Introduction
The landscape of medical device regulation in Brazil is evolving rapidly, particularly with the implementation of Unique Device Identification (UDI) requirements poised to reshape the industry. As manufacturers prepare to meet stringent compliance deadlines, the demand for expert guidance and support has never been more critical. This article explores ten key providers of UDI implementation support in Brazil, illustrating how these organizations can help navigate the complexities of regulatory compliance, enhance operational efficiency, and ultimately improve patient safety. With the stakes high and the timeline tightening, manufacturers must consider:
- How can they ensure they are well-prepared for the upcoming changes?
bioaccess: Leading CRO for UDI Implementation Support in Brazil
bioaccess® is recognized as a leading contract research organization (CRO) in Brazil, offering for Brazil providers for medical equipment manufacturers. With over 20 years of experience in and regulatory consulting, bioaccess® provides that offer , simplifying the adherence process. Their deep understanding of local regulations, combined with a robust network of more than 3,000 pre-qualified research sites, ensures efficient to meet . This includes registering items in the Siud database and applying standardized UDI labels starting July 2025.
The company boasts a , marked by faster patient recruitment and -10% to 15% above global averages-demonstrating its ability to navigate Brazil’s effectively. By leveraging bioaccess®’s expertise, medical equipment firms can confidently address UDI adherence with the help of , . Notably, bioaccess® has played a pivotal role in assisting Avantec Vascular with their in Latin America, showcasing their commitment to advancing clinical research in the region.
GS1: Global Standards for UDI Compliance in Brazil
GS1 is a pivotal global standards organization, offering for compliance with . Their comprehensive framework equips manufacturers with essential tools and to effectively navigate UDI requirements. By providing detailed guidelines and resources, GS1 enhances the precise identification and tracking of . This not only strengthens but also ensures .
The phased rollout of , with the assistance of UDI implementation support Brazil providers and aligned with international best practices, underscores GS1’s commitment to . Success stories from manufacturers who have adopted GS1 standards reveal significant advancements in , showcasing the tangible benefits of these guidelines in real-world applications. Expert opinions further highlight the importance of GS1’s role in facilitating a smoother transition to UDI compliance, ultimately fostering a safer healthcare environment.
Intertek: Expert UDI Compliance Services for Medical Devices
Intertek provides specialized UDI adherence services designed specifically for . Their comprehensive solutions include , testing, and certification, ensuring devices meet ANVISA’s UDI requirements. By emphasizing safety and traceability, Intertek offers , enabling clients to navigate the complex landscape effectively. This support is crucial as manufacturers gear up for impending , helping them maintain market competitiveness and enhance operational efficiency.
Notably, Intertek’s approach has led to a significant reduction in time to market. Some clients have achieved on their first attempt, avoiding costly delays and the need for re-testing. This success highlights the importance of collaborating with experienced regulatory consultants to streamline the UDI implementation support Brazil providers process.
FiveValidation: Solutions for UDI Validation Challenges
FiveValidation stands out in addressing the UDI validation challenges that encounter, thanks to its innovative SaaS platform. This scalable solution accelerates significantly, ensuring compliance with while effectively .
By leveraging advanced technology and deep industry expertise, FiveValidation empowers clients to , achieving than traditional methods.
With a strong focus on efficiency, the platform not only reduces the time spent on validation but also , making it an indispensable tool for manufacturers seeking UDI implementation support while navigating the complexities of UDI regulations.
Zebra Technologies: UDI Solutions for Enhanced Patient Safety
Zebra Technologies delivers cutting-edge that significantly bolster . Their and hardware facilitate precise capture and management of UDI data, ensuring compliance with regulatory standards. By enhancing traceability and , Zebra’s technology empowers to offer safer patient care. Notably, recent updates in 2026 have further refined these solutions, underscoring Zebra’s dedication to improving healthcare outcomes.
Statistics indicate that implementing UDI through Zebra’s systems has led to a marked reduction in healthcare errors, highlighting the critical need for . Experts assert that integrating UDI Scan Plus, with the assistance of , not only streamlines compliance but also , establishing it as an essential resource for healthcare institutions in Brazil aiming to elevate .
PureGlobal: Insights on Brazil’s UDI Compliance Requirements
PureGlobal serves as a crucial partner for medical equipment producers seeking to meet the . They offer in-depth guidance on the evolving , highlighting essential deadlines such as the , and for . By assisting clients in navigating the complexities of these regulations, PureGlobal provides to ensure that manufacturers are fully prepared for the upcoming UDI requirements, which are vital for enhancing product traceability and safety within the Brazilian market.
Ana Criado, our Director of , brings a wealth of expertise in , having collaborated with global companies and served as a professor in biomedical engineering. Her insights are instrumental in grasping the implications of UDI compliance. As Janaina dos Santos de Miranda notes, “Brazil’s Unique Identification (UDI) system is moving toward full enforcement, with .” is expected to significantly , fostering a more transparent and efficient system in Brazil.
Moreover, understanding the can greatly assist manufacturers in planning their .
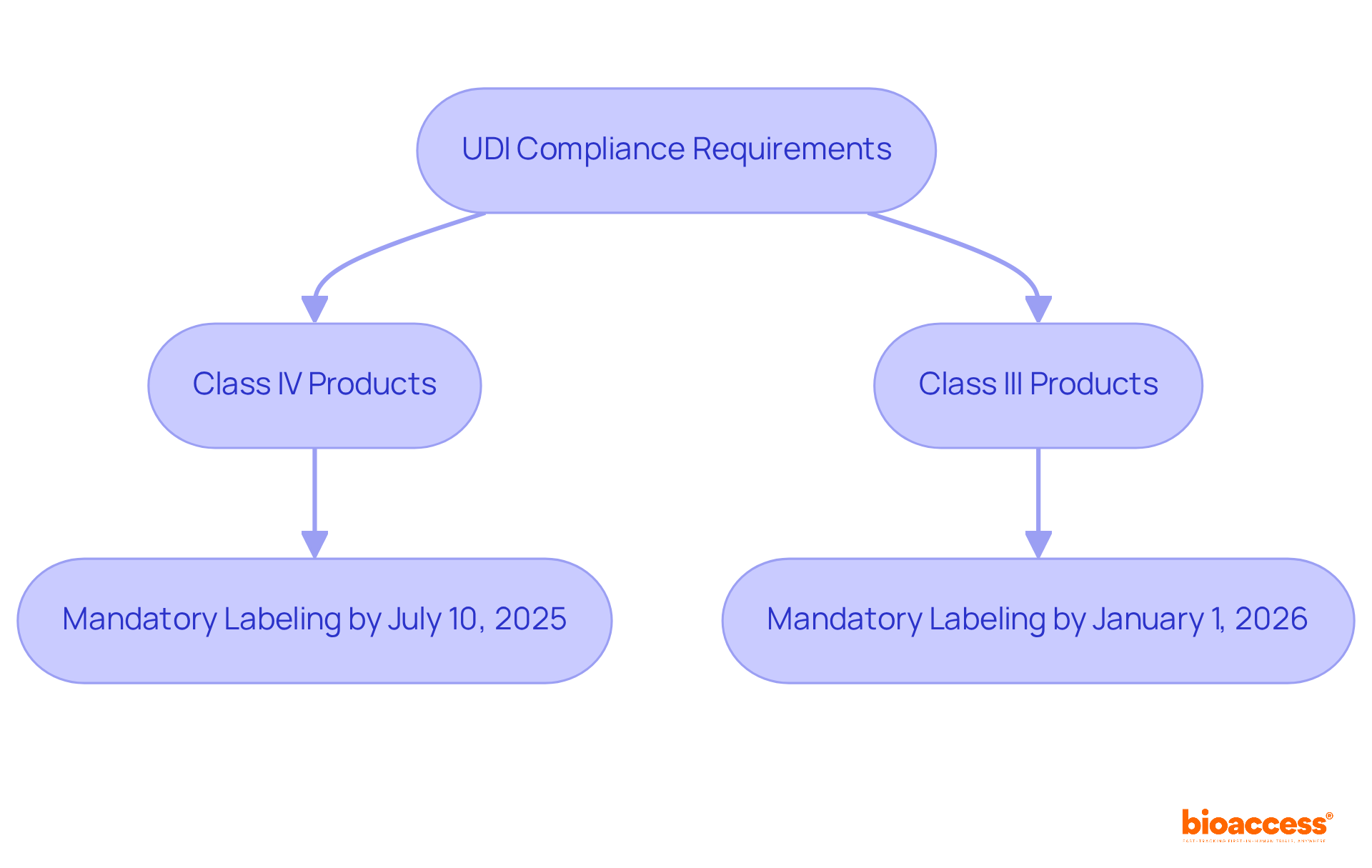
Rimsys: Global UDI Requirements and Timelines Overview
Rimsys delivers a vital overview of , crucial for medical product manufacturers in Brazil and beyond. Their platform streamlines UDI data management, ensuring compliance with both local and international regulations. With the European Union’s UDI system set to become , accompanied by a six-month grace period for existing products, is essential. Rimsys empowers clients with insights into these impending requirements, enabling them to adeptly navigate the shifting UDI landscape.
As the industry adapts to these changes, the importance of cannot be overstated. It , and guarantees adherence to regulatory standards. By leveraging Rimsys’ expertise, manufacturers can receive from Brazil providers to mitigate and maintain their foothold in Brazil’s competitive healthcare market.
Greenlight Guru: Ultimate Guide to UDI for Medical Devices
Greenlight Guru serves as an essential resource for with its , published in 2026. This guide covers a broad spectrum of topics, from the basics of UDI to , empowering companies to navigate the complex landscape of UDI regulations effectively. By shedding light on , Greenlight Guru simplifies UDI adherence efforts for its clients.
In a market where grasping the nuances of UDI is crucial for , their expertise stands out. The faces a significant challenge due to the . Greenlight Guru’s resources are invaluable for producers aiming to meet regulatory standards and . Notably, there are between 20 and 30 distinct routes to market for healthcare products in the U.S., highlighting the intricacy of UDI compliance. As Mike Drues points out, regulation hinges on the interpretation of words and the ability to defend those interpretations, underscoring the importance of Greenlight Guru’s guidance.
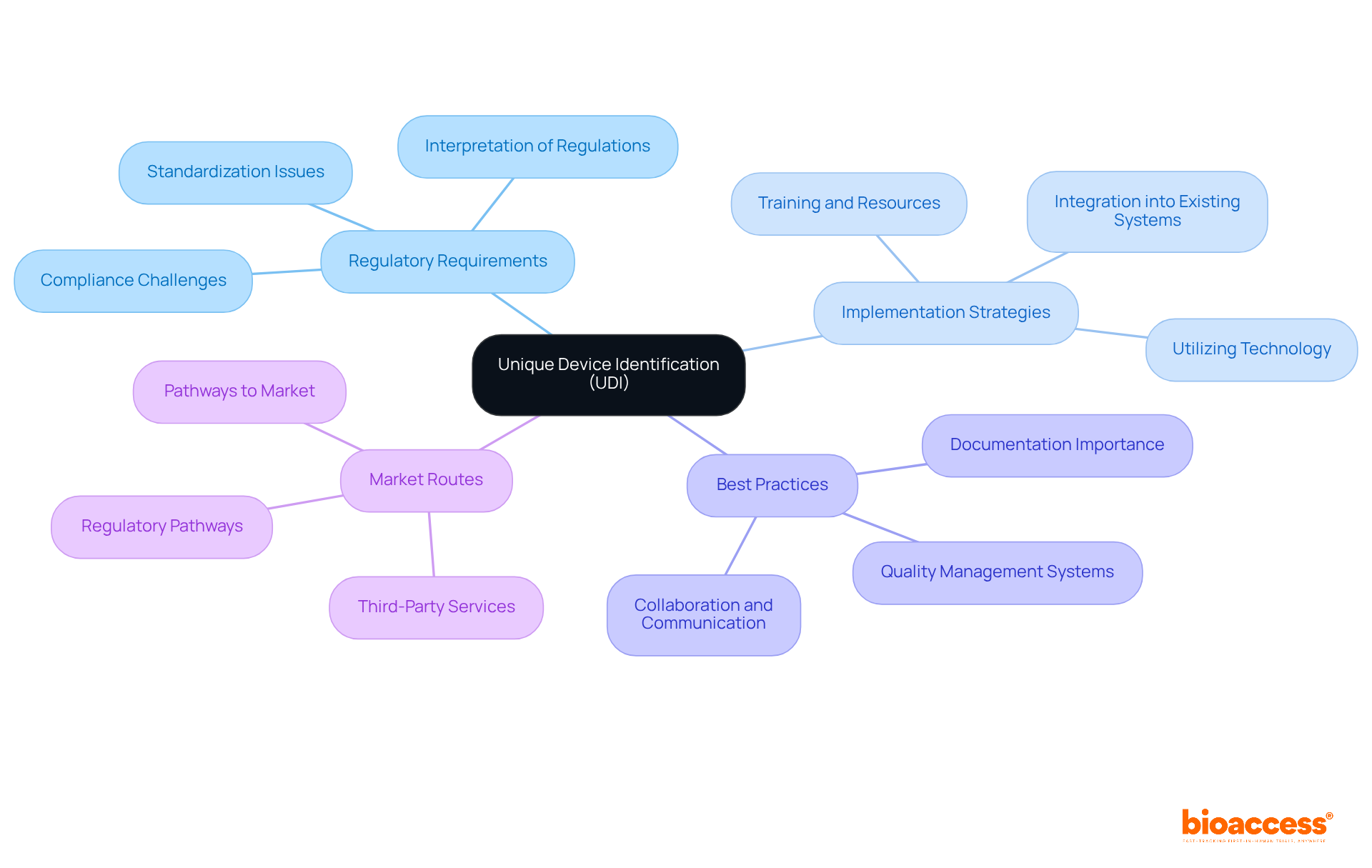
ANVISA: Regulatory Authority for UDI in Brazil
, serves as the regulatory authority overseeing the implementation of Unique Identification (UDI) requirements for health products. Established in 1999, ANVISA’s regulations, particularly , delineate the essential steps manufacturers must undertake to comply with UDI standards. This regulation has been pivotal in of healthcare products within the Brazilian market.
Recent updates, including those introduced in RDC 751/2022 and RDC 830/2023, have streamlined reporting and registration processes, granting additional time for compliance, especially for healthcare products categorized under risk levels II, III, and IV. These modifications reflect ANVISA’s dedication to facilitating the integration of UDI systems, recognizing the challenges that UDI implementation support Brazil providers encounter. Furthermore, ANVISA mandates to monitor the performance of healthcare products, ensuring ongoing safety and effectiveness. As a result, adherence rates for healthcare instruments have improved, with ethical approval rates for clinical trials in Brazil nearing 75%.
ANVISA’s proactive stance not only protects public health but also encourages innovation within the health technology sector, ensuring that medical devices adhere to rigorous safety, efficacy, and quality standards. In this context, bioaccess® emerges as a premier (CRO) in Latin America, offering comprehensive , including Early-Feasibility, , Pilot, Pivotal, and Post-Market Follow-Up Studies. Bioaccess® offers , assisting Medtech startups in navigating and achieving expedited clinical study outcomes with a tailored approach to address the unique challenges of the market.
As noted by the Bioaccess Content Team, “ANVISA was established in 1999 and plays a crucial role in Brazil’s healthcare landscape by fostering innovation in the health technology sector while prioritizing patient safety.” This collaboration between regulatory bodies and research organizations is essential for advancing clinical research and ensuring the safety of healthcare products.
LinkedIn: Networking Platform for UDI Implementation Updates
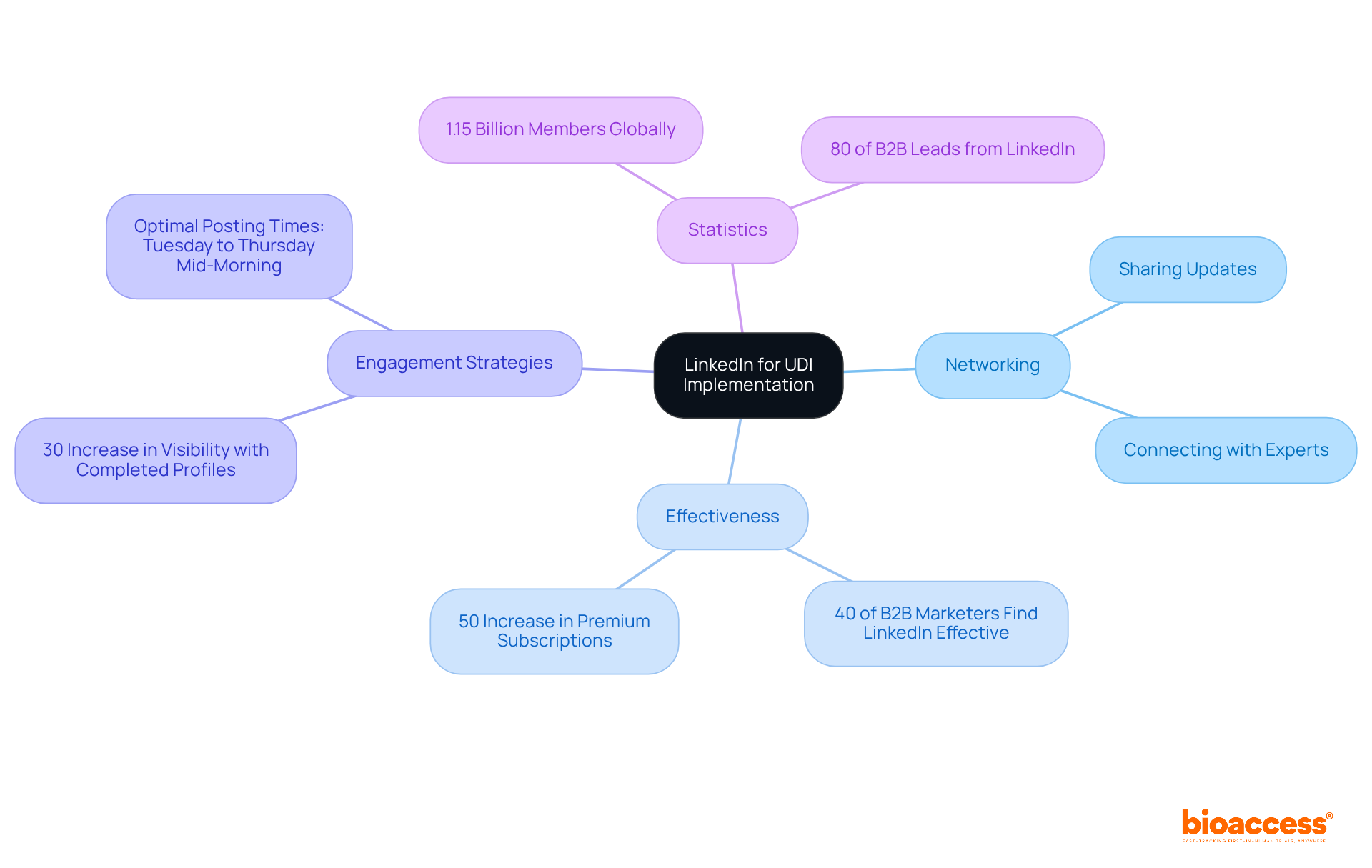
LinkedIn serves as an essential networking platform for professionals engaged in , offering a dynamic space for sharing updates, insights, and best practices related to UDI regulations. By connecting with industry experts and peers, manufacturers can stay informed about and collaborate on innovative solutions to meet effectively. This platform enhances the sharing of knowledge, enabling users to navigate the complexities of regulations with greater ease.
Moreover, LinkedIn stands as a hub for discussions on the latest UDI regulatory updates, ensuring users are equipped with the most current information and strategies to succeed in their initiatives. Notably, , underscoring its significance for UDI regulation professionals. The further indicates a growing demand for advanced networking tools, making it an even more valuable resource.
Users can also boost their visibility and engagement on the platform; a completed LinkedIn page can increase weekly views by 30%. To maximize engagement when sharing UDI adherence updates, it’s advisable to post during optimal times, particularly from Tuesday to Thursday around mid-morning. With 80% of originating from LinkedIn, the platform’s importance in the context of UDI compliance is undeniable.
Conclusion
As Brazil’s healthcare industry gears up for the critical UDI deadlines, the journey toward successful implementation is bolstered by a diverse range of specialized providers. Each of these players offers unique solutions tailored to navigate the complex regulatory landscape, ensuring compliance with the Unique Device Identification system. This system is vital for enhancing product traceability and patient safety.
Highlighted in this article are ten key contributors to the UDI landscape:
- bioaccess®’s extensive experience in clinical trials and regulatory consulting
- GS1’s global standards that streamline compliance
- Intertek’s regulatory consulting expertise
- FiveValidation’s innovative validation solutions
- Zebra Technologies’ advanced UDI systems
- PureGlobal
- Rimsys
- Greenlight Guru
- ANVISA
Engaging with these support providers is not merely beneficial; it is essential for manufacturers striving to maintain compliance and enhance operational efficiency. By leveraging expert insights and innovative solutions, the healthcare sector can foster a safer environment for patients and providers alike. Embracing these resources will pave the way for a successful transition to UDI compliance, underscoring the critical importance of collaboration and knowledge-sharing in this vital sector.
Frequently Asked Questions
What is bioaccess and what services do they provide?
bioaccess is a leading contract research organization (CRO) in Brazil that offers UDI implementation support for medical equipment manufacturers. They provide tailored solutions to simplify the adherence process, leveraging over 20 years of experience in clinical trials and regulatory consulting.
How does bioaccess support UDI implementation for Brazil providers?
bioaccess supports UDI implementation by helping providers register items in the Siud database and apply standardized UDI labels, ensuring compliance with critical deadlines set by ANVISA, starting July 2025.
What is the significance of bioaccess’s network in UDI implementation?
bioaccess has a robust network of over 3,000 pre-qualified research sites, which facilitates efficient UDI implementation support for Brazil providers, enhancing their ability to meet regulatory requirements.
What are the advantages of working with bioaccess for UDI compliance?
Working with bioaccess offers advantages such as faster patient recruitment, higher retention rates, and a deep understanding of Brazil’s regulatory landscape, which collectively enhance operational efficiency and market readiness.
Who has bioaccess assisted in clinical studies, and what does this indicate about their capabilities?
bioaccess has assisted Avantec Vascular with their first-in-human clinical study of an innovative vascular instrument in Latin America, showcasing their commitment to advancing clinical research and their capability in supporting complex studies.
What role does GS1 play in UDI compliance in Brazil?
GS1 is a global standards organization that provides UDI implementation support to Brazil providers, equipping manufacturers with tools and guidelines to navigate UDI requirements effectively and enhance product traceability.
How does GS1 contribute to patient safety and regulatory compliance?
By offering a comprehensive framework and resources, GS1 strengthens the identification and tracking of healthcare products, thereby improving patient safety and ensuring compliance with regulatory standards.
What is the phased rollout of UDI requirements in Brazil?
The phased rollout involves implementing UDI requirements in Brazil with the assistance of UDI implementation support from Brazil providers, aligned with international best practices to enhance medical device safety.
What benefits have manufacturers experienced by adopting GS1 standards?
Manufacturers who have adopted GS1 standards report significant advancements in traceability and adherence, demonstrating the tangible benefits of these guidelines in real-world applications.
What services does Intertek provide for UDI compliance in Brazil?
Intertek offers specialized UDI adherence services, including regulatory consulting, testing, and certification, ensuring that medical devices meet ANVISA’s UDI requirements.
How does Intertek help manufacturers with UDI implementation?
Intertek emphasizes safety and traceability, providing UDI implementation support that enables manufacturers to navigate the regulatory landscape effectively, maintain competitiveness, and enhance operational efficiency.
What outcomes have clients achieved by working with Intertek?
Clients have experienced a significant reduction in time to market, with some achieving regulatory approval on their first attempt, thus avoiding costly delays and re-testing.
List of Sources
- bioaccess: Leading CRO for UDI Implementation Support in Brazil
- ANVISA Publishes More UDI Guidance (https://emergobyul.com/news/anvisa-publishes-more-udi-guidance)
- bioaccessla.com (https://bioaccessla.com/blog/how-to-conduct-first-in-human-studies-cro-in-brazil-a-step-by-step-guide)
- meddeviceonline.com (https://meddeviceonline.com/doc/brazil-s-competitive-edge-infrastructure-and-operational-advantages-for-medical-device-trials-0001)
- Medical Device Innovation In Brazil: Success Stories And Future Opportunities (https://meddeviceonline.com/doc/medical-device-innovation-in-brazil-success-stories-and-future-opportunities-0001)
- Brazil Anvisa UDI System 2025 Launch: Medical Device Regulation Update | News | Pure Global (https://pureglobal.com/news/brazil-anvisa-udi-system-2025-launch-medical-device-regulation-update)
- GS1: Global Standards for UDI Compliance in Brazil
- Brazil ANVISA RDC 591/2021: Unique Device Identification (UDI) System for Medical Devices (https://elendilabs.com/en/articles/bra-unique-medical-device-identification)
- Unique Device Identification (UDI) – Healthcare | GS1 (https://gs1.org/industries/healthcare/udi)
- Brazil’s UDI System Nears Enforcement: What Medical Device Manufacturers Must Know for 2025 and Beyond | Blog | Pure Global (https://pureglobal.com/blog-posts/brazils-udi-system-nears-enforcement-what-medical-device-manufacturers-must-know-for-2025-and-beyond)
- GS1 Standards in Healthcare: Current & Future Uses (https://linkedin.com/pulse/gs1-standards-healthcare-current-future-uses-kevin-lewis-wnfcc)
- How GS1 Standards Improve Medical Device Safety & Efficiency (https://mavenprofserv.com/blog/the-gs1-advantage-for-medical-device)
- Intertek: Expert UDI Compliance Services for Medical Devices
- Medical Device Sensitization Testing Market, Global Outlook and Forecast 2025-2032 (https://statsmarketresearch.com/global-medical-device-sensitization-testing-forecast-market-8063313)
- Medical Devices Market Size Worth USD 1,209.42 Bn by 2035 Fueled by Advances in Diagnostic Technologies – Ortho Spine News (https://orthospinenews.com/2025/12/10/medical-devices-market-size-worth-usd-1209-42-bn-by-2035-fueled-by-advances-in-diagnostic-technologies)
- Case Study: Regulatory Consulting Services for Medical Devices (https://intertek.com/consulting/medical/case-study-regulatory-consulting-devices)
- Client Impact Stories (https://intertek.com/consulting/medical/client-impact-stories)
- FiveValidation: Solutions for UDI Validation Challenges
- Why speed in validation activities is a competitive advantage (https://fivevalidation.com/why-speed-in-validation-activities-is-a-competitive-advantage)
- 175+ Unmissable SaaS Statistics for 2026 (https://zylo.com/blog/saas-statistics)
- The big list of 2026 SaaS statistics that you should know | BetterCloud (https://bettercloud.com/monitor/saas-statistics)
- 85 SaaS Statistics, Trends and Benchmarks for 2026 – Vena (https://venasolutions.com/blog/saas-statistics)
- Zebra Technologies: UDI Solutions for Enhanced Patient Safety
- Zebra Study: 84% of U.S. and UK Hospital Leaders Prioritize Digitizing Inventory Management Solutions | Zebra (https://zebra.com/us/en/about-zebra/newsroom/press-releases/2024/zebra-study-84-of-us-and-uk-hospitals-leaders-prioritize-digitizing-inventory-management-solutions.html)
- What Is Unique Device Identification? | Zebra (https://zebra.com/us/en/resource-library/faq/what-Is-unique-device-identification-udi.html)
- Explore Key Trends in Zebra’s Hospital Materials Management Vision Study (https://id-integration.com/announcing-zebra-technologies-healthcare-vision-study-key-trends-and-insights)
- Zebra UDI Scan Plus – for greater patient safety in healthcare! | Jarltech.com (https://jarltech.com/en/news/474)
- Advancing Patient Safety Surrounding Medical Devices: Barriers, Strategies, and Next Steps in Health System Implementation of Unique Device Identifiers – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC9233486)
- PureGlobal: Insights on Brazil’s UDI Compliance Requirements
- Brazil’s UDI Update RDC 591/2021 (https://emergobyul.com/news/brazils-udi-update-rdc-5912021)
- Brazil’s UDI System Nears Enforcement: What Medical Device Manufacturers Must Know for 2025 and Beyond | Blog | Pure Global (https://pureglobal.com/blog-posts/brazils-udi-system-nears-enforcement-what-medical-device-manufacturers-must-know-for-2025-and-beyond)
- ANVISA Brazil Medical Device Registration | Pure Global (https://pureglobal.com/markets/brazil)
- Brazil Anvisa UDI System 2025 Launch: Medical Device Regulation Update | News | Pure Global (https://pureglobal.com/news/brazil-anvisa-udi-system-2025-launch-medical-device-regulation-update)
- Quick reference guide – global medical device UDI requirements and timelines (https://rimsys.io/blog/quick-reference-guide-global-udi-requirements-and-timelines)
- Rimsys: Global UDI Requirements and Timelines Overview
- Quick reference guide – global medical device UDI requirements and timelines (https://rimsys.io/blog/quick-reference-guide-global-udi-requirements-and-timelines)
- Rimsys POV: Updated EUDAMED timeline (https://rimsys.io/blog/rimsys-pov-new-eudamed-timeline)
- Global UDI Timeline | Innovit (https://innovit.com/udi-reference-center/global-udi/global-udi-timeline)
- Planning Your Enterprise’s UDI Strategy for EUDAMED and Beyond (https://rimsys.io/blog/planning-your-enterprises-udi-strategy-for-eudamed-and-beyond)
- Greenlight Guru: Ultimate Guide to UDI for Medical Devices
- Understanding the UDI System for Medical Devices (https://greenlight.guru/blog/understanding-the-udi-system-for-medical-devices)
- Meet a Guru: Brittani Smith (https://greenlight.guru/blog/meet-a-guru-brittani-smith)
- The Best of Jon & Mike Through the Years (https://greenlight.guru/blog/the-best-of-jon-mike-through-the-years)
- Ultimate Guide to UDI for Medical Devices – Free Download (https://greenlight.guru/downloads/udi-medical-devices)
- ANVISA: Regulatory Authority for UDI in Brazil
- ANVISA’s Latest Guidelines and Regulatory Updates for UDI Compliance – Domo Salute (https://domosalute.com.br/news/anvisas-latest-guidelines-and-regulatory-updates-for-udi-compliance)
- Understanding Anvisa Guidelines For Medical Device Trials An In Depth Tutorial | bioaccess® (https://bioaccessla.com/blog/understanding-anvisa-guidelines-for-medical-device-trials-an-in-depth-tutorial)
- Brazil Anvisa UDI System 2025 Launch: Medical Device Regulation Update | News | Pure Global (https://pureglobal.com/news/brazil-anvisa-udi-system-2025-launch-medical-device-regulation-update)
- Brazil Regulatory Updates (https://emergobyul.com/resources/brazil-regulatory-updates)
- LinkedIn: Networking Platform for UDI Implementation Updates
- 30 LinkedIn statistics that marketers must know in 2026 (https://sproutsocial.com/insights/linkedin-statistics)
- LinkedIn Statistics That Prove What’s Working in 2025 (https://botdog.co/blog-posts/linkedin-statistics)
- LinkedIn Statistics 2025: Full Guide for Pros & Recruiters (https://wavecnct.com/blogs/news/linkedin-statistics?srsltid=AfmBOorB8p1PYBust0bTtxgtGHPwHxehcPjbuVlgBdXX-LYuMSUUh0j6)
- buffer.com (https://buffer.com/resources/linkedin-statistics)
- 100 Essential LinkedIn Statistics and Facts for 2026 (https://cognism.com/blog/linkedin-statistics)

Leave a Reply