Introduction
In the rapidly evolving landscape of clinical trials, Electronic Data Capture (EDC) systems have emerged as indispensable tools, revolutionizing the way data is collected, managed, and utilized. With the ability to integrate traditional and digital data sources, modern EDC systems handle a staggering volume of data points, often reaching into the billions, especially in trials utilizing connected devices. This article delves into the multifaceted benefits of EDC systems, highlighting their role in enhancing data accuracy, real-time monitoring, and regulatory compliance.
It also explores key features that make these systems vital, such as user-friendly interfaces, robust security protocols, and advanced reporting tools. Furthermore, a comparative analysis of top EDC systems illustrates their unique strengths and suitability for various clinical trial needs. By providing a comprehensive overview of these advanced technologies, the article aims to underscore their critical impact on the efficiency, quality, and success of clinical research.
Benefits of EDC Systems in Clinical Trials
Electronic Information Capture (EDC) systems transform information collection and management in by combining both traditional and digital sources. Contemporary experiments usually employ five to ten information sources, with some extending to twenty, producing around 8.1 million information points per study. For experiments involving linked devices such as continuous glucose monitors, information points can skyrocket into the billions, greatly exceeding the quantities seen ten years earlier.
The main benefits of involve improved information precision by minimizing human mistakes, immediate information access for efficient oversight, and simplified adherence to . These systems also enable quicker and information reporting, accelerating the overall study process and ensuring more prompt results. By implementing a comprehensive , which includes optimal information collection and efficient information flow, sponsors and can effectively manage the influx of information and extract meaningful insights.
As SDC COO Faith Kolb indicates, the platform’s real-time reminders, support, and feedback through personal devices guarantee data quality and dependable results. This not only aids researchers and participants but also enhances studies for success. By investing in advanced EDC technologies, medical research can achieve a more streamlined regulatory submission process, ultimately driving the next generation of research solutions.
Key Features of EDC Systems
The most efficient EDC frameworks combine various crucial attributes aimed at enhancing . These systems feature user-friendly interfaces and customizable input forms, making the process easier for all participants involved. Strong security measures guarantee that confidential information stays safeguarded, an essential requirement considering the growing number of information sources in research studies, which can vary from five to ten, and occasionally up to twenty distinct sources, collecting millions of information points.
Equally important is the , which improves the effectiveness of information gathering and examination. For instance, contemporary EDC systems such as SDC Capture BYOD offer real-time notifications, assistance, and input via personal devices, ensuring information precision and dependable study results. This feature is valuable for both researchers and participants, optimizing trial success.
Moreover, are vital for maintaining data integrity and transparency, a requirement underscored by regulations such as the EU MDR Article 62 which mandates the protection of participant rights and the generation of scientifically valid and robust data. Support for multiple languages and regulatory standards further ensures that these frameworks can be utilized across diverse clinical environments, promoting global compliance and versatility.
Additionally, the use of within EDC systems automates the presentation of necessary information to patients, reducing the risk of non-compliance and legal issues. ‘Integration of Electronic Clinical Outcome Assessment (eCOA) solutions improves information quality by allowing participants to directly input their patient-reported outcomes, reducing errors from manual transcription.’.
Together, these features not only simplify research processes but also aid in the creation of that is vital for regulatory submissions and market entry, ultimately enhancing the progress of medical research and better patient outcomes.
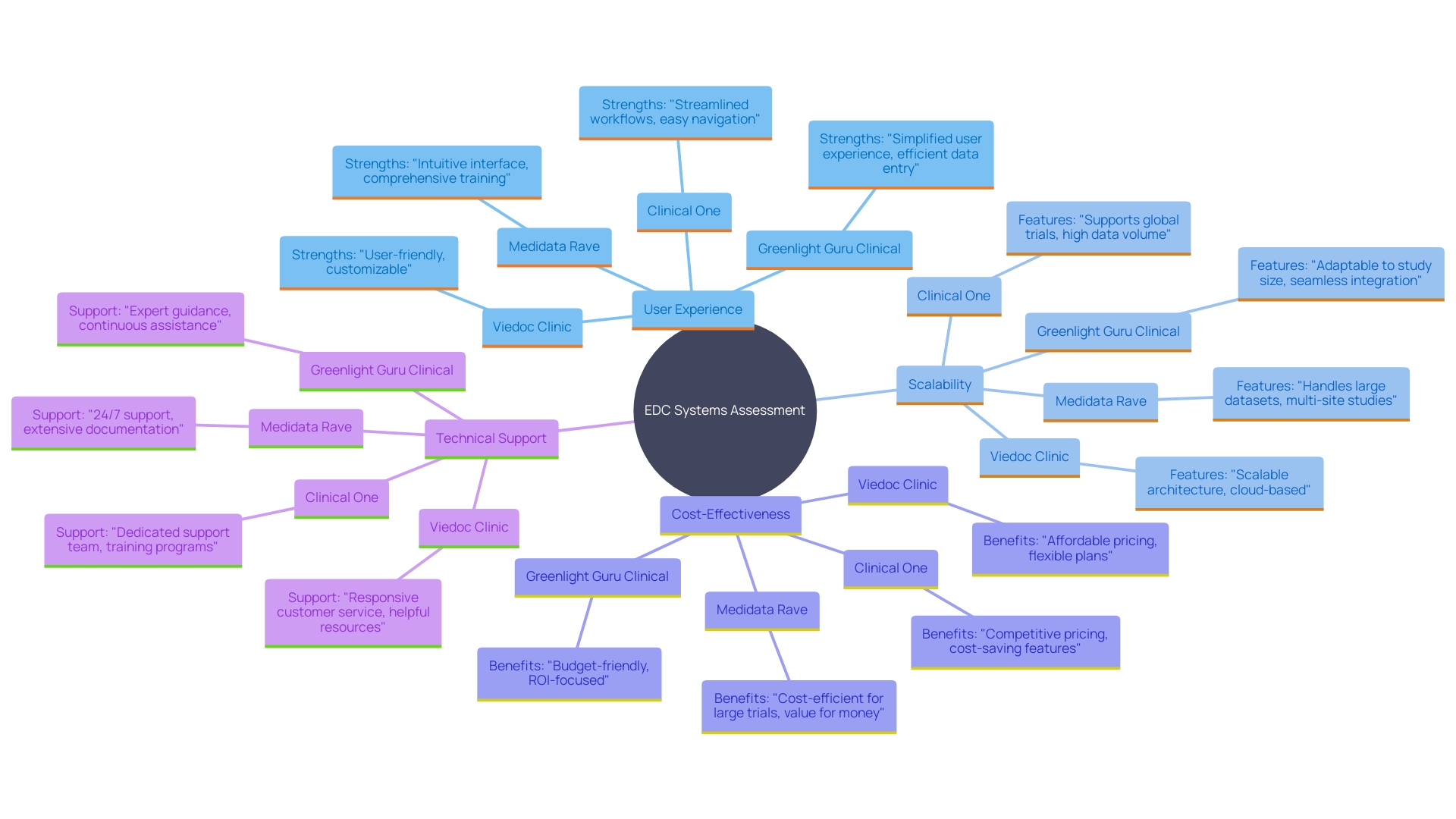
Comparative Analysis of Top EDC Systems
When assessing the best for medical studies, several crucial factors must be taken into account: , scalability, cost-effectiveness, and technical support. Medidata Rave excels with its comprehensive features and user-friendly design, as highlighted by its pending showcase at the NEXT New York clinical research conference. Viedoc Clinic is also lauded for its advanced features that simplify study management. On the other hand, Clinical One stands out by offering a unified platform that simplifies the integration of multiple trial components, addressing the need for a seamless from diverse sources.
In the realm of , solutions like Greenlight Guru Clinical are noteworthy for their intuitive user interfaces, which facilitate quick onboarding without requiring prior technical skills. ‘This ease of use is crucial for collaborating effectively with medical teams at study sites, thereby reducing the risk of non-compliance and ensuring a more streamlined pathway to regulatory submissions.’.
Considering that contemporary medical studies can encompass as many as 20 distinct information sources and produce millions of information points, a strong EDC system is crucial. For instance, Medidata’s Rave Lite, scheduled for release in early 2025, promises to help manage this influx of information efficiently. As progressively incorporate linked devices and real-time information, it becomes essential to have a thorough information management approach. This strategy should ensure optimal data collection and efficient to maintain high data quality and facilitate meaningful insights.
Ultimately, each EDC platform offers unique strengths, making it essential to evaluate them based on the specific needs of your trial. Whether it’s the extensive support and operational efficiency provided by Medidata or the intuitive use of Greenlight Guru Clinical, the right EDC solution can significantly enhance the success of your clinical research.
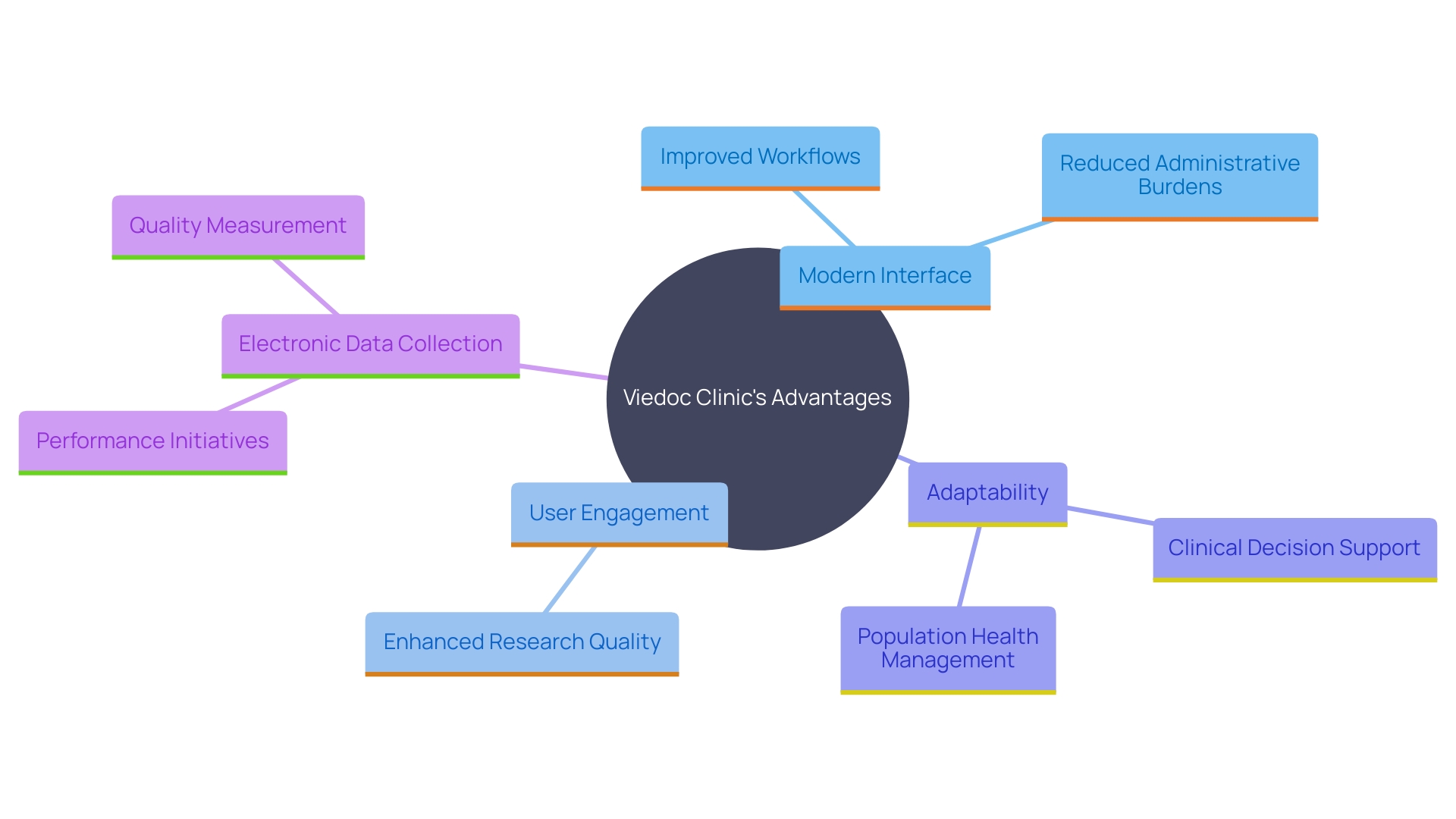
Viedoc Clinic: Modern and Streamlined Interface
Viedoc Clinic stands out for its modern, intuitive interface which significantly enhances and . The framework’s design facilitates easy navigation, providing a seamless experience for both investigators and site staff. Its adaptability to various study designs and rapid deployment capabilities make it a preferred choice for many . As per the 2023 Study Activation Survey, more than half of the research locations now employ electronic data collection (EDC) tools, along with other technologies, to handle growing study volumes more efficiently. This widespread adoption underscores the importance of like Viedoc Clinic to and reduce administrative burdens. As highlighted by specialists, utilizing not only speeds up timelines but also boosts adherence and openness, ultimately improving the overall quality of the research process.
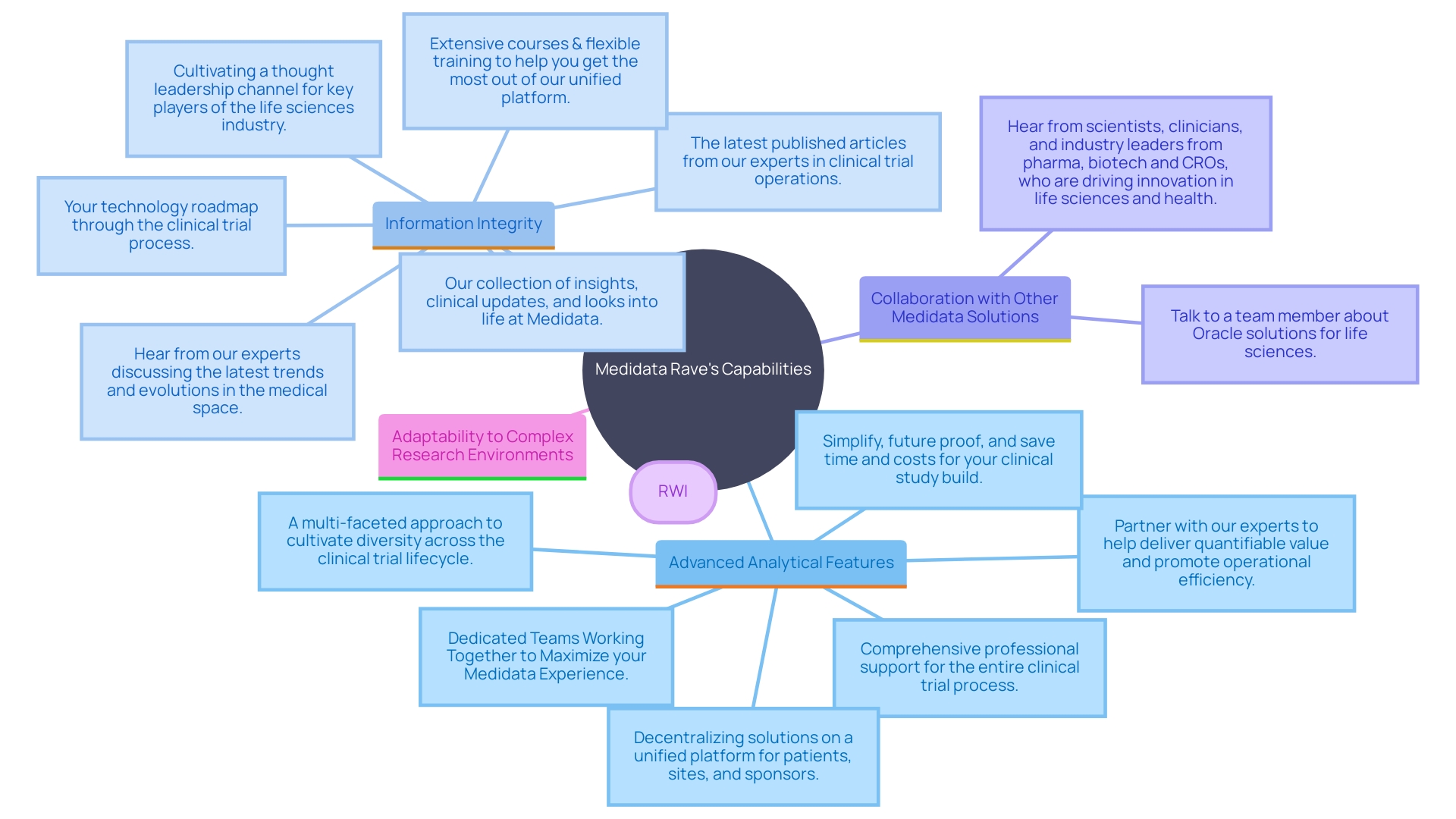
Medidata Rave: Advanced, Robust, and Secure
Medidata Rave excels in its capacity to manage and complex information sets with efficiency and precision. This platform’s advanced and strong protective measures guarantee thorough information integrity and adherence throughout the . Collaboration with other Medidata solutions provides a cohesive method for managing , simplifying procedures and improving operational effectiveness. The use of (RWI), gathered from actual patient experiences, further enriches the platform’s analytical capabilities, driving advancements in understanding disease and health states. As the research study environment becomes more intricate, Medidata Rave stays at the leading edge, offering the resources required to navigate and thrive in this data-heavy setting.
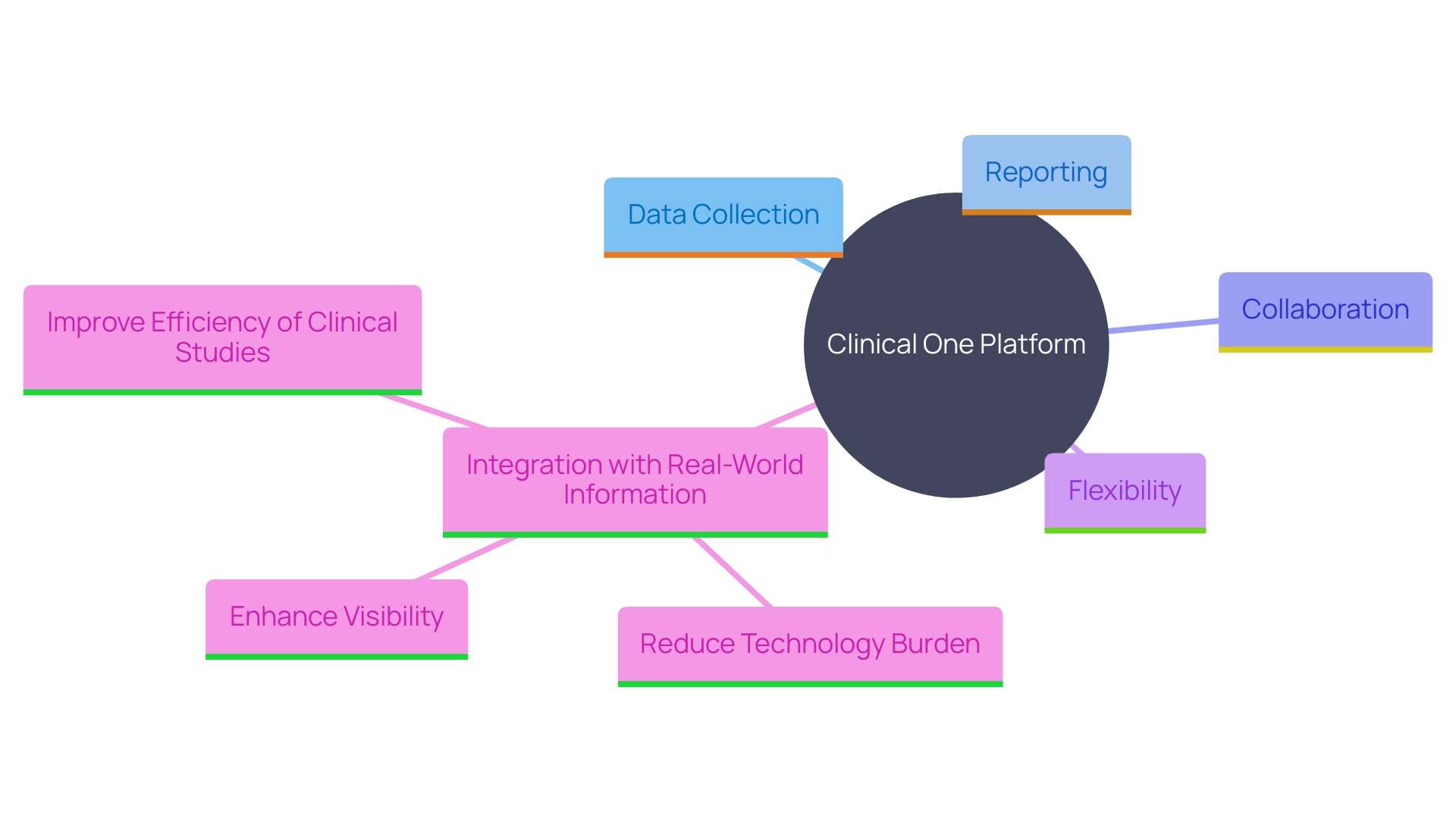
Clinical One offers a thorough platform for overseeing every element of research studies, from , which reduces technology burden by aggregating, connecting, and communicating across various essential systems. This unified approach simplifies processes, reduces training time, and enhances collaboration among study teams. The platform’s flexibility allows for seamless adjustments as study requirements evolve, making it suitable for different .
By providing a , Clinical One delivers a comprehensive overview of research locations, enabling stakeholders to navigate the technology ecosystem effortlessly. This capability is critical in reducing site burden, as it provides full visibility of studies, sponsors, and systems through . These dashboards enable research teams to handle priority tasks across studies, regardless of the provider or backer, ensuring a thorough and effective study process.
Furthermore, Clinical One tackles major obstacles in , such as the dependence on time- and labor-intensive information gathering techniques, by integrating with (RWI). This integration not only reduces the duplication of data already existing in electronic health records (EHR) but also captures a more complete picture of participant lives, including social determinants of health. This approach enhances the validity and generalizability of study findings, ultimately supporting the goal of producing evidence about medical interventions that are reflective of real-world scenarios.
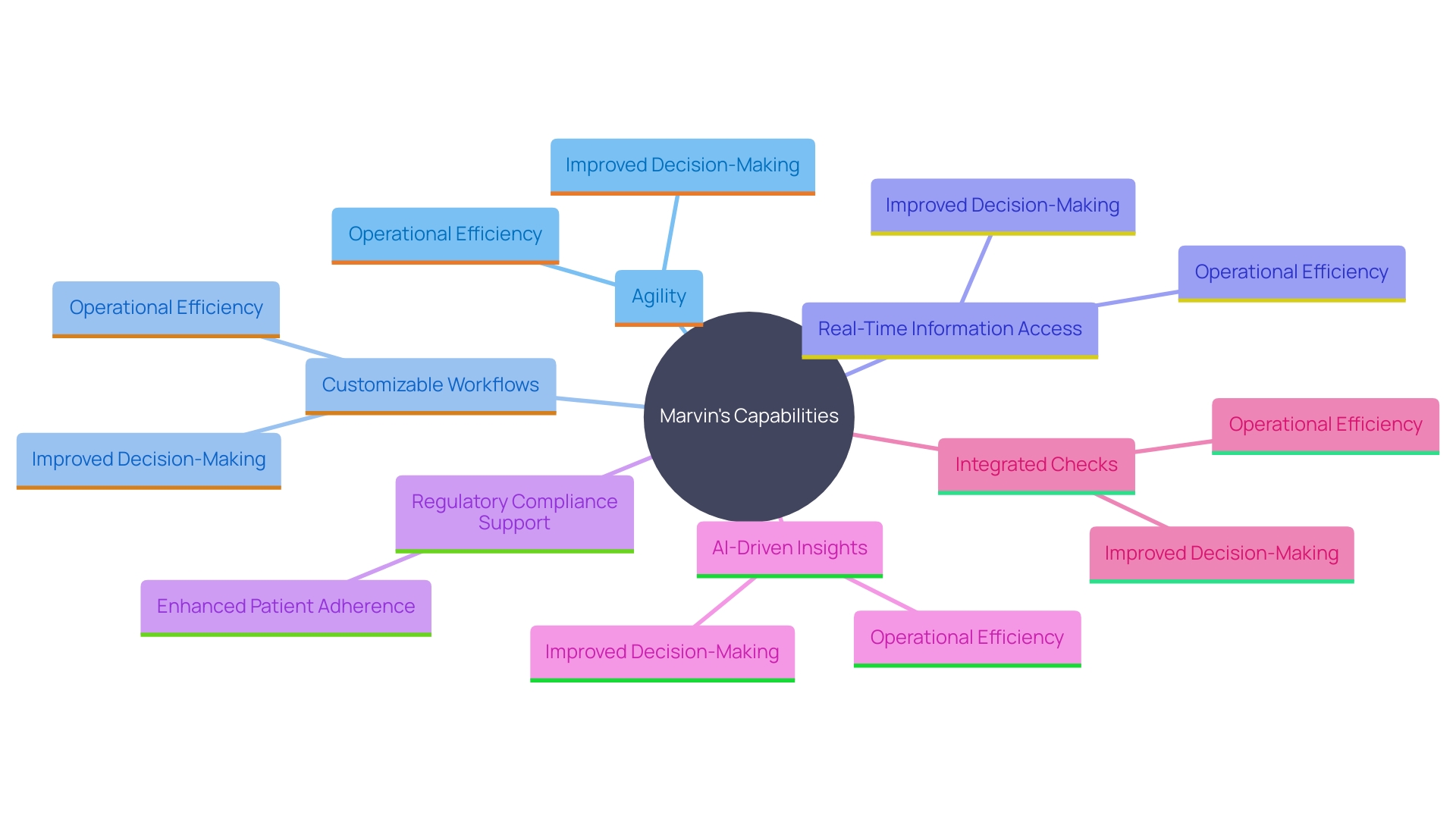
Marvin: Comprehensive and Agile EDC Solution
Marvin is distinctive for its agility and extensive features customized for research requirements. By offering and real-time information access, Marvin significantly enhances decision-making and . This is especially crucial considering the increasing intricacy of information in , where investigations frequently combine various information sources, including conventional and digital inputs. For example, contemporary experiments can gather up to 20 different sources and billions of information points, necessitating robust systems like Marvin.
Marvin’s strong support for is another key advantage, ensuring that all processes align with stringent industry standards. This capability is crucial as can result in significant costs and delays. Moreover, Marvin’s incorporation of assists in extracting meaningful insights from various information types, further enhancing its reliability and utility.
Additionally, Marvin’s integrated checks and reminders boost patient adherence and information quality, decreasing errors and enhancing the overall trustworthiness of . Such attributes are vital in today’s research environment, where effective information management and high-quality results are crucial. ‘Marvin’s extensive assistance for the whole process makes it a reliable option for organizations looking to optimize their research efforts and obtain quicker regulatory approvals and market preparedness.’.
Klindat: Secure and Intuitive Data Management
Klindat emphasizes both safety and usability, offering a highly efficient platform for information management in . The system’s intuitive design ensures seamless navigation, while advanced encryption and access controls protect sensitive information. This makes Klindat particularly attractive for trials with strict . Dr. Jeff Barrett, Chief Science Officer at Aridhia, emphasizes the significance of secure platforms: ‘A builds on the idea of a trusted research environment (TRE) by offering remote access to information and tools for analysis in a securely controlled workspace.’ This approach not only ensures that information and models are protected but also supports the —making information findable, accessible, interoperable, and reusable. ‘The effectiveness of such platforms has been demonstrated in recent assessments, including the evaluation of , which has renewed interest in innovative study designs.’. As Mr. Baara from EDETEK indicates, ‘By combining processes and , this method lessens the load on sponsors, locations, and patients,’ thus improving data quality and speeding up study timelines.
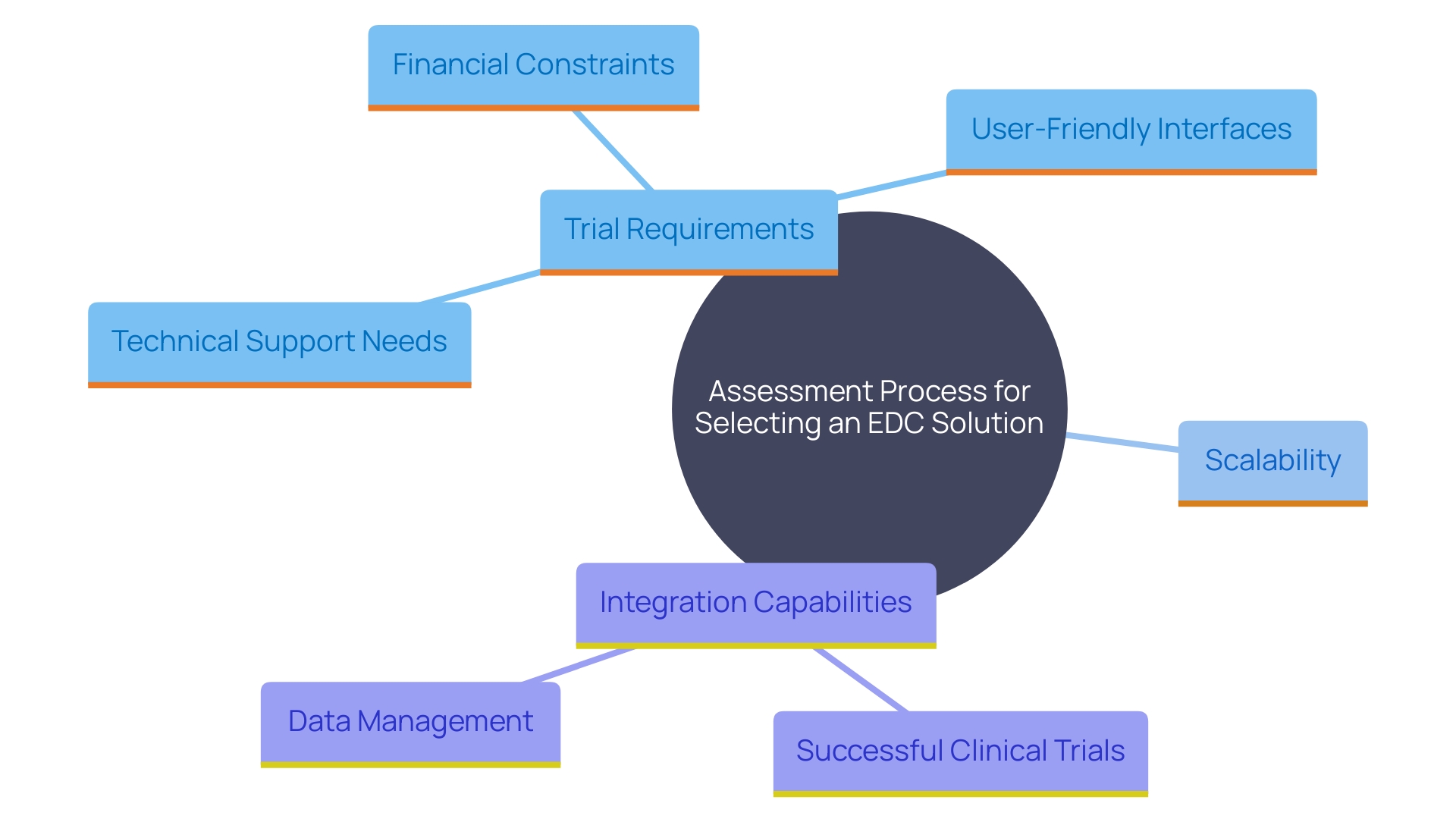
Choosing the Right EDC System for Your Clinical Trial
Selecting the appropriate EDC solution requires a comprehensive assessment of various essential elements, such as the particular requirements of the trial, financial limitations, and the degree of technical assistance needed. One must consider the , ease of use, and how seamlessly it integrates with existing tools. The importance of user-friendliness is underscored by the fact that like Greenlight Guru Clinical offer intuitive interfaces, allowing quick onboarding without requiring prior programming experience. This for ensuring collaboration with clinical teams at study sites, thereby reducing risks related to non-compliance.
Moreover, the capacity to scale studies is crucial. As Faith Kolb, SDC COO, emphasized, their platform’s , assistance, and feedback through personal devices are crucial in attaining quality information and dependable results. This method not only boosts patient enrollment and retention but also increases information precision, resulting in more successful studies.
The incorporation of technology in medical studies is creating a linked healthcare environment. ‘This modernized infrastructure supports not only information collection but also the recording of critical metadata.’. For instance, today’s can involve up to 20 different information sources, capturing approximately 8.1 million points of information per study. Certain research, particularly those engaging in ongoing information collection from linked devices, can accumulate tens of millions to over a billion points of information, highlighting the necessity for strong EDC frameworks.
Organizations should also conduct a detailed comparison of features and functionalities to ensure the selected EDC solution aligns with their research goals. As highlighted by industry experts, a new EDC solution can streamline the pathway to and market entry, ultimately generating more revenue. This is particularly significant for organizations transitioning from paper-based systems, as a modern EDC can significantly enhance data quality and operational efficiency.
Conclusion
In summary, Electronic Data Capture (EDC) systems have fundamentally transformed the clinical trial landscape by enhancing data collection, management, and analysis. Their ability to integrate multiple data sources, including traditional and digital inputs, allows for the handling of vast amounts of data—often reaching billions of data points in modern studies. The primary benefits of these systems include improved data accuracy, real-time monitoring, and streamlined regulatory compliance, all of which contribute to faster trial processes and more reliable outcomes.
Key features of EDC systems, such as user-friendly interfaces, robust security protocols, and advanced reporting tools, make them indispensable for clinical research. The integration capabilities with other trial management tools further enhance efficiency, ensuring that data collection and analysis are seamless. Moreover, the use of electronic consent and patient-reported outcomes significantly reduces errors and enhances data quality, which is critical for regulatory submissions.
A comparative analysis of top EDC systems reveals that each offers unique strengths tailored to varying trial needs. Whether it is the comprehensive support of Medidata Rave, the intuitive design of Greenlight Guru Clinical, or the unified approach of Clinical One, selecting the right EDC system is crucial for optimizing trial success. The ongoing evolution of clinical trials necessitates robust systems that can adapt to increasing complexity and volume, ultimately driving advancements in medical research and improving patient outcomes.
Discover how bioaccess™ can enhance your clinical trials with tailored EDC solutions. Contact us today to learn more!
Frequently Asked Questions
What are Electronic Data Capture (EDC) systems?
EDC systems are platforms that transform the collection and management of information in clinical studies by integrating both traditional and digital data sources.
How many information sources do contemporary experiments typically use?
Contemporary experiments usually employ between five to ten information sources, with some studies using up to twenty sources, resulting in approximately 8.1 million data points per study.
What are the main benefits of using EDC platforms?
The main benefits include improved data accuracy by reducing human errors, immediate access to information for efficient oversight, simplified compliance with regulatory requirements, and faster patient registration and data reporting, leading to quicker study outcomes.
How do EDC systems enhance data quality?
EDC systems provide real-time reminders, support, and feedback through personal devices, ensuring data quality and reliable results, which aids both researchers and participants.
What features should an effective EDC system have?
An efficient EDC system should feature user-friendly interfaces and customizable input forms, strong security measures to protect sensitive information, integration capabilities with other clinical study management tools, advanced reporting tools and audit trails for data integrity, and support for electronic consent (eConsent) and multiple languages.
Why is integration with other tools important for EDC systems?
Integration improves the effectiveness of data gathering and analysis by allowing seamless data flow between various clinical study management tools, facilitating better overall research processes.
What role does electronic consent (eConsent) play in EDC systems?
eConsent platforms automate the presentation of necessary information to patients, reducing non-compliance risks and legal issues associated with trial participation.
What considerations should be taken into account when selecting an EDC system?
When selecting an EDC system, consider user experience and ease of use, scalability to accommodate study growth, cost-effectiveness and budget constraints, technical support availability, and integration with existing tools and processes.
How does the use of EDC systems impact the success of clinical studies?
By streamlining data collection and management, EDC systems enhance data quality, improve regulatory submission processes, and ultimately lead to successful clinical research outcomes.
Can EDC systems help in regulatory submissions?
Yes, advanced EDC technologies can significantly streamline the regulatory submission process, ensuring compliance and increasing the likelihood of approval for medical interventions.
What is the significance of real-time data processing in EDC systems?
Real-time data processing allows for immediate access to information, enhancing oversight and enabling quicker responses to any issues that may arise during the study.
How do different EDC platforms compare?
Different EDC platforms offer unique strengths. For example, Medidata Rave is known for managing large-scale studies efficiently, Viedoc Clinic emphasizes user engagement with its intuitive design, Clinical One offers a unified platform for comprehensive study management, and Marvin focuses on customizable workflows and regulatory compliance.
What future trends are anticipated for EDC systems?
The continuous integration of advanced technologies, real-time data collection, and a focus on enhancing user experience are anticipated trends that will further improve the efficiency and effectiveness of EDC systems in clinical research.
List of Sources
- Benefits of EDC Systems in Clinical Trials
- greenlight.guru (https://greenlight.guru/blog/data-management-and-reporting-in-fda-regulated-clinical-trials)
- greenlight.guru (https://greenlight.guru/blog/electronic-data-capture-edc-system-buyers-guide)
- medicaldevice-network.com (https://medicaldevice-network.com/news/sdc-byod-epro-trials)
- hitconsultant.net (https://hitconsultant.net/2023/11/21/clinical-trial-data-strategy-shifting-to-innovative-data-collection)
- medidata.com (https://medidata.com/en/company/eclinical-solutions?utm_source=twitter&utm_medium=sm&utm_content=e-clinical-15-years&utm_campaign=gl-q422-partner)
- globallogic.com (https://globallogic.com/insights/white-papers/data-quality-solutions-for-stream-and-batch-data-processing?utm_source=whitepaper&utm_medium=x&utm_id=OrganicBG)
- Key Features of EDC Systems
- greenlight.guru (https://greenlight.guru/blog/data-management-and-reporting-in-fda-regulated-clinical-trials)
- jamanetwork.com (https://jamanetwork.com/journals/jama/fullarticle/2822037?utm_source=twitter&utm_campaign=content-shareicons&utm_content=article_engagement&utm_medium=social&utm_term=080624)
- grants.nih.gov (https://grants.nih.gov/grants/guide/notice-files/NOT-OD-24-063.html)
- greenlight.guru (https://greenlight.guru/blog/guide-clinical-data-management-medtech)
- hitconsultant.net (https://hitconsultant.net/2023/11/21/clinical-trial-data-strategy-shifting-to-innovative-data-collection)
- medicaldevice-network.com (https://medicaldevice-network.com/sponsored/game-changing-how-ai-and-automation-is-revolutionising-clinical-trial-admin)
- medicaldevice-network.com (https://medicaldevice-network.com/news/sdc-byod-epro-trials)
- greenlight.guru (https://greenlight.guru/blog/electronic-data-capture-edc-system-buyers-guide)
- cdc.gov (https://cdc.gov/mmwr/volumes/73/su/su7303a1.htm?s_cid=su7303a1_w)
- nexus.od.nih.gov (https://nexus.od.nih.gov/all/2024/02/28/seeking-ideas-on-using-common-data-elements-for-nih-supported-clinical-research)
- cdisc.org (https://cdisc.org/standards/in-development/digital-health-technologies)
- Comparative Analysis of Top EDC Systems
- hitconsultant.net (https://hitconsultant.net/2023/11/21/clinical-trial-data-strategy-shifting-to-innovative-data-collection)
- medidata.com (https://medidata.com/en/company/eclinical-solutions?utm_source=twitter&utm_medium=sm&utm_content=e-clinical-15-years&utm_campaign=gl-q422-partner)
- jamanetwork.com (https://jamanetwork.com/journals/jama/fullarticle/2822037?utm_source=twitter&utm_campaign=content-shareicons&utm_content=article_engagement&utm_medium=social&utm_term=080624)
- med-technews.com (https://med-technews.com/news/Digital-in-Healthcare-News/medidata-announces-rave-lite-to-support-growth-in-early-and-late-stage-clinical-trials)
- greenlight.guru (https://greenlight.guru/blog/electronic-data-capture-edc-system-buyers-guide)
- greenlight.guru (https://greenlight.guru/blog/guide-clinical-data-management-medtech)
- greenlight.guru (https://greenlight.guru/blog/guide-clinical-data-management-medtech)
- greenlight.guru (https://greenlight.guru/blog/electronic-data-capture-edc-system-buyers-guide)
- Viedoc Clinic: Modern and Streamlined Interface
- hitconsultant.net (https://hitconsultant.net/2024/10/02/5-ways-tech-is-enhancing-patient-engagement-in-clinical-trials)
- medidata.com (https://medidata.com/en/company/eclinical-solutions?utm_source=twitter&utm_medium=sm&utm_content=e-clinical-15-years&utm_campaign=gl-q422-partner)
- hitconsultant.net (https://hitconsultant.net/2024/01/17/the-new-era-of-clinical-trials-adopting-electronic-informed-consent)
- hitconsultant.net (https://hitconsultant.net/2023/11/06/survey-shows-burdens-on-clinical-research-sites-sharply-increasing-since-2018)
- academic.oup.com (https://academic.oup.com/jamia/advance-article/doi/10.1093/jamia/ocad249/7499277)
- hitconsultant.net (https://hitconsultant.net/2023/11/21/clinical-trial-data-strategy-shifting-to-innovative-data-collection)
- medcitynews.com (https://medcitynews.com/2023/12/connecting-the-clinical-ecosystem-to-alleviate-site-burden)
- jamanetwork.com (https://jamanetwork.com/journals/jama/fullarticle/2822037?utm_source=twitter&utm_campaign=content-shareicons&utm_content=article_engagement&utm_medium=social&utm_term=080624)
- academic.oup.com (https://academic.oup.com/jamia/advance-article/doi/10.1093/jamia/ocad249/7499277)
- bmcmedresmethodol.biomedcentral.com (https://bmcmedresmethodol.biomedcentral.com/articles/10.1186/s12874-024-02317-z)
- arxiv.org (https://arxiv.org/abs/2312.13423)
- Medidata Rave: Advanced, Robust, and Secure
- medidata.com (https://medidata.com/en/company/eclinical-solutions?utm_source=twitter&utm_medium=sm&utm_content=e-clinical-15-years&utm_campaign=gl-q422-partner)
- medtechintelligence.com (https://medtechintelligence.com/news_article/key-barriers-to-rwd-adoption)
- The Triad of Inclusivity, Innovation, and Scalability: Imperatives for Today’s Clinical Trial Teams – MedCity News (https://medcitynews.com/2024/07/the-triad-of-inclusivity-innovation-and-scalability-imperatives-for-todays-clinical-trial-teams)
- medcitynews.com (https://medcitynews.com/2024/01/making-the-most-of-real-world-data-in-clinical-trials)
- medcitynews.com (https://medcitynews.com/2023/11/virtual-data-rooms-are-securing-the-next-wave-of-medical-research-advancements)
- medicaldevice-network.com (https://medicaldevice-network.com/sponsored/how-saas-platforms-are-breaking-the-mold-for-clinical-trials)
- med-technews.com (https://med-technews.com/medtech-insights/medtech-regulatory-insights/simplifying-samd-regulatory-compliance-with-ai-driven-expert)
- medidata.com (https://medidata.com/en/company/eclinical-solutions?utm_source=twitter&utm_medium=sm&utm_content=e-clinical-15-years&utm_campaign=gl-q422-partner)
- oracle.com (https://oracle.com/life-sciences)
- Clinical One: Unified Platform for Simplified Trials
- The Triad of Inclusivity, Innovation, and Scalability: Imperatives for Today’s Clinical Trial Teams – MedCity News (https://medcitynews.com/2024/07/the-triad-of-inclusivity-innovation-and-scalability-imperatives-for-todays-clinical-trial-teams)
- hitconsultant.net (https://hitconsultant.net/2024/07/10/solving-for-tech-overload-in-clinical-trials)
- jamanetwork.com (https://jamanetwork.com/journals/jama/fullarticle/2822037?utm_source=twitter&utm_campaign=content-shareicons&utm_content=article_engagement&utm_medium=social&utm_term=080624)
- pharmavoice.com (https://pharmavoice.com/spons/trends-shaping-the-pharmaceutical-industry-in-2024/702703)
- clinicaltrialsarena.com (https://clinicaltrialsarena.com/news/ai-rwd-avoid-replication-clinical-trials)
- aridhia.com (https://aridhia.com/blog/bringing-platform-trials-closer-to-reality-by-enabling-with-digital-research-environment-dre-connectivity)
- greenlight.guru (https://greenlight.guru/blog/guide-clinical-data-management-medtech)
- medidata.com (https://medidata.com/en/company/eclinical-solutions?utm_source=twitter&utm_medium=sm&utm_content=e-clinical-15-years&utm_campaign=gl-q422-partner)
- jamanetwork.com (https://jamanetwork.com/journals/jama/fullarticle/2822037?utm_source=twitter&utm_campaign=content-shareicons&utm_content=article_engagement&utm_medium=social&utm_term=080624)
- Marvin: Comprehensive and Agile EDC Solution
- med-technews.com (https://med-technews.com/news/Digital-in-Healthcare-News/medidata-announces-rave-lite-to-support-growth-in-early-and-late-stage-clinical-trials)
- medicaldevice-network.com (https://medicaldevice-network.com/news/sdc-byod-epro-trials)
- prnewswire.com (https://prnewswire.com/news-releases/sanguine-biosciences-adds-advanced-health-data-integration-capabilities-that-further-enhance-translational-medical-research-302027966.html)
- hitconsultant.net (https://hitconsultant.net/2023/11/21/clinical-trial-data-strategy-shifting-to-innovative-data-collection)
- medidata.com (https://medidata.com/en/company/eclinical-solutions?utm_source=twitter&utm_medium=sm&utm_content=e-clinical-15-years&utm_campaign=gl-q422-partner)
- greenlight.guru (https://greenlight.guru/blog/electronic-data-capture-edc-system-buyers-guide)
- medicaldevice-network.com (https://medicaldevice-network.com/sponsored/game-changing-how-ai-and-automation-is-revolutionising-clinical-trial-admin)
- medcitynews.com (https://medcitynews.com/2023/12/connecting-the-clinical-ecosystem-to-alleviate-site-burden)
- rockhealth.com (https://rockhealth.com/insights/from-buzzword-to-business-case-constructing-ai-use-cases-for-pharma)
- elsevier.com (https://elsevier.com/connect/how-knowledge-graphs-can-supercharge-drug-repurposing)
- mckinsey.com (https://mckinsey.com/industries/healthcare/our-insights/the-ai-opportunity-how-payers-can-capture-it-now)
- Klindat: Secure and Intuitive Data Management
- aridhia.com (https://aridhia.com/blog/bringing-platform-trials-closer-to-reality-by-enabling-with-digital-research-environment-dre-connectivity)
- rethinkingclinicaltrials.org (https://rethinkingclinicaltrials.org/news/grand-rounds-february-23-2024-virtual-vigilance-monitoring-of-decentralized-clinical-trials-adrian-hernandez-md-christopher-j-lindsell-phd)
- hitconsultant.net (https://hitconsultant.net/2023/11/21/clinical-trial-data-strategy-shifting-to-innovative-data-collection)
- flatiron.com (https://flatiron.com/clinical-research-solutions/clinical-pipe)
- medidata.com (https://medidata.com/en/company/eclinical-solutions?utm_source=twitter&utm_medium=sm&utm_content=e-clinical-15-years&utm_campaign=gl-q422-partner)
- greenlight.guru (https://greenlight.guru/blog/guide-clinical-data-management-medtech)
- medcitynews.com (https://medcitynews.com/2023/12/connecting-the-clinical-ecosystem-to-alleviate-site-burden)
- infomeddnews.com (https://infomeddnews.com/edetek-news-91624)
- infomeddnews.com (https://infomeddnews.com/viz-ai-news-52324)
- Choosing the Right EDC System for Your Clinical Trial
- jamanetwork.com (https://jamanetwork.com/journals/jama/fullarticle/2822037?utm_source=twitter&utm_campaign=content-shareicons&utm_content=article_engagement&utm_medium=social&utm_term=080624)
- cdisc.org (https://cdisc.org/standards/in-development/digital-health-technologies)
- medicaldevice-network.com (https://medicaldevice-network.com/news/sdc-byod-epro-trials)
- med-technews.com (https://med-technews.com/news/Digital-in-Healthcare-News/medidata-announces-rave-lite-to-support-growth-in-early-and-late-stage-clinical-trials)
- medidata.com (https://medidata.com/en/company/eclinical-solutions?utm_source=twitter&utm_medium=sm&utm_content=e-clinical-15-years&utm_campaign=gl-q422-partner)
- greenlight.guru (https://greenlight.guru/blog/electronic-data-capture-edc-system-buyers-guide)
- hitconsultant.net (https://hitconsultant.net/2023/11/21/clinical-trial-data-strategy-shifting-to-innovative-data-collection)
- medcitynews.com (https://medcitynews.com/2023/12/connecting-the-clinical-ecosystem-to-alleviate-site-burden)
- jamanetwork.com (https://jamanetwork.com/journals/jama/fullarticle/2822037?utm_source=twitter&utm_campaign=content-shareicons&utm_content=article_engagement&utm_medium=social&utm_term=080624)