedical.com/blog/medical-device-design-for-testability)
- medicaldevice-network.com (https://medicaldevice-network.com/news/rishi-sunak-addresses-existential-ai-threats-ahead-of-uk-safety-summit)
- arxiv.org (https://arxiv.org/abs/2311.16506)
- Design Validation: Ensuring Devices Meet User Needs and Intended Use
- newsweek.com (https://newsweek.com/quality-assurance-medtech-safeguarding-excellence-innovation-1861147)
- greenlight.guru (https://greenlight.guru/blog/reducing-medical-device-risk-with-usability-testing)
- medicaldevice-network.com (https://medicaldevice-network.com/news/thornhill-anaesthesia-production-boost)
- medicaldevice-network.com (https://medicaldevice-network.com/news/dalhousie-professor-mri-grant)
- statista.com (https://statista.com/chart/31247/smart-home-adoption-in-the-united-states)
- greenlight.guru (https://greenlight.guru/blog/human-centered-design-medical-devices)
- linkedin.com (https://linkedin.com/posts/ajosephprofile_fda-medicaldevices-designcontrols-activity-7168268183764717569-MPAa?utm_source=share&utm_medium=member_desktop&lipi=urn:li:page:d_flagship3_pulse_read%3BDwZ7TdkYSeyNcVSmwyMcHA%3D%3D)
- hardianhealth.com (https://hardianhealth.com/insights/early-economic-modelling-of-ai-as-medical-device-answering-the-why)
- Design Transfer: Transitioning from Development to Production
- medicaldevice-network.com (https://medicaldevice-network.com/news/baird-class-iii-needle)
- medicaldevice-network.com (https://medicaldevice-network.com/news/ge-healthcare-and-airstrip-sign-us-distribution-agreement)
- medcitynews.com (https://medcitynews.com/2023/12/medical-device-fda-healthcare)
- medtechintelligence.com (https://medtechintelligence.com/ate/harnessing-the-potential-of-digital-quality-transforming-manufacturing-outcomes-for-long-term-success)
- starfishmedical.com (https://starfishmedical.com/blog/medical-device-design-for-testability)
- medicaldevice-network.com (https://medicaldevice-network.com/uncategorized/establishment-labs-patent-activity)
- medicaldevice-network.com (https://medicaldevice-network.com/news/recor-medical-paradise-urdn-system-us)
- hardianhealth.com (https://hardianhealth.com/insights/health-institution-exemptions-for-samd-in-great-britain)
- starfishmedical.com (https://starfishmedical.com/blog/medical-device-entrepreneur-traits)
- starfishmedical.com (https://starfishmedical.com/blog/commercializing-medical-devices-with-optics)
- Design Changes: Managing and Documenting Post-Production Modifications
- medtechintelligence.com (https://medtechintelligence.com/news_article/medical-devices-new-guidance-for-industry-and-notified-bodies)
- linkedin.com (https://linkedin.com/posts/ajosephprofile_fda-medicaldevices-designcontrols-activity-7168268183764717569-MPAa?utm_source=share&utm_medium=member_desktop&lipi=urn:li:page:d_flagship3_pulse_read%3BDwZ7TdkYSeyNcVSmwyMcHA%3D%3D)
- blog.shockwavemedical.com (https://blog.shockwavemedical.com/case-spotlight-ivl-first-calcium-modification-for-female-patients)
- medtechintelligence.com (https://medtechintelligence.com/feature_article/designing-compliant-wireless-medical-devices)
- megit.com (https://megit.com/case-study-llr-pcl-quality-auditing-and-incident-reporting?utm_source=twitter&utm_medium=post&utm_campaign=llrpcl-case-study&utm_content=20240501–cta-link)
- medtechintelligence.com (https://medtechintelligence.com/news_article/medical-devices-new-guidance-for-industry-and-notified-bodies)
- Integrating Risk Management into Design Controls
- greenlight.guru (https://greenlight.guru/blog/ai-automation-risk-medtech)
- starfishmedical.com (https://starfishmedical.com/blog/fda-guidance-medical-device-cybersecurity)
- medicaldevice-network.com (https://medicaldevice-network.com/features/medical-device-network-review-2023)
- iaea.org (https://iaea.org/newscenter/news/how-are-universal-ionizing-radiation-symbols-used-around-the-world)
- greenlight.guru (https://greenlight.guru/blog/project-management-in-medtech)
- IoT Devices Are a Leading Vulnerability in Healthcare Data Breaches | IoT For All (https://iotforall.com/iot-devices-vulnerability-healthcare-data-breaches)
- medtechintelligence.com (https://medtechintelligence.com/ate/harnessing-the-potential-of-digital-quality-transforming-manufacturing-outcomes-for-long-term-success)
- Compliance with FDA 21 CFR 820 and ISO 13485:2016
- raps.org (https://raps.org/news-and-articles/news-articles/2024/2/fda-warns-industry-about-potential-data-integrity?utm_campaign=Regulatory-Focus&utm_source=twitter&utm_medium=social)
- fda.gov (https://fda.gov/news-events/press-announcements/fda-roundup-february-16-2024)
- tuv.com (https://tuv.com/india/en/lp/tec.html)
- fda.gov (https://fda.gov/drugs/information-consumers-and-patients-drugs/overview-our-role-regulating-and-approving-drugs-video-series)
- medtechintelligence.com (https://medtechintelligence.com/news_article/medical-devices-new-guidance-for-industry-and-notified-bodies)
- rimsys.io (https://rimsys.io/blog/fda-listed-cleared-approved-granted)
- starfishmedical.com (https://starfishmedical.com/blog/medical-device-design-for-testability)
- starfishmedical.com (https://starfishmedical.com/blog/fda-guidance-medical-device-cybersecurity)
- Best Practices for Implementing Design Controls
- learn.microsoft.com (https://learn.microsoft.com/en-us/gaming/sustainability/case-studies/case-studies-halo)
- dev.to (https://dev.to/pedrohnq/unraveling-the-strategy-pattern-a-practical-case-study-349g)
- medtechintelligence.com (https://medtechintelligence.com/news_article/medical-devices-new-guidance-for-industry-and-notified-bodies)
- pressroom.aami.org (https://pressroom.aami.org/posts/pressreleases/fda-gives-full-recognition-to-aami-cybersecur)
- fda.gov (https://fda.gov/news-events/press-announcements/fda-roundup-november-21-2023)
- medtechsafety.com (https://medtechsafety.com)
- thoughtworks.com (https://thoughtworks.com/what-we-do/software-engineering/engineering-effectiveness/empowering-engineering-effectiveness-commissioned-study)
- fivesgroup.com (https://fivesgroup.com)




![Relationships and Concepts in [Company Name]'s Operations Relationships and Concepts in [Company Name]'s Operations](https://vectorly-files.s3.eu-central-1.amazonaws.com/7e5170f3-eeed-4ae0-a415-e37c583adb20.jpg)








. Each box represents a key action, and the decision point shows whether self-certification or Notified Body involvement is required based on the type of device. Follow the arrows to see the steps needed for CE marking. Each box represents a key action, and the decision point shows whether self-certification or Notified Body involvement is required based on the type of device.](https://images.tely.ai/telyai/jmpvayoq-follow-the-arrows-to-see-the-steps-needed-for-ce-marking-each-box-represents-a-key-action-and-the-decision-point-shows-whether-self-certification-or-notified-body-involvement-is-required-based-on-the-type-of-device.webp)