Introduction
The landscape of clinical trials is constantly evolving, with Clinical Research Organizations (CROs) playing a vital role in orchestrating these complex studies. The selection of a top-tier CRO is crucial, as it directly impacts the management, efficacy, and overall success of clinical research.
In this article, we will explore the top 10 CRO companies that have distinguished themselves through their ability to manage multifaceted dimensions of clinical trials and their commitment to adapt and innovate in a field that demands constant vigilance and improvement. These companies have demonstrated expertise in specific therapeutic areas, global reach and capacity, innovative approaches to clinical trials, strong regulatory compliance, advanced technology and data analytics, a patient-centric approach, collaborative partnerships, proven track record of success, customized solutions for sponsors, and a commitment to quality and excellence. Join us as we delve into the world of clinical trials and discover the leading CRO companies driving advancements in medical research.
Top 10 CRO Companies for Clinical Trials
The landscape of is ever-evolving, with at the forefront of orchestrating these complex studies. The significance of selecting a cannot be understated, as it directly impacts the management, efficacy, and overall success of clinical research.
For patients with rare diseases, like the one from rural Pennsylvania facing the prospect of international travel for a trial in Turkey, the CRO’s role extends beyond the trial itself, encompassing the and guidance through daunting challenges such as visa acquisition and language barriers. Moreover, industry experts like Ken Getz have observed a paradigm shift in .
Emphasizing that the historical focus on ‘great science’ is now harmoniously paired with ”. This new trend is crucial in enhancing the efficiency and cost-effectiveness of , aided by the integration of technologies like electronic data capture.
Treehill’s insights further illuminate the critical nature of decision-making in clinical research. With approximately 80% of , it’s clear that a meticulous approach by CROss in planning and executing each phase of a trial can significantly alter its course. This need for ‘bulletproofing’ decisions underscores the value of expertise and dedication that leading CROs bring to the table. In light of these perspectives, the top 10 CRO companies have distinguished themselves not only by their ability to manage the multifaceted dimensions of but also by their commitment to adapt and innovate in a field that demands constant vigilance and improvement.
[Company Name]: Expertise in [Specific Therapeutic Area]
Navigating the complex landscape of necessitates a CRO with specialized expertise in distinct therapeutic areas. [Company Name] distinguishes itself with an in-depth understanding and extensive experience in [Specific Therapeutic Area].
This expertise is not just about academic knowledge; it’s about how this knowledge can be applied to (RWD) and . As highlighted by Flatiron’s research, which drew commentary from FDA members, the quality of RWD hinges on its relevance and reliability in addressing specific scientific questions.
This insight is critical for [Company Name], as it ensures that are not only designed with precision but also yield results that are meaningful for clinical application. Moreover, the challenges addressed in a JAMA special communication—specifically the disconnect between —underscore the importance of integrating with . [Company Name]’s team is at the forefront of this integration, striving to overcome the that have traditionally hamstrung the scope and impact of trials. By prioritizing the alignment of with the broader context of , [Company Name] ensures that their trials contribute to high-quality, actionable outcomes, addressing the that persists despite the tens of thousands of RCTs conducted annually.
[Company Name]: Global Reach and Capacity
Global scalability is imperative for , enabling them to address the pressing issue of . [Company Name] leverages their longstanding trust within communities, as exemplified by , to enhance .
By utilizing their established physical locations, such as stores and pharmacies, they can integrate into everyday health facilities. This approach not only facilitates diverse but also bridges care gaps through accessible screening and diagnostic services.
Moreover, [Company Name]’s international presence is not just about geographical coverage, but also about addressing the complexities patients face when participating in trials abroad. For instance, a patient with a rare disease from rural Pennsylvania may face the daunting task of navigating international travel to join a trial in Turkey.
The questions of obtaining visas, handling foreign documentation, and coordinating travel are significant barriers that [Company Name] aims to mitigate. Their infrastructure is designed to streamline these processes, ensuring that each step is as seamless as possible for participants, regardless of the trial’s location. Their commitment to optimizing is further reflected in their meticulous approach to decision-making. By closely examining the myriad of decisions made years in advance, [Company Name] strives to ensure that every aspect of the trial, from design to execution, is robust and effective. This level of attention to detail is geared towards maximizing the efficiency and impact of their large-scale clinical trials.
[Company Name]: Innovative Approaches to Clinical Trials
like [Company Name] are revolutionizing the landscape of by integrating state-of-the-art technologies such as artificial intelligence (AI) and machine learning (ML). These tools are not only refining the way trials are designed but also streamlining the recruitment process and bolstering the analysis of complex data.
For instance, a patient with a rare disease in rural Pennsylvania may face overwhelming challenges to participate in a trial overseas. The complexity of international travel, language barriers, and document management can deter participation, highlighting the necessity for innovative solutions to make trials more accessible and efficient.
As noted by Gregory Curfman from JAMA, the traditional separation between results in inefficiencies and limited trial scopes. By leveraging advanced technologies, [Company Name] is addressing these critical issues, ensuring that trials are not only more inclusive but also that their findings are more readily applicable to clinical practice. This approach is informed by the realization that many past trial decisions, which impact future outcomes, could have been optimized with more careful planning and analysis, as reflected in the insights from transaction advisory experiences. Through such , [Company Name] is enhancing the very fabric of , contributing to more robust and practical medical guidelines, and ultimately, fostering better patient outcomes.
[Company Name]: Strong Regulatory Compliance
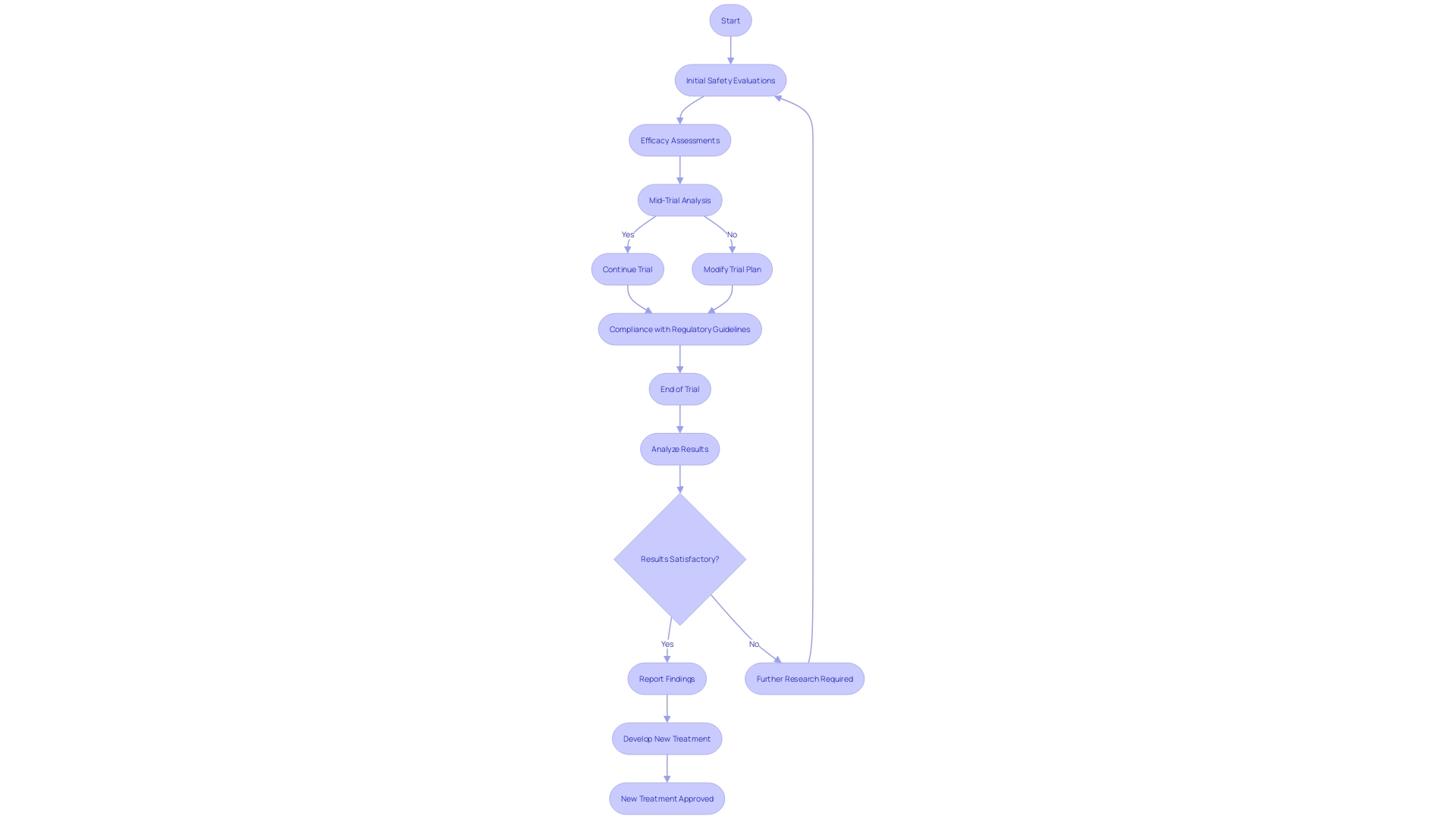
Clinical trial companies like [Company Name] are pivotal in the discovery of , ensuring that new treatments are both safe and effective. The meticulous adherence to is not just a formality but a cornerstone in guaranteeing the integrity and reliability of .
With the healthcare industry’s increasing incorporation of AI and machine learning, the regulatory landscape is evolving. International bodies like the FDA and EMA are introducing stringent guidelines to navigate the risks associated with these technologies, emphasizing a “” and demanding transparency.
[Company Name] has demonstrated a deep understanding of these complex regulations, effectively implementing that align with the latest standards. Their commitment is reflected in the rigorous conduct of across all phases, from initial safety evaluations in small groups of healthy volunteers to broader assessments of efficacy and side effects in targeted patient populations. As the industry acknowledges the challenges posed by innovative technologies, [Company Name]’s steadfast compliance ensures that they remain at the forefront, shaping the future of patient treatment and care.
[Company Name]: Advanced Technology and Data Analytics
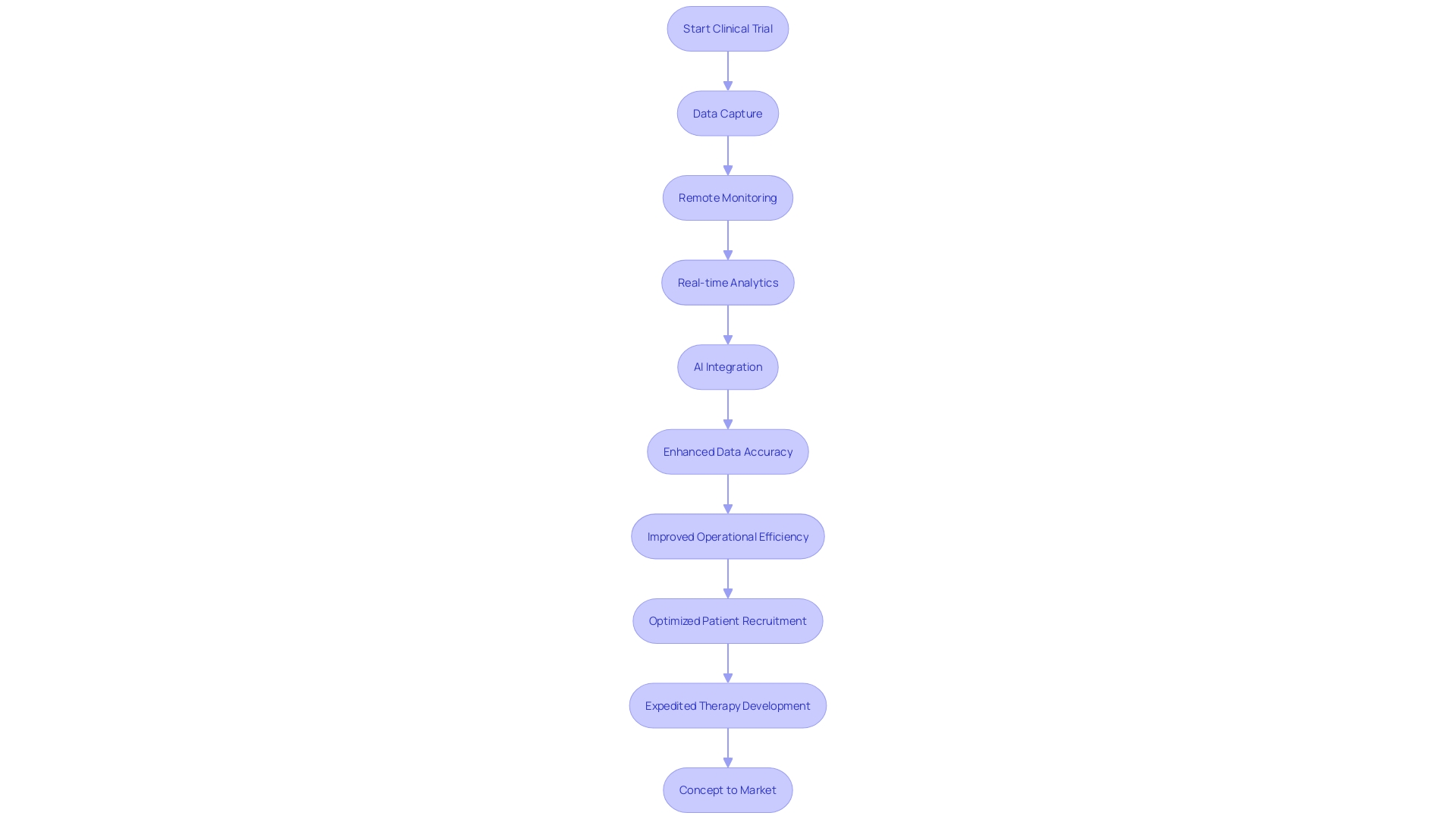
In the realm of medical advancements, are pivotal in harnessing technology to manage the surge of data generated during . As the volume of medical data grows exponentially—doubling every 70 days compared to every 50 years in 1950— are adopting to keep pace.
A Phase 3 trial now produces about 3.6 million data points, a threefold increase from a decade ago. To address this, Company Name integrates , remote monitoring, and to enhance data accuracy and operational efficiency.
The utilization of Artificial Intelligence (AI) begins at the inception of drug development, aiding in the identification of potential drug candidates and offering insights into the most effective targets for specific conditions. Ai’s predictive capabilities are instrumental in modeling , foreseeing potential challenges, and optimizing study designs. This advanced approach not only mitigates the risk of trial failures but also expedites —a critical hurdle in the clinical trial process. Consequently, these technological advancements enable swifter decision-making, accelerating the journey of new therapies from concept to market, ultimately elevating patient care and improving quality of life.
[Company Name]: Patient-Centric Approach
are the cornerstone of medical advancements, and at the heart of these trials are the patients whose participation is vital. [Company Name] emphasizes a in , recognizing that the involvement of patients in the planning and design of trials is essential.
As noted by Daniel J Herron, Vice President of Digital Offerings, Regulated Industries at RWS, involves ‘actively involving patients in the planning and design of trials, so their perspectives and needs are considered.’ This involves providing information that is not only accessible but also easy to comprehend, catering to a diverse population with different lived experiences.
By embracing , [Company Name] ensures that the nuances of varying conditions, backgrounds, and identities are acknowledged and addressed in their studies. The approach extends beyond just meeting regulatory requirements; it’s about fostering an environment where patients feel valued, their time respected, and their contributions to science appreciated. This ethos has a tangible impact, as it leads to a more positive trial experience and enhances , ultimately resulting in more robust and meaningful research outcomes.
[Company Name]: Collaborative Partnerships
At [Company Name], we understand that the success of hinges on and methodical planning. Our approach is to engage with investigators, sponsors, regulatory authorities, and ethics committees from the outset, aligning our efforts to ensure that every clinical trial we undertake is poised for success.
This proactive engagement is grounded in a hypothesis-driven framework that emphasizes the significance and impact of the clinical trial, as well as its readiness for implementation at the time of award. The planning phase of our includes a thorough presentation of the rationale, drawing on prior studies and pre-clinical data that underscore the necessity and viability of the research.
We meticulously define the , ensuring that the trial is aptly designed to test the proposed hypotheses. Acquisition and management of study agents are carefully outlined, alongside protocols for ensuring adherence to , GCLP, and GMP standards across all trial sites.
As we navigate the complexities of , particularly in the realm of rare diseases with unique and distinct needs, we are reminded of the urgency and scale of challenges ahead. The words of an industry expert resonate with our mission: ‘Start early. What are some of the challenges remaining in this field? … Funding to support natural history work has been very limited in the past; that is changing, and it’s improving, but still in a way, we’re seeing a couple of studies launch at a time, when what we really need are broad approaches where we can advance a lot of learning across many diseases at once.’ It is with this understanding that [Company Name] commits to fostering robust partnerships and detailed planning, ensuring not only the successful execution of but also contributing to the broader advancement of medical knowledge.
[Company Name]: Proven Track Record of Success
In the landscape of clinical research, the caliber of a (CRO) is often gauged by its history of success and the quality of data it produces. [Company Name] stands out with a formidable track record, showcasing a comprehensive portfolio of that have been executed to perfection.
Their approach integrates not just scientific rigor but also []([Company Name]), which has been increasingly recognized as pivotal in the industry. As Ken Getz noted, the dual focus on ” and ‘great execution’ is essential, a philosophy that [Company Name] embodies through their meticulous planning and use of like electronic data capture.
Moreover, their dedication to refining the clinical trial process is evident in their commitment to thorough , as highlighted by the insights from Treehill’s advisory experience. This ensures that each phase of the trial is not only well-conceived but also resilient to future challenges. Sponsors can entrust [Company Name] with the management of their , confident in their ability to navigate complex logistics and produce reliable, high-quality data.
[Company Name]: Customized Solutions for Sponsors
At CMIC Group, the pioneering (CRO) in Japan, the focus is on delivering end-to-end solutions that are as unique as the they support. Recognizing that the effectiveness of a product is intrinsically tied to the ‘ design—which ultimately shapes the product’s market indication—CMIC provides bespoke services that span the entire . From to market entry solutions, they ensure that the nuanced needs of their clients are met with precision.
Drawing on over three decades of industry innovation, CMIC’s tailored approach extends to pharmaceutical companies, medical device manufacturers, academia, and beyond. Their commitment is to ‘meet customers where they are’ with exactly what is needed to advance their products. Acknowledging the criticality of decision-making in the early stages of , CMIC places a strong emphasis on working through decisions with sponsors.
As articulated by industry experts, a significant proportion of decisions made years in advance could be better optimized with a CRO’s careful guidance and expertise. By dedicating time and energy to ‘bulletproof’ these pivotal choices, CMIC helps to ensure that each link in the chain is strengthened, reflecting a deep understanding of the client’s timeline and the market’s unmet needs. This approach not only meets sponsors’ requirements but also anticipates the strategic moves that will define the in the real world.
[Company Name]: Commitment to Quality and Excellence
At the core of [Company Name]’s operations lies a rigorous dedication to top-tier quality and excellence. Their global presence, with subsidiaries across more than 50 countries and business activities spanning over 150 countries, is a testament to their []([Company Name]).
[Company Name] has implemented comprehensive to ensure that their services meet the highest standards, mirroring the meticulous approach taken by LEDVANCE in the general lighting sector. Much like LEDVANCE’s commitment to conveying complex topics through accessible eLearning platforms, [Company Name] strives to deliver clear, reliable, and precise data that propels .
By integrating smart solutions and advanced technology into their operations, similar to LEDVANCE’s smart home and smart building solutions, [Company Name] not only enhances the quality of their work but also ensures that it is inclusive and reaches a global audience. The translation of their materials into multiple languages reflects this inclusivity, making their research and findings accessible to a diverse international community. Such attention to detail and quality underpins [Company Name]’s reputation as a trusted partner in the advancement of .
![Relationships and Concepts in [Company Name]'s Operations Relationships and Concepts in [Company Name]'s Operations](https://vectorly-files.s3.eu-central-1.amazonaws.com/7e5170f3-eeed-4ae0-a415-e37c583adb20.jpg)
Conclusion
In conclusion, the top 10 CRO companies highlighted in this article are leading the way in clinical trials. They excel in managing complex studies and demonstrate a commitment to adapt and innovate.
These CROs have expertise in specific therapeutic areas, ensuring precise trial design and meaningful results. They integrate trials with clinical practice, addressing the disconnect between trialists and clinicians for high-quality outcomes.
Global reach and capacity are crucial, allowing these CROs to address healthcare accessibility issues. They streamline processes for participants facing challenges in trials abroad, ensuring a seamless experience.
Innovative approaches to clinical trials are revolutionizing medical research. These CROs leverage advanced technologies like AI and ML to enhance efficiency and inclusivity.
Strong regulatory compliance is paramount to ensure safe and effective treatments. The leading CROs demonstrate an understanding of complex regulations and implement quality control measures aligned with the latest standards.
Advanced technology and data analytics play a significant role in improving data management and decision-making speed. These CROs integrate electronic data capture systems, remote monitoring, real-time analytics, and AI to enhance operational efficiency.
A patient-centric approach is emphasized by actively involving patients in trial planning and design. These CROs provide accessible information that caters to diverse populations’ needs while fostering an environment where patients feel valued.
Collaborative partnerships are essential for successful trials. The leading CROs engage with investigators, sponsors, regulatory authorities, ethics committees from the outset aligning efforts for each trial’s success. Proven track records of success distinguish these top CROs as they showcase comprehensive portfolios of executed trials integrating scientific rigor with strategic operational management. Customized solutions tailored to sponsors’ needs set these CROs apart within the pharmaceutical value chain. They work closely with sponsors early on in drug development stages to optimize choices that strengthen each link in the development chain. At their core lies a commitment to top-tier quality and excellence reflected in global presence and comprehensive quality assurance systems. These CROs strive for clear, reliable data delivery while ensuring inclusivity through translation into multiple languages. Overall, these top 10 CRO companies drive advancements in medical research through their expertise, global reach, innovative approaches, regulatory compliance measures advanced technology utilization patient-centricity collaborative partnerships proven track records customized solutions commitment quality excellence.
Frequently Asked Questions
What is a Clinical Research Organization (CRO)?
A CRO is a company that provides support for the management of clinical trials, coordinating various aspects such as logistics, regulatory compliance, and data management to ensure the success of clinical research.
Why is selecting a top-tier CRO important?
Choosing a leading CRO is crucial because it directly influences the management, efficacy, and success of clinical trials. A top-tier CRO can provide expertise in trial design, patient recruitment, and regulatory compliance, all of which are essential for producing reliable data and achieving trial objectives.
How do CROs assist patients, especially those with rare diseases?
CROs offer logistical support and guidance for patients, including help with international travel, visa acquisition, and overcoming language barriers, making participation in trials more feasible for individuals from diverse backgrounds.
What recent trends are influencing clinical trial design?
There is a growing emphasis on combining ‘great science’ with ‘great execution’ to enhance the efficiency and cost-effectiveness of clinical trials. Technologies like electronic data capture are increasingly integrated into trial processes.
How do CROs ensure the quality of Real-World Data (RWD)?
CROs utilize expertise in specific therapeutic areas to ensure that clinical trials yield meaningful results relevant to clinical practice, addressing issues such as the disconnect between trialists and clinicians.
What role do CROs play in global scalability for clinical trials?
CROs leverage their global presence to enhance accessibility, utilizing established community networks for patient recruitment and facilitating processes like visa acquisition for international trial participation.
How are advanced technologies changing the landscape of clinical trials?
CROs are integrating artificial intelligence (AI) and machine learning (ML) to streamline trial design, recruitment, and data analysis, which helps in optimizing study designs and improving patient outcomes.
Why is regulatory compliance crucial for CROs?
Adherence to regulatory guidelines is essential for ensuring the safety and effectiveness of new treatments. CROs must navigate evolving regulations, especially with the introduction of AI and ML technologies in clinical trials.
How do CROs handle the increasing volume of data in clinical trials?
CROs adopt sophisticated technologies, such as electronic data capture and real-time analytics, to manage the exponential growth of data generated during trials, enhancing data accuracy and operational efficiency.
What is a patient-centric approach in clinical trials?
A patient-centric approach involves actively involving patients in the planning and design of clinical trials to ensure their perspectives and needs are considered, ultimately leading to more meaningful research outcomes and improved patient experiences.
How do CROs foster collaborative partnerships?
CROs engage with various stakeholders, including investigators, sponsors, and regulatory authorities, from the outset to ensure that every clinical trial is well-planned and aligned with its objectives for success.
What is the significance of a proven track record for CROs?
A CRO’s history of successful clinical trials and high-quality data production is often a key indicator of its capability and reliability in managing complex clinical research projects.
How do CROs provide customized solutions for sponsors?
CROs offer tailored services that cater to the unique needs of their clients, ensuring that their clinical trials are designed to meet specific market demands and regulatory requirements.
What commitment to quality and excellence do leading CROs demonstrate?
Leading CROs implement rigorous quality assurance systems and utilize advanced technologies to maintain high standards in their operations, ensuring that their research findings are reliable and accessible to a global audience.
List of Sources
- Top 10 CRO Companies for Clinical Trials
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- pharmavoice.com (https://www.pharmavoice.com/news/red-jacket-ken-getz-a-champion-of-clinical-trials/697633/)
- biospace.com (https://www.biospace.com/article/treehill-partners-ali-pashazadeh-on-how-to-improve-clinical-trial-design/?utm_source=dlvr.it&utm_medium=twitter)
- [Company Name]: Expertise in [Specific Therapeutic Area]
- appliedclinicaltrialsonline.com (https://www.appliedclinicaltrialsonline.com/view/scope-2024-alex-deyle-of-flatiron-health-discusses-rwd-in-clinical-trials)
- raps.org (https://www.raps.org/News-and-Articles/News-Articles/2024/6/Experts,-FDA-officials-discuss-future-of-clinical?utm_campaign=Regulatory-Focus&utm_source=twitter&utm_medium=social)
- [Company Name]: Global Reach and Capacity
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- biospace.com (https://www.biospace.com/article/treehill-partners-ali-pashazadeh-on-how-to-improve-clinical-trial-design/?utm_source=dlvr.it&utm_medium=twitter)
- statnews.com (https://www.statnews.com/2024/06/14/walgreens-ramita-tandon-diversity-recruiting-customers-into-clinical-trials/?utm_campaign=rss)
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- [Company Name]: Innovative Approaches to Clinical Trials
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- biospace.com (https://www.biospace.com/article/treehill-partners-ali-pashazadeh-on-how-to-improve-clinical-trial-design/?utm_source=dlvr.it&utm_medium=twitter)
- raps.org (https://www.raps.org/News-and-Articles/News-Articles/2024/6/Experts,-FDA-officials-discuss-future-of-clinical?utm_campaign=Regulatory-Focus&utm_source=twitter&utm_medium=social)
- [Company Name]: Strong Regulatory Compliance
- rwmalonemd.substack.com (https://rwmalonemd.substack.com/p/indication-labeling-and-fraud)
- pharmalive.com (https://www.pharmalive.com/balancing-innovation-with-patient-safety-navigating-regulatory-guidelines-in-clinical-research/)
- scientia.global (https://www.scientia.global/mr-anthony-keyes-understanding-and-improving-clinical-trial-compliance/)
- medpagetoday.com (https://www.medpagetoday.com/opinion/second-opinions/106064)
- [Company Name]: Advanced Technology and Data Analytics
- medtechintelligence.com (https://medtechintelligence.com/column/the-power-of-ai-to-enhance-clinical-trials/)
- forbes.com (https://www.forbes.com/sites/forbesbusinessdevelopmentcouncil/2023/09/14/the-next-generation-of-clinical-trials-ai-and-new-ways-to-use-data/)
- infomeddnews.com (https://infomeddnews.com/about-medical-device-news-magazine-2024/)
- [Company Name]: Patient-Centric Approach
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- en.cmicgroup.com (https://en.cmicgroup.com)
- pmlive.com (https://pmlive.com/blogs/how-patient-engagement-improves-outcomes-in-chronic-illness/)
- clinicaltrialsarena.com (https://www.clinicaltrialsarena.com/sponsored/why-patient-centricity-is-the-answer-to-the-clinical-trial-enrolment-gap/)
- acsjournals.onlinelibrary.wiley.com (https://acsjournals.onlinelibrary.wiley.com/doi/10.1002/cncr.35145)
- [Company Name]: Collaborative Partnerships
- cgtlive.com (https://www.cgtlive.com/view/partnership-patient-advocacy-groups-key-clinical-trial-end-point-selection)
- grants.nih.gov (https://grants.nih.gov/grants/guide/pa-files/PAR-24-066.html?utm_source=dlvr.it&utm_medium=twitter)
- [Company Name]: Proven Track Record of Success
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- pharmavoice.com (https://www.pharmavoice.com/news/red-jacket-ken-getz-a-champion-of-clinical-trials/697633/)
- biospace.com (https://www.biospace.com/article/treehill-partners-ali-pashazadeh-on-how-to-improve-clinical-trial-design/?utm_source=dlvr.it&utm_medium=twitter)
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- [Company Name]: Customized Solutions for Sponsors
- en.cmicgroup.com (https://en.cmicgroup.com)
- rwmalonemd.substack.com (https://rwmalonemd.substack.com/p/indication-labeling-and-fraud)
- biospace.com (https://www.biospace.com/article/treehill-partners-ali-pashazadeh-on-how-to-improve-clinical-trial-design/?utm_source=dlvr.it&utm_medium=twitter)
- [Company Name]: Commitment to Quality and Excellence
- medhealthoutlook.com (https://medhealthoutlook.com/three-best-practices-to-combat-clinical-trial-pain-points-scott-gray-ceo-of-clincierge/)
- raps.org (https://www.raps.org/News-and-Articles/News-Articles/2024/6/Experts,-FDA-officials-discuss-future-of-clinical?utm_campaign=Regulatory-Focus&utm_source=twitter&utm_medium=social)
- elearningindustry.com (https://elearningindustry.com/a-lighting-companys-path-to-training-success-with-a-custom-built-lms)
- pfizer.com (https://www.pfizer.com/about/purpose)

Leave a Reply