Introduction
In the complex world of Medtech and Biopharma, following FDA annual report guidance is not just a regulatory obligation; it’s a vital element that upholds the integrity and transparency of clinical research. Organizations can gain a competitive advantage by grasping the key components outlined in the FDA’s guidance for 2024, which aims to improve compliance and public health outcomes. Yet, as the regulatory landscape evolves, how can companies adeptly navigate the challenges these requirements present to secure timely approvals and achieve successful product commercialization?
bioaccess: Accelerating Clinical Research in Compliance with FDA Guidance
bioaccess® harnesses its extensive experience in navigating FDA regulations to streamline . By ensuring strict adherence to , bioaccess® accelerates the approval and enrollment stages, allowing Medtech and Biopharma innovators to bring their offerings to market more swiftly. Our comprehensive services encompass:
- Feasibility assessments
- Investigator selection
- Meticulous project management
All of which enhance the integrity of . This unwavering commitment to not only fosters trust with stakeholders and regulatory bodies but also establishes bioaccess® as the premier CRO in Latin America for expedited .
In the rapidly evolving Medtech landscape, the challenges of clinical research can be daunting. However, bioaccess® stands ready to address these hurdles, providing tailored solutions that meet the unique needs of each client. By collaborating with us, innovators can navigate the complexities of with confidence, knowing they have a partner dedicated to their success.
As we look to the future, the importance of collaboration in clinical research cannot be overstated. Together, we can . Reach out to bioaccess® today to explore how we can support your clinical research endeavors.
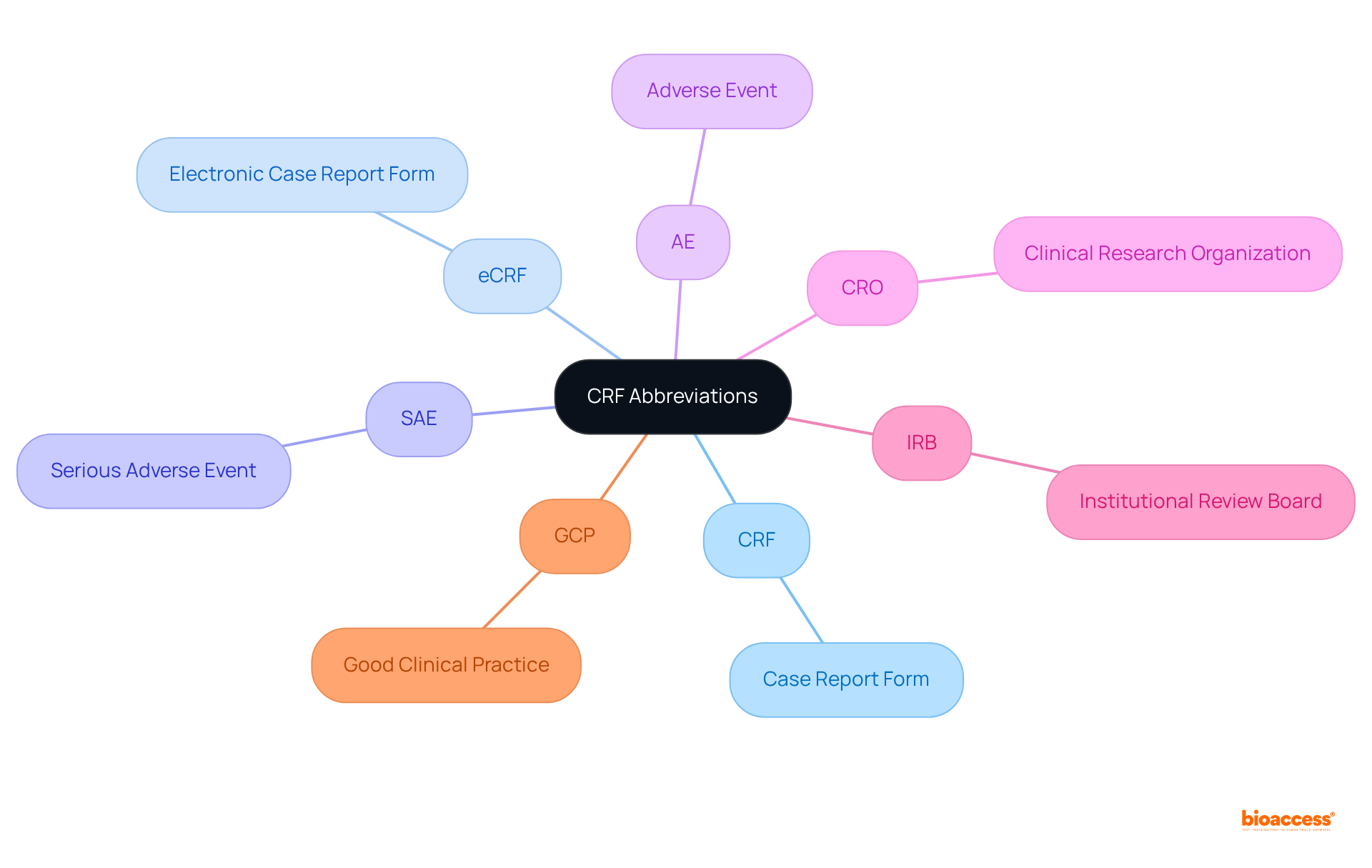
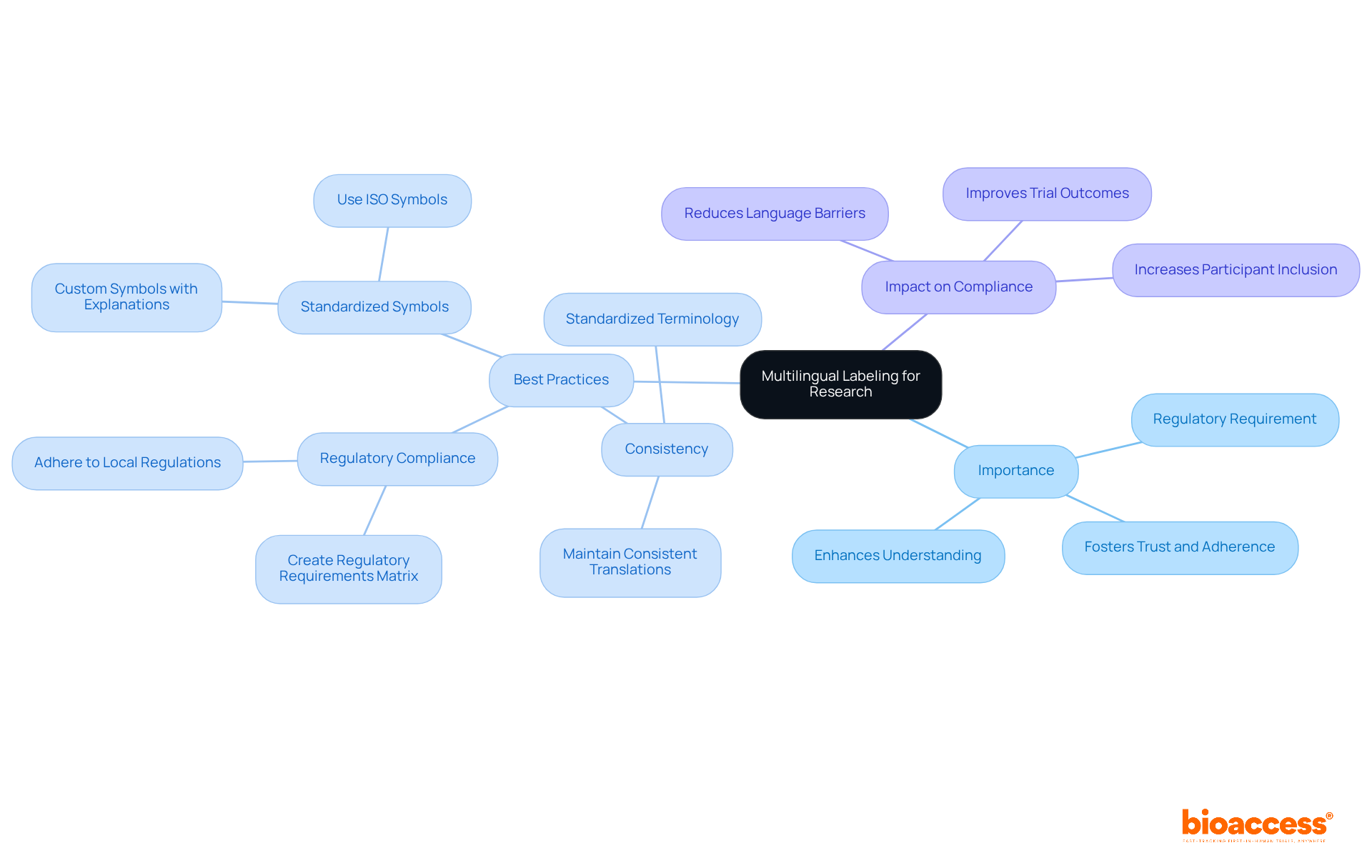
Key Components of the FDA Annual Report Guidance for 2024
The for 2024 outlines several critical components that organizations must address to ensure compliance and effective communication with the agency. This guidance is and enhancing public health outcomes.
- Summary of : A thorough overview of both ongoing and completed trials is essential for transparency and accountability. This transparency fosters trust and ensures that stakeholders are informed about the progress and results of clinical investigations.
- : Detailed documentation of adverse events and safety monitoring is crucial for evaluating the safety and efficacy of the item. Such reports not only protect participants but also provide valuable data for regulatory assessments.
- : Companies are required to present robust data that supports the efficacy and safety of their offerings, reinforcing the importance of sound statistical practices. This data is vital for substantiating claims and ensuring that products meet regulatory standards.
- : Comprehensive documentation demonstrating adherence to FDA regulations throughout the trial process is vital for maintaining credibility and facilitating timely approvals. Organizations must prioritize compliance to navigate the complexities of the regulatory landscape effectively.
These components are not only fundamental for compliance but also play a significant role in enhancing public health outcomes by ensuring that critical information is readily accessible to the FDA, in accordance with the . By addressing these areas, organizations can contribute to a more transparent and effective environment.
Implications of FDA Guidance for Medtech and Biopharma Industries
The implications of the on the Medtech and Biopharma industries are significant and multifaceted. Businesses must navigate an evolving regulatory environment that directly affects development timelines and market strategies. Key implications include:
- Increased Transparency: demand more comprehensive documentation and proactive communication with the FDA, fostering a culture of accountability and clarity in the approval process.
- Faster Approvals: are designed to expedite product approvals, thereby improving patient access to innovative therapies. For instance, the is currently between 140 to 175 days, reflecting ongoing efforts to enhance efficiency.
- Innovation Pressure: The FDA motivates companies to innovate while ensuring adherence to regulations, establishing a delicate balance between speed and regulatory observance. This is particularly evident as the agency adapts to the rapid growth of AI and digital health technologies, necessitating new frameworks for evaluation.
In this context, partnering with bioaccess® can significantly enhance your . With over 20 years of experience in Medtech, bioaccess® specializes in , including (EFS), (FIH), Pilot Studies, Pivotal Studies, and Post-Market Clinical Follow-Up Studies (PMCF). Their customized method and adaptability, along with extensive services like feasibility studies, site selection, , trial setup, import permits, project management, and reporting, are intended to assist companies in managing the intricacies of FDA guidance efficiently. These factors collectively shape the strategic direction of companies within these sectors, compelling them to align their development processes with the FDA’s annual report guidance.
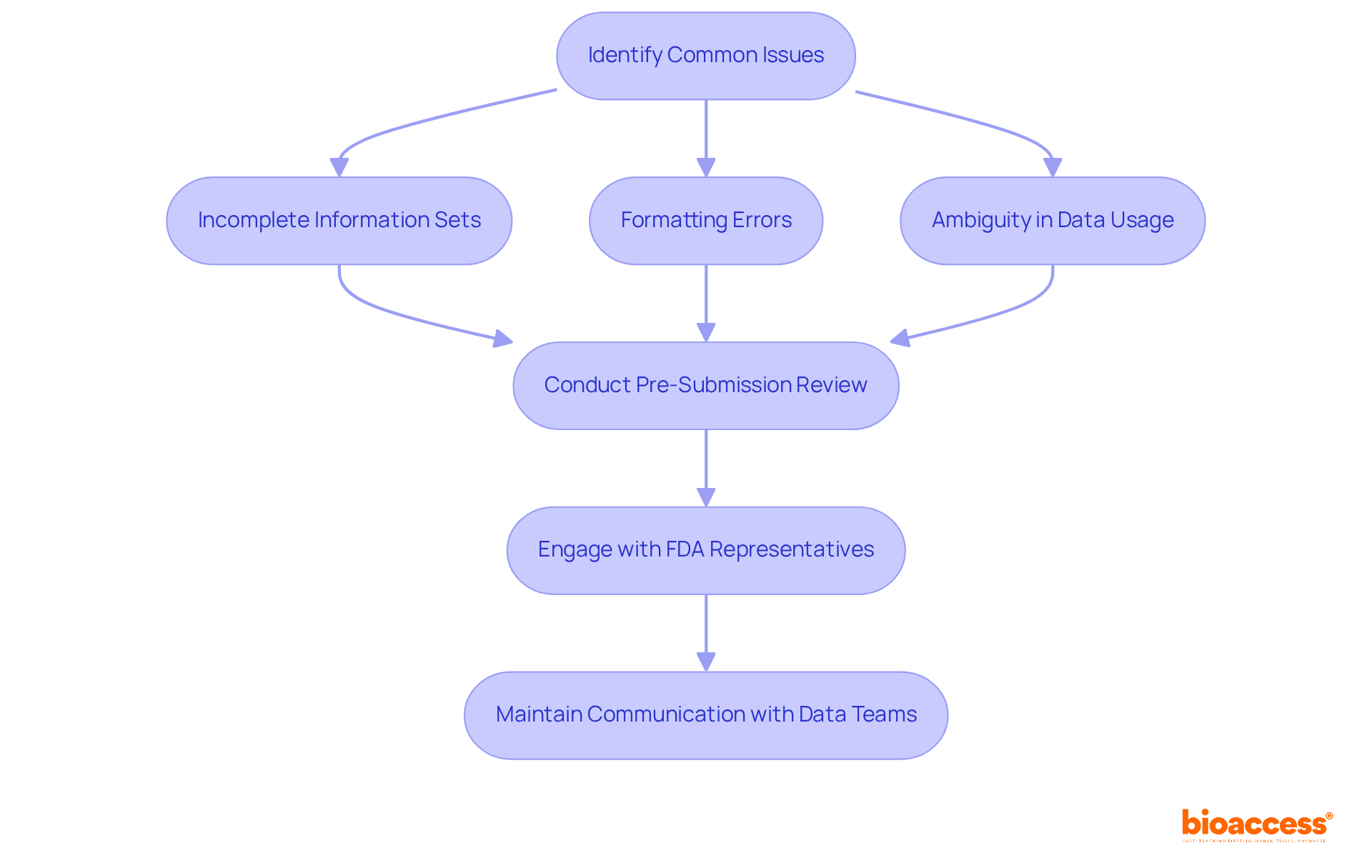
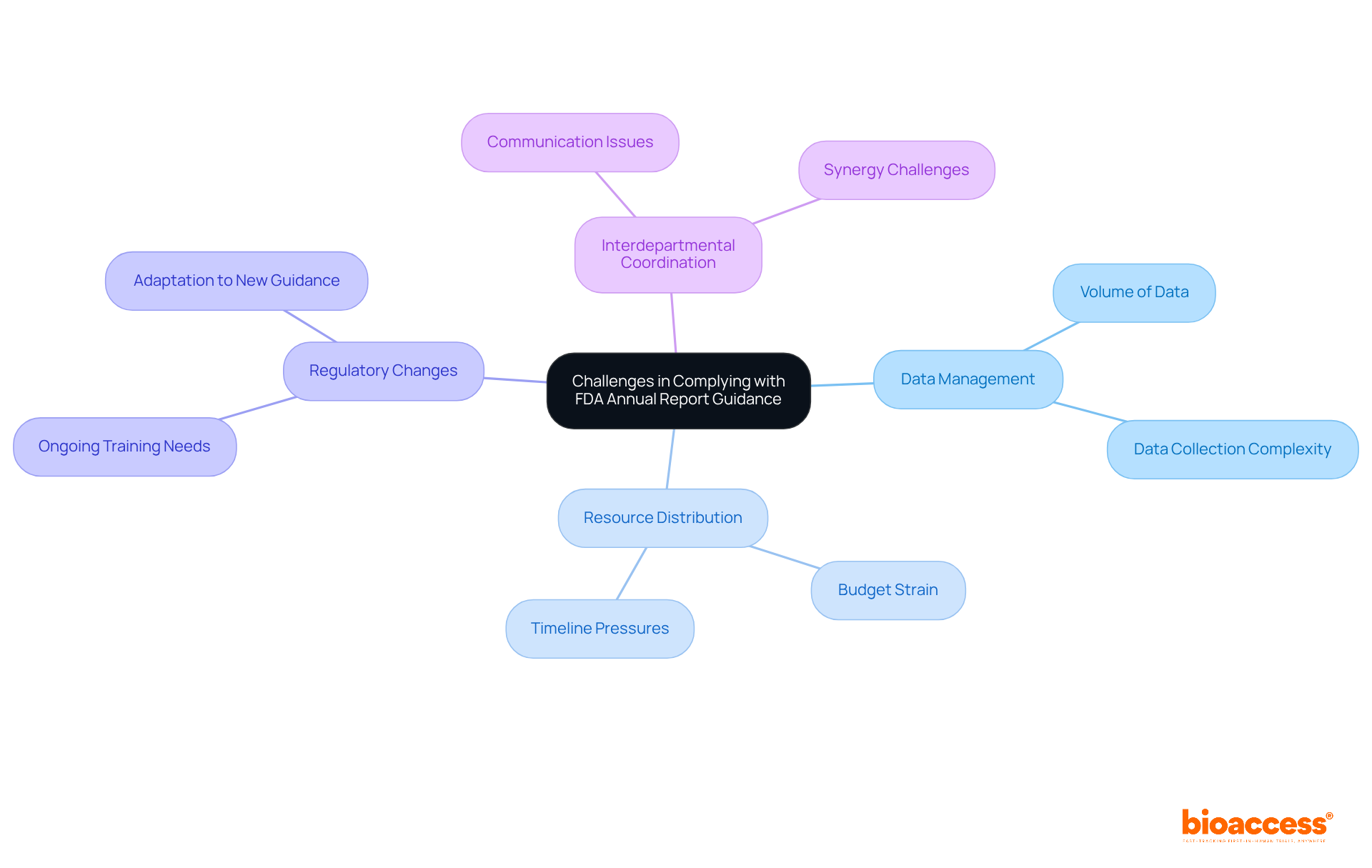
Challenges in Complying with FDA Annual Report Guidance
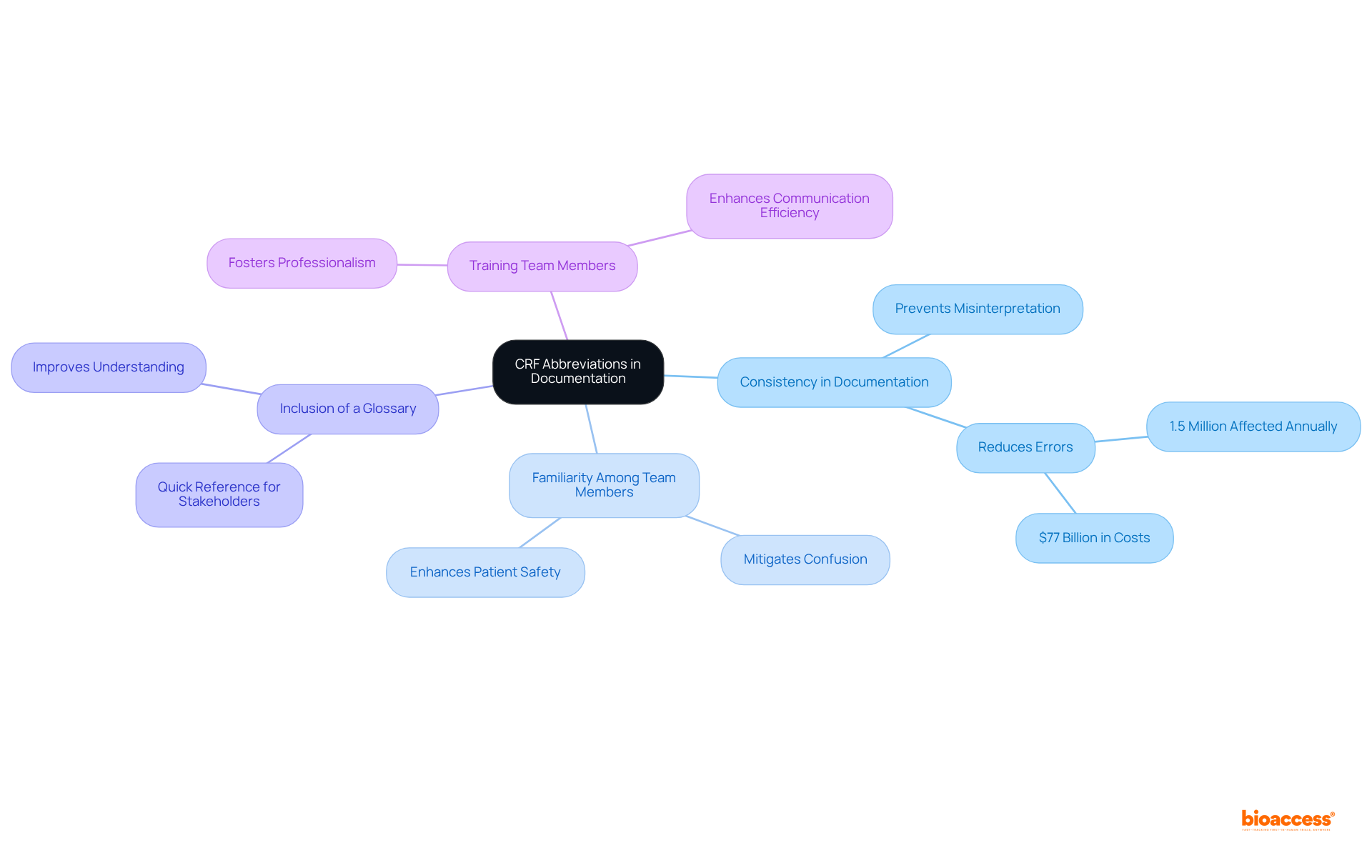
Organizations face significant challenges in adhering to the annual report guidance set by the FDA, particularly in data management. The sheer volume of data generated during larger clinical trials can be overwhelming, complicating collection and management processes. Additionally, is crucial; ensuring adequate resources for compliance can strain budgets and timelines, potentially jeopardizing overall project success.
present another obstacle, as the . This requires ongoing training and adaptation within organizations to maintain compliance. Moreover, effective interdepartmental coordination is essential. Communication between clinical, regulatory, and quality assurance teams is vital, yet achieving this synergy can be difficult.
To navigate these complexities, organizations must engage in and invest in a robust . Implementing tailored in line with can streamline processes, enhance data integrity, and ultimately facilitate adherence to regulatory requirements. By prioritizing these strategies, organizations can better position themselves for success in the evolving landscape of clinical research.
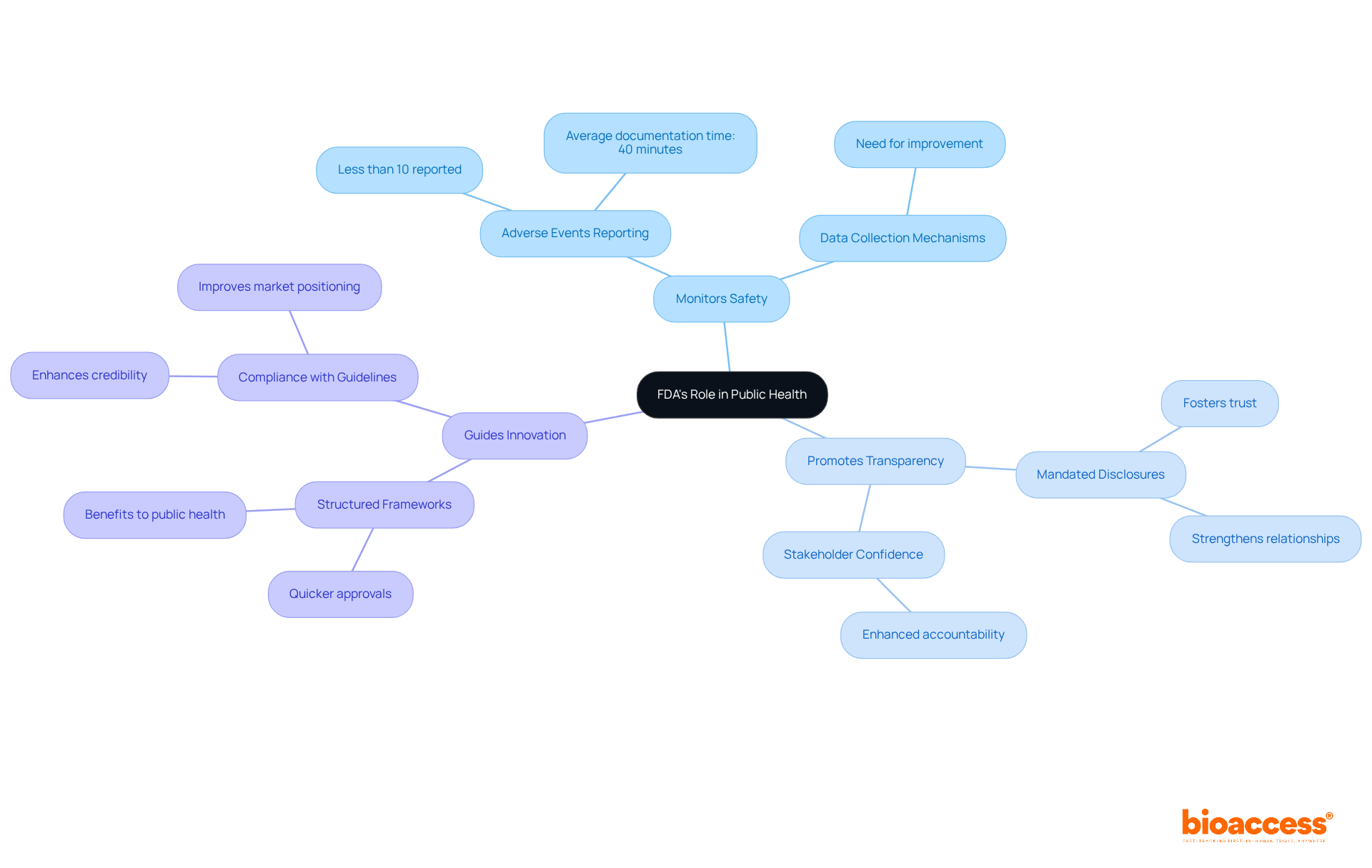
The Role of FDA in Public Health and Annual Reporting
The in safeguarding public well-being by ensuring the safety and effectiveness of medical products. This regulatory body is essential for maintaining trust in healthcare systems and .
- : The FDA systematically collects data on adverse events, which is vital for identifying potential safety issues. Alarmingly, statistics show that less than 10% of significant adverse events are reported by healthcare professionals. This highlights the urgent need for improved data collection mechanisms to enhance .
- : The FDA mandates that companies disclose extensive information about their products, fostering trust within the healthcare system. This transparency is crucial for and strengthening relationships with regulatory authorities.
- : By providing a structured framework for developing new therapies, the FDA prioritizes . Companies that adhere to these guidelines not only enhance their credibility but also position themselves for quicker approvals, ultimately benefiting public health outcomes.
This regulatory oversight is vital for sustaining public trust in medical goods and advancing public health efforts, as it cultivates a culture of adherence and responsibility within the sector.
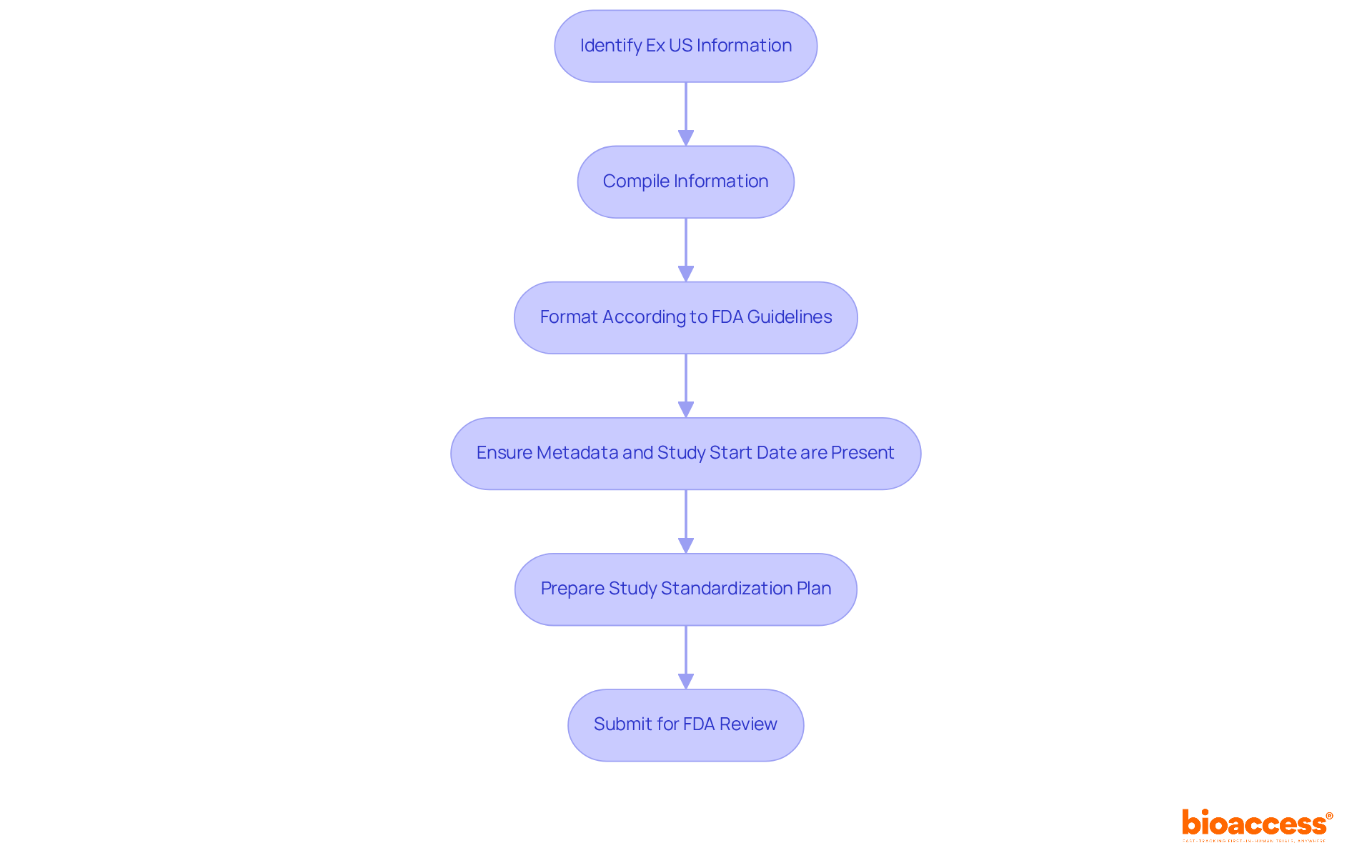
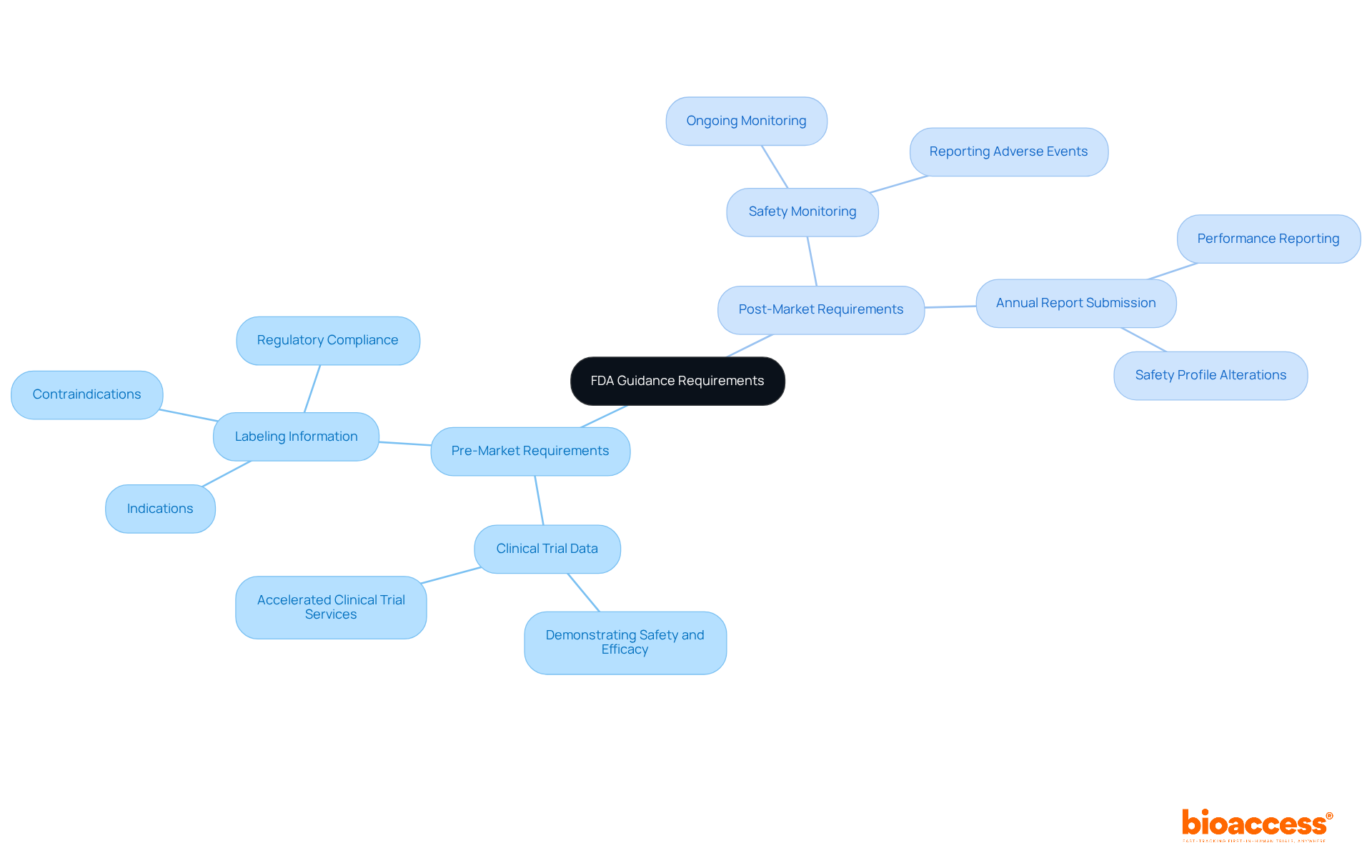
Pre-Market and Post-Market Information Requirements in FDA Guidance
outlines essential information requirements for both pre-market and post-market phases, which bioaccess® expertly facilitates for Medtech, Biopharma, and Radiopharma startups in Latin America and beyond.
Pre-Market Requirements:
- Submission of demonstrating safety and efficacy, supported by bioaccess®’s accelerated .
- Comprehensive labeling information, including indications and contraindications, ensuring compliance with regulatory standards.
Post-Market Requirements:
- Ongoing of adverse events, a critical aspect of bioaccess®’s commitment to maintaining .
- Submission of outlining the performance of items and any alterations in safety profile, facilitated by bioaccess® to ensure that items remain safe and effective throughout their lifecycle.
These requirements are crucial for ensuring that throughout their lifecycle. Bioaccess® is dedicated to helping clients navigate these complexities efficiently, reinforcing the importance of collaboration in clinical research.
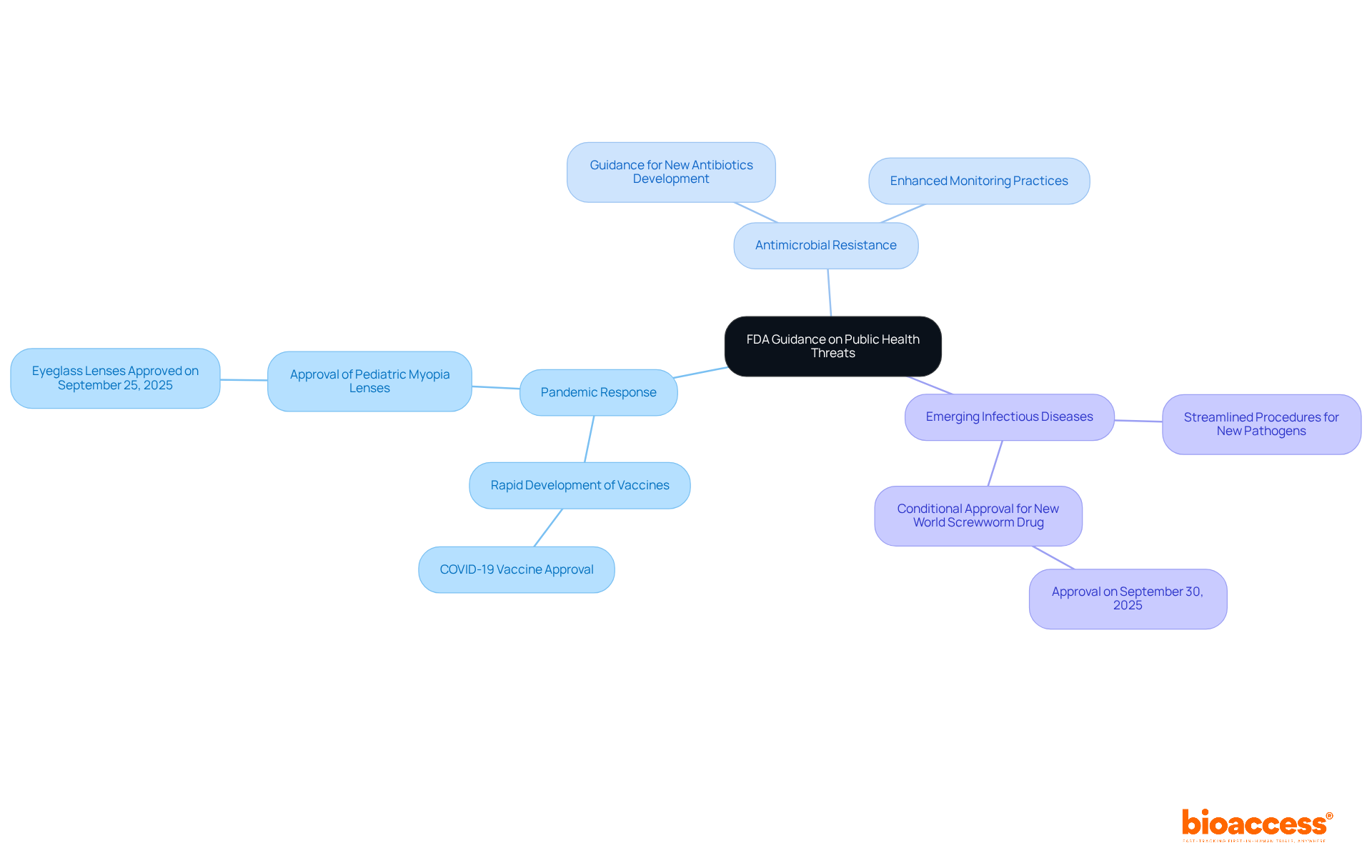
Addressing Emerging Public Health Threats through FDA Guidance
The FDA continually evolves its guidance to effectively tackle , focusing on several key areas:
- Pandemic Response: The agency has demonstrated remarkable agility in the rapid development and approval of vaccines and therapeutics during health crises, significantly shortening the time from research to market. For example, the rapid approval of COVID-19 vaccines illustrates this dedication to public well-being. Furthermore, on September 25, 2025, the of the first eyeglass lenses created to slow the advancement of pediatric myopia, highlighting its continuous efforts to tackle various medical issues.
- : In response to the growing concern of , the FDA has issued guidance aimed at fostering the development of new antibiotics and enhancing monitoring practices. This initiative is crucial for ensuring that effective treatments remain available as resistance patterns evolve.
- : The FDA has established streamlined procedures for items targeting newly identified pathogens, ensuring that innovative solutions can be quickly deployed in response to outbreaks. For instance, the conditional endorsement of the initial medication for the prevention and management of New World Screwworm infestations in cattle on September 30, 2025, emphasizes the particular wellness threats.
These proactive strategies highlight the , enabling timely access to essential medical products and strengthening its role as a vital participant in protecting well-being during crises. As FDA Commissioner Marty Makary stated on September 25, 2025, “Encouraging retailers to stop selling illegal vapes” reflects the agency’s commitment to addressing emerging public welfare threats comprehensively. Moreover, the a demonstrates its involvement with important public welfare issues. Overall, these measures ensure that the FDA remains attentive to public health requirements, facilitating timely access to essential medical items.
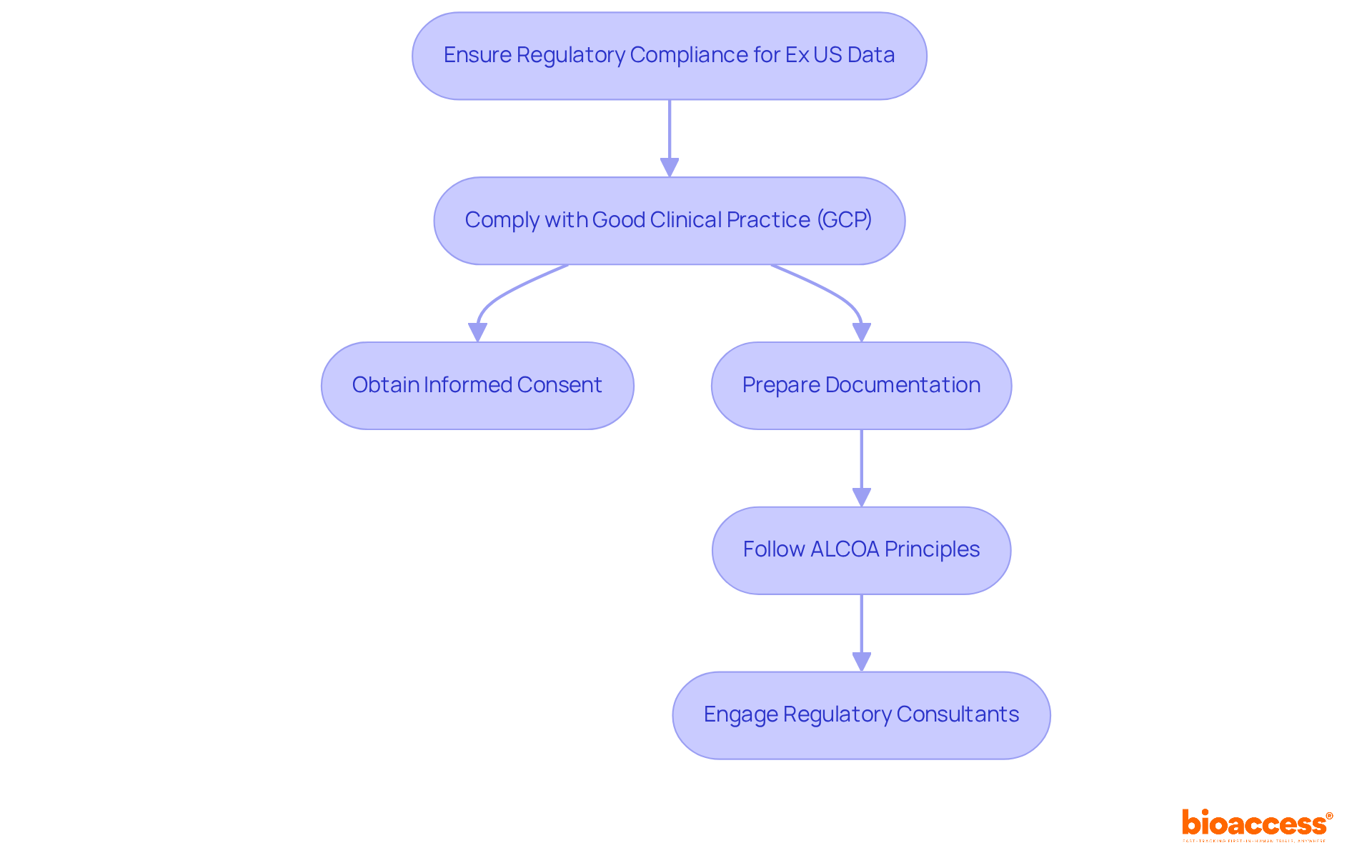
Importance of Ethical Approvals in Accelerating Clinical Trials
are essential for accelerating . They protect participants by ensuring that the rights and welfare of are prioritized. Furthermore, they enhance credibility; ethical oversight builds trust with stakeholders and the public, facilitating smoother trial operations. Additionally, they streamline processes; efficient ethical review processes can significantly reduce timelines for trial initiation. By prioritizing ethical considerations, organizations can enhance the overall quality and acceptance of their .
: protecting participants ensures their rights are upheld; enhancing credibility builds trust with stakeholders; and streamlining processes helps speed up trial initiation. Each slice of the pie represents a key benefit of ethical approvals: protecting participants ensures their rights are upheld; enhancing credibility builds trust with stakeholders; and streamlining processes helps speed up trial initiation.](https://images.tely.ai/telyai/qhnwszrr-each-slice-of-the-pie-represents-a-key-benefit-of-ethical-approvals-protecting-participants-ensures-their-rights-are-upheld-enhancing-credibility-builds-trust-with-stakeholders-and-streamlining-processes-helps-speed-up-trial-initiation.webp)
Understanding FDA Guidance for Successful Product Commercialization
Successful product commercialization hinges on a thorough understanding of FDA guidance, which includes several critical elements:
- : Identifying the correct regulatory pathway is essential for Medtech and Biopharma products. In 2025, navigating these pathways efficiently can significantly impact the speed and success of approval.
- : Crafting that align with FDA requirements is vital for facilitating market entry. Research shows that 57% of drug launch failures arise from limited market access, highlighting the necessity for strategic alignment.
- : Establishing robust post-market monitoring systems is crucial to ensure ongoing compliance and safety. This proactive approach not only meets regulatory expectations but also fosters trust with stakeholders.
By aligning commercialization strategies with the , companies can greatly enhance their chances of success in a competitive marketplace. Effective , especially with payers, should commence 18-24 months before product launch to refine value messaging and improve . This strategic foresight is essential, as 34% of drugs launched between 2012 and 2021 , often due to a lack of understanding of market and customer needs. Therefore, a rigorous approach to developing and evolving is paramount for achieving sustainable success.
, [market access strategies](https://deloitte.com/us/en/insights/industry/life-sciences/pharmaceutical-market-access.html), and [post-market surveillance](https://deloitte.com/us/en/insights/industry/life-sciences/pharmaceutical-market-access.html). Each sub-point provides additional insights and statistics to enhance understanding of these areas. At the center, you'll find the main theme of FDA guidance. The branches explore key areas crucial for product success: regulatory pathways, market access strategies, and post-market surveillance. Each sub-point provides additional insights and statistics to enhance understanding of these areas.](https://images.tely.ai/telyai/tknaivaw-at-the-center-youll-find-the-main-theme-of-fda-guidance-the-branches-explore-key-areas-crucial-for-product-success-regulatory-pathways-market-access-strategies-and-post-market-surveillance-each-sub-point-provides-additional-insights-and-statistics-to-enhance-understanding-of-these-areas.webp)
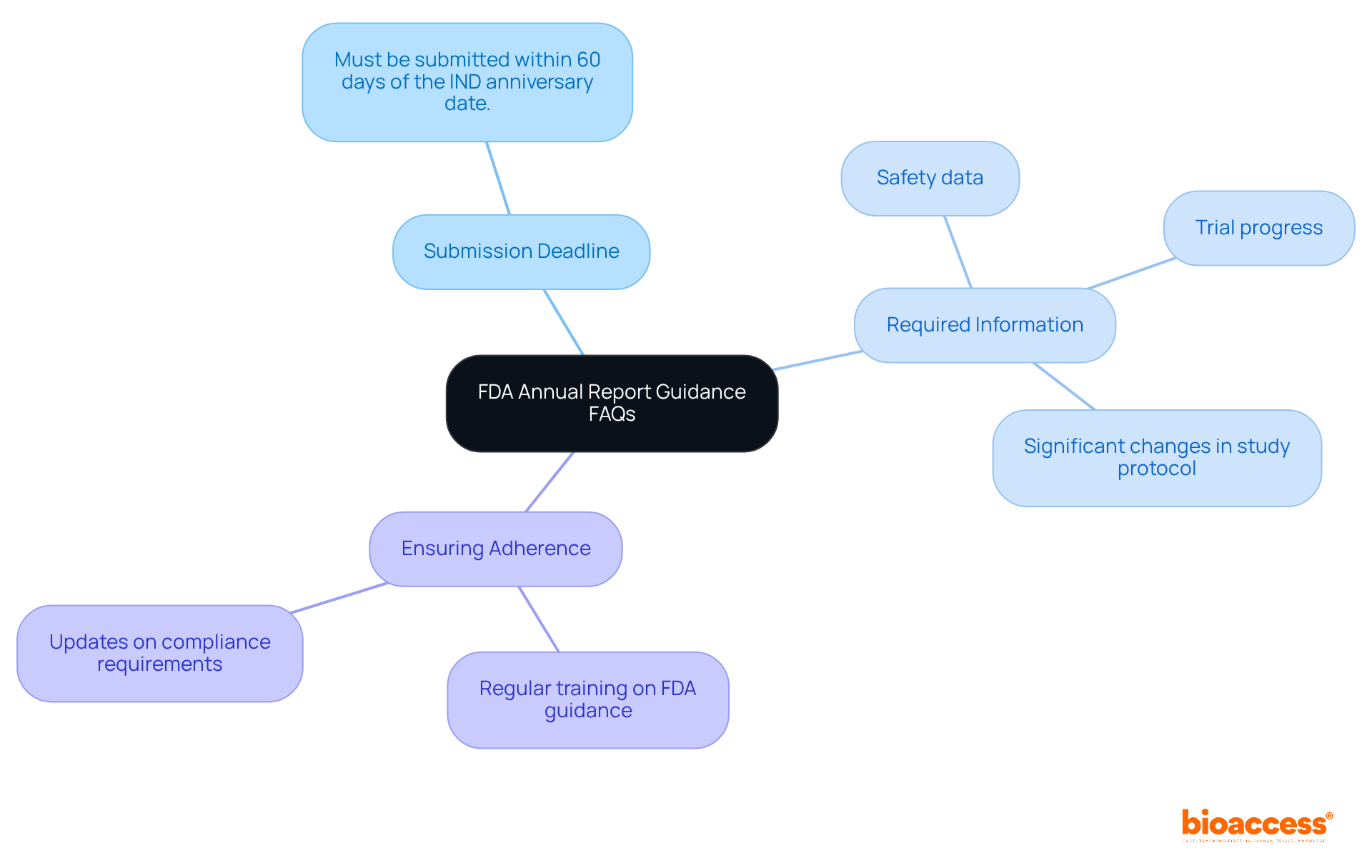
Frequently Asked Questions about FDA Annual Report Guidance
Common questions regarding FDA include:
- What is the deadline for submission? Annual reports must be submitted within 60 days of the anniversary date of the IND.
- What information is required? Reports should include , , and any .
- How can organizations ensure adherence? Regular training and updates on can help maintain compliance.
By addressing these FAQs, organizations can better navigate the complexities of .
Conclusion
The annual report guidance from the FDA is essential for organizations striving to ensure compliance and enhance communication within the clinical research landscape. By following these guidelines, companies not only uphold regulatory integrity but also play a vital role in improving public health outcomes through transparency and accountability in their clinical trials.
Key insights from the guidance underscore the necessity of comprehensive documentation, including:
- Summaries of clinical trials
- Safety reports
- Thorough statistical analyses
These components are crucial for navigating the intricate regulatory environment, speeding up product approvals, and fostering innovation in the Medtech and Biopharma sectors. As organizations grapple with challenges in data management and regulatory compliance, strategic planning and interdepartmental coordination become indispensable for success.
Ultimately, the FDA’s guidance acts as a pivotal framework that safeguards public health while driving innovation in medical products. By adopting these guidelines, companies can boost their operational efficiencies and ensure timely access to life-saving therapies. Collaborating with experienced partners, such as bioaccess®, can further streamline compliance and accelerate the journey from research to market, reinforcing the essential role of collaboration in advancing healthcare solutions.
Frequently Asked Questions
What is bioaccess® and what services does it provide?
bioaccess® is a clinical research organization (CRO) that specializes in navigating FDA regulations to streamline clinical research processes. Its services include feasibility assessments, investigator selection, and meticulous project management, all aimed at enhancing the integrity of clinical trials.
How does bioaccess® help accelerate clinical research?
By ensuring strict adherence to FDA guidelines, bioaccess® accelerates the approval and enrollment stages of clinical research, allowing Medtech and Biopharma innovators to bring their products to market more swiftly.
Why is regulatory compliance important in clinical research?
Regulatory compliance is crucial for fostering trust with stakeholders and regulatory bodies, maintaining the integrity of clinical trials, and facilitating timely approvals, which ultimately enhances public health outcomes.
What are the key components of the FDA annual report guidance for 2024?
The key components include a summary of clinical trials, safety reports detailing adverse events, robust statistical analysis to support efficacy and safety claims, and comprehensive documentation of regulatory compliance throughout the trial process.
How does the annual report guidance impact the Medtech and Biopharma industries?
The guidance leads to increased transparency, faster product approvals, and a pressure for innovation while ensuring adherence to regulations, which affects development timelines and market strategies.
What types of studies does bioaccess® manage?
bioaccess® manages a range of studies including Early-Feasibility Studies (EFS), First-In-Human Studies (FIH), Pilot Studies, Pivotal Studies, and Post-Market Clinical Follow-Up Studies (PMCF).
How can partnering with bioaccess® benefit clinical trial management?
Partnering with bioaccess® provides access to over 20 years of experience in Medtech, along with customized methods, adaptability, and extensive services to efficiently manage the complexities of FDA guidance in clinical trials.
List of Sources
- Key Components of the FDA Annual Report Guidance for 2024
- bioaccessla.com (https://bioaccessla.com/blog/understanding-fda-annual-report-guidance-for-2024-key-insights)
- Implications of FDA Guidance for Medtech and Biopharma Industries
- iqvia.com (https://iqvia.com/insights/the-iqvia-institute/reports-and-publications/reports/global-trends-in-r-and-d-2025)
- iqvia.com (https://iqvia.com/locations/united-states/blogs/2025/04/a-new-fda-preparing-uncertain-landscape)
- Factors Influencing Clearance Time for Medical Devices (https://mddionline.com/medical-device-regulations/factors-influencing-fda-clearance-time-for-medical-devices-evolution-of-a-critical-regulatory-pathway)
- Challenges in Complying with FDA Annual Report Guidance
- reuters.com (https://reuters.com/business/healthcare-pharmaceuticals/us-fda-sends-warning-letter-philips-over-three-device-production-facilities-2025-10-28)
- The Role of FDA in Public Health and Annual Reporting
- bioaccessla.com (https://bioaccessla.com/blog/understanding-fda-annual-report-guidance-for-2024-key-insights)
- Addressing Emerging Public Health Threats through FDA Guidance
- Press Announcements (https://fda.gov/news-events/fda-newsroom/press-announcements)
- Understanding FDA Guidance for Successful Product Commercialization
- deloitte.com (https://deloitte.com/us/en/insights/industry/life-sciences/pharmaceutical-market-access.html)


 with different educational qualifications. A larger slice indicates a higher number of CRAs with that degree. Each slice represents the percentage of Clinical Research Associates with different educational qualifications. A larger slice indicates a higher number of CRAs with that degree.](https://images.tely.ai/telyai/ckymahul-each-slice-represents-the-percentage-of-clinical-research-associates-with-different-educational-qualifications-a-larger-slice-indicates-a-higher-number-of-cr-as-with-that-degree.webp)