Introduction
Navigating the intricate landscape of medical device regulations in Latin America is essential for companies looking to penetrate this rapidly growing market, valued at an impressive $11 billion. Grasping key standards such as ISO 13485 and ISO 14971 is not merely advantageous; it is vital for ensuring compliance, enhancing product safety, and securing market access. Yet, with each country presenting its own distinct regulatory hurdles, how can manufacturers effectively align their strategies to meet these diverse requirements while also prioritizing patient safety and product efficacy?
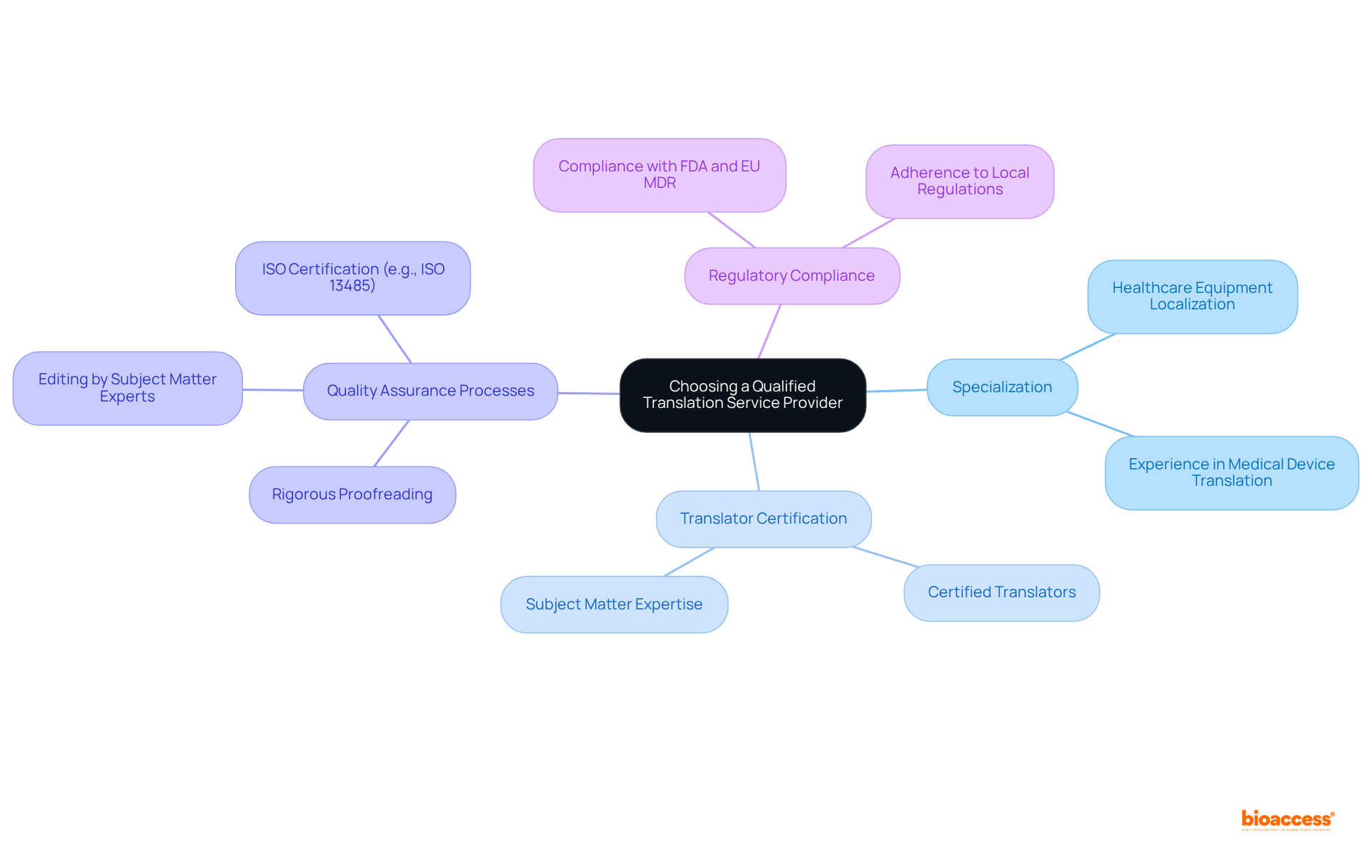
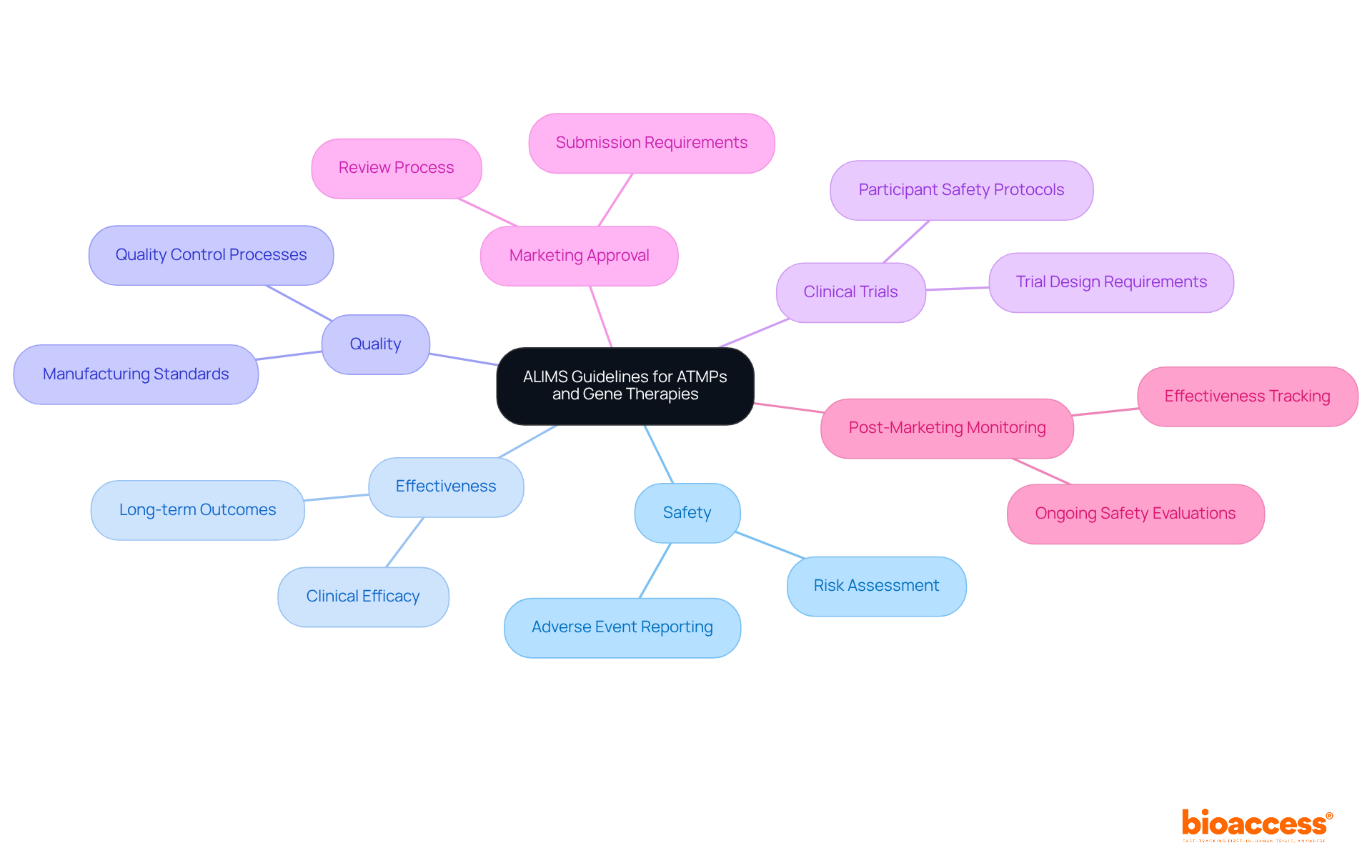
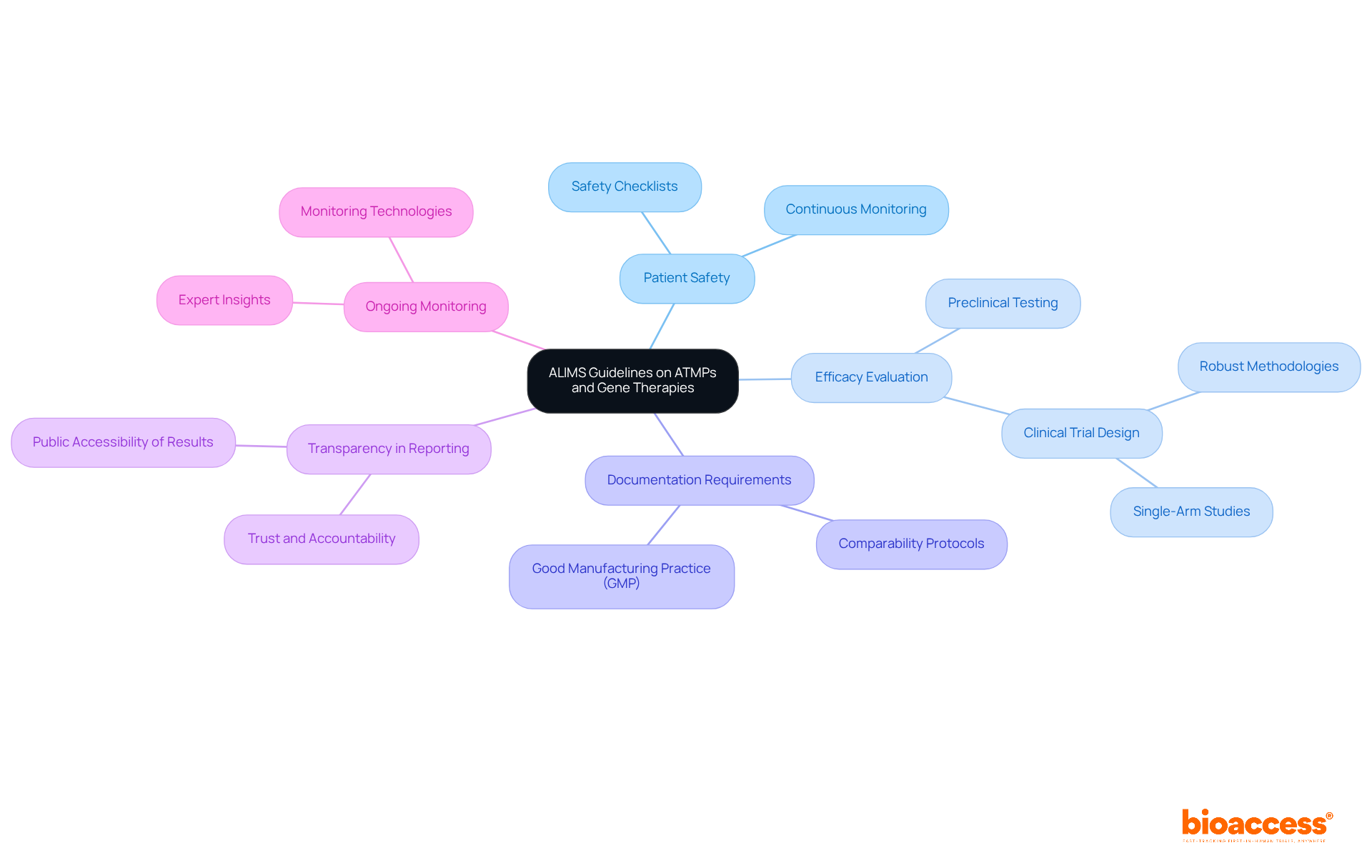
ISO 13485: Quality Management Systems for Medical Devices
ISO 13485 establishes essential requirements for a . This standard is crucial for ensuring that organizations consistently meet customer and compliance expectations. In Latin America, , which is in line with the of various countries in the region. For instance, in Colombia, INVIMA (Colombia National Food and Drug Surveillance Institute) plays a pivotal role. INVIMA supervises the marketing and production of , ensuring adherence to and granting approval for the import and export of products. Its designation as a Level 4 health authority by PAHO/WHO underscores its capability in regulating , making compliance with ISO 13485 even more essential for companies aiming to enter this lucrative market.
Valued at $11 billion and rapidly expanding, the in Latin America underscores the necessity of compliance with like ISO 13485. Recent updates to the standard emphasize a , which is critical for maintaining safety and efficacy. Organizations are now required to conduct regular and strengthen controls over suppliers and outsourced processes, reflecting the evolving compliance landscape. Effectively implementing ISO 13485 not only helps meet regulations but also enhances market appeal and consumer confidence, making it an indispensable element for companies in the healthcare sector.
As highlighted by Katherine Ruiz, a specialist in regulatory affairs for and in vitro diagnostics in Colombia, is often seen as the ‘gold standard’ for confirming safety and consistency in healthcare items. This certification not only aids in complying with local regulations, including those imposed by INVIMA, but also improves the overall quality and safety of , ultimately benefiting both patients and healthcare professionals.
ISO 14971: Risk Management for Medical Devices
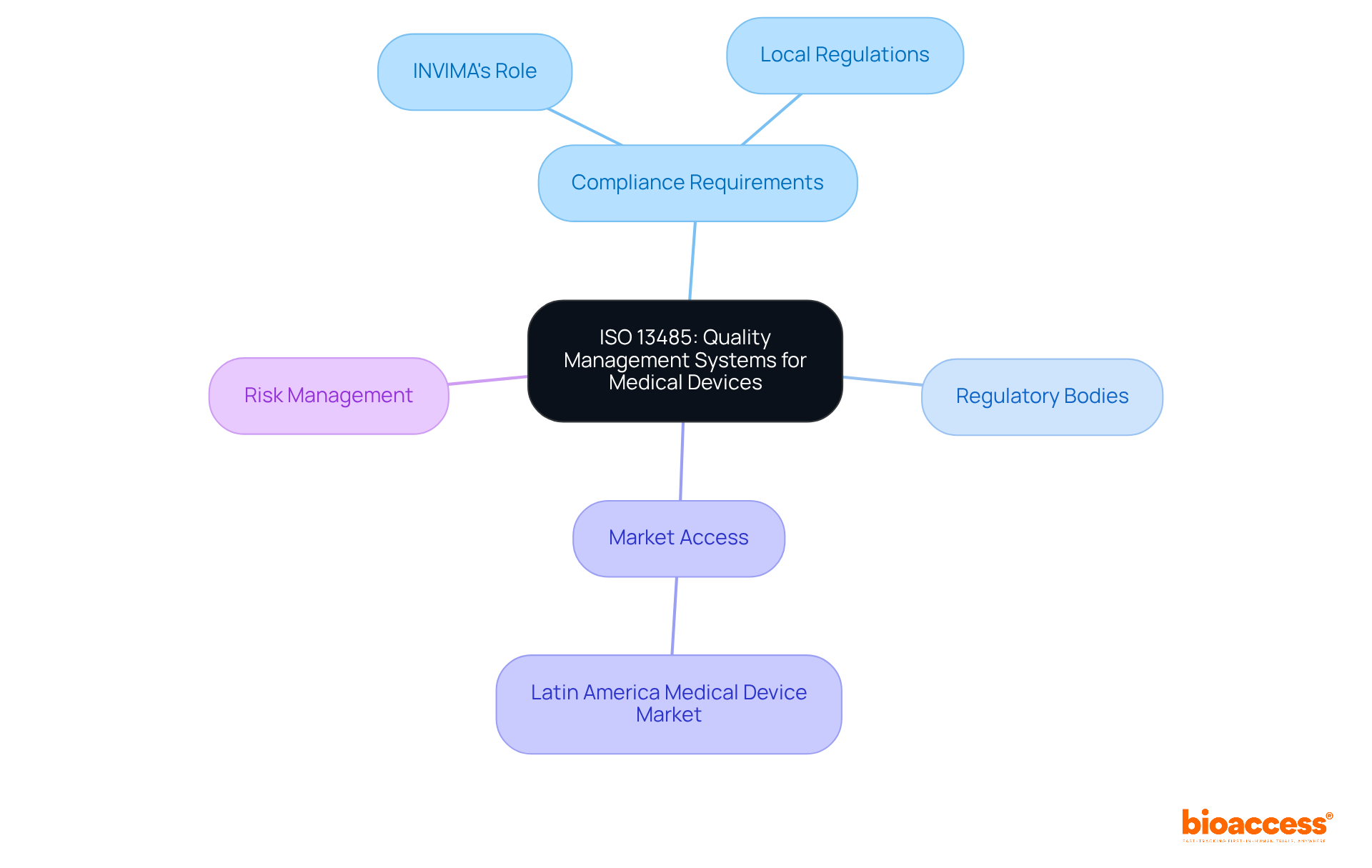
, focusing on the identification of hazards, thorough risk assessment, and the implementation of effective . This standard mandates that manufacturers meticulously document their , ensuring transparency and compliance with oversight authorities. By systematically evaluating and mitigating potential risks throughout the , ISO 14971 is essential in .
Recent developments in compliance underscore the urgent need for manufacturers to adopt robust strategies. Integrating methodologies such as Failure Modes and Effects Analysis (FMEA) and Hazard and Operability Studies (HAZOP) not only facilitates efficient risk evaluation but also aligns with . This guarantees that meet stringent safety criteria, which is crucial in today’s dynamic Medtech landscape.
As the regulatory environment for healthcare equipment continues to evolve, adherence to ISO 14971 is paramount for producers aiming to navigate the complexities of effectively. By embracing these standards, manufacturers can enhance their credibility and ensure that their products not only comply with regulations but also prioritize .
ASTM Standards: Shaping Medical Device Compliance
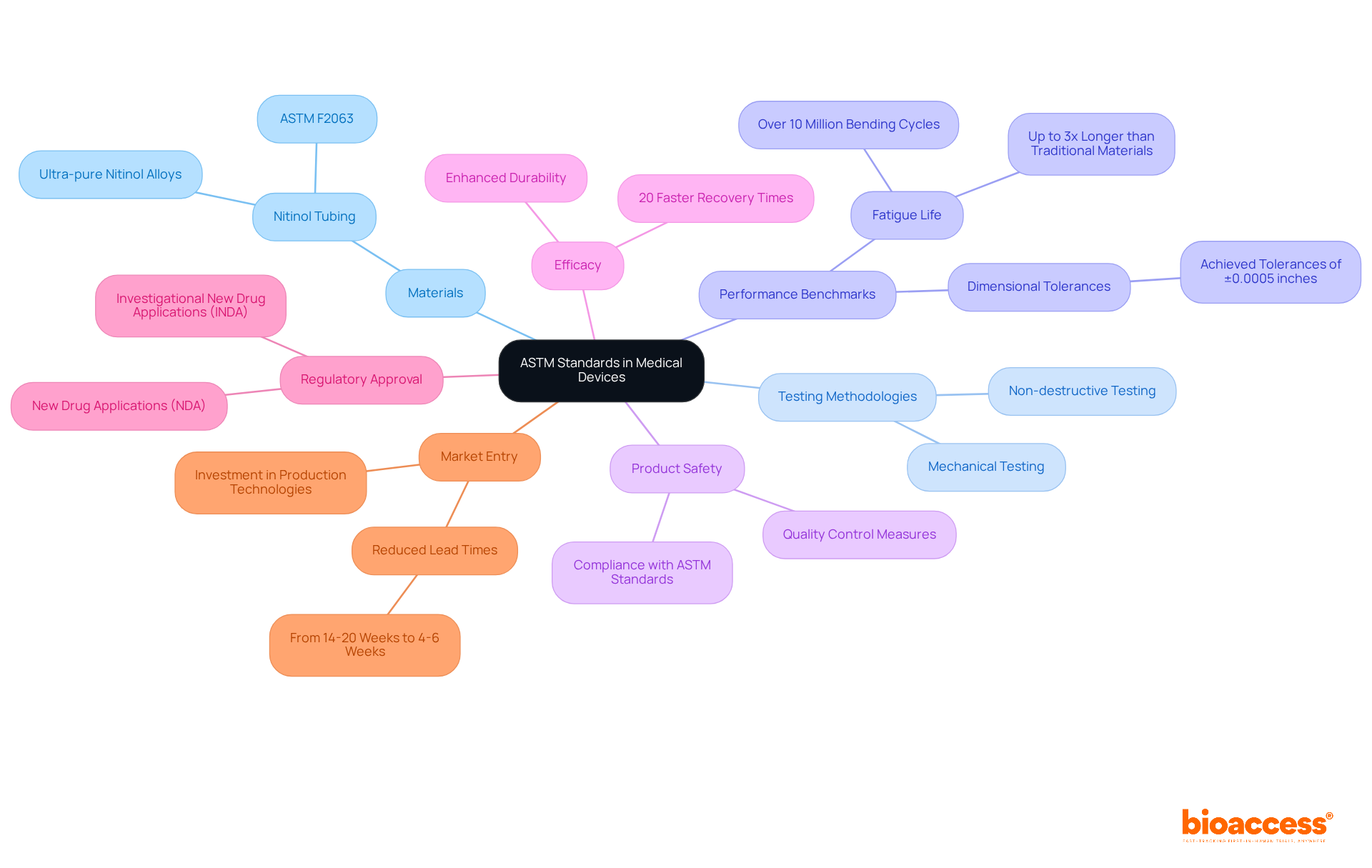
ASTM International plays a pivotal role in shaping voluntary consensus standards that govern critical aspects of , such as materials, testing methodologies, and performance benchmarks. By adhering to these standards, manufacturers significantly enhance , paving a smoother path to . For instance, the serves as a cornerstone for nitinol tubing, ensuring that products not only meet stringent safety requirements but also encourage innovation in healthcare applications.
As producers invest in , not only boosts the reliability of healthcare products but also accelerates their market entry. Staying informed about the is essential for companies aiming to meet compliance obligations and maintain a of healthcare product development.
How prepared is your organization to navigate these standards and leverage them for success?
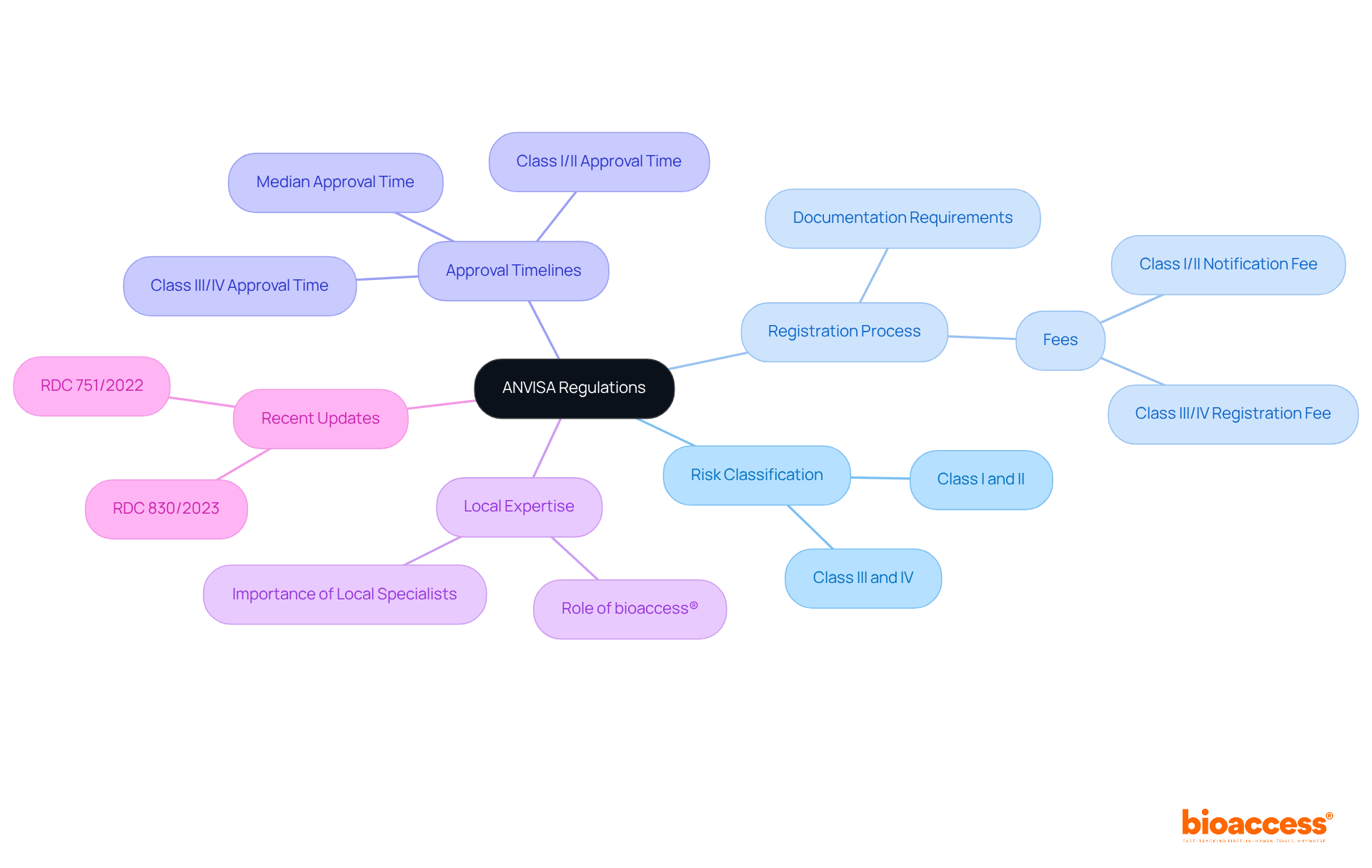
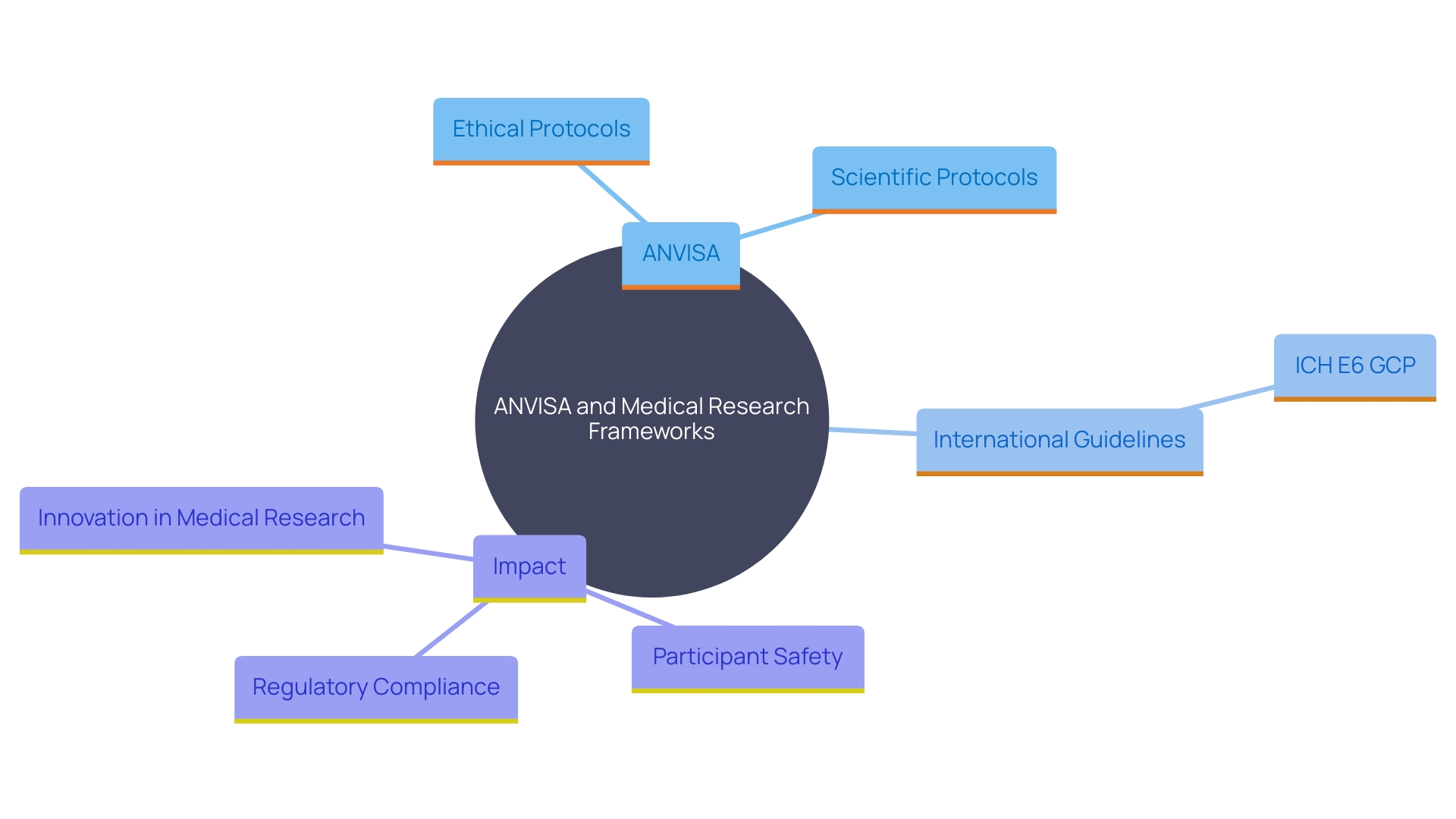
ANVISA Regulations: Navigating Brazil’s Medical Device Market
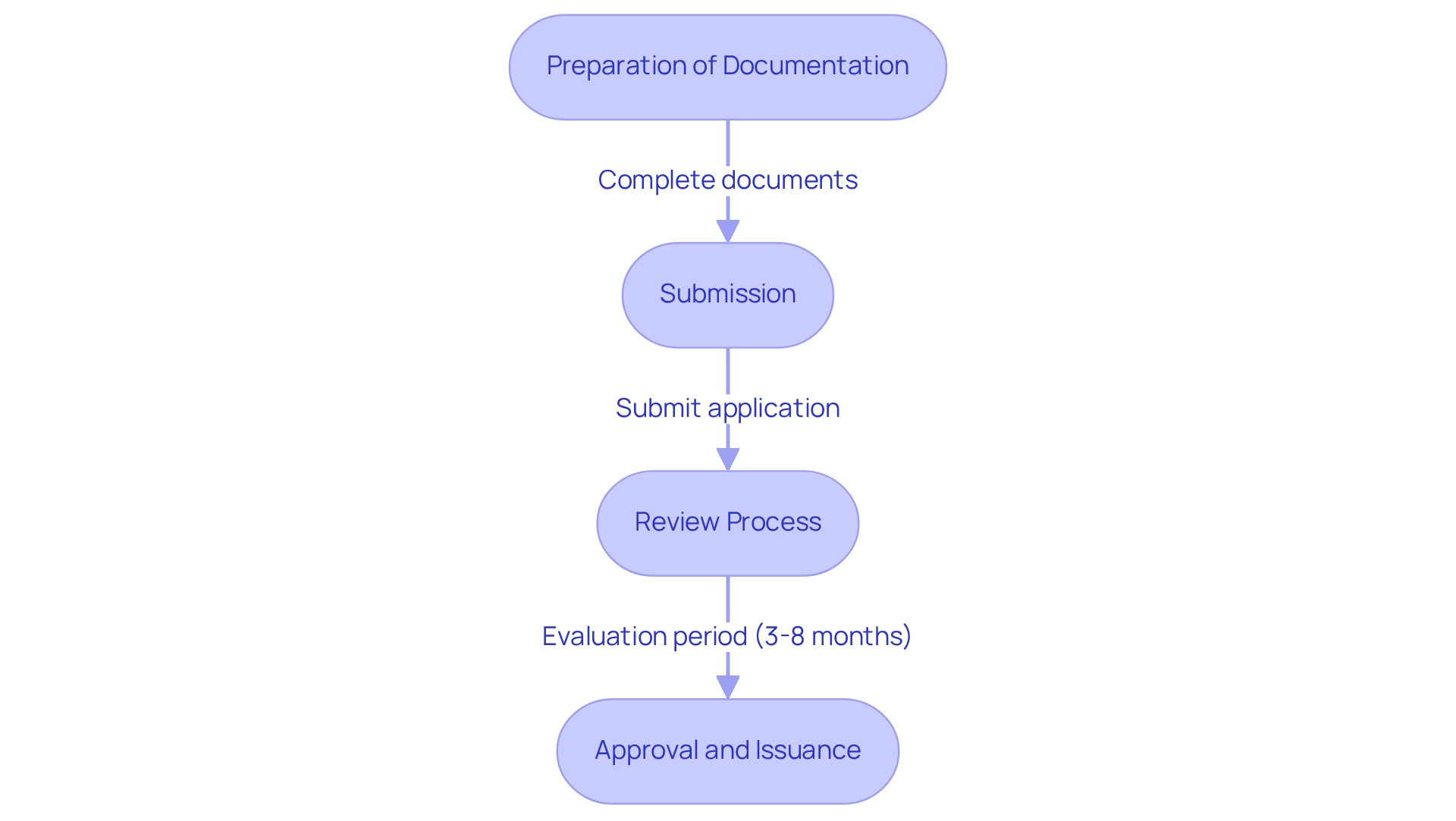
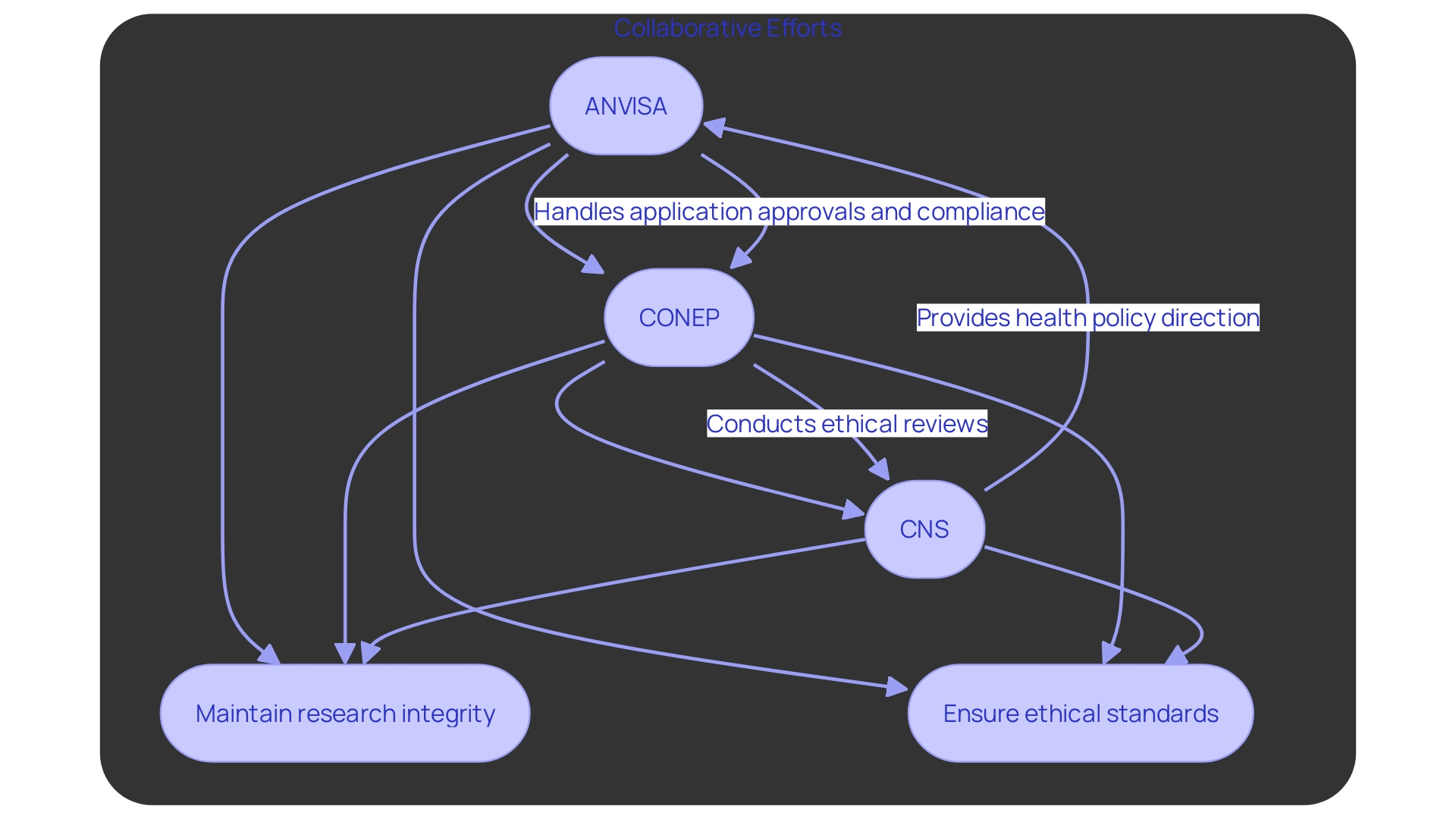
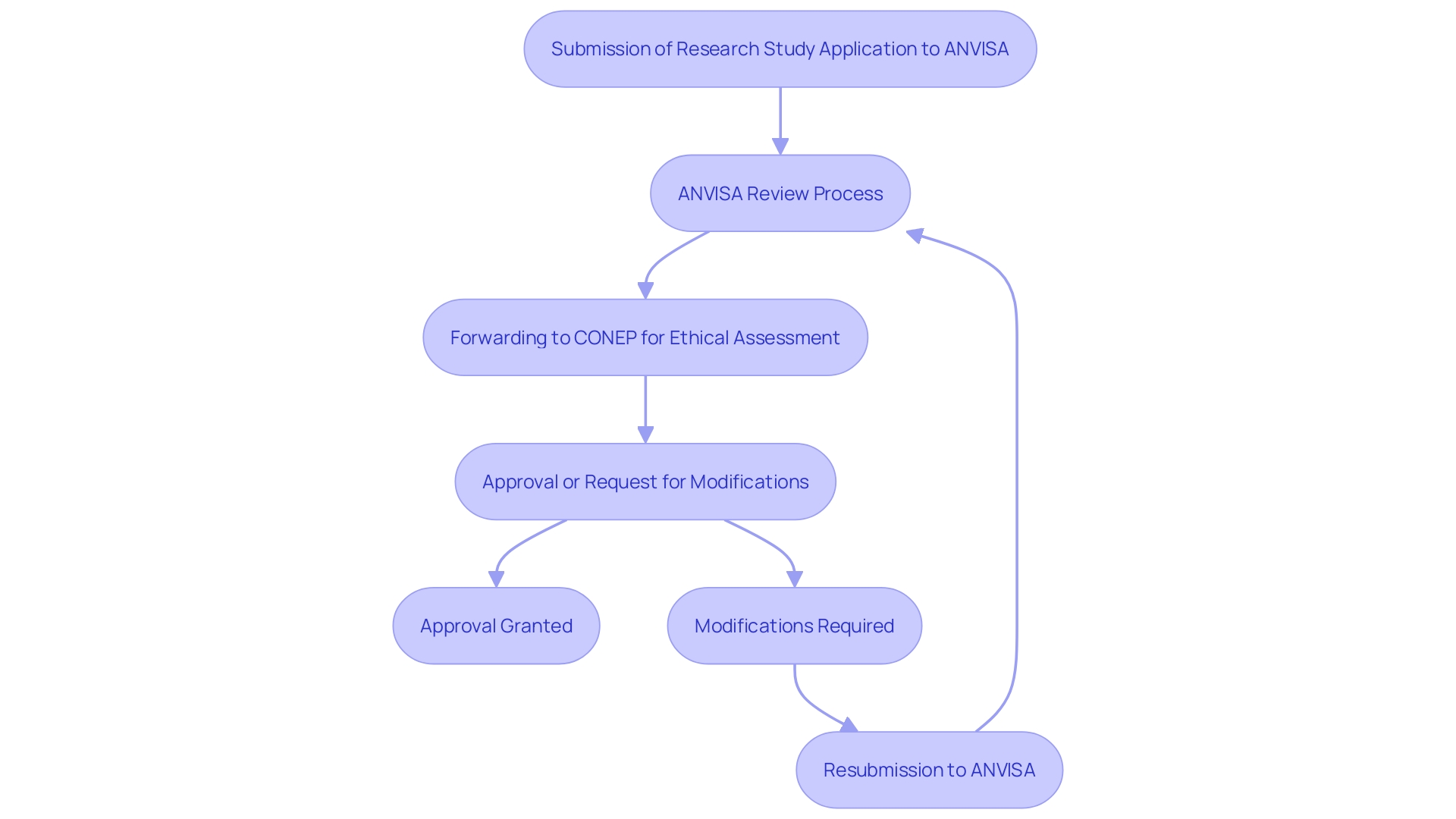
(National Health Surveillance Agency) plays a crucial role in regulating medical instruments in Brazil under the guidelines of . This agency enforces a comprehensive framework that encompasses risk classification, registration, and ongoing . The approval process can be quite extensive; for instance, average timelines for Class III and IV items typically range from 4 to 12 months, with the overall median approval time for products being 795 days.
To ensure , manufacturers must have a solid grasp of and prepare thorough documentation. Engaging with local specialists, such as the team at , which boasts over 20 years of experience in , can significantly enhance . Their insights into navigating the complexities of the compliance environment are invaluable. Recent updates, including and streamlined processes outlined in RDC 830/2023, underscore the necessity of staying informed about the and evolving regulations.
By adopting a proactive approach and leveraging local expertise from , companies can effectively navigate Brazil’s and ensure compliance with ‘s standards. Collaboration with experienced partners is not just beneficial; it’s essential for success in this dynamic landscape.
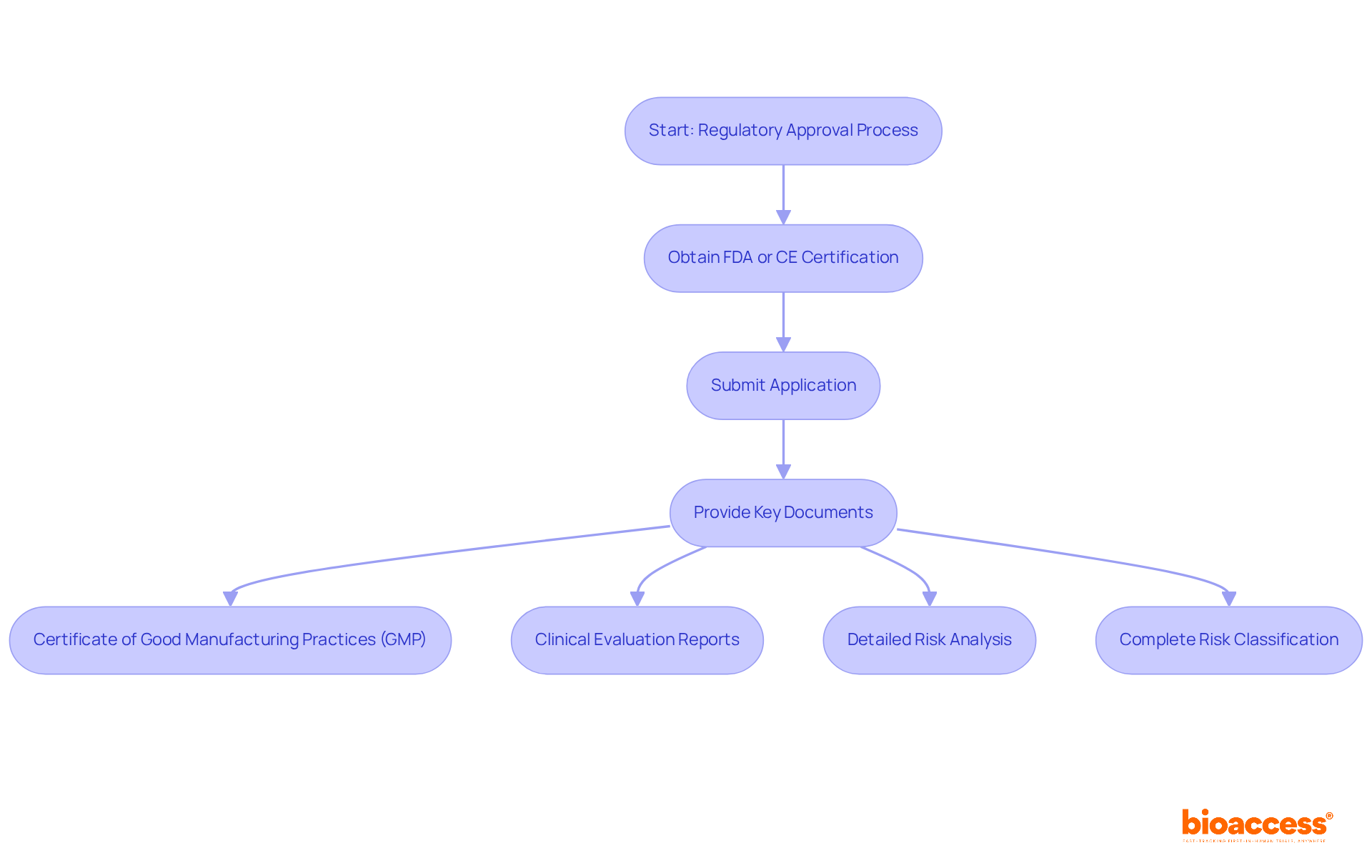
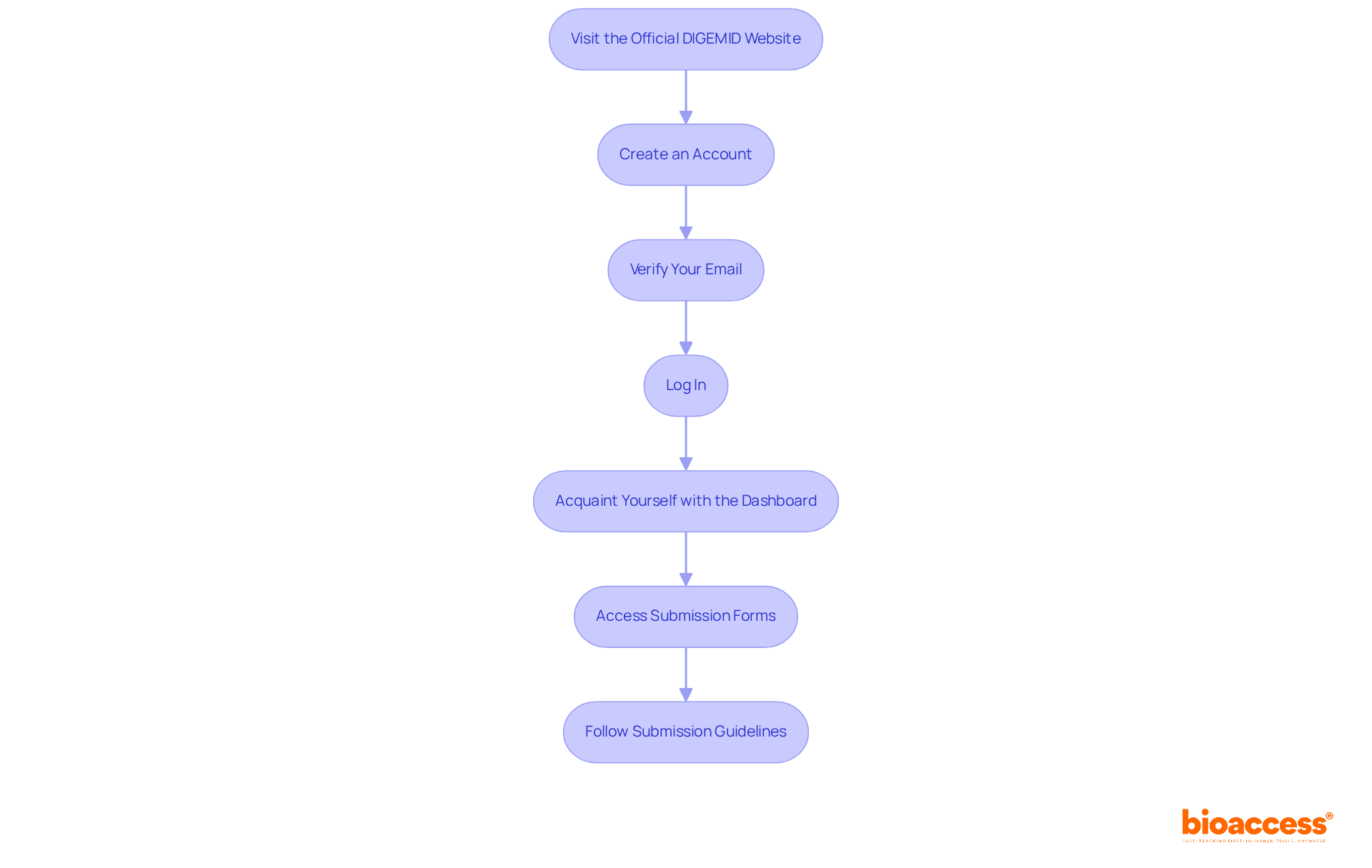
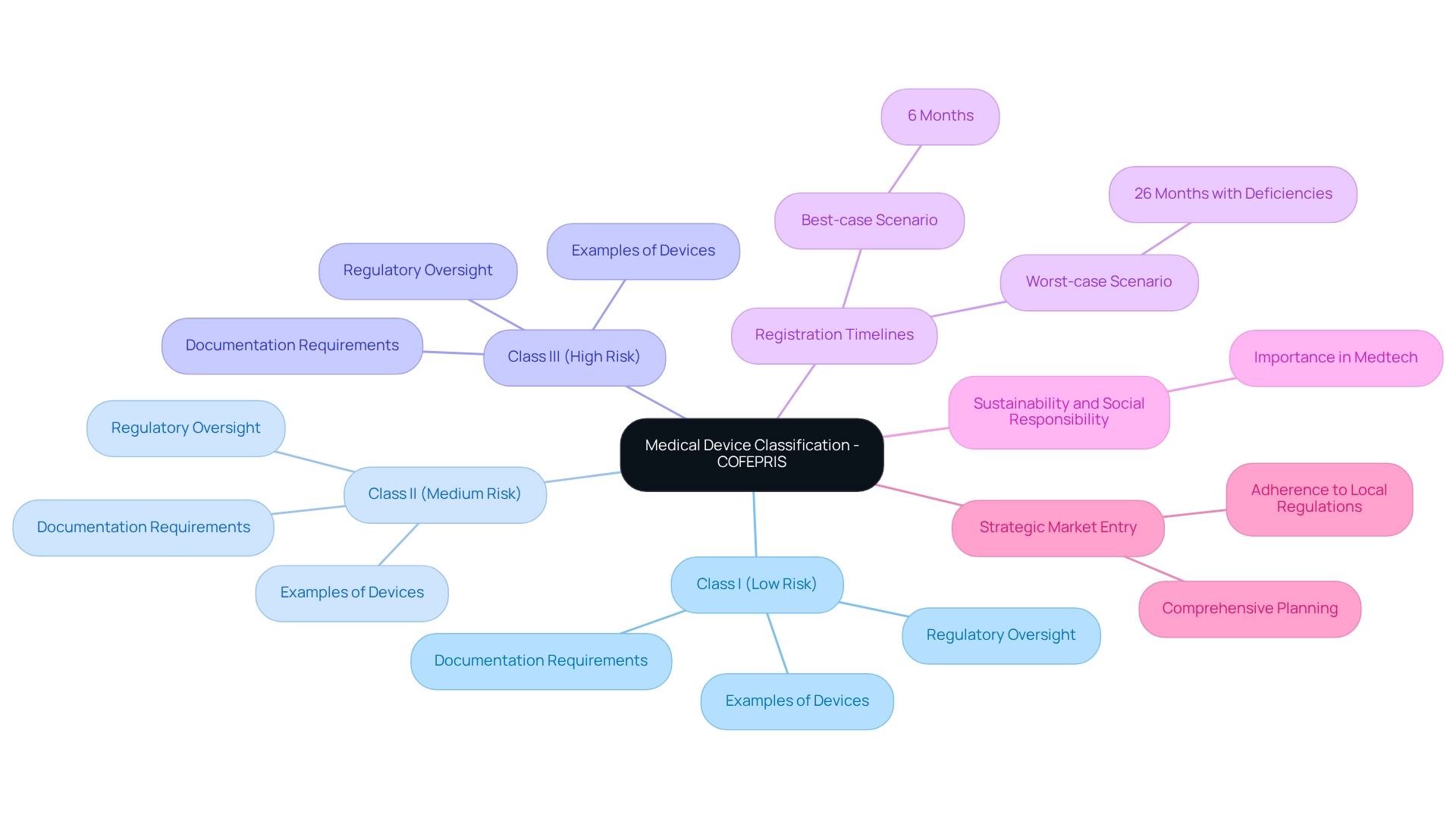
Mexico’s Regulatory Approval Process: Key Considerations
COFEPRIS, Mexico’s oversight agency, has introduced an that possess FDA or CE certification. This initiative not only aims to attract foreign manufacturers but also facilitates quicker , a crucial factor in today’s competitive Medtech landscape. However, , which includes thorough and .
Key documents such as:
- A
- A detailed risk analysis
are essential for compliance. Understanding the nuances of is vital for in Mexico, as they can significantly influence approval timelines and market entry strategies. Are you prepared to tackle these challenges and seize the opportunities presented by this ?
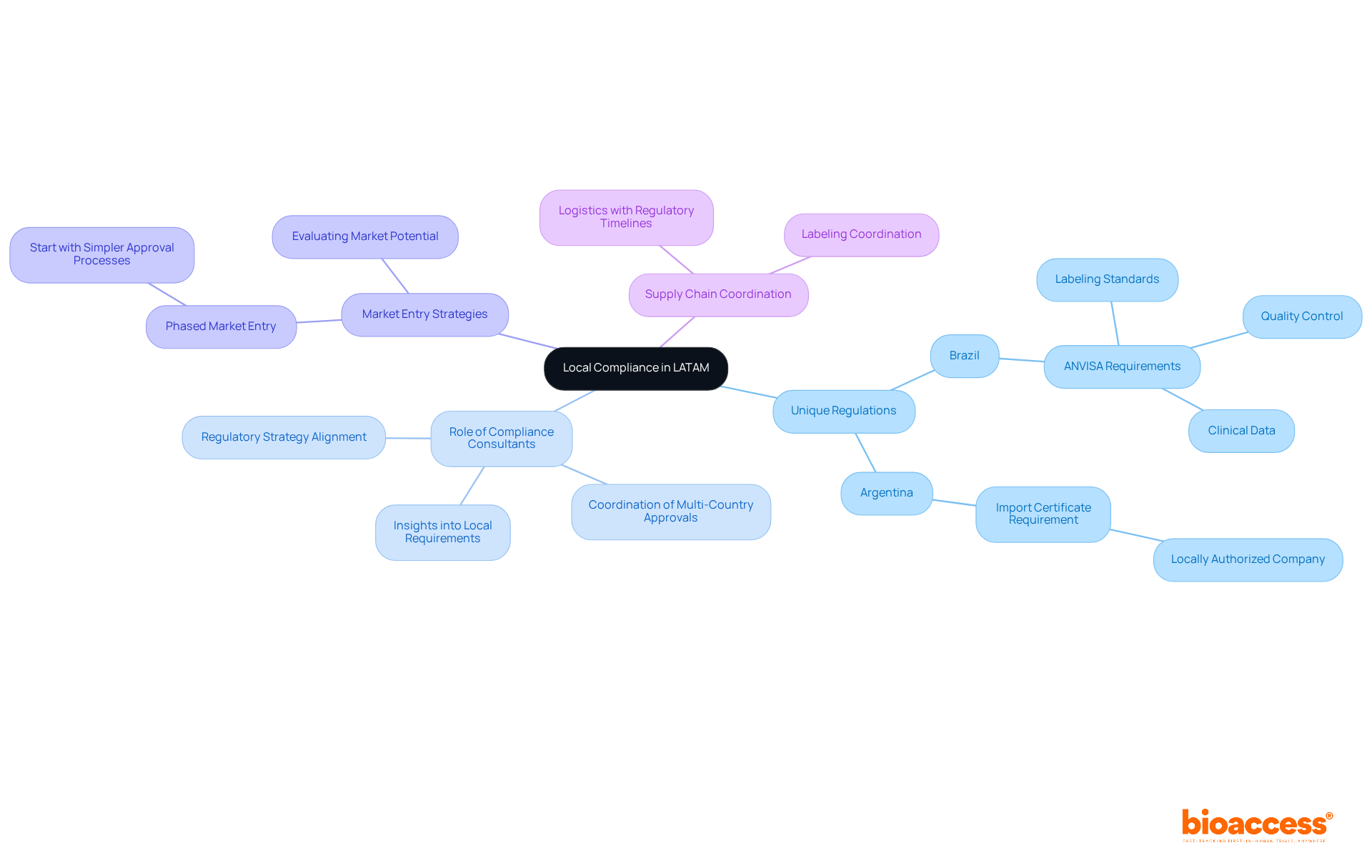
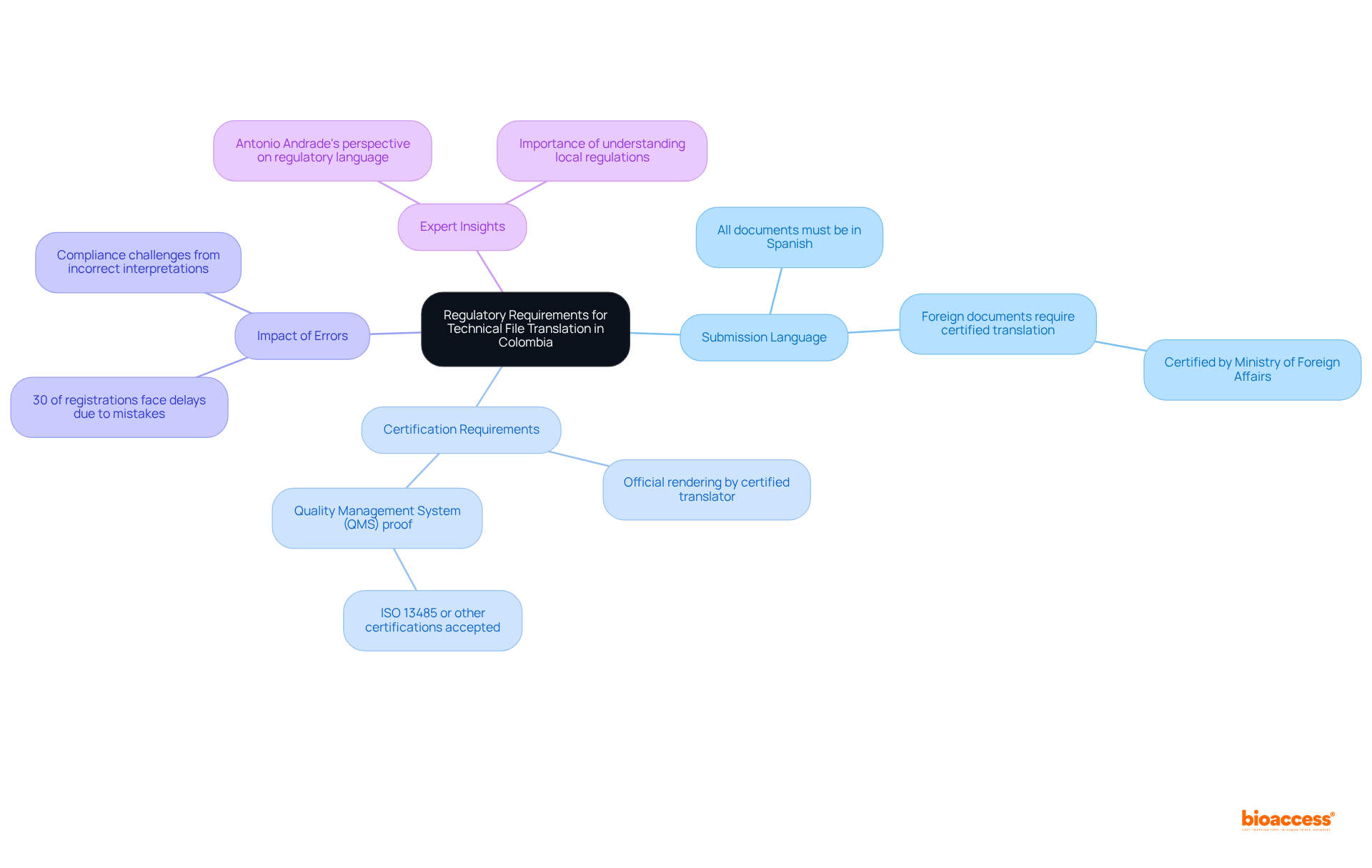
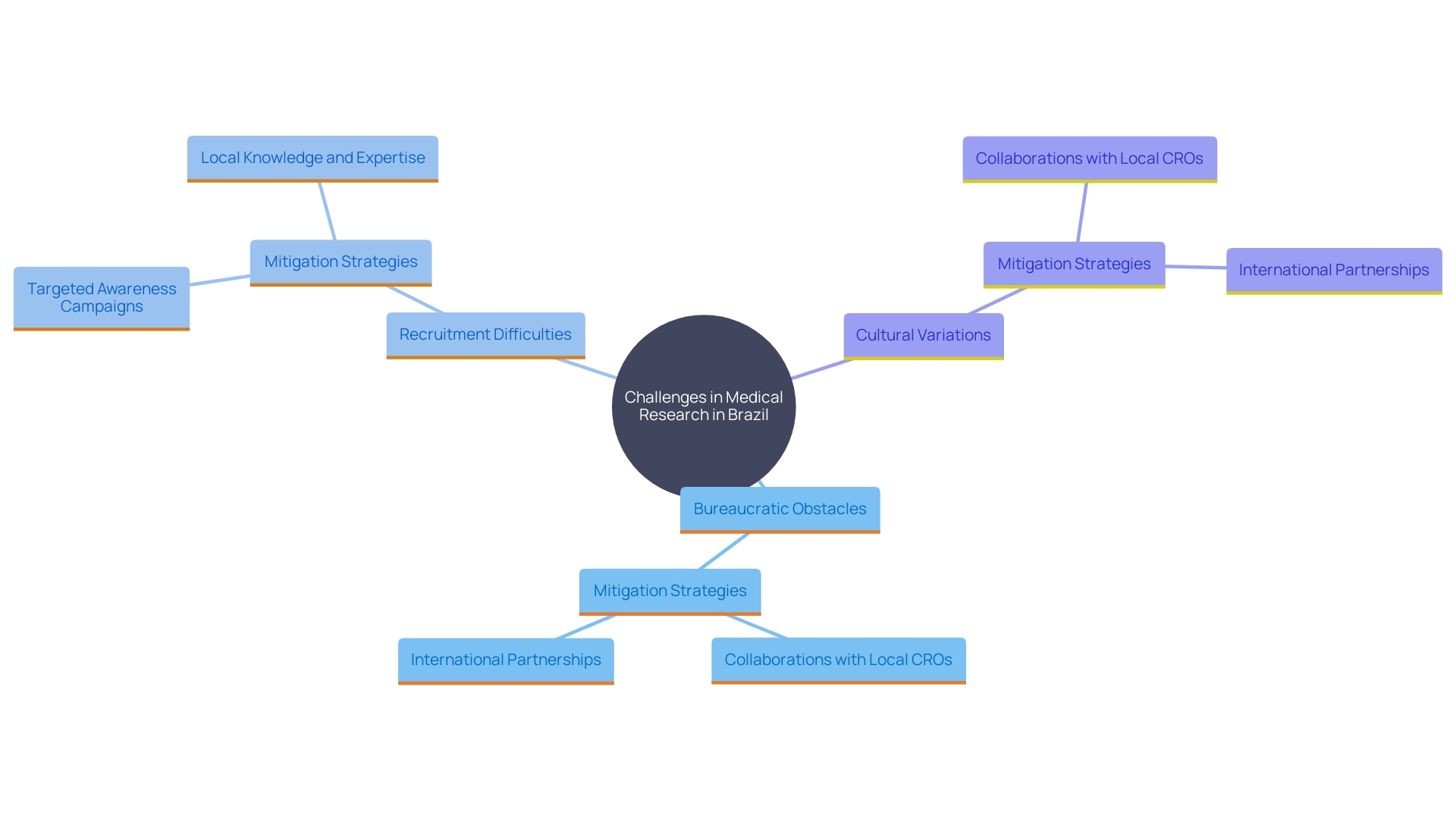
Local Compliance: Understanding Diverse LATAM Regulations
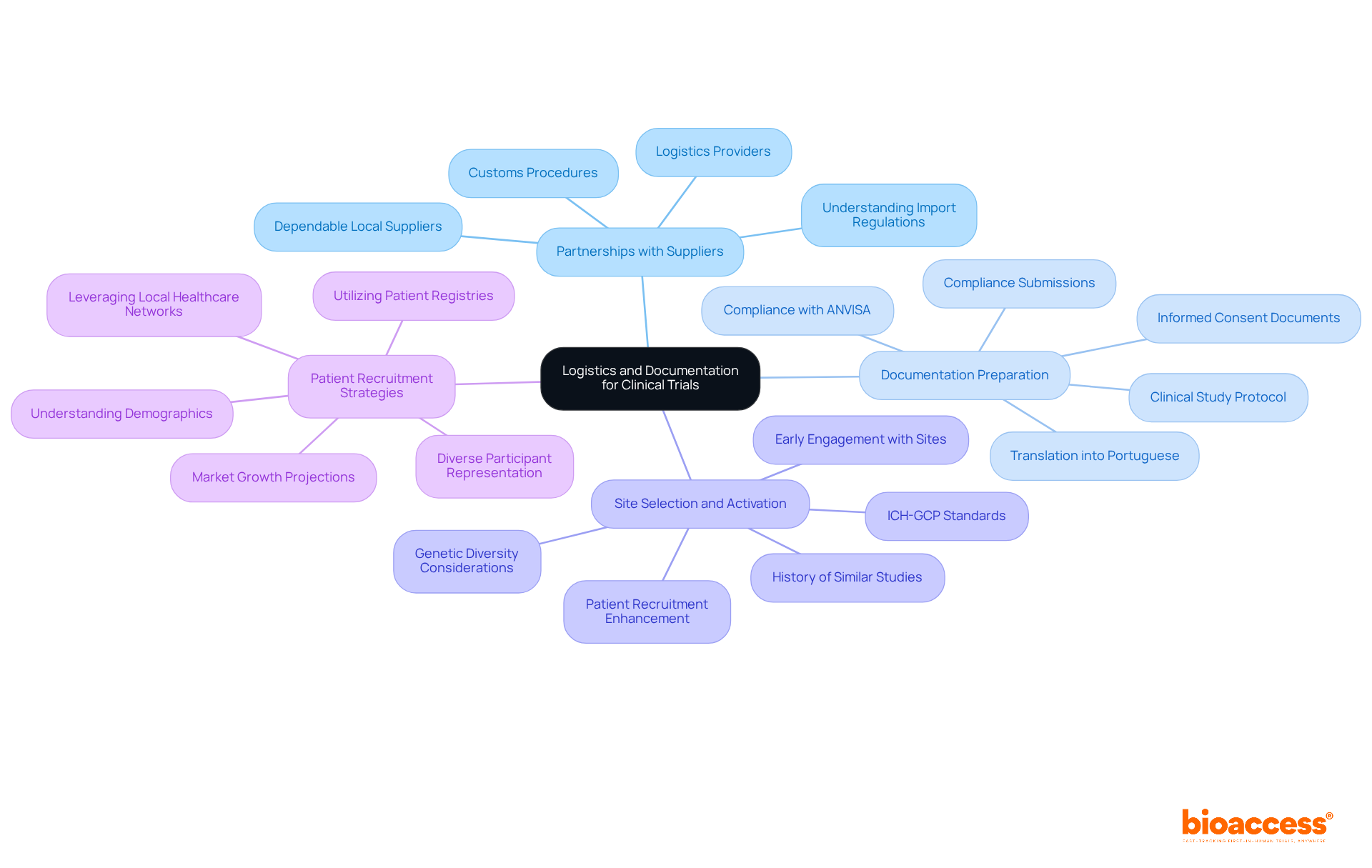
Navigating the compliance environment in is crucial for healthcare equipment producers. Each nation presents unique authorities and adherence criteria that must be understood for successful . Understanding , including , , and , is essential for compliance with . Recent statistics reveal that a significant percentage of medical device manufacturers lack familiarity with the , highlighting the urgent need for .
To effectively tackle these complexities, engaging is vital. These experts offer insights into specific requirements, such as , as well as Argentina’s stipulation that . Aligning compliance strategies with business objectives not only maximizes market efficiency but also enhances impact.
Companies should consider a phased , beginning with countries that have simpler approval processes. This strategy allows for a more manageable transition into the diverse legal landscape of Latin America. Furthermore, coordinating supply chain logistics with compliance timelines is essential to avoid delays and ensure adherence. By leveraging local knowledge and conducting thorough market analysis, manufacturers can successfully navigate the legal environment and seize the significant opportunities within Latin America’s multi-billion dollar healthcare market, particularly by complying with .
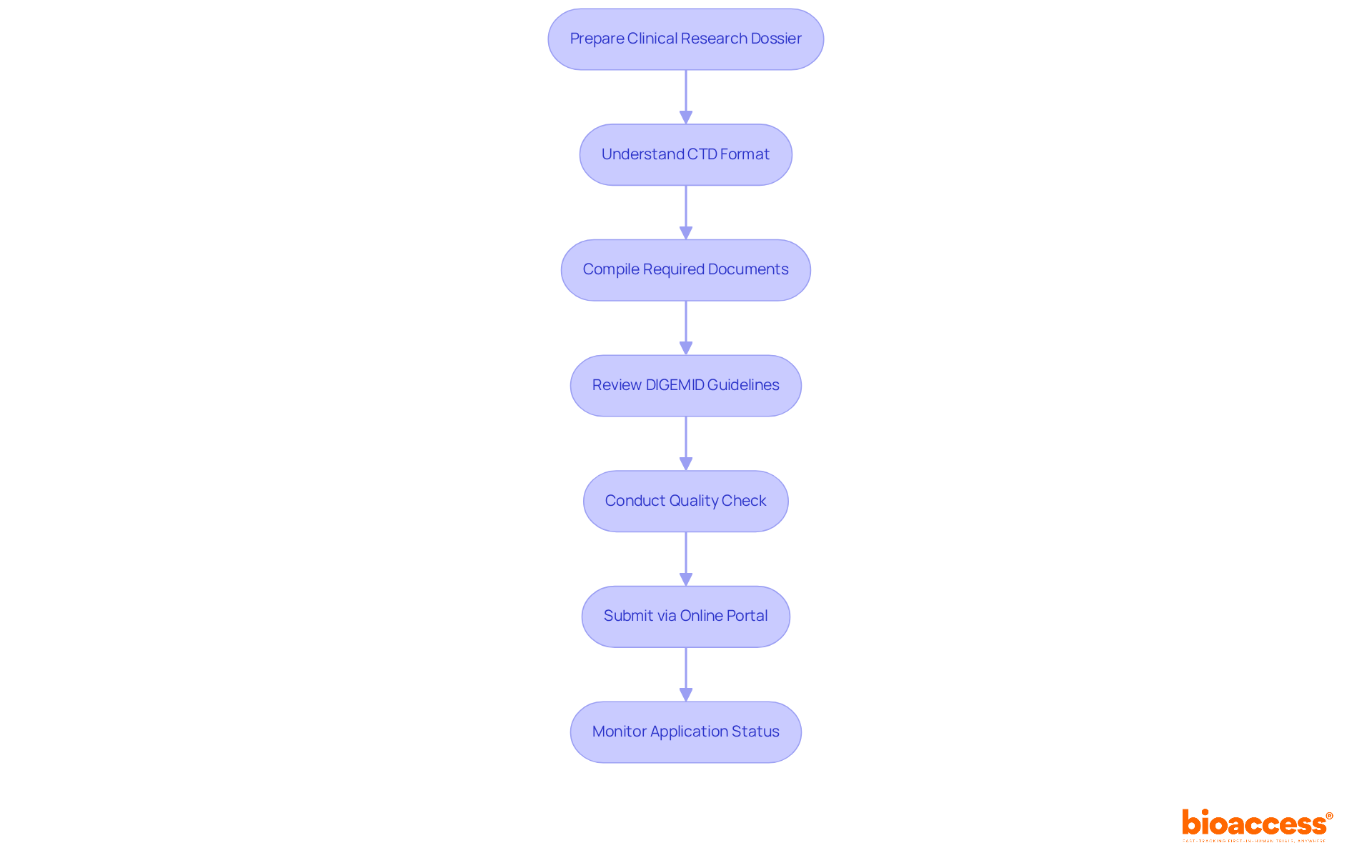
Clinical Trial Regulations: Essential for LATAM Research
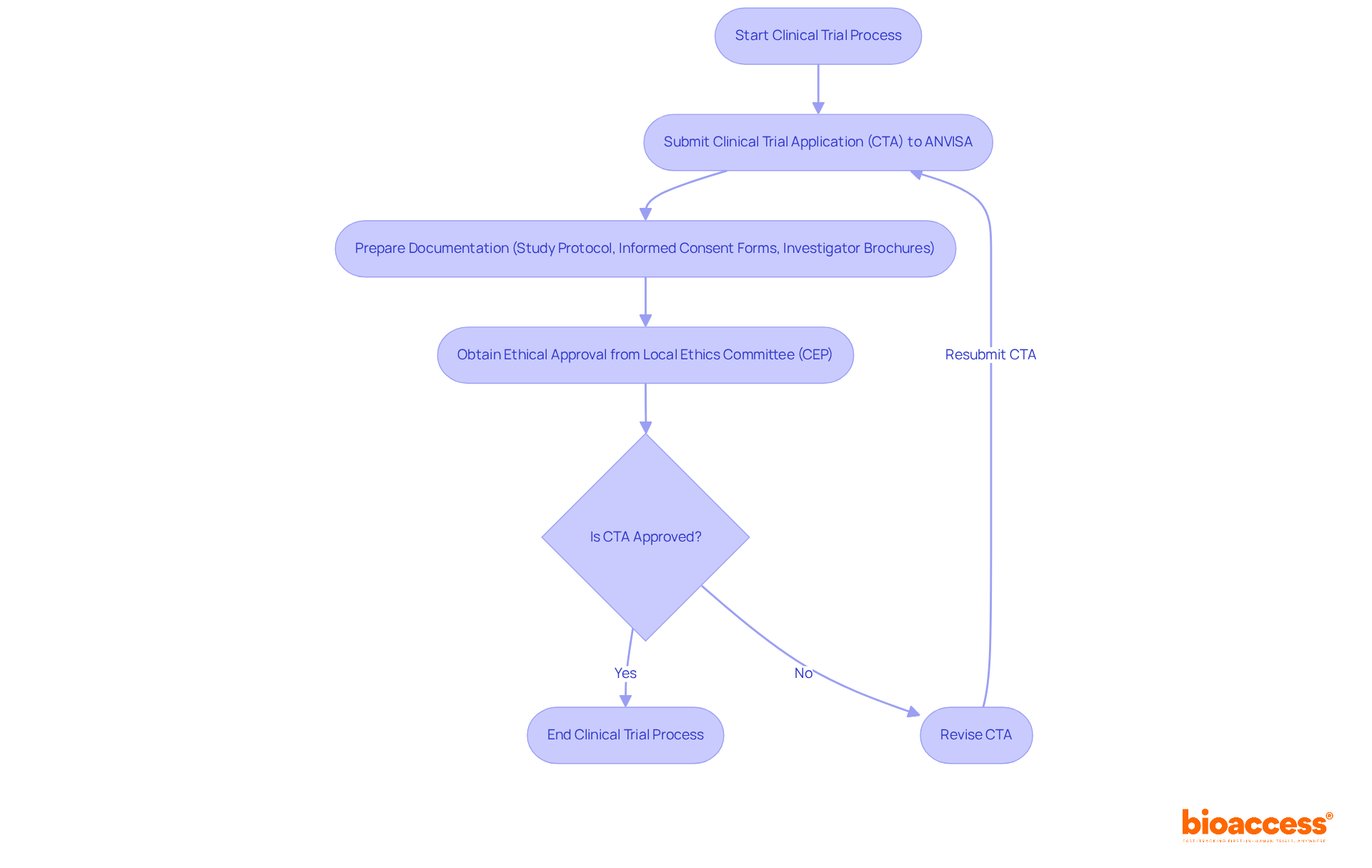
exhibit , yet they universally mandate adherence to ethical standards, informed consent, and thorough reporting. Researchers must present detailed protocols to local ethics committees and oversight authorities for approval, a process that can take an average of 90-120 days in Colombia and 3-4 months in Chile. Understanding these regulations is crucial for , ensuring participant safety, and maintaining data integrity.
Colombia stands out in the Latin American landscape due to its recent advancements in the legal framework, including that have increased funding from $3-4 million to over $50 million annually. This positions the country as a favorable location for , offering . Moreover, the continuous digital transformation of governance frameworks in Latin America is anticipated to streamline the navigation of compliance requirements, improving operational efficiency and transparency for researchers and participants alike.
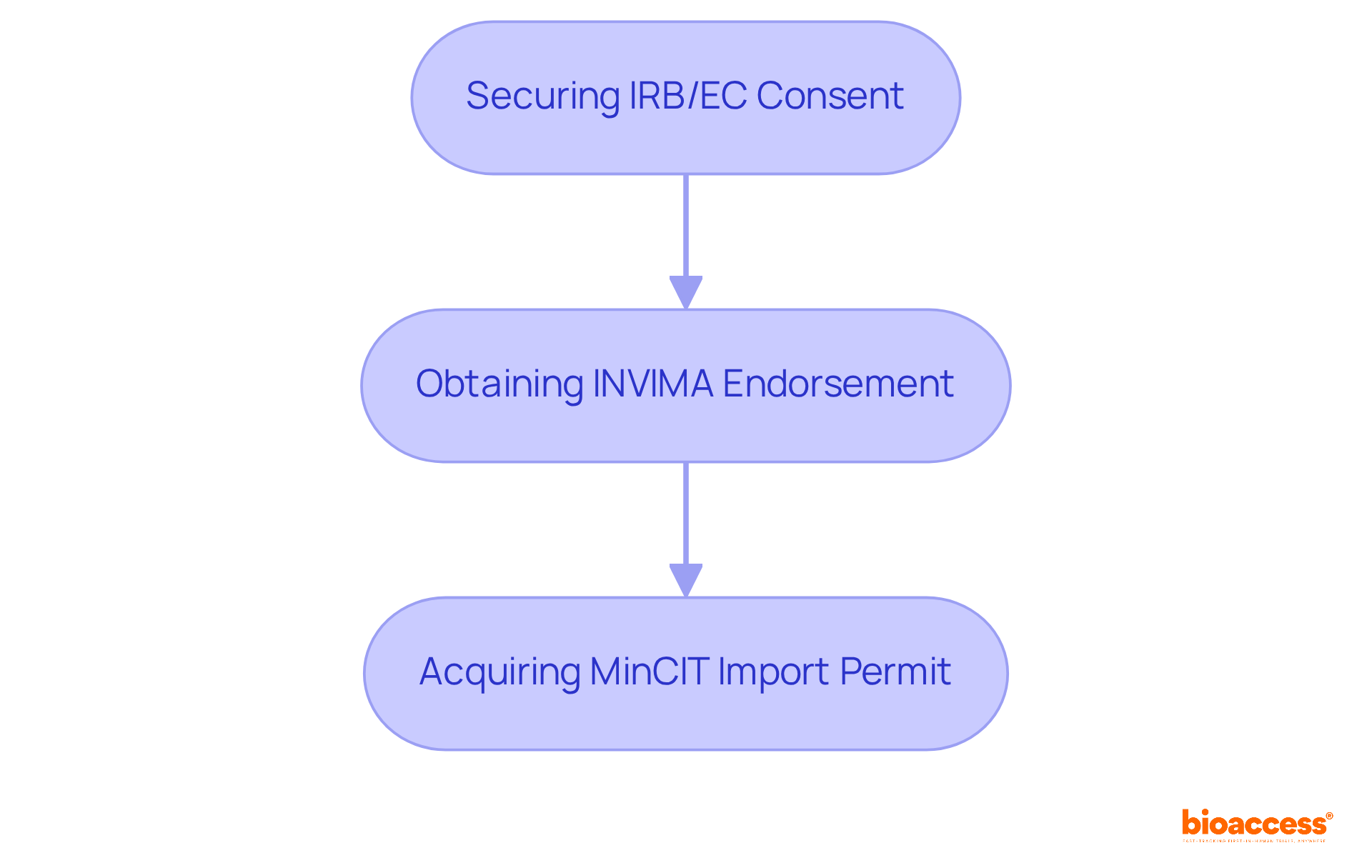
The process for obtaining involves several key steps:
- Securing IRB/EC consent
- Obtaining INVIMA endorsement
- Acquiring a MinCIT import permit
, with requiring careful consideration of cultural and linguistic differences among diverse populations. For instance, in Colombia, the formal and informal ‘you’ forms in Spanish can influence communication with participants, necessitating culturally sensitive approaches to ensure understanding and compliance. As the region continues to evolve, maintaining high ethical standards will be essential for fostering trust and ensuring the integrity of clinical trials. Furthermore, bioaccess® provides specialized services to assist these processes, ensuring a more straightforward route to compliance and successful trial execution.
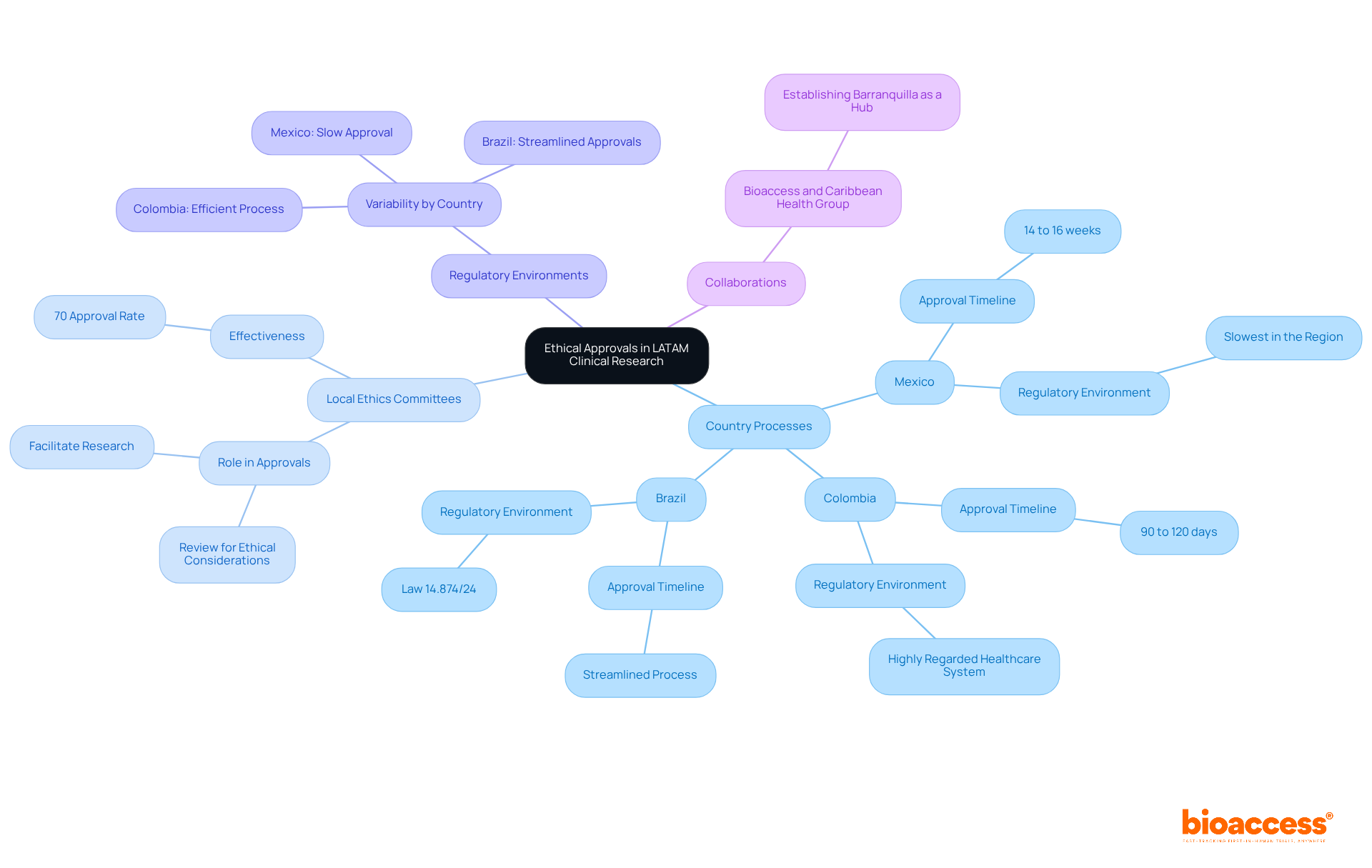
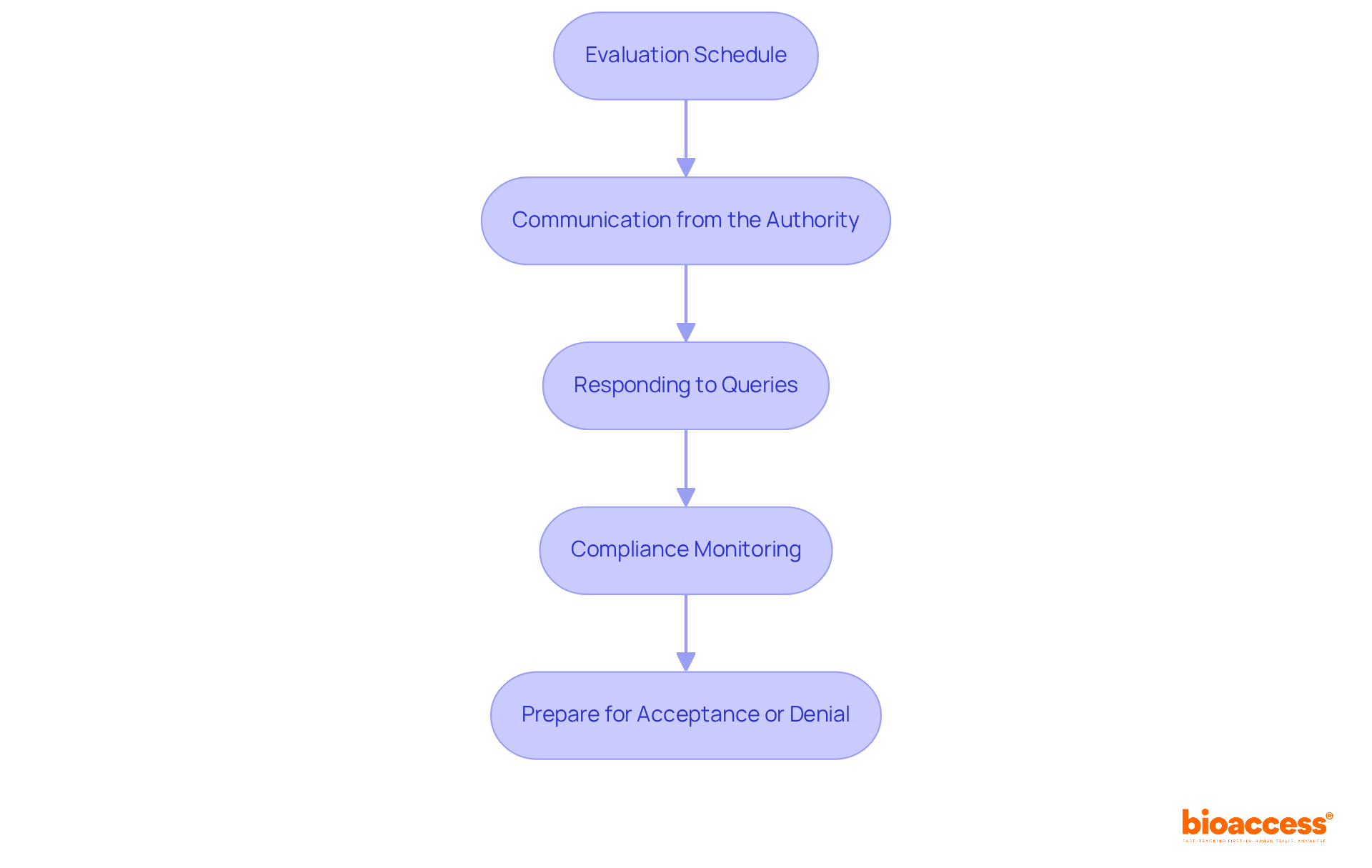
Ethical Approvals: Navigating LATAM Clinical Research
Obtaining ethical consent is a critical step in conducting that comply with . Researchers must submit their study protocols to , which play a vital role in reviewing proposed research for ethical considerations and participant safety. The authorization process varies significantly among nations; for instance, , while Colombia generally completes its review in 90 to 120 days, making it a more efficient choice for trial initiation. is highly regarded, with hospitals recognized for their quality, further enhancing its appeal for conducting .
Recent updates indicate that , allowing to independently endorse protocols. This change is expected to reduce authorization times significantly. In contrast, Mexico’s regulatory environment remains one of the slowest in the region, underscoring the need for when approaching there.
Statistics reveal that approximately 70% of in Latin America successfully obtain , highlighting the effectiveness of in facilitating research. Collaborations, such as those between bioaccess® and Caribbean Health Group, aim to establish Barranquilla as a hub for medical studies, further improving recruitment efficiency and compliance.
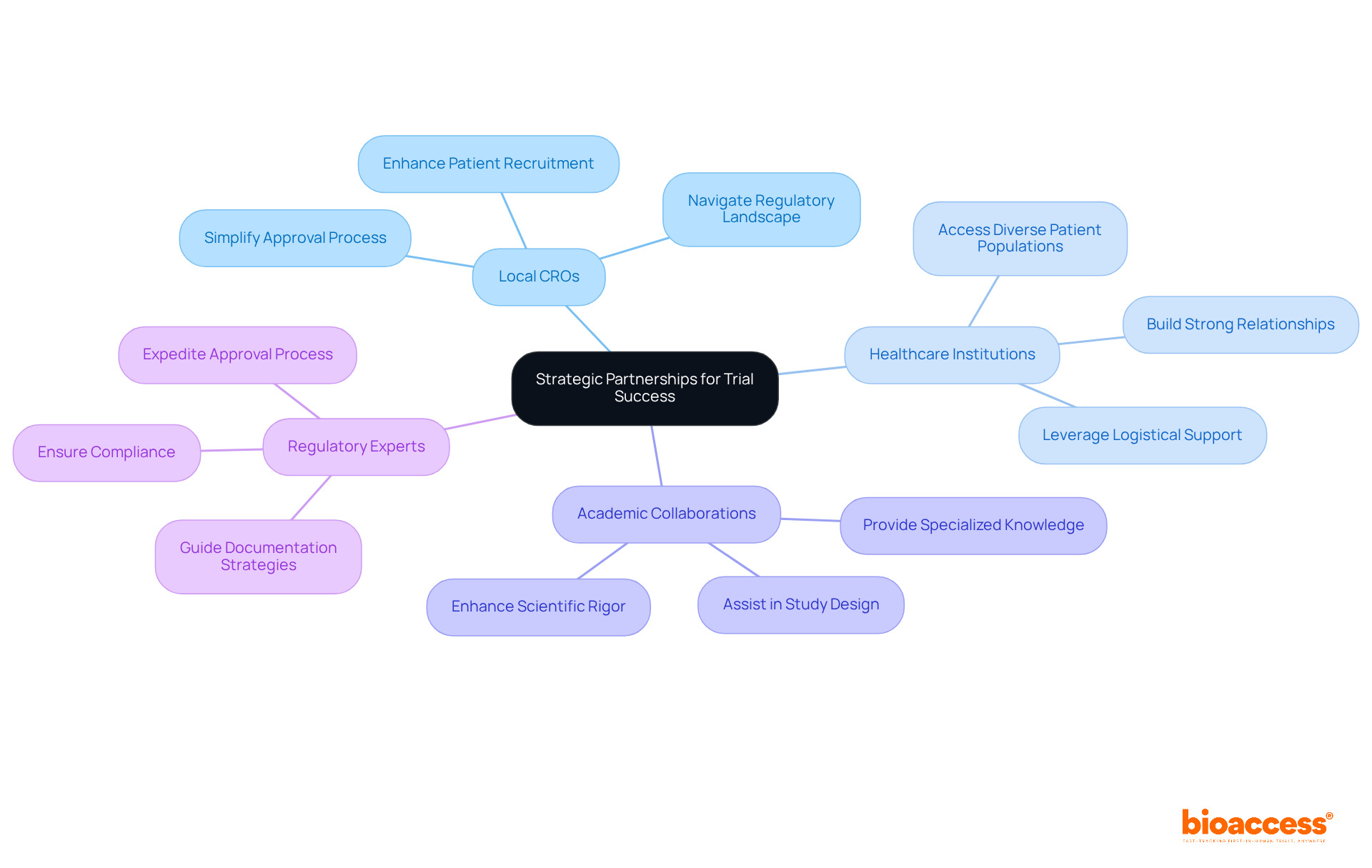
Navigating these complexities requires researchers to engage with local experts who understand the nuances of the ethical review process and the in each country. This localized approach not only ensures adherence to standards but also with participants, ultimately leading to more successful .
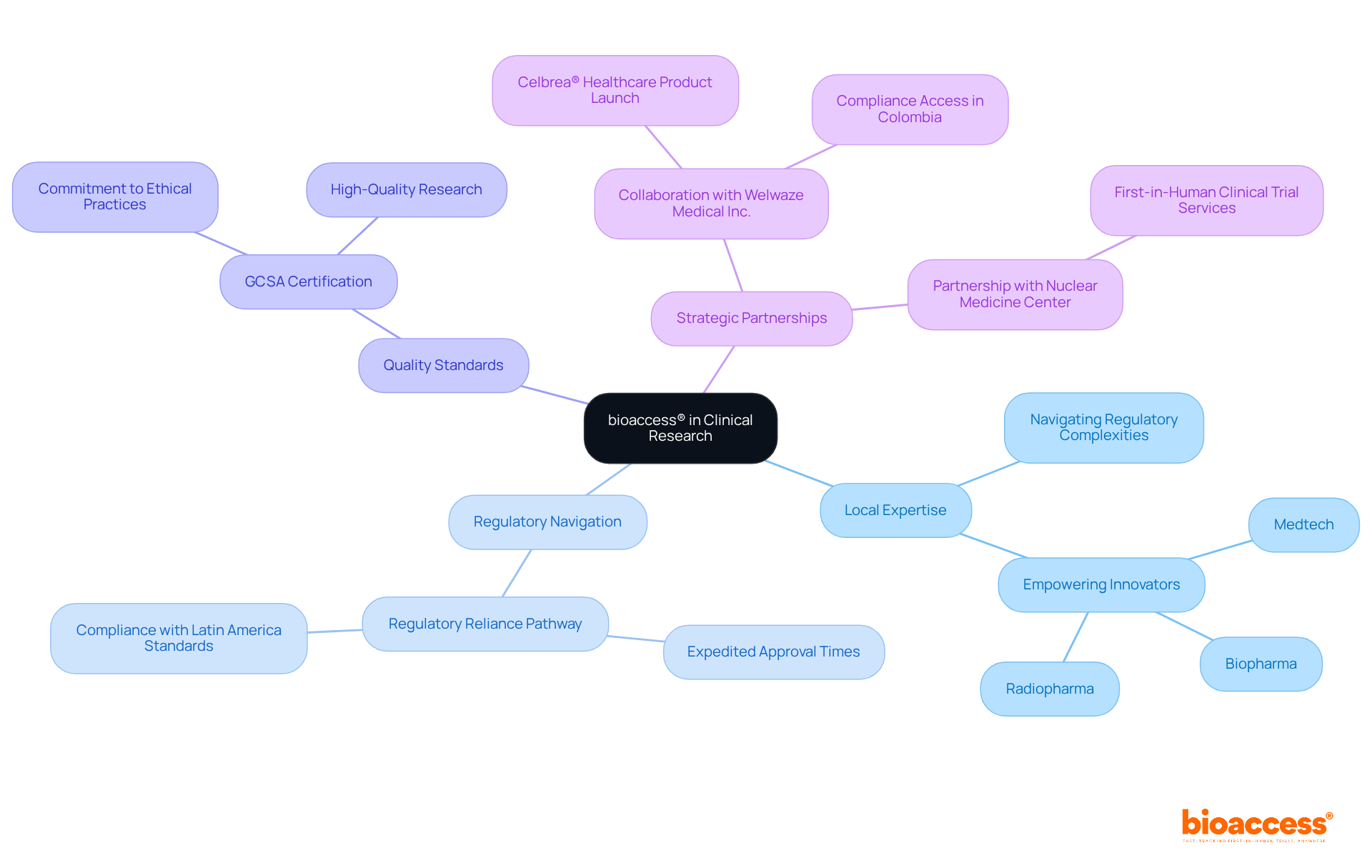
bioaccess: Accelerating Clinical Research and Compliance in LATAM
bioaccess® stands out in delivering comprehensive across Latin America, leveraging local expertise to . By concentrating on early-phase research, bioaccess® empowers Medtech, Biopharma, and Radiopharma innovators to secure quicker authorizations and enhance patient enrollment. Their deep understanding of regional regulations, particularly the newly introduced ‘Regulatory Reliance’ pathway by COFEPRIS, establishes them as a vital partner for companies looking to excel in the LATAM market while complying with . This pathway significantly , streamlining processes for clients.
Moreover, bioaccess® adheres to quality standards such as the , underscoring their commitment to ethical practices and high-quality research. Recent collaborations, including a strategic partnership with Welwaze Medical Inc. for the Celbrea® healthcare product launch and compliance access in Colombia, highlight bioaccess®’s dedication to advancing clinical research and ensuring adherence. This ultimately for . As the number of continues to grow, bioaccess® remains at the forefront, providing invaluable support to its clients.
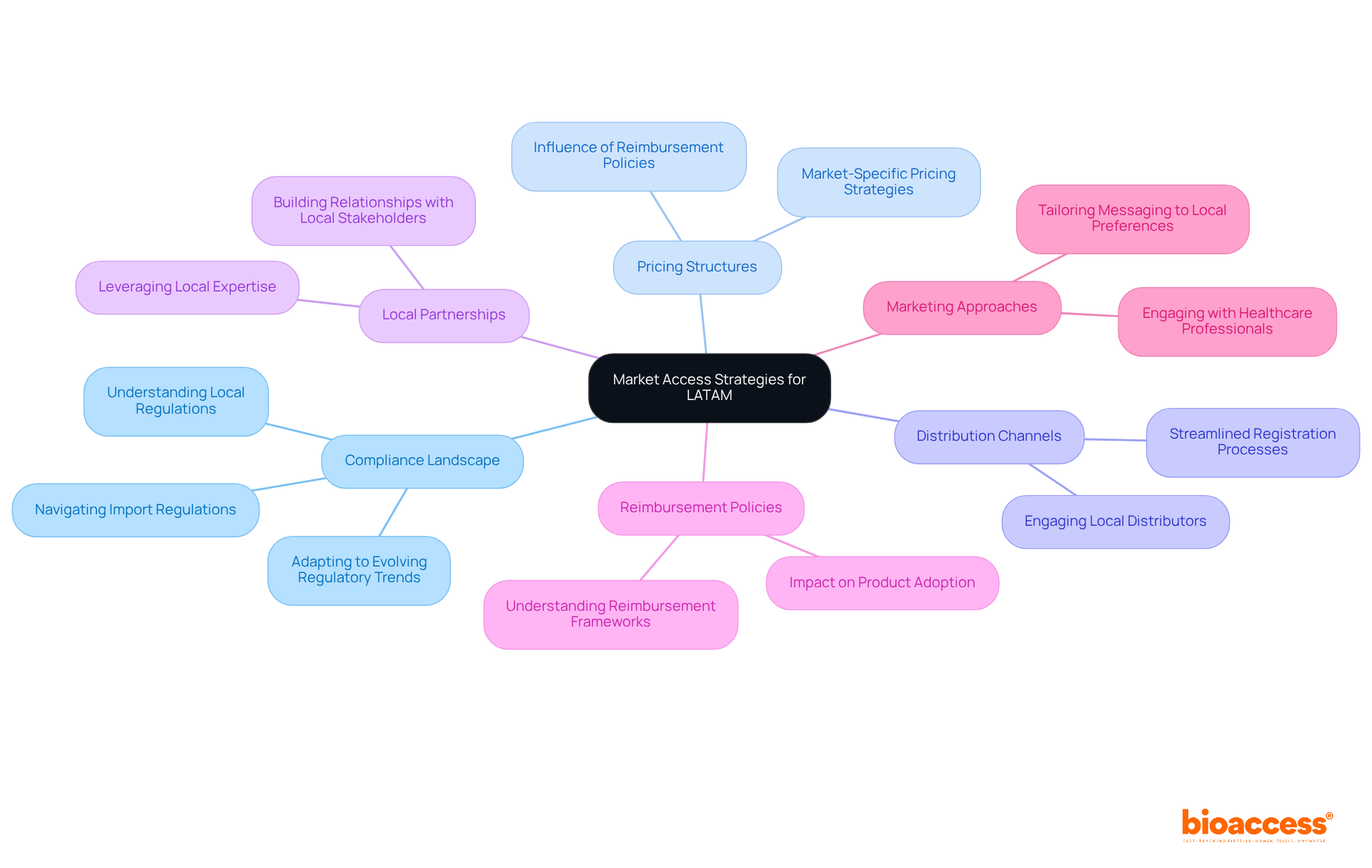
Market Access Strategies: Commercializing Medical Devices in LATAM
To successfully commercialize in Latin America, companies must craft robust that adeptly navigate the region’s unique compliance landscape, pricing structures, and distribution channels in accordance with . Engaging is not just beneficial; it’s essential. These partners offer invaluable insights into market dynamics and facilitate a smoother entry into the . Moreover, understanding is critical, as they can significantly influence product adoption and market penetration.
Tailoring marketing approaches to align with regional preferences is vital; this ensures that messaging resonates with local stakeholders. Staying informed about evolving is equally important for adapting strategies and maintaining compliance. As the healthcare sector in LATAM continues to expand, leveraging and fostering strong partnerships will be key to unlocking the region’s substantial .
In this context, consider how your organization can enhance its . Are you equipped to navigate the complexities of the LATAM healthcare landscape? By prioritizing collaboration and local insights, you can position your company for success in this dynamic market.
Conclusion
Navigating the landscape of medical device standards in Latin America is crucial for companies aiming to establish a foothold in this rapidly growing market. Critical standards like ISO 13485 and ISO 14971 play a pivotal role in ensuring quality management and risk management in medical devices. Compliance with these standards not only meets regulatory requirements but also enhances product safety and marketability, highlighting their significance for successful market entry.
Key insights emphasize the influence of local regulatory bodies, such as ANVISA in Brazil and COFEPRIS in Mexico, in shaping the approval processes for medical devices. Understanding the diverse regulations across Latin America is essential, as each country presents unique challenges and requirements that manufacturers must navigate. Engaging with local experts and compliance consultants can significantly streamline this process, ensuring that all necessary documentation and submissions are handled efficiently.
As the healthcare sector in Latin America continues to evolve, the importance of adhering to medical device standards cannot be overstated. Companies should prioritize collaboration with local partners and stay informed about regulatory changes to optimize their market access strategies. By embracing these standards and leveraging local expertise, organizations can unlock the potential of the Latin American medical device market, ultimately contributing to improved healthcare outcomes across the region.
Frequently Asked Questions
What is ISO 13485 and why is it important for the medical device industry?
ISO 13485 establishes essential requirements for a quality management system (QMS) specifically for the medical device industry. It is crucial for ensuring that organizations consistently meet customer and compliance expectations, particularly in Latin America where compliance is often a prerequisite for market access.
How does ISO 13485 relate to market access in Latin America?
In Latin America, compliance with ISO 13485 is often necessary for market access, aligning with the medical device standards of various countries in the region. For example, in Colombia, INVIMA oversees the marketing and production of health products, making compliance with ISO 13485 essential for companies entering the market.
What recent updates have been made to ISO 13485?
Recent updates to ISO 13485 emphasize a risk-based approach throughout the product lifecycle, requiring organizations to conduct regular risk evaluations and strengthen controls over suppliers and outsourced processes. This reflects the evolving compliance landscape in the medical device industry.
How does ISO 13485 certification benefit healthcare companies?
ISO 13485 certification is seen as the ‘gold standard’ for confirming safety and consistency in healthcare items. It aids in complying with local regulations, improves overall quality and safety of health products, and ultimately benefits both patients and healthcare professionals.
What is ISO 14971 and its significance for medical devices?
ISO 14971 establishes a framework for managing risks associated with healthcare equipment, focusing on hazard identification, risk assessment, and implementation of risk control measures. It is essential for safeguarding patient safety and ensuring product reliability throughout the product lifecycle.
What methodologies are recommended under ISO 14971 for risk management?
Methodologies such as Failure Modes and Effects Analysis (FMEA) and Hazard and Operability Studies (HAZOP) are recommended for efficient risk evaluation and aligning with evolving regulatory requirements.
Why is compliance with ASTM standards important for medical device manufacturers?
ASTM International shapes voluntary consensus standards that govern critical aspects of healthcare devices, such as materials and testing methodologies. Compliance with these standards enhances product safety and efficacy, facilitating smoother regulatory approval and market entry.
How can manufacturers benefit from staying informed about ASTM standards?
Staying informed about the latest ASTM standards is essential for manufacturers to meet compliance obligations, enhance the reliability of healthcare products, and maintain a competitive edge in the evolving landscape of healthcare product development.
List of Sources
- ISO 13485: Quality Management Systems for Medical Devices
- ISO 13485 Certification Services for Medical Devices (https://tuvsud.com/en-us/services/auditing-and-system-certification/iso-13485)
- urmconsulting.com (https://urmconsulting.com/blog/iso-13485-medical-devices-quality-management-system-explained)
- ISO 13485: Comprehensive Overview of the Medical Device Quality Management Standard (https://14644.dk/iso-13485-comprehensive-overview-of-the-medical-device-quality-management-standard)
- Latin America Market Entry Strategies for Medical Device Companies (https://complianceonline.com/resources/latin-america-market-entry-strategies-for-medical-device-companies.html)
- medicaldevicehq.com (https://medicaldevicehq.com/articles/guide-to-quality-management-for-iso-13485)
- ISO 14971: Risk Management for Medical Devices
- How ISO 14971 Helps Medical Device Firms Better Meet QMS Risk Management Requirements – IntellaQuest (https://intellaquest.com/how-iso-14971-helps-medical-device-firms-better-meet-qms-risk-management-requirements)
- Elements of Medical Device Risk Management ISO 14971 – Ventura Solutions (https://ventura-solutions.com/elements-of-medical-device-risk-management-iso-14971)
- FDA’s Risk Management Expectations for Medical Devices: What Manufacturers Need to Know (https://gardner.law/news/fda-risk-management-expectations-medical-devices)
- ASTM Standards: Shaping Medical Device Compliance
- Key Updates on ASTM F2063 Nitinol Tubing Standards in 2025 (https://blog.accupathmed.com/astm-f2063-nitinol-tubing-2025-updates-medical-wearables)
- ANVISA Regulations: Navigating Brazil’s Medical Device Market
- Brazil ANVISA Announces Priorities for the 2026-2027 Year (https://emergobyul.com/news/brazil-anvisa-announces-priorities-2026-2027-year)
- Checking your browser – reCAPTCHA (https://pmc.ncbi.nlm.nih.gov/articles/PMC7704494)
- Brazil Anvisa Regulatory Agenda 2026–2027 | News | Pure Global (https://pureglobal.com/news/brazil-anvisa-regulatory-agenda-2026-2027)
- ANVISA Brazil Medical Device Registration | Pure Global (https://pureglobal.com/markets/brazil)
- Understanding Anvisa Guidelines For Medical Device Trials An In Depth Tutorial | bioaccess® (https://bioaccessla.com/blog/understanding-anvisa-guidelines-for-medical-device-trials-an-in-depth-tutorial)
- Mexico’s Regulatory Approval Process: Key Considerations
- Mexico’s Short Regulatory Timetable Attracts Medtech (https://mddionline.com/medical-device-regulations/mexico-s-shortened-regulatory-timetable-attracts-medical-device-manufacturers)
- bioaccessla.com (https://bioaccessla.com/blog/9-key-class-ii-device-rules-cofepris-for-successful-registration)
- Mexico COFEPRIS 2025 Abbreviated Pathway for Medical Devices | News | Pure Global (https://pureglobal.com/news/mexico-cofepris-2025-abbreviated-pathway-for-medical-devices)
- Local Compliance: Understanding Diverse LATAM Regulations
- How to Navigate Regulatory Compliance in Latin America: A Comprehensive Guide – LAMA (https://lamaaccess.com/how-to-navigate-regulatory-compliance-in-latin-america-a-comprehensive-guide)
- Clinical Trial Regulations: Essential for LATAM Research
- languageconnections.com (https://languageconnections.com/clinical-trials-in-latin-america)
- Clinical Trials – Considerations for Latin America (https://clinicaltrialsarena.com/news/clinical-trials-considerations-for-latin-america-5813786-2)
- bioaccessla.com (https://bioaccessla.com/blog/top-5-latin-america-clinical-trial-success-stories-you-need-to-know)
- bioaccessla.com (https://bioaccessla.com/blog/understanding-regulatory-requirements-for-latin-america-trials-a-comprehensive-overview-of-regulatory-requirements-for-latin-america-trials)
- Ethical Approvals: Navigating LATAM Clinical Research
- bioaccessla.com (https://bioaccessla.com/blog/understanding-regulatory-requirements-for-latin-america-trials-a-comprehensive-overview-of-regulatory-requirements-for-latin-america-trials)
- Clinical Trials in Mexico: Addressing the Challenges (https://languageconnections.com/clinical-trials-in-mexico-addressing-the-challenges)
- Clinical Trials – Considerations for Latin America (https://clinicaltrialsarena.com/news/clinical-trials-considerations-for-latin-america-5813786-2)
- bioaccess: Accelerating Clinical Research and Compliance in LATAM
- bioaccessla.com (https://bioaccessla.com/news)
- Why Global Certification is the Future of Clinical Research in Latin America – Global Medical Device Podcast powered by Greenlight Guru (https://podcast.greenlight.guru/episode/414-clinical-research-certifications-and-what-it-means-for-your-trials)
- PRG & bioaccess Partner to Expand Clinical Trials in Latin America — Physicians Research Group (https://prgresearch.com/press/blog-post-title-one-xxk3g-wsd73)
- amavita.health (https://amavita.health/news/synapse-global-theranostics-launches-to-bridge-precision-medicine-gap-in-latin-america)
- Market Access Strategies: Commercializing Medical Devices in LATAM
- LATAM Market Entry – Pharma Rx & MD (https://chameleon-pharma.com/opportunities-in-latam-a-growing-market-for-pharma-rx-and-medical-devices-towards-2040)
- Unveiling the Latin American health market (https://worldhealthexpo.com/insights/healthcare-management/unveiling-the-latin-american-health-market)
- americasmi.com (https://americasmi.com/about-ami/case-studies)
- Mexico’s Medical Device Regulatory Landscape in 2026 (https://emergobyul.com/events/mexicos-medical-device-regulatory-landscape-2026)




 and see how each branch connects to important details like their role, advancements, effectiveness statistics, risks, and surgeons' views. Explore the central idea of surgical staplers and see how each branch connects to important details like their role, advancements, effectiveness statistics, risks, and surgeons' views.](https://images.tely.ai/telyai/gdmadftk-explore-the-central-idea-of-surgical-staplers-and-see-how-each-branch-connects-to-important-details-like-their-role-advancements-effectiveness-statistics-risks-and-surgeons-views.webp)





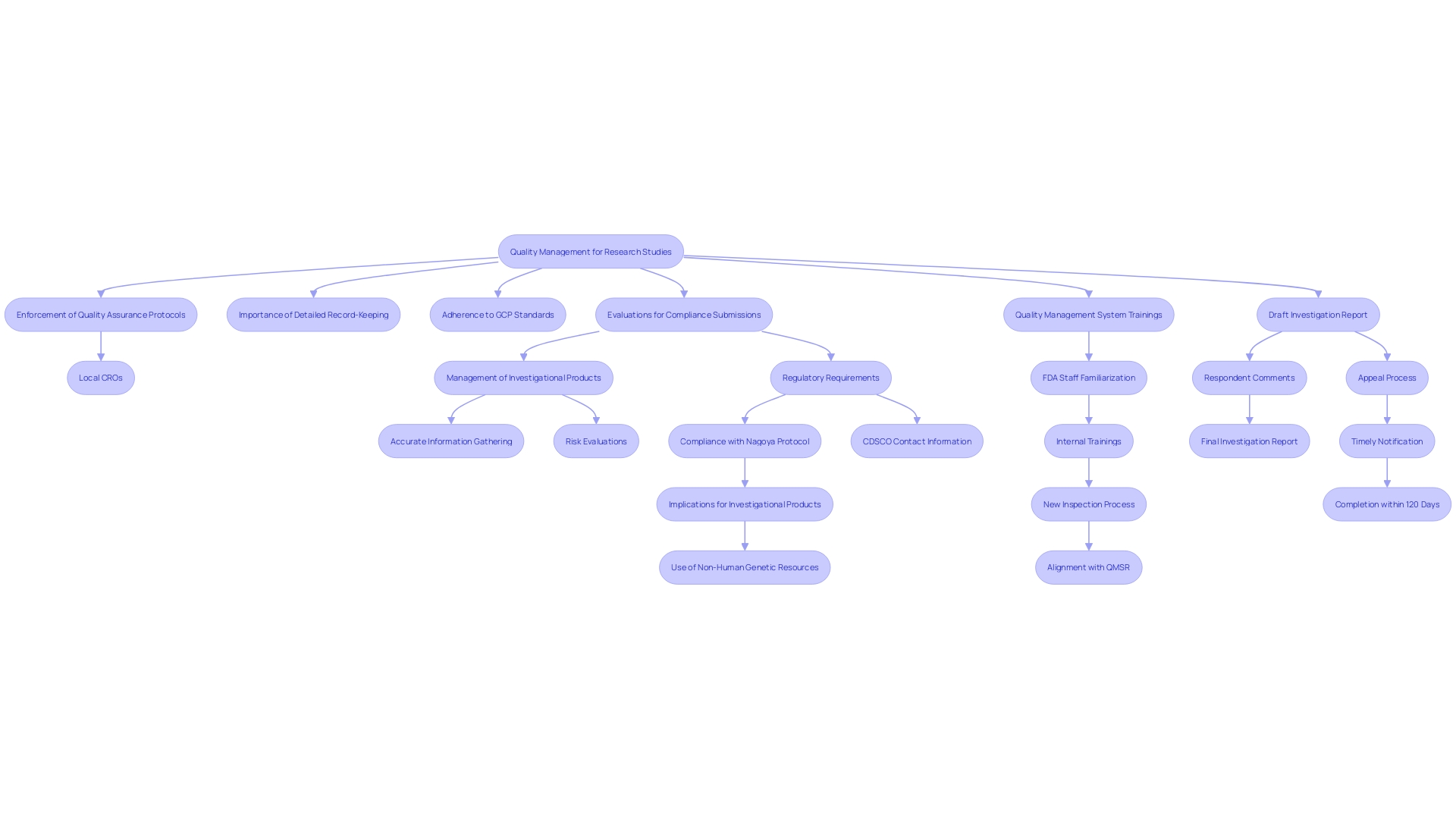
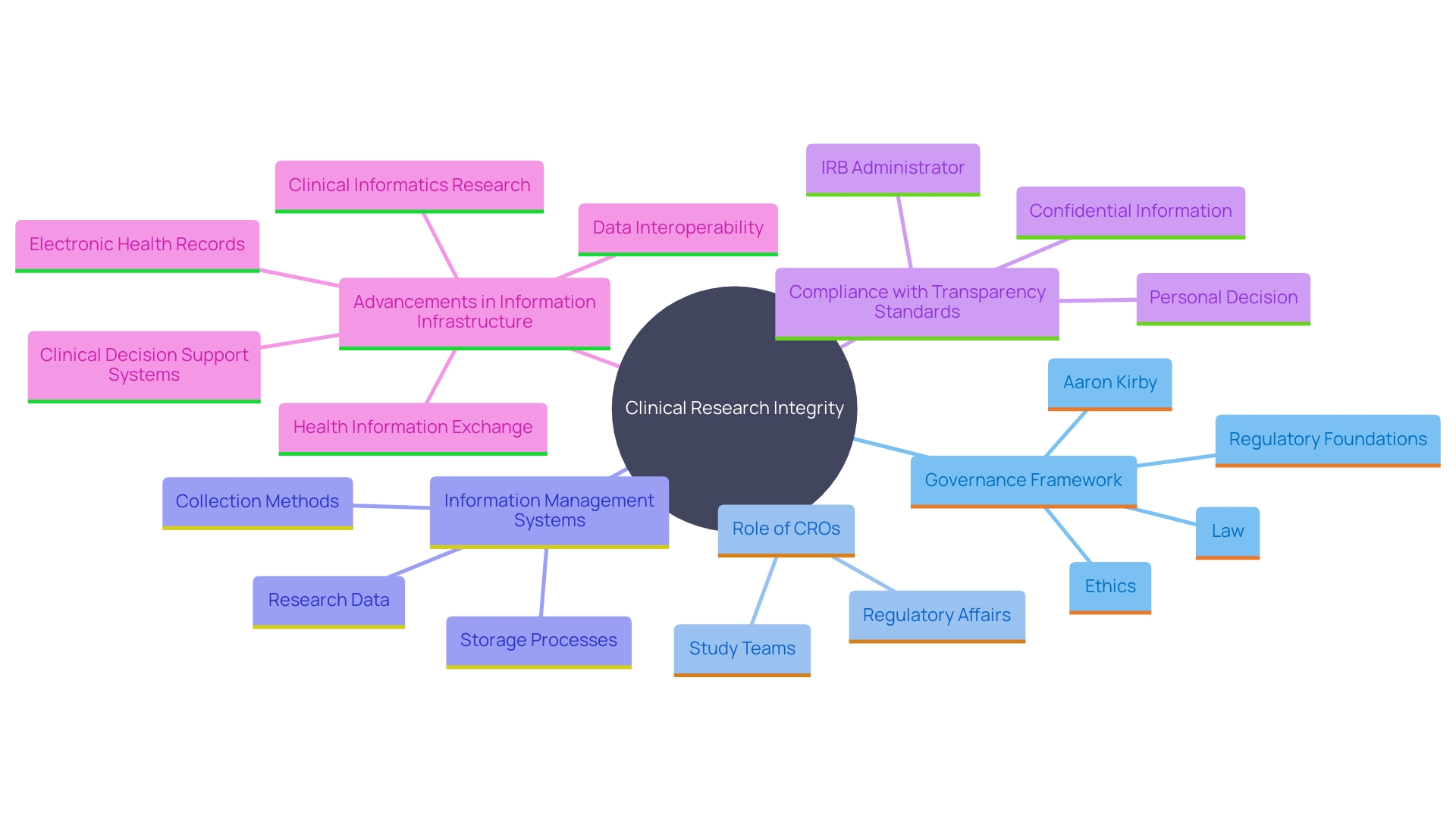
. It highlights the relationships between experience, regulatory knowledge, patient recruitment, [quality management systems](https://bioaccessla.com/blog/10-essential-rec-medical-abbreviations-for-clinical-research-success), and collaboration. This mind map illustrates the key factors to consider when selecting a Contract Research Organization (CRO) for medical device studies. It highlights the relationships between experience, regulatory knowledge, patient recruitment, quality management systems, and collaboration.](https://tely.blob.core.windows.net/telyai/this-mind-map-illustrates-the-key-factors-to-consider-when-selecting-a-contract-research-organization-cro-for-medical-device-studies-it-highlights-the-relationships-between-experience-regulatory-knowledge-patient-recruitment-quality-management-systems-and-collaboration.jpg)