Introduction
Class III medical devices stand at the forefront of transforming patient care, presenting innovative solutions that significantly enhance treatment outcomes and improve quality of life. This article explores ten remarkable examples of these devices, ranging from implantable cardioverter defibrillators to artificial pancreases, illustrating how advancements in technology are reshaping the medical landscape. However, as these devices revolutionize healthcare, critical questions emerge regarding their accessibility, efficacy, and the disparities in their utilization among diverse patient populations.
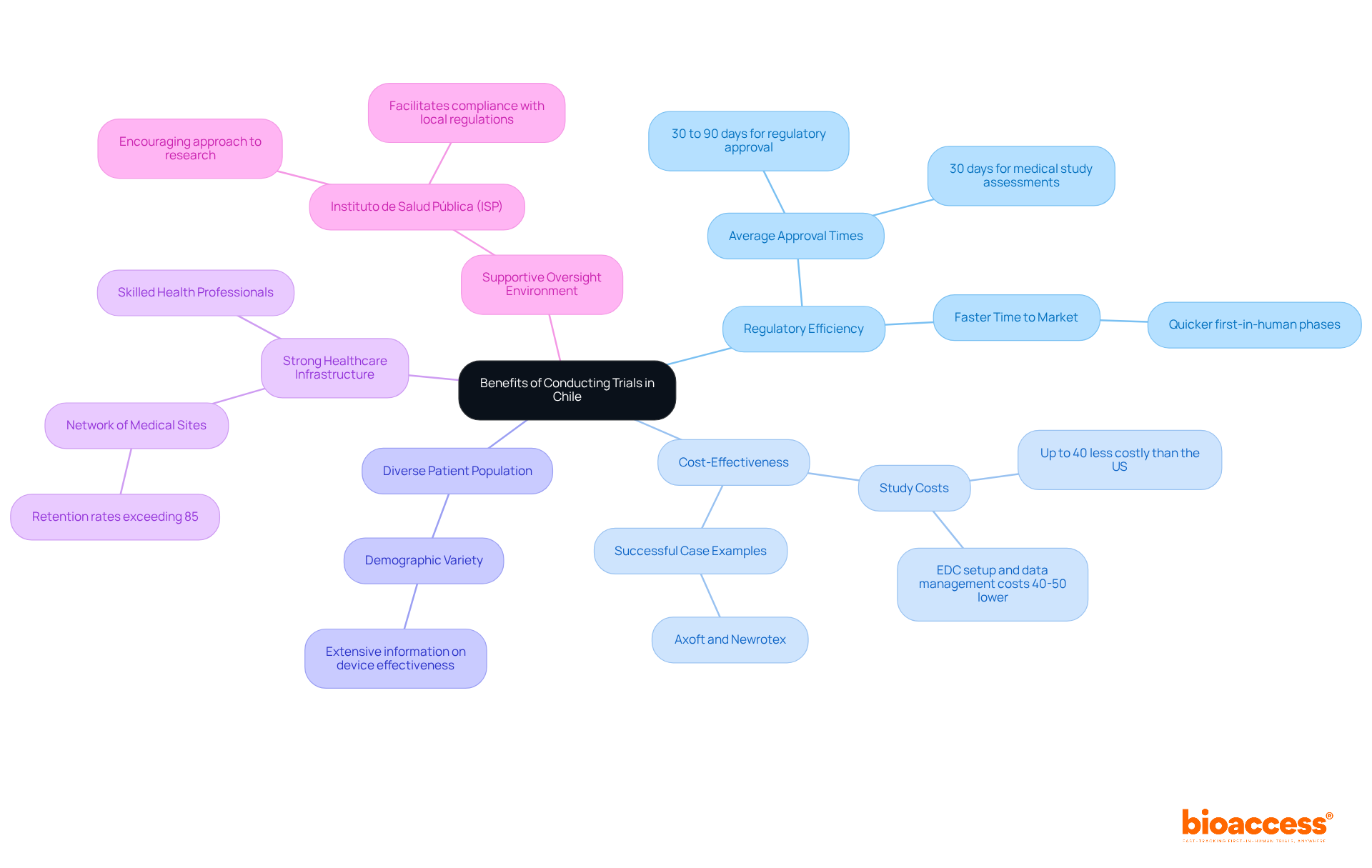
bioaccess®: Accelerate Clinical Research for Class III Medical Devices
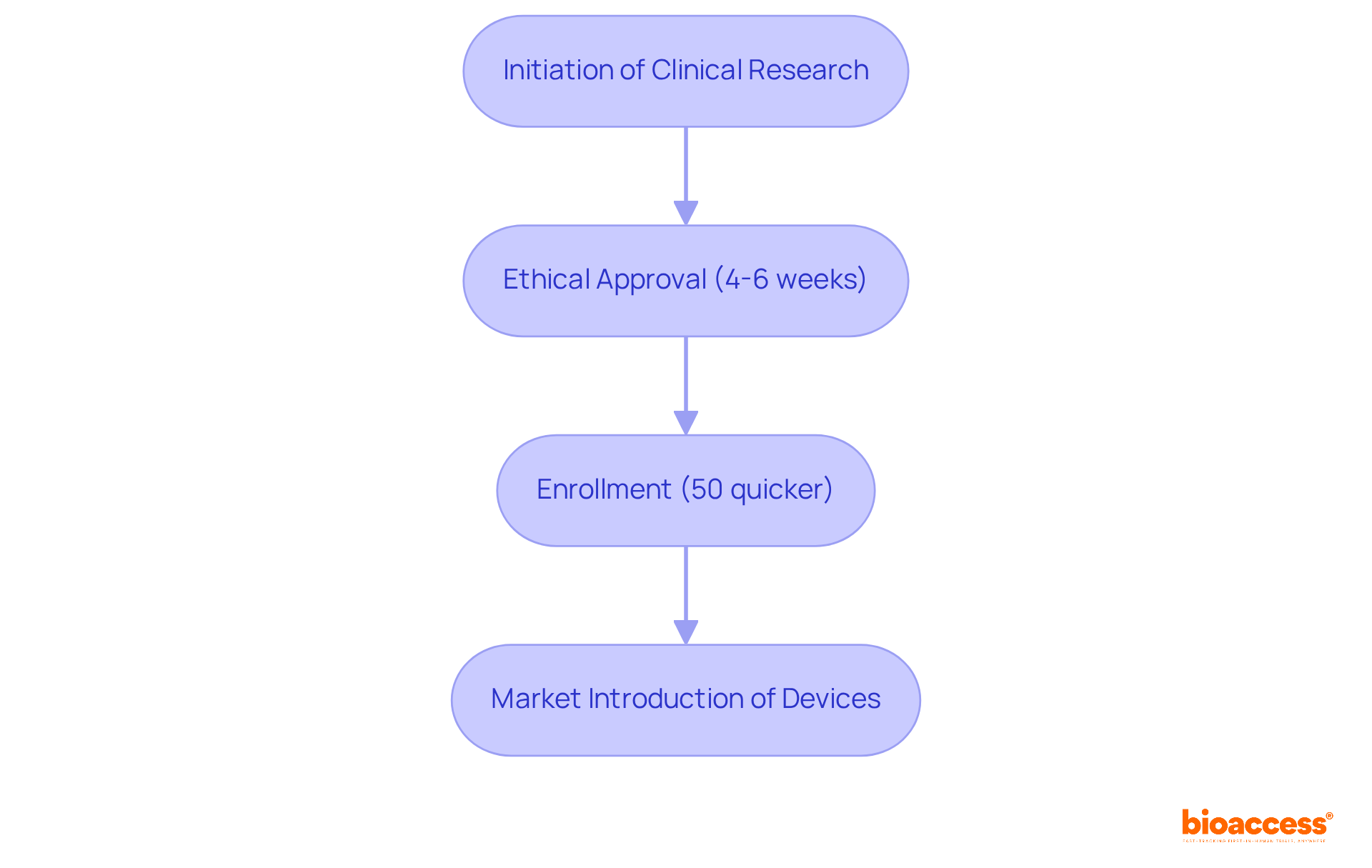
bioaccess® excels in expediting clinical research for examples of , leveraging its extensive expertise and regional advantages. The organization secures ethical approvals in an impressive 4-6 weeks—significantly faster than the average timelines observed in traditional markets. This efficiency is complemented by , enabling Medtech and Biopharma innovators to bring to market with remarkable speed.
The importance of cannot be overstated; they are crucial for ensuring participant safety and maintaining trust in the research process. Successful instances from Latin America illustrate how can enhance the overall efficiency of clinical trials, ultimately benefiting individuals by providing and the latest advancements in medical technology.
By utilizing its distinct capabilities, bioaccess® not only accelerates the but also plays a pivotal role in .
Implantable Cardioverter Defibrillator: Life-Saving Heart Rhythm Management
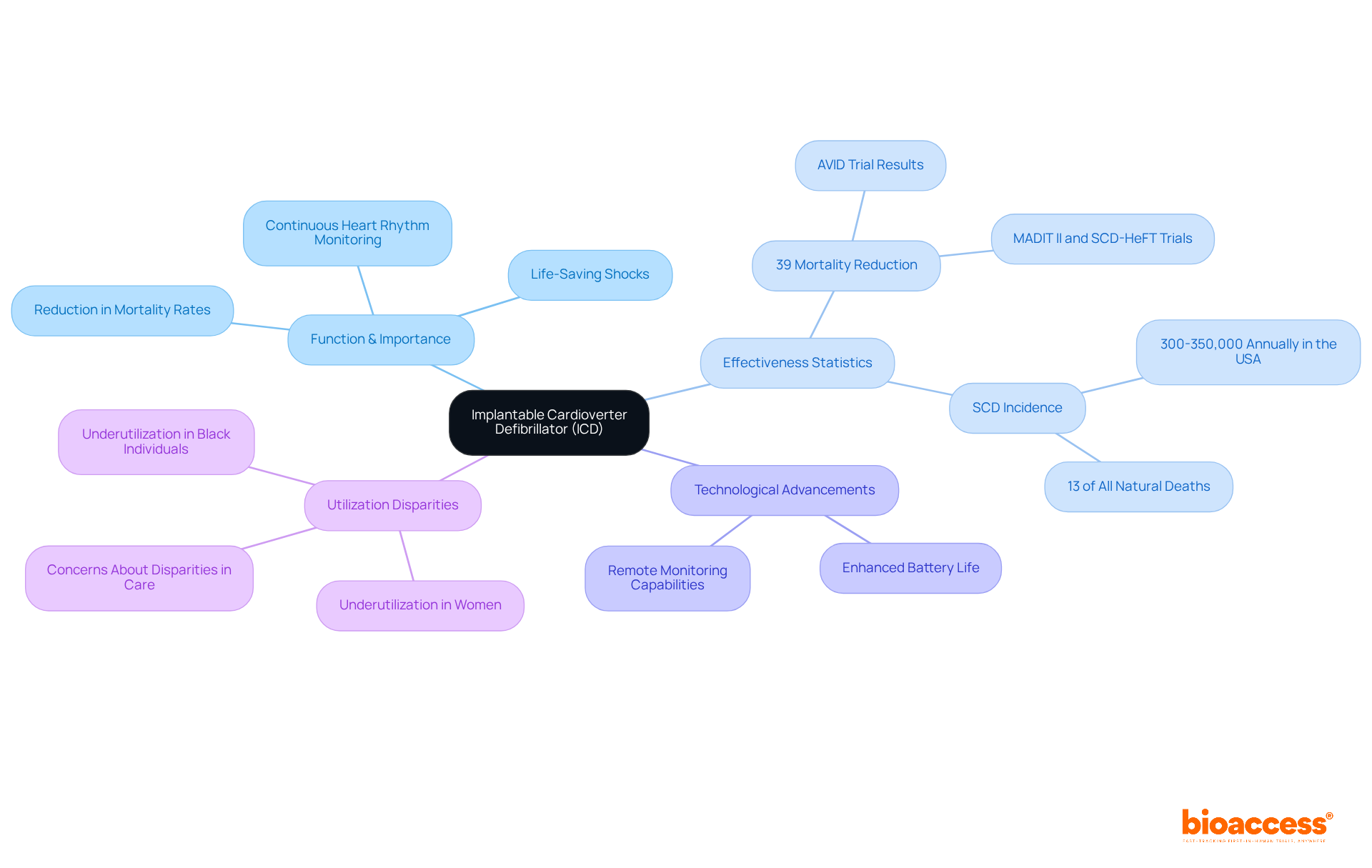
The is crucial in managing individuals at risk of . By continuously monitoring heart rhythms and delivering life-saving shocks when necessary, ICDs significantly reduce mortality rates. Research indicates that individuals with documented sustained experience a notable survival advantage from ICD therapy, particularly in high-risk groups such as those with ischemic cardiomyopathy and a left ventricular ejection fraction (LVEF) of ≤35%.
The AVID trial demonstrated a , underscoring its effectiveness in these populations. Furthermore, the incidence of in the USA is approximately 300-350,000 annually, emphasizing the urgent need for effective interventions like ICDs.
Recent advancements in , including remote monitoring capabilities and enhanced battery life, further improve safety and comfort for patients. These innovations facilitate real-time data transmission to , enabling timely interventions and personalized care. Consequently, ICDs are becoming essential tools in contemporary cardiology, contributing to improved outcomes and survival rates in cases of .
However, it is vital to acknowledge that ICDs are underutilized among certain groups, such as women and Black individuals, raising concerns about disparities in care. Moreover, while , potential complications, such as inappropriate shocks, must be considered in managing individuals.
Total Artificial Heart: Revolutionary Solution for End-Stage Heart Failure
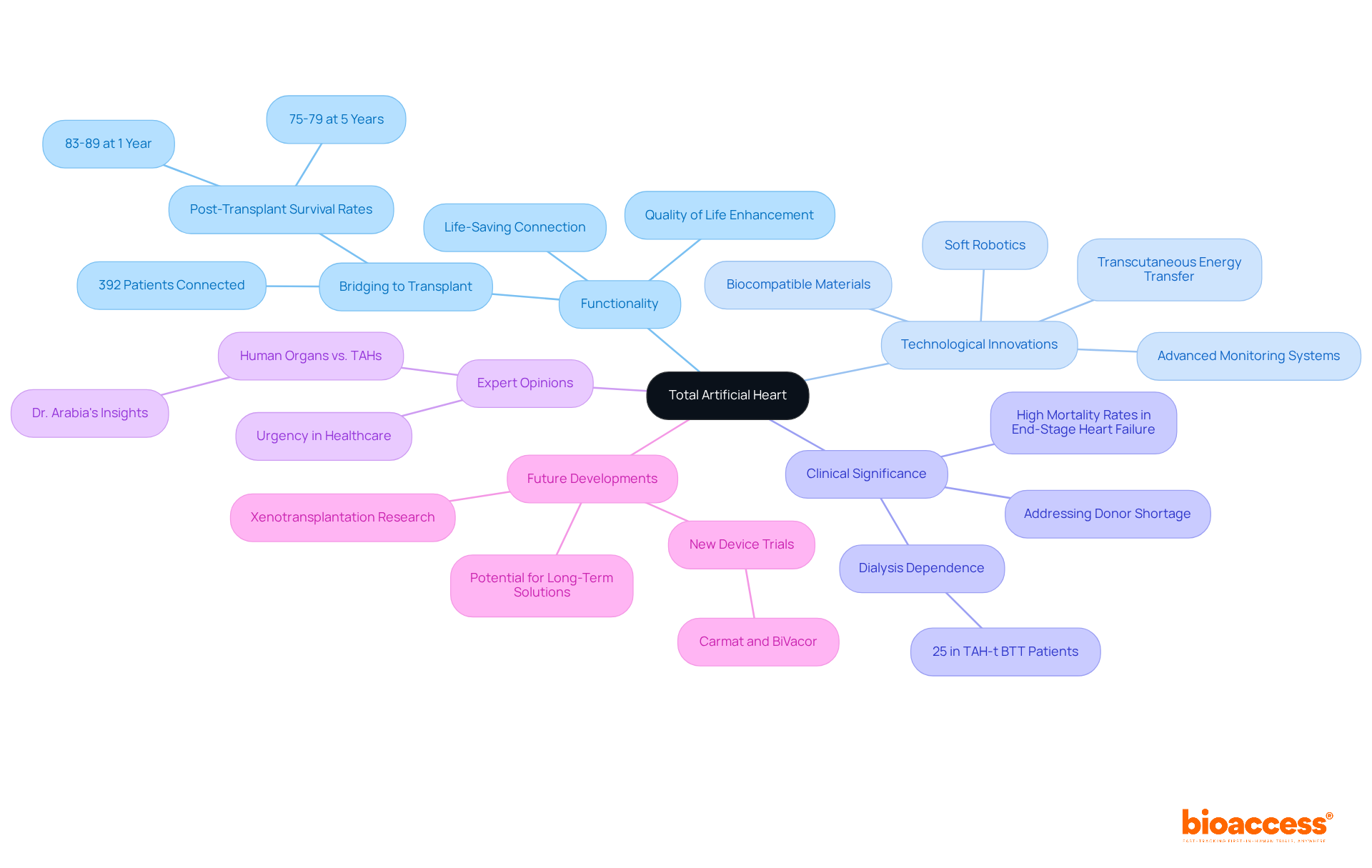
The (TAH) serves as a critical life-saving connection for individuals facing while awaiting transplantation. By fully substituting the organ’s function, TAHs provide essential support and significantly enhance . Recent innovations in TAH technology, including biocompatible materials and , are paving the way for broader applications and improved outcomes for patients. Notably, 392 individuals were connected to transplant with a (TAH-t), underscoring the importance of TAHs in .
Experts emphasize the urgency of TAHs within healthcare. Dr. Arabia notes that while remain the optimal choice, TAHs present a vital alternative for patients experiencing severe ventricular failure when transplants are not readily available. Furthermore, research indicates that individuals who navigate the early postoperative stage with TAHs exhibit comparable long-term survival rates to those who undergo conventional organ transplants.
As the demand for cardiac transplants continues to exceed supply, TAHs emerge as a promising solution to address this critical gap. With ongoing research and development, including and advanced soft robotics, the future of TAHs looks hopeful, offering new possibilities to many patients grappling with the challenges of end-stage heart failure.
Cochlear Implant: Restoring Hearing for the Hearing Impaired
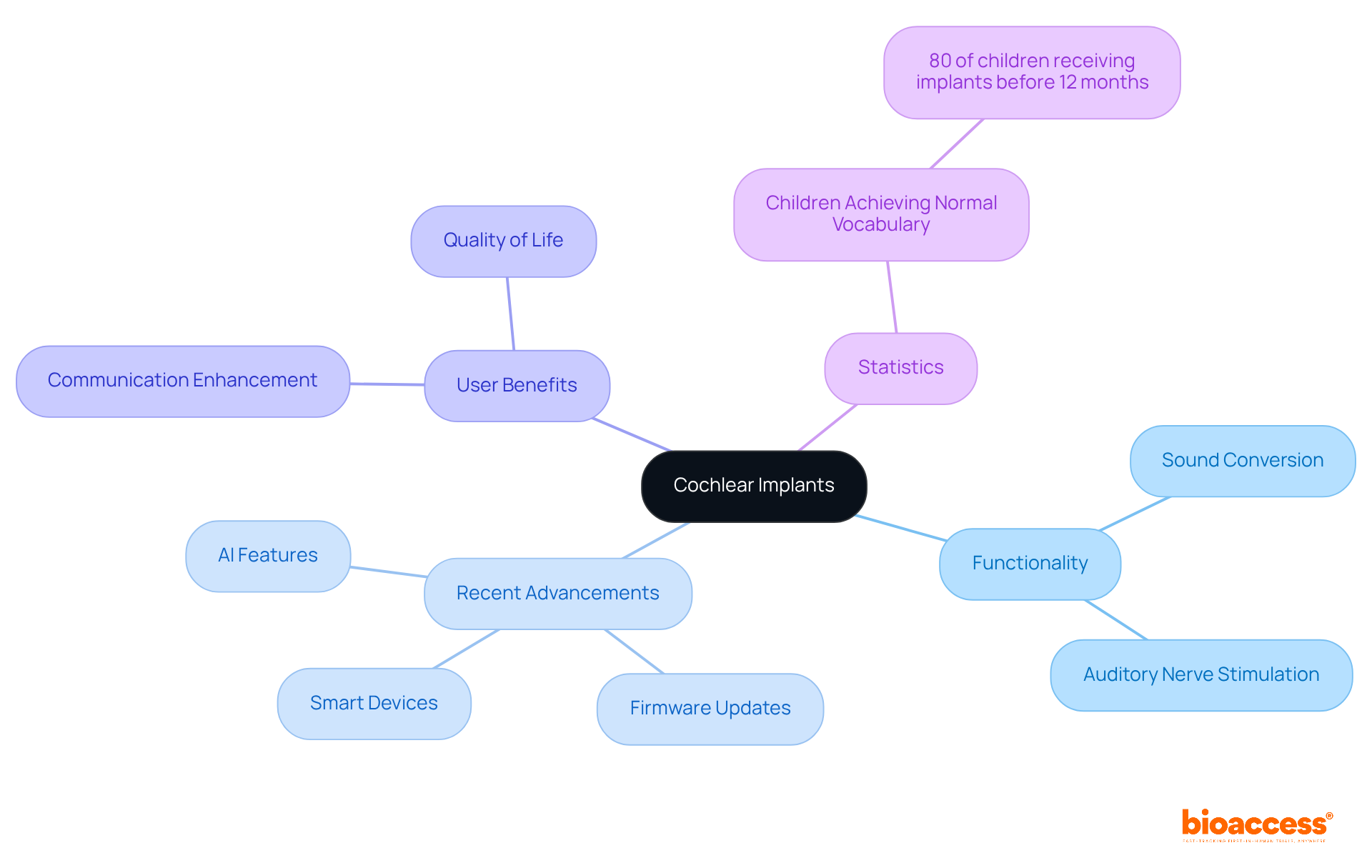
Cochlear implants are that restore hearing in individuals experiencing severe to profound . These devices operate by converting sound into electrical signals that directly stimulate the auditory nerve, enabling users to perceive sound and significantly enhancing their communication abilities.
Recent advancements in cochlear implant systems, particularly the introduction of , have further elevated user experience and . For example, the , the first cochlear implant equipped with its own firmware, allows for continuous updates and personalized settings, guaranteeing that users reap the benefits of the latest innovations throughout their lives. This system also includes and the capability to securely store personalized hearing settings (MAPs), thereby enhancing convenience and efficiency.
Statistics indicate that up to 80% of children receiving cochlear implants before the age of 12 months demonstrate normal receptive vocabulary by the time they enter school, underscoring the . Furthermore, improve sound processing, facilitating better speech recognition in noisy environments and enhancing overall auditory experiences. Context-aware sound processing further fine-tunes device settings based on environmental cues, optimizing the listening experience.
These advancements not only empower users to engage more fully in conversations but also contribute to their social integration and overall quality of life. As Lori Miller, a recipient of the , articulated, “With my cochlear implant, life has changed dramatically for me. Most importantly, I have been able to hear my family, and that is my highest priority.
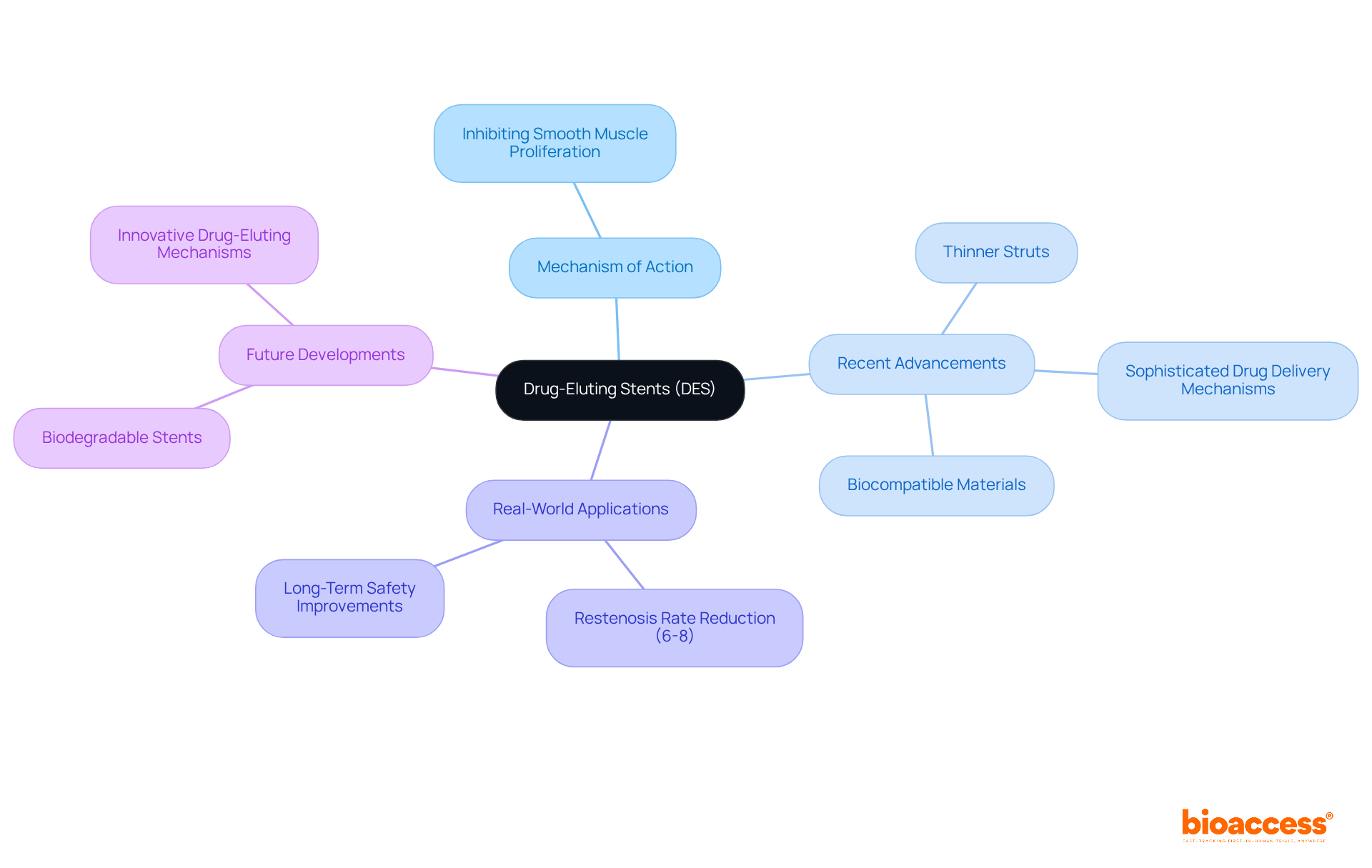
Drug-Eluting Stent: Minimizing Restenosis in Coronary Artery Disease
(DES) are crucial in the fight against restenosis, a common complication following coronary artery interventions. These stents are meticulously designed to release medications that inhibit , thereby significantly reducing the risk of artery re-narrowing.
, including thinner struts and sophisticated , have further bolstered their efficacy and safety. For example, newer DES incorporate and enhanced polymer coatings that minimize inflammatory responses, addressing associated with earlier iterations.
Real-world applications reveal that DES have effectively lowered , with studies indicating a reduction in the incidence of re-narrowing to as low as 6-8% within the first year.
Continuous technological advancements, such as the development of biodegradable stents and , are set to transform the treatment landscape for , promising improved outcomes for patients and fewer complications.
Ventricular Assist Device: Supporting Heart Function in Critical Patients
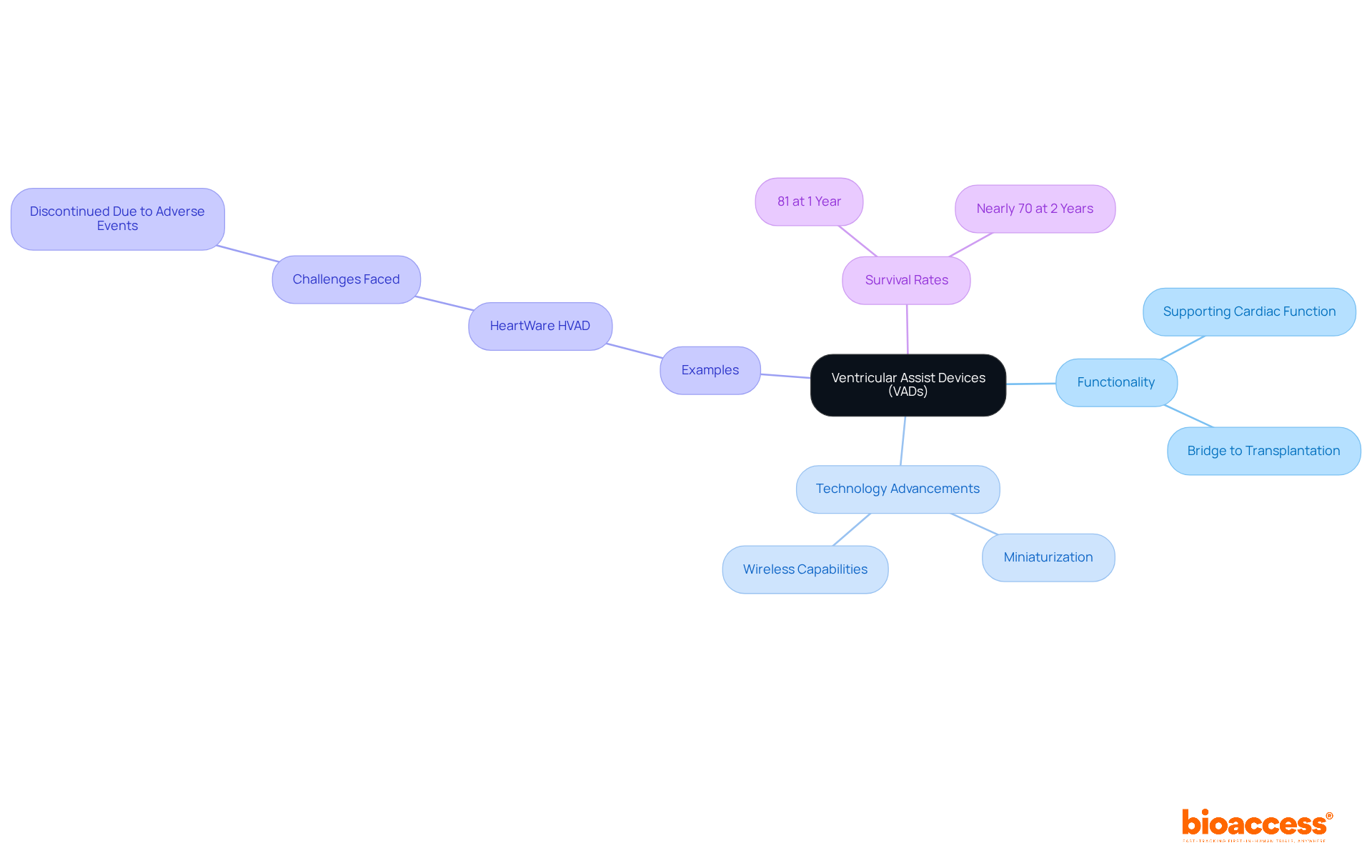
are designed to support cardiac function in individuals suffering from severe . By assisting the heart in pumping blood, VADs significantly alleviate symptoms and enhance quality of life, serving either as a bridge to transplantation or as a long-term therapeutic solution. Recent advancements in , particularly in miniaturization and wireless capabilities, have expanded their applicability and effectiveness.
For instance, the HeartWare HVAD, weighing only 145 grams, exemplifies how smaller designs can lead to less invasive procedures and quicker recovery times for patients. However, it was discontinued in June 2021 due to adverse events, underscoring the ongoing challenges faced in VAD therapy.
Furthermore, innovations such as are paving the way for devices that eliminate the need for external driveline components, thereby reducing infection risks and enhancing user comfort. As Dr. Limael E. Rodriguez states, ‘ have transformed the management of late-stage ,’ highlighting the profound impact these devices have on patient care.
Additionally, statistics reveal that the is:
- 81% at 1 year
- Nearly 70% at 2 years
This reinforces the effectiveness of VADs. The continuous evolution of VAD systems plays a crucial role in improving outcomes, establishing them as an essential element in the management of .
Infusion Pump: Precision Delivery of Medications and Nutrients
Infusion pumps are indispensable that ensure the precise administration of medications and nutrients to patients. By automating the infusion process, these devices significantly mitigate the risk of human error, ensuring consistent dosing. Recent advancements in have introduced , such as and , which bolster monitoring and control over medication delivery.
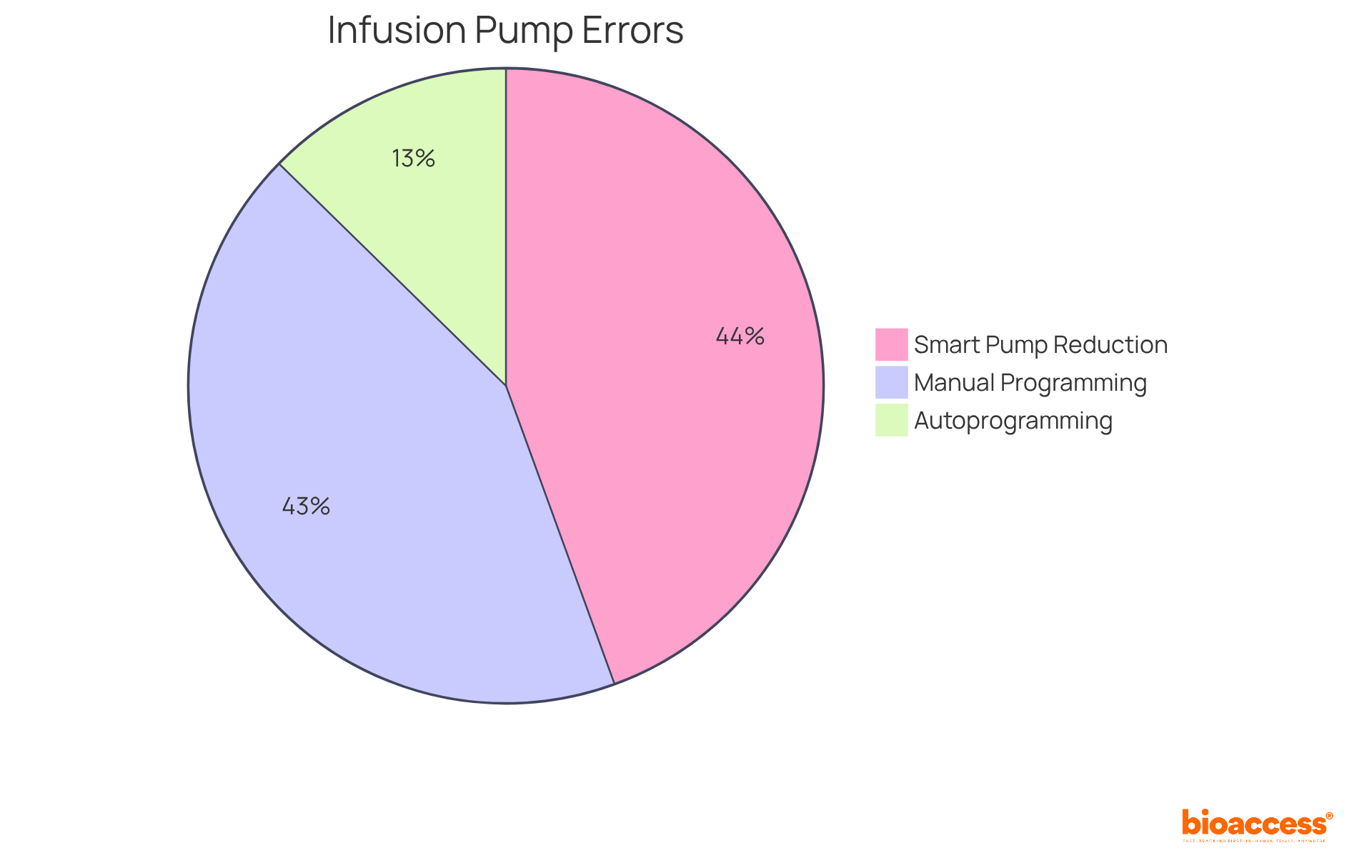
For instance, studies indicate that implementing smart pumps can lead to an 80% reduction in , underscoring their effectiveness in minimizing medication delivery mistakes. Furthermore, real-world applications of these systems have illustrated their ability to streamline workflows and enhance safety, making them essential tools in modern healthcare environments.
Notably, manual programming has been linked to 77.2% of administration and user documentation errors, compared to just 22.8% for autoprogramming, emphasizing the critical need for . Additionally, has resulted in a 16% reduction in , further validating the claims regarding the efficacy of these systems. Comprehensive support from providers like Med One ensures that healthcare facilities can fully leverage the advantages of infusion pump technology.
Pacemaker: Regulating Heartbeats for Improved Cardiac Function
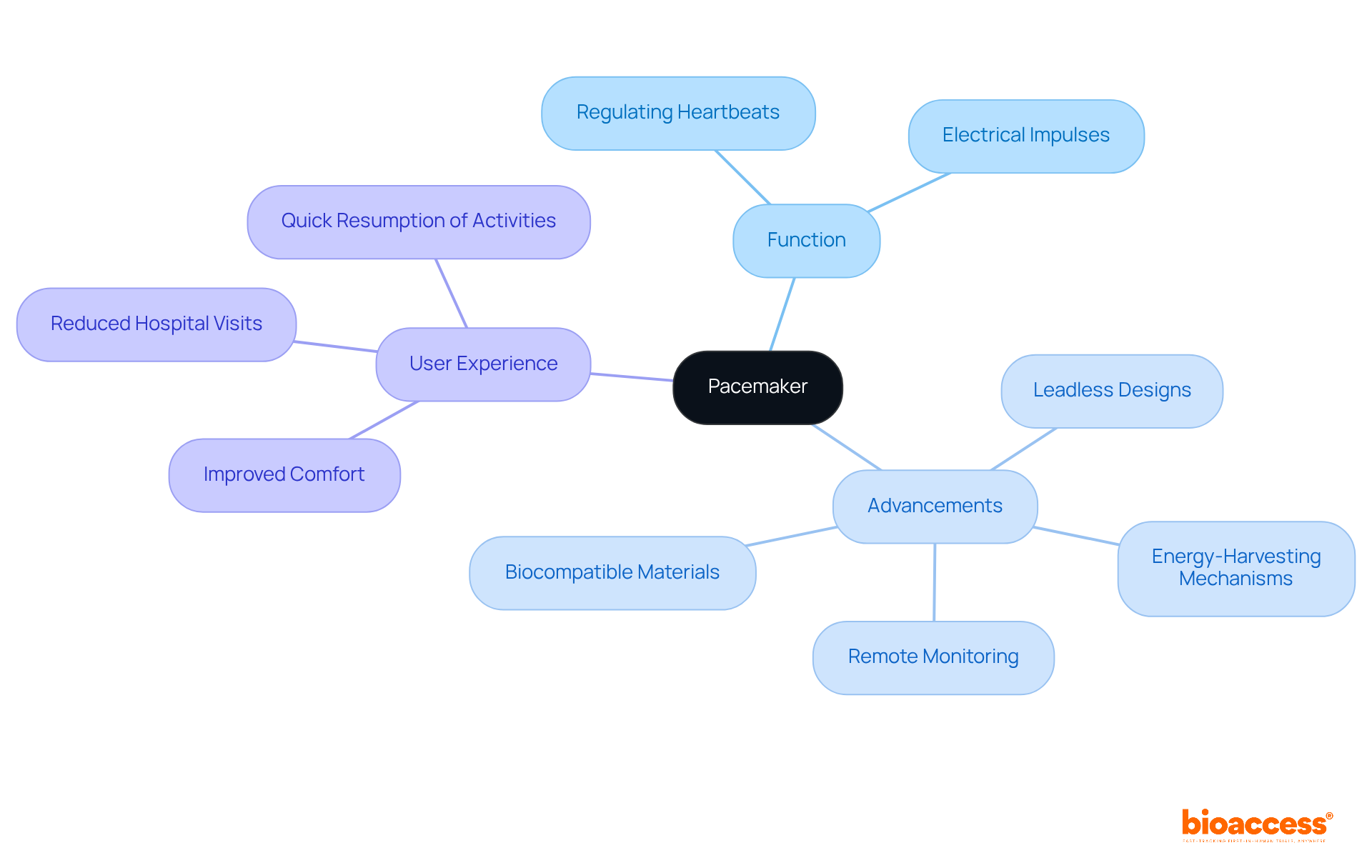
Pacemakers serve a critical function in managing heartbeats for individuals experiencing arrhythmias. By delivering precise electrical impulses to the heart, these devices guarantee a steady and effective heartbeat. Recent , such as , are significantly . These innovations not only enhance the user experience but also address key challenges within the , reflecting a commitment to .
Intraocular Lens: Restoring Vision Post-Cataract Surgery
Intraocular lenses (IOLs) represent a pivotal advancement in cataract surgery, serving as . , particularly in multifocal and toric designs, have markedly improved the correction of diverse vision issues.
- at various distances, thereby reducing the reliance on glasses and enhancing overall visual experiences. A compelling study indicates that 95% of individuals reported their night driving vision was as good as or better than before surgery after receiving multifocal IOLs.
- Similarly, , offering .
- Continuous advancements in IOL materials and designs are not only elevating visual outcomes but also increasing satisfaction, with recent studies revealing that .
These developments are significantly transforming the , positioning IOLs as an indispensable component of modern cataract care. It is advisable for individuals to select surgeons who possess expertise in premium lenses to ensure optimal outcomes.
Artificial Pancreas: Innovative Management for Diabetes Patients
The artificial pancreas represents a , for individuals with type 1 diabetes. By continuously monitoring blood glucose levels and adjusting insulin delivery in real-time, this device significantly enhances glucose control, thereby reducing the risk of both hyperglycemia and hypoglycemia.
Recent innovations in artificial pancreas technology, including the integration of and advanced algorithms, have further optimized health outcomes. For example, the employs a proportional-integral-derivative (PID) controller, effectively managing insulin delivery and significantly decreasing hypoglycemic events. Conversely, the MiniMed 780 G system utilizes fuzzy logic to replicate clinical decision-making, demonstrating improved safety and efficacy compared to earlier models.
Real-world applications of , such as the iLet bionic pancreas, illustrate the potential to simplify , enabling users to avoid making up to 288 decisions daily regarding insulin dosing. As Ed Damiano, a pivotal figure in the development of the iLet, remarked, “The analogy I like to make with the iLet is going from a manual to a fully self-driving car.”
These advancements not only but also , rendering less burdensome and more effective.
Conclusion
The exploration of Class III medical devices reveals their transformative impact on patient care, showcasing innovations that enhance treatment outcomes and improve quality of life. These devices, ranging from implantable cardioverter defibrillators to artificial pancreases, are at the forefront of medical technology, driving advancements that address critical health challenges faced by patients today.
Key insights from the article highlight the significant roles of various Class III devices. For instance:
- Implantable cardioverter defibrillators save lives by managing heart rhythms.
- Total artificial hearts offer hope to those awaiting transplants.
- Cochlear implants restore hearing.
- Drug-eluting stents prevent restenosis.
These examples showcase the breadth of solutions available. Moreover, advancements in infusion pumps, pacemakers, intraocular lenses, and artificial pancreases underscore the continuous evolution of these technologies, ensuring that patient safety and efficacy remain paramount.
As the healthcare landscape evolves, the importance of these innovations cannot be overstated. Embracing the latest advancements in Class III medical devices not only enhances clinical outcomes but also empowers patients to lead healthier lives. Stakeholders in the medical field are encouraged to support ongoing research and development, ensuring that the benefits of these technologies extend to all individuals in need, ultimately shaping a future where advanced medical solutions are accessible and effective for everyone.
Frequently Asked Questions
What is bioaccess® and what role does it play in clinical research for Class III medical devices?
bioaccess® is an organization that specializes in expediting clinical research for Class III medical devices. It leverages its expertise and regional advantages to secure ethical approvals in 4-6 weeks and achieve enrollment rates that are 50% quicker than traditional markets.
Why are ethical approvals important in clinical trials?
Ethical approvals are crucial for ensuring participant safety and maintaining trust in the research process. They help streamline the clinical trial process and provide individuals with access to the latest advancements in medical technology.
What is an Implantable Cardioverter Defibrillator (ICD) and how does it function?
An ICD is a medical device that manages individuals at risk of sudden cardiac arrest by continuously monitoring heart rhythms and delivering life-saving shocks when necessary. It significantly reduces mortality rates, particularly in high-risk groups.
What evidence supports the effectiveness of ICD therapy?
The AVID trial demonstrated a 39% decrease in overall mortality with ICD therapy, highlighting its effectiveness for individuals with documented sustained ventricular arrhythmias.
What recent advancements have been made in ICD technology?
Recent advancements include remote monitoring capabilities and enhanced battery life, which improve safety and comfort for patients by facilitating real-time data transmission to healthcare providers.
What challenges exist regarding the utilization of ICDs?
ICDs are underutilized among certain groups, such as women and Black individuals, raising concerns about disparities in care. Additionally, potential complications, such as inappropriate shocks, must be considered in managing individuals.
What is a Total Artificial Heart (TAH) and its significance in healthcare?
A TAH is a life-saving device for individuals facing end-stage cardiac failure while awaiting transplantation. It fully substitutes the heart’s function, providing essential support and improving quality of life.
How does the performance of TAHs compare to conventional organ transplants?
Research indicates that individuals navigating the early postoperative stage with TAHs exhibit comparable long-term survival rates to those who undergo conventional organ transplants.
What are the future prospects for Total Artificial Hearts?
Ongoing research and development, including explorations into xenotransplantation and advanced soft robotics, suggest a hopeful future for TAHs, offering new possibilities for patients with end-stage heart failure.
List of Sources
- bioaccess®: Accelerate Clinical Research for Class III Medical Devices
- agingresearch.org (https://agingresearch.org/blog/new-york-times-article-on-clinical-trials-features-quote-from-the-alliance-for-aging-research)
- bioaccessla.com (https://bioaccessla.com/br/blog/10-essential-insights-on-medical-devices-class-3-for-researchers)
- bioaccess® | MedTech, Biopharma & Radiopharma CRO in Latin America (https://bioaccessla.com/br/blog/10-key-insights-on-clinical-trials-for-medical-devices)
- Factors influencing the time to ethics and governance approvals for clinical trials: a retrospective cross-sectional survey – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10693024)
- Quotes About Medical Research (https://changingthepresent.org/pages/quotes-about-medical-research)
- Implantable Cardioverter Defibrillator: Life-Saving Heart Rhythm Management
- academic.oup.com (https://academic.oup.com/europace/article/14/6/787/547781)
- acc.org (https://acc.org/latest-in-cardiology/ten-points-to-remember/2024/12/11/16/02/revisiting-icd-therapy)
- sciencedirect.com (https://sciencedirect.com/science/article/pii/S0735109721063178)
- acc.org (https://acc.org/latest-in-cardiology/articles/2025/01/07/17/44/the-extravascular-implantable-cardioverter-defibrillator)
- Total Artificial Heart: Revolutionary Solution for End-Stage Heart Failure
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC9771925)
- cardiovascularbusiness.com (https://cardiovascularbusiness.com/topics/clinical/cardiac-surgery/total-artificial-hearts-exploring-future-heart-transplant-technology)
- thecardiologyadvisor.com (https://thecardiologyadvisor.com/features/total-artificial-heart)
- news-medical.net (https://news-medical.net/news/20250605/Breakthrough-soft-robotics-could-redefine-artificial-heart-technology.aspx)
- ncbi.nlm.nih.gov (https://ncbi.nlm.nih.gov/books/NBK234445)
- Cochlear Implant: Restoring Hearing for the Hearing Impaired
- cochlear.com (https://cochlear.com/us/en/corporate/media-center/media-releases/2025/cochlear-launches-worlds-first-and-only-smart-cochlear-implant-system)
- hearingreview.com (https://hearingreview.com/hearing-products/implants-bone-conduction/cochlear-implants/cochlear-launches-smart-cochlear-implant-system)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC12079510)
- slideserve.com (https://slideserve.com/victoria/applications-and-advancements-in-biomechanical-engineering)
- biotechdispatch.com.au (https://biotechdispatch.com.au/news/cochlear-lauunches-the-worlds-first-smart-hearing-implant-system)
- Drug-Eluting Stent: Minimizing Restenosis in Coronary Artery Disease
- pubmed.ncbi.nlm.nih.gov (https://pubmed.ncbi.nlm.nih.gov/35863852)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC6159420)
- jamanetwork.com (https://jamanetwork.com/journals/jama/fullarticle/2816074)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC3567608)
- Ventricular Assist Device: Supporting Heart Function in Critical Patients
- medicalupdate.pennstatehealth.org (https://medicalupdate.pennstatehealth.org/cardiology/technological-advances-lead-to-improved-survival-with-permanent-left-ventricular-assist-devices)
- sciencedirect.com (https://sciencedirect.com/science/article/pii/S0735109722001711)
- annalscts.com (https://annalscts.com/article/view/16809/html)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC3600882)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC3099197)
- Infusion Pump: Precision Delivery of Medications and Nutrients
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC9283846)
- medonegroup.com (https://medonegroup.com/aboutus/blog/infusion-pumps-vital-tools-in-modern-healthcare)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC9359779)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC10085524)
- ncbi.nlm.nih.gov (https://ncbi.nlm.nih.gov/books/NBK555506)
- Pacemaker: Regulating Heartbeats for Improved Cardiac Function
- link.springer.com (https://link.springer.com/article/10.1007/s40124-024-00334-6)
- nature.com (https://nature.com/research-intelligence/nri-topic-summaries/leadless-pacemaker-technology-and-outcomes-micro-27917)
- iuhealth.org (https://iuhealth.org/thrive/living-with-afib-how-pacemakers-improve-quality-life)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC12074492)
- Intraocular Lens: Restoring Vision Post-Cataract Surgery
- drranimenon.com (https://drranimenon.com/advancements-in-cataract-intraocular-lens-iol-technology-enhancing-vision-and-quality-of-life)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC10772437)
- coheneyeinstitute.com (https://coheneyeinstitute.com/blog/game-changing-cataract-lenses-for-2025)
- Checking your browser – reCAPTCHA (https://pmc.ncbi.nlm.nih.gov/articles/PMC12168278)
- crstoday.com (https://crstoday.com/articles/2011-jan/top-quotations)
- Artificial Pancreas: Innovative Management for Diabetes Patients
- niddk.nih.gov (https://niddk.nih.gov/health-information/diabetes/overview/managing-diabetes/artificial-pancreas)
- bu.edu (https://bu.edu/articles/2023/how-a-bionic-pancreas-could-revolutionize-type-1-diabetes-management)
- tandfonline.com (https://tandfonline.com/doi/full/10.1080/17434440.2022.2150546)
- sciencedirect.com (https://sciencedirect.com/science/article/pii/S2214623724000437)