Introduction
ISO 14971 stands as a pivotal framework for risk management within the medical device industry, steering manufacturers through the complexities of hazard identification and mitigation. By adhering to this globally recognized standard, companies not only bolster the safety and efficacy of their products but also foster trust among stakeholders throughout the healthcare spectrum.
As the medical landscape in Latin America continues to evolve, manufacturers face the pressing question: how can they effectively navigate the intricacies of compliance while ensuring patient safety and market accessibility?
This article explores the significance of ISO 14971 in LATAM, examining its historical development, key components, and the substantial impact it has on clinical trials and regulatory practices in the region.
Define ISO 14971: The Framework for Risk Management in Medical Devices
stands as a globally recognized standard that delineates the for medical instruments. It offers a systematic approach for manufacturers to identify, assess, manage, and supervise risks throughout the product lifecycle. This standard is essential in ensuring that are safe for both patients and users, as it emphasizes .
By adopting , manufacturers not only meet regulatory requirements but also significantly bolster the of their products. This framework cultivates trust among stakeholders – including regulatory bodies, healthcare providers, and patients – by ensuring that potential hazards are systematically identified and mitigated.
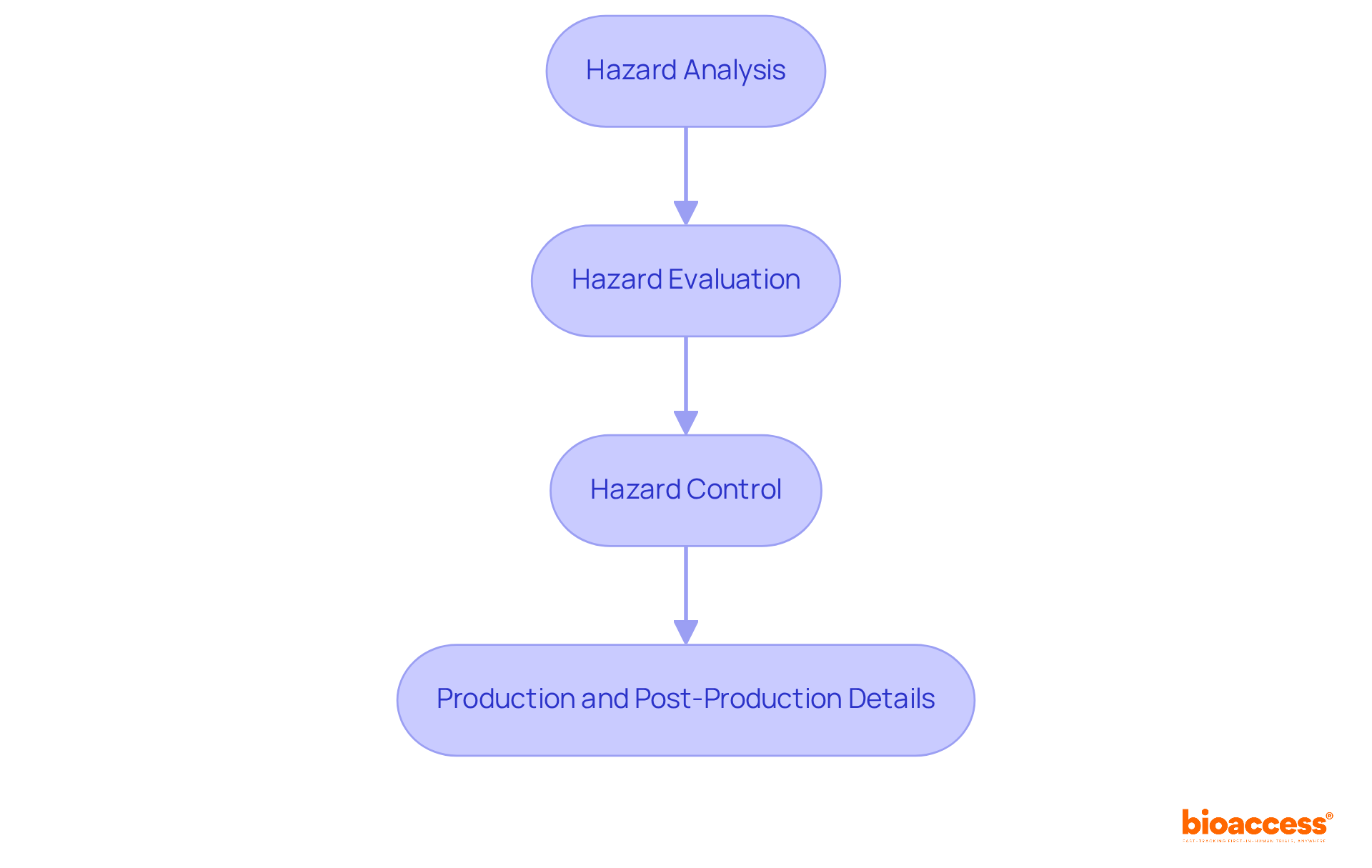
The outlined in includes critical steps:
- Hazard analysis
- Hazard evaluation
- Hazard control
- Production and post-production details
Organizations that have embraced report improved outcome control, underscoring the importance of . Furthermore, integrating hazard oversight into the product development process has been shown to streamline , facilitating quicker market access.
Experts emphasize the in the medical equipment sector. Professionals like Ana Criado, Director of Regulatory Affairs and CEO of Mahu Pharma, assert that compliance with this standard is crucial for maintaining high safety standards. As the medical equipment landscape continues to evolve, the emphasis on robust safety practices, as outlined in , remains a cornerstone of product development and regulatory compliance.
Trace the Evolution of ISO 14971: Historical Context and Development
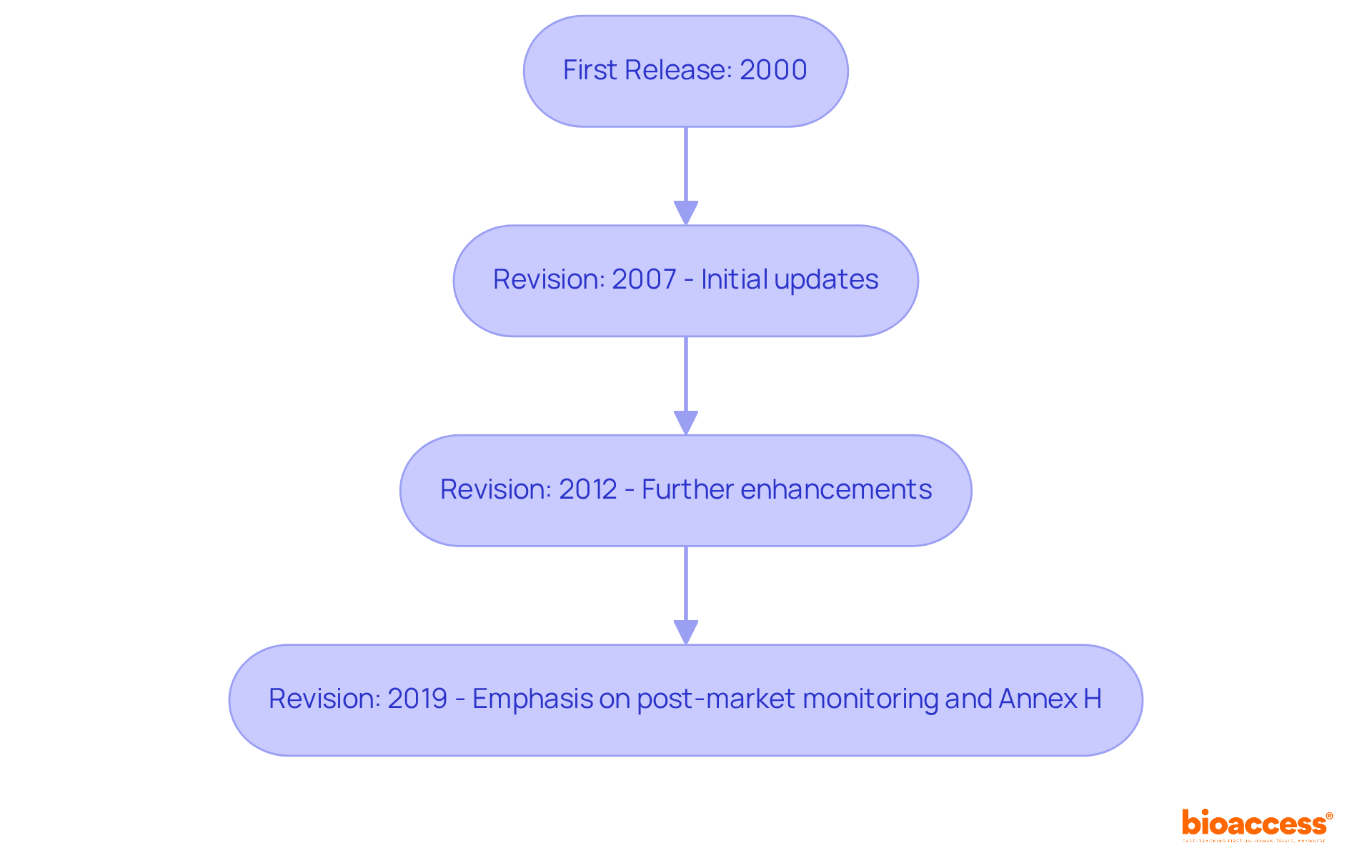
, first released in 2000, was designed to meet the increasing demand for a standardized approach to managing uncertainties in the medical equipment sector. This essential standard has evolved through several revisions, with the published in 2019. Each update reflects contributions from industry experts and advancements in technology, highlighting the dynamic nature of and the challenges they present. Notably, the 2019 revision places a stronger emphasis on and the critical need for .
A significant addition in this revision is Annex H, which evaluates and addresses issues related to devices that previously did not comply with . This inclusion underscores the standard’s commitment to improving compliance. Moreover, ISO/TR 24971:2020 offers practical examples for conducting a , serving as a valuable resource for manufacturers. This evolution illustrates the necessity of adapting LATAM relevance practices to ensure and meet in an ever-changing healthcare landscape.
Statistics indicate that adhering to the revised :2019 can significantly reduce . Manufacturers are now required to integrate proactive data collection into their risk mitigation strategies. Case studies reveal that organizations implementing these changes have improved their oversight practices, leading to enhanced product safety outcomes and better alignment with regulatory standards. As Ed Bills, a leading expert in medical equipment hazard oversight, emphasizes, ‘Maintaining the safety oversight file current throughout the product life cycle is essential for ensuring safety and effectiveness.
Examine Key Components of ISO 14971: Risk Management Processes and Applications
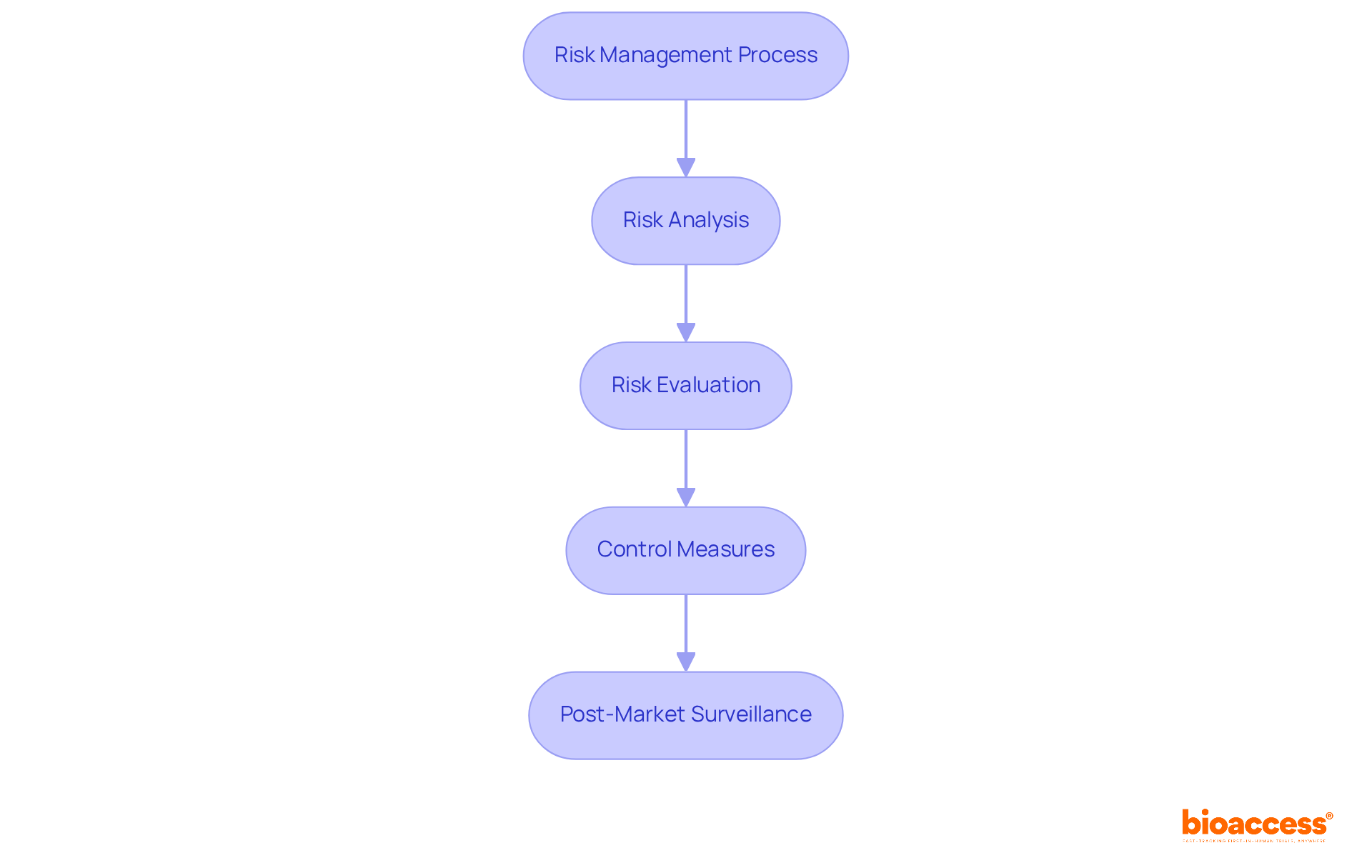
plays a crucial role in establishing a robust management process for , encompassing analysis, evaluation, control, and . At the forefront is , which identifies potential hazards linked to , such as design flaws or . Following this, the evaluation of these hazards assesses their significance against predefined criteria, enabling manufacturers to prioritize which risks demand immediate attention.
Implementing effective control measures is vital for mitigating identified hazards. This could involve redesigning products to enhance , conducting additional testing, or improving processes to ensure risks are minimized to acceptable levels. For example, a medical equipment company might automate production processes to reduce human error, thereby enhancing overall safety.
Equally important is , which ensures continuous monitoring of a device’s performance and safety after its release into the market. Regular evaluations of post-market data can reveal new concerns, prompting timely actions to maintain compliance and protect patient safety.
Current best practices in analysis and evaluation advocate for a dynamic approach, with assessments conducted multiple times a year, particularly in response to evolving regulatory standards or product modifications. This proactive strategy not only aligns with requirements but also highlights the importance of LATAM relevance in bolstering the reliability of available to consumers.
Industry experts emphasize that effective communication of hazard control activities to all stakeholders is essential for raising awareness of potential threats and their mitigations. Moreover, team involvement in hazard identification is critical for comprehensive coverage. Tools like (FMEA) and and Critical Control Points (HACCP) are commonly employed for threat evaluation, enhancing the efficiency of the process.
By embracing a comprehensive strategy, manufacturers can significantly enhance patient safety and product reliability, ultimately leading to improved health outcomes.
Highlight the Importance of ISO 14971 in LATAM Clinical Trials: Relevance and Impact
In Latin America, the risk management is essential for ensuring that medical equipment meets rigorous safety and efficacy standards throughout . With the region’s and continuing to grow, the LATAM relevance enables manufacturers to navigate the complex regulatory environment efficiently and access diverse markets. For instance, Colombia boasts an impressive average clinical trial approval duration of just 90 to 120 days, significantly enhanced by robust safety oversight practices aligned with ISO 14971.
Bioaccess offers a comprehensive process for advancing , which includes:
- Feasibility studies
- Site selection
- Compliance reviews
- Trial setup
- Import permits
- Project oversight
- Reporting
By implementing these thorough management strategies, companies can bolster their credibility with regulatory authorities and healthcare providers, leading to expedited approvals and smoother market entry. Recent updates to ISO 14971 emphasize the importance of risk management , highlighting the necessity of continuous , both vital for maintaining compliance and ensuring patient safety.
Moreover, within the medical sector, building trust among patients and healthcare professionals alike. As Latin America positions itself as a prime destination for clinical research, the LATAM relevance will be a crucial factor in the success of . This commitment to quality not only enhances market access but also gives manufacturers a competitive edge, where is increasingly viewed as the ‘gold standard’ for market entry.
Conclusion
ISO 14971 stands as a crucial framework for risk management in the medical device industry, especially in Latin America. It establishes a structured approach to identifying, assessing, and controlling risks, ensuring that medical devices not only comply with regulatory requirements but also uphold the highest safety and efficacy standards. By adopting ISO 14971, stakeholders – including healthcare providers and patients – can trust that thorough hazard management is prioritized throughout the product lifecycle.
This article explores the historical evolution of ISO 14971, emphasizing its revisions and the increasing focus on post-market surveillance and continuous risk evaluation. Key components of the standard, such as:
- Hazard analysis
- Evaluation
- Control
- Post-market monitoring
are outlined as essential for maintaining product safety. Insights from industry experts highlight the necessity of integrating these practices into product development, particularly in a rapidly evolving healthcare landscape.
In summary, the significance of ISO 14971 in Latin America is paramount. As the region’s medical equipment market expands, manufacturers must prioritize compliance with this standard to ensure patient safety and enhance their competitive edge. By embracing the robust risk management practices outlined in ISO 14971, companies can facilitate quicker market access and contribute to a culture of safety and quality in healthcare. This commitment to high standards will ultimately lead to improved health outcomes and foster innovation in the medical device sector across LATAM.
Frequently Asked Questions
What is ISO 14971?
ISO 14971 is a globally recognized standard that outlines the hazard control process for medical devices, providing a systematic approach for manufacturers to identify, assess, manage, and supervise risks throughout the product lifecycle.
Why is ISO 14971 important for medical device manufacturers?
ISO 14971 is important because it helps manufacturers ensure that medical devices are safe for patients and users. It emphasizes thorough evaluation and oversight practices, which bolster the safety and effectiveness of products while meeting regulatory requirements.
What are the key steps in the hazard assessment process outlined in ISO 14971?
The key steps in the hazard assessment process include hazard analysis, hazard evaluation, hazard control, and production and post-production details.
How does adopting ISO 14971 benefit organizations?
Organizations that adopt ISO 14971 report improved outcome control, which helps reduce the likelihood of adverse events. Additionally, integrating hazard oversight into product development can streamline regulatory submissions, facilitating quicker market access.
What do experts say about the importance of ISO 14971 in the medical equipment sector?
Experts emphasize that compliance with ISO 14971 is crucial for maintaining high safety standards in the medical equipment sector. It remains a cornerstone of product development and regulatory compliance as the landscape continues to evolve.
List of Sources
- Define ISO 14971: The Framework for Risk Management in Medical Devices
- Failure mode effect analysis use and limitations in medical device risk management (https://sciencedirect.com/science/article/pii/S2199853124002336)
- The Importance of ISO 14971 in Medical Device Risk Management (https://sobelconsult.com/success-in-product-design-the-importance-of-iso-14971)
- greenlight.guru (https://greenlight.guru/blog/iso-14971-risk-management)
- Stats for Medical Device ARTG Applications in FY25 | Gary Burgess (https://linkedin.com/posts/gary-burgess-ra_stats-for-medical-device-artg-applications-activity-7398520988830789632-Mwwm)
- Inside Look into ISO 14971:2019 & ISO TR 24971:2020 from the Author’s Point of View (https://greenlight.guru/blog/iso-14971-2019-iso-tr-24971-2020-author-point-of-view)
- Trace the Evolution of ISO 14971: Historical Context and Development
- Inside Look into ISO 14971:2019 & ISO TR 24971:2020 from the Author’s Point of View (https://greenlight.guru/blog/iso-14971-2019-iso-tr-24971-2020-author-point-of-view)
- ISO 14971: 2019: 3 Key Changes from ISO 14971: 2007 | RQM+ (https://rqmplus.com/blog/key-changes-from-iso-149712019-from-iso-149712007)
- From Military to MedTech: The Unexpected Path to ISO 14971 Risk Management with Edwin Bills (https://greenlight.guru/blog/from-military-to-medtech-the-unexpected-path-to-iso-14971-risk-management-with-edwin-bills)
- Analyzing the Changes to ISO 14971:2019 – The Auditor (https://theauditoronline.com/analyzing-the-changes-to-iso-149712019)
- A comprehensive guide to ISO 14971: Risk management for medical devices (https://qualio.com/blog/iso-14971)
- Examine Key Components of ISO 14971: Risk Management Processes and Applications
- Medical Device Risk Analysis | ISO 14971 Compliance Guide (https://elexes.com/medical-device-risk-analysis-guide)
- The Importance of Risk Management in Medical Device Development (https://meddux.com/blog/importance-risk-management-medical-device-development)
- c2a-sec.com (https://c2a-sec.com/60-healthcare-and-medical-device-cybersecurity-risk-statistics-for-2025)
- medicaldevicehq.com (https://medicaldevicehq.com/articles/the-illustrated-guide-to-risk-management-for-medical-devices-and-iso-14971)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC9507292)
- Highlight the Importance of ISO 14971 in LATAM Clinical Trials: Relevance and Impact
- ketryx.com (https://ketryx.com/blog/iso-14971-a-comprehensive-guide-to-risk-management-in-medical-devices)
- bioaccessla.com (https://bioaccessla.com/es/blog/10-key-latin-america-medical-device-standards-you-must-know)
- Latin America Medical Device Market Size | 2019-2033 (https://datacuberesearch.com/latin-america-medical-device-market)
- Latin America Medical Devices Market Size & Share, 2034 (https://marketdataforecast.com/market-reports/latin-america-medical-devices-market)
- greenlight.guru (https://greenlight.guru/blog/iso-14971-risk-management)

Leave a Reply