Introduction
In the rapidly evolving landscape of clinical research, the design of electronic case report forms (eCRFs) is crucial for ensuring data integrity and regulatory compliance. Effective strategies can significantly enhance the quality and efficiency of eCRFs, ultimately leading to faster and more reliable clinical trials. However, a pressing challenge persists: how can stakeholders balance the need for customization with the necessity of adhering to stringent industry standards? This article delves into ten innovative strategies that not only streamline eCRF design but also empower researchers to confidently navigate the complexities of modern clinical studies.
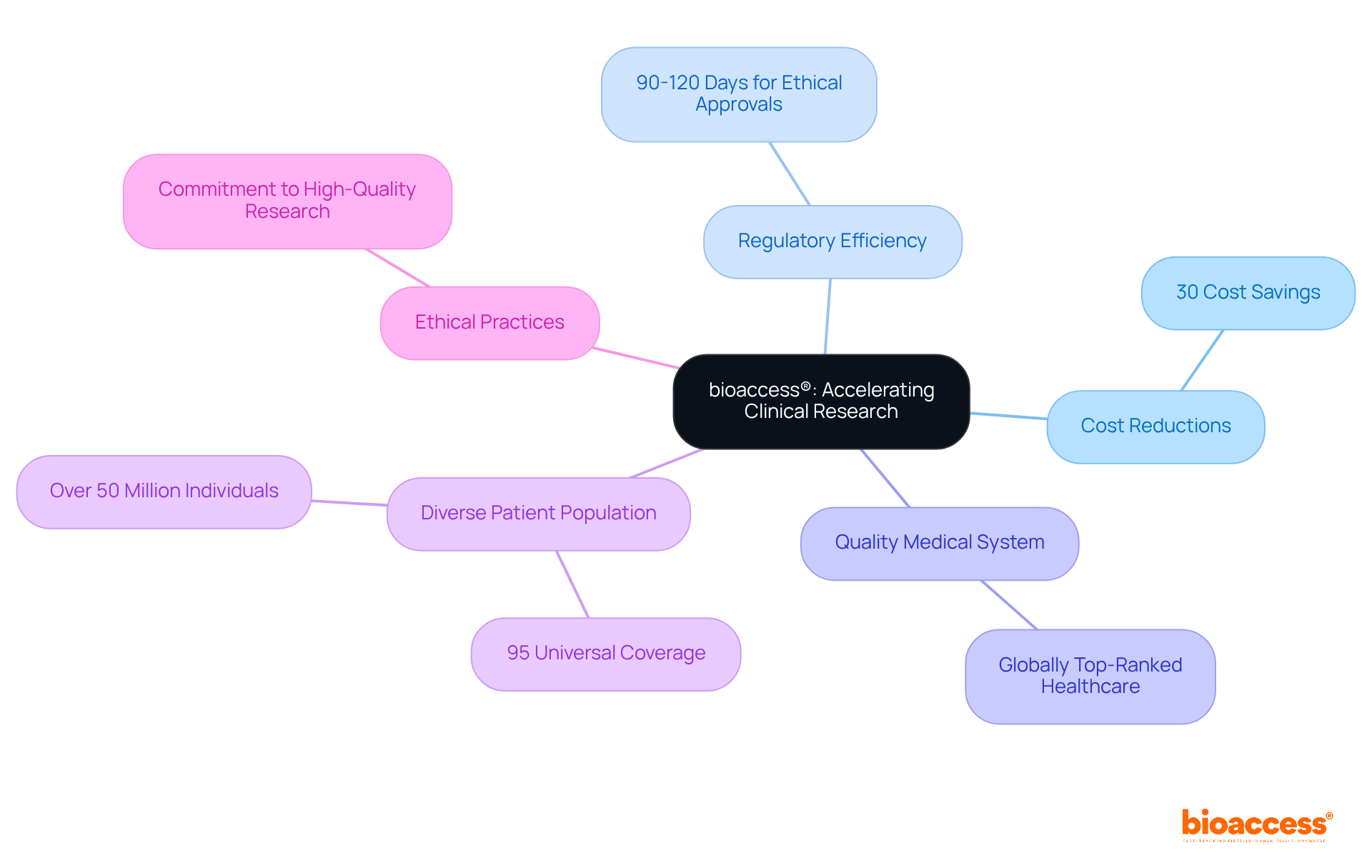
bioaccess®: Accelerate eCRF Design with Expert Clinical Research Services
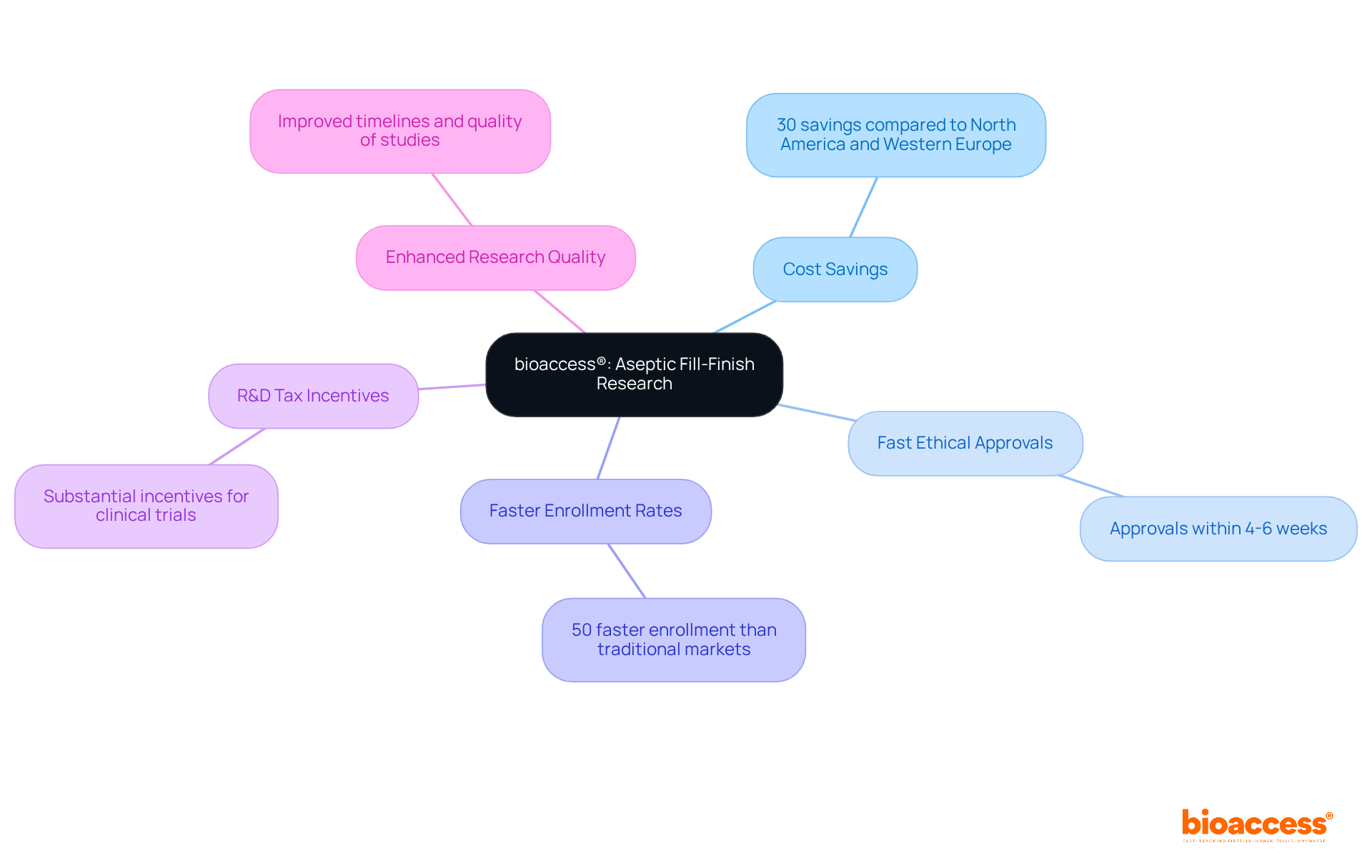
bioaccess® leverages its extensive research expertise to accelerate the development of . With a pioneering approach to medical flexibility, bioaccess® ensures that these forms are while being customized to meet the unique requirements of innovators in Medtech, Biopharma, and Radiopharma. This strategic approach not only expedites the approval process but also streamlines information collection, significantly enhancing the quality of .
Research indicates that , enabling faster market entry for new therapies. Furthermore, the implementation of has been shown to , with error rates dropping below 5%, thereby fostering trust among stakeholders and participants.
By emphasizing compliance and efficiency, bioaccess® establishes itself as a leader in the successful implementation of , ultimately driving superior outcomes in .
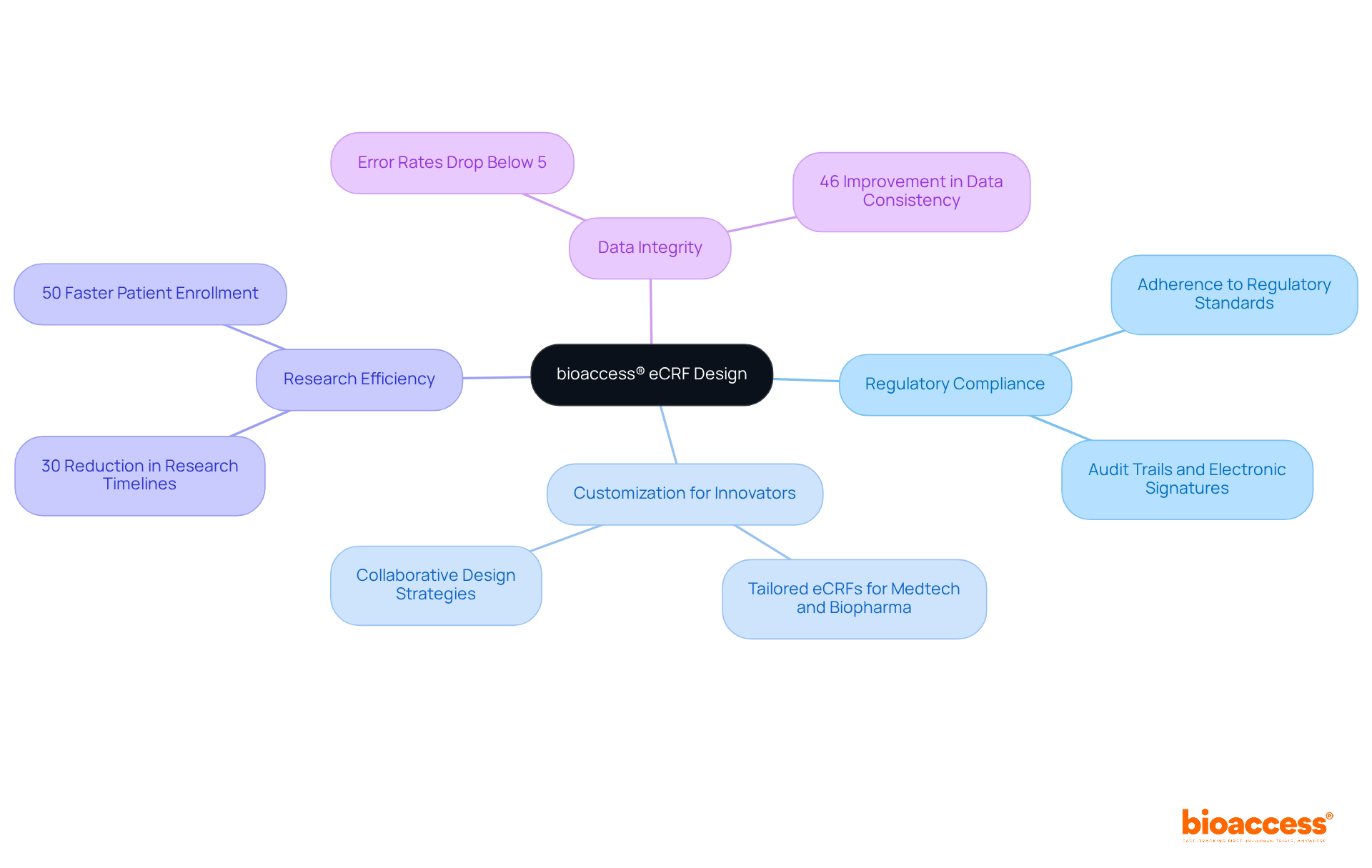
Maximize Semantic Interoperability in eCRF Design
To enhance , it is essential that (eCR) designs adhere to established , particularly those set forth by the . This adherence not only guarantees that the information collected is readily shareable but also ensures its comprehensibility across various platforms and stakeholders.
By utilizing standardized terminologies and formats, researchers can streamline the processes of and analysis, ultimately leading to improved . Moreover, efficient significantly reduce the likelihood of errors in , underscoring the critical need for the application of .
Furthermore, the synergistic application of both semantic and syntactic standards bolsters interoperability, making it a crucial component in the development of effective .
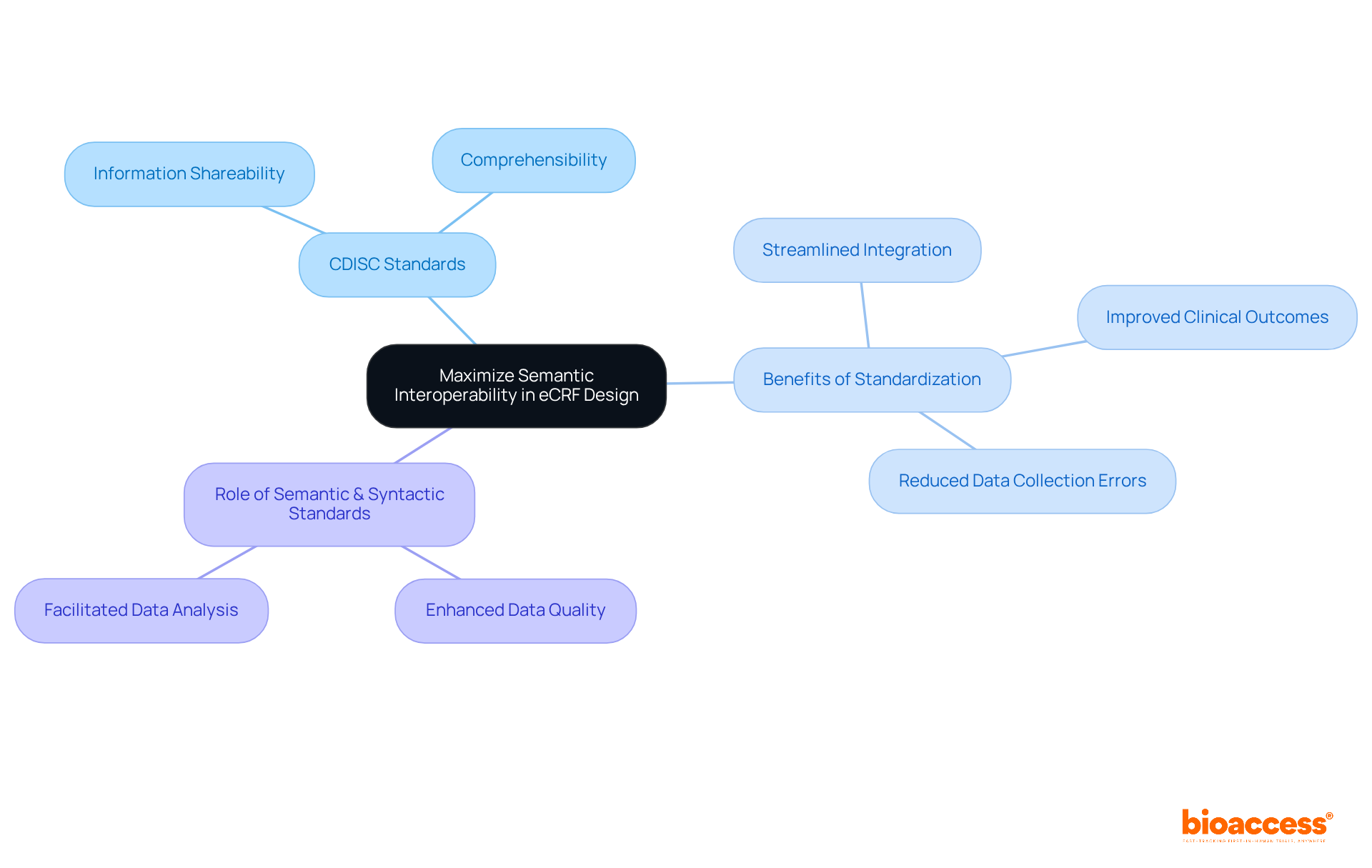
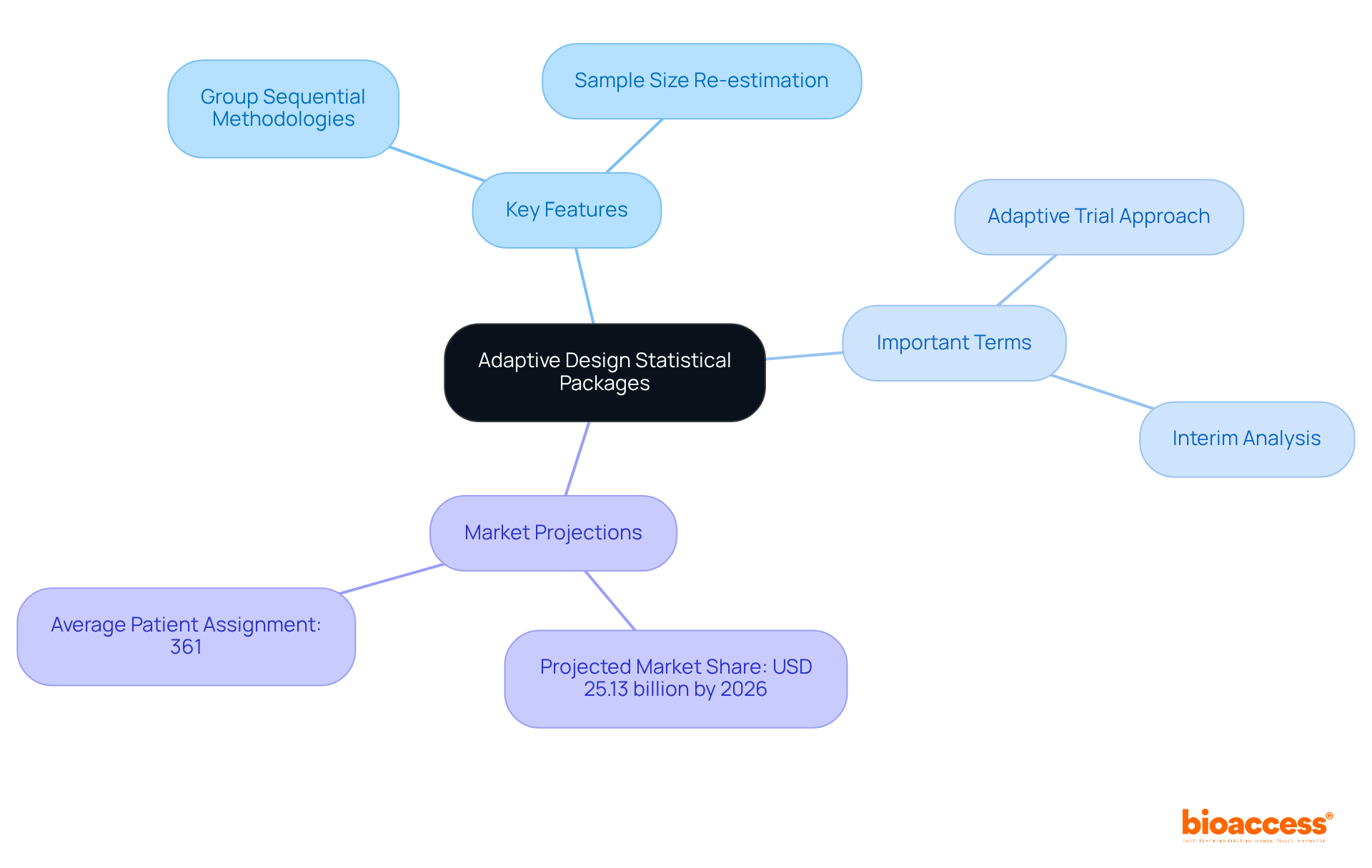
Utilize Core Data Elements (CDEs) for Standardized Data Collection
Integrating into the design of is essential for achieving uniform information collection across . CDEs are predefined information fields that ensure , facilitating effective comparison and analysis. Research indicates that utilizing can significantly enhance the reliability of findings.
For instance, studies have shown that can reduce entry mistakes to 0%, compared to a 5% error rate associated with traditional paper forms. By leveraging CDEs, researchers not only bolster the integrity of their data but also streamline the , ultimately leading to quicker and more efficient trials.
This approach aligns with the growing emphasis on , where over 500 organizations have adopted CDISC standards to . As highlighted by industry experts, standardizing information collection is vital for improving and ensuring that innovative therapies reach patients more swiftly.
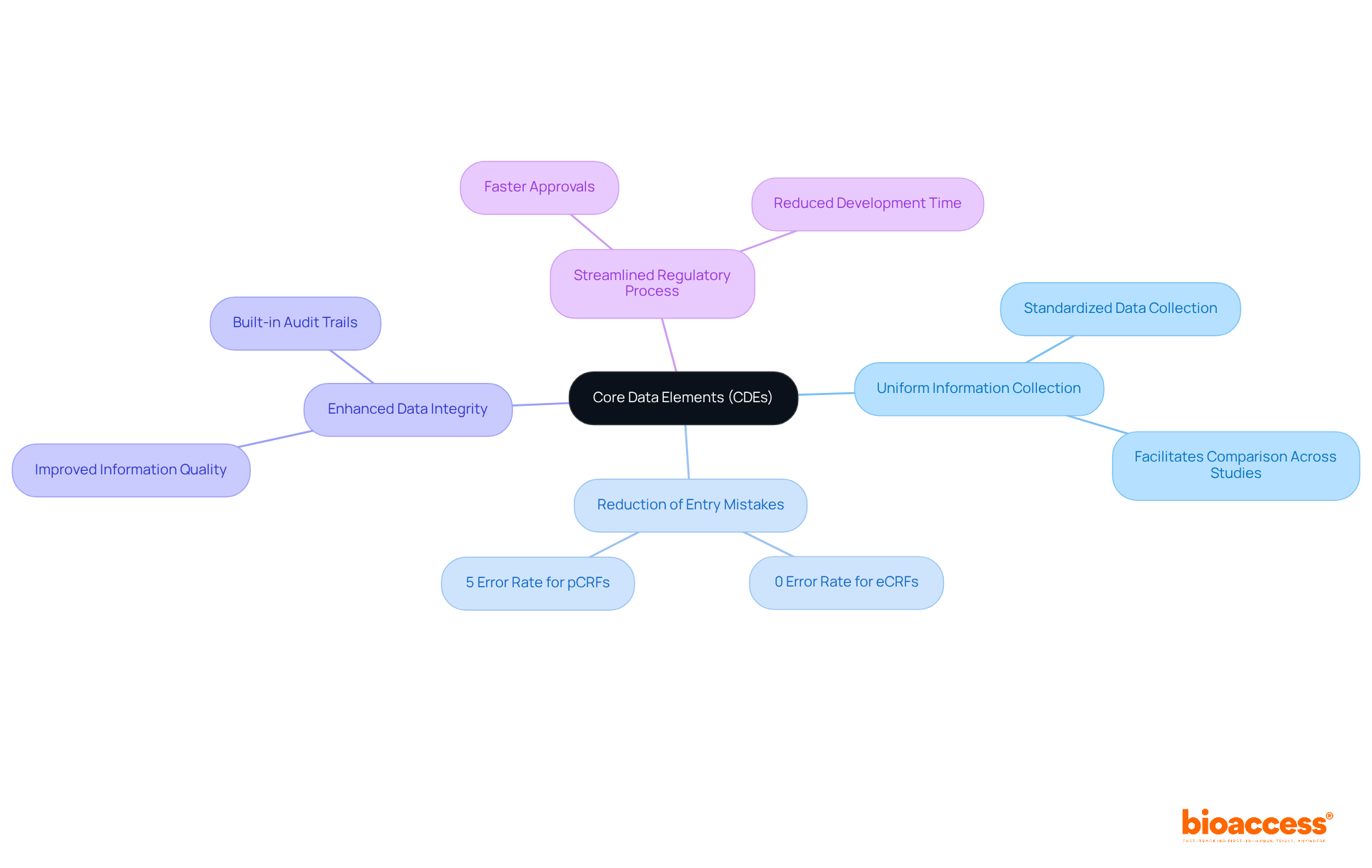
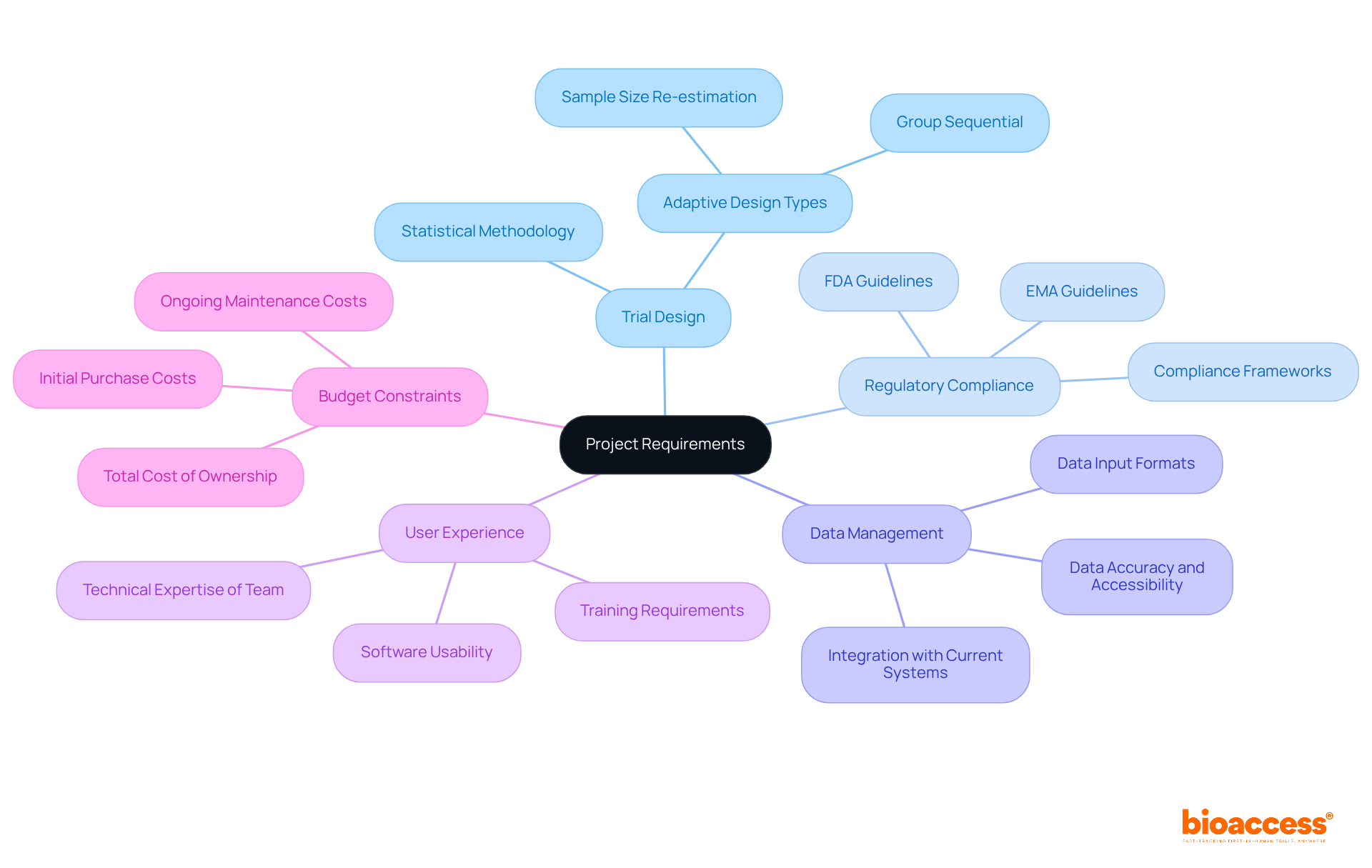
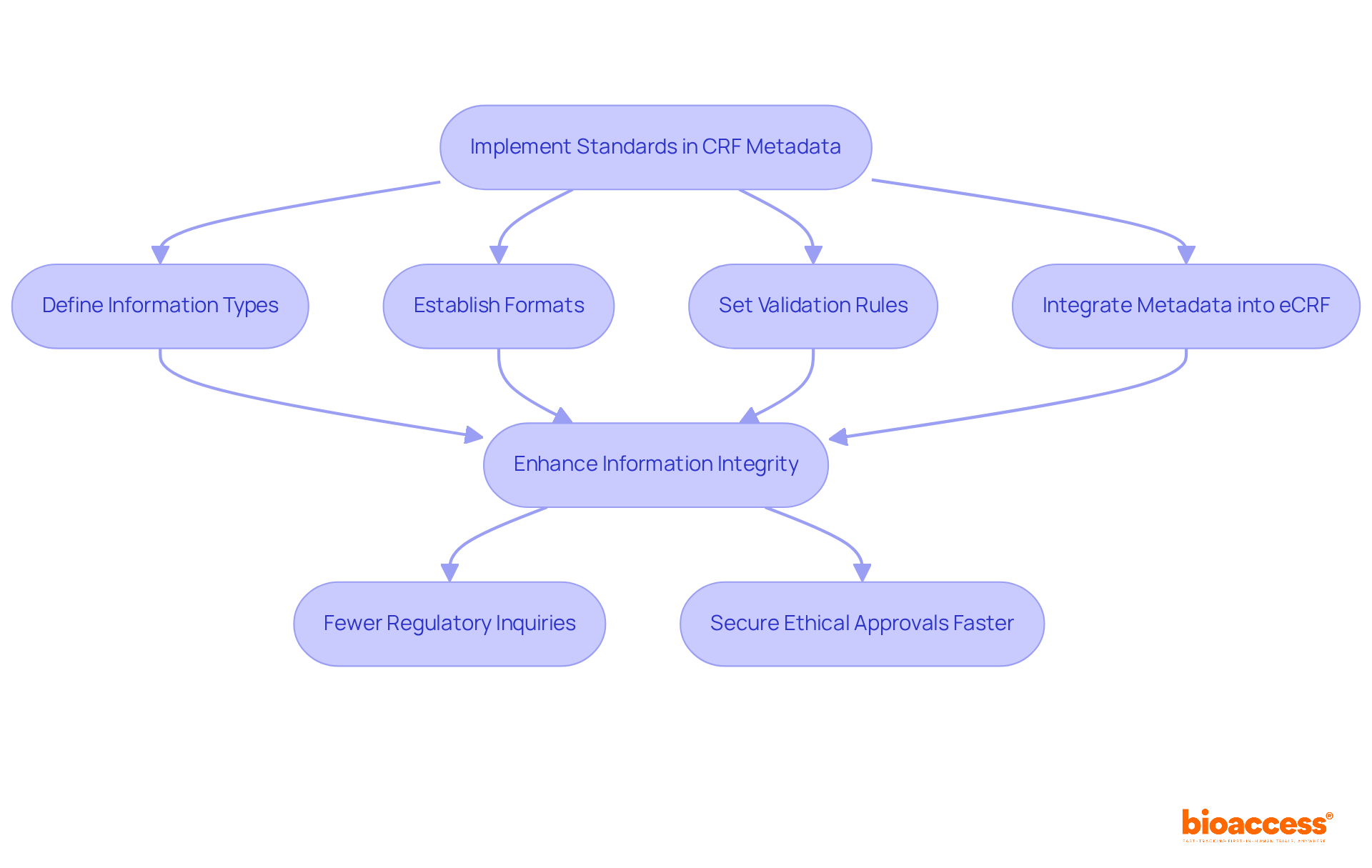
Implement Standards in CRF Metadata for Compliance
Establishing rigorous standards for is crucial for adhering to . This necessitates a clear definition of information types, formats, and validation rules that comply with industry standards. By integrating robust metadata into the electronic case report structure, researchers can significantly enhance information integrity, which is essential for ensuring compliance and facilitating smoother audits and assessments by regulatory authorities.
Trials that impose stringent metadata standards can experience up to 30% fewer during audits, underscoring the importance of meticulous information management. Furthermore, aligning the with metadata standards not only simplifies the but also elevates overall data quality, ultimately leading to more effective trials.
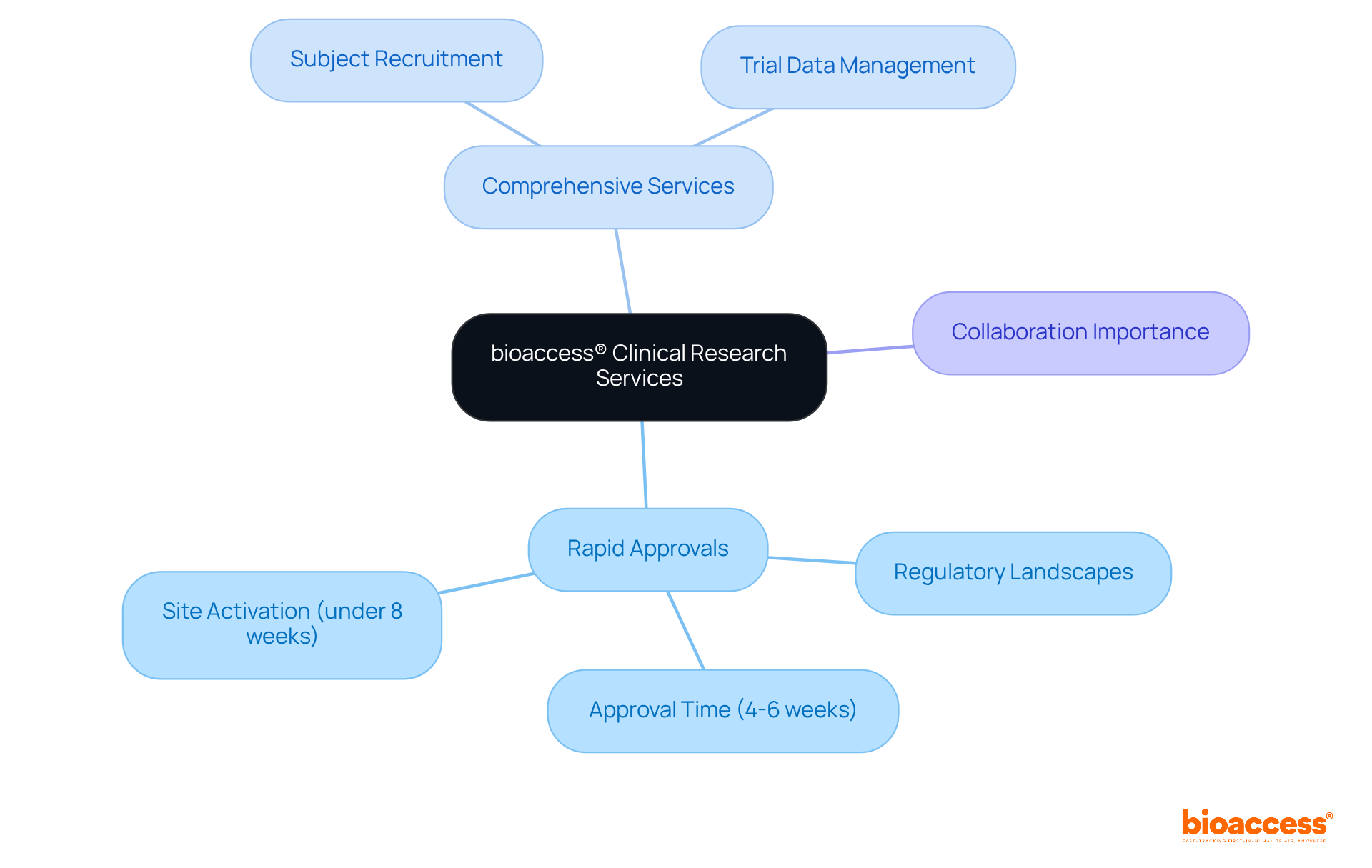
Notably, organizations can secure ethical approvals in as little as 4-6 weeks with , while the average duration of recruitment with electronic case reports is 22.4 ± 9 months, demonstrating their efficiency compared to traditional methods. Additionally, bioaccess offers extensive , including:
- Site selection
- Compliance reviews
- Trial setup
- Import permits
- Project management
- Reporting
These services are essential for ensuring that these standards are met. As Qualityze aptly states, ‘The electronic case report in clinical trials is not merely a collection tool—it’s an essential enabler of high-quality, audit-ready, and efficient studies.
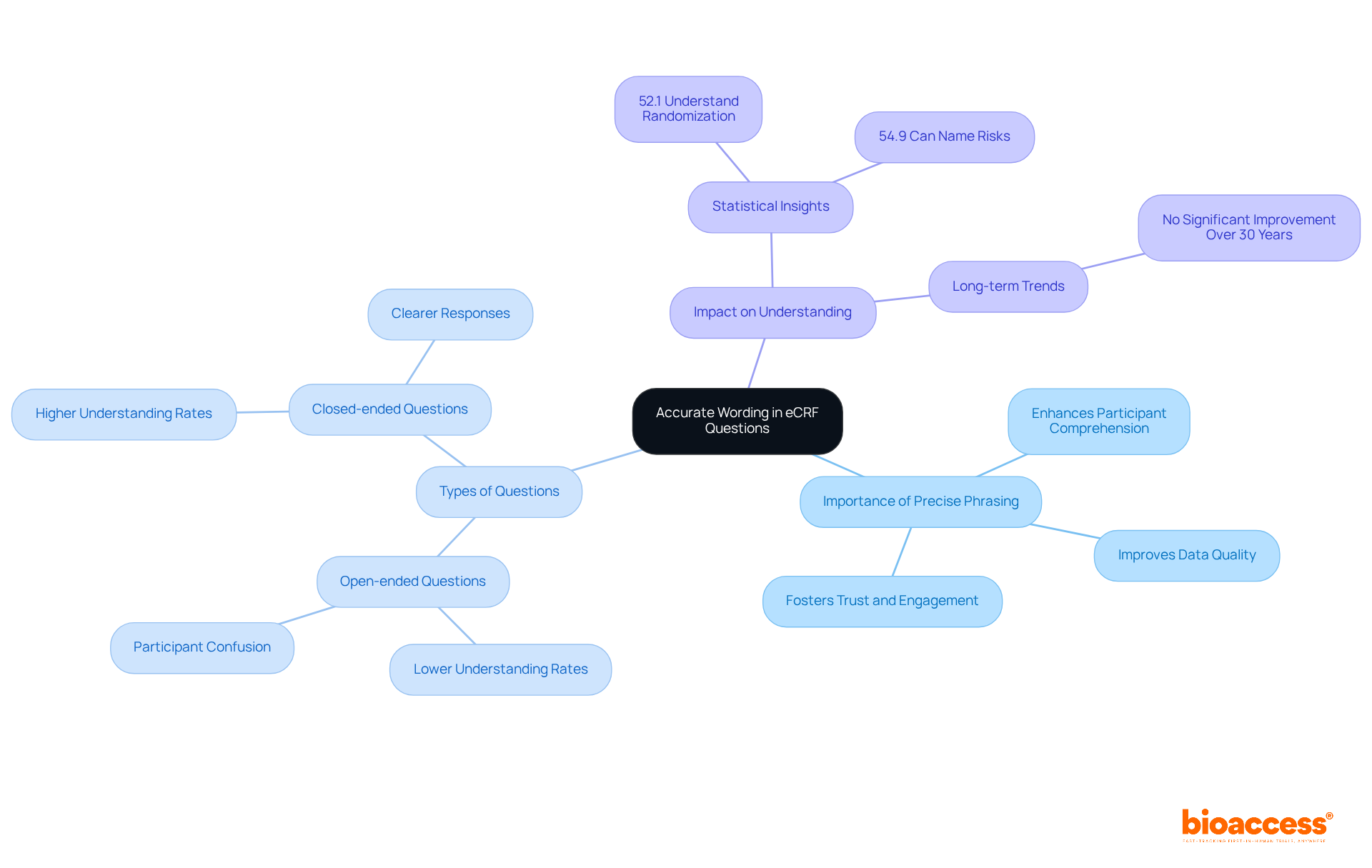
Ensure Accurate Wording in eCRF Questions
Precise phrasing in the is crucial for gathering . Questions must be clear, concise, and free of jargon to avoid participant confusion. Research shows that the wording of questions significantly impacts understanding; for example, only 52.1% of , and those evaluated with open-ended questions often struggle to comprehend key elements of . Conversely, the use of closed-ended questions correlates with higher rates of understanding regarding the study’s purpose.
By employing , researchers can enhance , leading to information that more accurately reflects their responses. This approach not only improves the quality of the data collected in the but also fosters trust and engagement among participants, ultimately bolstering the .
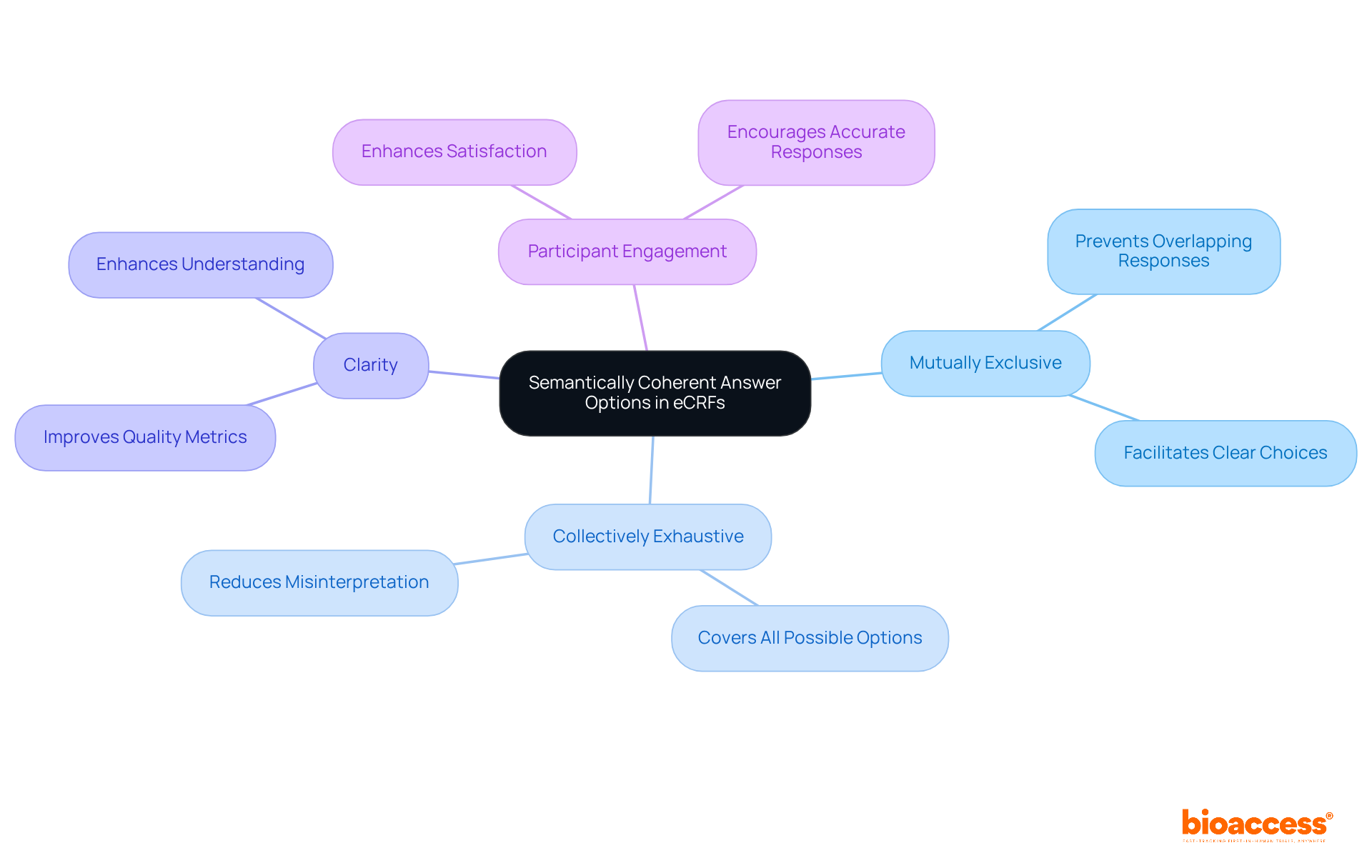
Provide Semantically Coherent Answer Options in eCRFs
Ensuring semantically coherent answer options in the is paramount for obtaining accurate . Answer choices must be mutually exclusive and collectively exhaustive, empowering participants to select the most relevant option. This clarity not only but also significantly boosts participant engagement and satisfaction throughout the study.
Research indicates that can lead to a substantial improvement in , as participants are less likely to misinterpret questions. Furthermore, experts underscore that cultivates a better understanding of the survey, ultimately resulting in more reliable .
For instance, the ORCHESTRA project analyzed over 3,700 variables to identify similar information across studies, highlighting the critical importance of . By prioritizing clarity in answer design, researchers can create a more engaging experience for participants, which is essential for the success of .
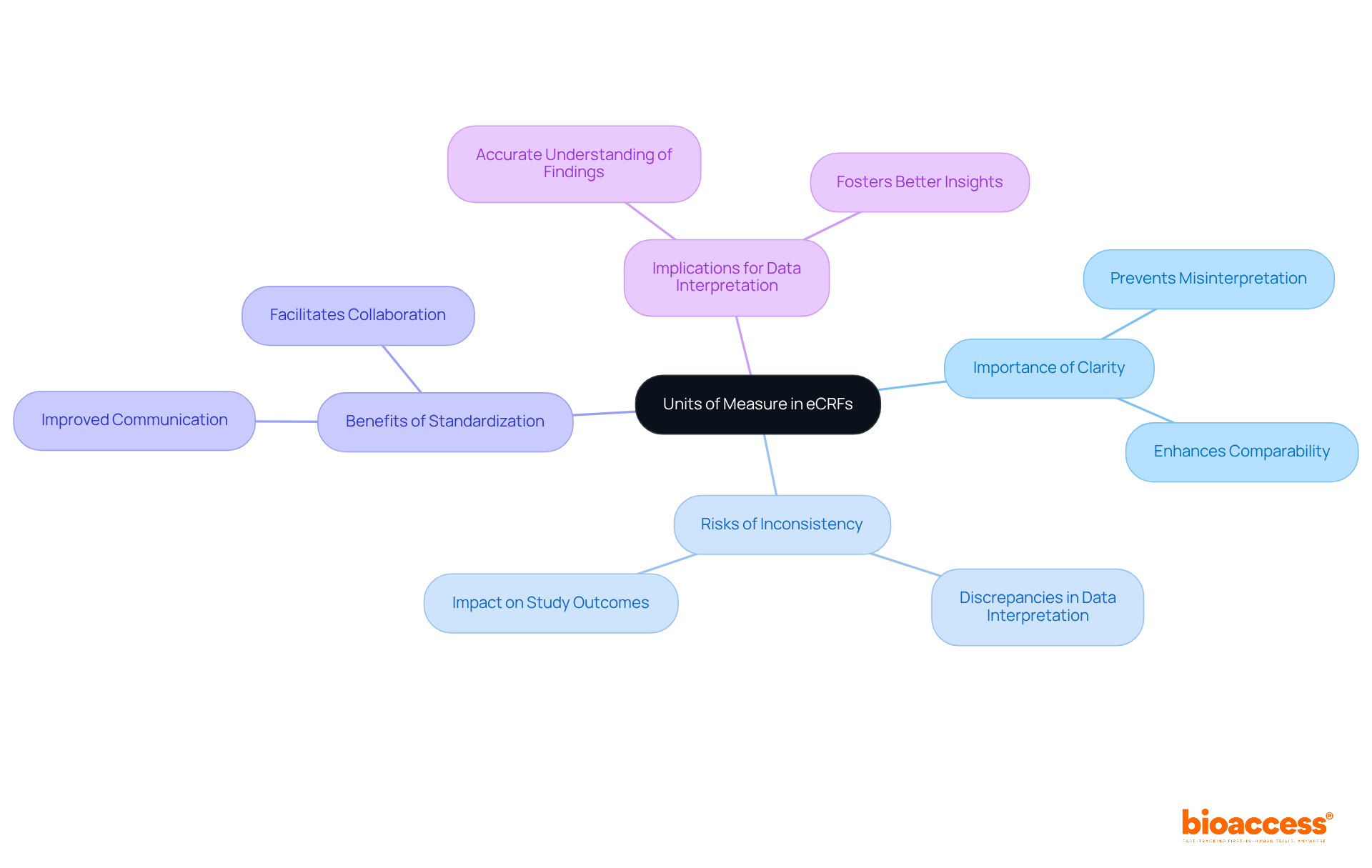
Clearly State Units of Measure in eCRFs
In (eCRFs), specifying the is essential. This includes clarifying whether measurements are in and providing any necessary conversion factors. Clear definitions of units not only prevent misinterpretation of information but also enhance the across various studies.
For instance, in a , using inconsistent units can lead to significant discrepancies in , ultimately affecting study outcomes. By adhering to , researchers can ensure that their findings are accurately understood and effectively communicated, fostering better collaboration and insights within the scientific community.
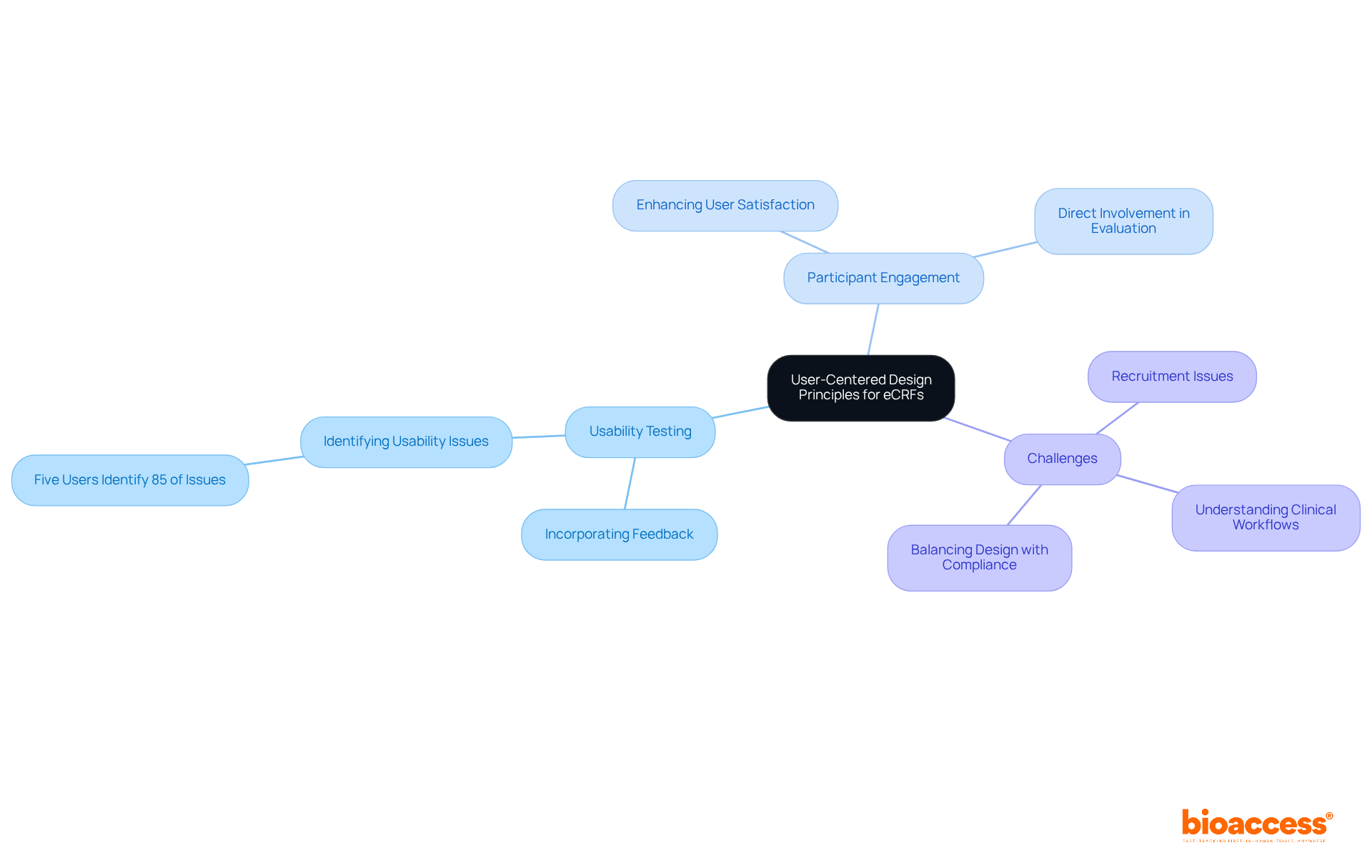
Adopt User-Centered Design Principles for eCRFs
Applying principles in the creation of electronic case reports is essential for enhancing usability in . This approach emphasizes the importance of understanding the needs and preferences of both participants and researchers. By conducting thorough and actively incorporating feedback, eCRFs can be designed to be . Such enhancements not only simplify information gathering but also significantly improve the , fostering greater engagement and satisfaction.
Usability specialists assert that careful planning can yield more , ultimately enhancing data quality and outcomes in . As highlighted by Don Norman, shortcomings in human-machine interaction frequently stem from a lack of understanding of fundamental principles, underscoring the significance of user-centered approaches in developing . Moreover, research indicates that five users can identify 85% of during ongoing testing, illustrating the critical role of in refining the electronic case report (eCRF) structure.
However, it is important to acknowledge the challenges associated with implementing user-centered approaches, such as conducting detailed studies of clinical workflows and recruiting participants for usability studies. Recognizing these challenges provides a balanced perspective on the topic and emphasizes the need for continued focus on in .
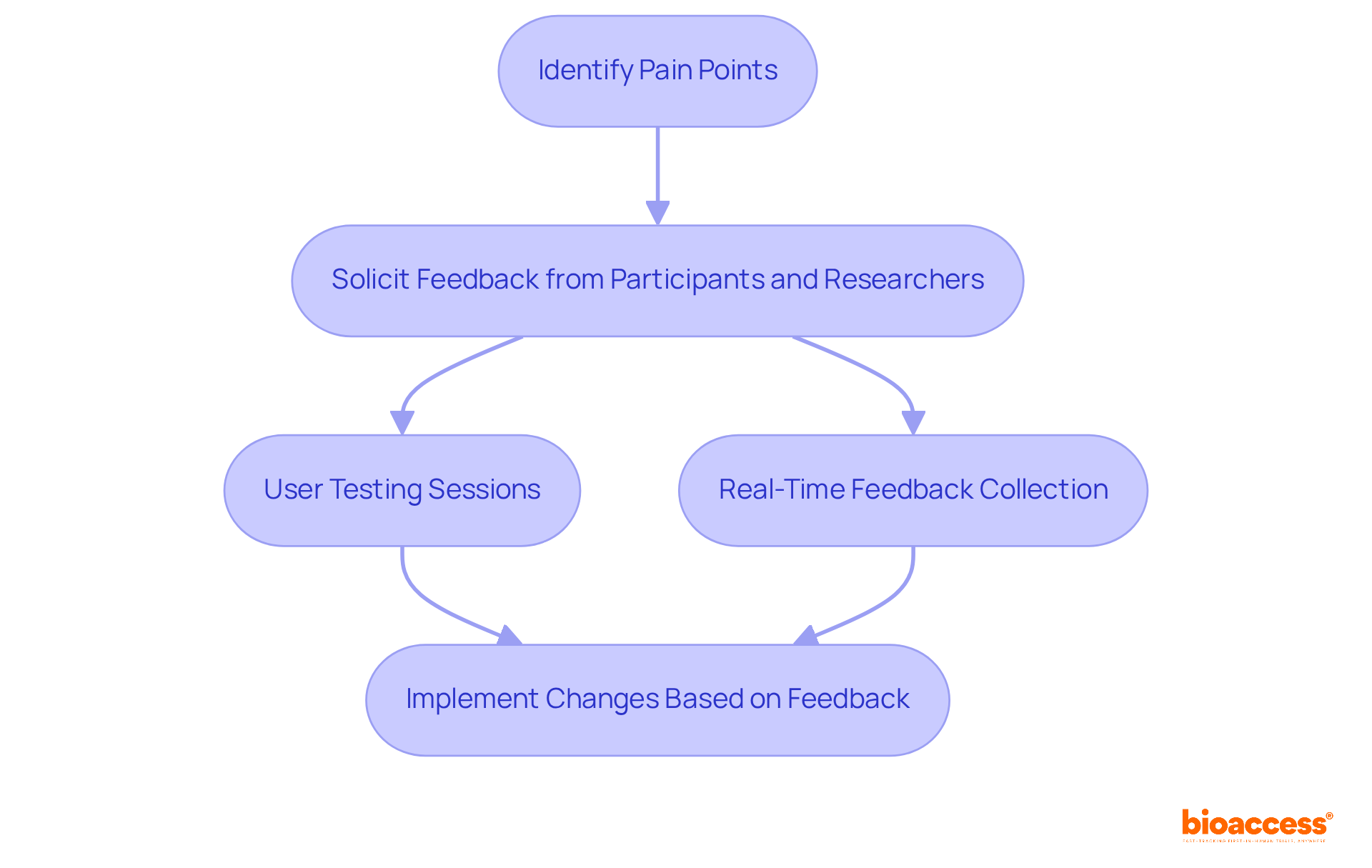
Incorporate Feedback Loops for Continuous eCRF Improvement
Incorporating is essential for fostering . By actively soliciting input from both participants and researchers, designers can pinpoint pain points and identify areas for enhancement. This iterative method not only guarantees that remain efficient and relevant but also significantly enhances the quality of information and participant satisfaction. Statistics indicate that the implementation of electronic case reports can lead to a substantial reduction in and improve data quality, ultimately influencing innovation success rates in .
To implement continuous improvement in , teams should establish a that promotes open communication and collaboration. This may include:
- Regular user testing sessions, which involve testing designs with real users to gather qualitative and quantitative feedback.
- Real-time feedback collection, both of which are crucial for aligning eCRF designs with user needs and regulatory requirements.
As highlighted by industry specialists at , is vital for effective feedback cycles, fostering innovation and ensuring that align with the evolving demands of .
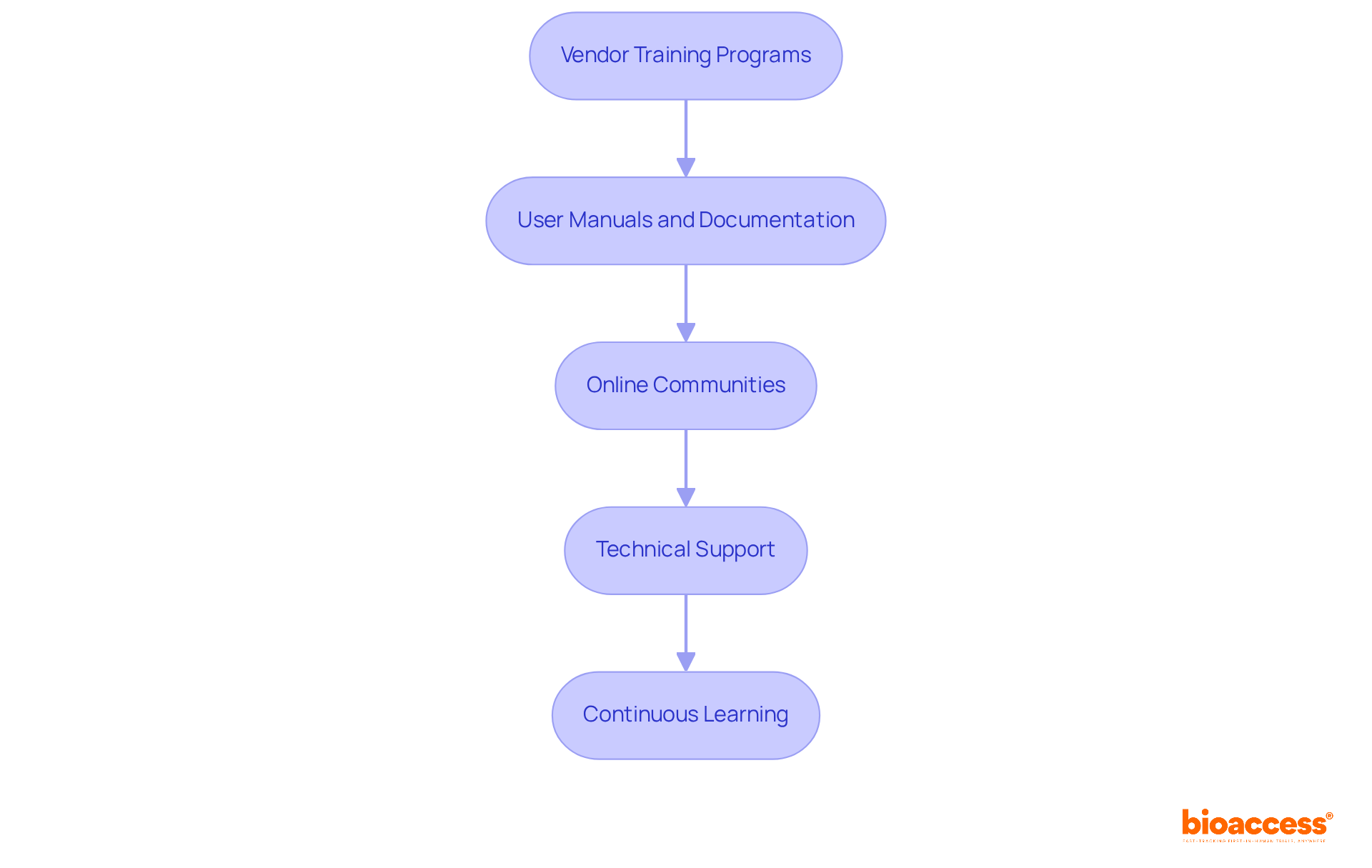
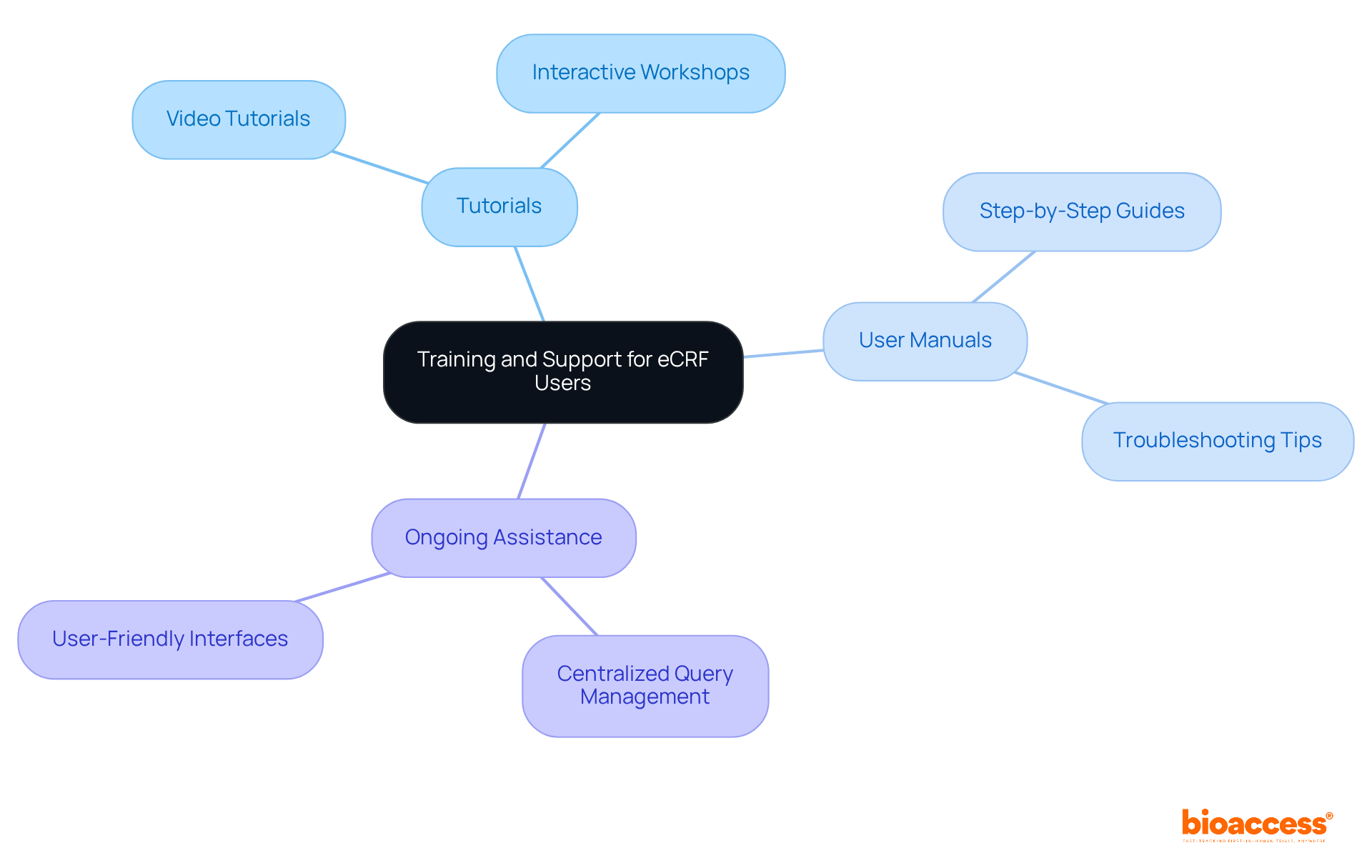
Provide Training and Support for Effective eCRF Use
Providing comprehensive training and support for eCRF users is essential for . This involves offering:
- Tutorials
- User manuals
- Ongoing assistance, including and
These resources empower users to navigate the system with confidence. By ensuring that all stakeholders are proficient in and the features of , researchers can significantly enhance the efficiency of information collection and improve overall study outcomes.
Furthermore, the integration of within enhances the quality of information, while role-based access control and extensive audit trails guarantee security and compliance. and troubleshooting tips serve as invaluable resources, enabling users to resolve issues independently and maintain throughout the study.
Ultimately, investing in robust user support systems is crucial for optimizing the performance of and achieving successful .
Conclusion
Enhancing electronic case report (eCRF) design is pivotal for improving clinical research outcomes and ensuring regulatory compliance. By adopting strategic approaches that emphasize standardization, user-centered design, and continuous improvement, researchers can significantly streamline data collection processes and foster trust among stakeholders. The meticulous focus on design not only accelerates the approval process but also enhances the overall quality of clinical studies.
This article outlines several key strategies, including:
- The integration of Core Data Elements (CDEs) for standardized data collection
- The importance of semantic interoperability
- The necessity of precise wording in eCRF questions
Additionally, implementing robust metadata standards, providing comprehensive training, and establishing feedback loops for continuous improvement are crucial steps that can lead to significant reductions in error rates and research timelines. Each of these strategies contributes to creating a more efficient, reliable, and user-friendly environment for clinical research.
In conclusion, the significance of well-designed electronic case reports cannot be overstated. By prioritizing best practices in eCRF design, researchers can not only enhance data integrity and compliance but also ensure that innovative therapies reach patients more swiftly. Embracing these strategies is essential for driving progress in clinical research and ultimately improving patient outcomes. Engaging with services like those offered by bioaccess® can further empower organizations to achieve excellence in eCRF implementation, paving the way for successful clinical trials and meaningful advancements in healthcare.
Frequently Asked Questions
What is bioaccess® and how does it contribute to electronic case report (eCRF) design?
bioaccess® is a clinical research service provider that accelerates the development of electronic case reports by leveraging extensive research expertise. It ensures that eCRFs are aligned with regulatory standards and customized to meet the unique needs of Medtech, Biopharma, and Radiopharma innovators, which expedites the approval process and enhances information collection quality.
How do well-structured electronic case report forms impact research timelines?
Well-structured electronic case report forms can facilitate a 30% reduction in overall research timelines, allowing for faster market entry of new therapies.
What improvements are associated with tailored electronic case report forms?
Tailored electronic case report forms improve data integrity, reducing error rates to below 5%, which fosters trust among stakeholders and participants.
What is the significance of semantic interoperability in eCRF design?
Semantic interoperability ensures that electronic case report designs adhere to established information standards, such as those set by the Clinical Data Interchange Standards Consortium (CDISC). This guarantees that collected information is shareable and comprehensible across various platforms and stakeholders.
How do standardized terminologies and formats affect clinical outcomes?
Utilizing standardized terminologies and formats streamlines information integration and analysis processes, ultimately leading to improved clinical outcomes and a reduced likelihood of errors in data collection.
What are Core Data Elements (CDEs) and why are they important?
Core Data Elements (CDEs) are predefined information fields that ensure essential details are captured consistently across clinical studies. They enhance the reliability of findings and facilitate effective comparison and analysis.
How do electronic case reports compare to traditional paper forms in terms of data entry errors?
Electronic case reports can reduce entry mistakes to 0%, while traditional paper forms have a 5% error rate, highlighting the advantages of using electronic formats.
How does the use of CDEs streamline the regulatory approval process?
By leveraging Core Data Elements, researchers bolster data integrity and standardization, which streamlines the regulatory approval process and leads to quicker and more efficient trials.
What is the role of CDISC standards in the Medtech industry?
Over 500 organizations in the Medtech industry have adopted CDISC standards to enhance data integrity and regulatory compliance, emphasizing the importance of standardization in improving clinical trial outcomes.
List of Sources
- bioaccess®: Accelerate eCRF Design with Expert Clinical Research Services
- bioaccessla.com (https://bioaccessla.com/blog/10-benefits-of-e-crf-clinical-trials-for-research-directors)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC3909932)
- bioaccessla.com (https://bioaccessla.com/blog/understanding-crf-in-research-definition-evolution-and-importance)
- bioaccessla.com (https://bioaccessla.com/blog/10-essential-e-cr-fs-for-streamlined-clinical-research-success)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC5709849)
- Maximize Semantic Interoperability in eCRF Design
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC11931322)
- bioaccessla.com (https://bioaccessla.com/blog/8-key-insights-on-cdm-medical-abbreviation-for-clinical-research)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC8971042)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC12308985)
- Utilize Core Data Elements (CDEs) for Standardized Data Collection
- bioaccessla.com (https://bioaccessla.com/blog/10-essential-insights-on-clinical-trial-case-report-form-design)
- bioaccessla.com (https://bioaccessla.com/br/blog/10-strategies-for-effective-crf-clinical-trial-design)
- appliedclinicaltrialsonline.com (https://appliedclinicaltrialsonline.com/view/global-standardization-clinical-research-data)
- medrio.com (https://medrio.com/blog/clinical-trial-electronic-data-capture-strategies)
- transceleratebiopharmainc.com (https://transceleratebiopharmainc.com/initiatives/clinical-data-standards)
- Implement Standards in CRF Metadata for Compliance
- quanticate.com (https://quanticate.com/blog/ecrf-design-in-clinical-trials)
- mahalo.health (https://mahalo.health/insights/how-ecrfs-modernize-clinical-data-management)
- qualityze.com (https://qualityze.com/blogs/ecrf-electronic-case-report-form)
- bioaccessla.com (https://bioaccessla.com/blog/10-essential-e-cr-fs-for-streamlined-clinical-research-success)
- Ensure Accurate Wording in eCRF Questions
- Participants’ understanding of informed consent in clinical trials over three decades: systematic review and meta-analysis – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC4371493)
- researchgate.net (https://researchgate.net/publication/267102679_Impact_of_Source_Data_Verification_on_data_quality_in_clinical_trials_An_empirical_post-hoc_analysis_of_three_phase_3_randomised_clinical_trials)
- Checking your browser – reCAPTCHA (https://pmc.ncbi.nlm.nih.gov/articles/PMC10775420)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC6904374)
- Participants’ understanding of informed consent in clinical trials: A systematic review and updated meta-analysis (https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0295784)
- Provide Semantically Coherent Answer Options in eCRFs
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC11931322)
- prolific.com (https://prolific.com/resources/survey-data-quality-factors-that-matter-to-researchers)
- 12 Data Quality Metrics to Measure Data Quality in 2026 (https://lakefs.io/data-quality/data-quality-metrics)
- 9 Key Data Quality Metrics You Need to Know in 2025 (https://atlan.com/data-quality-metrics)
- Clearly State Units of Measure in eCRFs
- careerfoundry.com (https://careerfoundry.com/en/blog/data-analytics/inspirational-data-quotes)
- 70 Research Quotes to Inspire Your Work – Qualtrics (https://qualtrics.com/articles/strategy-research/research-quotes)
- nature.com (https://nature.com/articles/s41597-024-04312-x)
- 100 Essential Data Storytelling Quotes (https://effectivedatastorytelling.com/post/100-essential-data-storytelling-quotes)
- Adopt User-Centered Design Principles for eCRFs
- pubmed.ncbi.nlm.nih.gov (https://pubmed.ncbi.nlm.nih.gov/32815381)
- healthcareitnews.com (https://healthcareitnews.com/news/frustrations-linger-around-electronic-health-records-and-user-centered-design)
- bioaccessla.com (https://bioaccessla.com/blog/10-essential-insights-on-clinical-trial-case-report-form-design)
- cloudbyz.com (https://cloudbyz.com/resources/clinical-operations/comprehensive-guide-to-electronic-case-report-forms-ecrf-faqs-benefits-and-best-practices)
- Incorporate Feedback Loops for Continuous eCRF Improvement
- bioaccessla.com (https://bioaccessla.com/blog/10-essential-e-cr-fs-for-streamlined-clinical-research-success)
- How Feedback Loops Sharpen Real-Time Decision-Making (https://certaintynews.com/article/how-feedback-loops-sharpen-real-time-decision-making)
- medium.com (https://medium.com/@UXVerse/the-importance-of-feedback-loops-in-the-ux-design-process-db35c1c4aeea)
- Design feedback loops: Examples and best practices for creatives (https://ziflow.com/blog/design-feedback-loop-examples)
- minervaresearchsolutions.com (https://minervaresearchsolutions.com/electronic-case-report-forms)
- Provide Training and Support for Effective eCRF Use
- Comprehensive Guide to Electronic Data Capture (EDC) Systems | ICON news and blogs (https://careers.iconplc.com/blogs/2024-12/comprehensive-guide-to-electronic-data-capture-edc-systems)
- mahalo.health (https://mahalo.health/insights/how-ecrfs-modernize-clinical-data-management)
- ccrps.org (https://ccrps.org/clinical-research-blog/how-gcp-training-enhances-data-accuracy-in-clinical-trials)