Introduction
Understanding the intricacies of remote audits is essential for navigating compliance within Brazil’s health regulatory landscape. As organizations adapt to ANVISA’s evolving guidelines, particularly in light of recent global shifts, the stakes for ensuring a successful and secure remote audit have never been higher.
What critical steps must organizations take to not only meet these regulatory standards but also enhance their operational integrity in the process? This article delves into key strategies and best practices that empower organizations to excel in their remote audit endeavors while maintaining compliance with ANVISA’s rigorous expectations.
Understand ANVISA’s Regulatory Framework for Remote Audits
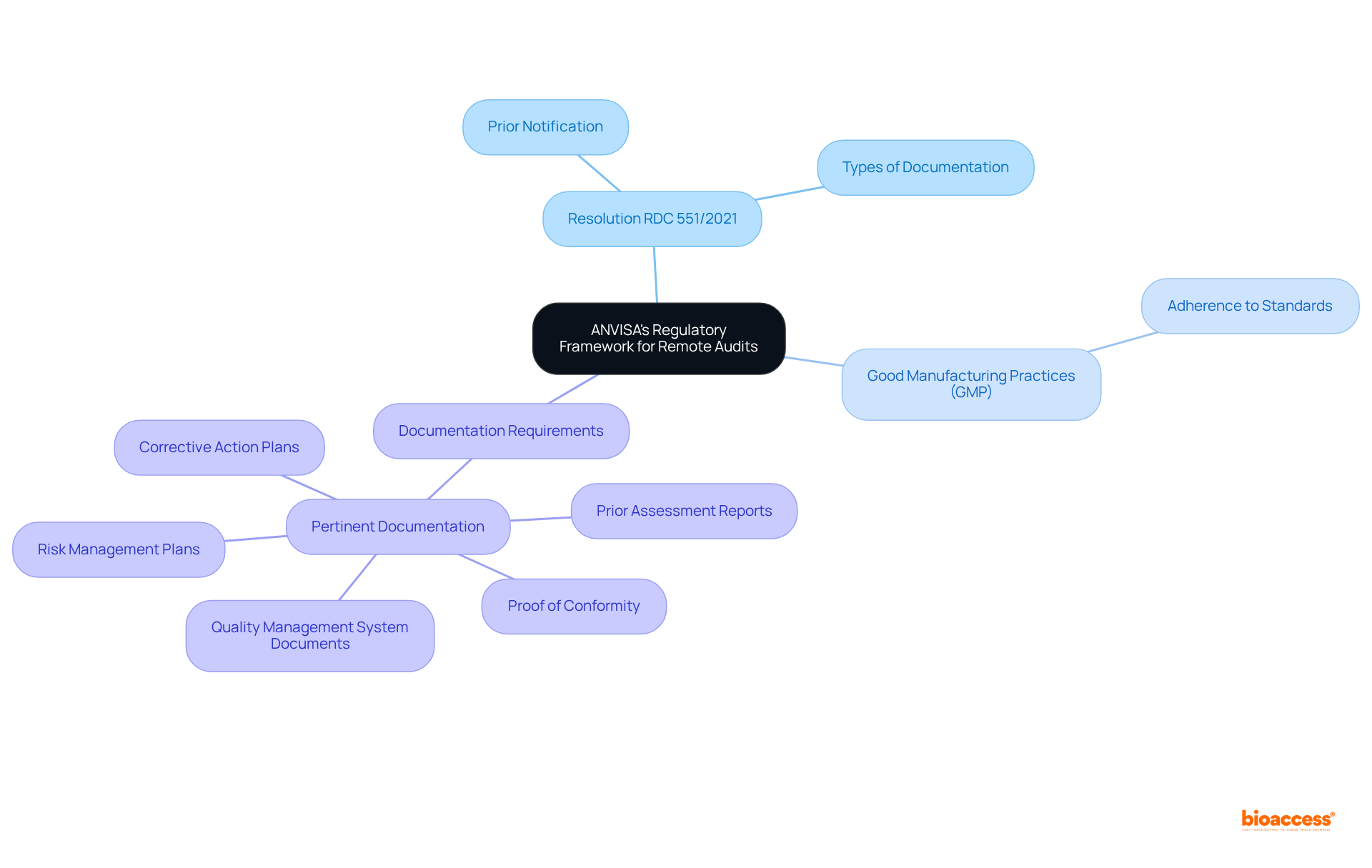
To effectively manage the evaluations, it is essential to familiarize oneself with the regulatory framework governing these processes. ANVISA, the Brazilian Health Regulatory Agency, has established specific guidelines for , particularly in response to recent global shifts towards virtual assessments. Key regulations include:
- : This regulation outlines the requirements for remote evaluations, including the necessity for prior notification and the that must be made available. Understanding the can significantly enhance the effectiveness of the .
- : Familiarity with GMP is crucial, as adherence to these standards is a primary concern during evaluations. It is important to understand the relevant GMP guidelines related to your specific sector, since following these practices can streamline the evaluation process.
- : ANVISA mandates that all pertinent documentation must be accessible during the evaluation. This includes , prior assessment reports, and any corrective action plans. Additionally, organizations should prepare specific documents such as risk management plans and proof of conformity with relevant technical standards, which are vital for demonstrating compliance with regulatory expectations.
By comprehending these regulations, organizations can better prepare for the evaluation and ensure alignment with ANVISA’s expectations for a . Integrating insights from case studies, such as ANVISA’s , can further illustrate the practical implications of these regulations and enhance the overall understanding of the evaluation process.
Prepare Essential Documentation and Protocols for the Audit
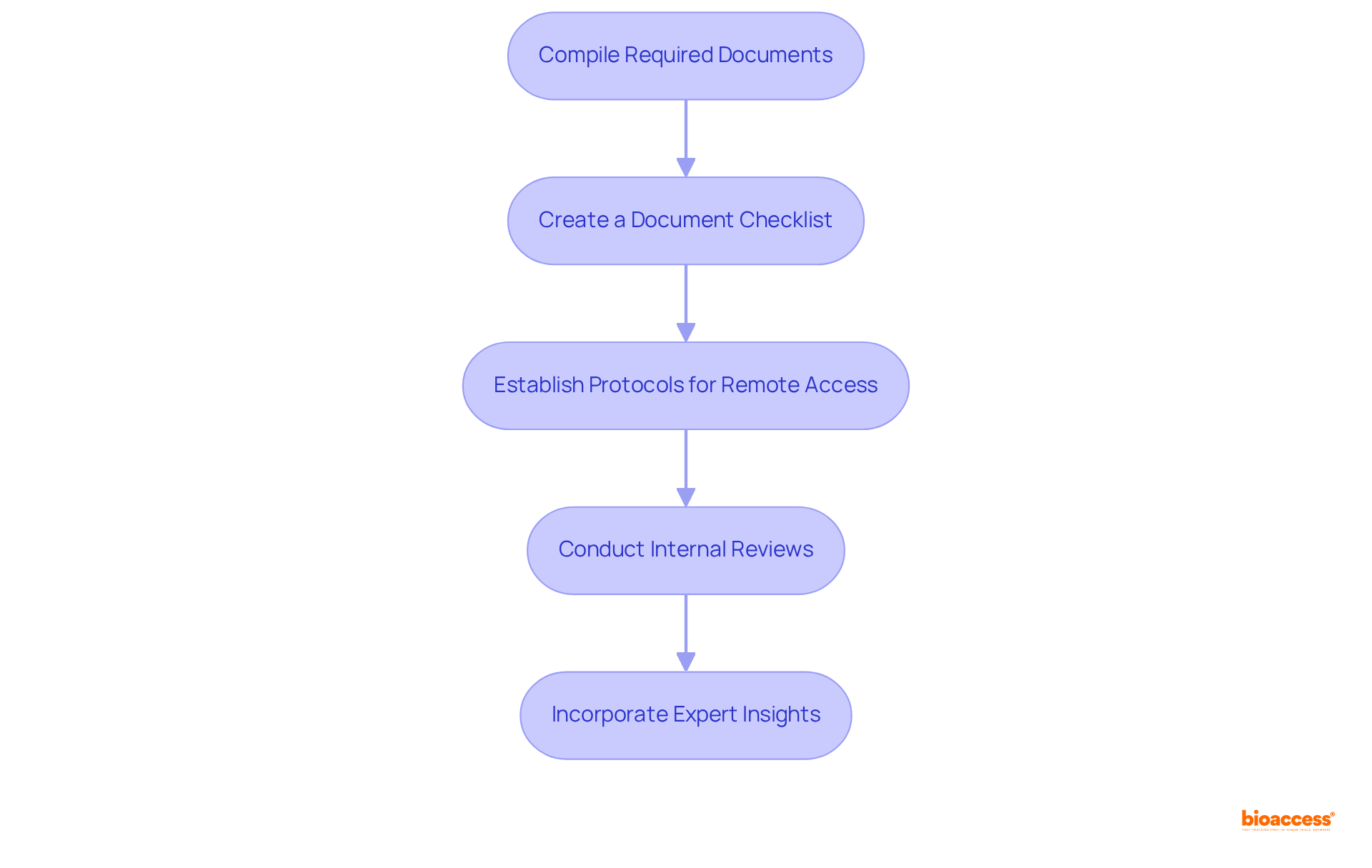
Preparation is essential for a successful . To ensure that all necessary documentation and protocols are in place, follow these critical steps:
- : Gather all relevant documents, including documentation, previous audit reports, , training records for staff involved in the audit, and corrective action plans from previous audits.
- : Develop a checklist to ensure that all required documents are accounted for. This organized method simplifies the review process and reduces the chance of missing essential items.
- : Store all documents in a secure, accessible location. Employing cloud-based solutions is advised, as approximately 70% of organizations now utilize such technologies for documentation purposes, enabling easy sharing and collaboration during the review.
- as part of the to ensure that all documentation is up to date and adheres to regulations before the examination. This proactive measure aids in recognizing and correcting any gaps prior to the formal evaluation.
- : As Ana Prelec, Head of Quality Assurance Department, emphasizes, “Preparation is vital as it is a factor that you have control of.” This underscores the significance of thorough preparation in ensuring a seamless review process.
By following these steps, organizations can demonstrate a well-structured and compliant appearance during the remote evaluation, ultimately improving their chances of a favorable result.
Execute the Remote Audit: Best Practices and Tools
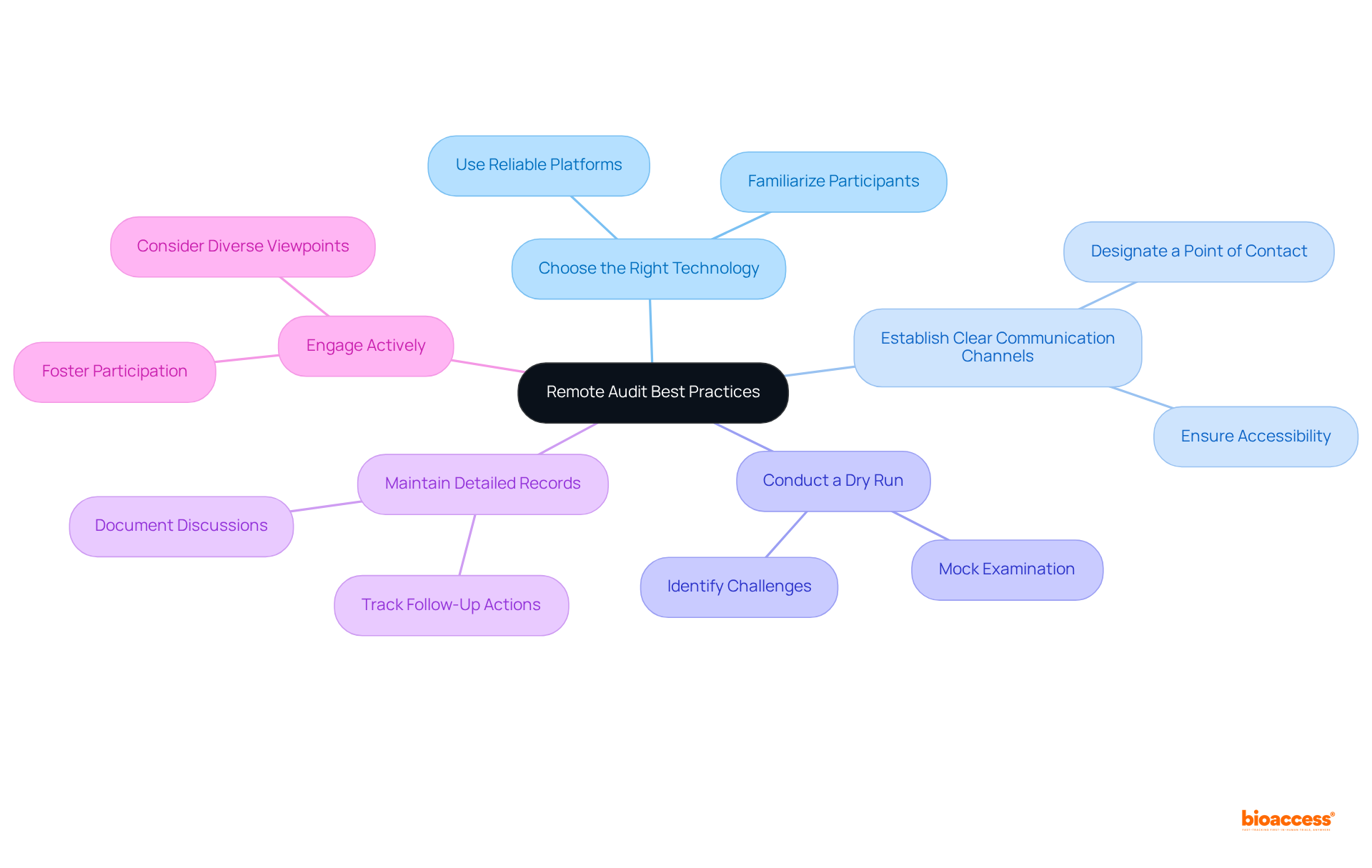
Conducting a remote evaluation necessitates meticulous planning and the right technological tools. Essential to follow include:
- Choose the Right Technology: Select reliable , which facilitate and effective screen sharing. It is imperative that all participants are well-acquainted with the technology prior to the assessment to minimize disruptions.
- Establish Channels: Designate a specific point of contact for the review and ensure that all team members know how to reach them. This approach enables quick resolution of any issues that may arise during the evaluation.
- : Prior to the actual review, execute a mock examination to assess both the technology and the processes involved. This practice aids in identifying potential challenges and allows for necessary adjustments, ensuring a smoother experience.
- Maintain : Throughout the review, meticulously document discussions, findings, and any necessary follow-up actions. This documentation is crucial for addressing any issues that may surface post-audit and for .
- Engage Actively: Foster during the review. This not only demonstrates commitment but also ensures that diverse viewpoints are considered, enhancing the evaluation process.
By adhering to these , organizations can significantly enhance their experience with secure , ensuring compliance with ANVISA’s rigorous standards. For instance, Quattris successfully managed a project with an annual revenue of approximately $700 million, underscoring the importance of efficient evaluations in high-stakes environments. Furthermore, statistics indicate that remote evaluations utilizing video conferencing tools have achieved success rates exceeding 85%, highlighting the effectiveness of these technologies in maintaining integrity and efficiency. As Quattris stated, “To guarantee the seamless implementation of the project, we designated a committed project manager along with a technical team leader for each location,” emphasizing the critical role of organized oversight in remote evaluations.
Address Findings and Ensure Continuous Compliance Post-Audit
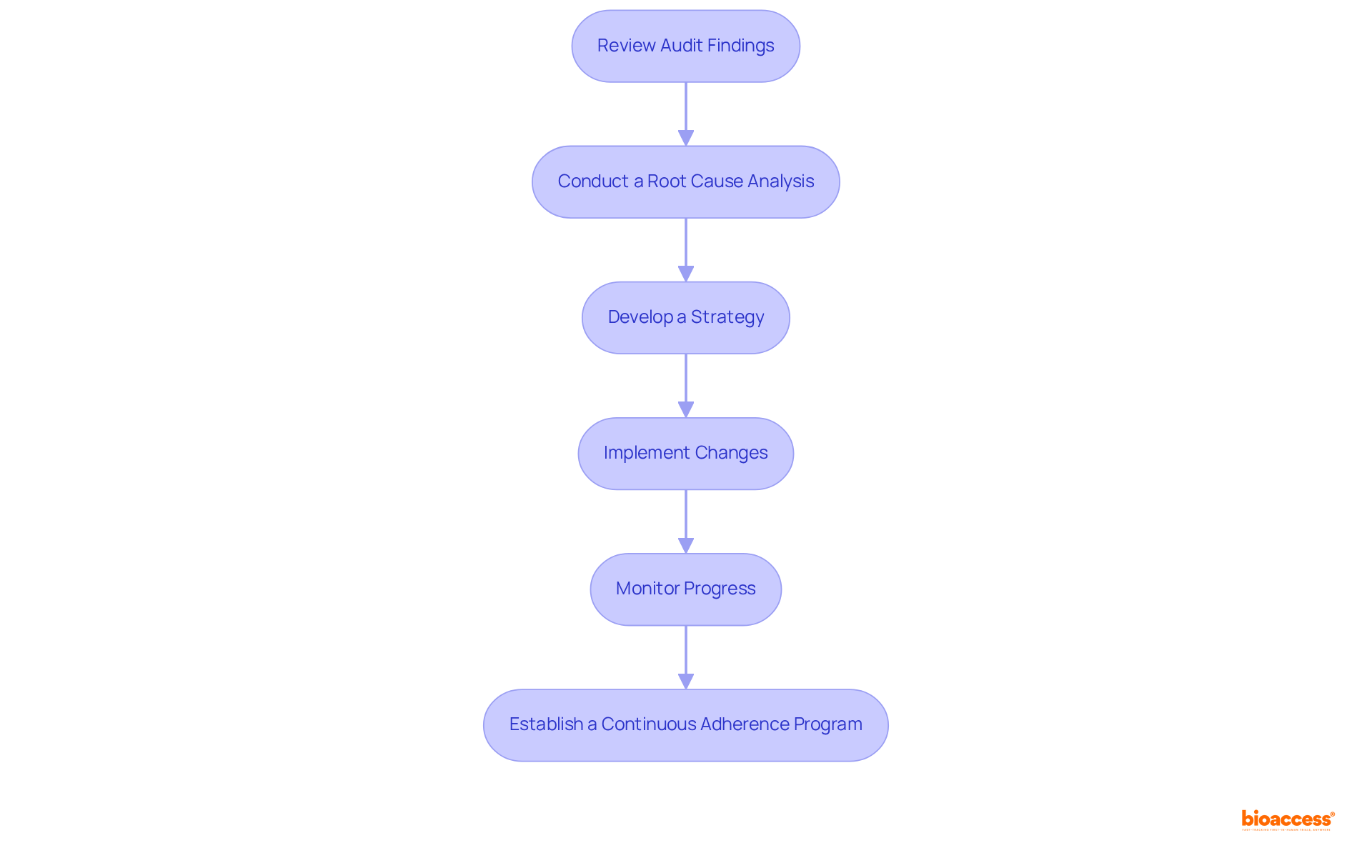
After a distant evaluation, addressing discoveries and ensuring ongoing adherence is crucial for organizational integrity. The following steps outline an effective :
- Review Audit Findings: Thoroughly analyze the to pinpoint areas of non-compliance and opportunities for improvement. Prioritize these findings based on their potential impact on .
- Conduct a : Before creating a plan, perform a to identify the underlying issues that led to the findings. This step is vital for guaranteeing that the tackle the core issues efficiently.
- Develop a Strategy: Formulate a comprehensive strategy tailored to each finding. This plan should outline specific , assign responsibilities to team members, and establish clear timelines for completion. A well-organized strategy is essential, as demonstrated by case studies where organizations that established clear guidelines experienced enhanced accountability and quicker resolution of issues.
- Implement Changes: Promptly execute the plan, ensuring that all team members understand their roles and the significance of compliance. Effective communication is key to fostering a culture of accountability.
- : Continuously track the implementation of and evaluate their effectiveness. Organizations should maintain detailed records of corrective actions to mitigate risks and ensure that changes are not only made but are also sustained over time. Regular follow-up evaluations or reviews can assist in this regard.
- Establish a : Create a robust program that includes regular training sessions, internal audits, and updates to documentation. This proactive approach not only meets ANVISA regulations but also facilitates a , thereby enhancing overall operational efficiency. It is important to recognize that the process of applying corrective actions should be iterative, with ongoing monitoring of progress and results.
By systematically addressing findings and fostering a culture of continuous compliance, organizations can navigate regulatory landscapes more effectively and improve their operational frameworks.
Conclusion
Understanding the complexities of secure remote audits under ANVISA’s regulations is crucial for organizations aiming to achieve compliance and operational excellence. Familiarity with ANVISA’s regulatory framework is essential, as is the effective preparation of necessary documentation, execution of audits following best practices, and addressing findings post-evaluation. By embracing these steps, organizations can navigate the audit process more effectively and enhance their overall compliance posture.
Key insights discussed include:
- The necessity of thorough preparation, such as compiling required documents and establishing secure protocols for remote access.
- Utilizing reliable technology and maintaining clear communication during the audit process are significant factors highlighted in this discussion.
- Addressing audit findings with a strategic approach ensures continuous compliance.
Collectively, these elements contribute to a streamlined and successful remote audit experience.
In a rapidly evolving regulatory landscape, organizations must prioritize adherence to ANVISA’s guidelines to maintain integrity and operational efficiency. Proactive measures, such as implementing a continuous compliance program and conducting regular training, foster a culture of accountability and preparedness. By committing to these practices, organizations not only meet regulatory expectations but also position themselves for long-term success in the industry.
Frequently Asked Questions
What is ANVISA?
ANVISA is the Brazilian Health Regulatory Agency responsible for overseeing health regulations in Brazil, including the management of remote audits.
What is the purpose of the regulatory framework for remote audits established by ANVISA?
The regulatory framework aims to provide guidelines for conducting secure remote audits in Brazil, particularly in response to the increasing shift towards virtual assessments.
What is Resolution RDC 551/2021?
Resolution RDC 551/2021 outlines the requirements for remote evaluations, including the necessity for prior notification and the types of documentation that must be made available during the audit process.
Why is familiarity with Good Manufacturing Practices (GMP) important for remote audits?
Familiarity with GMP is crucial because adherence to these standards is a primary concern during evaluations, and understanding the relevant GMP guidelines can help streamline the evaluation process.
What documentation is required during an ANVISA remote audit?
ANVISA requires that all pertinent documentation be accessible during the evaluation, including quality management system documents, prior assessment reports, corrective action plans, risk management plans, and proof of conformity with relevant technical standards.
How can organizations prepare for an ANVISA remote audit?
Organizations can prepare by understanding ANVISA’s regulations, ensuring all required documentation is accessible, and familiarizing themselves with GMP guidelines relevant to their sector.
How did the COVID-19 pandemic affect ANVISA’s regulatory framework?
The pandemic prompted ANVISA to adjust its regulations to accommodate remote audits, illustrating the agency’s flexibility in response to global shifts in assessment practices.
List of Sources
- Understand ANVISA’s Regulatory Framework for Remote Audits
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC8475452)
- ANVISA RDC 751: Technical Dossier Compliance Guide – Freyr (https://freyrsolutions.com/blog/anvisas-rdc-751-technical-dossier-regulatory-compliance)
- Prepare Essential Documentation and Protocols for the Audit
- martifarm.com (https://martifarm.com/how-to-prepare-for-remote-audits-7-tips-to-ease-stress-and-achieve-compliance)
- thefdagroup.com (https://thefdagroup.com/blog/remote-auditing-best-practices-checklist)
- fdamap.com (https://fdamap.com/blog/fdas-guidance-for-remote-audits-good-intentions-bad-perceptions)
- simplerqms.com (https://simplerqms.com/remote-auditing-best-practices)
- Execute the Remote Audit: Best Practices and Tools
- quattris.com (https://quattris.com/case_studies_category/manufacturing)
- Address Findings and Ensure Continuous Compliance Post-Audit
- linfordco.com (https://linfordco.com/blog/corrective-action-plans-for-audit-findings)
- yellowbook-cpe.com (https://yellowbook-cpe.com/action-plan-for-audit-findings.html)
- smith-howard.com (https://smith-howard.com/how-to-handle-and-respond-to-findings-in-a-single-audit)

Leave a Reply