Introduction
The role of a Clinical Research Associate (CRA) has become increasingly vital in the fast-evolving landscape of medical research, serving as the crucial link between sponsors and research sites. Aspiring CRAs have the opportunity to gain a wealth of knowledge regarding the responsibilities, educational pathways, and practical experiences essential for thriving in this competitive field.
But what does it truly take to navigate the complexities of becoming a CRA?
How can one effectively position themselves for success amidst the growing demand for these professionals?
Exploring these essential steps not only illuminates the career path but also reveals the opportunities that await those ready to embark on this journey.
Understand the Role and Responsibilities of a Clinical Research Associate
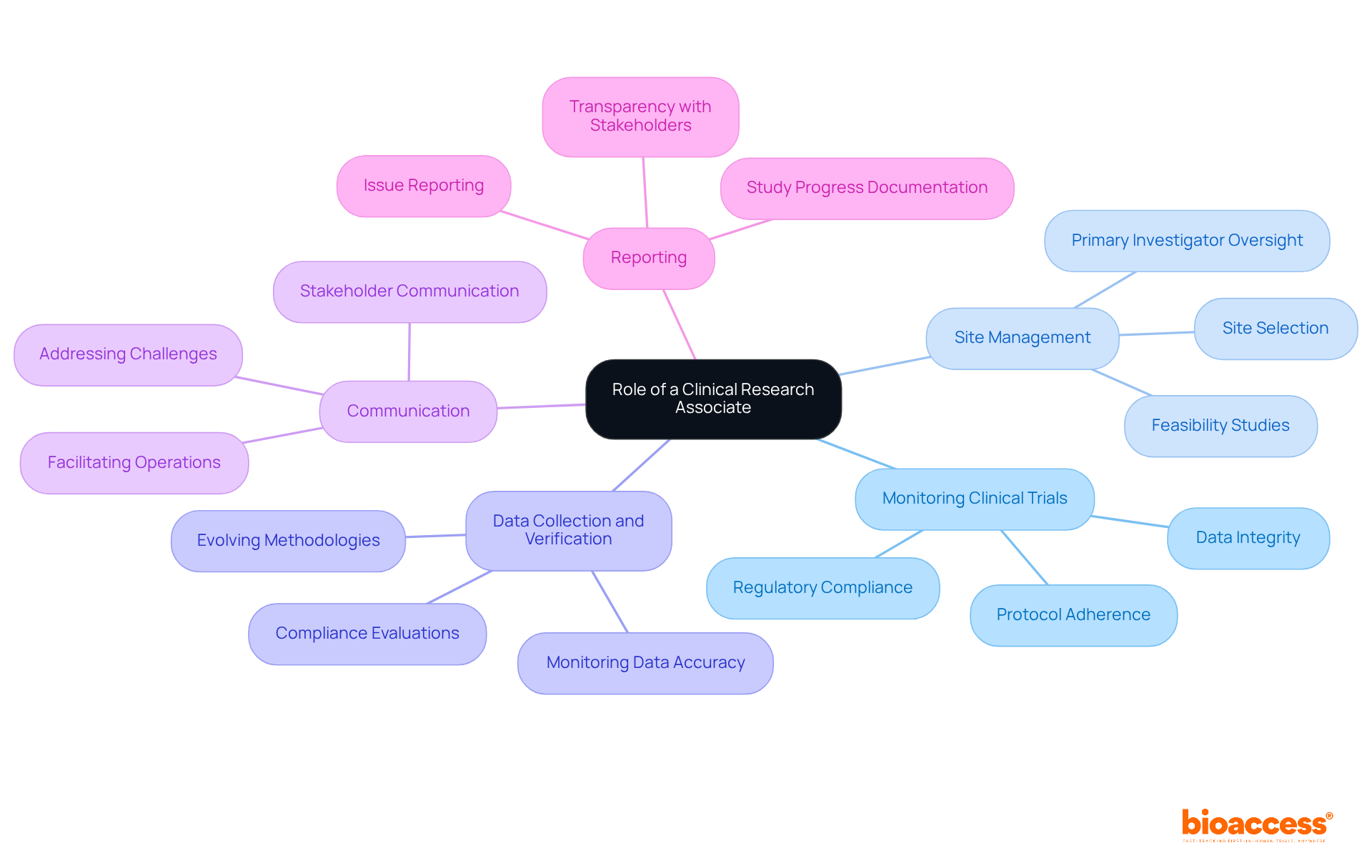
A Clinical Research Associate (CRA) plays a pivotal role in the research process, serving as a vital link between sponsors and research sites. Their key responsibilities encompass several critical areas:
- Monitoring : CRAs ensure that trials comply with protocols, regulatory requirements, and . This adherence is essential for maintaining the integrity of the research.
- Site Management: They are responsible for selecting and overseeing , ensuring that each site is adequately equipped with the necessary resources and trained personnel to conduct the study effectively. This includes performing feasibility studies and selecting primary investigators (PIs), which are crucial for the success of the study.
- Data Collection and Verification: By , clinical research associates guarantee its accuracy and integrity—elements vital for the study’s success. This role is increasingly complex, requiring CRAs to stay informed about evolving research methodologies and technologies, along with to meet country-specific requirements.
- Communication: Maintaining open lines of communication with investigators, sponsors, and regulatory bodies is essential for facilitating smooth operations and addressing challenges that may arise during the study, including regulatory hurdles and recruitment issues.
- Reporting: Clinical research associates prepare comprehensive documents detailing study progress and any encountered issues, providing valuable insights to stakeholders and ensuring transparency throughout the process. This includes reporting on study status, inventory, and adverse events, all of which are critical for project management and monitoring.
In 2025, the , with the clinical trial market expected to surpass 70 billion dollars. Understanding these responsibilities and the associated challenges is crucial for anyone , as it aligns with the skills and interests necessary for success in this dynamic field.
Explore Educational Requirements and Certification Options
To thrive as a Associate (CRA), knowing requires a solid educational foundation and relevant certifications.
Educational Background: Most CRAs possess . Notably, approximately 53% of CRAs hold a bachelor’s degree, while over 34% have . In fact, professionals with master’s degrees earn 93% higher average salaries compared to those with undergraduate qualifications, making advanced education a valuable investment.
Certifications: Pursuing certifications can greatly benefit your career trajectory. Key certifications include:
- : Offered by the Association of Professionals (ACRP), this credential showcases your expertise and dedication to the field, often leading to better job opportunities and advancement.
- : Also from ACRP, this certification is beneficial for those who will collaborate closely with trial sites, enhancing your comprehension of trial management.
- : Completing GCP training is frequently a prerequisite for many CRA positions, ensuring compliance with regulatory standards.
The medical research sector is expected to attain $52.0 billion by 2026, highlighting the increasing need for and the significance of certifications in a competitive market. Moreover, over 40% of interns in medical research transition into permanent positions after their internships, emphasizing the possible routes for aspiring CRAs. will equip you with the knowledge and credentials necessary to understand how to become a associate and excel in the competitive area of research in healthcare.
 with different educational qualifications. A larger slice indicates a higher number of CRAs with that degree. Each slice represents the percentage of Clinical Research Associates with different educational qualifications. A larger slice indicates a higher number of CRAs with that degree.](https://images.tely.ai/telyai/ckymahul-each-slice-represents-the-percentage-of-clinical-research-associates-with-different-educational-qualifications-a-larger-slice-indicates-a-higher-number-of-cr-as-with-that-degree.webp)
Gain Practical Experience Through Internships and Entry-Level Positions
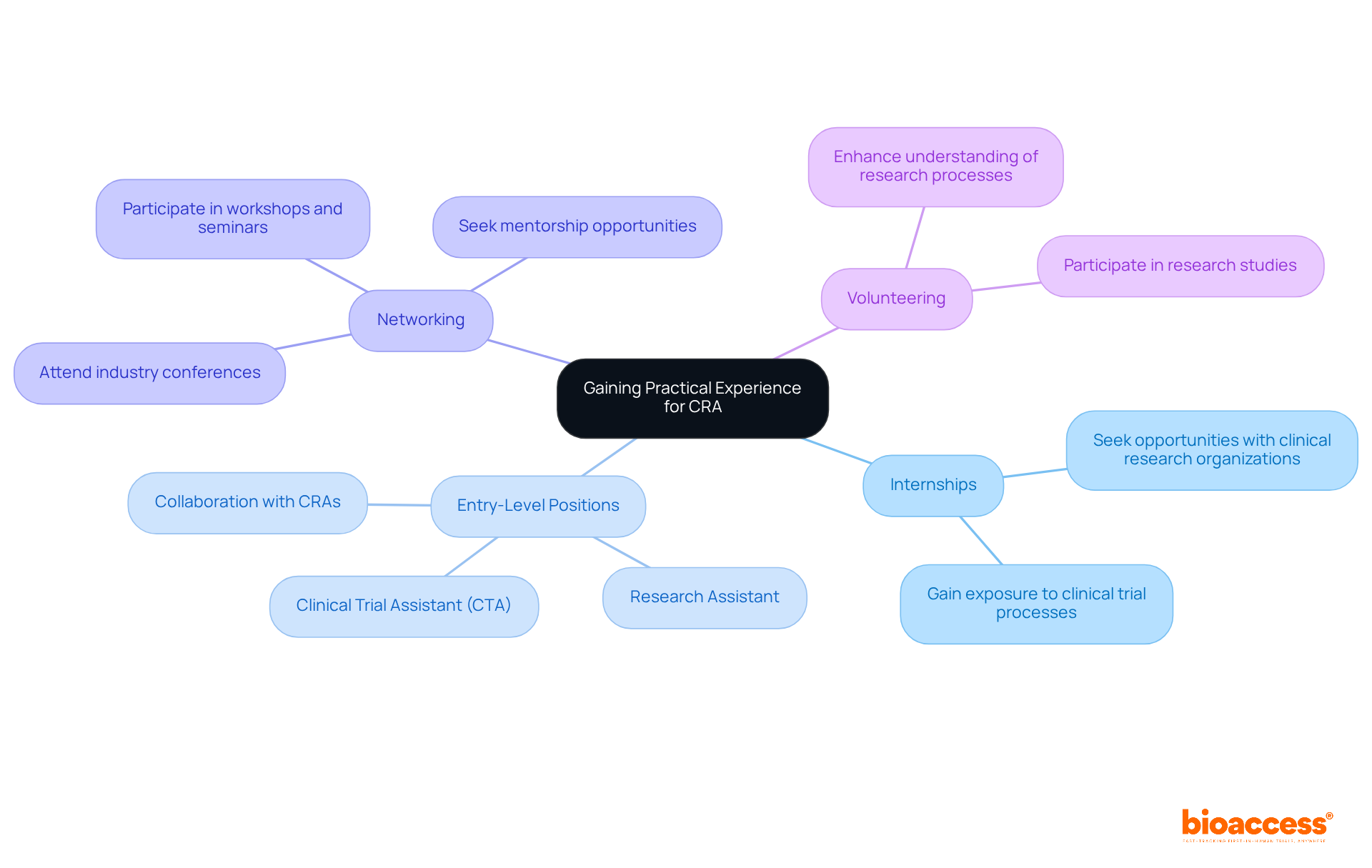
Gaining practical experience is a critical step in learning how to become a (CRA). To navigate this path effectively, consider the following strategies:
- Internships: Seek internship opportunities with , pharmaceutical companies, or academic institutions. These positions provide invaluable exposure to the , significantly enhancing your understanding of the industry.
- : Explore roles such as (CTA) or Research Assistant. Notably, a substantial percentage of s began their careers as s, illustrating a common route into the profession. These roles allow for close collaboration with CRAs, facilitating a deeper understanding of research fundamentals in healthcare.
- Networking: to connect with professionals in the field. Networking can lead to mentorship opportunities and job referrals, both crucial for career advancement.
- Volunteer: If feasible, volunteer for . This hands-on experience enhances your grasp of the processes involved and positions you as a more attractive candidate.
By actively pursuing these experiences, you will establish a robust foundation for understanding how to become a . The medical research market is projected to surpass $80 billion by 2025, underscoring the growing demand for skilled professionals in this field. As one expert noted, “research professionals in the medical field are in high demand with strong competition among employers.
Navigate the Job Application Process and Secure a CRA Position
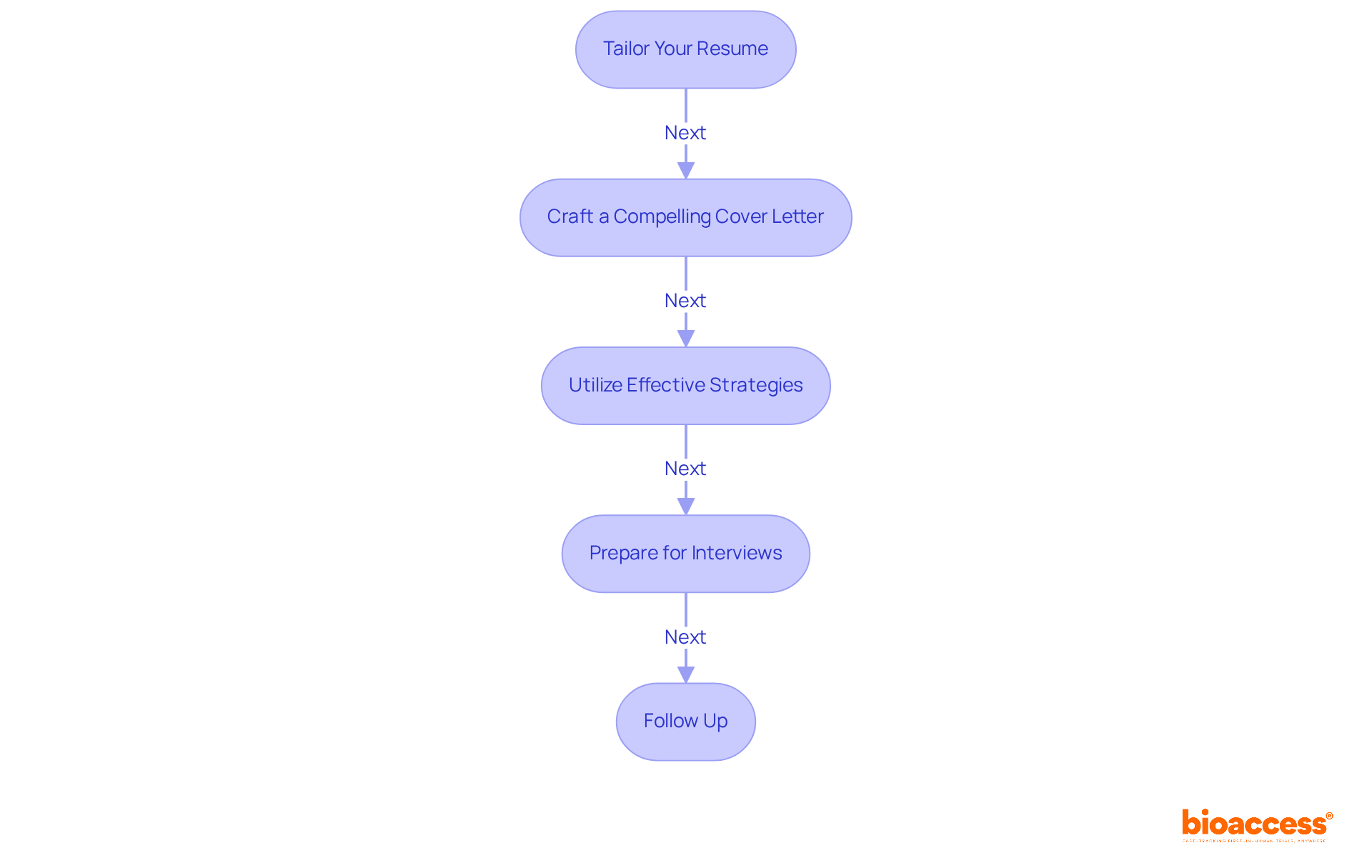
Navigating the job application process is a critical step toward learning how to become a (CRA). With the right approach, you can enhance your application significantly. Follow these :
- Tailor Your Resume: Highlight your relevant education, certifications, and practical experience. Utilize to align your resume with the employer’s needs. Resumes that closely match job titles can .
- Craft a Compelling Cover Letter: Your cover letter should effectively convey your enthusiasm for and clarify why you are a fitting candidate for the role. Personalization is crucial; address it to a specific person if possible and include relevant examples that demonstrate your qualifications. .
- Utilize Effective Strategies: Incorporate quantifiable achievements in your cover letter to showcase your impact. For instance, mention specific projects you contributed to and the outcomes achieved. This approach can significantly enhance your appeal to hiring managers, as .
- : Research common interview questions for CRAs and practice your responses. Be ready to discuss your experience, problem-solving abilities, and knowledge of . Demonstrating your knowledge and enthusiasm can set you apart from other candidates.
- : After interviews, send a thank-you email to express your appreciation for the opportunity and reiterate your interest in the position. This simple gesture can reinforce your enthusiasm and keep you top of mind for hiring managers.
By diligently following these steps, you will enhance your chances of successfully understanding how to become a and advancing your career in clinical research.
Conclusion
Becoming a Clinical Research Associate (CRA) is a rewarding journey that necessitates a clear understanding of the role, educational background, practical experience, and strategic job application techniques. This guide outlines the essential steps to embark on this career path, emphasizing the significance of each aspect in ensuring success within the dynamic field of clinical research.
Key points discussed encompass the multifaceted responsibilities of CRAs, including:
- Monitoring clinical trials
- Managing research sites
- Ensuring data integrity
Furthermore, a solid educational foundation, encompassing relevant degrees and certifications, is highlighted as a crucial factor in enhancing job prospects. Gaining practical experience through internships and entry-level positions is equally emphasized, as these opportunities provide invaluable insights into the clinical trial process while helping to build a professional network.
Ultimately, the demand for Clinical Research Associates is on the rise, presenting a wealth of opportunities for those willing to invest in their education and experience. Aspiring CRAs are encouraged to actively pursue internships, tailor their job applications, and continuously seek professional development to distinguish themselves in a competitive market. By following these steps, individuals can position themselves for a successful and fulfilling career in clinical research, contributing to advancements in healthcare and improved patient outcomes.
Frequently Asked Questions
What is the role of a Clinical Research Associate (CRA)?
A Clinical Research Associate (CRA) serves as a vital link between sponsors and research sites, playing a crucial role in the research process.
What are the key responsibilities of a CRA?
The key responsibilities of a CRA include monitoring clinical trials, managing research sites, collecting and verifying data, maintaining communication with stakeholders, and reporting on study progress and issues.
How do CRAs ensure compliance during clinical trials?
CRAs ensure compliance by monitoring trials to adhere to protocols, regulatory requirements, and Good Clinical Practice (GCP) guidelines, which are essential for maintaining the integrity of the research.
What does site management involve for a CRA?
Site management involves selecting and overseeing clinical research locations, ensuring they are equipped with necessary resources and trained personnel, performing feasibility studies, and selecting primary investigators (PIs).
Why is data collection and verification important for CRAs?
Data collection and verification are important because CRAs guarantee the accuracy and integrity of the data, which are vital for the success of the study.
What role does communication play in a CRA’s responsibilities?
Communication is essential for CRAs to maintain open lines with investigators, sponsors, and regulatory bodies, facilitating smooth operations and addressing challenges that may arise during the study.
What types of reports do CRAs prepare?
CRAs prepare comprehensive documents detailing study progress, encountered issues, study status, inventory, and adverse events, ensuring transparency and providing valuable insights to stakeholders.
What is the projected demand for Clinical Research Associates in the future?
The demand for Clinical Research Associates is projected to increase significantly by 2025, with the clinical trial market expected to surpass 70 billion dollars.
List of Sources
- Understand the Role and Responsibilities of a Clinical Research Associate
- The Pathway to Becoming a Clinical Research Associate (https://about.citiprogram.org/blog/the-pathway-to-becoming-a-clinical-research-associate)
- 2025 Clinical Research Workforce Report (https://ccrps.org/clinical-research-blog/2025-clinical-research-workforce-report)
- Key Findings from BDO’s 2024 Clinical Research Organization Global Compensation & Turnover Survey (https://bdo.com/insights/tax/key-findings-from-bdos-2024-clinical-research-organization-global-compensation-turnover-survey)
- lindushealth.com (https://lindushealth.com/blog/cra-vs-crc-demystifying-the-roles-in-clinical-research)
- An Examination of the Role of the Clinical Research Associate and Factors Impacting Performance and Experience | Applied Clinical Trials Online (https://appliedclinicaltrialsonline.com/view/an-examination-of-the-role-of-the-clinical-research-associate-and-factors-impacting-performance-and-experience)
- Explore Educational Requirements and Certification Options
- careervira.com (https://careervira.com/job-role/science-and-biotech-clinical-research-associate-cra-for-early-career-in-au)
- qualtrics.com (https://qualtrics.com/blog/research-quotes)
- careers.iconplc.com (https://careers.iconplc.com/blogs/2024-11/how-to-become-a-clinical-research-associate-cra)
- CRA Turnover Within Contract Research Organizations Post-COVID-19: A Cross-Sectional Study – ACRP (https://acrpnet.org/2025/02/14/cra-turnover-within-contract-research-organizations-post-covid-19-a-cross-sectional-study)
- lindushealth.com (https://lindushealth.com/blog/cra-vs-crc-demystifying-the-roles-in-clinical-research)
- Gain Practical Experience Through Internships and Entry-Level Positions
- Case Study Report | PDF | Pharmaceutical Drug | Pharmaceutical Industry (https://scribd.com/document/139268588/Case-Study-Report)
- utulsa.edu (https://utulsa.edu/news/inspire-interns)
- childrenshospitals.org (https://childrenshospitals.org/news/childrens-hospitals-today/2024/10/pilot-internship-program-seeks-to-grow-vital-research-role)
- 2025 Clinical Research Workforce Report (https://ccrps.org/clinical-research-blog/2025-clinical-research-workforce-report)
- Navigate the Job Application Process and Secure a CRA Position
- 165 Resume Statistics: Cover Letters and Valuable Skills (https://flair.hr/en/blog/resume-statistics)
- Top 80 Cover Letter Statistics, Data & Trends in 2025 (https://blog.9cv9.com/top-80-cover-letter-statistics-data-trends-in-2025)
- medium.com (https://medium.com/@raysims/are-cover-letters-still-important-for-job-applications-51bcb4cee2b8)
- jobscan.co (https://jobscan.co/blog/interview-rates-study)
- 170+ Must-Know Resume Statistics for Job Seekers in 2026 (https://enhancv.com/blog/resume-statistics)

Leave a Reply