Introduction
While Brazil’s diverse and culturally rich population offers a unique opportunity for clinical trials, the complexities of patient recruitment present significant challenges. By leveraging local insights and community engagement, sponsors can not only increase participation rates but also ensure that studies reflect the demographics of the population they aim to serve.
Navigating the intricate web of regional disparities and regulatory landscapes poses significant challenges for sponsors. Let’s dive into some best practices and innovative strategies that can truly bridge the gap between clinical research and local communities, ultimately reshaping the future of clinical trials in Brazil.
Addressing these challenges is not just beneficial; it is essential for the integrity and success of clinical research in Brazil.
Engage Local Communities with Cultural Acumen
To enhance patient involvement in research studies, particularly for patient recruitment clinical trial Brazil, a deep understanding of local cultures is not just beneficial; it’s essential. Connecting with local communities while being culturally aware fosters trust and significantly boosts participation rates. For instance, utilizing local languages in hiring materials and involving community leaders as advocates can bridge communication gaps and build rapport. Conducting informational sessions in community centers clarifies the research process and addresses potential participants’ concerns, fostering an inviting atmosphere.
Many successful hiring initiatives have tapped into local festivals and health fairs to spread the word about medical studies, leading to significant rises in enrollment rates. These culturally customized methods not only enhance patient recruitment clinical trial Brazil but also aid in the overall success of studies by ensuring diverse representation in participant populations. Furthermore, strategies such as providing financial compensation for time and travel expenses can alleviate burdens associated with participation, further encouraging involvement.
Community involvement is crucial for fostering trust and awareness about research studies. Collaborating with local leaders and patient advocacy groups helps educate the public, while digital platforms and social media campaigns can reach broader audiences, targeting specific demographics. By showing regard for local traditions and actively engaging community members, sponsors can foster a more welcoming environment that promotes involvement, ultimately resulting in more comprehensive and representative research outcomes. Furthermore, grasping the compliance environment, including adherence to ANVISA guidelines and ICH-GCP standards, is essential for successful execution of the study. Utilizing the Unified Health System (SUS) can significantly aid in patient recruitment clinical trial Brazil by offering extensive access to healthcare services and generating a vast pool of potential individuals for research studies. With the support of bioaccess®, which provides crucial clinical trial insights and market access strategies, Medtech startups can navigate the complexities of regulatory pathways and enhance their participant engagement strategies in Brazil.
Tailor Recruitment Strategies to Regional Contexts
In Brazil, the promise of clinical research is often hindered by significant regional disparities in healthcare access and cultural attitudes. Tailoring hiring strategies to these specific contexts can greatly improve enrollment success. For instance, in urban areas where healthcare access is more common, digital engagement methods like targeted social media campaigns can effectively involve potential participants. Conversely, rural regions may benefit more from traditional outreach methods, including partnerships with local healthcare providers who can assist in patient referrals to research studies.
Understanding the local healthcare landscape, including public health systems and private clinics, is crucial for shaping these staffing strategies. Partnering with local physicians not only enhances credibility but also fosters trust within the community. By examining local demographics and healthcare trends, research sponsors can develop focused recruitment strategies that resonate with community members. This strategic approach ultimately leads to increased patient recruitment for clinical trials in Brazil and more comprehensive data collection, thereby strengthening Brazil’s status as a cost-effective and efficient site for early-stage studies in the MedTech and Biopharma fields.
Did you know Brazil represents about 2% of global research studies, with costs approximately 65% lower than those in the United States? Recent advancements in regulations, including Law #14.874/2024, have streamlined ethical approval processes, further enhancing Brazil’s appeal for research studies. Adhering to ICH-GCP standards and collaborating with authorities such as ANVISA and INVIMA ensures that studies meet essential criteria, establishing a robust foundation for effective patient enrollment and data reliability. By utilizing these regulatory pathways and insights from services like Global Trial Accelerators™, which include customized patient engagement strategies and regulatory navigation assistance, study sponsors can improve patient recruitment for clinical trials in Brazil more effectively. By embracing tailored strategies, research sponsors can not only enhance enrollment but also contribute to the advancement of Brazil’s MedTech and Biopharma sectors.
Utilize Technology and Innovative Approaches for Recruitment
In Brazil’s rapidly evolving clinical trial landscape, the challenge of patient recruitment clinical trial Brazil presents both obstacles and opportunities for sponsors. Incorporating technology into patient engagement strategies can significantly enhance efficiency and reach. Digital platforms, such as social media, mobile applications, and telemedicine, not only enhance patient engagement but also simplify the participant selection process. For instance, utilizing electronic health records (EHRs) facilitates the identification of potential participants based on specific criteria, accelerating enrollment. Additionally, telehealth solutions for initial consultations can reduce barriers to participation, especially for patients in remote areas.
Using innovative methods like artificial intelligence (AI) for patient matching and engagement can really boost enrollment efforts. AI can analyze extensive datasets to identify eligible candidates and predict their likelihood of participation, enabling more targeted outreach. By adopting these technological advancements, clinical trial sponsors can enhance enrollment rates and improve the overall patient experience, making participation more accessible and appealing.
The incorporation of AI in hiring strategies not only streamlines processes but also aligns with compliance frameworks in Latin America, such as adherence to ICH-GCP standards and acceptance of FDA-bridgeable data. This ensures that patient recruitment clinical trial Brazil efforts are efficient and meet the necessary compliance requirements, facilitating a smoother pathway to approval in jurisdictions where approval timelines can be significantly shorter compared to traditional markets.
With bioaccess®’s capabilities, including pre-negotiated site contracts, sponsors can achieve 50% faster enrollment and realize $25K savings per patient, optimizing patient recruitment clinical trial Brazil’s advantages for early-stage research. Furthermore, through the Global Trial Accelerators™, sponsors acquire vital insights into essential studies, updates on compliance, and strategies for market entry, thereby boosting their operational effectiveness. Embracing these technological advancements not only streamlines the patient recruitment clinical trial Brazil process but also positions sponsors for success in a competitive market, ultimately reshaping the future of clinical research in Brazil.
Address Common Challenges in Patient Recruitment
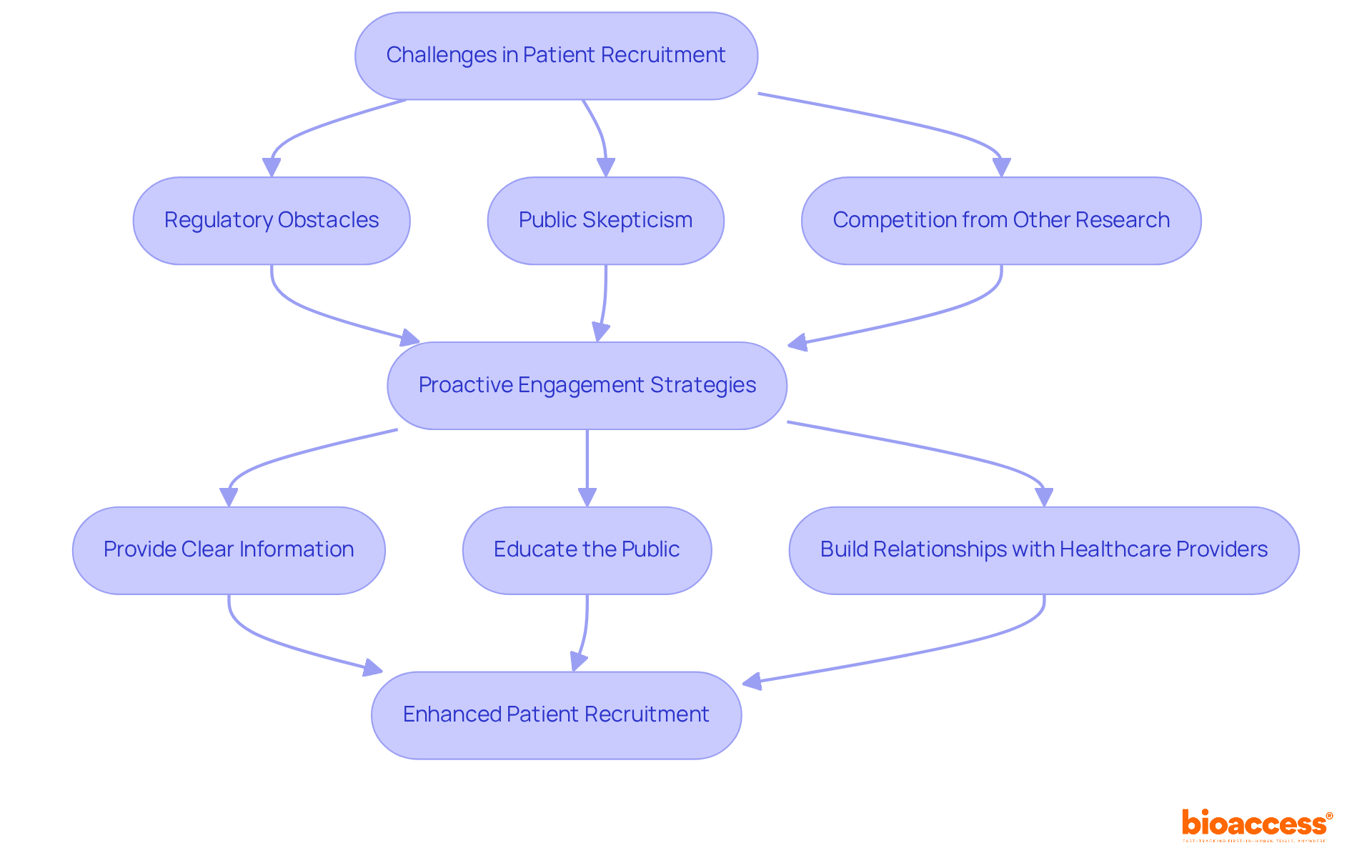
Navigating the complexities of patient recruitment clinical trial Brazil is no small feat. Patient recruitment clinical trial Brazil poses distinct challenges, including regulatory obstacles, public skepticism regarding studies, and competition from other research efforts. Study sponsors must proactively engage with potential participants. They should offer clear and transparent information about the study’s objectives, procedures, and potential benefits. Educational efforts focused on clarifying research studies are crucial for reducing fears and misunderstandings among the public.
Building strong relationships with local healthcare providers is key. It not only promotes referrals but also enhances the credibility of the studies. Sponsors should also explore flexible participation options, such as remote monitoring and telehealth follow-ups, to better accommodate patients’ needs and preferences. By anticipating and addressing these common challenges, clinical study sponsors can foster a more supportive environment for patient recruitment. This proactive approach not only enhances recruitment but also paves the way for more successful clinical trials.
In Brazil, oversight bodies such as ANVISA play a crucial role in the approval process, with timelines for submissions typically ranging from 30 to 90 days. Adherence to ICH-GCP standards is essential, guaranteeing that studies align with international quality criteria. By leveraging these regulatory pathways and focusing on patient-centric approaches, sponsors can enhance patient recruitment clinical trial Brazil efforts and improve overall trial efficiency.
Conclusion
Understanding local cultures is not just beneficial; it’s essential for enhancing patient recruitment in clinical trials across Brazil. By prioritizing cultural acumen and community engagement, sponsors significantly improve participation rates. This ensures that research reflects the populations they aim to serve. Such an approach builds trust and leads to more comprehensive, representative data, ultimately benefiting the broader healthcare landscape.
The article outlines several key strategies for effective recruitment, including:
- Leveraging local languages
- Collaborating with community leaders
- Utilizing digital tools to reach wider audiences
It emphasizes the importance of adapting recruitment methods to regional contexts, whether through traditional outreach in rural areas or digital campaigns in urban settings. Moreover, the article highlights the role of regulatory frameworks and technological innovations, such as AI and telemedicine, in streamlining the recruitment process and enhancing the overall participant experience.
As Brazil emerges as a strategic hub for early-stage clinical trials, navigating the complexities of patient recruitment presents significant challenges. By focusing on community engagement, cultural sensitivity, and innovative technological solutions, sponsors can overcome these common challenges and harness Brazil’s cost efficiency and regulatory advantages. The call to action is clear: proactive engagement with local communities and adaptation of recruitment strategies will not only pave the way for successful trials but also reshape the future of healthcare in Brazil.
Frequently Asked Questions
Why is cultural understanding important for patient recruitment in clinical trials in Brazil?
A deep understanding of local cultures is essential for enhancing patient involvement in research studies. It fosters trust and significantly boosts participation rates by connecting with local communities in culturally aware ways.
How can local languages and community leaders aid in patient recruitment?
Utilizing local languages in hiring materials and involving community leaders as advocates can bridge communication gaps and build rapport with potential participants, making them more likely to engage in the research process.
What strategies can be employed to clarify the research process to potential participants?
Conducting informational sessions in community centers can clarify the research process and address participants’ concerns, creating a welcoming atmosphere for engagement.
How do local festivals and health fairs contribute to patient recruitment?
Many successful hiring initiatives have leveraged local festivals and health fairs to promote medical studies, leading to significant increases in enrollment rates by reaching potential participants in familiar and communal settings.
What role does financial compensation play in encouraging participation?
Providing financial compensation for time and travel expenses can alleviate the burdens associated with participation, further encouraging individuals to join clinical trials.
How can community involvement enhance trust and awareness about research studies?
Collaborating with local leaders and patient advocacy groups helps educate the public about research studies, while digital platforms and social media campaigns can effectively reach broader audiences and target specific demographics.
What compliance requirements must be considered for successful study execution in Brazil?
Understanding the compliance environment, including adherence to ANVISA guidelines and ICH-GCP standards, is crucial for the successful execution of clinical trials in Brazil.
How can the Unified Health System (SUS) assist in patient recruitment for clinical trials?
The Unified Health System (SUS) can provide extensive access to healthcare services, generating a vast pool of potential participants for research studies in Brazil.
How does bioaccess® support Medtech startups in Brazil?
Bioaccess® offers crucial clinical trial insights and market access strategies, helping Medtech startups navigate regulatory complexities and enhance their participant engagement strategies in Brazil.
List of Sources
- Engage Local Communities with Cultural Acumen
- Patient Recruitment and Retention in Brazilian Clinical Trials (https://kuickresearch.com/patient-recruitment-and-retention-in-brazilian-clinical-trials–clinical)
- More patient-centric clinical trials | Science Arena (https://sciencearena.org/en/news/decentralized-clinical-trials-diversity)
- Quotes on Culture and Culturally Sensitive Approaches (https://unfpa.org/resources/quotes-culture-and-culturally-sensitive-approaches)
- Current scenario and future perspectives of clinical research in Brazil: a national survey – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10898894)
- Tailor Recruitment Strategies to Regional Contexts
- Unlocking Brazil’s Clinical Trial Opportunity: A Strategic Roadmap for Healthcare Leaders (https://lek.com/insights/life-sciences-pharma/unlocking-brazils-clinical-trial-opportunity-strategic-roadmap)
- Table 4 Quotes on recruiters’ perception of recruitment from interviews… (https://researchgate.net/figure/Quotes-on-recruiters-perception-of-recruitment-from-interviews-with-recruiters_tbl3_327675032)
- 10 Inspiring Patient Experience Quotes | Relias (https://relias.com/blog/patient-experience-quotes)
- Utilize Technology and Innovative Approaches for Recruitment
- 25+ useful clinical trial recruitment statistics for better results (https://antidote.me/blog/25-useful-clinical-trial-recruitment-statistics-for-better-results)
- Study: Social Media Outperforms Traditional Methods for Recruiting Patients with Depression in Clinical Trials | Applied Clinical Trials Online (https://appliedclinicaltrialsonline.com/view/study-social-media-outperforms-traditional-methods-recruiting-patients-depression-clinical-trials)
- Unsung but Impactful Voices of Clinical Research: Thomas Hohing, Recruitment Specialist at the University of Pennsylvania – TrialX (https://trialx.com/unsung-but-impactful-voices-of-clinical-research-heroes-featuring-thomas-hohing)
- 2025 Trends In Patient Recruitment: From Disruption To Precision (https://clinicalleader.com/doc/trends-in-patient-recruitment-from-disruption-to-precision-0001)
- The Role of Social Media in Enhancing Clinical Trial Recruitment: Scoping Review – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC7652693)
- Address Common Challenges in Patient Recruitment
- Brazil Clinical Trial Patient Recruitment Services Market Size & Outlook, 2033 (https://grandviewresearch.com/horizon/outlook/clinical-trial-patient-recruitment-services-market/brazil)
- More patient-centric clinical trials | Science Arena (https://sciencearena.org/en/news/decentralized-clinical-trials-diversity)
- 5 Common Clinical Trial Recruitment Challenges And Tips To Overcome Them. (https://mbsciences.com/clinical-trial-recruitment-challenges)
- Patient Recruitment Strategies for Clinical Trials (https://freyrsolutions.com/blog/patient-recruitment-strategies-for-clinical-trials)

Leave a Reply