Introduction
Navigating the complexities of registering health-related products in Chile is no small feat, particularly given the stringent regulations imposed by the Instituto de Salud Pública (ISP). This guide distills the process of acquiring the ISP Chile Registration Bundle into three straightforward steps, enabling businesses to comply efficiently with essential legal requirements. However, potential pitfalls often lurk in the documentation and submission phases. So, how can one ensure a smooth and successful registration experience?
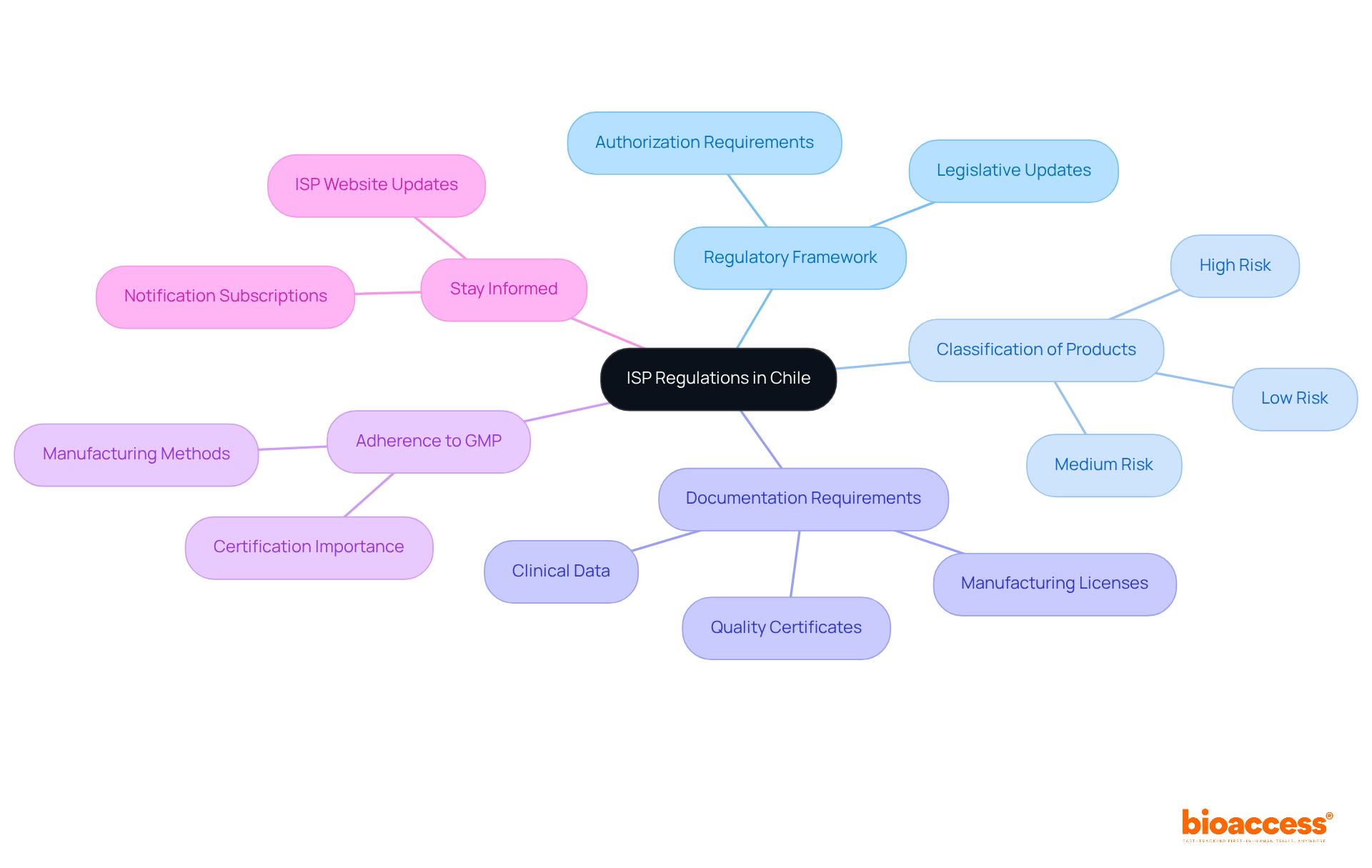
Understand ISP Regulations in Chile
To successfully navigate the to purchase the isp chile registration bundle, understanding the regulations established by the Instituto de Salud Pública (ISP) is crucial. The ISP oversees the approval of health-related products in Chile, including pharmaceuticals and medical devices. Here are the key points to consider:
- : Familiarize yourself with the latest legislative updates and guidelines from the ISP. This includes understanding which products require authorization and the specific documentation needed.
- : Products are categorized based on their risk levels. Knowing whether your product is classified as high, medium, or low risk will significantly influence the procedures and requirements you must follow.
- : Prepare the necessary documents, which typically include quality certificates, manufacturing licenses, and . Ensure that all documents meet ISP standards to avoid any delays in the process.
- Adherence to : Align your manufacturing methods with GMP guidelines, as this is essential for certification.
- : Regularly check the ISP website or subscribe to notifications to stay updated on any changes in regulations or procedures that may affect your enrollment process.
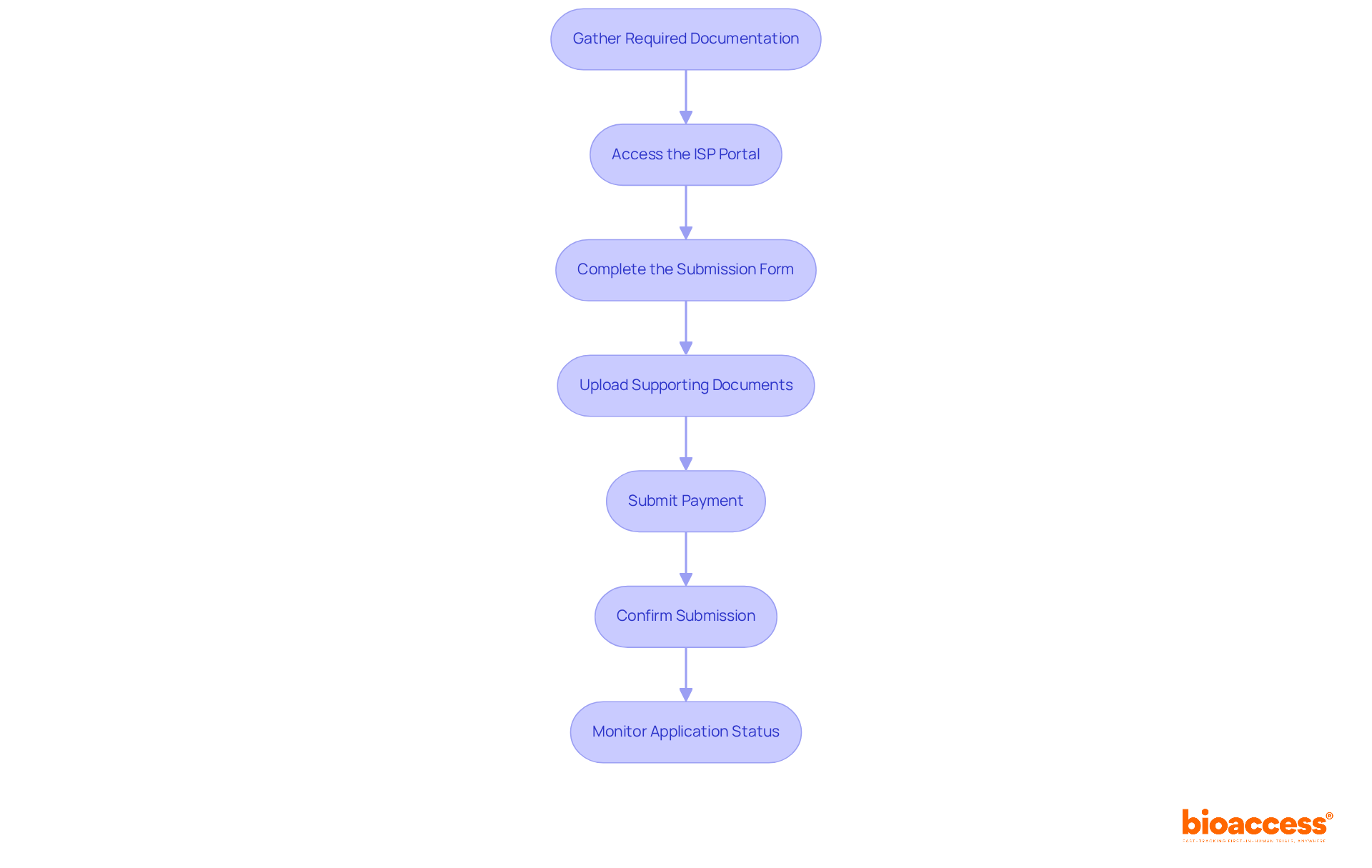
Follow the Step-by-Step Purchase Process
To successfully purchase the , follow this streamlined process:
- Gather Required : Ensure you have all necessary documents ready, including product specifications, , and any prior approvals from other .
- Access the ISP Portal: Navigate to the ISP’s official website and locate the . If you do not have an account, create one to proceed.
- Complete the : Accurately fill out the online with detailed information about your product. Ensure that all details align with the you have prepared.
- Upload : Attach all required documents to your submission. Verify that each document is complete and formatted according to ISP guidelines.
- Submit Payment: After finishing the form, proceed to the . The , so select the appropriate option.
- Confirm Submission: Once payment is made, verify that your request has been submitted successfully. You should receive a confirmation email with a tracking number for your submission.
- Monitor Application Status: Regularly check the ISP portal for updates on your application status. Be prepared to respond promptly to any requests for additional information or clarification from the ISP.
By following these steps, you can efficiently navigate the ISP sign-up procedure and successfully purchase the , ensuring compliance and speeding up your product’s .
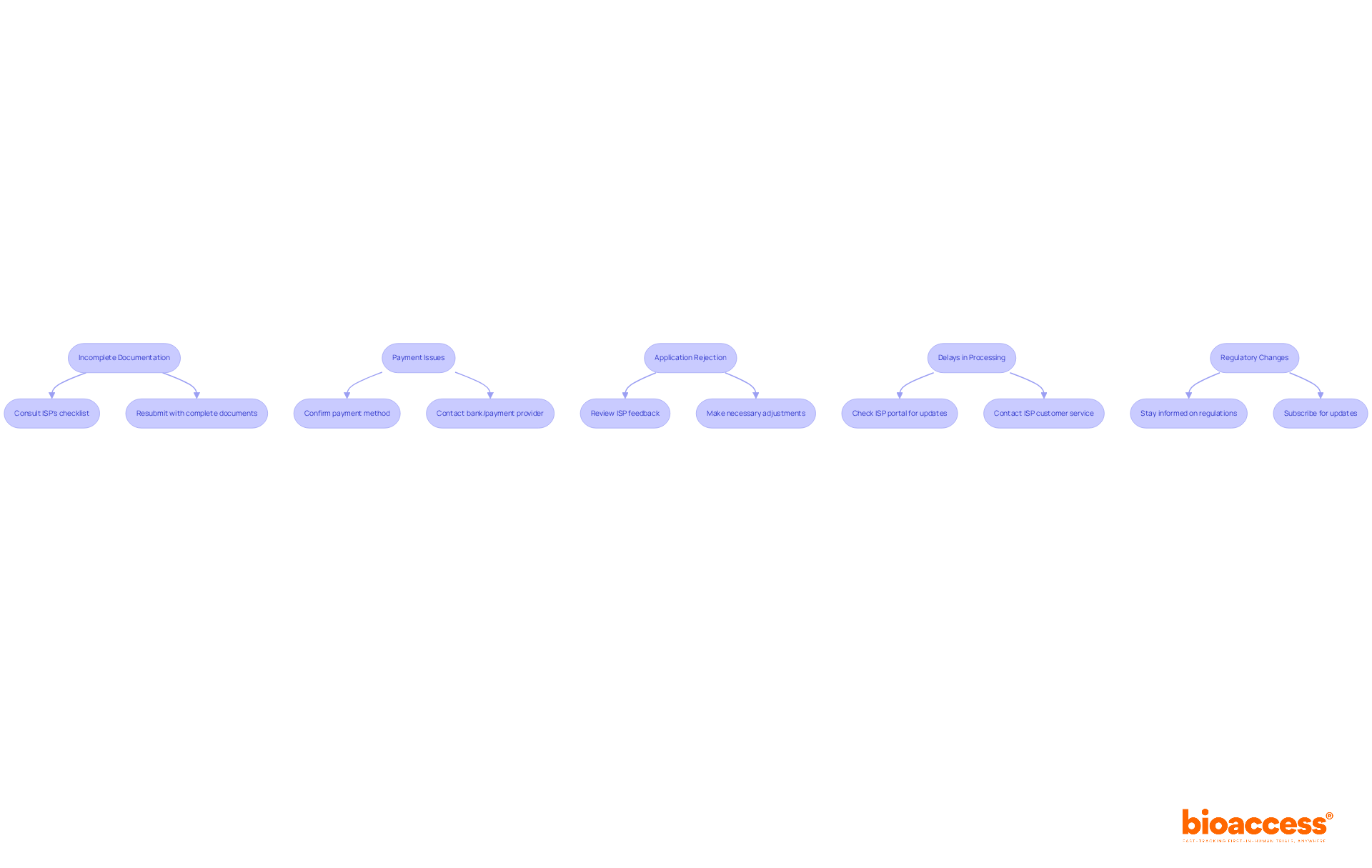
Troubleshoot Common Registration Issues
Even with meticulous preparation, . Here are common issues and effective troubleshooting strategies:
- Incomplete Documentation: . In fact, missing a single document or providing incorrect information can lead to immediate rejection of the submission. To avoid this, consult the thoroughly and ensure all required documents are included. If your request is returned, promptly resubmit it with the complete documentation. Consulting experts like Registrationwala can also help ensure your paperwork is complete and accurate.
- : Payment processing problems can obstruct your request. Confirm that your payment method is accepted by the ISP and check with your bank or payment provider if necessary. Ensure you have sufficient funds to complete the transaction.
- : In the event of rejection, carefully review the feedback provided by the ISP. Address the specific concerns raised and make the necessary adjustments before resubmitting your request. Statistics indicate that applicants face challenges due to , highlighting the importance of thoroughness.
- : If your request is taking longer than expected, check the ISP portal for updates. If no information is available, contact ISP customer service for clarification on your request’s status. Understanding the typical processing times can help set realistic expectations.
- : Stay informed about any modifications in ISP regulations that may affect your submission. Regularly check the ISP’s website or subscribe to their updates to ensure compliance with the latest requirements. Being proactive about can prevent unnecessary delays and complications in your application process.
Conclusion
Navigating the process of purchasing the ISP Chile Registration Bundle may seem daunting, but with a clear understanding of the regulations and a step-by-step approach, success is well within reach. This guide underscores the necessity of familiarizing yourself with ISP regulations, preparing the required documentation, and following the outlined procedures to ensure compliance and efficiency in the registration process.
Understanding the regulatory framework established by the Instituto de Salud Pública (ISP) is critical. It’s essential to classify products based on their risk levels. Proper documentation preparation, adherence to Good Manufacturing Practices (GMP), and staying updated on regulatory changes are vital steps that can significantly streamline the enrollment process. Moreover, addressing common issues like incomplete documentation or payment problems can prevent delays and rejections, ensuring a smoother experience.
Ultimately, mastering the ISP registration process not only facilitates the entry of health-related products into the Chilean market but also highlights the importance of compliance in the health sector. By taking proactive measures and staying informed, businesses can effectively navigate the complexities of ISP regulations. For those aiming for success in this endeavor, thorough preparation and diligence are key-take action today to ensure your registration process is as seamless as possible.
Frequently Asked Questions
What is the role of the Instituto de Salud Pública (ISP) in Chile?
The ISP oversees the approval of health-related products in Chile, including pharmaceuticals and medical devices.
Why is it important to understand ISP regulations when purchasing the ISP Chile registration bundle?
Understanding ISP regulations is crucial for successfully navigating the enrollment procedure and ensuring compliance with the necessary requirements.
What should I familiarize myself with regarding ISP regulations?
You should familiarize yourself with the latest legislative updates and guidelines from the ISP, including which products require authorization and the specific documentation needed.
How are products classified by the ISP?
Products are categorized based on their risk levels: high, medium, or low risk, which influences the procedures and requirements that must be followed.
What documentation is required for ISP registration?
Necessary documents typically include quality certificates, manufacturing licenses, and clinical data, all of which must meet ISP standards to avoid delays.
What are Good Manufacturing Practices (GMP) and why are they important?
GMP guidelines are standards for manufacturing methods that must be aligned with to achieve certification from the ISP.
How can I stay informed about changes in ISP regulations?
You can stay informed by regularly checking the ISP website or subscribing to notifications for updates on regulations or procedures that may affect your enrollment process.
List of Sources
- Follow the Step-by-Step Purchase Process
- Chile medical device registration process | MDRC (https://mdrc-consulting.com/chile-medical-device-registration-process-en)
- Importing medicines and cosmetics in Chile — ISP regulations – ComexPoint (https://comexpoint.com/en/blog/chile-isp-regulations)
- Chile – Rx & OTC drugs registration (https://chameleon-pharma.com/insights-into-the-rx-and-otc-drug-registration-in-chile)
- How to Register Medical Devices in Chile: Comp… | bioaccess® (https://bioaccessla.com/blog/medical-device-registration-chile-isp-requirements)
- pharmaregulatory.in (https://pharmaregulatory.in/introduction-to-isp-and-its-regulatory-authority-in-chile-regulatory-affairs-and-pharmacovigilance)
- Troubleshoot Common Registration Issues
- 7 Common Network Issues and How to Resolve Them Fast (https://cbtnuggets.com/blog/technology/networking/7-common-network-issues-and-how-to-resolve-them-fast)
- Top Challenges Faced During the ISP License Application Process (https://registrationwala.wordpress.com/2025/04/28/top-challenges-faced-during-the-isp-license-application-process)
- 7 Common Challenges for Local ISPs and Splynx Solutions (https://splynx.com/blog/business-automation/7-most-common-challenges-faced-by-local-isps-how-splynx-helps-to-solve-them)
- Chile: fixed internet provider market share 2024| Statista (https://statista.com/statistics/217499/market-share-of-fixed-line-users-in-chile-by-provider?srsltid=AfmBOoopFAVUIVDpUXe7SCalxhIImjTwucy3TovxS5V5SJDtC7nVspbP)
- The 5 Most Common Mistakes in ISP Management and How to Avoid Them – ISPadmin (https://ispadmin.eu/en/the-5-most-common-mistakes-in-isp-management-and-how-to-avoid-them)

Leave a Reply