Introduction
The European Medicines Agency (EMA) serves as a pivotal institution in the healthcare landscape of Europe, overseeing the safety and efficacy of medicinal products across its member countries. This article explores the profound impact of the EMA on clinical research, emphasizing how its regulatory frameworks not only streamline approval processes but also foster innovation in medical studies.
However, navigating the complexities of compliance presents a dual-edged sword; while it poses challenges for researchers, it simultaneously opens doors to new opportunities for enhancing research efficiency.
How can clinical researchers leverage the EMA’s guidelines to optimize their studies while overcoming the hurdles of diverse national regulations?
Define the European Medicines Agency and Its Role in Clinical Research
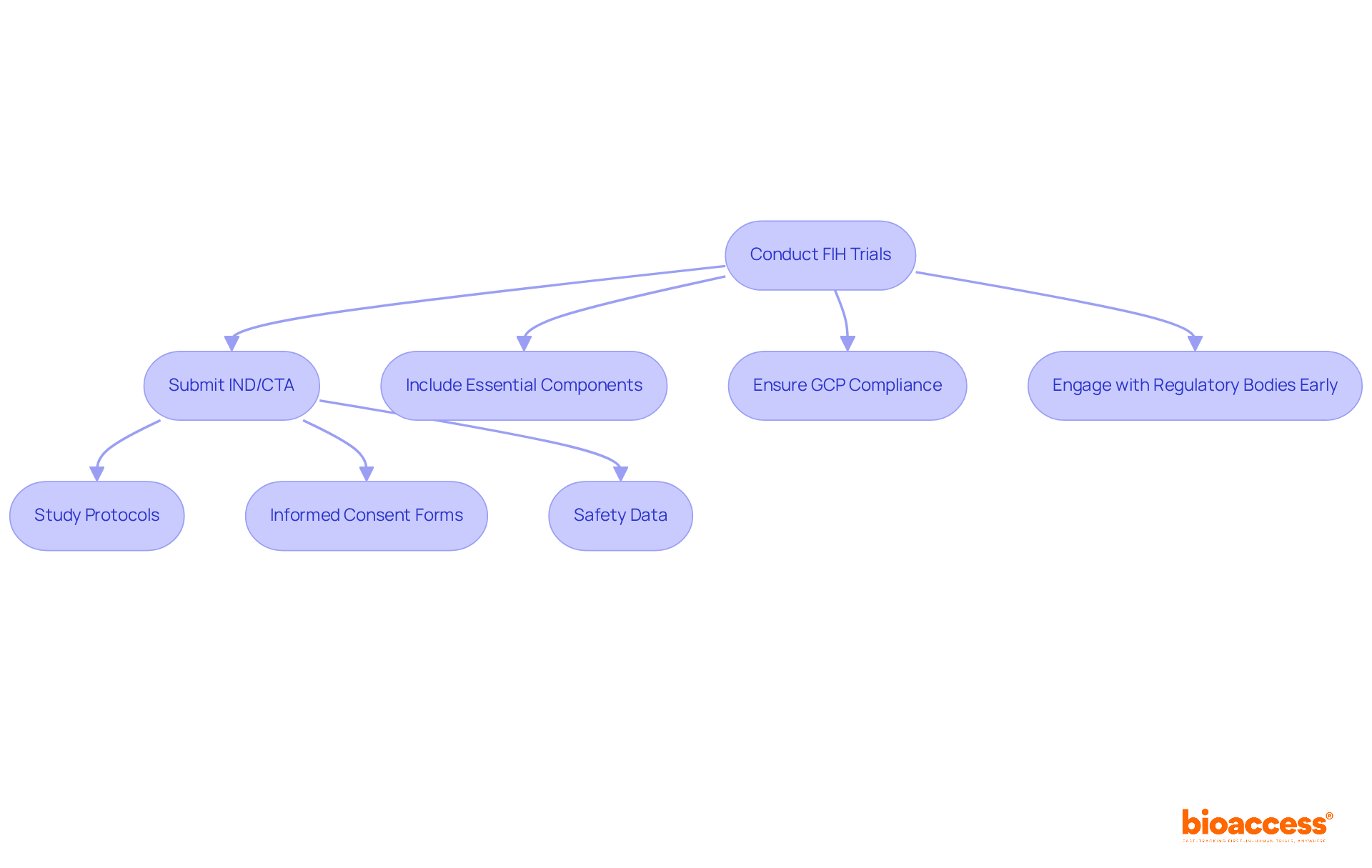
The (EMA), which serves as a cornerstone of the healthcare framework in countries, has been overseeing the , supervision, and since its establishment in 1995. Its primary mission is to ensure that medicinal products are safe, effective, and of high quality, thereby safeguarding across the countries. The EMA assesses , coordinating thorough s that comply with international standards of good practice in medicine. This alignment of regulatory standards not only simplifies the approval process but also fosters a cooperative atmosphere that enhances medical research and innovation.
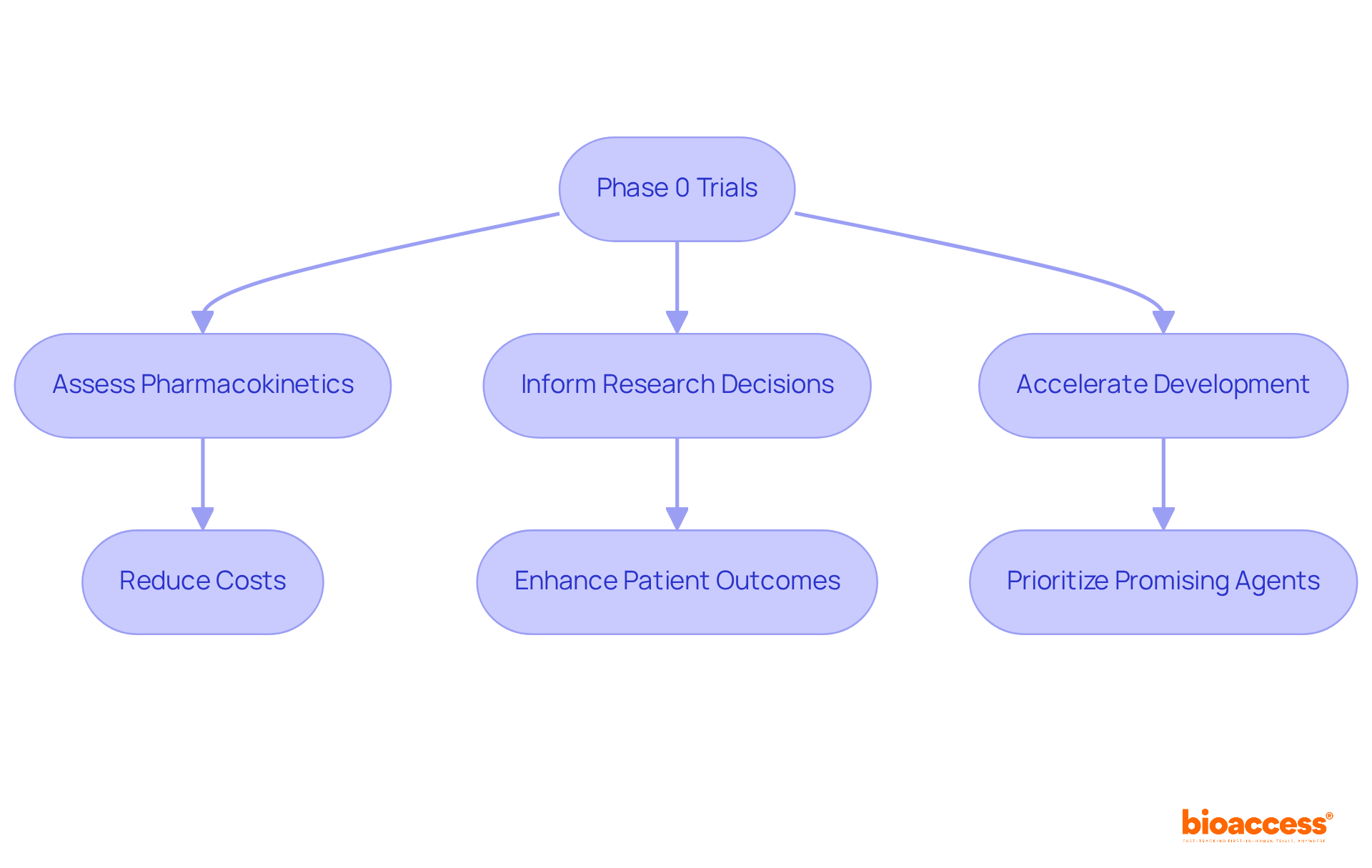
The influence of the EMA on is significant. By providing guidance to medicine developers and facilitating pre-submission meetings, the agency improves application quality and reduces delays. Consequently, the EMA has released study findings for more than 100 recently evaluated medicines, ensuring transparency and public examination. This commitment to transparency is further exemplified by the (CTIS), which serves as a single access point for study data submission and evaluation, thereby improving the efficiency of the oversight process.
Moreover, the EMA’s role transcends oversight; it actively supports by evaluating whether the benefits of a medicine outweigh its risks for specific patient groups. This assessment is vital, particularly for life-threatening diseases where treatment options may be scarce. The agency’s collaborative approach, which includes healthcare professionals and patients in , guarantees that diverse perspectives are considered, ultimately leading to .
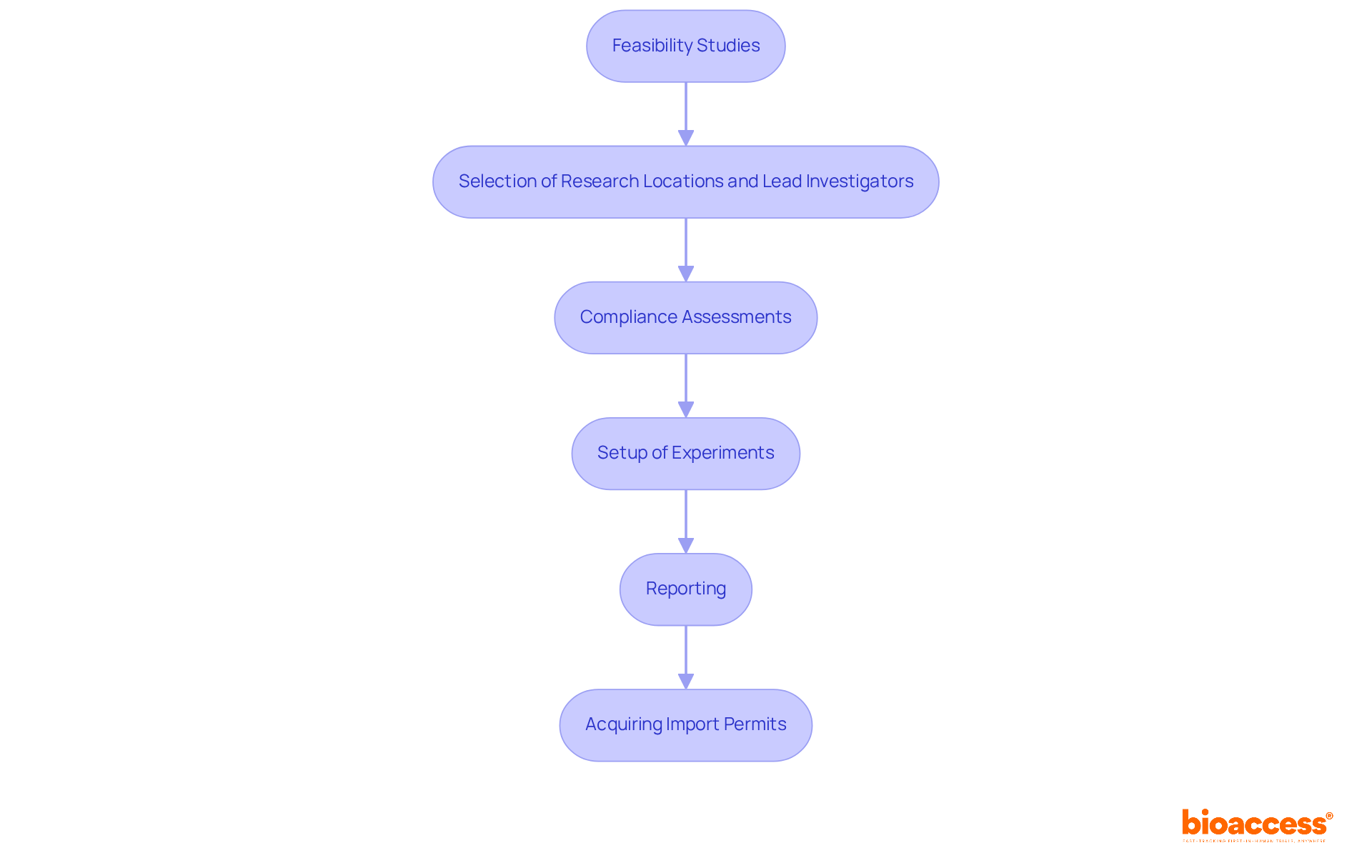
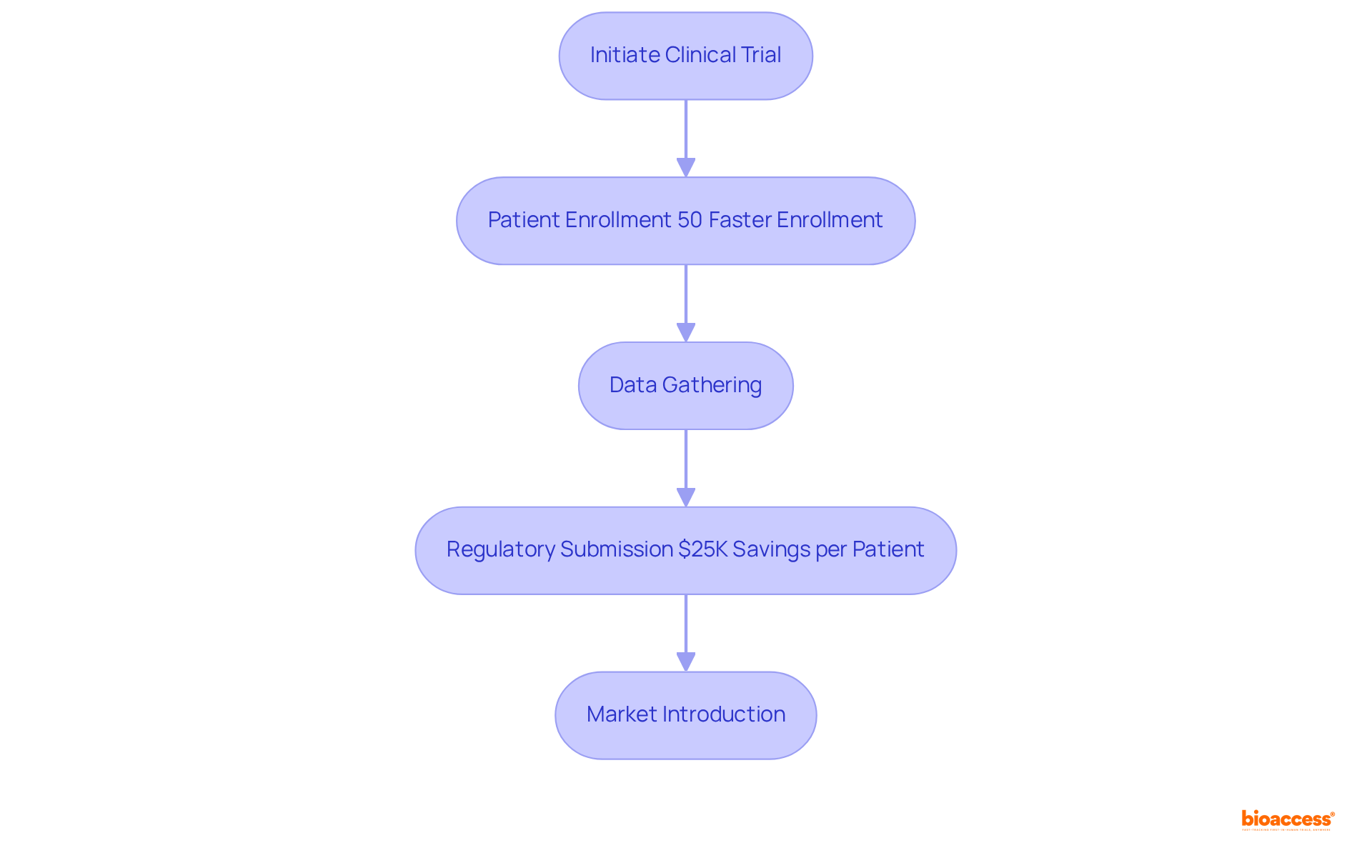
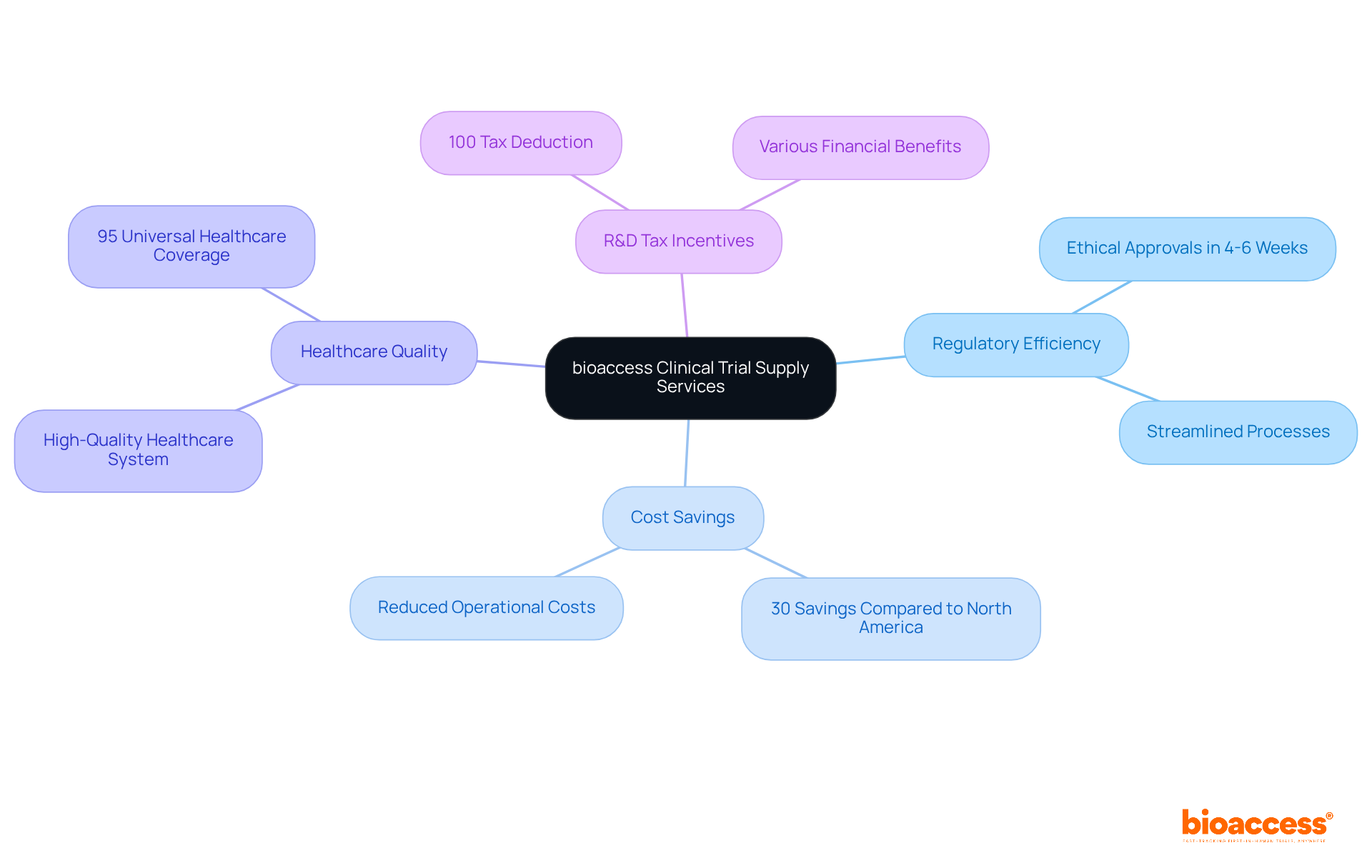
In the realm of research studies, companies like to enhance their service offerings, including feasibility assessments, compliance evaluations, study setup, and project management. This comprehensive strategy enables bioaccess® to enroll 50% faster than Western sites, achieving significant cost savings of $25K per patient. The impact of medtech research on local economies is substantial, contributing to job creation, economic growth, and improved healthcare outcomes while effectively managing the compliance environment.
In conclusion, the EMA’s essential functions in medicine evaluation and safety monitoring not only facilitate the development of innovative therapies but also play a crucial role in enhancing in countries. Its initiatives, such as the ACT EU, aim to modernize research practices while ensuring participant protection and data integrity, further solidifying the EMA’s status as a leader in the global healthcare landscape.
Explore EMA Countries and Their Regulatory Frameworks
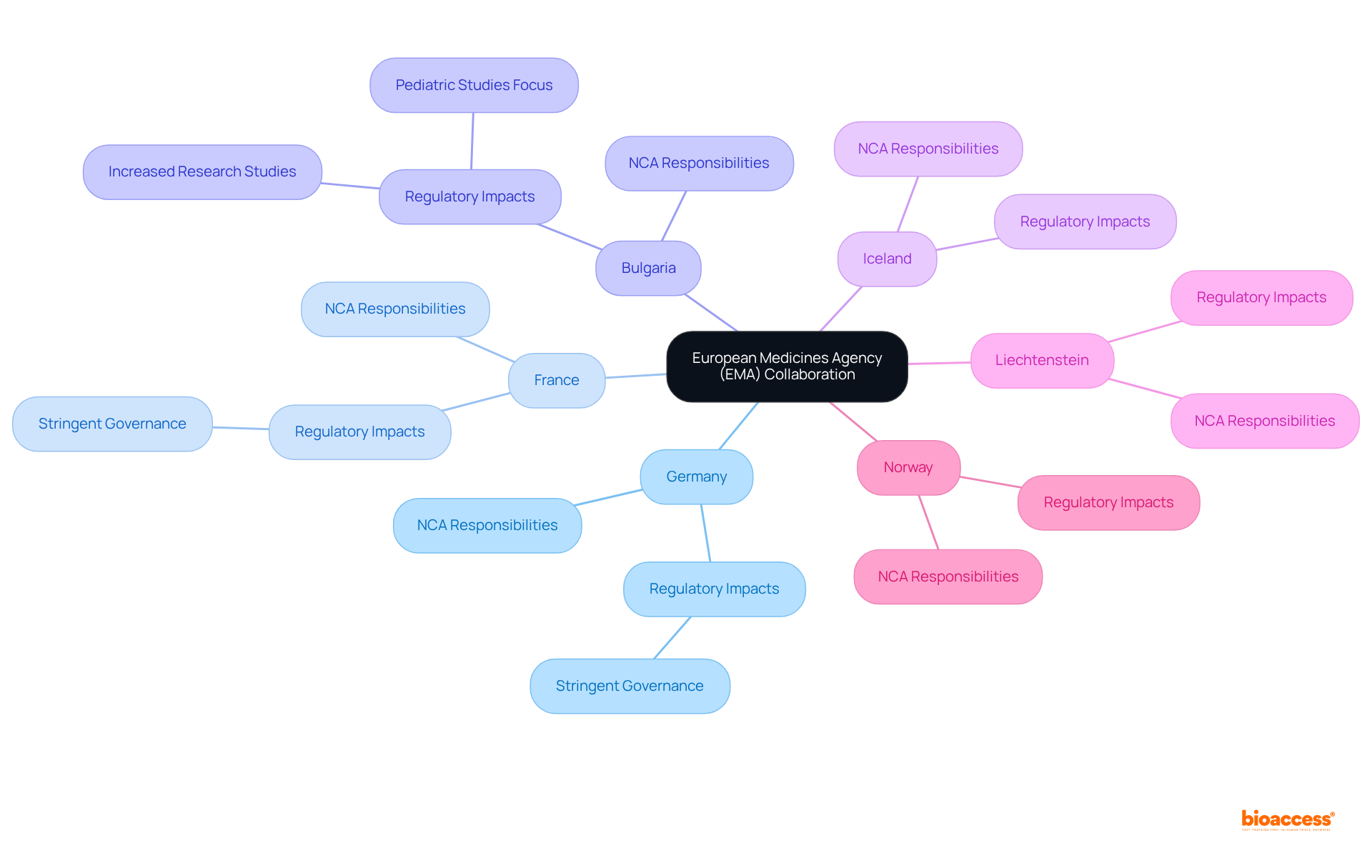
The (EMA) collaborates with the authorities of the countries, which include the 27 EU member states, along with Iceland, Liechtenstein, and Norway, to establish a robust network for medicine oversight. Each country is governed by its own national competent authority (NCA), which holds the responsibility for the authorization of medicinal products. While the EMA sets overarching guidelines, individual NCAs may impose specific requirements that reflect national healthcare priorities and needs.
This diversity of regulations significantly impacts , approval schedules, and . For example:
- Germany and France are , which can affect the feasibility and execution of research studies.
- In contrast, countries like Bulgaria have made strides in enhancing their , as evidenced by a of the EU (EC) 536/2014.
- This regulation has bolstered transparency and safety in research processes, positioning Bulgaria as an attractive location for sponsors.
- Moreover, Bulgaria’s prominence in pediatric studies, ranking second among the EU-EECs, highlights the nation’s .
Ultimately, the interplay between EMA guidelines and national regulations in countries shapes the , necessitating a nuanced understanding of each nation’s regulatory framework to optimize research strategies.
Analyze the Impact of EMA Regulations on Clinical Research Efficiency
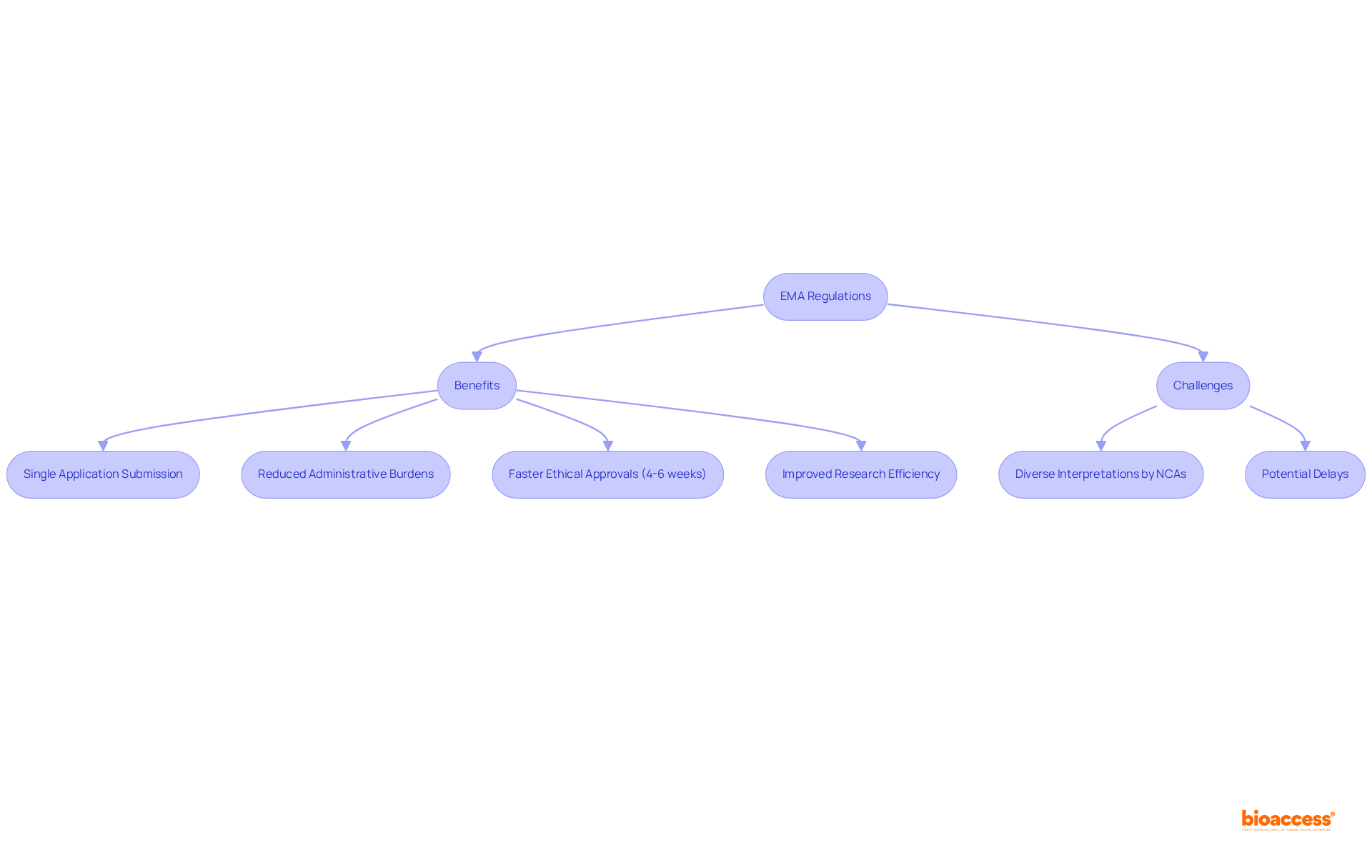
The regulations from the are pivotal in throughout Europe. The (CTR) was established to simplify and standardize the approval process for research studies among member states. By allowing sponsors to submit a single application for multiple , the CTR significantly reduces administrative burdens, resulting in . For example, , marking a substantial improvement over previous processes.
Despite these advancements, challenges remain, particularly due to the diverse interpretations of regulations by various (NCAs) in , which can result in delays. Nevertheless, the EMA’s dedication to transparency and collaboration cultivates an innovative environment, enabling researchers to navigate the more effectively.
Statistics indicate that the CTR has led to a , enhancing the overall effectiveness of research studies. By 2025, the full implementation of the CTR is expected to refine research procedures, making Europe a more attractive location for conducting medical studies. This oversight structure not only accelerates timelines but also encourages the , projected to reach approximately USD 886.5 billion by 2032, reflecting the increasing demand for effective research solutions.
Identify Challenges and Opportunities in EMA Compliance for Clinical Researchers
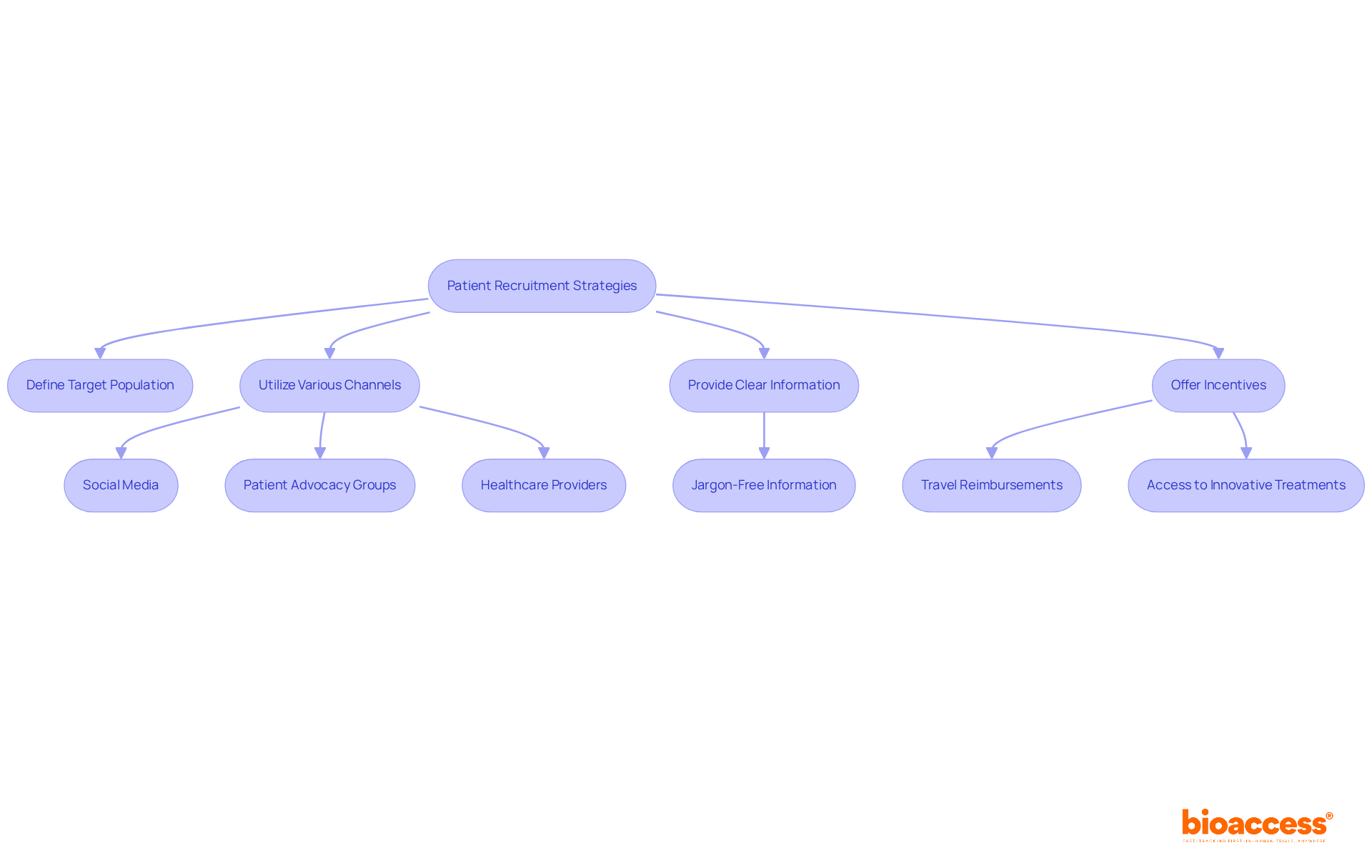
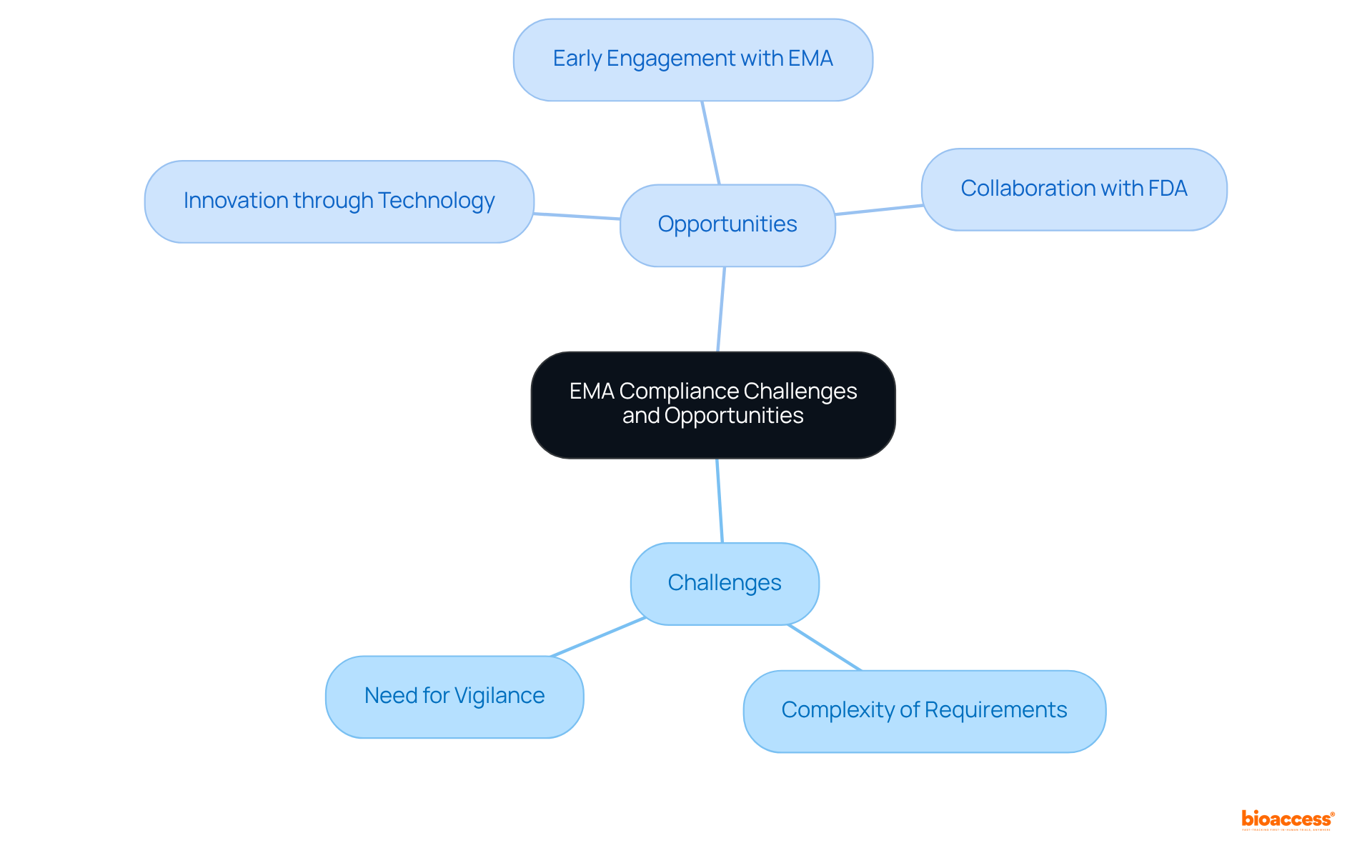
Navigating compliance with presents both significant challenges and opportunities for . The complexity of , which can vary widely across nations, stands as a primary obstacle. Researchers must remain vigilant regarding the latest guidelines to ensure their studies adhere to and regulatory expectations. Yet, these challenges also pave the way for innovation. By harnessing technology and data analytics, researchers can , enhance patient recruitment strategies, and improve . Engaging with the EMA early in the research process can yield valuable , fostering more efficient study designs and successful outcomes. Furthermore, the on has improved inspection coverage and understanding of procedures, providing a framework for researchers to optimize their compliance strategies.
Conclusion
The European Medicines Agency (EMA) plays a pivotal role in shaping the landscape of healthcare and clinical research across its member countries. By establishing rigorous standards for the evaluation and monitoring of medicines, the EMA not only ensures the safety and efficacy of medical products but also fosters an environment conducive to innovation and research. This framework is essential for maintaining public health and enhancing the quality of care available to patients in EMA countries.
Key insights throughout the article highlight the EMA’s influence on research efficiency, particularly through the implementation of the Clinical Studies Regulation (CTR), which streamlines approval processes and reduces administrative burdens. The collaboration between the EMA and national competent authorities ensures that diverse regulatory frameworks are harmonized, allowing for a more effective approach to clinical trials. Furthermore, the involvement of stakeholders, including healthcare professionals and patients, underscores the importance of a multifaceted perspective in evaluating medical products.
As the EMA continues to adapt and modernize its practices, there lies an opportunity for researchers to leverage these regulations to enhance their study designs and compliance strategies. Engaging with the EMA early in the research process can unlock valuable insights, paving the way for innovative solutions that address the complexities of clinical research. The ongoing evolution of the EMA’s framework not only promises to improve the efficiency of clinical trials but also reinforces the significance of regulatory compliance in advancing healthcare outcomes across Europe.
Frequently Asked Questions
What is the European Medicines Agency (EMA)?
The European Medicines Agency (EMA) is a regulatory agency established in 1995 that oversees the scientific evaluation, supervision, and safety monitoring of medicines in European Medicines Agency countries. Its primary mission is to ensure that medicinal products are safe, effective, and of high quality.
What role does the EMA play in clinical research?
The EMA assesses marketing authorization submissions, coordinates scientific evaluations, and ensures compliance with international standards. It provides guidance to medicine developers, facilitates pre-submission meetings, and improves application quality, which helps reduce delays in the approval process.
How does the EMA ensure transparency in its operations?
The EMA promotes transparency by releasing study findings for over 100 recently evaluated medicines and through the Clinical Studies Information System (CTIS), which serves as a single access point for study data submission and evaluation.
How does the EMA support public health?
The EMA evaluates whether the benefits of a medicine outweigh its risks for specific patient groups, particularly for life-threatening diseases. It collaborates with healthcare professionals and patients to ensure diverse perspectives are considered in the evaluation process.
What is the impact of the EMA on research study success?
The EMA significantly influences research study success by improving application quality and reducing delays, thus facilitating the development of innovative therapies and enhancing public health outcomes.
How do companies like bioaccess® utilize the EMA’s framework?
Companies like bioaccess® leverage the EMA’s framework to enhance service offerings such as feasibility assessments, compliance evaluations, study setup, and project management, allowing them to enroll treatment-naive cardiology or neurology cohorts 50% faster than Western sites.
What economic benefits are associated with medtech research influenced by the EMA?
Medtech research contributes to job creation, economic growth, and improved healthcare outcomes while effectively managing the compliance environment.
What initiatives does the EMA have to modernize research practices?
The EMA has initiatives like ACT EU aimed at modernizing research practices while ensuring participant protection and data integrity, reinforcing its role as a leader in the global healthcare landscape.
List of Sources
- Define the European Medicines Agency and Its Role in Clinical Research
- ema.europa.eu (https://ema.europa.eu/en/about-us/what-we-do/authorisation-medicines/how-ema-evaluates-medicines-human-use)
- citoday.com (https://citoday.com/news/european-medicines-agencys-clinical-trials-regulation-takes-effect)
- globalregulatorypartners.com (https://globalregulatorypartners.com/case_studies/case-study-13-new-drug-registration-in-usa-china-and-japan)
- Explore EMA Countries and Their Regulatory Frameworks
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC5809349)
- tandfonline.com (https://tandfonline.com/doi/full/10.1080/13102818.2023.2226741)
- clinicaltherapeutics.com (https://clinicaltherapeutics.com/article/S0149-2918(23)00307-7/fulltext)
- Number of clinical trials by year, country, region and income group (https://who.int/observatories/global-observatory-on-health-research-and-development/monitoring/number-of-clinical-trials-by-year-country-who-region-and-income-group)
- europeancommission.medium.com (https://europeancommission.medium.com/quotes-by-jacques-delors-the-architect-of-the-united-europe-we-know-today-d7cc21d708f4)
- Analyze the Impact of EMA Regulations on Clinical Research Efficiency
- Clinical Trials Statistics and Facts (2026) (https://media.market.us/clinical-trials-statistics)
- Quotes About Medical Research (https://changingthepresent.org/pages/quotes-about-medical-research)
- antidote.me (https://antidote.me/blog/what-clinical-trial-statistics-tell-us-about-the-state-of-research-today)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC10173933)
- ema.europa.eu (https://ema.europa.eu/en/news/clinical-trials-regulation-becomes-fully-applicable)
- Identify Challenges and Opportunities in EMA Compliance for Clinical Researchers
- Good clinical practice | European Medicines Agency (EMA) (https://ema.europa.eu/en/human-regulatory-overview/research-development/compliance-research-development/good-clinical-practice)
- Compliance: Overview | European Medicines Agency (EMA) (https://ema.europa.eu/en/human-regulatory-overview/compliance-overview)
- TOP 25 COMPLIANCE QUOTES (of 106) | A-Z Quotes (https://azquotes.com/quotes/topics/compliance.html)
- Key Compliance Statistics & Insights For 2025 | Zluri (https://zluri.com/blog/key-compliance-statistics-and-insights-for-2024)
- indeed.com (https://indeed.com/career-advice/career-development/quotes-on-opportunities)


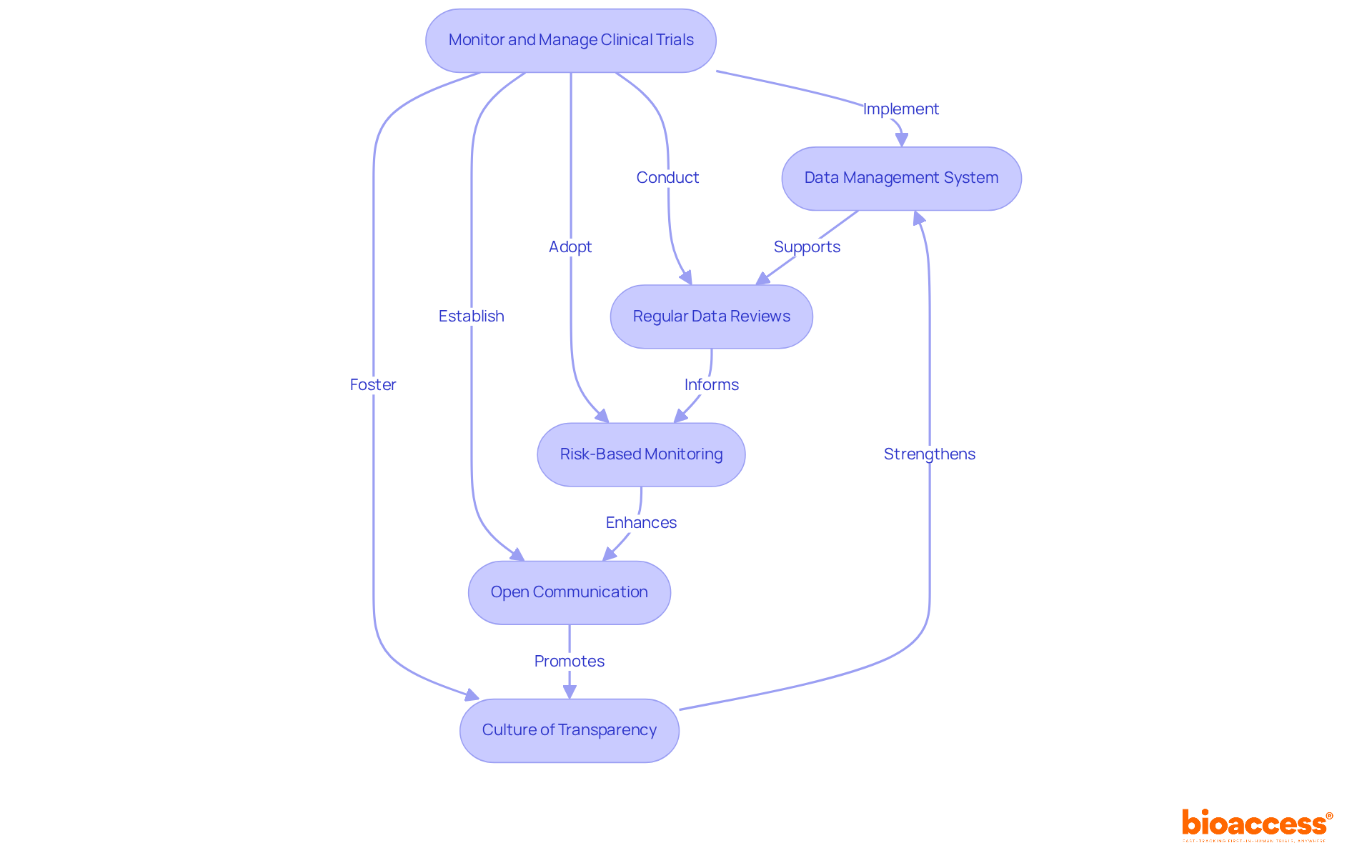
 process. Follow the arrows to see how monitoring and communication lead to safety and compliance. Each box represents a critical step in the post-market surveillance process. Follow the arrows to see how monitoring and communication lead to safety and compliance.](https://images.tely.ai/telyai/jfnquvoo-each-box-represents-a-critical-step-in-the-post-market-surveillance-process-follow-the-arrows-to-see-how-monitoring-and-communication-lead-to-safety-and-compliance.webp)




 — the larger the slice, the greater the savings from that category. Each slice of the pie shows how much each cost factor contributes to the total savings in virtual clinical trials — the larger the slice, the greater the savings from that category.](https://images.tely.ai/telyai/vsfswxcc-each-slice-of-the-pie-shows-how-much-each-cost-factor-contributes-to-the-total-savings-in-virtual-clinical-trials-the-larger-the-slice-the-greater-the-savings-from-that-category.webp)
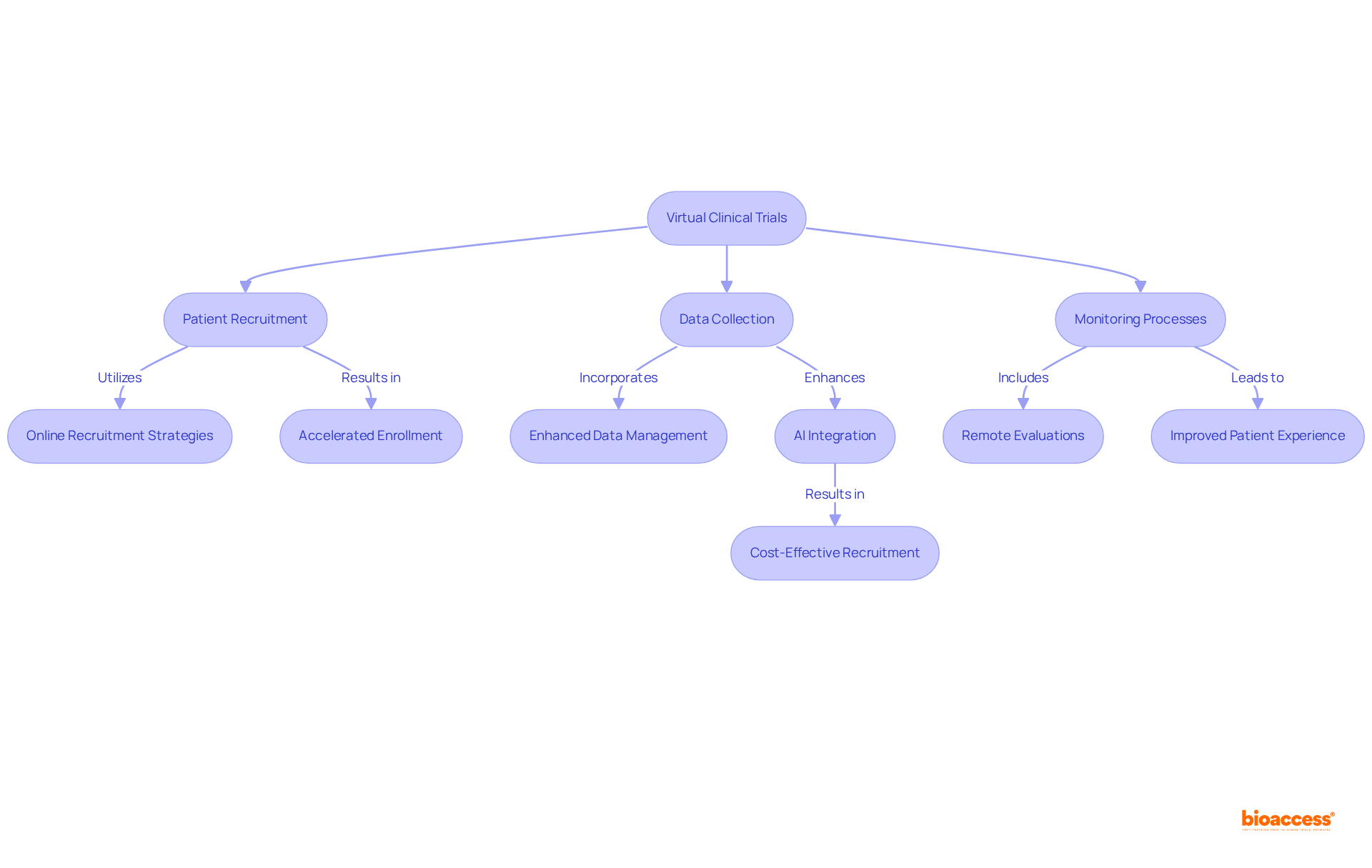


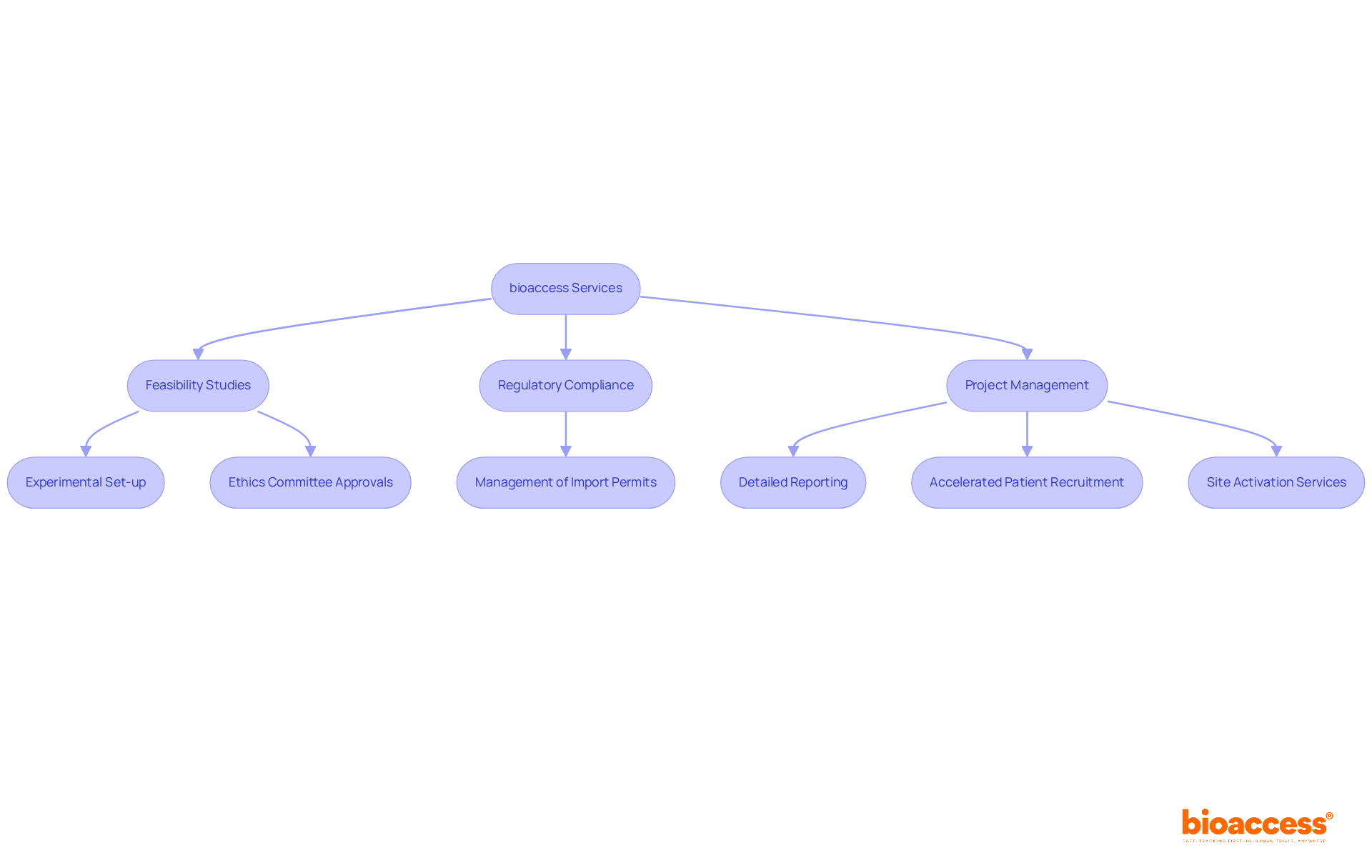
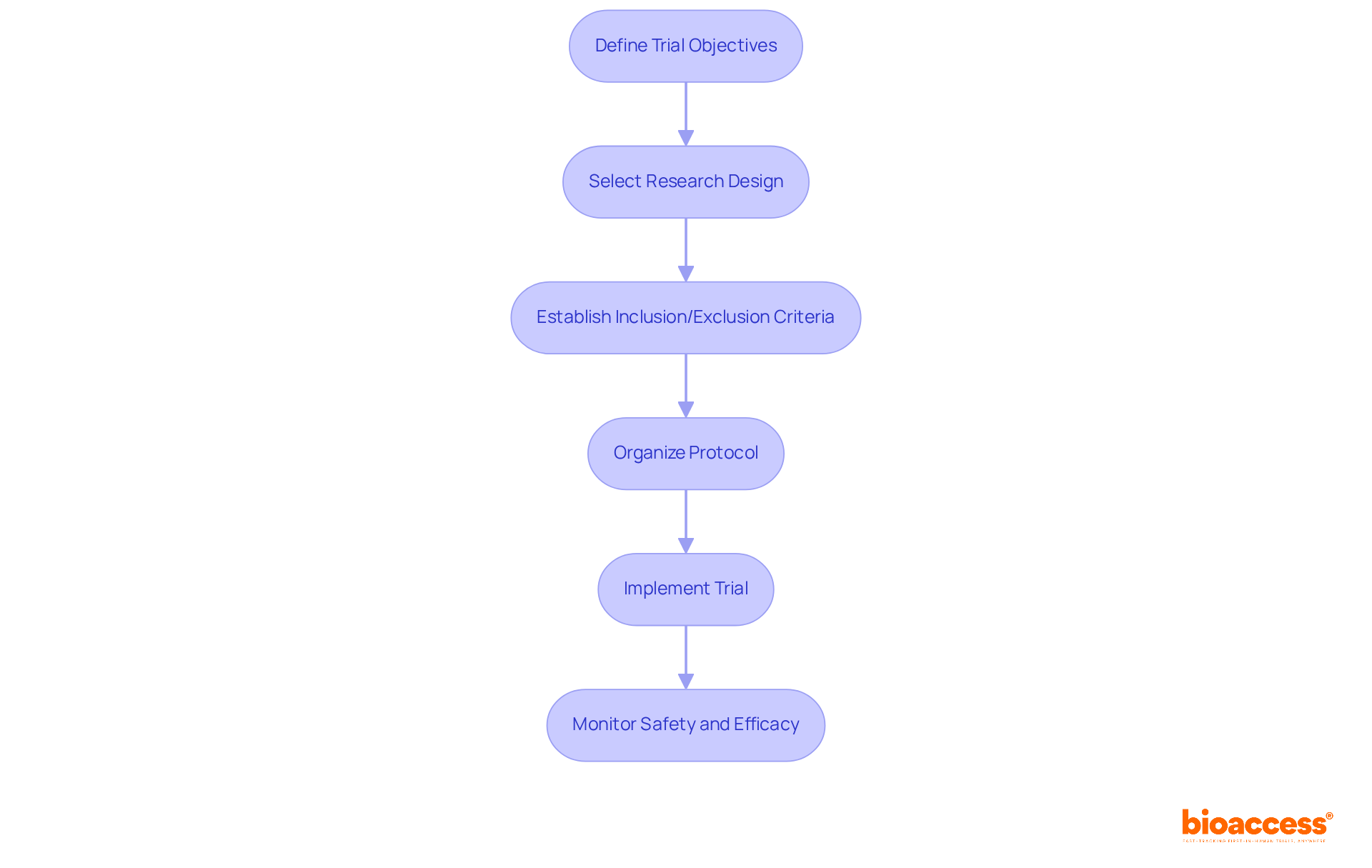
 and study efficiency. Each box shows a step in the clinical study management process. Follow the arrows to see how each step leads into the next, ultimately enhancing patient recruitment and study efficiency.](https://images.tely.ai/telyai/cmiyxnfq-each-box-shows-a-step-in-the-clinical-study-management-process-follow-the-arrows-to-see-how-each-step-leads-into-the-next-ultimately-enhancing-patient-recruitment-and-study-efficiency.webp)