Introduction
In the dynamic landscape of medical technology, navigating regulatory frameworks is pivotal for success, particularly in Brazil, where ANVISA, the Brazilian Health Regulatory Agency, stands as the guardian of public health. This agency not only ensures that medical devices meet stringent safety and efficacy standards but also shapes the pathways for innovation in healthcare.
With recent updates to its regulatory agenda, ANVISA is adapting to emerging technologies and evolving market demands, making it essential for Medtech companies to grasp its classification system and compliance requirements.
As the industry witnesses a surge in transformative technologies like robotics, understanding ANVISA’s role becomes increasingly critical for organizations aiming to thrive in this competitive environment.
This article delves into the intricacies of ANVISA’s regulations, offering insights into effective strategies for compliance, documentation preparation, and best practices for engaging with the agency to facilitate successful market entry.
Understand ANVISA: The Regulatory Authority for Medtech in Brazil
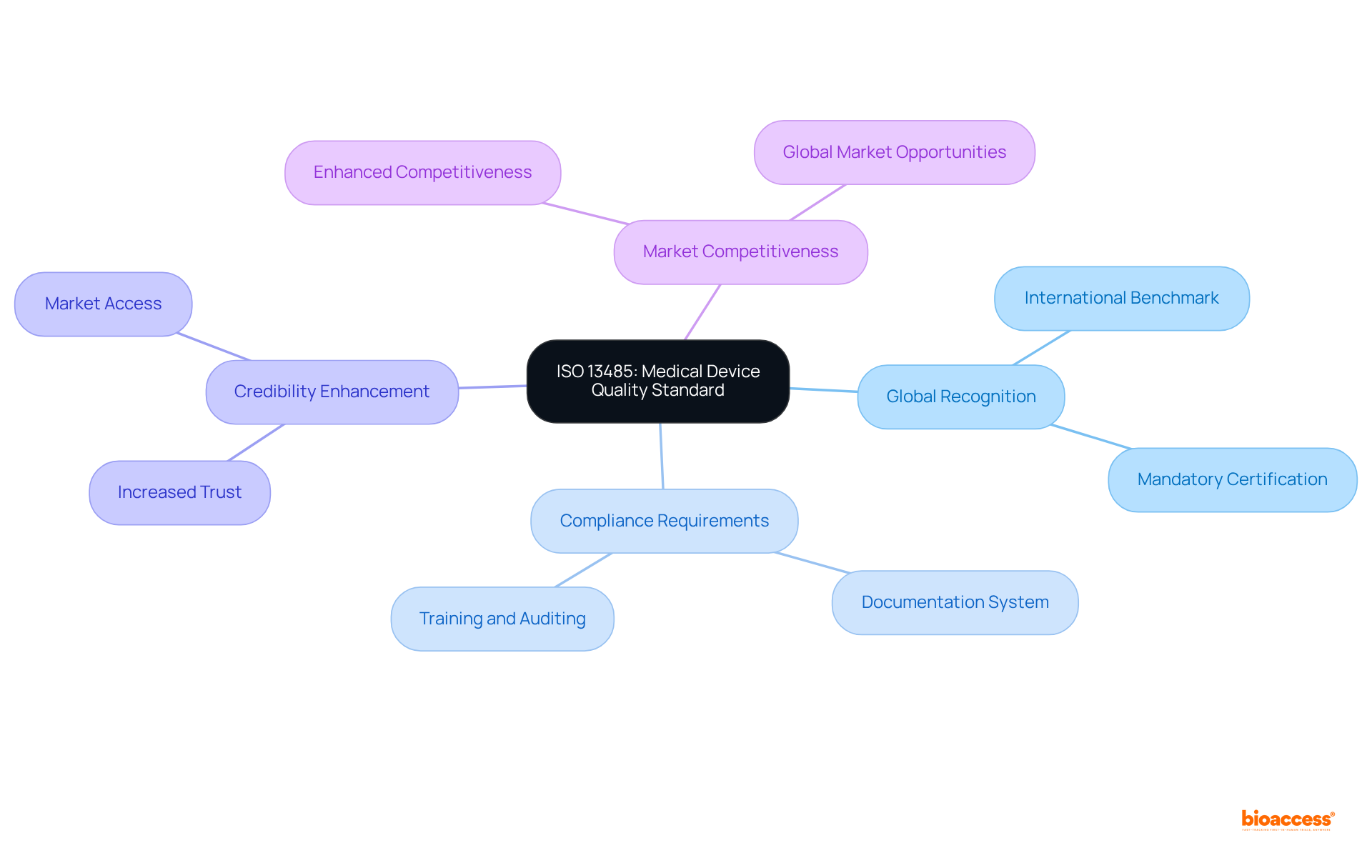
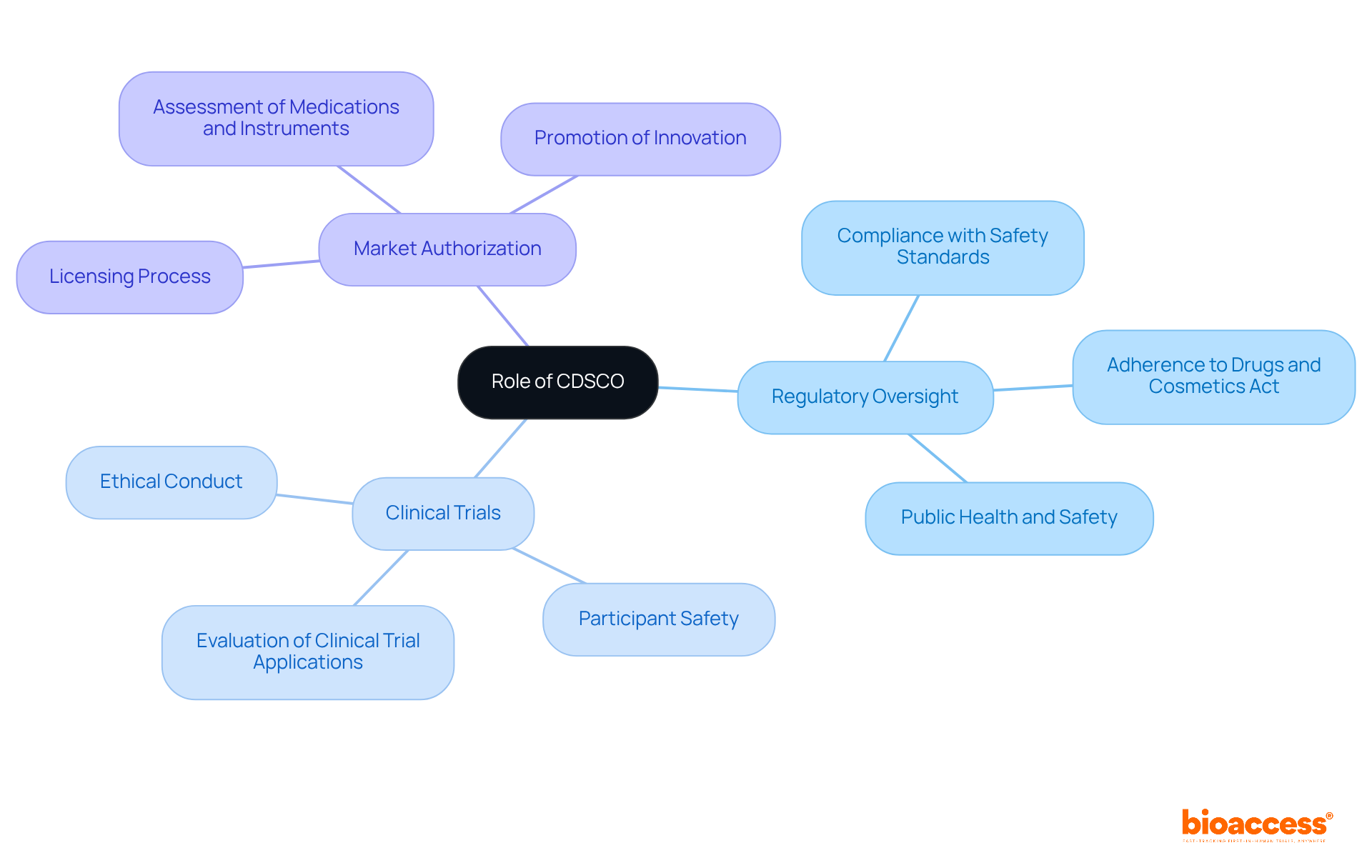
The Brazilian Health Regulatory Agency serves as a pivotal organization, overseeing health-related products, pharmaceuticals, and equipment in Brazil. Established to safeguard public health, this regulatory body mandates that all , efficacy, and quality standards prior to market entry. Understanding the agency’s classification system is vital, as it categorizes items based on their associated risk levels. This classification significantly influences the approval process and the specific requirements for each item, thereby shaping timelines and strategies for compliance.
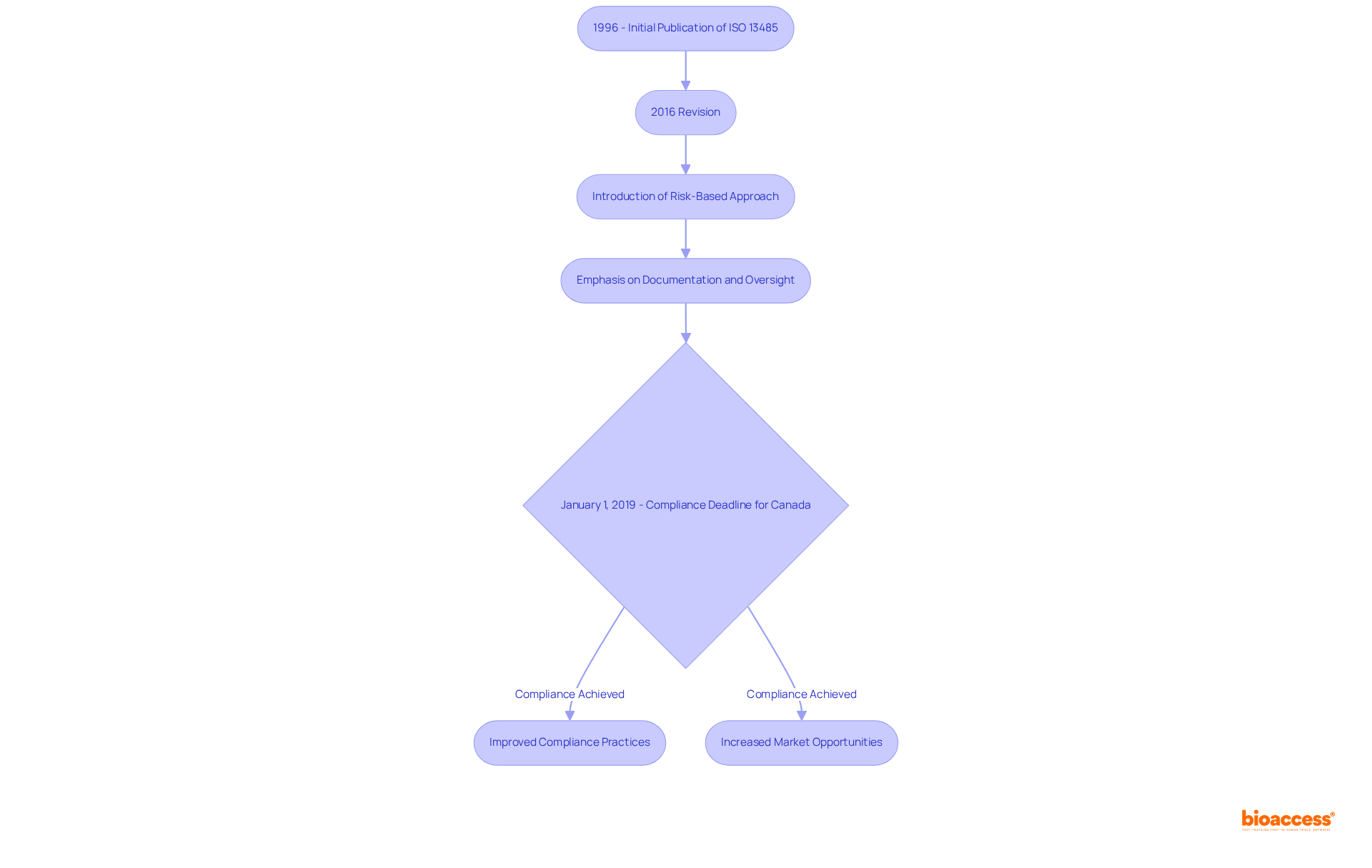
In 2025, updated its oversight agenda, underscoring the need for ongoing adaptation to emerging technologies and market demands. Recent statistics reveal that the regulatory agency necessitates data from subjects to be excluded from pharmacokinetic and statistical analyses, demonstrating a commitment to maintaining rigorous data integrity in . This is crucial for ensuring that healthcare devices not only comply with safety standards but also deliver effective solutions in the healthcare sector.
The rise of robotics as a transformative technology in healthcare further highlights the shifting landscape of healthcare devices and their regulatory ramifications. Expert opinions underscore the essential role of the , emphasizing its influence on the successful introduction of innovative medical technologies. Compliance narratives from various Medtech firms illustrate the importance of adhering to , showcasing how following these guidelines can lead to successful market entry and enhanced patient safety.
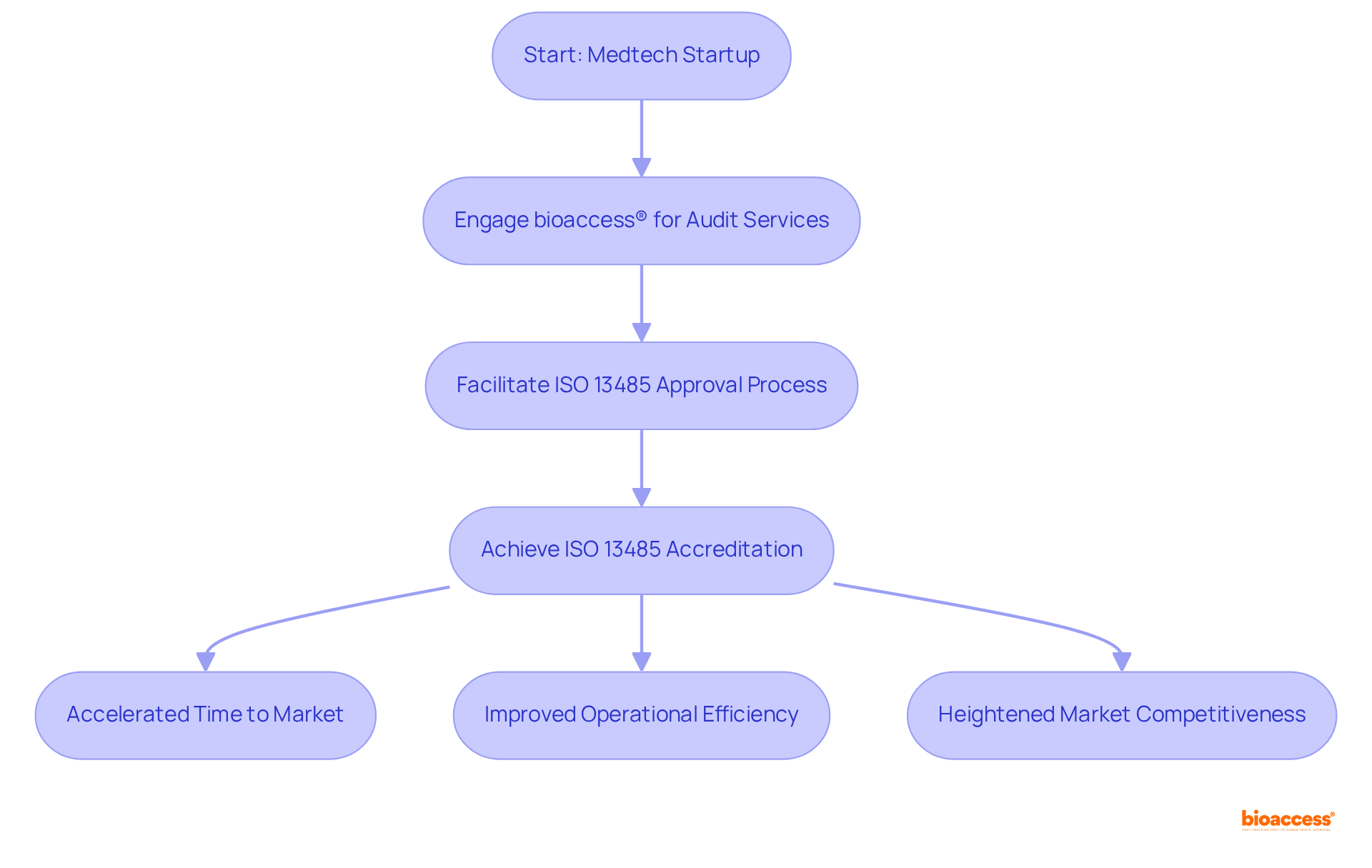
For instance, bioaccess®, a leading CRO in Latin America, has demonstrated a successful track record in conducting studies for . With over 20 years of experience in Medtech, bioaccess® provides , including:
- Site selection
- Compliance reviews
- Trial setup
- Import permits
- Project management
- Reporting
These services are indispensable for navigating the intricacies of regulatory approval. As the landscape of continues to evolve, staying informed about regulatory updates and requirements is crucial for any organization aiming to thrive in the Brazilian market.
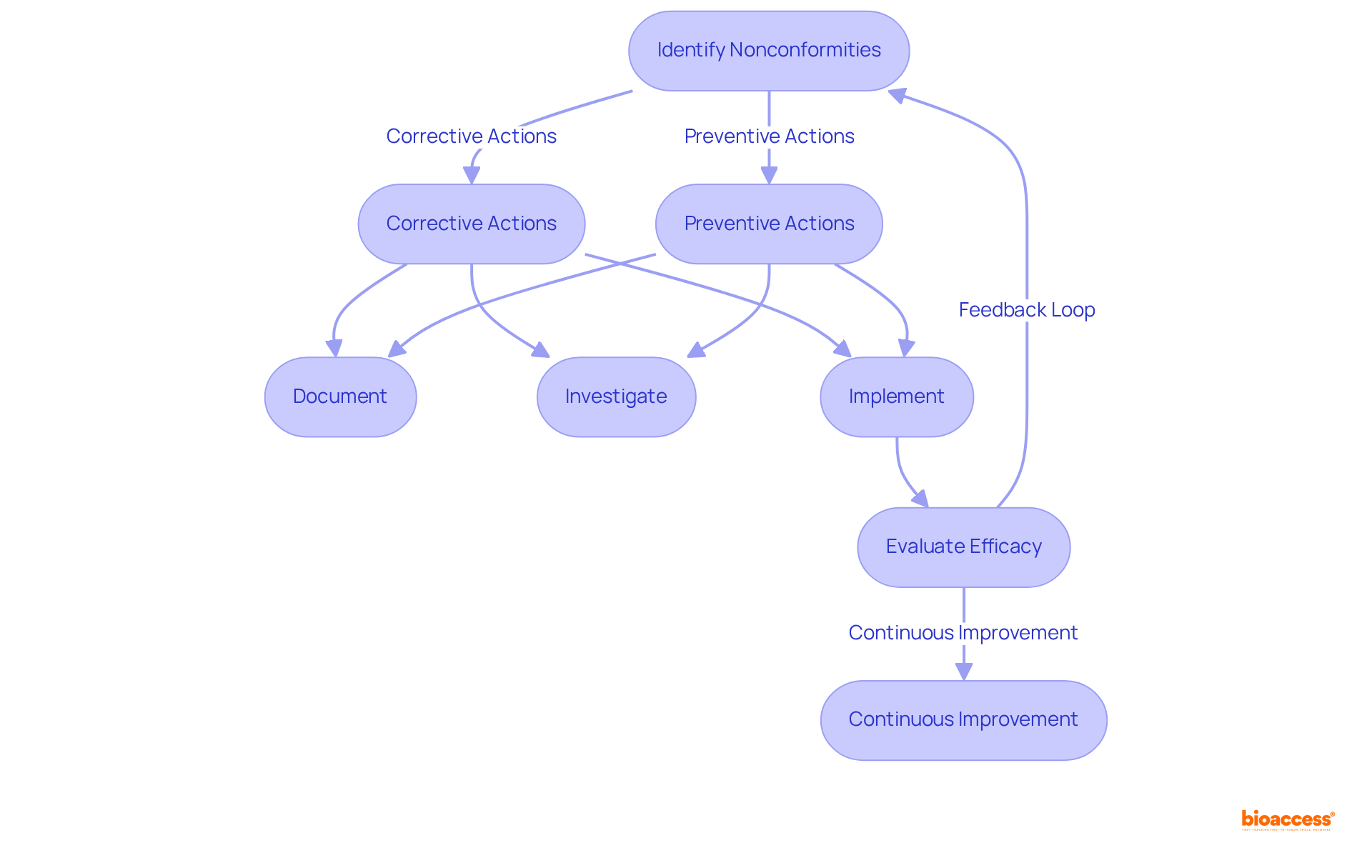
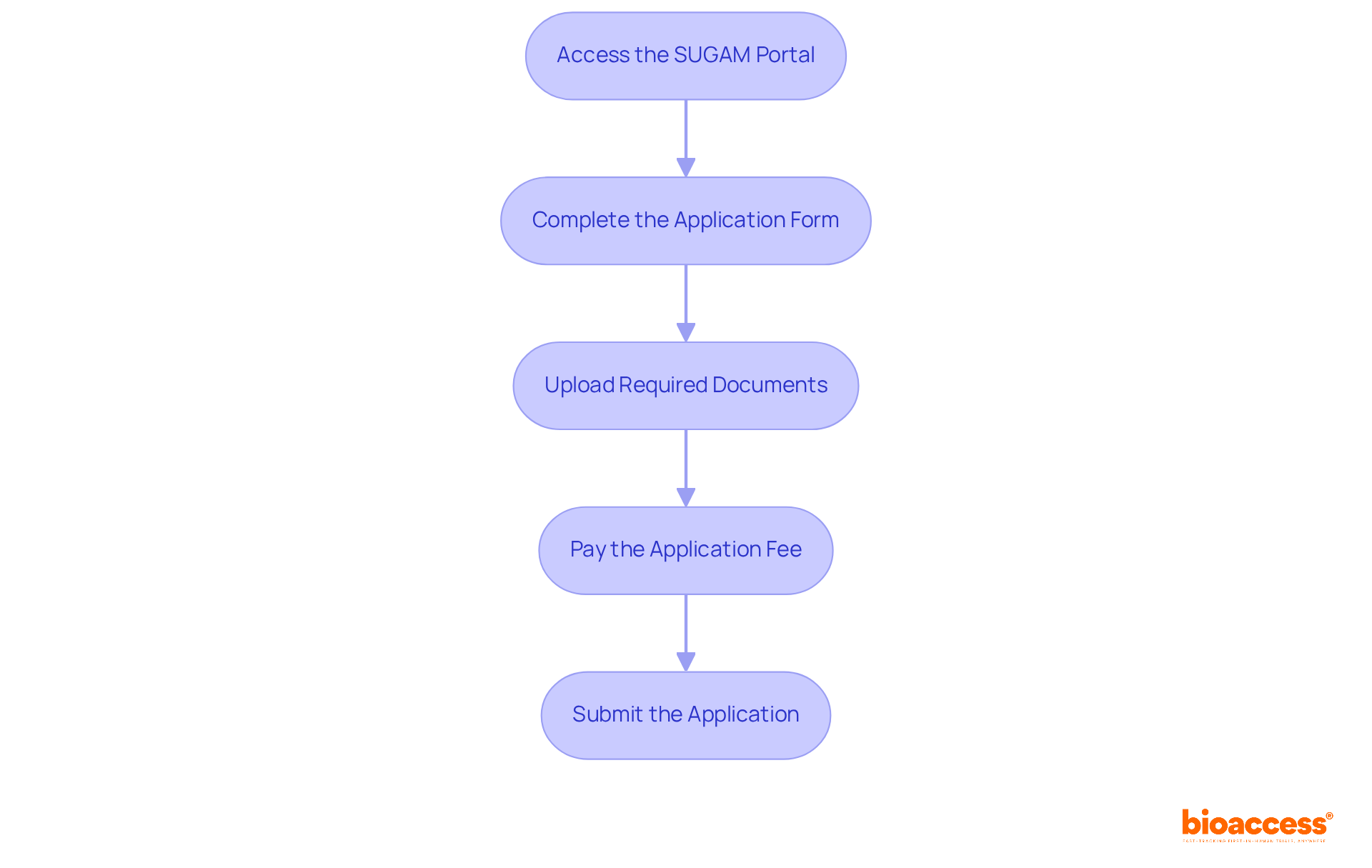
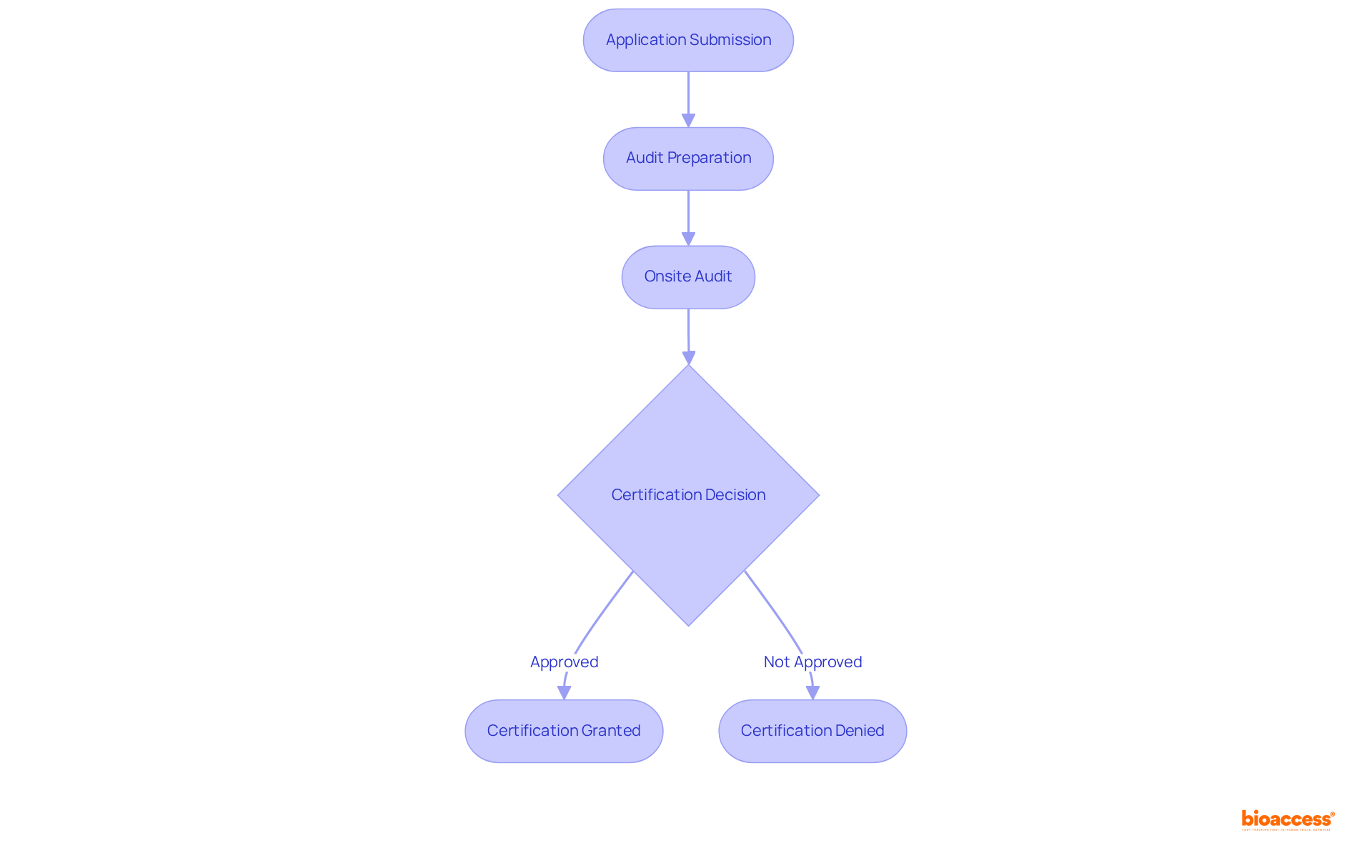
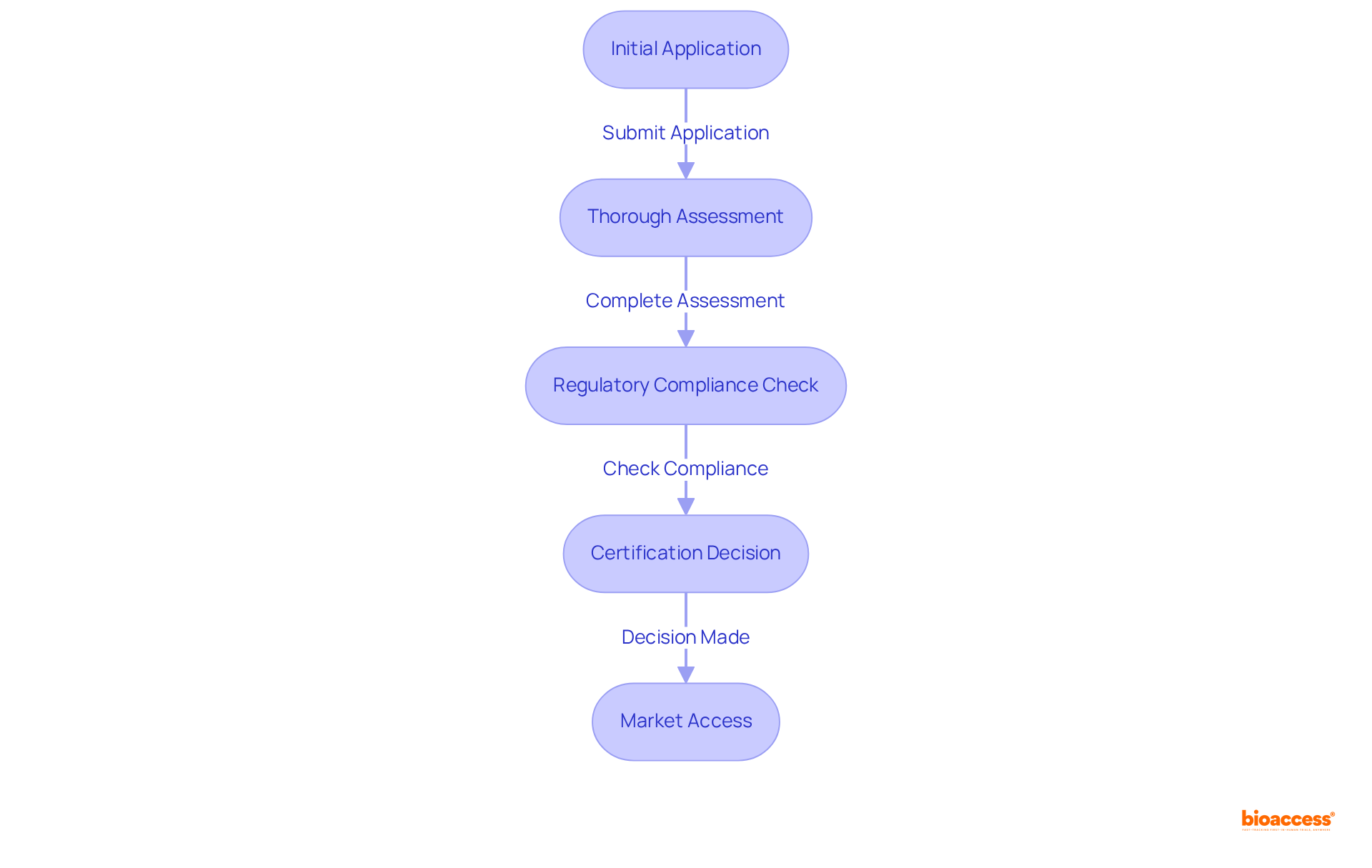
Follow the Step-by-Step Process for ANVISA Compliance
To achieve compliance with the , it is essential to follow these critical steps:
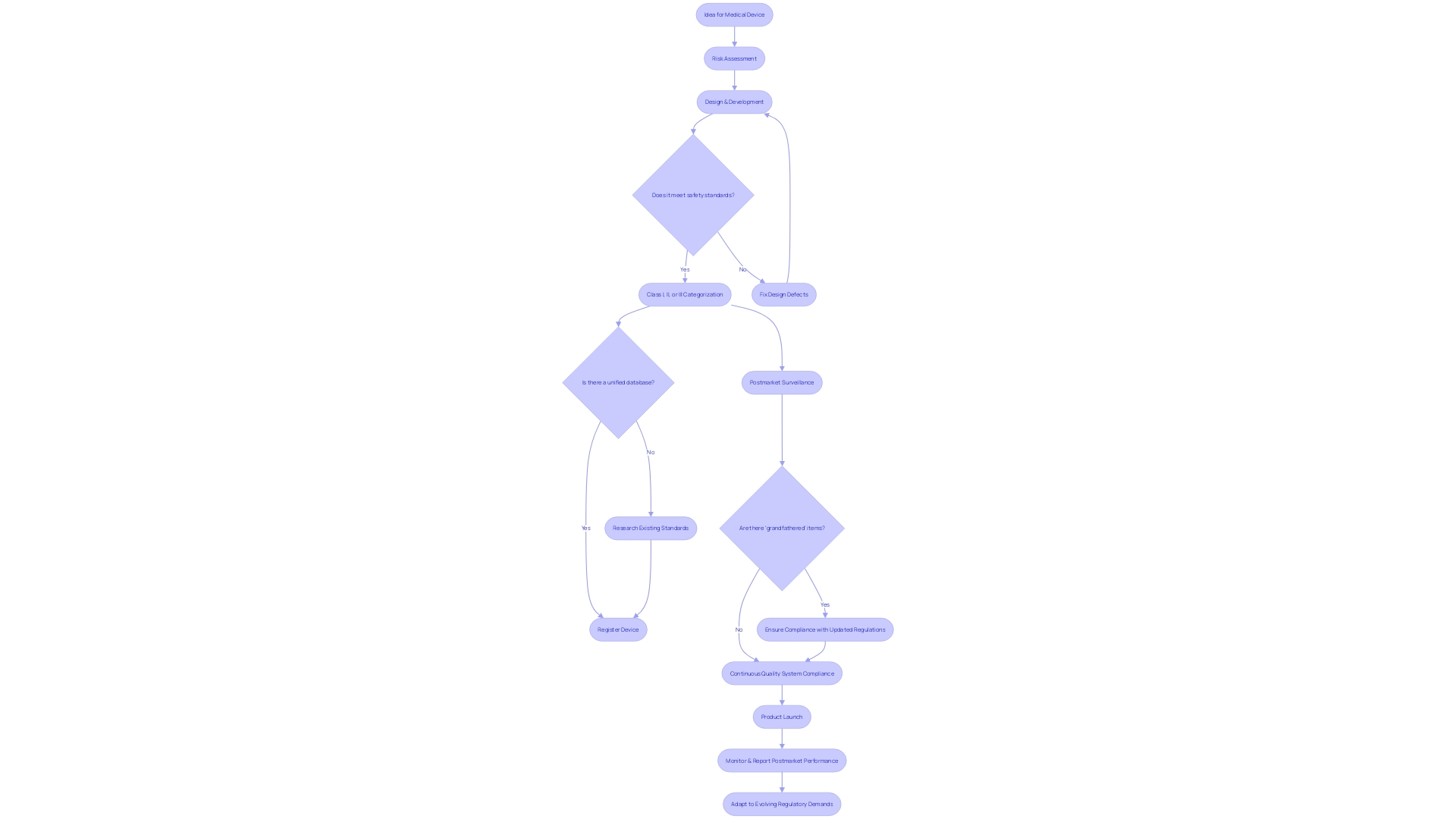
- Determine Device Classification: Assess your based on its intended use and associated risk level. This classification is vital, as it determines the compliance pathway you must follow. Notably, Class III and IV registrations are valid for ten years, while Class I and II registrations do not expire. Understanding these classifications is crucial, as they significantly influence your , especially in light of .
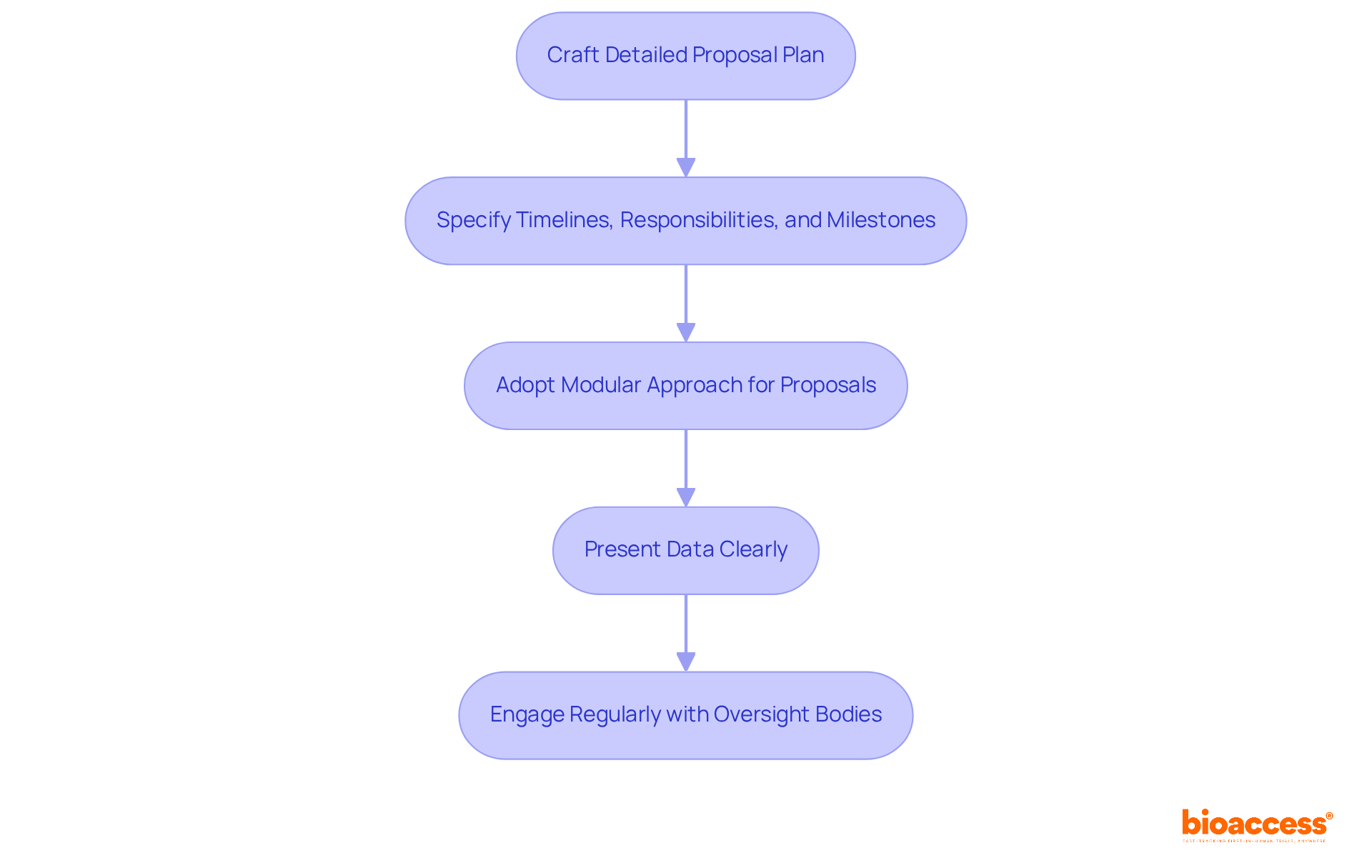
- Prepare for Submission: Compile all necessary documentation, including technical files, , and . Thorough preparation can streamline the approval process by adhering to . At bioaccess®, we offer extensive assistance in preparing these documents to meet the strict criteria set by the regulatory agency.
- Submit Application: Complete the registration application through their electronic platform (e-PL) and provide your documentation. This step initiates the formal review process. Our team at bioaccess® can assist you in navigating this process efficiently while following the , ensuring that all submissions are accurate and complete.
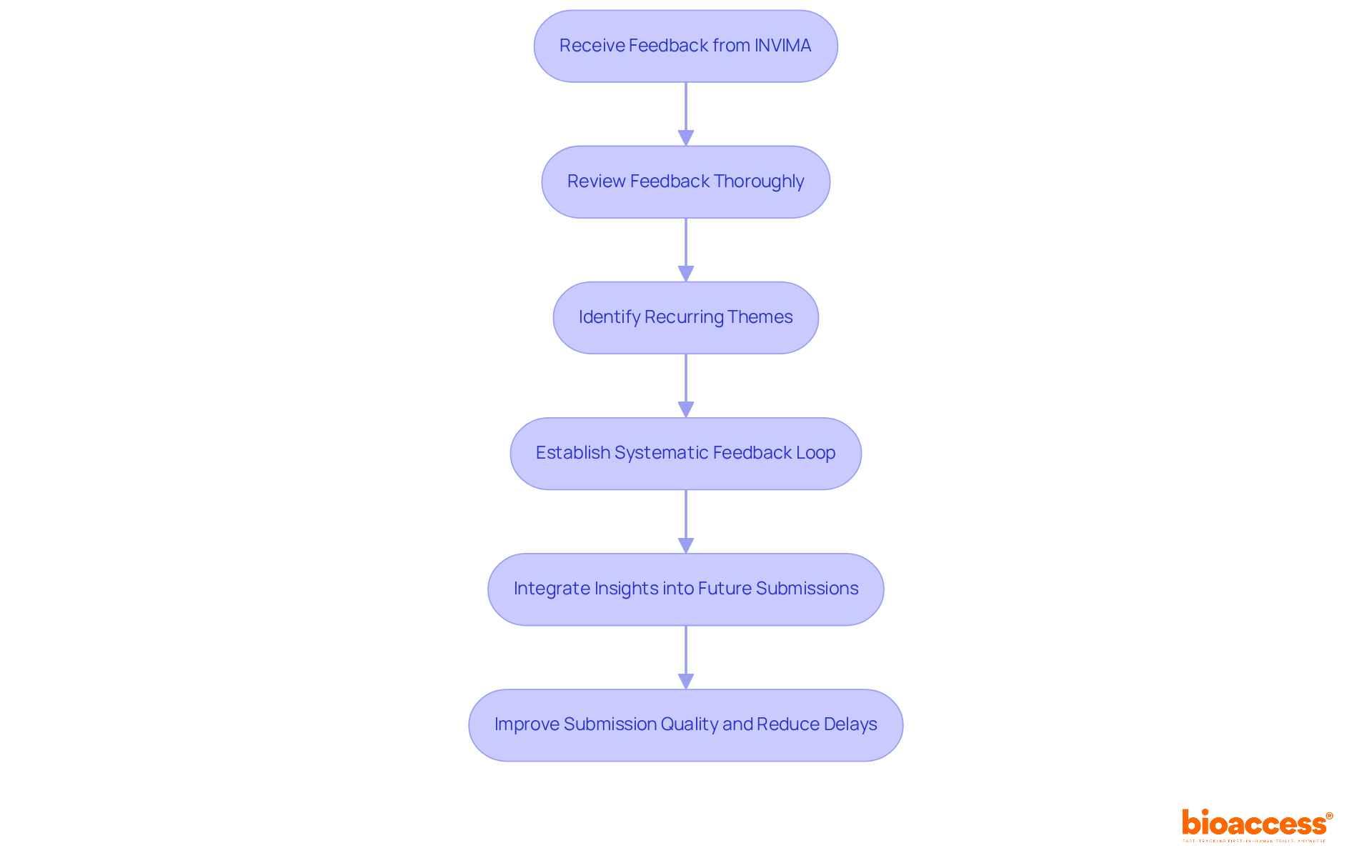
- Respond to Queries: Be prepared to address any inquiries or requests for additional information from ANVISA during the review. Prompt and clear communication can facilitate a smoother evaluation. With our proficiency in compliance matters, including Katherine Ruiz’s specialized knowledge in Colombia, we can assist you in crafting effective replies to any inquiries.
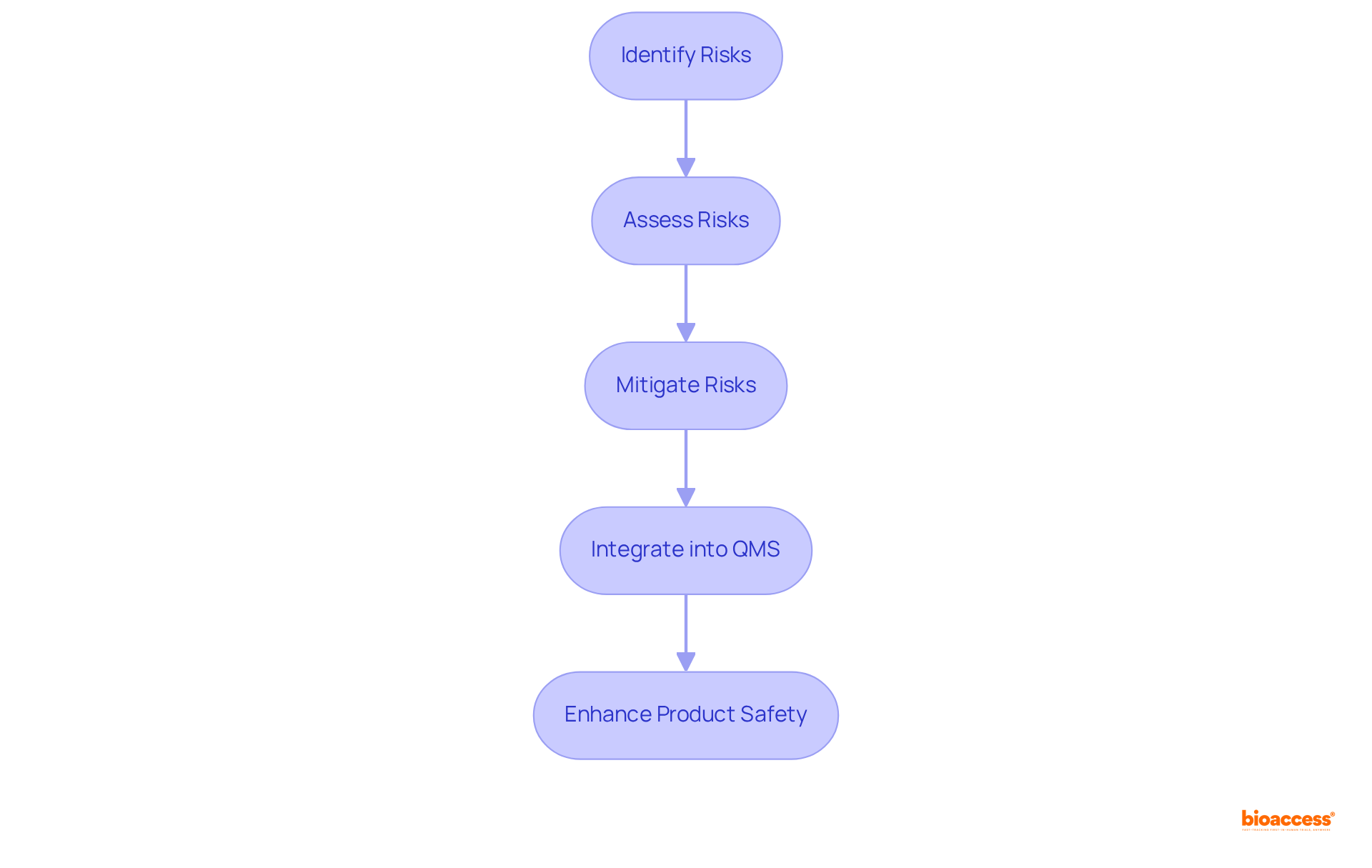
- Receive Approval: Upon receiving approval, ensure compliance with any and maintain your QMS. This ongoing dedication is essential for maintaining compliance with the and ensuring product safety. bioaccess® offers continuous assistance to guarantee that your products remain compliant with local regulations after approval.
Moreover, it is crucial to stay updated on regulatory changes. For instance, the illustrates the organization’s commitment to updating regulations in alignment with current medical standards. By following these steps and remaining aware of such developments, you can effectively navigate the regulatory compliance landscape. Leveraging efficient regulatory approval strategies tailored to the Brazilian market, as noted by Emergo by UL, can further enhance your compliance efforts.
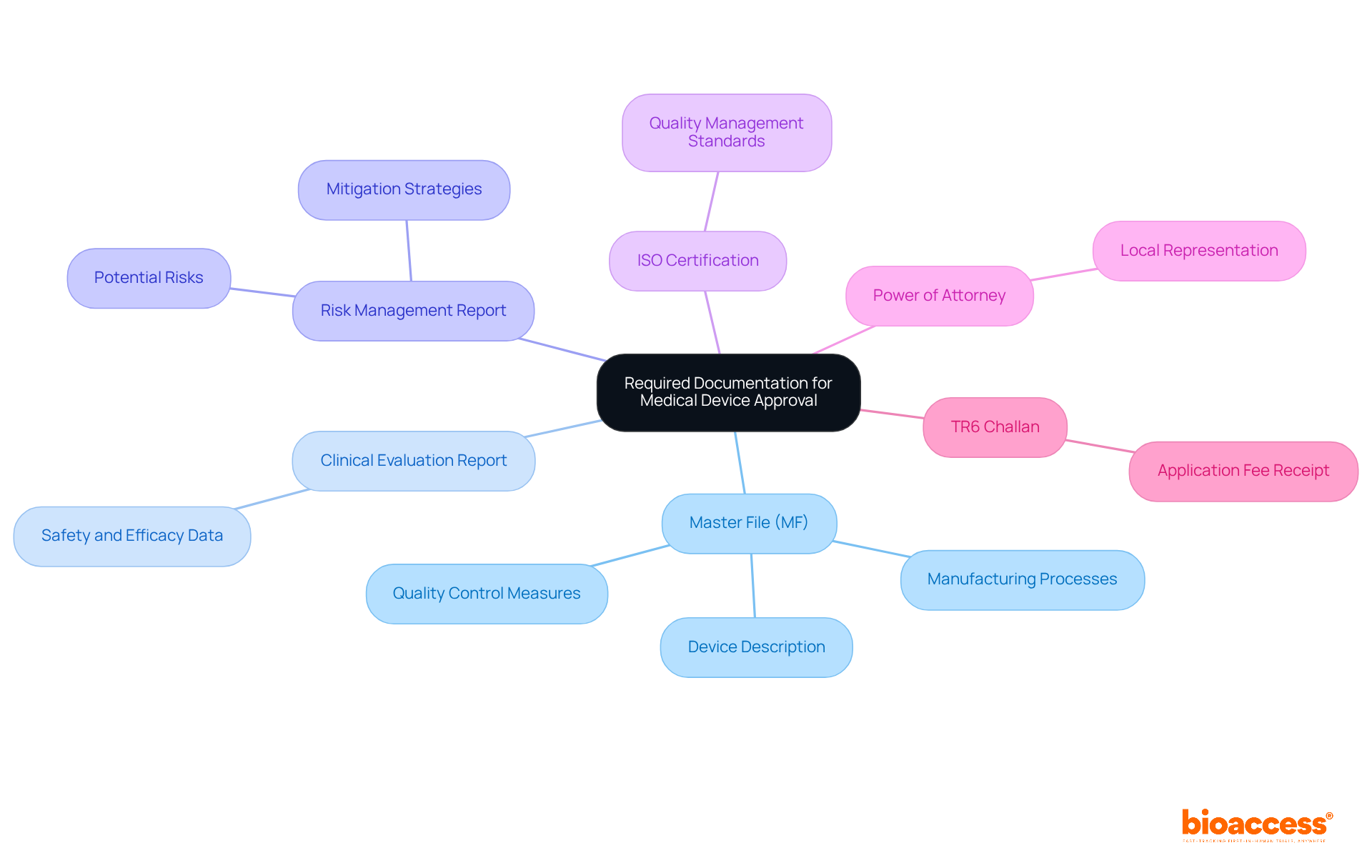
Prepare Essential Documentation for ANVISA Submission
When preparing documentation for ANVISA submission, it is essential to include the following components:
- : This document must provide comprehensive details about the medical device, encompassing design specifications, manufacturing processes, and intended use. A well-prepared is crucial, as it significantly influences the approval process and can expedite market entry. Medtech companies must present a (CSA) to ANVISA, which includes comprehensive study protocols and informed consent forms, underscoring the necessity of thorough documentation. Leveraging the expertise of bioaccess®, a leading , can enhance the quality of your , ensuring it meets all regulatory expectations.
- : Evidence demonstrating the apparatus’s safety and efficacy is paramount. This can be achieved through robust clinical studies or thorough literature reviews. Recent statistics suggest that a well-documented section can improve the chances of approval by demonstrating the product’s performance in real-world scenarios. Bioaccess® focuses on overseeing , such as Early-Feasibility and First-In-Human studies, to produce the essential for regulatory submissions.
- : Include certifications that affirm compliance with . This documentation assures the regulatory agency of the product’s quality and reliability, which is especially crucial for customized medical equipment. Adherence to the regulatory body’s guidelines is stressed for guaranteeing the safety and effectiveness of these devices, and bioaccess® can help ensure that your quality management documentation is thorough and meets standards.
- : All labels and instructions for use must adhere to ANVISA’s specific requirements. Proper labeling not only ensures compliance but also aids in user understanding and safety. Bioaccess® can offer advice on labeling practices that comply with official standards.
- Declaration of Conformity: This verifies that the equipment meets all relevant regulatory standards and serves as a vital assurance of compliance, particularly when considering the ANVISA guidelines for medtech compliance, which highlight the importance of careful preparation of these documents for the successful submission of healthcare products. Regulatory affairs specialists stress that standardizing documentation practices can significantly streamline the approval process, ultimately benefiting both manufacturers and patients. As mentioned by Peng Lu, ‘Standardizing the use of specific outcomes and outcome measures for research studies will assist in the creation of health guidelines and future indirect comparisons among interventions.’ Furthermore, understanding the different approval paths for medical devices based on classification, as outlined in the case study titled ‘,’ is critical for grasping the implications of documentation preparation on market entry in Brazil.
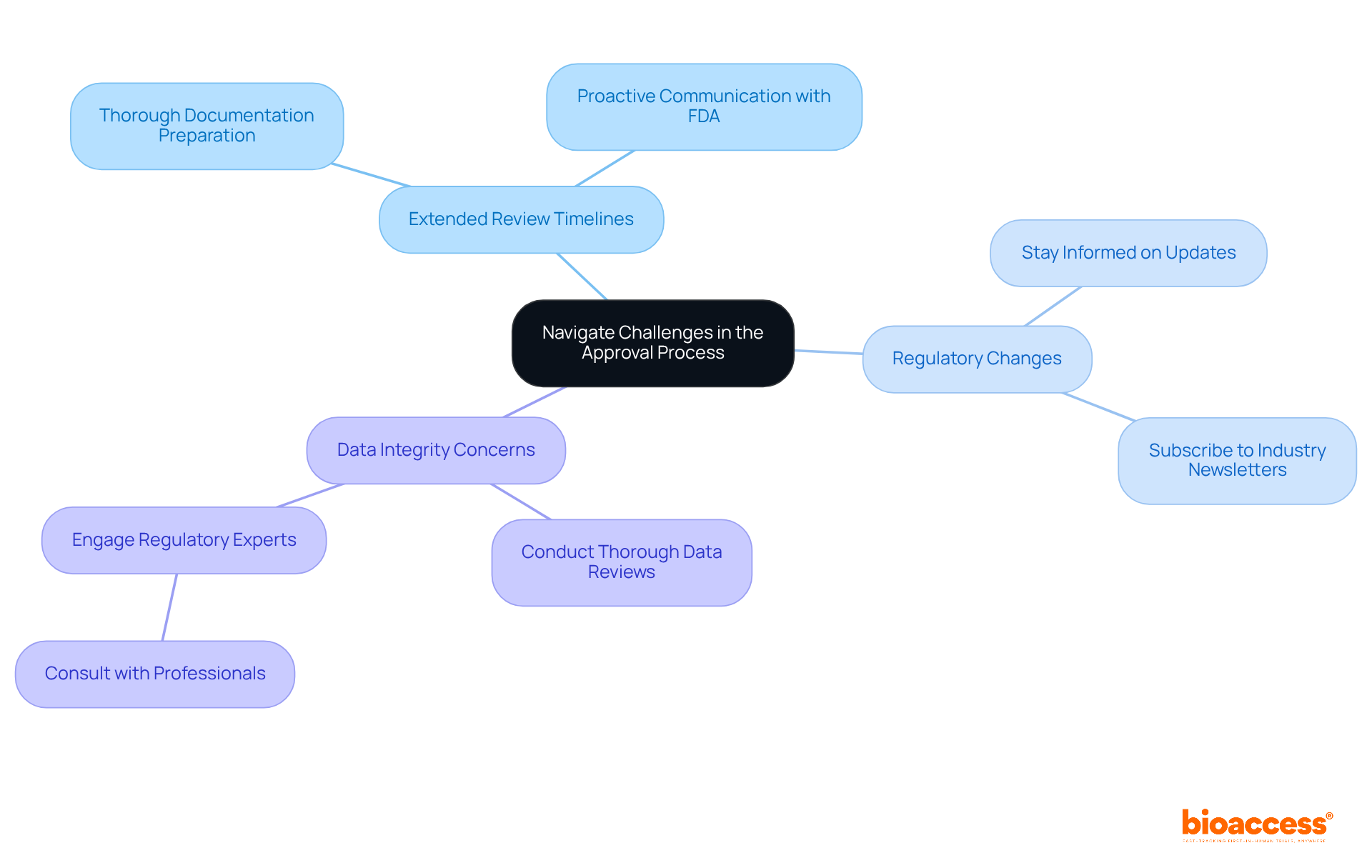
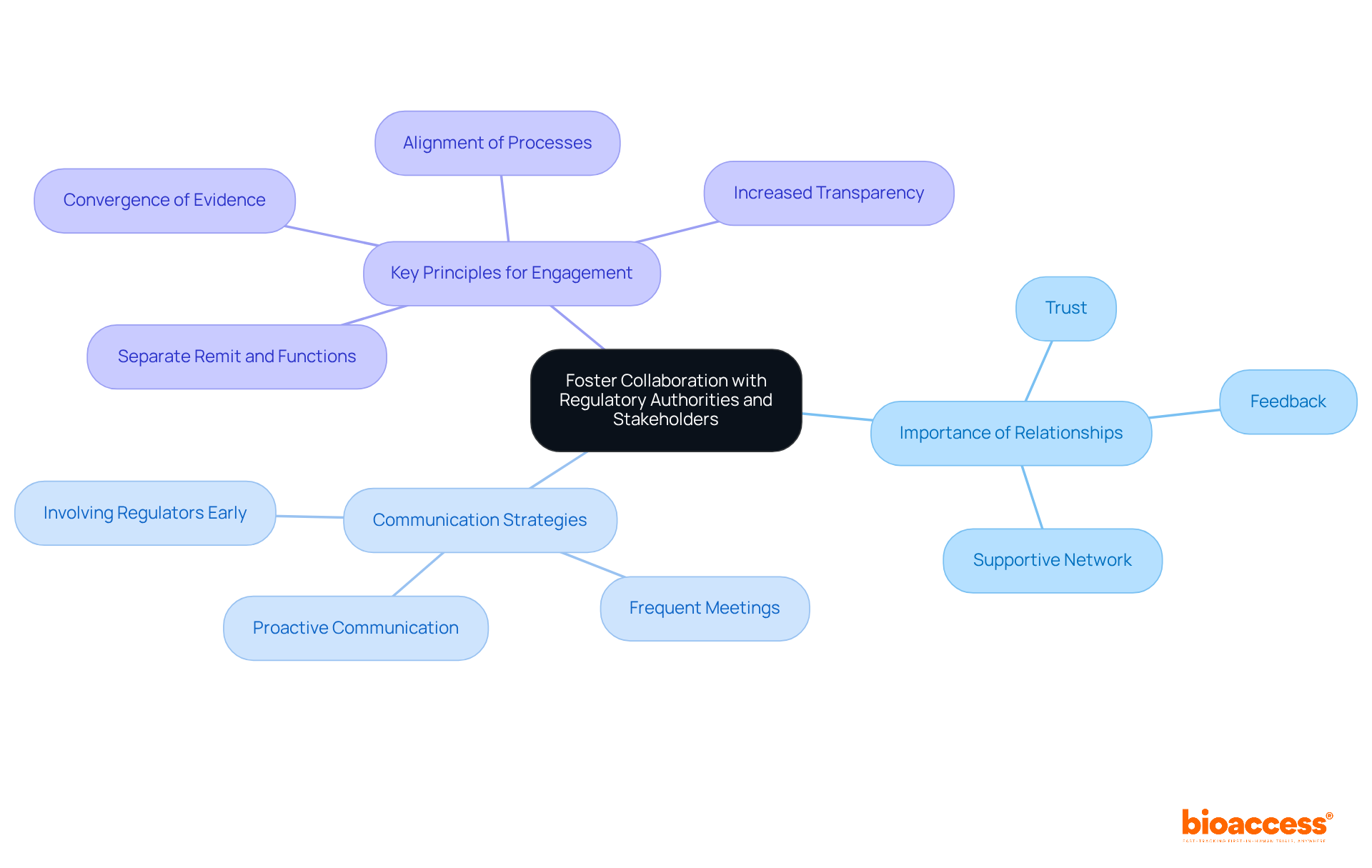
Engage with ANVISA: Best Practices for Effective Communication
To engage effectively with the , consider the following best practices:
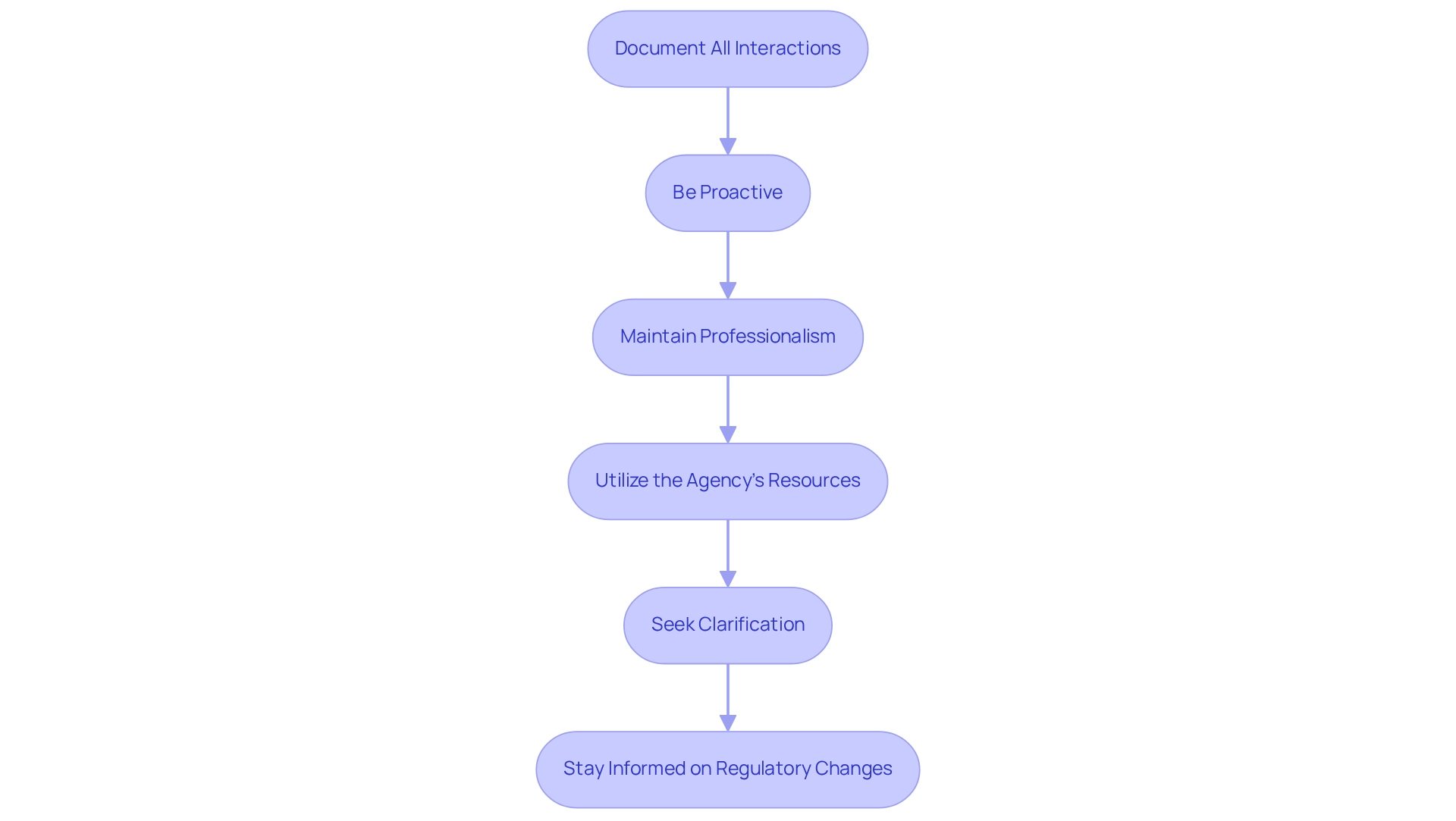
- Document All Interactions: Maintain comprehensive records of all communications with the , including emails, phone calls, and meetings. This documentation is crucial for tracking progress and ensuring accountability.
- Be Proactive: Anticipate potential questions or concerns from the and address them in your submissions. can significantly enhance the efficiency of the review process.
- Maintain Professionalism: Always communicate in a respectful and professional manner. This approach fosters a positive relationship with the agency, which is essential for successful interactions. As highlighted by Leonardo Abranches, enhancing collaboration can assist the agency in addressing its current staffing crisis while establishing a foundation for long-term advancements in oversight efficiency.
- Utilize the Agency’s Resources: Leverage the official website and available materials for guidance on submission processes and . Staying informed can streamline .
- Seek Clarification: If any aspect of the regulations is unclear, do not hesitate to contact the for clarification. can prevent misunderstandings and facilitate smoother interactions.
- Stay Informed on Regulatory Changes: With Directors from the agency discussing vital at their February 2024 meeting, it is essential to remain aware of any updates that could impact your submissions. Comprehending the function of INVIMA, , which monitors the management of health products and guarantees adherence to safety standards, can offer significant context for your engagements with the . By applying these strategies, organizations can strengthen their communication with the , ultimately resulting in more effective oversight processes and better outcomes for . Additionally, leveraging frameworks like the Agreement on Trade and Economic Cooperation (ATEC) can deepen , turning challenges into opportunities for smoother product approval processes.
Conclusion
Navigating the regulatory landscape established by ANVISA is crucial for Medtech companies striving for success in Brazil’s competitive healthcare market. Understanding ANVISA’s classification system represents the first critical step, as it determines the regulatory pathway and compliance requirements for medical devices. With the recent updates to ANVISA’s regulatory agenda, it is evident that staying informed about these changes and adapting to emerging technologies is more important than ever.
This article outlines a systematic approach to achieving compliance, highlighting the significance of thorough documentation, which includes:
- Technical dossiers
- Clinical data
- Quality management system certifications
Each component is essential in expediting the approval process and ensuring that devices meet the necessary safety and efficacy standards. Furthermore, engaging effectively with ANVISA through proactive communication and maintaining professionalism can significantly enhance the likelihood of successful submissions.
Ultimately, the key to thriving in Brazil’s Medtech landscape lies in aligning with ANVISA’s guidelines and nurturing a collaborative relationship with the agency. By adhering to best practices in documentation and communication, organizations can navigate the complexities of regulatory approval while contributing to the advancement of innovative medical solutions that enhance patient safety and public health. As the industry continues to evolve, those who prioritize compliance and effective engagement with ANVISA will be well-positioned to seize opportunities and drive meaningful change in the healthcare sector.
Frequently Asked Questions
What is the role of the Brazilian Health Regulatory Agency (ANVISA)?
ANVISA oversees health-related products, pharmaceuticals, and equipment in Brazil, ensuring that all medical products meet strict safety, efficacy, and quality standards before entering the market.
Why is understanding ANVISA’s classification system important?
ANVISA’s classification system categorizes items based on their associated risk levels, which significantly influences the approval process and specific requirements for each item, affecting timelines and compliance strategies.
What recent updates did ANVISA implement in 2025?
In 2025, ANVISA updated its oversight agenda to adapt to emerging technologies and market demands, emphasizing the importance of maintaining rigorous data integrity in clinical trials.
How does ANVISA ensure data integrity in clinical trials?
ANVISA requires data from subjects to be excluded from pharmacokinetic and statistical analyses, demonstrating its commitment to maintaining high standards of data integrity.
What is the significance of robotics in healthcare regulation?
The rise of robotics as a transformative technology in healthcare highlights the changing landscape of healthcare devices and their regulatory implications, showcasing the essential role of ANVISA in introducing innovative medical technologies.
How do Medtech firms benefit from adhering to ANVISA guidelines?
Compliance with ANVISA guidelines is crucial for Medtech firms, as it can lead to successful market entry and enhanced patient safety.
What services does bioaccess® provide for regulatory submissions?
Bioaccess® offers comprehensive clinical trial management services, including feasibility studies, site selection, compliance reviews, trial setup, import permits, project management, and reporting.
Why is it important for organizations to stay informed about regulatory updates in Brazil?
Staying informed about regulatory updates and requirements is crucial for any organization aiming to thrive in the Brazilian market, especially as the landscape of healthcare equipment regulation continues to evolve.
List of Sources
- Understand ANVISA: The Regulatory Authority for Medtech in Brazil
- biopharmaservices.com (https://biopharmaservices.com/blog/bioequivalence-studies-for-anvisa-navigating-requirements)
- linkedin.com (https://linkedin.com/pulse/guardians-quality-brief-guide-anvisa-fgeaf)
- Follow the Step-by-Step Process for ANVISA Compliance
- demarest.com.br (https://demarest.com.br/en/boletim-de-life-sciences-e-healthcare-marco-de-2025)
- emergobyul.com (https://emergobyul.com/services/brazil-anvisa-medical-device-classification-consulting)
- Prepare Essential Documentation for ANVISA Submission
- Understanding Clinical Trial Requirements In Brazil For Medtech An In Depth Tutorial | bioaccess® (https://bioaccessla.com/blog/understanding-clinical-trial-requirements-in-brazil-for-medtech-an-in-depth-tutorial)
- Approval of medical devices in Brazil (https://blog.johner-institute.com/regulatory-affairs/approval-from-medical-devices-in-brazil)
- regdesk.co (https://regdesk.co/anvisa-qa-on-personalised-devices-overview)
- Engage with ANVISA: Best Practices for Effective Communication
- emergobyul.com (https://emergobyul.com/news/brazil-anvisa-announces-priorities-2024-2025-year)
- uschamber.com (https://uschamber.com/international/strengthening-anvisa-a-pivotal-moment-for-u-s-brazil-cooperation)
- 4 Quotes that Underscore the Importance of Compliance (https://compliancebridge.com/4-quote-that-underscore-importance-of)



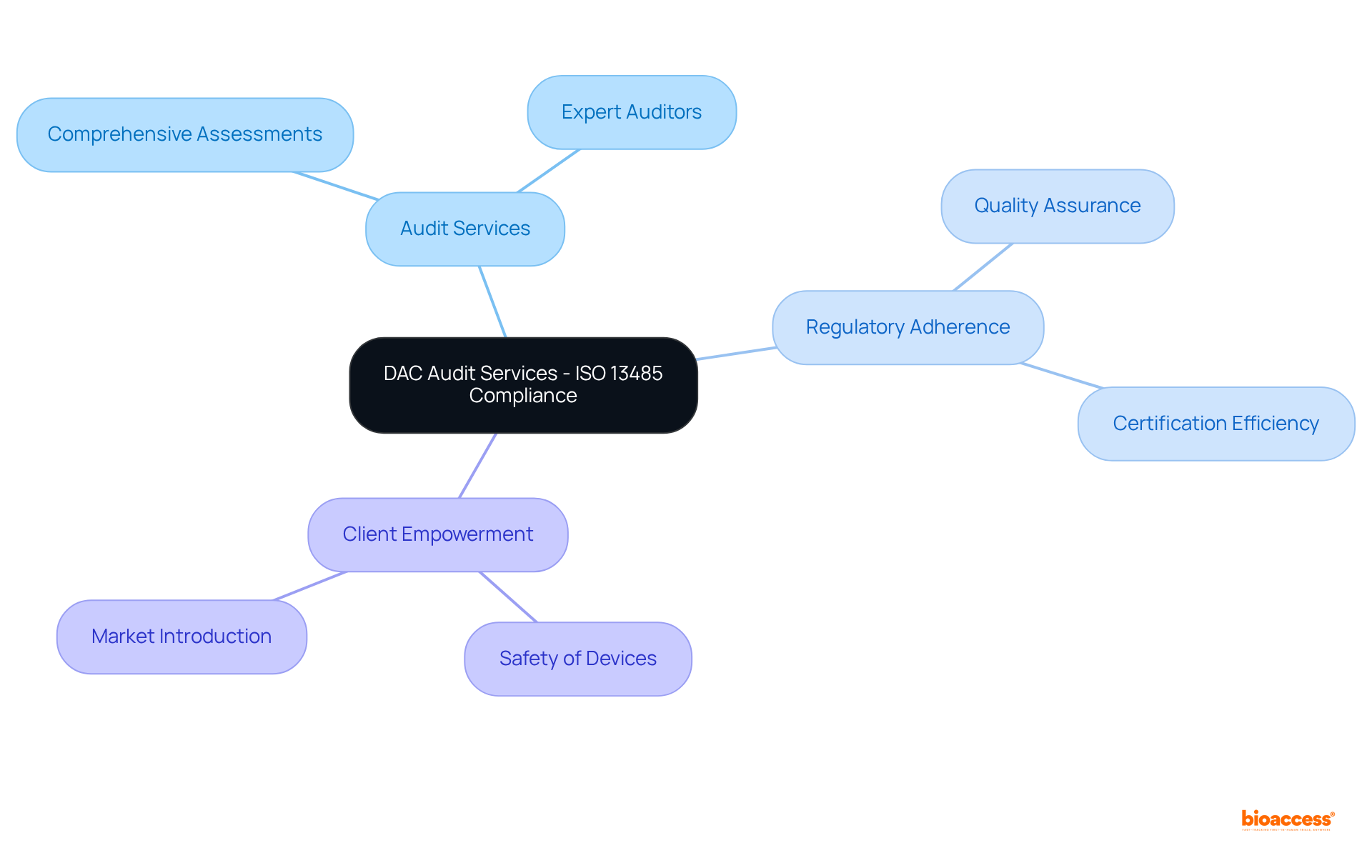



 in clinical trials, highlighting their importance in ensuring reliable and valid research outcomes. This mind map illustrates the interconnected concepts surrounding quality assurance (QA) and quality control (QC) in clinical trials, highlighting their importance in ensuring reliable and valid research outcomes.](https://tely.blob.core.windows.net/telyai/this-mind-map-illustrates-the-interconnected-concepts-surrounding-quality-assurance-qa-and-quality-control-qc-in-clinical-trials-highlighting-their-importance-in-ensuring-reliable-and-valid-research-outcomes.jpg)

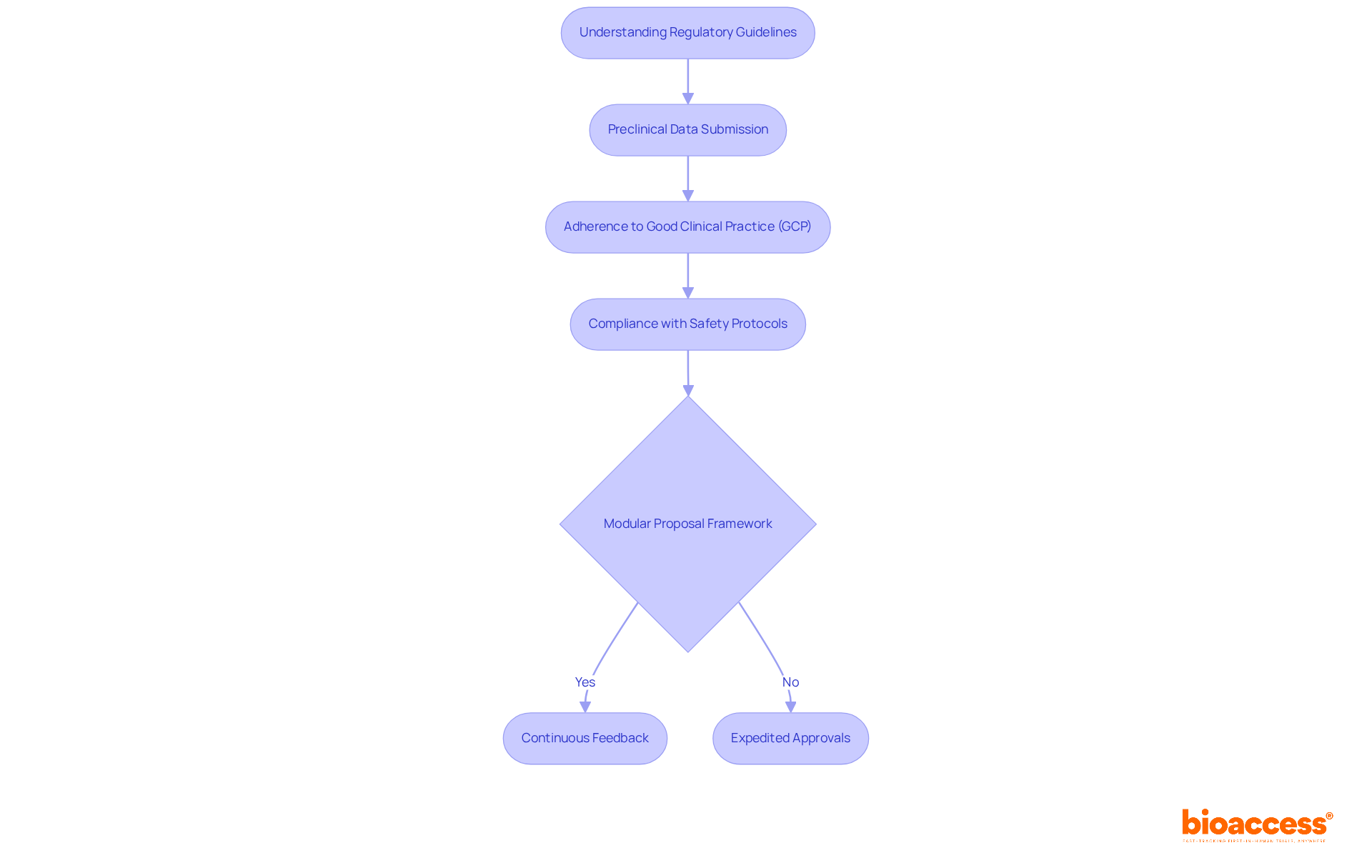


 (CDI) Process Flowchart: Clinical Documentation Improvement (CDI) Process](https://tely.blob.core.windows.net/telyai/flowchart-clinical-documentation-improvement-cdi-process.jpg)