Introduction
In the dynamic landscape of Argentina’s medical technology sector, navigating the complexities of market access is paramount for success. Medtech companies face a myriad of challenges, from stringent regulatory requirements imposed by ANMAT to the necessity of local partnerships that can enhance product acceptance. This article delves into seven essential strategies that can empower these firms to streamline their market entry, optimize clinical trials, and ultimately thrive in a rapidly evolving healthcare environment.
How can companies effectively leverage local expertise and innovative approaches to overcome barriers and seize opportunities in this burgeoning market?
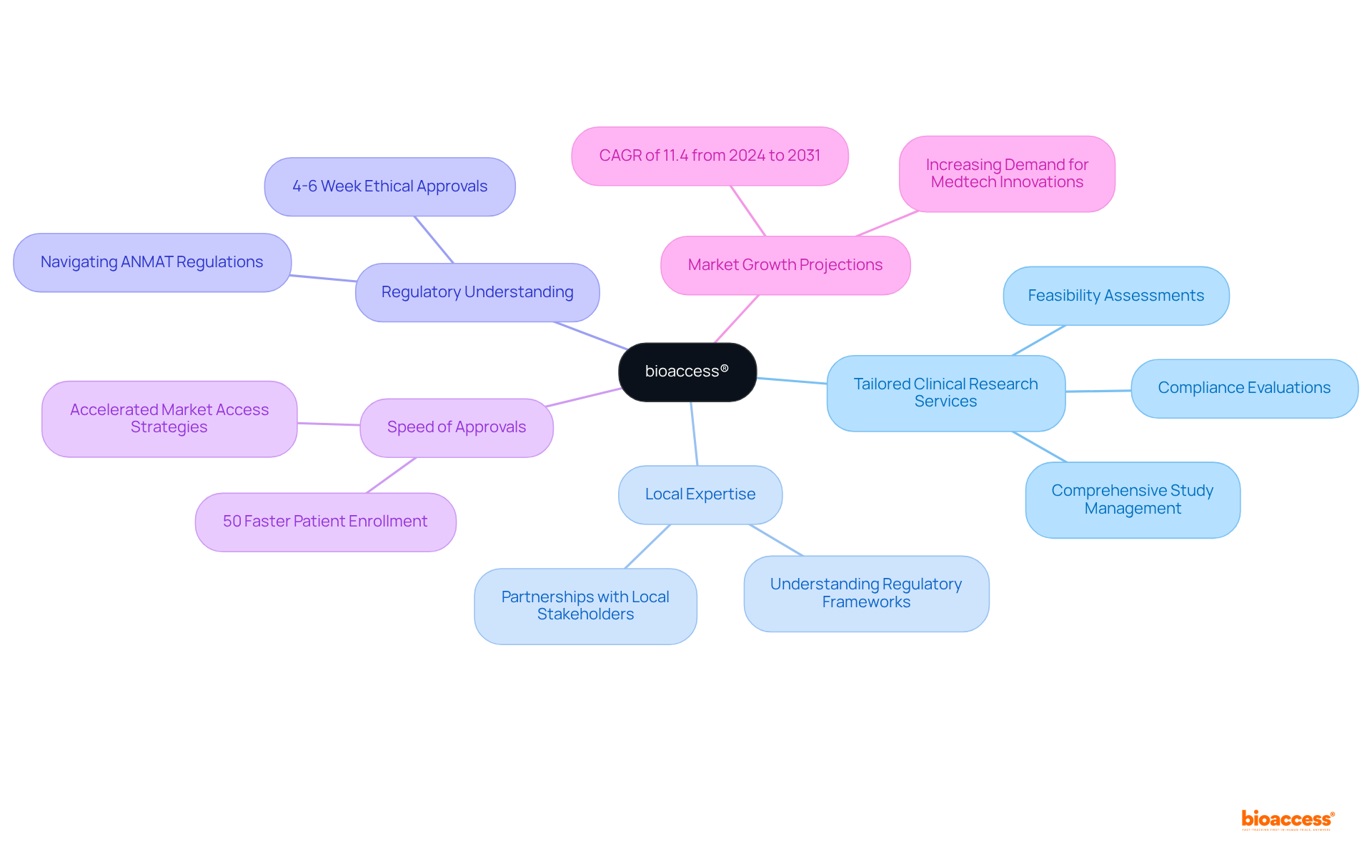
bioaccess®: Accelerate Market Access with Expert Clinical Research Services
bioaccess® distinguishes itself by providing tailored clinical research services that improve for medtech companies in Argentina (ANMAT). By leveraging local expertise and a , bioaccess® ensures the effective implementation of , which facilitates accelerated approvals and supports (ANMAT). Their unwavering commitment to ethical practices and adherence to ANMAT regulations, coupled with effective , positions them as a reliable partner for medical technology innovators.
With an exceptional ability to secure and to expedite than conventional sectors, bioaccess® empowers organizations to implement (ANMAT) with unmatched speed and efficiency. This capability is vital in a competitive landscape where (ANMAT) can significantly impact a product’s success. As the South American healthcare technology sector is projected to expand at a CAGR of 11.4% from 2024 to 2031, leveraging bioaccess®’s comprehensive solutions—including pre-qualified networks and centralized monitoring—can provide a looking to thrive by implementing effective (ANMAT) in this growing market. Dushyanth Surakanti, Founder & CEO of Sparta Biomedical, emphasizes the effectiveness of bioaccess® during its inaugural human trial in Colombia, highlighting the critical role of local expertise in minimizing patient recruitment times.
ANMAT Regulations: Navigate Compliance for Successful Market Entry
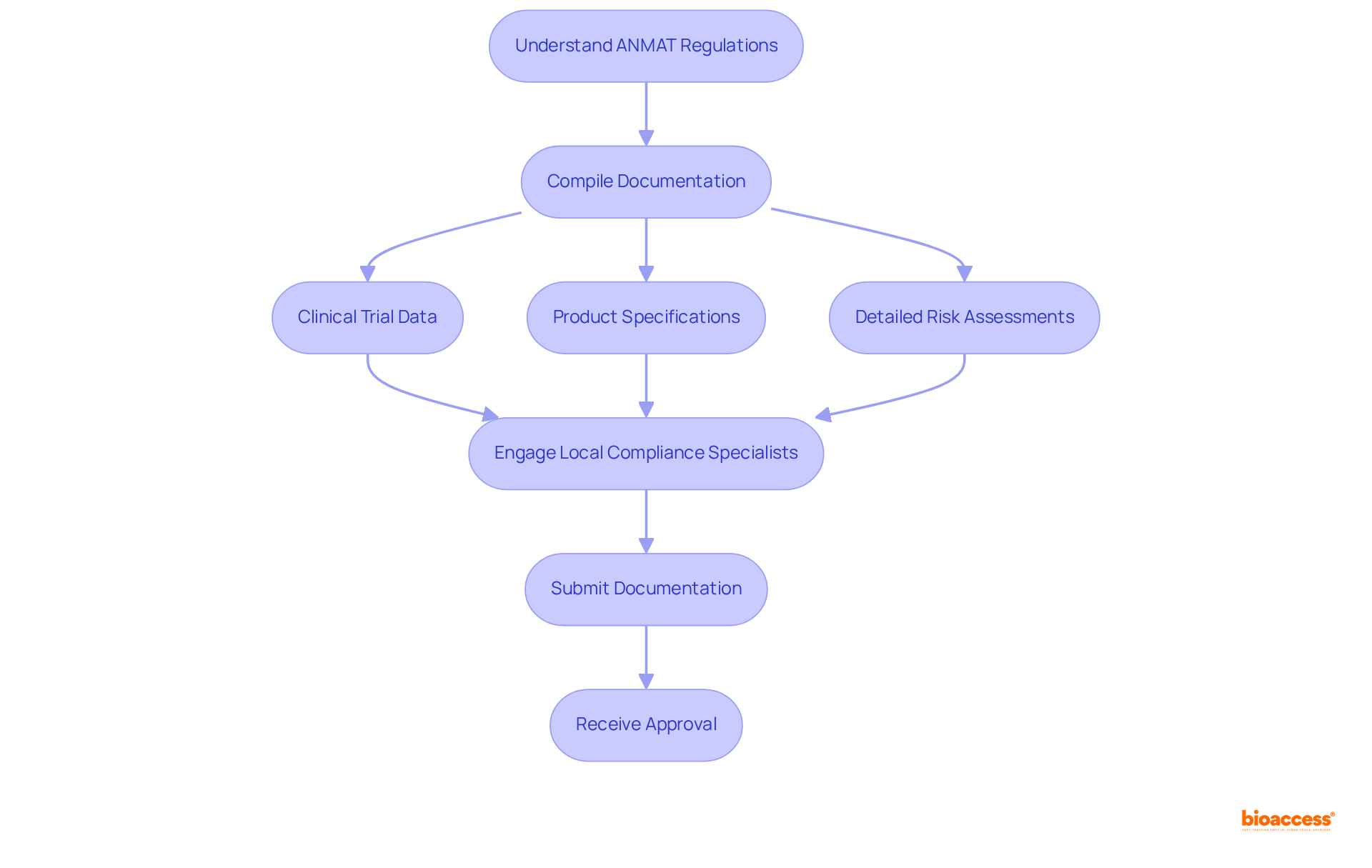
Navigating the compliance environment set by is crucial for Medtech firms seeking successful market entry in Argentina. ANMAT governs the approval process for medical devices, ensuring compliance with rigorous safety and efficacy standards.
To align with , companies must compile , including:
- Product specifications
- Detailed risk assessments
Involving , like those from bioaccess®, can greatly simplify the submission process, efficiently and dependably providing approval and aiding patient recruitment. Bioaccess® focuses on linking cutting-edge medical technology, Biopharma, and Radiopharma startups with leading clinical research locations, thereby improving the effectiveness of and approvals.
Based on industry insights, numerous medical technology firms encounter , which may result in delays in product approvals. Statistics indicate that is a common hurdle for Medtech firms in Argentina, with a significant percentage reporting difficulties in meeting ANMAT’s stringent documentation standards. By adopting a proactive approach and leveraging local expertise from bioaccess®, organizations can enhance their chances of successfully navigating the ANMAT approval process. Reach out to bioaccess® today to discover how we can support you in and speeding up your entry into the Argentine healthcare sector.
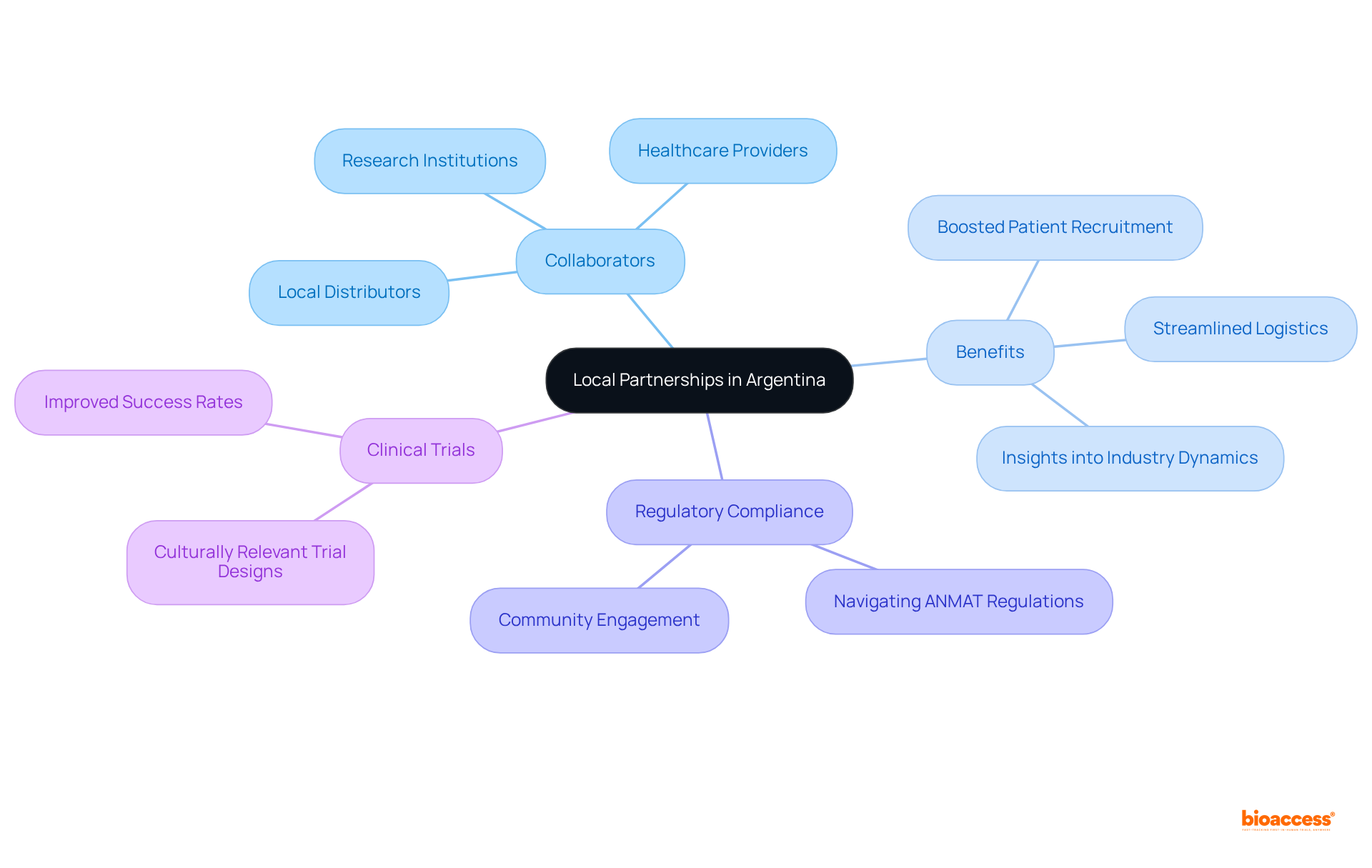
Local Partnerships: Strengthen Your Market Access Strategy in Argentina
Forming local alliances is crucial for aiming to enhance their access strategy in . Collaborating with , , and research institutions yields invaluable insights into industry dynamics and patient requirements, essential for effectively customizing products and services.
These partnerships streamline logistics and significantly boost , vital in a landscape where demand for is rising. By leveraging the expertise and established networks of local partners, businesses can implement effective in (ANMAT) to navigate the complexities of the Argentine healthcare system more efficiently, ensuring compliance with the regulations.
Successful collaborations have demonstrated improved clinical trial success rates, as culturally relevant trial designs resonate better with local populations, leading to higher enrollment and retention. As the Argentine healthcare sector continues to progress, the influence of these will become increasingly important, preparing businesses for success in a competitive environment.
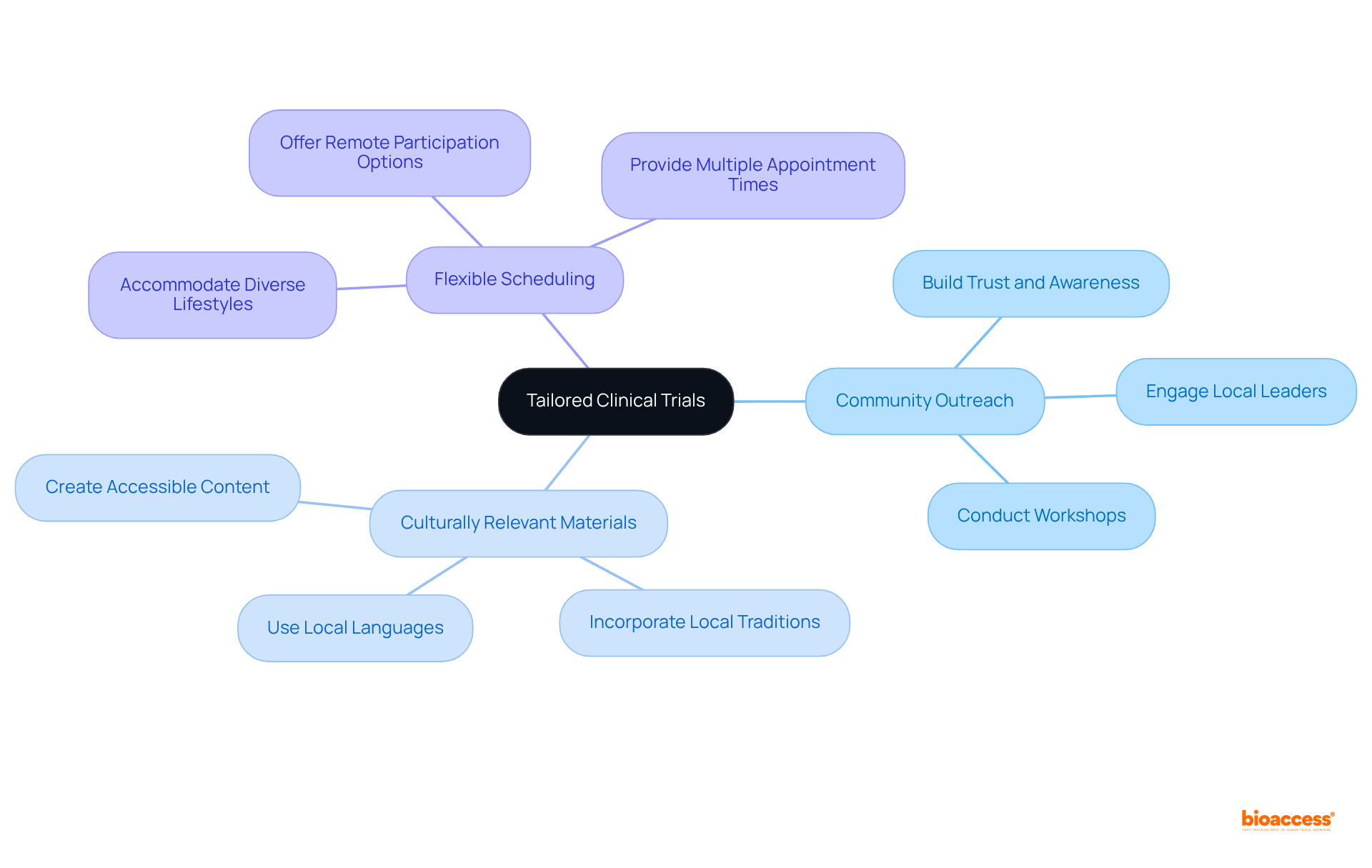
Tailored Clinical Trials: Optimize Recruitment and Retention in Argentina
Designing that reflect the cultural and demographic nuances of the Argentine population is crucial for optimizing recruitment and retention. By incorporating local traditions and preferences into trial designs, medical technology firms can significantly enhance and adherence.
Effective strategies include:
- that build trust and awareness
- The use of culturally relevant materials that resonate with participants
- that accommodate diverse lifestyles
Furthermore, maintaining with participants throughout the trial fosters a sense of partnership and commitment, ultimately leading to improved retention rates and more . This culturally informed approach not only enhances participant experience but also contributes to the in Argentina.
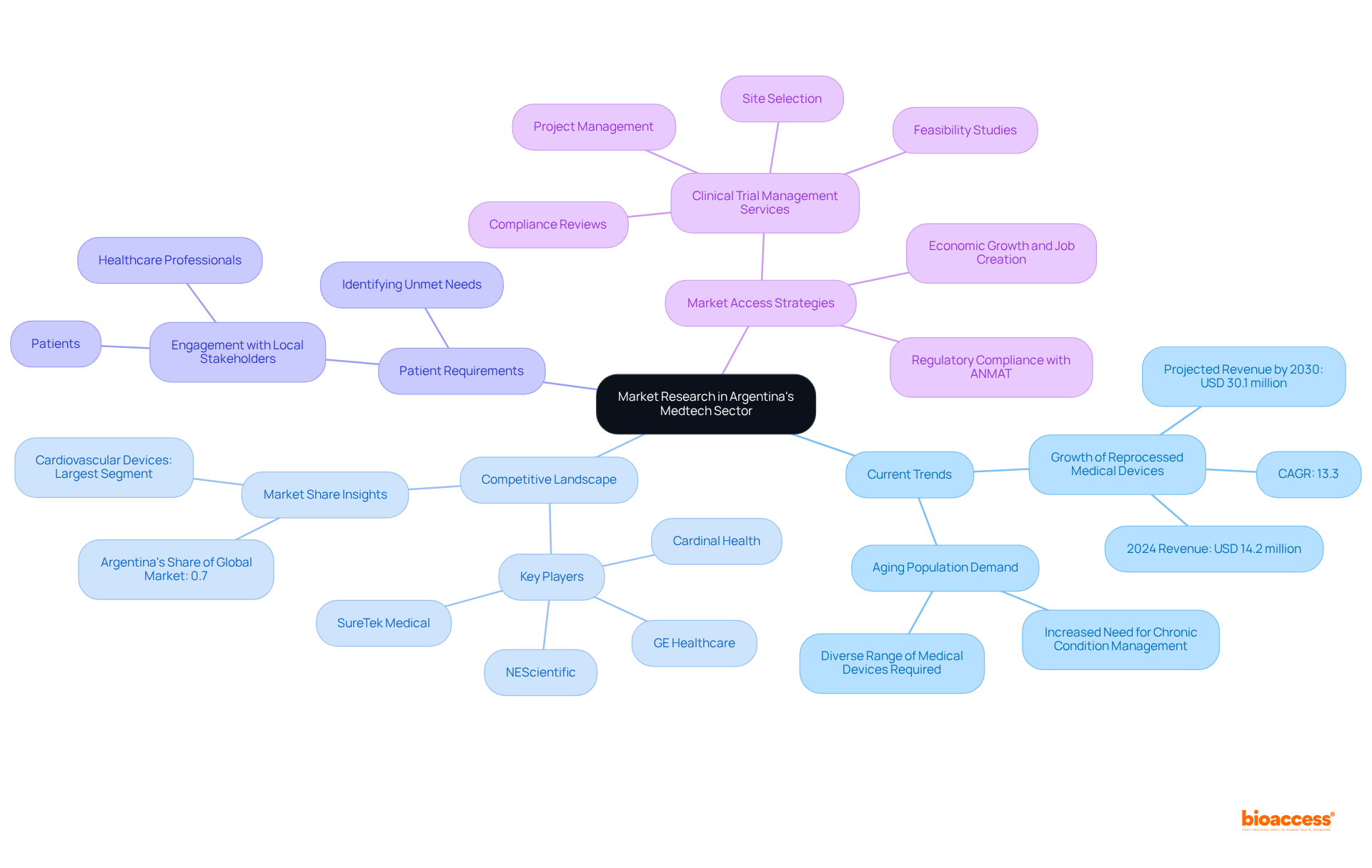
Market Research: Identify Opportunities in Argentina’s Medtech Sector
Conducting comprehensive research on the industry is essential for Medtech firms aiming to uncover within the evolving healthcare sector. This research must include a .
By utilizing both qualitative and quantitative research methods, companies can obtain vital insights that inform product development and marketing strategies. For example, the Argentine and is projected to reach USD 30.1 million by 2030, indicating a compound annual growth rate of 13.3%. Engaging with , including healthcare professionals and patients, enhances understanding of industry dynamics and helps identify unmet needs that can effectively address.
Furthermore, as the aging population in Argentina increases the demand for medical devices, businesses that effectively utilize can position themselves advantageously in a sector poised for growth. Market analysts indicate that the , which presents a significant opportunity that can be leveraged through . To leverage these insights, organizations should consider developing for medtech companies in Argentina (ANMAT) that specifically cater to the needs of this demographic.
Additionally, employing extensive —such as feasibility studies, site selection, compliance reviews, trial setup, import permits, project management, and reporting—as part of can significantly enhance a company’s ability to navigate the . These services not only ensure adherence to ANMAT regulations but also stimulate local economic growth through job creation and improved healthcare outcomes, ultimately fostering international collaboration and innovation within the .
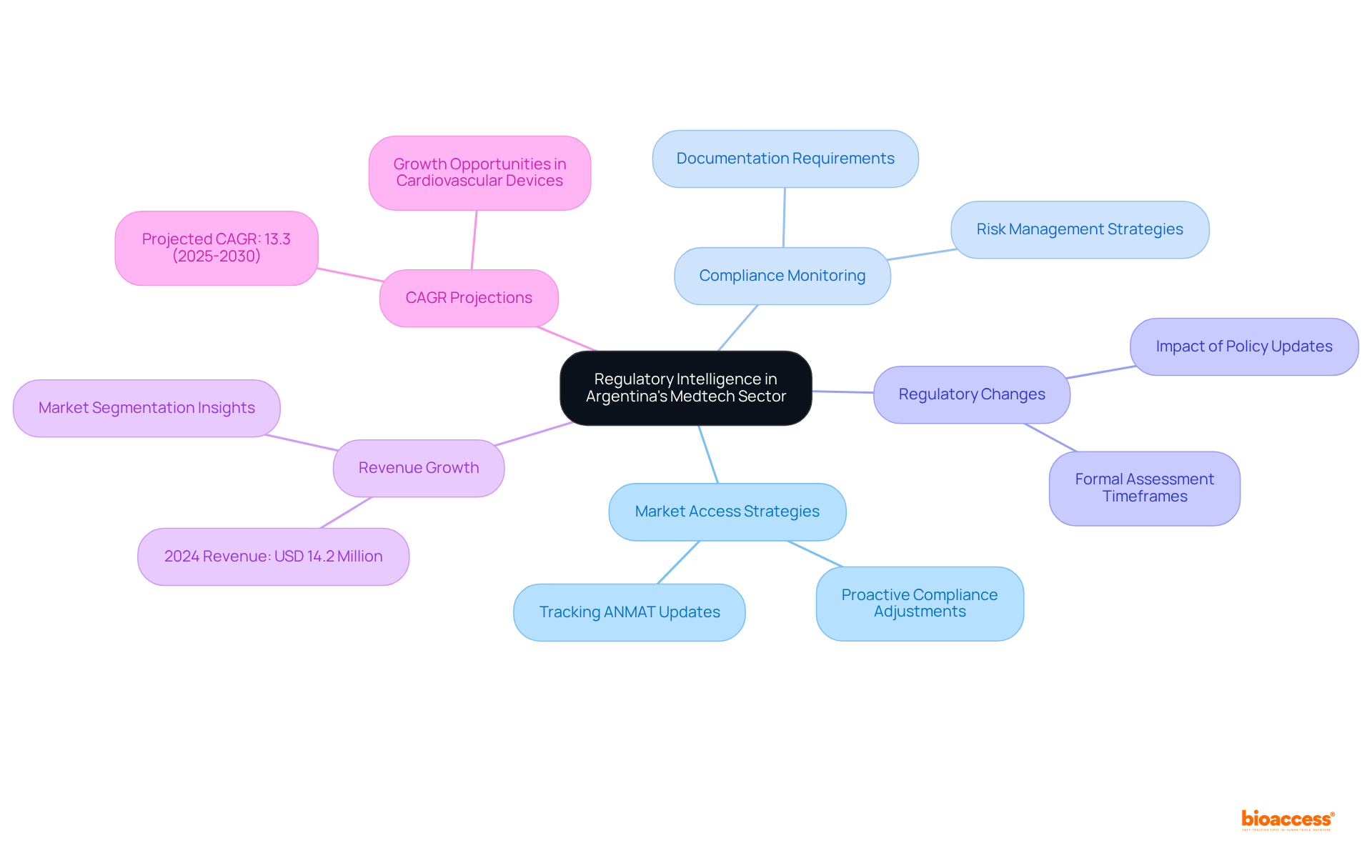
Regulatory Intelligence: Stay Ahead of Changes in Argentina’s Medtech Regulations
In Argentina’s rapidly evolving medical technology landscape, being attuned to and policy updates is crucial for compliance and maintaining a competitive edge. involves the ongoing monitoring of updates from the National Administration of Drugs, Food and Medical Devices (ANMAT) and other pertinent authorities to inform . This proactive strategy enables companies to foresee changes that could influence as well as .
Establishing a robust framework for tracking allows medtech companies to effectively adjust their . For instance, while the formal assessment timeframe for is set at 180 days, it often extends to nearly a year. can significantly . In 2024, Argentina’s , underscoring the potential for growth amidst shifting regulations.
Moreover, with the industry anticipated to expand at a compound annual growth rate (CAGR) of 13.3% from 2025 to 2030, organizations utilizing can not only navigate adherence challenges but also seize new opportunities. By staying updated on , healthcare technology firms can ensure their products meet the latest standards, thereby enhancing their competitive position and fostering innovation in the healthcare sector.
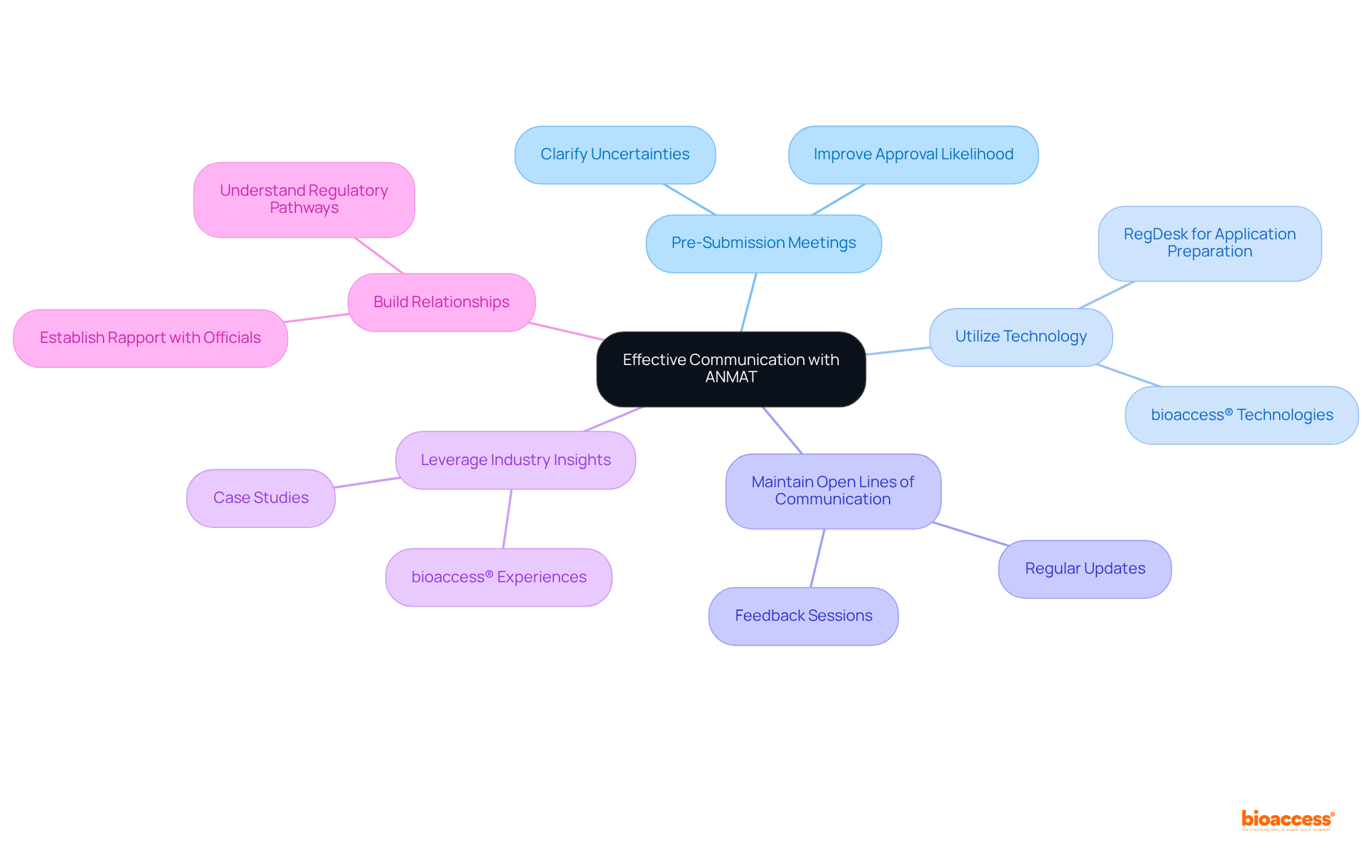
Effective Communication: Engage with ANMAT and Other Regulatory Bodies
Effective communication with ANMAT and other is essential for implementing . Establishing a transparent dialogue clarifies expectations and streamlines the approval process. Companies should prioritize regular updates and feedback sessions with governing bodies to address concerns and ensure alignment with compliance requirements. This proactive approach fosters a collaborative relationship, enhancing credibility and facilitating a more efficient oversight process.
To optimize engagement, companies can adopt several successful strategies:
- Conduct Pre-Submission Meetings: Engaging in clarifies uncertainties and improves the likelihood of approval.
- Utilize Technology: Tools like RegDesk have significantly reduced application preparation time, enabling . bioaccess® employs similar technologies to enhance efficiency in application processes.
- Maintain Open Lines of Communication: Regular interactions with assist in understanding evolving guidelines and expectations, ensuring that businesses remain compliant.
- Leverage Industry Insights: Gaining knowledge from case studies and the experiences of other companies offers valuable lessons in navigating the compliance landscape. For instance, bioaccess® has demonstrated successful approaches in managing and compliance pathways, aiding the rapid advancement of medical devices from to commercialization.
- Build Relationships: Establishing rapport with oversight officials can lead to more favorable outcomes and smoother processes. As bioaccess® exemplifies, understanding the facilitates the swift progression of medical devices from to commercialization.
By applying these strategies, including bioaccess®’s expertise in , , and Post-Market Clinical Follow-Up Studies, healthcare technology firms can enhance their (anmat), capitalizing on the region’s multi-billion dollar healthcare market.
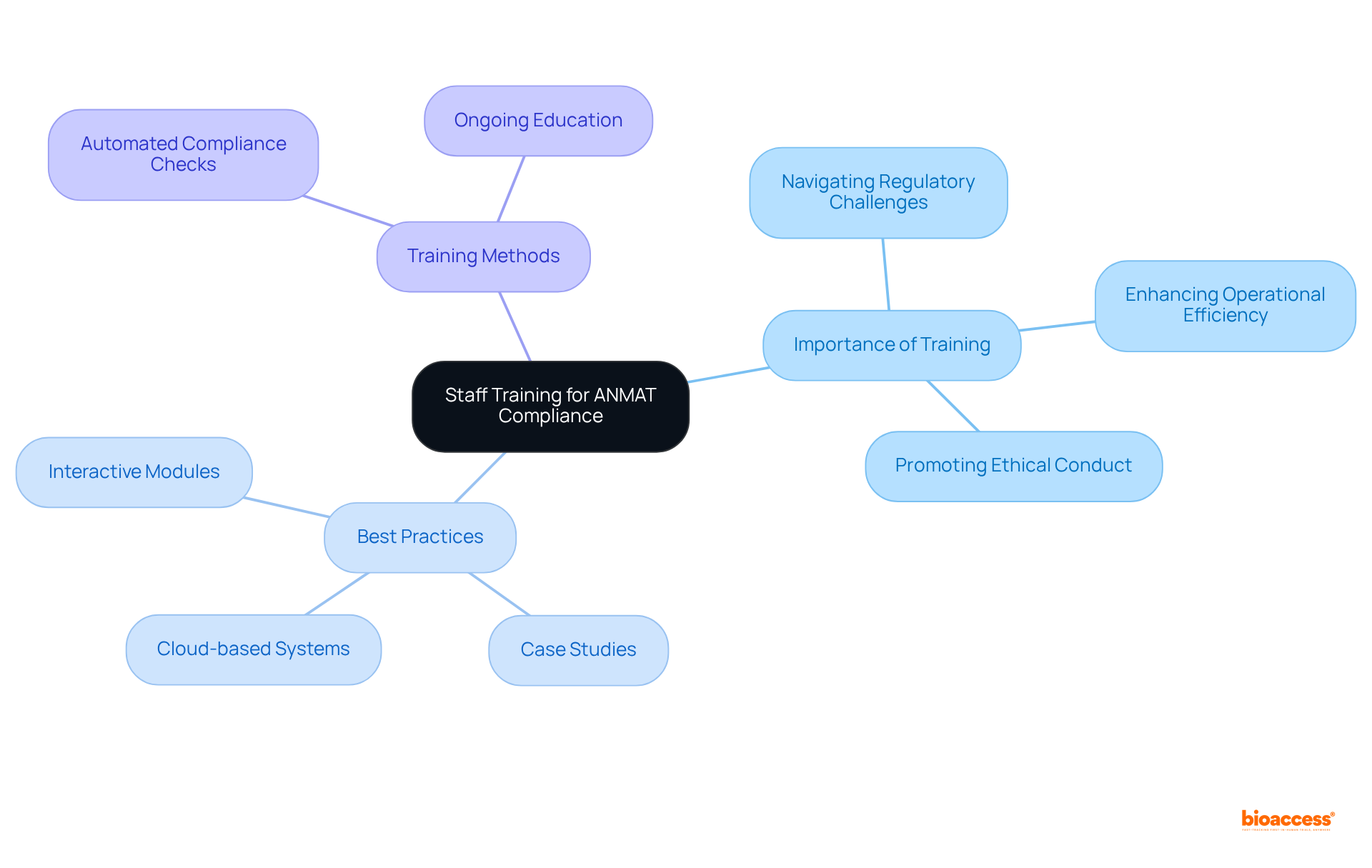
Staff Training: Equip Your Team for Regulatory Compliance Success
Equipping your team with the essential knowledge and skills to navigate is crucial for implementing for medtech companies in Argentina. Implementing regular training sessions that focus on , , and best practices is vital. These sessions should be designed to promote a culture of ongoing learning, ensuring that staff stays informed about the latest legal developments and prepared to address compliance challenges effectively.
on include:
- Utilizing interactive and role-specific training modules that cater to the diverse needs of employees.
- Incorporating to enhance understanding and retention of critical compliance information.
- Leveraging cloud-based training management systems for flexible access to training materials, enabling employees to learn at their own pace while tracking their progress.
Training programs for in Argentina should highlight for medtech companies in Argentina (ANMAT), emphasizing the integration of and to keep pace with evolving industry standards. This includes understanding the feasibility and selection of research locations, compliance reviews, and trial setup processes, which are essential for effective access. By embracing a , companies can greatly improve operational efficiency and lessen the chance of compliance setbacks, ultimately preparing themselves for successful entry into the marketplace.
Leverage Technology: Streamline Your Market Access Processes
In the Medtech sector, the utilization of technology is paramount for streamlining access procedures. —such as electronic data capture systems, project management software, and compliance platforms—significantly enhance data management, improve communication, and facilitate submissions, particularly within Argentina’s evolving landscape. These tools can drastically alleviate while simultaneously increasing submission accuracy. For instance, bioaccess® enables to be enrolled 50% faster than Western sites, achieving $25K in savings per patient with —no rework, no delays. Organizations that have adopted these technologies report and .
As the sector faces increasing compliance challenges, the adoption of these is not merely advantageous but essential for success in 2025 and beyond. Industry leaders emphasize that organizations must adapt to these technological advancements to remain competitive; failure to do so could jeopardize their standing in the industry. With over 40% of enterprises anticipated to encounter difficulties if they do not innovate, incorporating emerges as a critical strategy for healthcare firms aiming to thrive in Argentina’s multi-billion dollar medical sector, especially when considering (ANMAT). Furthermore, as highlighted by industry specialists, the next five years will prove more transformative than the previous fifteen, underscoring the necessity for medical technology firms to embrace these innovations.
Leadership commitment is vital; as Adam Singfield asserts, “You can’t delegate for your company… You and your executives have to own it!” This statement reinforces the notion that the successful implementation of necessitates active involvement from top management.
Continuous Monitoring: Adapt Your Market Access Strategies for Success
Ongoing observation of is crucial for achieving lasting success in the Medtech sector. Organizations must establish distinct metrics to evaluate the effectiveness of their strategies, focusing on such as:
- Penetration rates
Gathering feedback from healthcare professionals, patients, and regulatory bodies is vital for refining these strategies. As industry specialists emphasize, and cultural nuances is essential for .
Regularly examining enables organizations to pinpoint areas for improvement and adjust their strategies accordingly. This proactive approach cultivates a culture of agility and responsiveness, empowering Medtech firms to adeptly navigate the for medtech companies in Argentina (ANMAT) within the .
By embracing flexibility and utilizing insights from performance metrics, companies can sustain a and drive successful market access in 2025 and beyond.
Conclusion
Implementing effective market access strategies for medtech companies in Argentina is essential for navigating the complexities of the healthcare landscape governed by ANMAT regulations. This multifaceted approach encompasses:
- Local partnerships
- Tailored clinical trials
- Comprehensive market research
- Proactive regulatory intelligence
By leveraging local expertise and technology, organizations can enhance their chances of successful product approvals and timely market entry.
Key insights discussed throughout the article highlight the importance of forming local alliances to gain valuable insights into industry dynamics and patient needs. Optimizing clinical trial designs to resonate with the Argentine population and maintaining open communication with regulatory bodies are crucial. Furthermore, investing in staff training and utilizing digital tools can significantly streamline compliance processes, ultimately improving operational efficiency and market access timelines.
As the Argentine medtech sector continues to grow, embracing these strategies is not just advantageous but necessary for sustained success. Companies are encouraged to continuously monitor their market access efforts, adapting strategies based on performance metrics and regulatory changes. By fostering a culture of agility and responsiveness, medtech firms can secure their position in a rapidly evolving market, ensuring they meet the healthcare needs of the population while achieving their business objectives.
Frequently Asked Questions
What services does bioaccess® provide for medtech companies in Argentina?
bioaccess® offers tailored clinical research services that improve market access strategies for medtech companies by leveraging local expertise and understanding of regulatory frameworks to facilitate clinical trials and accelerate approvals.
How quickly can bioaccess® secure ethical approvals for clinical trials?
bioaccess® can secure ethical approvals in just 4-6 weeks.
How does bioaccess® expedite patient enrollment for clinical trials?
bioaccess® expedites patient enrollment at a rate 50% faster than conventional sectors, enabling organizations to implement market access strategies more efficiently.
What is the projected growth rate of the South American healthcare technology sector?
The South American healthcare technology sector is projected to expand at a CAGR of 11.4% from 2024 to 2031.
What documentation is required to comply with ANMAT regulations?
Companies must compile comprehensive documentation including clinical trial data, product specifications, and detailed risk assessments to align with ANMAT regulations.
How can local compliance specialists assist medtech firms?
Local compliance specialists, like those from bioaccess®, can simplify the submission process, provide reliable approvals, and aid in patient recruitment.
What challenges do medtech firms face regarding ANMAT compliance?
Many medtech firms encounter difficulties in understanding changing regulatory standards, leading to delays in product approvals due to stringent documentation requirements.
Why are local partnerships important for medtech companies in Argentina?
Local partnerships with distributors, healthcare providers, and research institutions provide valuable insights into industry dynamics, streamline logistics, and significantly boost patient recruitment for clinical trials.
How do local collaborations influence clinical trial success rates?
Successful collaborations lead to culturally relevant trial designs that resonate better with local populations, resulting in higher enrollment and retention rates in clinical trials.
List of Sources
- bioaccess®: Accelerate Market Access with Expert Clinical Research Services
- bioaccessla.com (https://bioaccessla.com/blog/latam-regulatory-strategist-comparison-chart-for-medtech-innovations)
- meddeviceonline.com (https://meddeviceonline.com/doc/how-chile-is-shaping-medical-device-clinical-trials-in-latin-america-0001)
- bioaccessla.com (https://bioaccessla.com/blog/why-medtech-companies-choose-latin-america-key-factors-explored)
- bioaccess® | MedTech, Biopharma & Radiopharma CRO in Latin America (https://bioaccessla.com/blog/how-to-conduct-clinical-research-in-argentina-for-medical-devices-a-step-by-step-guide)
- ANMAT Regulations: Navigate Compliance for Successful Market Entry
- ANMAT Argentina Medical Device Registration | Pure Global (https://pureglobal.com/markets/argentina)
- Current Trends in Argentina Medical Device Tri… | bioaccess® (https://bioaccessla.com/blog/current-trends-in-argentina-medical-device-trials-and-regulations)
- gpcgateway.com (https://gpcgateway.com/common/news_details/MjA5Mw/Mjc/lib)
- bioaccessla.com (https://bioaccessla.com/blog/how-to-design-clinical-trials-for-medical-devices-in-argentina-a-step-by-step-guide)
- qservegroup.com (https://qservegroup.com/eu/en/services/regulatory-affairs/global-registration/argentina-medical-device-registrations)
- Local Partnerships: Strengthen Your Market Access Strategy in Argentina
- bioaccessla.com (https://bioaccessla.com/blog/argentina-specific-medtech-trial-strategies-for-effective-execution)
- bioaccess® | MedTech, Biopharma & Radiopharma CRO in Latin America (https://bioaccessla.com/blog/understanding-medical-device-research-in-argentina-an-in-depth-tutorial)
- medtechintelligence.com (https://medtechintelligence.com/feature_article/state-latin-american-market-medical-devices/2)
- statista.com (https://statista.com/outlook/hmo/medical-technology/medical-devices/argentina)
- trade.gov (https://trade.gov/country-commercial-guides/argentina-medical-products)
- Tailored Clinical Trials: Optimize Recruitment and Retention in Argentina
- bioaccessla.com (https://bioaccessla.com/blog/enhancing-patient-diversity-in-argentine-clinical-trials)
- alzres.biomedcentral.com (https://alzres.biomedcentral.com/articles/10.1186/s13195-015-0122-5)
- bioaccessla.com (https://bioaccessla.com/blog/top-7-challenges-in-latin-america-trials-essential-insights-you-must-know)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC7342338)
- Can decentralized clinical trials solve attrition? (https://iqvia.com/blogs/2021/03/can-decentralized-clinical-trials-solve-attrition)
- Market Research: Identify Opportunities in Argentina’s Medtech Sector
- mordorintelligence.com (https://mordorintelligence.com/industry-reports/argentina-icu-beds-market)
- grandviewresearch.com (https://grandviewresearch.com/horizon/outlook/reprocessed-medical-devices-market/argentina)
- statista.com (https://statista.com/outlook/hmo/medical-technology/medical-devices/argentina)
- Argentina’s Medical Device Market Will Be Supported By Incre (https://fitchsolutions.com/bmi/medical-devices/argentinas-medical-device-market-will-be-supported-increases-health-budget-2025-despite-currency-depreciation-18-12-2024)
- Regulatory Intelligence: Stay Ahead of Changes in Argentina’s Medtech Regulations
- statista.com (https://statista.com/outlook/hmo/medical-technology/medical-devices/argentina?currency=USD)
- statista.com (https://statista.com/outlook/hmo/medical-technology/argentina)
- Mastering Regulatory Pathways For Approval In … | bioaccess® (https://bioaccessla.com/blog/mastering-regulatory-pathways-for-approval-in-argentina)
- grandviewresearch.com (https://grandviewresearch.com/horizon/outlook/reprocessed-medical-devices-market/argentina)
- Effective Communication: Engage with ANMAT and Other Regulatory Bodies
- Mastering Regulatory Pathways For Approval In … | bioaccess® (https://bioaccessla.com/blog/mastering-regulatory-pathways-for-approval-in-argentina)
- qreg.co.uk (https://qreg.co.uk/argentina-regulatory-requirements)
- Regulatory Considerations For Trials In Argent… | bioaccess® (https://bioaccessla.com/blog/regulatory-considerations-for-trials-in-argentina-key-insights-and-processes)
- researchgate.net (https://researchgate.net/publication/299670317_The_Regulatory_Framework_and_Case_Studies_from_Argentina)
- How To Conduct Pivotal Studies For Medical Dev… | bioaccess® (https://bioaccessla.com/blog/how-to-conduct-pivotal-studies-for-medical-device-approval-in-argentina-a-comprehensive-guide)
- Staff Training: Equip Your Team for Regulatory Compliance Success
- compliancequest.com (https://compliancequest.com/training-compliance-management)
- aurevia.com (https://aurevia.com/learning/regulatory-essentials-in-health-tech)
- simbo.ai (https://simbo.ai/blog/the-role-of-compliance-training-in-the-medical-technology-industry-educating-professionals-to-ensure-ethical-interactions-2703882)
- 6 Important Quotes from Medtech Leaders (https://mddionline.com/business/6-important-quotes-from-medtech-leaders)
- The Top 5 Challenges in Medical Device Regulatory Compliance and How to Overcome Them – Nerac (https://nerac.com/the-top-5-challenges-in-medical-device-regulatory-compliance-and-how-to-overcome-them)
- Leverage Technology: Streamline Your Market Access Processes
- iqvia.com (https://iqvia.com/locations/united-states/blogs/2024/02/top-10-us-market-access-trends-for-2024)
- 24 Inspiring Digital Transformation Quotes | Vista Projects (https://vistaprojects.com/digital-transformation-quotes)
- quixy.com (https://quixy.com/blog/top-digital-transformation-quotes)
- meditechinsights.com (https://meditechinsights.com/market-access-solutions-market)
- 60 Game-Changing Quotes on Digital Transformation (https://deliberatedirections.com/digital-transformation-quotes)
- Continuous Monitoring: Adapt Your Market Access Strategies for Success
- 12 Best Quotes on the Future of Healthcare (https://klara.com/blog/12-best-quotes-on-the-future-of-healthcare)
- meditechinsights.com (https://meditechinsights.com/market-access-solutions-market)
- statista.com (https://statista.com/outlook/hmo/medical-technology/worldwide)
- MedTech Industry Growth: Latest Market Stats (https://patentpc.com/blog/medtech-industry-growth-latest-market-stats)

Leave a Reply