Introduction
Understanding the complexities of Institutional Review Board (IRB) approval in Bolivia is crucial for researchers committed to conducting ethical clinical trials. With the increasing emphasis on ethical standards in research, grasping the IRB’s role and the necessary documentation can significantly influence the success of studies involving human subjects. Researchers often face obstacles such as incomplete submissions and cultural sensitivities that can delay the approval process.
What steps can researchers take to prepare and streamline their IRB processes for quicker approvals while ensuring their clinical trials remain ethical? By addressing these challenges head-on, researchers can enhance their chances of securing timely IRB approvals and maintaining the integrity of their studies.
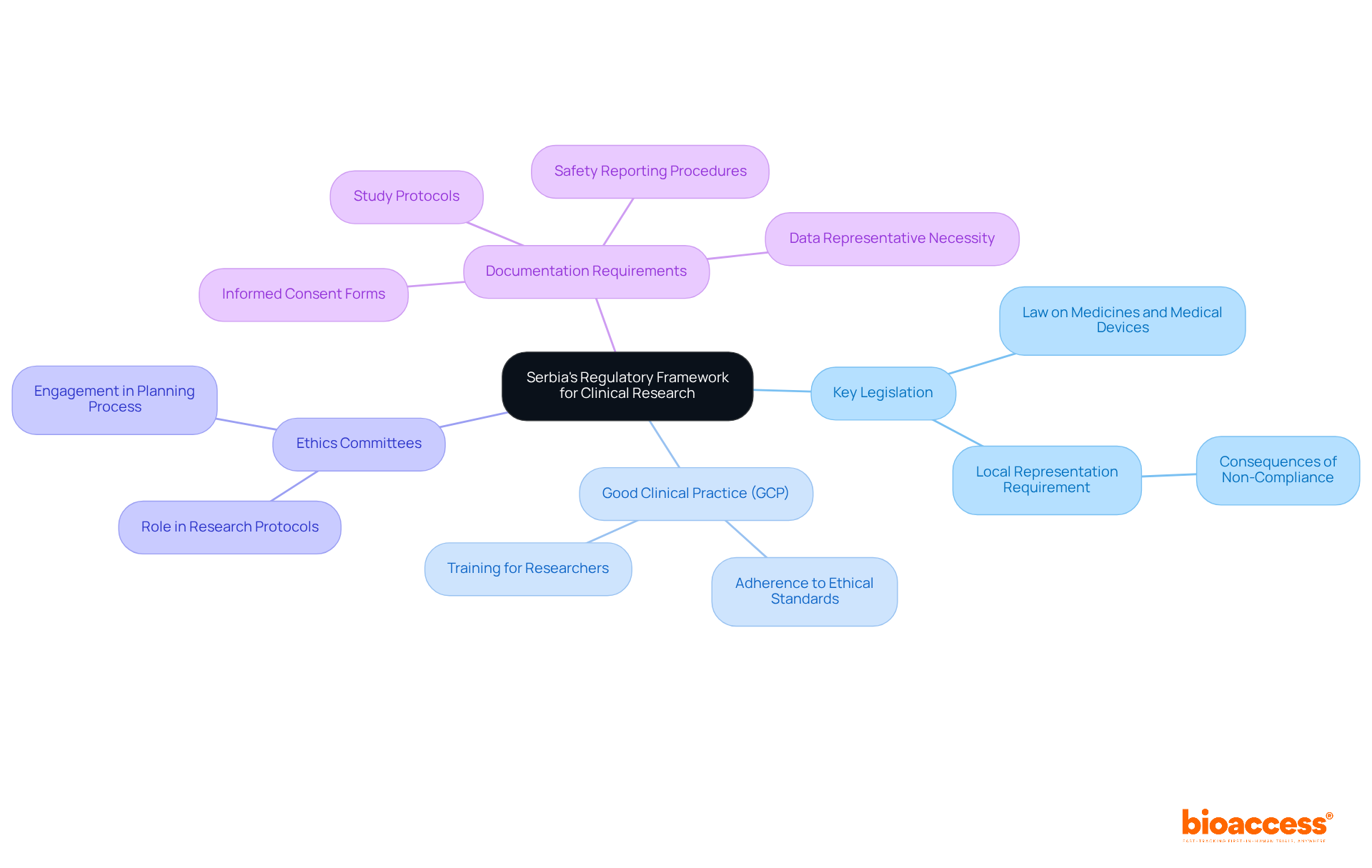
Understand the Role of Institutional Review Boards (IRBs) in Bolivia
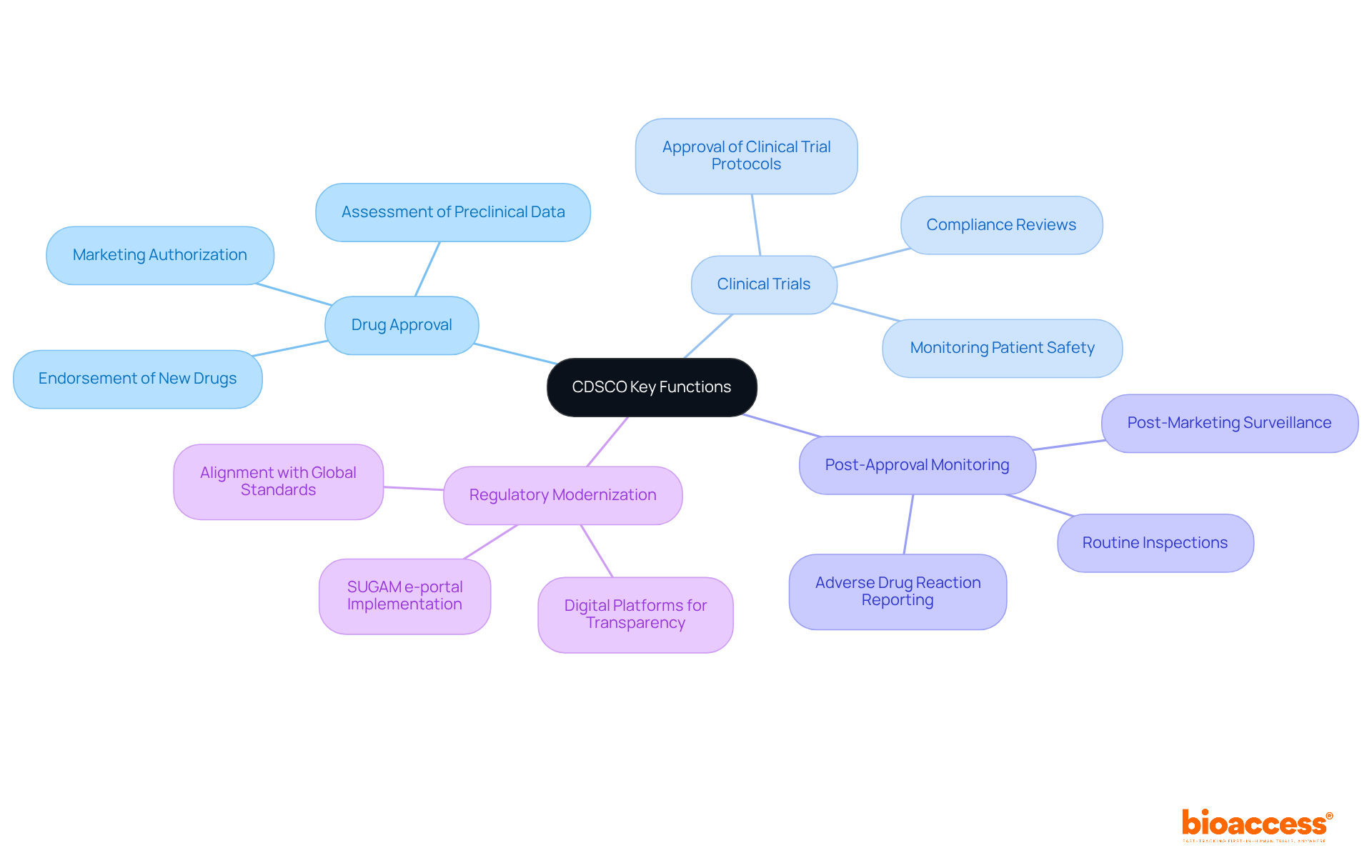
In Bolivia, the process of IRB approval Bolivia is pivotal in shaping the landscape of clinical trials involving human subjects. Their primary responsibility is to ensure that all proposals meet ethical standards while safeguarding the rights and welfare of participants. IRBs evaluate study protocols, informed consent documents, and materials related to participant recruitment. They assess the potential risks and benefits of research, ensuring adherence to local regulations and international guidelines such as ICH-GCP.
Recognizing the IRB’s role is crucial for researchers. It directly influences both the approval timeline and the overall success of their studies. It’s vital for researchers to connect with IRBs early in their planning. This ensures their protocols meet ethical standards and regulatory requirements. Navigating the IRB process can often feel overwhelming for researchers, leading to potential setbacks. However, this proactive approach can significantly streamline the approval procedure and mitigate delays.
By 2026, the presence of IRB approval Bolivia will allow several IRBs to actively operate, significantly enhancing ethical standards in clinical trials. The efforts of the Research Ethics Committee in Bolivia have improved ethical oversight and boosted participation in international collaborations, demonstrating a commitment to community engagement. The IRB process generally entails submitting a comprehensive proposal, which encompasses the project’s objectives, methodology, and ethical considerations. Researchers should familiarize themselves with the specific IRB requirements in their region, as these can vary between institutions and may include additional documentation or local ethical approvals. Furthermore, research initiatives in Bolivia are supported by funding from the National Institute for Health Research (NIHR), underscoring the importance of ethical standards in advancing clinical research. Understanding and navigating the IRB approval Bolivia process is not just a regulatory hurdle; it is a critical step toward ensuring the integrity and success of clinical research in Bolivia.
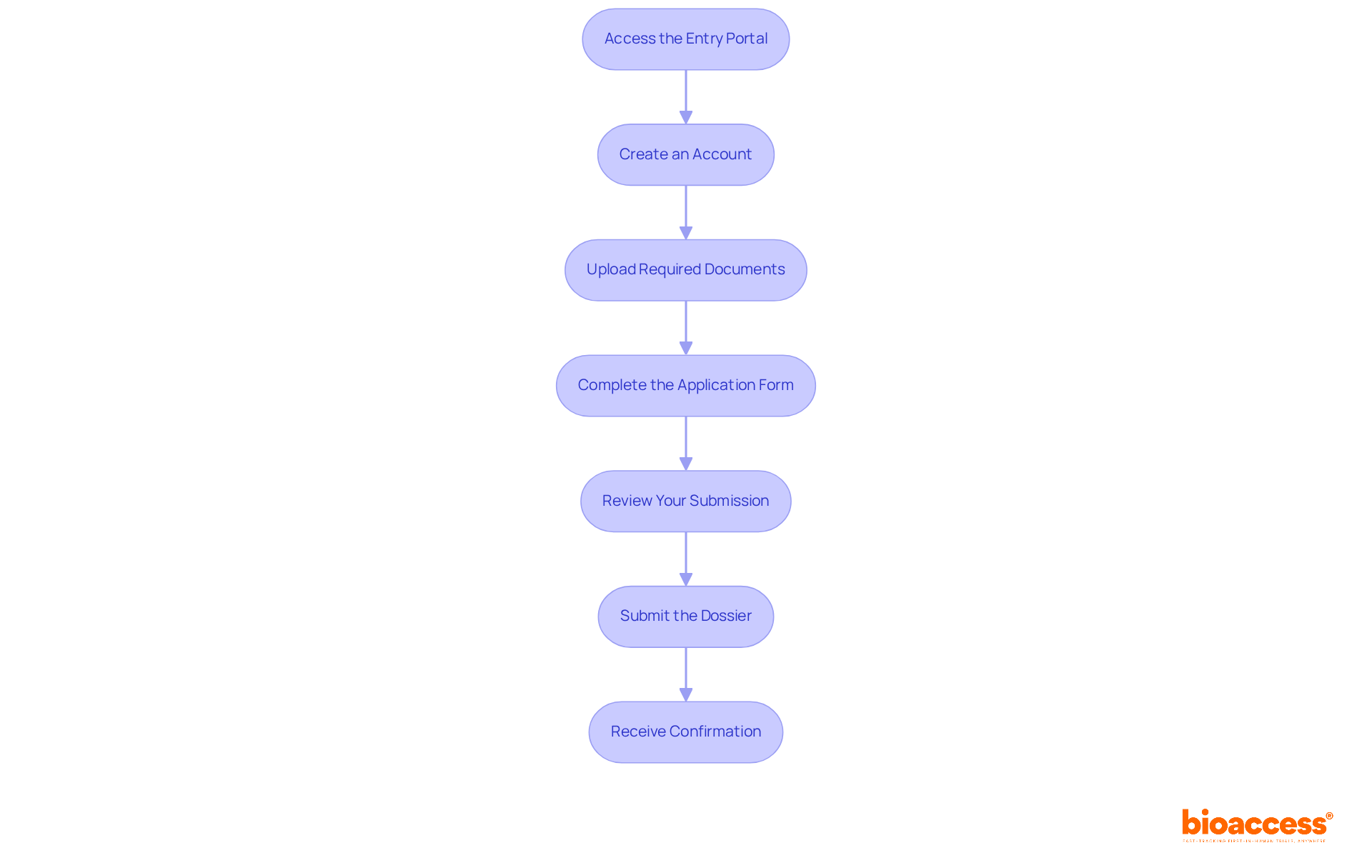
Prepare Required Documentation for IRB Submission
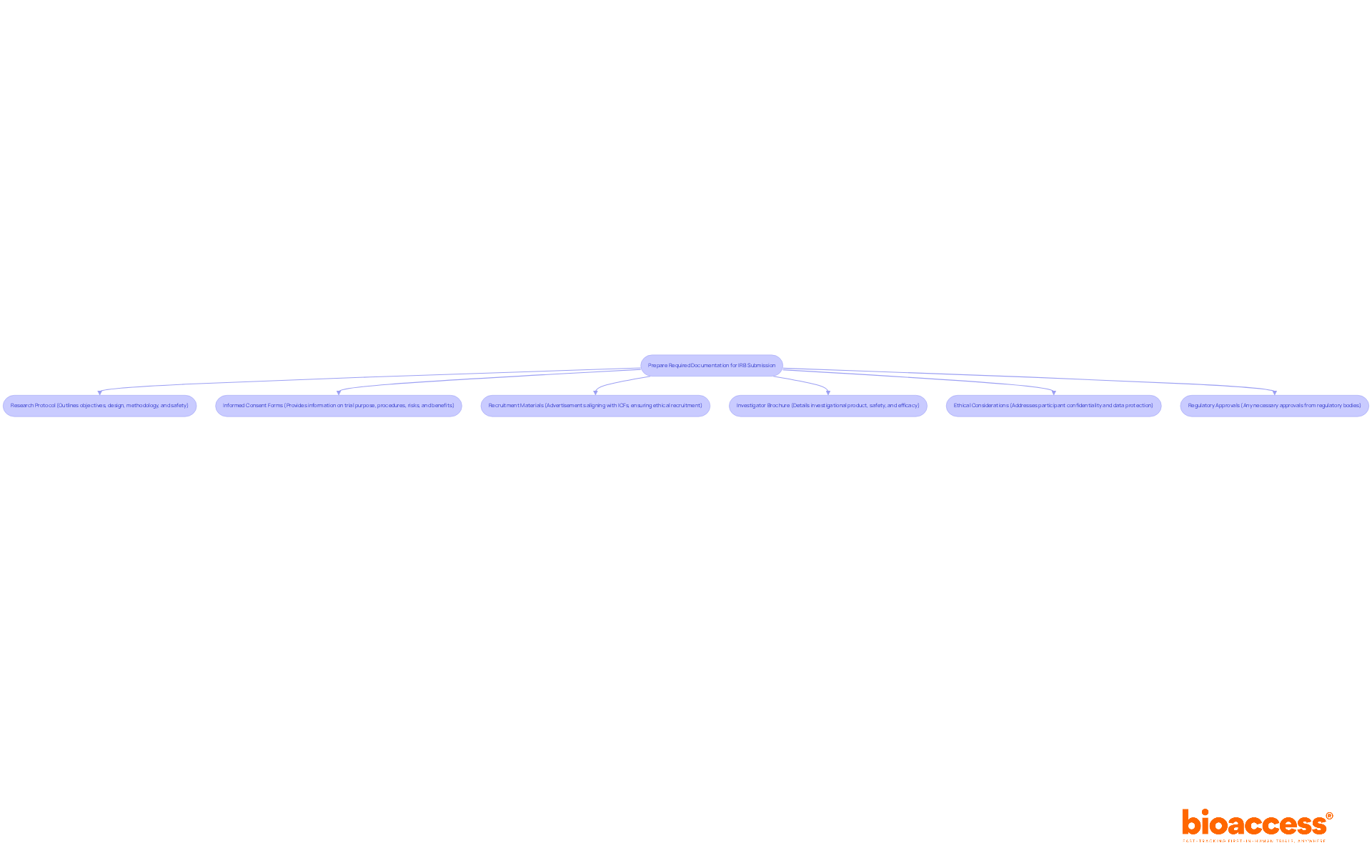
Navigating the irb approval bolivia process can be daunting, yet it is essential for advancing clinical research. To successfully obtain irb approval bolivia, researchers must prepare a comprehensive set of documents that demonstrate the ethical and scientific validity of their work. The following documents are typically required:
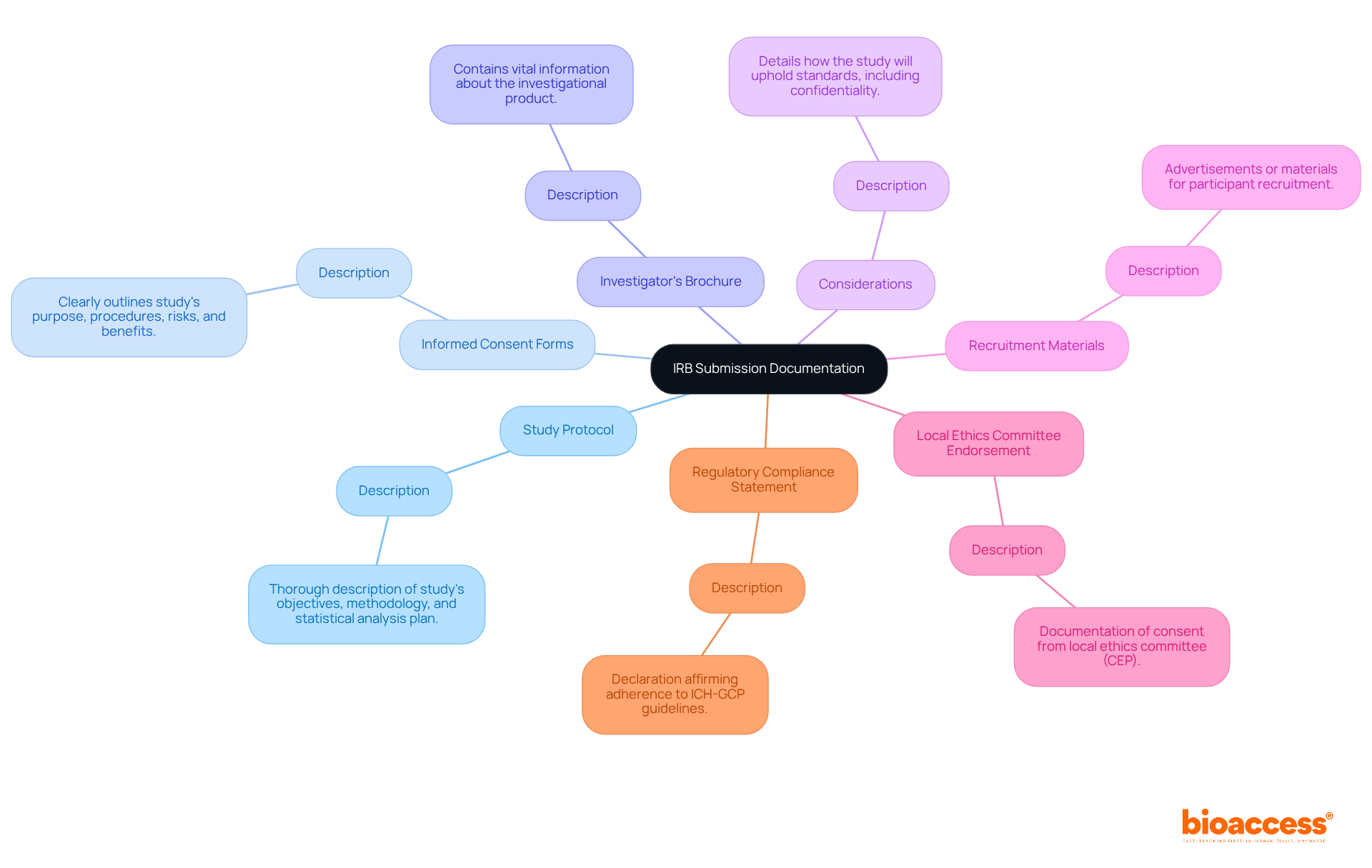
- Research Protocol: This document outlines the project’s objectives, design, methodology, and statistical analysis plan. It should clearly articulate how the research will be conducted and how participant safety will be ensured, adhering to ICH-GCP guidelines. Furthermore, researchers should indicate whether the research will necessitate expedited or full-board review depending on the level of risk involved.
- Informed Consent Forms (ICFs): These documents must offer potential participants clear and comprehensive information regarding the trial, including its purpose, procedures, risks, and benefits. The language should be accessible to the target population, ensuring that participants can make informed decisions.
- Recruitment Materials: Any advertisements or materials used to recruit participants must be submitted for review. These should align with the information provided in the ICFs and must not contain coercive language, promoting ethical recruitment practices.
- Investigator Brochure: This document offers comprehensive details regarding the investigational product, encompassing preclinical and clinical data that back its application in the proposed trial. It is essential for demonstrating the product’s safety and efficacy.
- Ethical Considerations: A section detailing how the study will address ethical issues, including participant confidentiality and data protection measures, is crucial for IRB evaluation.
- Regulatory Approvals: If applicable, include any approvals from other regulatory bodies, such as INVIMA or AGEMED, which may be required for specific types of research. This guarantees adherence to local regulations and can accelerate the approval timeline.
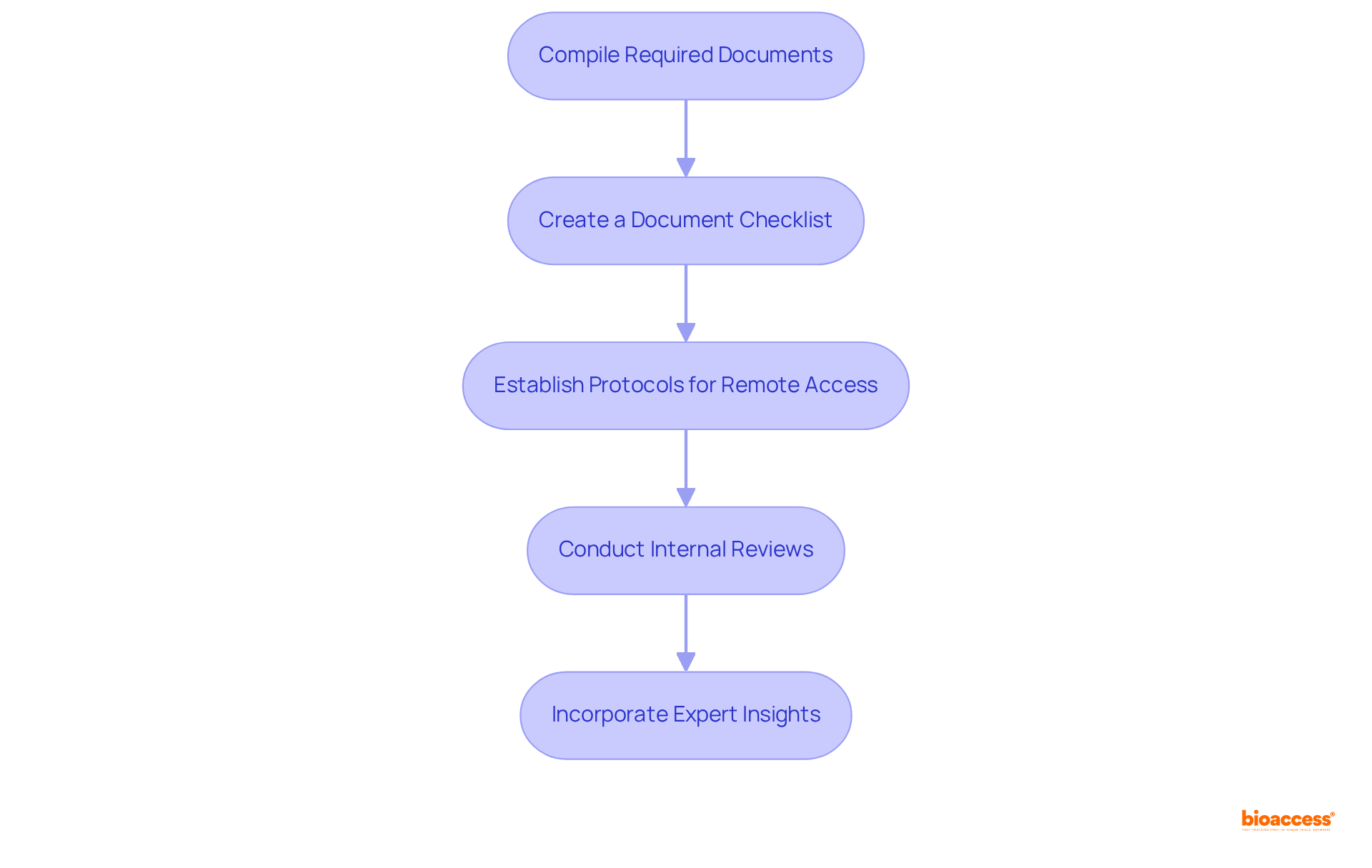
It’s crucial for researchers to meticulously prepare and format all documents according to the IRB’s guidelines. Using a checklist can help ensure that all essential materials are included, thereby speeding up the review and reducing the risk of delays due to incomplete submissions. By ensuring thorough documentation and ethical practices, researchers can not only meet regulatory demands but also enhance the integrity of their studies.
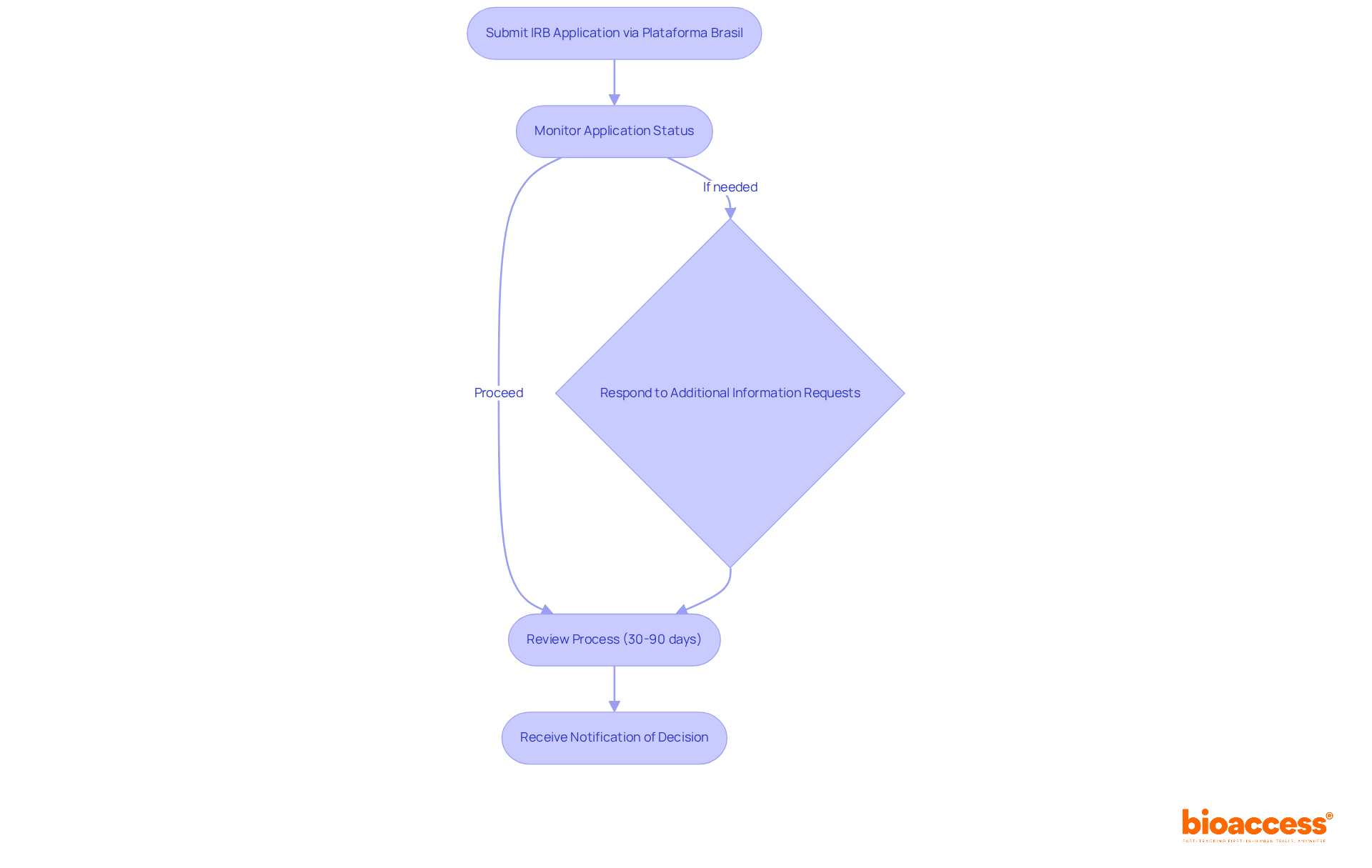
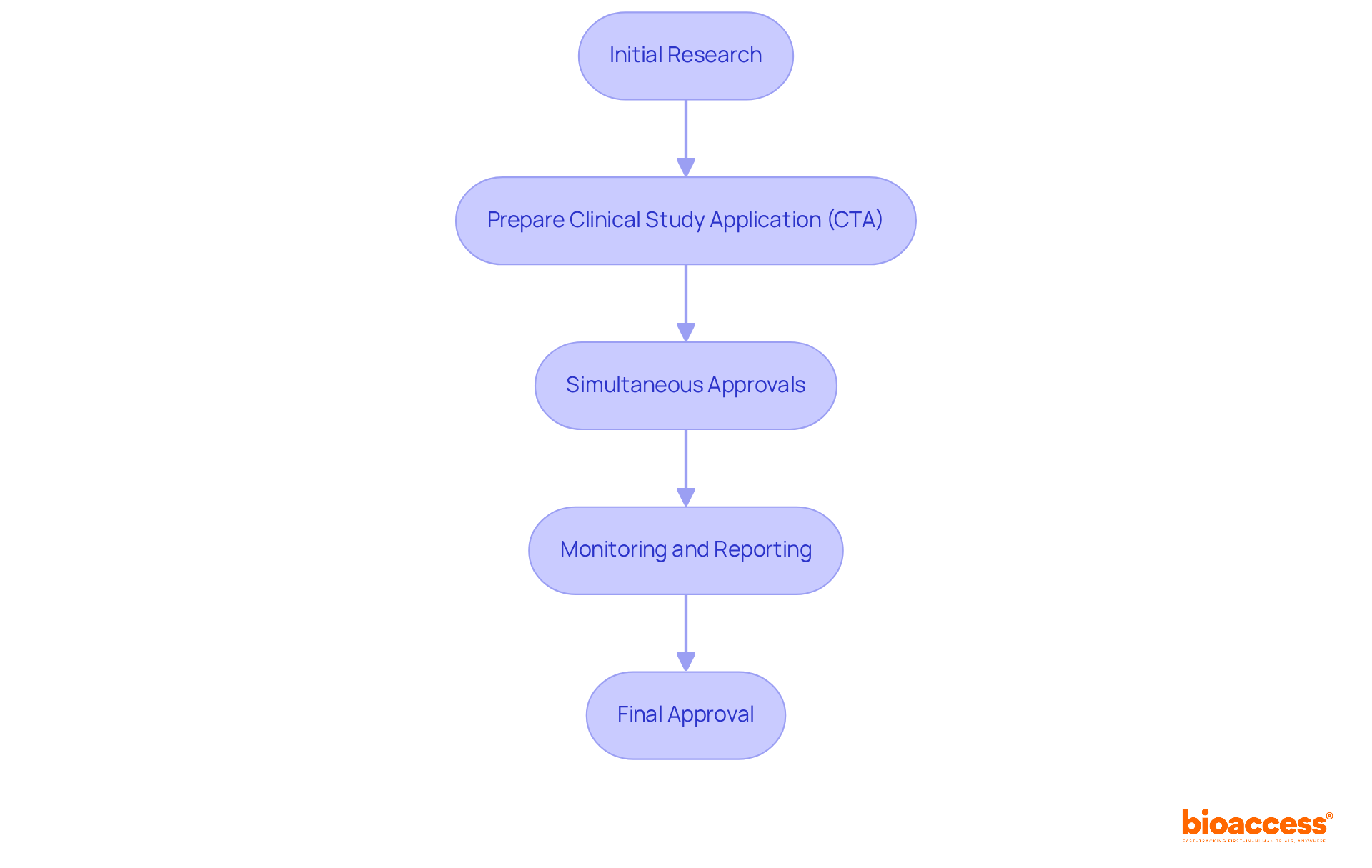
Navigate the IRB Submission Timeline and Review Process
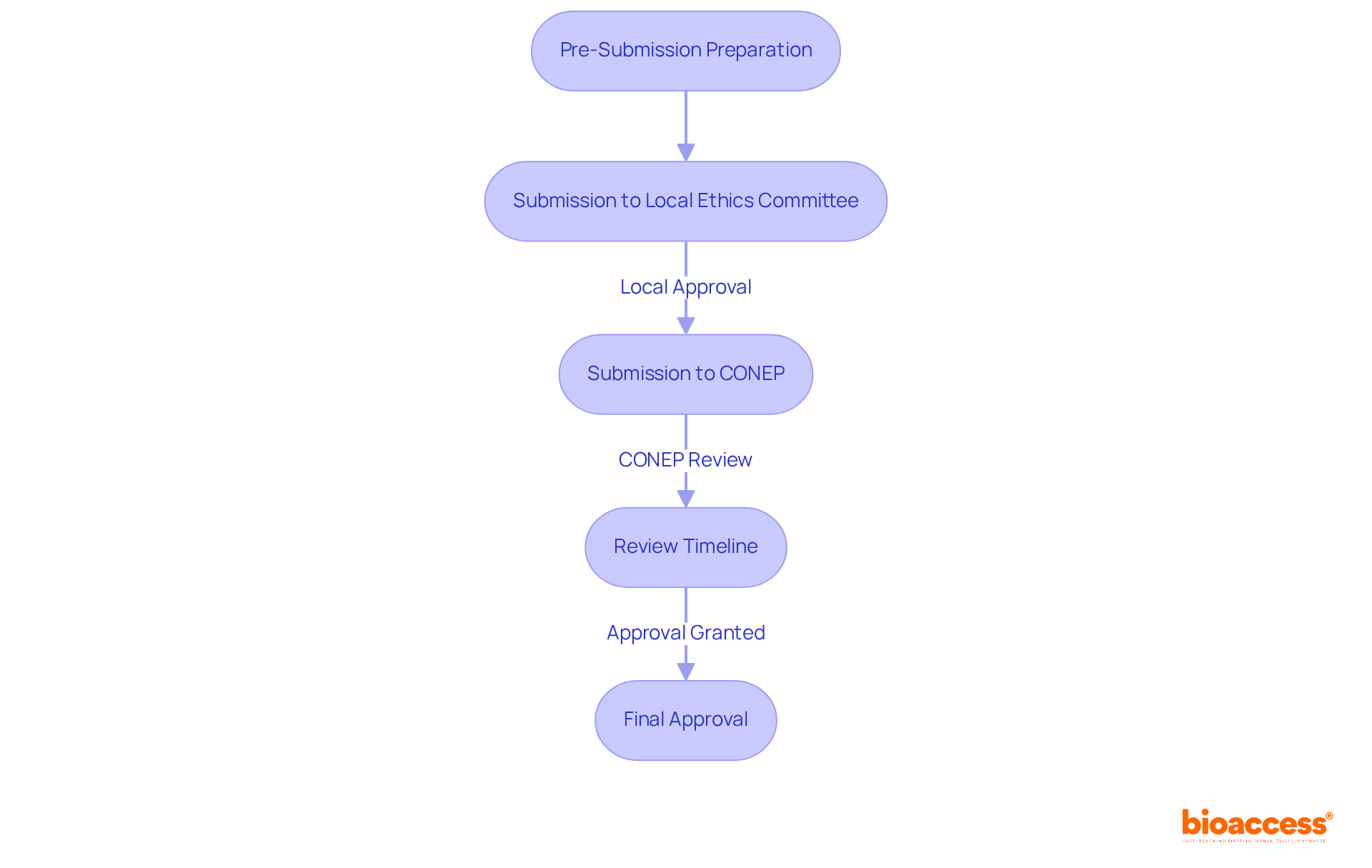
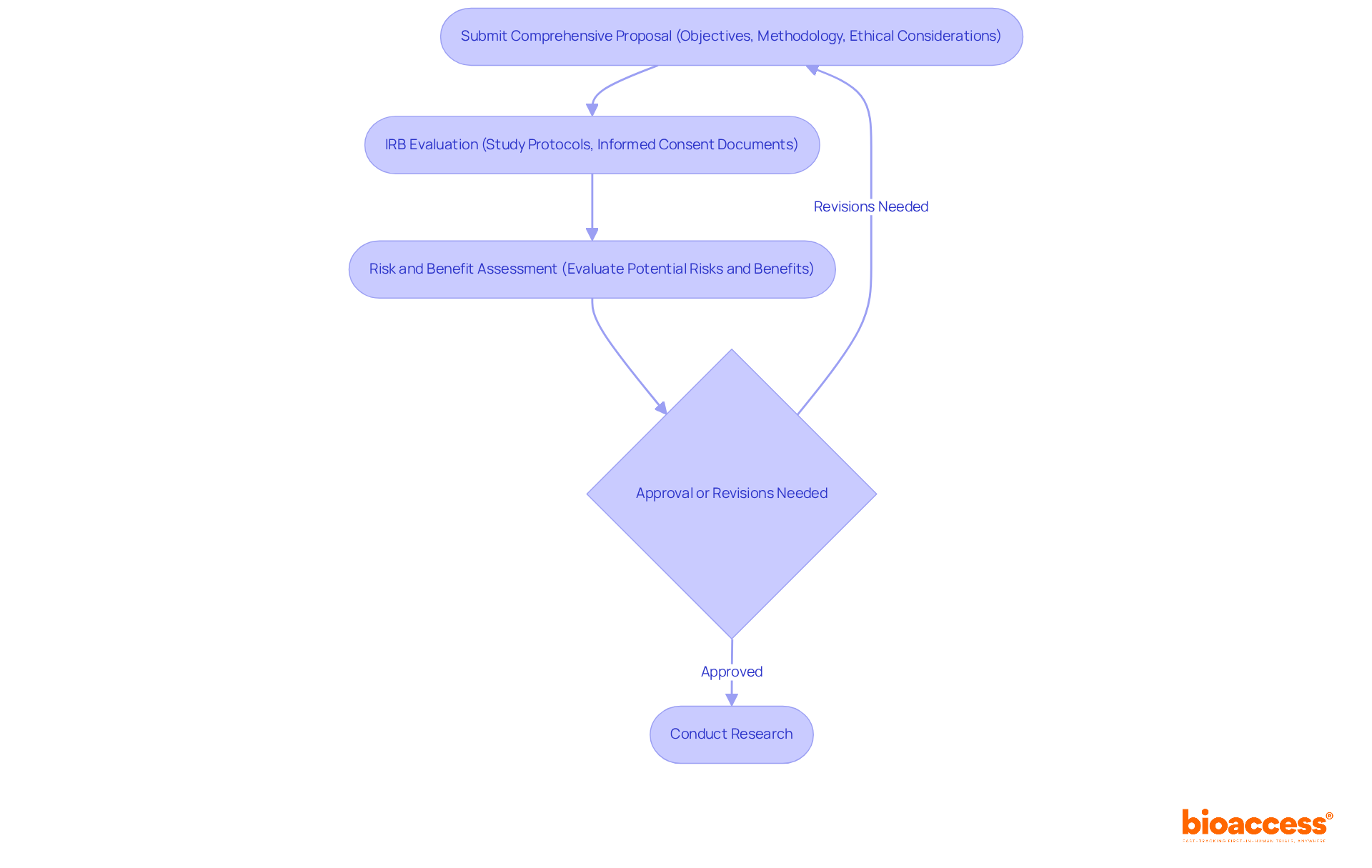
Navigating the irb approval bolivia process can be a complex journey, influenced by various factors that researchers must understand. Here’s a detailed timeline and what researchers can expect:
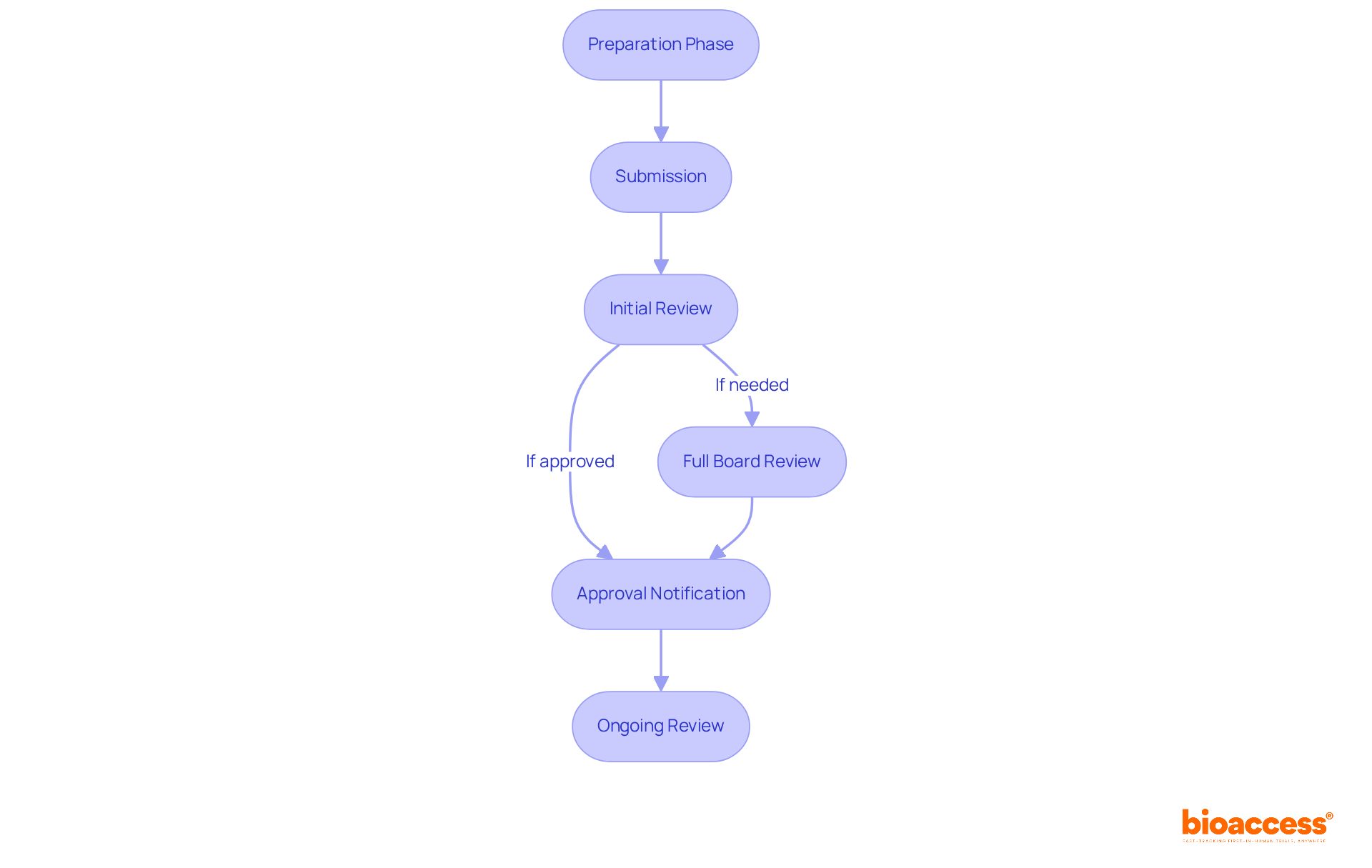
- Preparation Phase: Researchers need to set aside ample time for documentation preparation. This process can span from a few weeks to several months, depending on the project’s complexity and the team’s readiness.
- Submission: After completing the necessary documentation, it is submitted to the IRB. Guaranteeing that all necessary documents are included is essential to prevent delays in the evaluation.
- Initial Review: Typically, the IRB takes about 5-10 business days for an initial review. During this period, they assess the completeness of the submission and may request additional information or clarifications, which can impact the overall timeline.
- Full Board Review: If a full board review is required, expect an additional delay of 2-4 weeks. This delay can significantly impact project timelines, forcing researchers to adjust their schedules. The IRB will convene to discuss the research and make a decision regarding approval, particularly for projects involving vulnerable populations or complex methodologies.
- Approval Notification: Once the review is complete, the IRB will notify the researchers of their decision. If approved, researchers can begin their research; if changes are needed, the IRB will offer feedback for necessary revisions.
- Ongoing Review: Following initial approval, researchers must submit progress reports and any amendments to the IRB for ongoing review, ensuring continued compliance with ethical standards.
By grasping this timeline, researchers can avoid costly delays and streamline their research efforts. This proactive strategy can lead to more efficient research methods, ultimately enhancing the success of first-in-human trials that require irb approval in Bolivia.
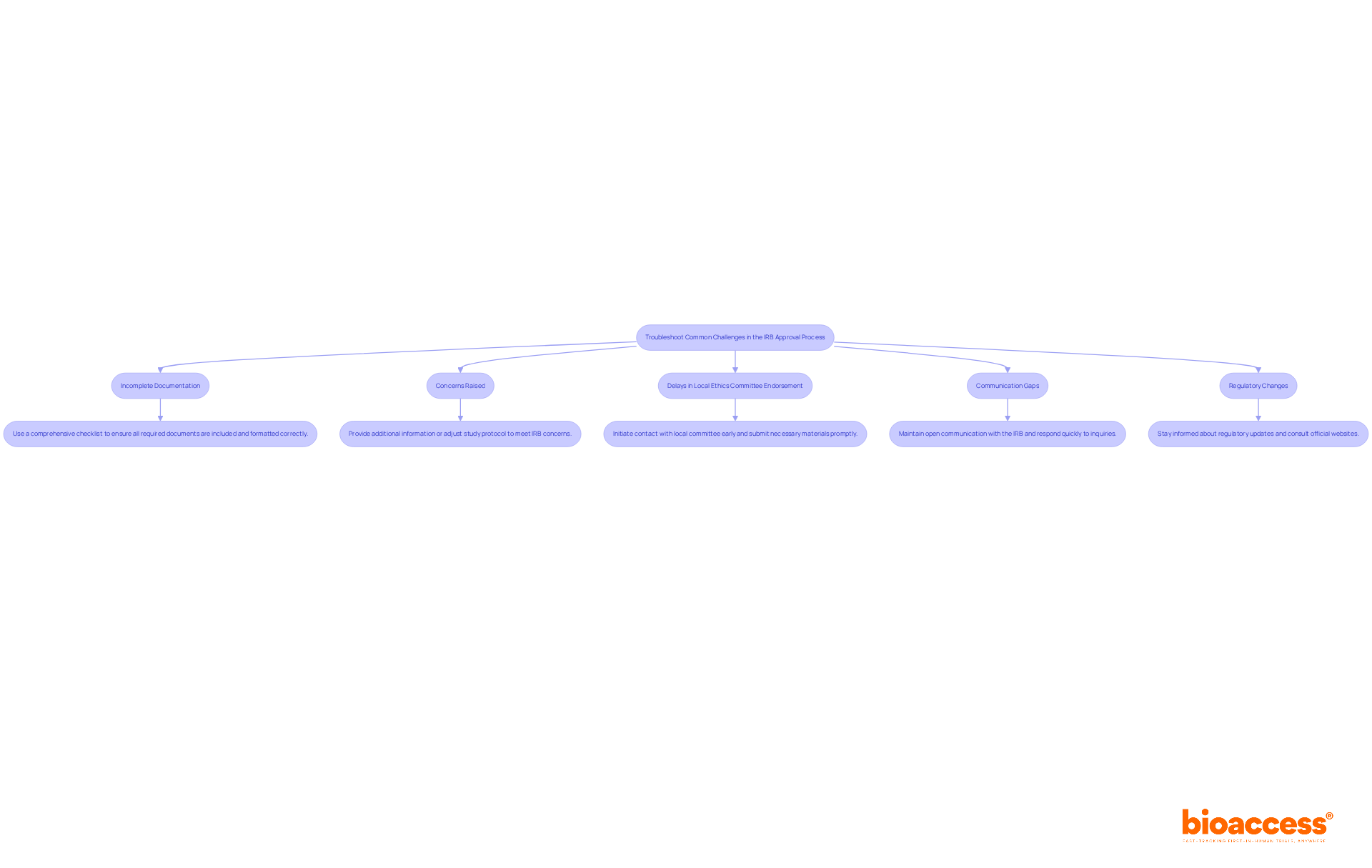
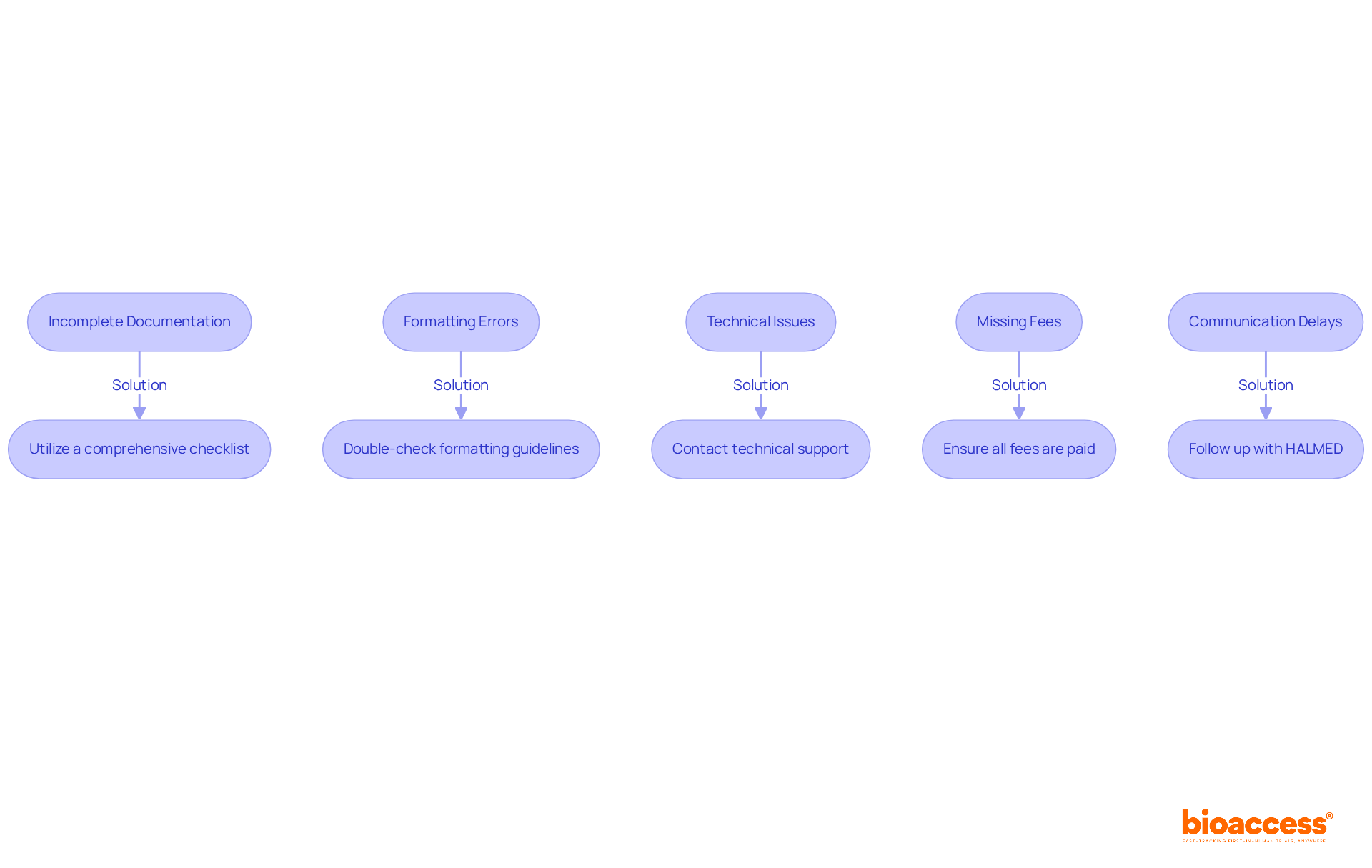
Address Common Challenges in the IRB Approval Process
The challenges associated with IRB approval in Bolivia are significant and can impede research progress. Here are some common issues and strategies to address them, particularly with the support of bioaccess®:
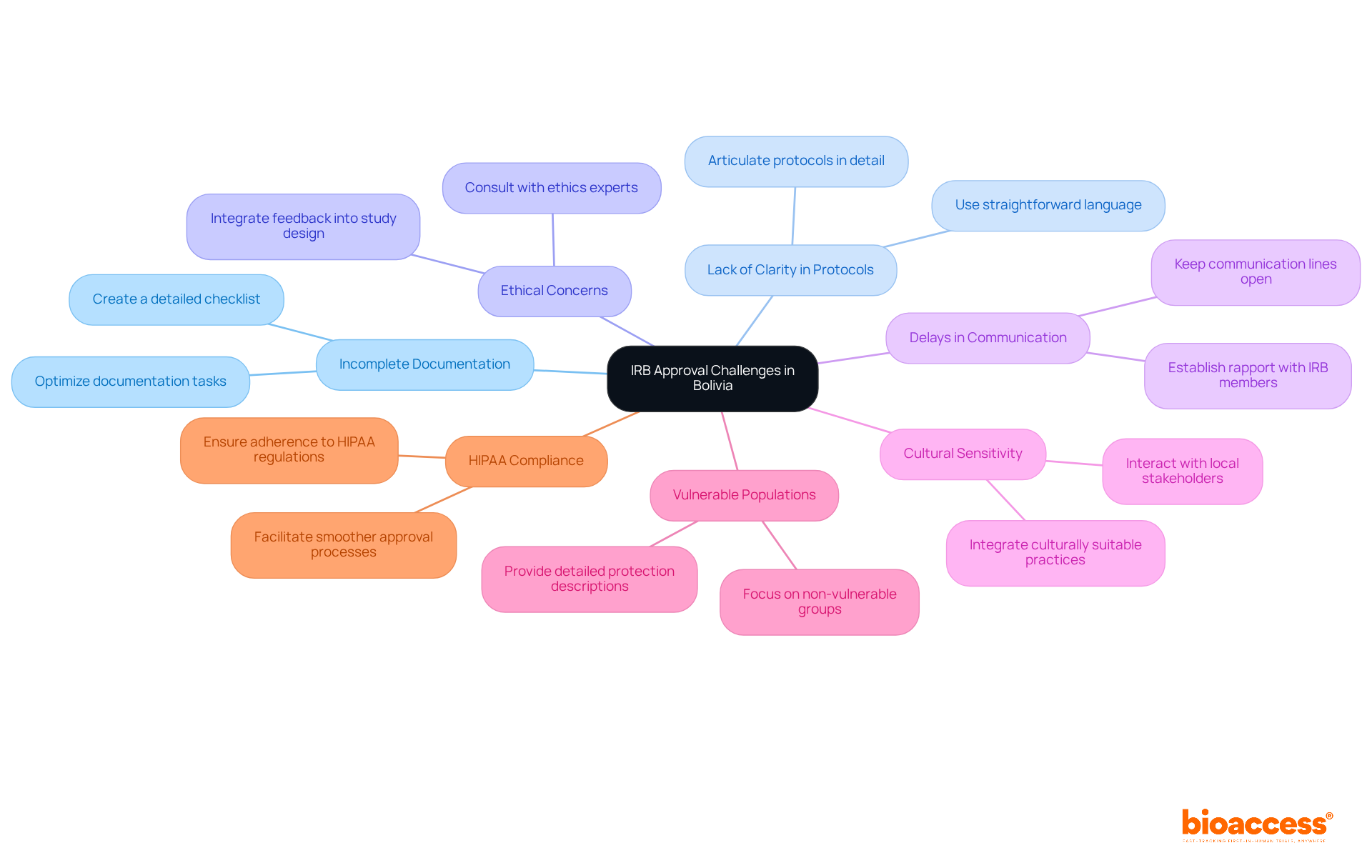
- Incomplete Documentation: A significant reason for delays is the submission of incomplete or improperly formatted documents. Creating a detailed checklist based on the IRB’s requirements can help researchers ensure that all documents are thoroughly reviewed before submission. This proactive approach can significantly reduce the risk of delays. bioaccess® has assisted clients in optimizing their documentation tasks, leading to quicker approvals.
- Lack of Clarity in Protocols: IRBs often request clarifications if the research protocol lacks clarity. To avoid this, researchers should articulate their protocols in detail, using straightforward language and avoiding jargon. Clear protocols enable smoother communication with the IRB and accelerate the review timeline. One client noted that with bioaccess®’s assistance, their protocol was approved without any requests for clarification, saving valuable time.
- Ethical Concerns: Potential ethical issues, such as inadequate informed consent processes or insufficient risk mitigation strategies, can lead to delays. Researchers should proactively address these concerns by consulting with ethics experts and integrating their feedback into the study design. This not only enhances the protocol but also demonstrates a commitment to ethical research practices. bioaccess®’s team includes ethics professionals who have successfully guided clients through complex ethical landscapes, ensuring compliance with local regulations.
- Delays in Communication: Delays in communication can lead to prolonged approval timelines, causing frustration for researchers. To mitigate this, researchers must keep communication lines open with the IRB. Promptly responding to requests for additional information and following up on submission statuses can make a significant difference. Establishing a rapport with IRB members can also facilitate quicker resolutions. bioaccess® enables effective communication channels, ensuring that researchers receive timely updates and support throughout the approval stage.
- Cultural Sensitivity: Understanding local cultural norms and ethical considerations is crucial in Bolivia. Researchers should interact with local stakeholders and integrate culturally suitable practices into their study design. This approach not only enhances acceptance by the IRB but also fosters trust within the community, which is vital for successful patient recruitment. bioaccess® emphasizes the importance of cultural competence in its operational strategies, helping researchers navigate these nuances effectively.
- Vulnerable Populations: Studying vulnerable populations can add months to the IRB approval process. Researchers should consider focusing on individuals who do not belong to vulnerable groups to avoid complications and expedite their submissions. bioaccess® can offer insights into the demographics of potential participants, assisting in the selection of suitable populations for trials.
- HIPAA Compliance: The IRB assesses adherence to HIPAA regulations in studies involving human subjects. Researchers must ensure that their investigations comply with these guidelines to facilitate a smoother approval. bioaccess® assists in ensuring that all research protocols comply with HIPAA and other relevant regulations, further expediting the approval timeline.
By embracing these strategies, researchers can not only streamline their IRB approval process in Bolivia but also enhance the integrity of their studies in the country.
Conclusion
Navigating the IRB approval process in Bolivia is not just a step; it’s a critical gateway for ethical clinical trials. This complex process is crucial for safeguarding participant rights and ensuring ethical standards. Understanding the role of Institutional Review Boards and meticulously preparing documentation will empower researchers to navigate the submission timeline effectively and secure approval.
Throughout this guide, we’ve highlighted essential points, including:
- The importance of early engagement with IRBs
- The meticulous preparation of required documents
- The awareness of common challenges that may arise during the approval process
Addressing these elements proactively can streamline the journey toward obtaining IRB approval, ultimately facilitating timely and effective clinical research.
The significance of navigating the IRB approval process in Bolivia cannot be overstated. It not only ensures compliance with ethical standards but also positions researchers to take advantage of the strategic benefits that Latin America offers for early-stage clinical trials, such as speed and cost efficiency. Those who master this process will not only ensure compliance but also unlock the potential for groundbreaking advancements in healthcare.
Frequently Asked Questions
What is the role of Institutional Review Boards (IRBs) in Bolivia?
In Bolivia, IRBs are responsible for ensuring that clinical trial proposals meet ethical standards and protect the rights and welfare of participants. They evaluate study protocols, informed consent documents, and recruitment materials, assessing potential risks and benefits while adhering to local regulations and international guidelines like ICH-GCP.
Why is it important for researchers to engage with IRBs early in their planning?
Engaging with IRBs early helps ensure that research protocols meet ethical standards and regulatory requirements, which can streamline the approval process and reduce potential delays in the study timeline.
What is the expected impact of IRB approval in Bolivia by 2026?
By 2026, the presence of IRB approval in Bolivia is expected to enhance ethical standards in clinical trials, improve ethical oversight, and increase participation in international collaborations, demonstrating a commitment to community engagement.
What does the IRB approval process entail for researchers in Bolivia?
The IRB approval process involves submitting a comprehensive proposal that includes the project’s objectives, methodology, and ethical considerations. Researchers must familiarize themselves with specific IRB requirements, which can vary between institutions.
What support is available for research initiatives in Bolivia?
Research initiatives in Bolivia are supported by funding from the National Institute for Health Research (NIHR), highlighting the importance of ethical standards in advancing clinical research.
How can the IRB process affect the success of clinical trials in Bolivia?
Understanding and navigating the IRB approval process is critical for ensuring the integrity and success of clinical research in Bolivia, as it directly influences both the approval timeline and the overall feasibility of studies.
List of Sources
- Understand the Role of Institutional Review Boards (IRBs) in Bolivia
- The Research Ethics Committee in Bolivia Strengthens Efforts to Promote International-Scale Research – NIHR (https://nihrlatamcentre.com/the-research-ethics-committee-in-bolivia-strengthens-efforts-to-promote-international-scale-research)
- Prepare Required Documentation for IRB Submission
- Required Documents for IRB Submissions – Solutions IRB (https://solutionsirb.com/required-documents-for-irb-submissions)
- Is Institutional Review Board (IRB) Approval Necessary for Usability Tests? (https://emergobyul.com/news/institutional-review-board-irb-approval-necessary-usability-tests)
- How to Draft a Research Plan for IRB Review: A Step-by-Step Guide | WCG (https://wcgclinical.com/insights/drafting-a-research-plan-for-irb-review-and-research-conduct)
- Regulatory Submissions | Clinical Trials | dicentra.com (https://dicentra.com/clinical-trials/regulatory-submissions)
- Navigate the IRB Submission Timeline and Review Process
- 4 Steps For A Successful First-In-Human Clinical Trial In Bolivia | bioaccess® (https://bioaccessla.com/blog/4-steps-for-a-successful-first-in-human-clinical-trial-in-bolivia)
- Is Institutional Review Board (IRB) Approval Necessary for Usability Tests? (https://emergobyul.com/news/institutional-review-board-irb-approval-necessary-usability-tests)
- Precision receives CTA approval to expand ELIMINATE-B study (https://clinicaltrialsarena.com/news/precision-cta-approval-eliminate-b-study)
- Early Feasibility Studies in Latin America (https://greenlight.guru/blog/early-feasibility-studies-in-latin-america)
- Address Common Challenges in the IRB Approval Process
- Avoiding Hurdles in Your IRB Application (https://statisticssolutions.com/avoiding-hurdles-in-your-irb-application)