Introduction
Data monitoring committees (DMCs) stand as the guardians of integrity and safety in clinical research, serving as independent evaluators of study data. These expert groups not only ensure compliance with ethical standards but also provide essential oversight that can determine the fate of clinical trials.
As the landscape of medical research becomes increasingly complex, how can these committees effectively navigate the challenges of participant safety and data integrity while making timely decisions?
Understanding the multifaceted functions and significance of DMCs is crucial for anyone involved in clinical trials, as their recommendations can profoundly impact both research outcomes and participant welfare.
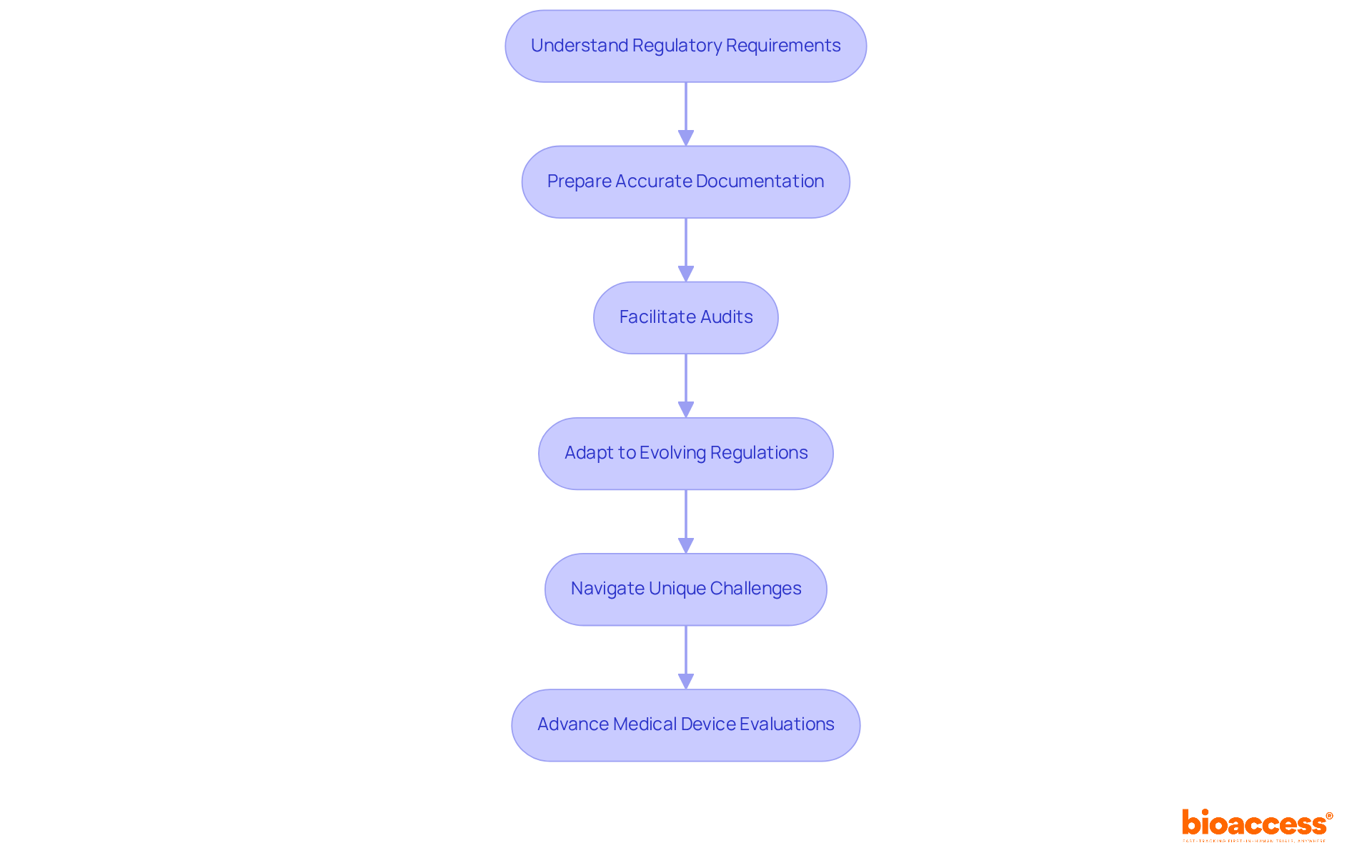
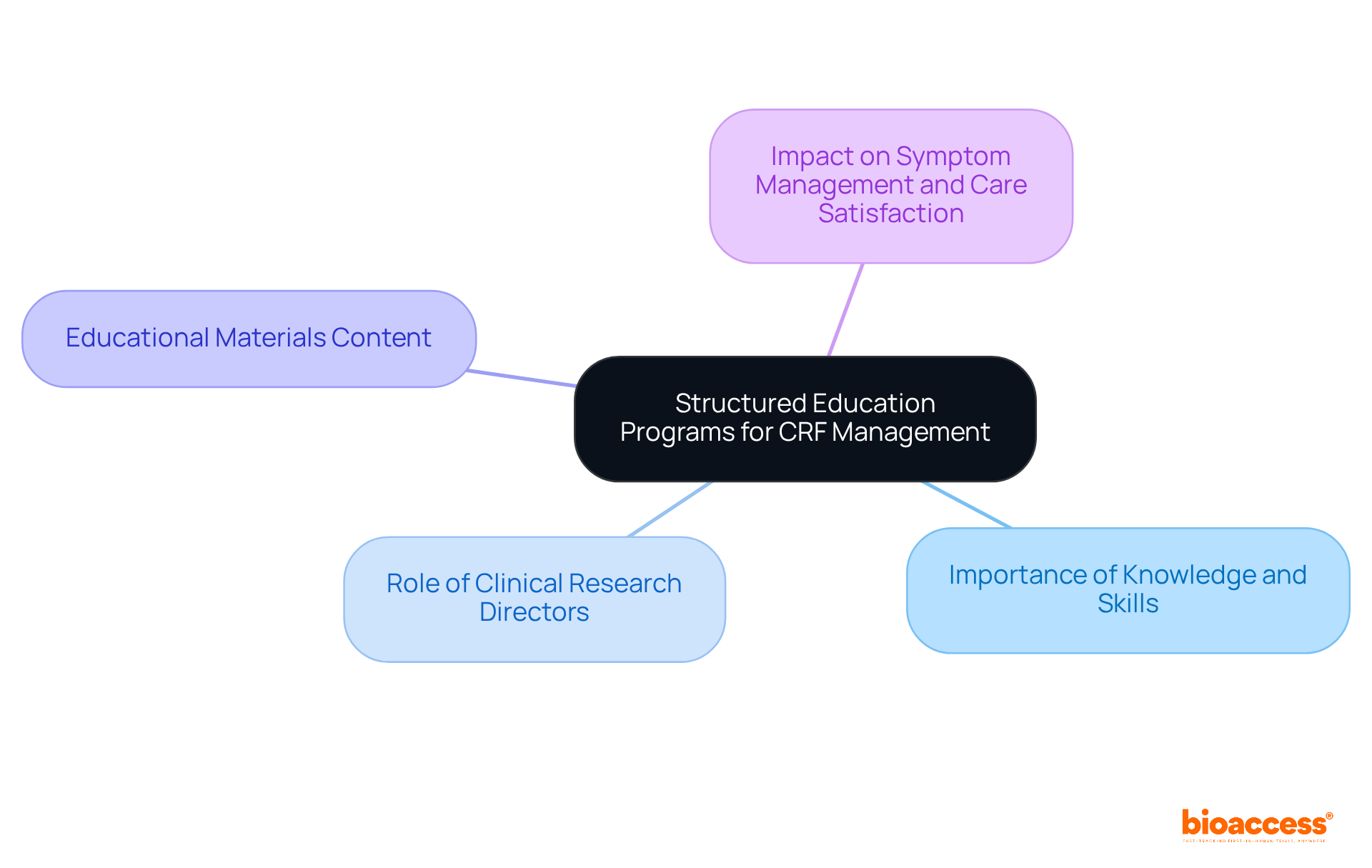
Define Data Monitoring Committee (DMC)
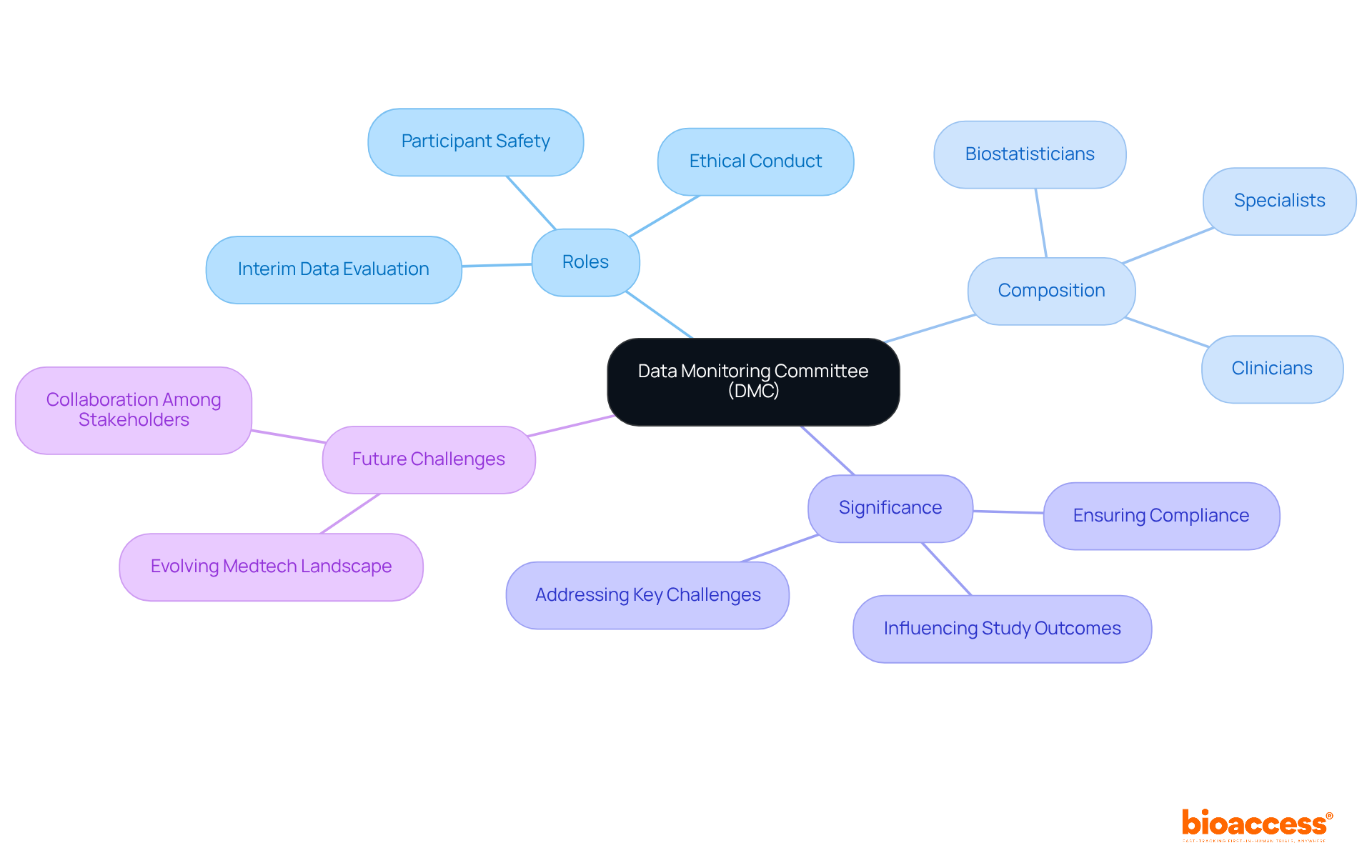
A (DMC), also known as a (DSMB), plays a crucial role in overseeing the well-being and effectiveness of . These independent groups of experts are dedicated to ensuring ethical conduct and prioritizing through regular evaluations of interim data. Comprising a diverse array of professionals – clinicians, biostatisticians, and specialists in relevant fields – DMCs assess the progress of studies and provide recommendations on whether to continue, modify, or terminate research based on their analyses.
The significance of s cannot be overstated, particularly in 2026. They are pivotal in identifying and ensuring compliance with . Their recommendations can profoundly influence study outcomes, reinforcing their status as a cornerstone of responsible . As the Medtech landscape evolves, the role of DMCs in addressing key challenges becomes increasingly vital. Collaboration among stakeholders is essential to navigate these complexities effectively.
In conclusion, the oversight provided by the is indispensable for upholding the integrity of and safeguarding participant interests. As we move forward, fostering collaboration and understanding the critical functions of DMCs will be essential for advancing .
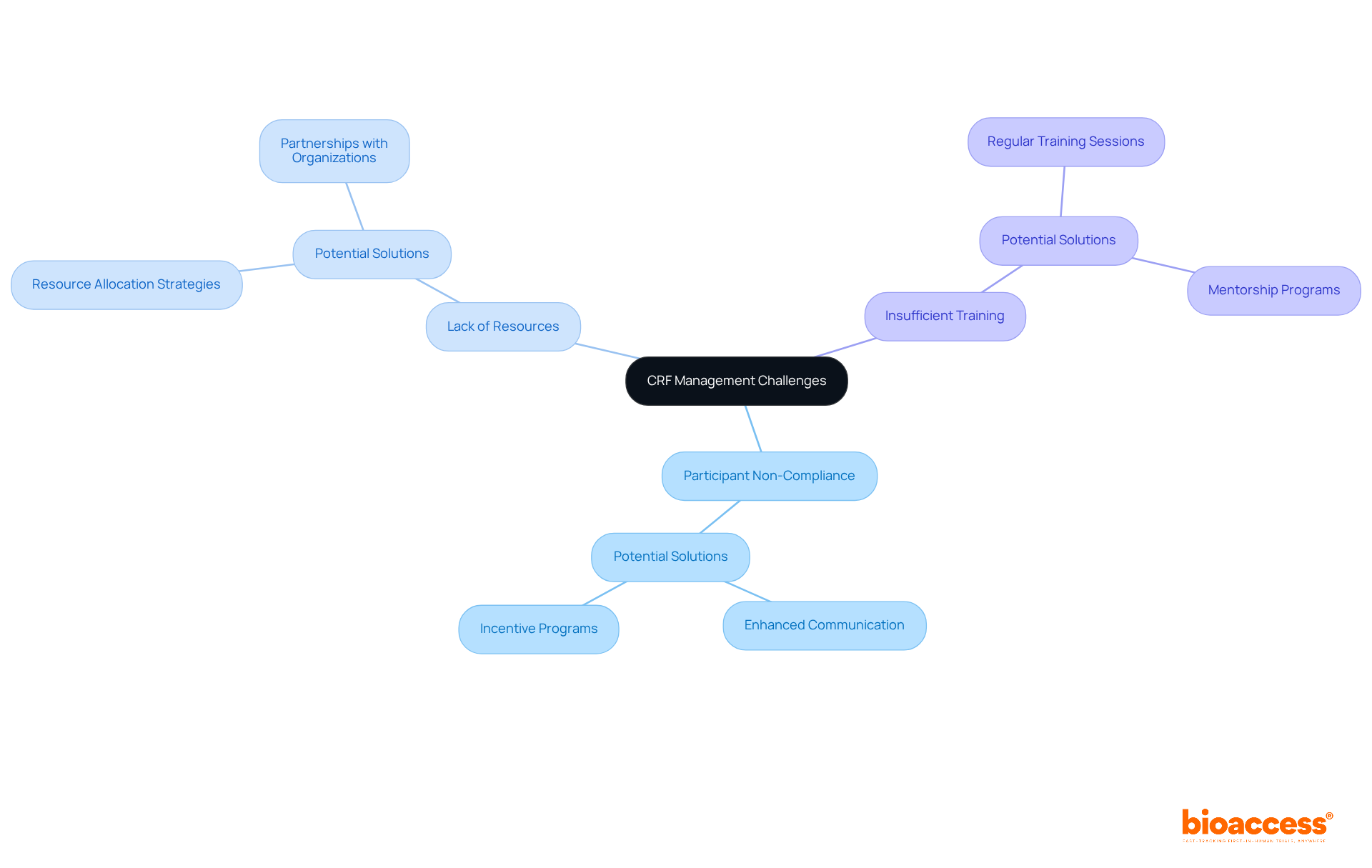
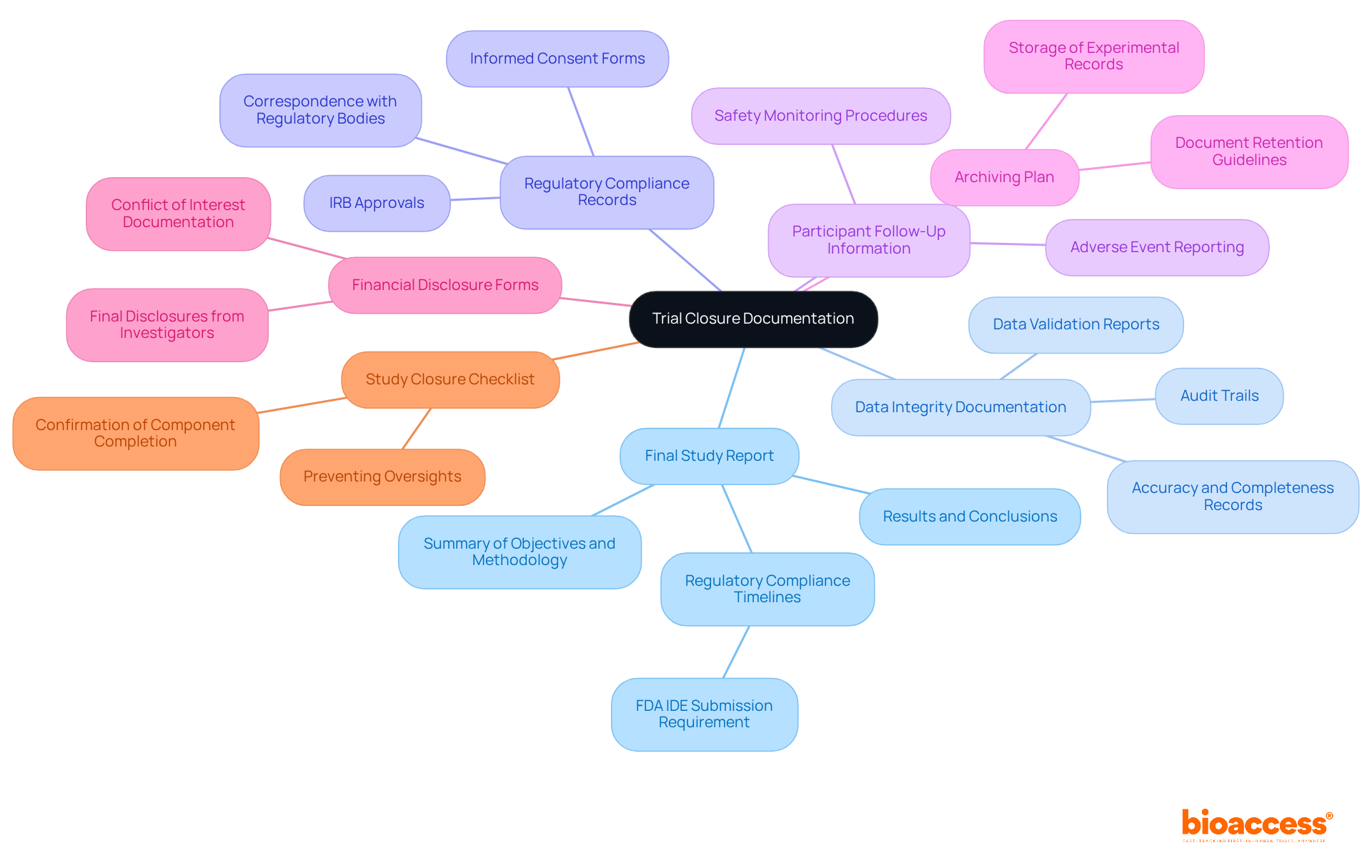
Context and Importance of DMCs in Clinical Trials
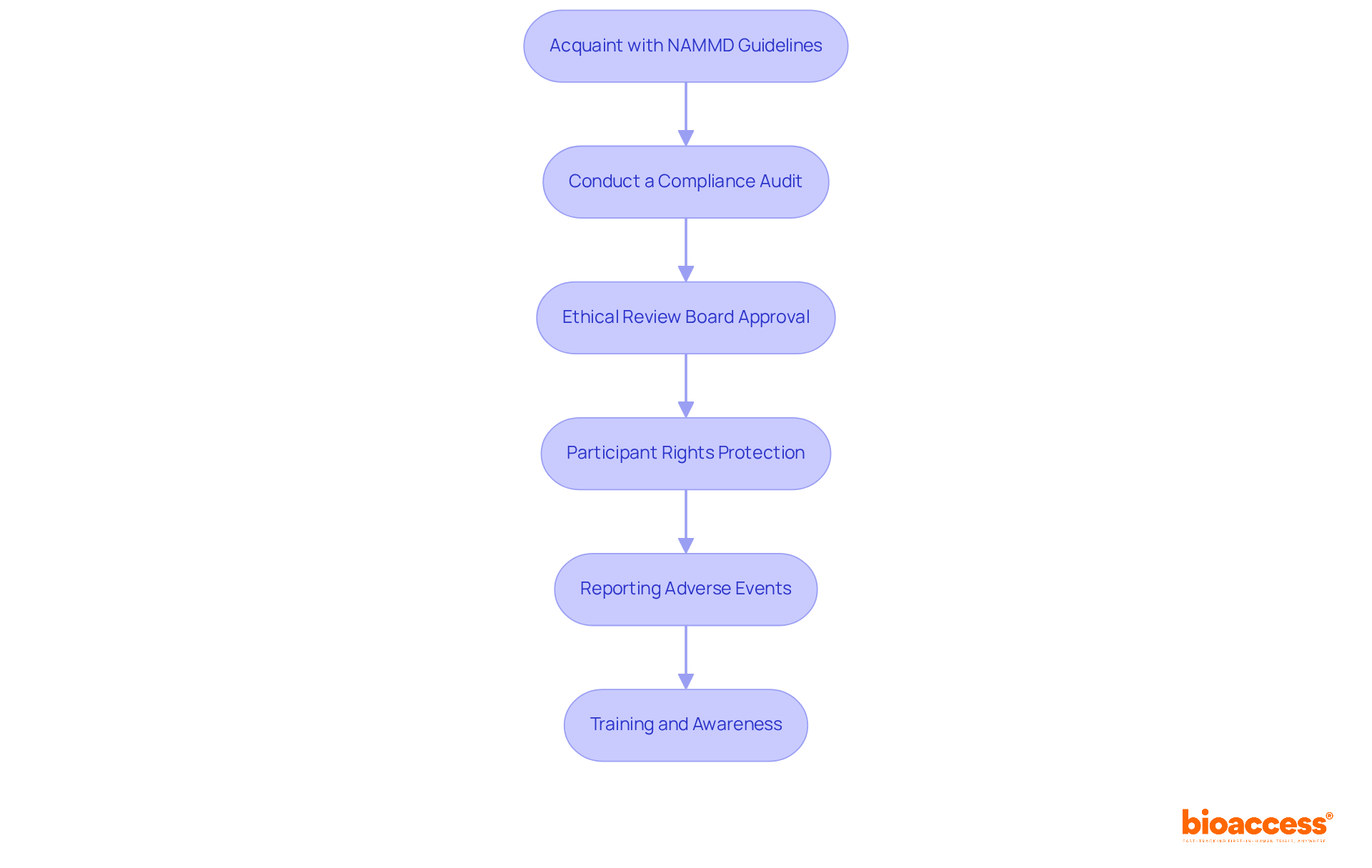
s (DMCs) play a crucial role in due to the necessity for as research becomes more complex and extensive. Regulatory bodies like the FDA advocate for the establishment of s to in . The provides unbiased evaluations of study data, facilitating the early identification and mitigation of . By diligently monitoring interim results, the can offer informed recommendations that prioritize participant safety while ensuring these suggestions are both actionable and confidential.
Their role is particularly critical in high-risk interventions, where the conducts a careful assessment of potential benefits versus harms. Moreover, the must have access to all pertinent information to make , and a clearly defined committee charter is vital for outlining standard operating procedures and ensuring effective oversight. The presence of a not only but also , ultimately benefiting both contributors and the broader medical community.
Notably, over half of studies utilizing interim monitoring strategies are halted prematurely, underscoring the effectiveness of s in safeguarding the interests of all involved.
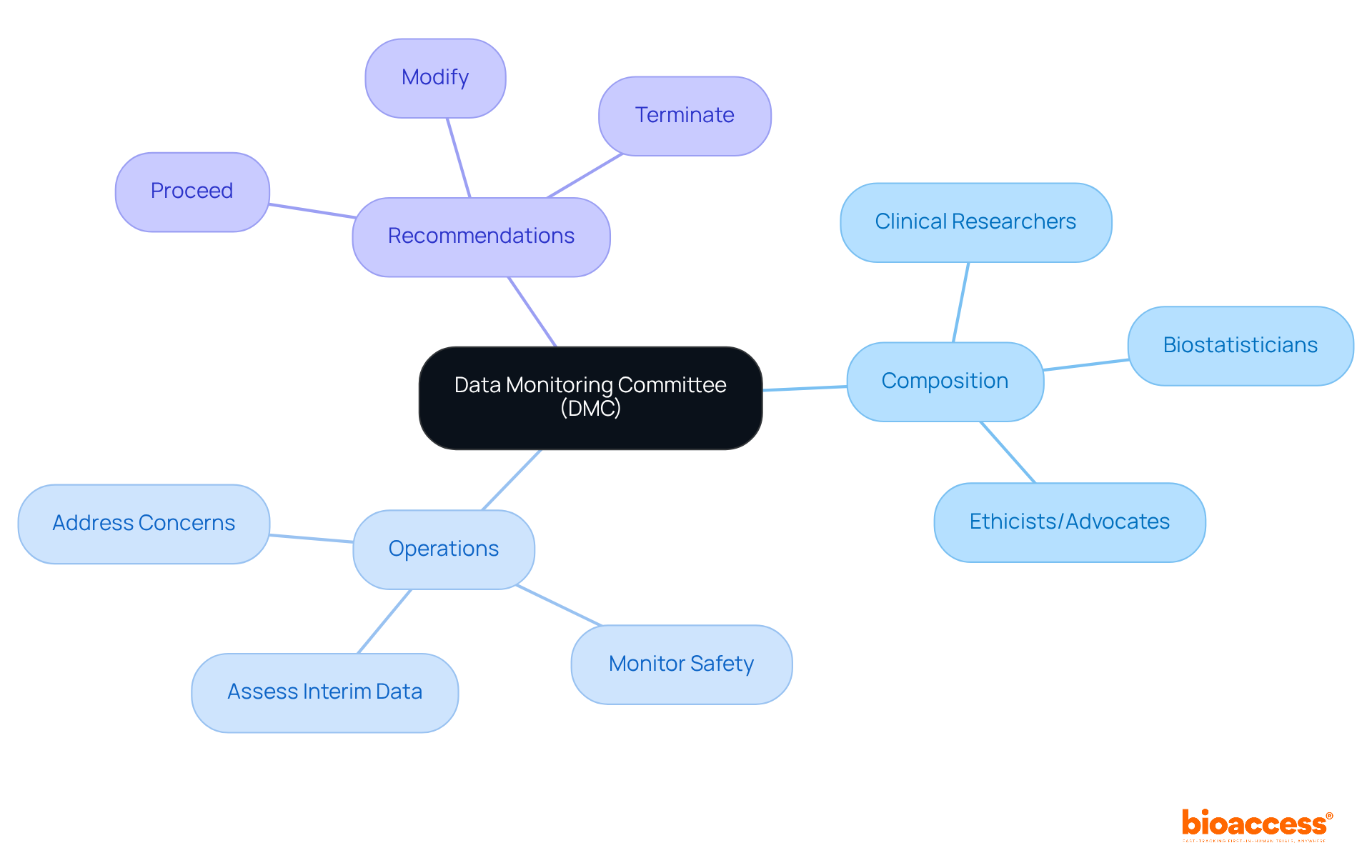
Composition and Operations of Data Monitoring Committees
A (DMC) is essential in , consisting of a multidisciplinary team of 3 to 7 . This team typically includes:
- ers
- Biostatisticians
- Sometimes ethicists or representatives from patient advocacy groups
Such a varied composition is essential for ensuring that diverse perspectives are considered when evaluating study data, particularly in research involving at-risk populations.
The operations of a DMC are governed by a comprehensive charter that delineates its responsibilities, procedures, and decision-making processes. These committees convene at scheduled intervals to:
- Assess unblinded interim data
- Monitor
- Address any emerging concerns
Their independence from the study sponsor is crucial, allowing for free from potential conflicts of interest.
The recommendations made by the can significantly influence the trajectory of a clinical study. They may advise on whether to:
- Proceed
- Modify
- Terminate the research based on the data reviewed
This advisory role is vital for upholding the integrity and . Furthermore, the the establishment of a in trials assessing new interventions, underscoring its importance in and ensuring scientific integrity.
However, interpreting interim data presents challenges due to statistical fluctuations, which DMCs must navigate with care. This complexity highlights the necessity for their , ensuring that the research remains both ethical and scientifically sound.
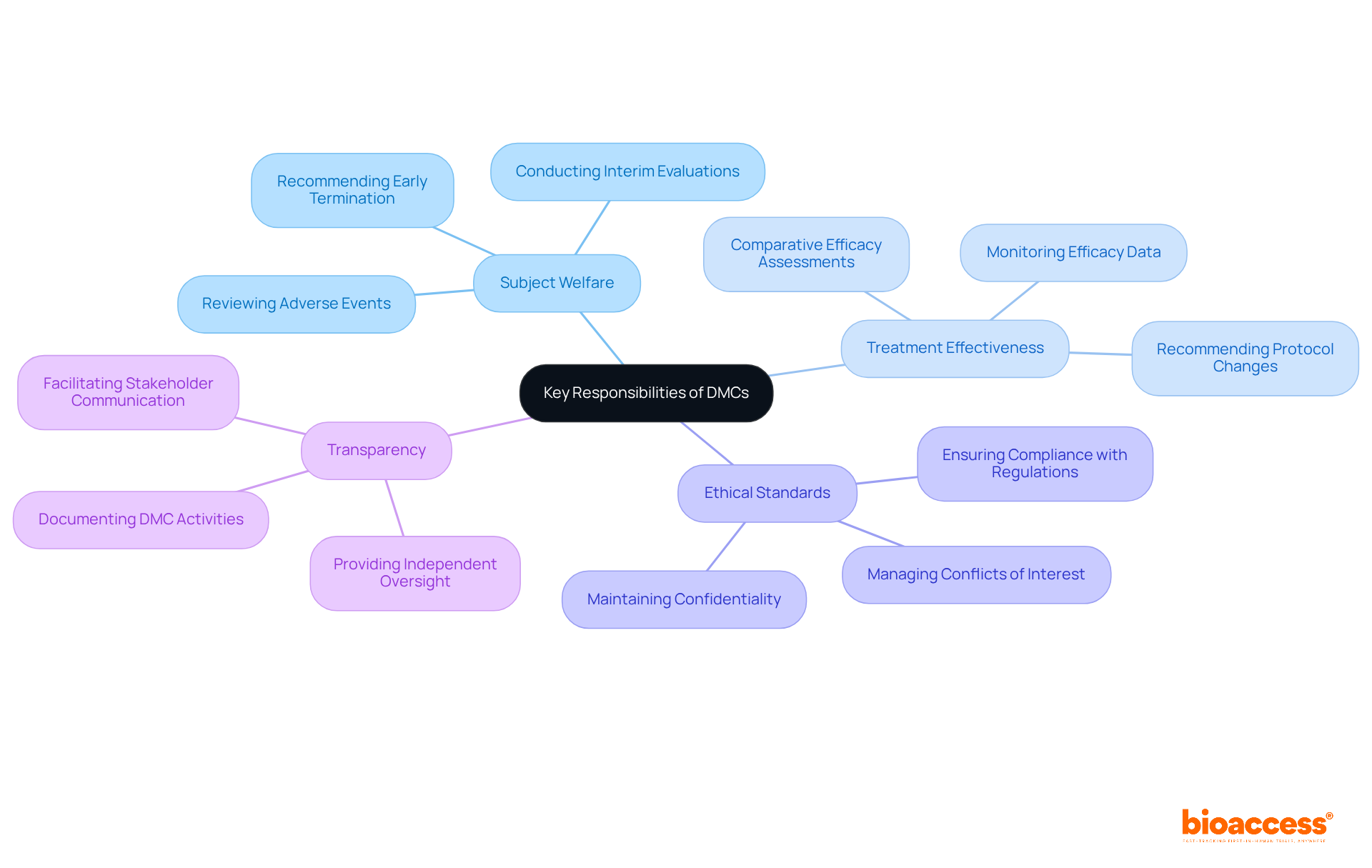
Key Responsibilities and Activities of DMCs
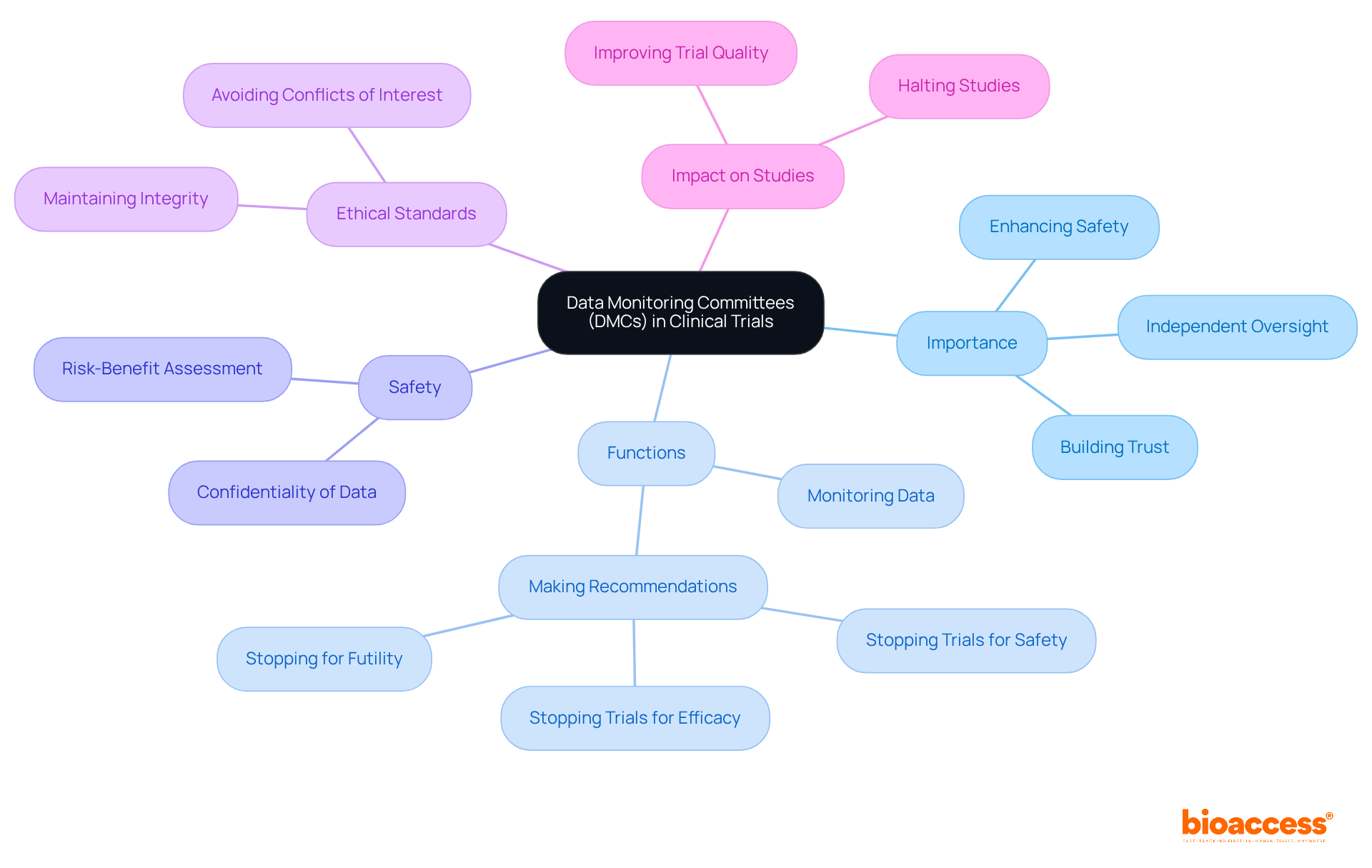
s (DMCs) are essential in , as they are responsible for overseeing subject welfare, evaluating , and ensuring adherence to ethical standards. They conduct at specified intervals, determining whether the benefits of an intervention outweigh its risks. For instance, the consistently reviews and other risk information to identify emerging concerns, which is vital for . When significant safety issues arise, the (DMC) has the authority to , such as adjusting dosages or halting enrollment altogether. Their non-binding suggestions to sponsors, based on thorough evaluations, facilitate informed decisions regarding the continuation or modification of the study.
Moreover, the is essential in fostering transparency and trust within the arena. By providing , they reassure stakeholders – including participants, sponsors, and regulatory bodies – that trials are conducted responsibly. The FDA underscores the importance of the , particularly in research involving substantial risks or vulnerable populations, mandating their establishment under specific conditions. Typically composed of 5-6 independent experts, including at least one medical doctor with expertise in the relevant therapeutic area, the ensures that a diverse range of knowledge informs their evaluations.
Statistics indicate that the (DMC) performs interim evaluations to monitor both protection and effectiveness, with a lower threshold for halting studies for safety than for efficacy. This proactive approach enables management companies to identify potential risks early, ensuring that participant safety remains the primary focus throughout the research process. Furthermore, these management companies must maintain independence from sponsors to prevent conflicts of interest, which is crucial for upholding the integrity of their operations. Overall, the structured activities of the , including the necessity for a comprehensive DMC charter outlining responsibilities and procedures, are vital for preserving the integrity of and protecting the interests of all stakeholders involved.
Conclusion
In the realm of clinical research, the data monitoring committee (DMC) stands as a crucial guardian, ensuring that participant safety and ethical standards are upheld throughout the study process. Their independent oversight and expert evaluations not only guide clinical trials but also significantly influence outcomes, reinforcing the integrity of the research.
This article has explored the multifaceted responsibilities and operations of DMCs, emphasizing their essential role in monitoring participant welfare, evaluating treatment effectiveness, and ensuring compliance with regulatory standards. Composed of diverse experts from various fields, these committees enhance their capability to provide unbiased assessments. Their proactive approach to identifying risks and recommending necessary adjustments underscores their significance in maintaining the ethical conduct of clinical trials.
As the landscape of clinical research evolves, the importance of data monitoring committees cannot be overstated. Their work not only protects participants but also fosters trust in the research process among stakeholders. Embracing a collaborative approach and understanding the critical functions of DMCs will be essential for advancing clinical studies and ensuring that the highest standards of safety and ethics are consistently met.
Frequently Asked Questions
What is a Data Monitoring Committee (DMC)?
A Data Monitoring Committee (DMC), also known as a Data Safety Monitoring Board (DSMB), is an independent group of experts that oversees the well-being and effectiveness of clinical studies by evaluating interim data to ensure ethical conduct and participant safety.
Who comprises a Data Monitoring Committee?
A DMC is made up of a diverse array of professionals, including clinicians, biostatisticians, and specialists in relevant fields, who collectively assess the progress of clinical studies.
What are the main responsibilities of a DMC?
The main responsibilities of a DMC include conducting regular evaluations of interim data, assessing study progress, and providing recommendations on whether to continue, modify, or terminate research based on their analyses.
Why are DMCs significant in clinical research?
DMCs are significant because they identify safety issues, ensure compliance with regulatory standards, and their recommendations can profoundly influence study outcomes, making them a cornerstone of responsible clinical research.
How does the role of DMCs evolve with the Medtech landscape?
As the Medtech landscape evolves, the role of DMCs becomes increasingly vital in addressing key challenges, highlighting the importance of collaboration among stakeholders to navigate complexities effectively.
What is the importance of collaboration in the context of DMCs?
Collaboration among stakeholders is essential for effectively navigating the complexities of clinical research and for fostering an understanding of the critical functions of DMCs to advance clinical studies.
List of Sources
- Define Data Monitoring Committee (DMC)
- Seven Roles of a Data Monitoring Committee in Clinical Trials | PPD (https://ppd.com/blog/the-top-seven-things-to-understand-about-data-monitoring-committee-in-clinical-trials)
- Roles of Statisticians in Data Safety Monitoring Boards (DSMBs) and Interim Analysis (https://phuse.global/Communications/PHUSE_Blog/roles-of-statisticians-in-data-safety-monitoring-boards-dsm-bs-and-interim-analysis)
- clinicaltrialsalliance.org.au (https://clinicaltrialsalliance.org.au/statistical-requirements-for-a-data-safety-monitoring-board-dsmb)
- What is a Data Monitoring Committee? (https://advarra.com/blog/what-is-a-data-monitoring-committee)
- Data Monitoring Committees – CTTI (https://ctti-clinicaltrials.org/about/ctti-projects/data-monitoring-committees)
- Context and Importance of DMCs in Clinical Trials
- Data Monitoring Committee (DMC): Roles in Clinical Trials Explained (https://ccrps.org/clinical-research-blog/data-monitoring-committee-dmc-roles-in-clinical-trials-explained)
- Data Monitoring Committees in Practice | Applied Clinical Trials Online (https://appliedclinicaltrialsonline.com/view/data-monitoring-committees-practice)
- Data monitoring committees: Promoting best practices to address emerging challenges – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC5380168)
- Importance and role of independent data monitoring committees (IDMCs) in oncology clinical trials (https://bmjopen.bmj.com/content/11/10/e047294)
- Data Monitoring Committees — Expect the Unexpected | NEJM (https://nejm.org/doi/full/10.1056/NEJMra1510066)
- Composition and Operations of Data Monitoring Committees
- Data monitoring committee – Wikipedia (https://en.wikipedia.org/wiki/Data_monitoring_committee)
- Data Monitoring Committee – Clinical Research Explained | VIARES (https://viares.com/blog/clinical-research-explained/data-monitoring-committee)
- What is a Data Monitoring Committee? (https://advarra.com/blog/what-is-a-data-monitoring-committee)
- Establishing a data monitoring committee for clinical trials – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC4118004)
- Key Responsibilities and Activities of DMCs
- Seven Roles of a Data Monitoring Committee in Clinical Trials | PPD (https://ppd.com/blog/the-top-seven-things-to-understand-about-data-monitoring-committee-in-clinical-trials)
- The Role of Data Monitoring Committees (DMC) in Clinical Trials (https://quanticate.com/blog/data-monitoring-committees-clinical-trials)
- Key Insights on Data Monitoring Committees: Summarizing the FDA Guidance (https://phastar.com/knowledge-centre/blogs/key-insights-on-data-monitoring-committees-summarizing-the-fda-guidance)
- Responsibilities of Data Monitoring Committees: Consensus Recommendations | WCG (https://wcgclinical.com/insights/dmc-best-practices)