Introduction
The cardiovascular clinical trial landscape in Chile is rife with complexities that can either hinder or propel sponsors toward success. With a regulatory framework designed to protect participants while promoting research, understanding the nuances of approval processes, patient recruitment strategies, and early feasibility studies is crucial for success.
What strategies can sponsors adopt to turn these local advantages into tangible benefits for their trial outcomes? By mastering these local intricacies, sponsors can significantly improve their trial outcomes and accelerate their market entry.
Understand Regulatory Frameworks for Clinical Trials in Chile
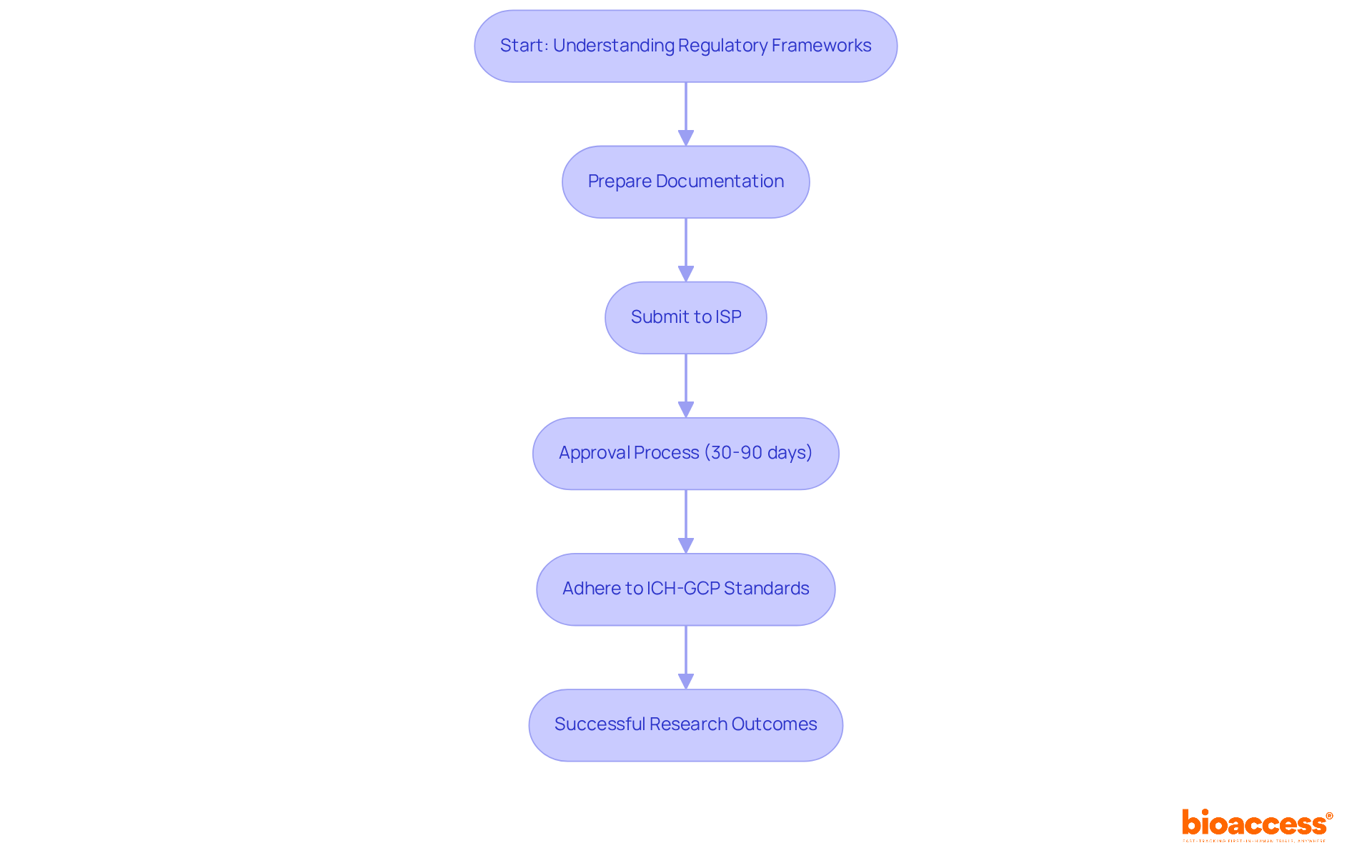
Navigating Chile’s regulatory landscape for research studies can be complex, yet it is essential for successful outcomes. The framework established by Law No. 20.120 governs research involving human subjects, with the Instituto de Salud Pública (ISP) overseeing the approval process, which typically takes between 30 to 90 days. Adhering to International Council for Harmonisation Good Clinical Practice (ICH-GCP) standards is vital, ensuring that studies are conducted ethically and scientifically sound.
Navigating the approval process can be daunting without a clear understanding of submission pathways. This involves preparing necessary documentation such as the study protocol and informed consent forms. This understanding not only accelerates approvals but also enhances the trustworthiness of your research data, making it more acceptable to international authorities like the FDA.
Law No. 20.120 has led to significant improvements in the research landscape, including enhanced protections and a focus on aligning studies with local health needs. Successful approvals of research studies under this law illustrate its effectiveness in promoting investigation while safeguarding participant welfare and ethical benchmarks. By mastering these regulations, sponsors can unlock the full potential of their research initiatives in Latin America.
Implement Early Feasibility Studies to Optimize Trial Design
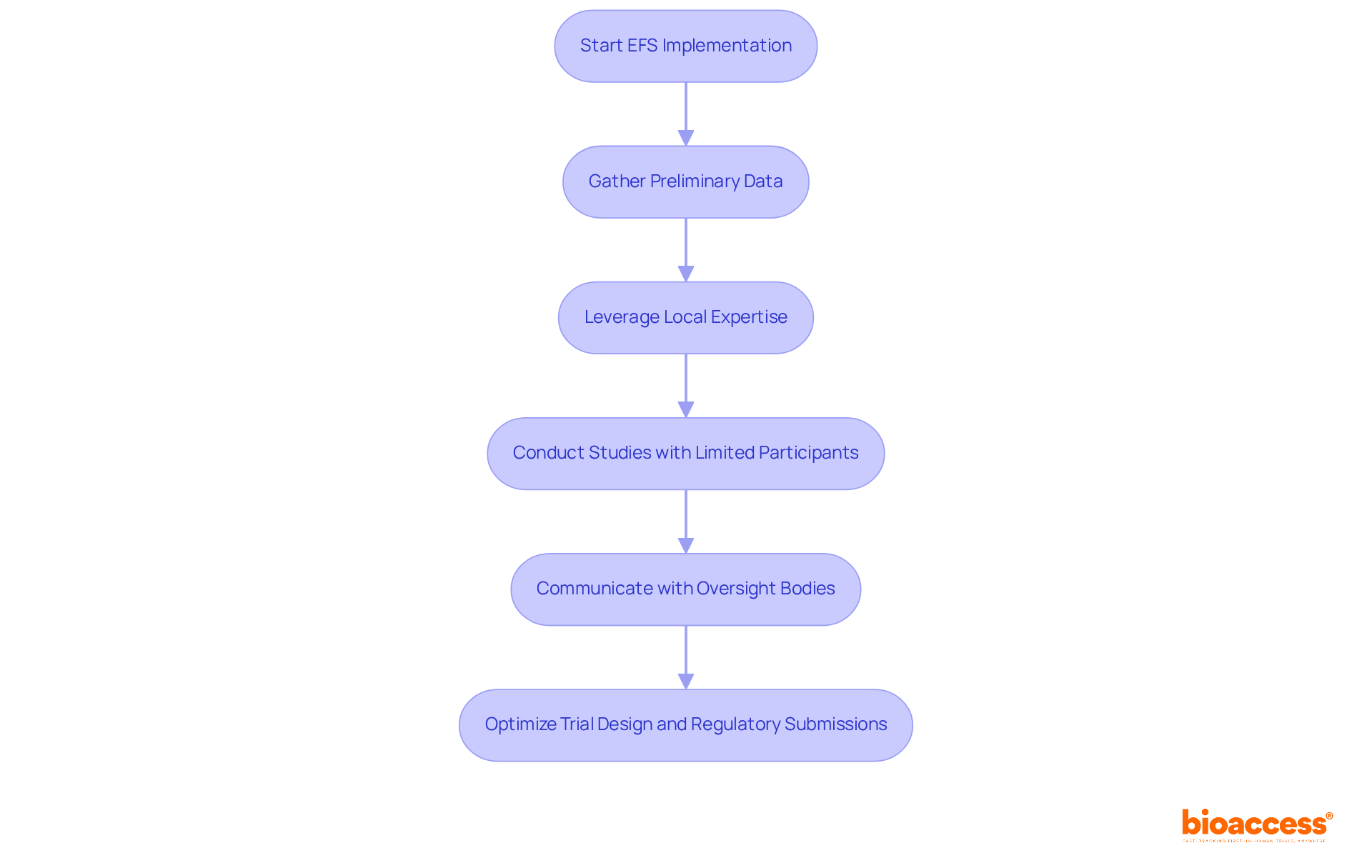
Early feasibility studies (EFS) are essential for evaluating the safety and functionality of new medical devices or therapies, setting the stage for successful clinical evaluations. In the country, conducting an EFS allows sponsors to gather preliminary data on device performance and patient safety, which is crucial for enhancing the design of subsequent studies. These studies generally enroll a limited number of participants, often ten or fewer, and focus on specific endpoints essential for submissions to authorities.
Sponsors can efficiently execute EFS by leveraging the diverse patient population and local expertise in the region. This often leads to results that are significantly faster than those from traditional trial designs. This faster process reduces risks and boosts the chances of approval by delivering strong preliminary data that backs the safety and effectiveness of the intervention.
The oversight framework in Chile, supported by agencies such as the Instituto de Salud Pública (ISP), facilitates a streamlined pathway for EFS, allowing for reduced preclinical data requirements compared to pivotal studies. This flexibility is particularly advantageous for MedTech and Biopharma companies aiming to navigate compliance challenges while optimizing trial design.
Furthermore, the EFS program promotes ongoing communication with oversight bodies, which can result in timely adjustments of clinical protocols based on early findings. By adopting best practices in EFS, sponsors can enhance their regulatory submissions, speeding up the journey to market for innovative medical technologies. Significantly, conducting studies in Latin America can result in considerable cost reductions, with pre-negotiated site contracts providing $25K savings per patient and overall expenses being 30% lower than in other areas. Moreover, the strategic benefits of conducting studies in Colombia, Brazil, and Mexico further boost the potential for successful early phase research.
Develop Targeted Patient Recruitment Strategies for Effective Participation
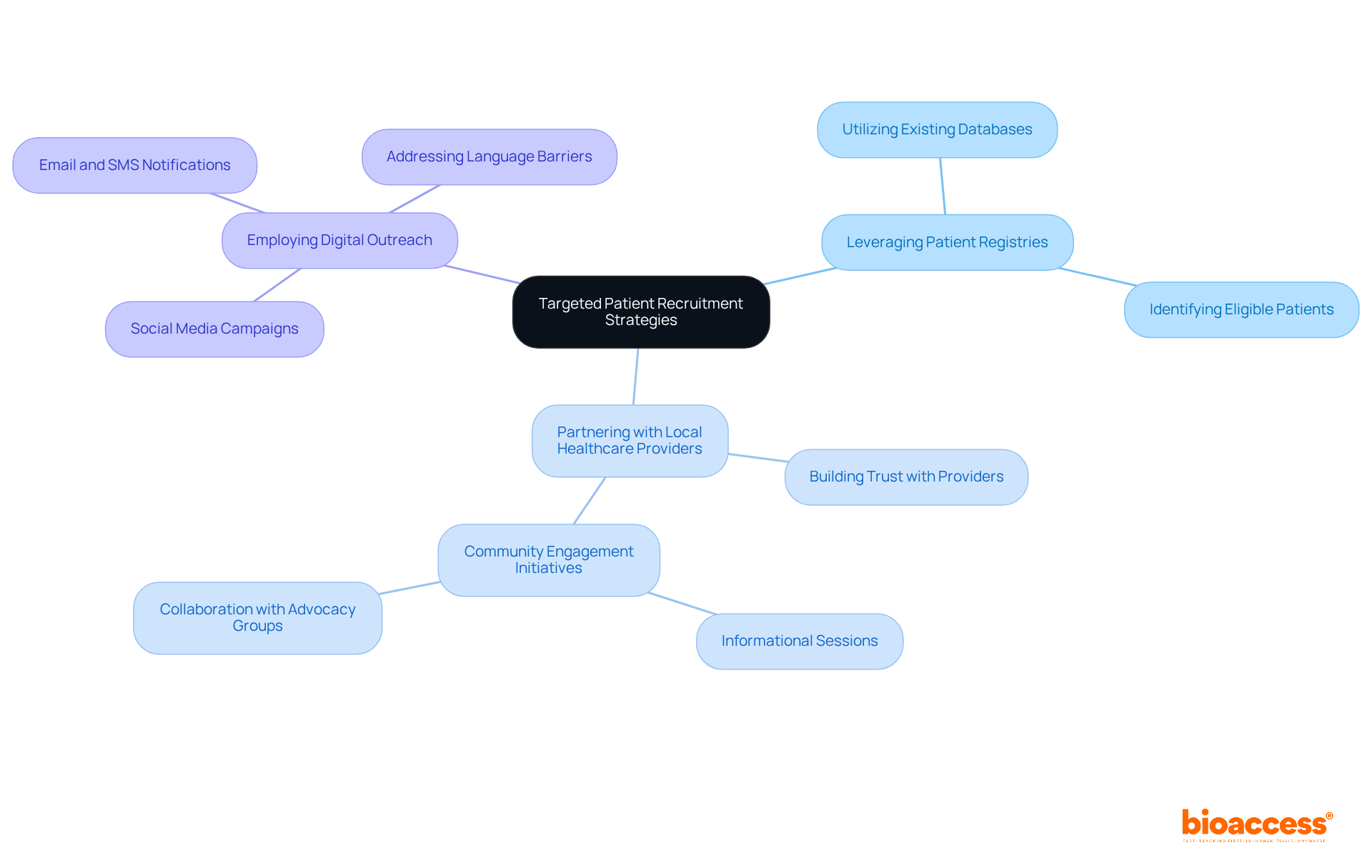
In the competitive landscape of first-in-human research, efficient patient enrollment is not just important; it’s essential for success. Effective strategies include:
- Leveraging patient registries.
- Partnering with local healthcare providers.
- Employing digital outreach methods to significantly raise awareness about the study.
Imagine facing a staggering 80% of studies delayed due to recruitment issues, emphasizing the necessity for focused strategies.
To build trust and encourage participation, it’s essential to address barriers like language and cultural differences. Customizing recruitment messages to connect with the intended demographic can lead to enhanced enrollment rates, ensuring studies are completed on schedule and within budget. For instance, successful patient recruitment examples in a cardiovascular clinical trial in Chile have shown that community engagement initiatives, such as informational sessions and partnerships with local advocacy groups, can yield higher participation rates.
Furthermore, adherence to local compliance necessities, such as those established by the Instituto de Salud Pública (ISP) and conformity to ICH-GCP standards, is crucial for preserving the integrity of the study process. By implementing these strategies, sponsors can turn the tide on recruitment delays, enhancing their chances of meeting recruitment goals and obtaining the necessary data for submission processes. Ultimately, the right strategies can transform recruitment challenges into opportunities for groundbreaking research.
Leverage Local Clinical Trial Sites for Faster Execution
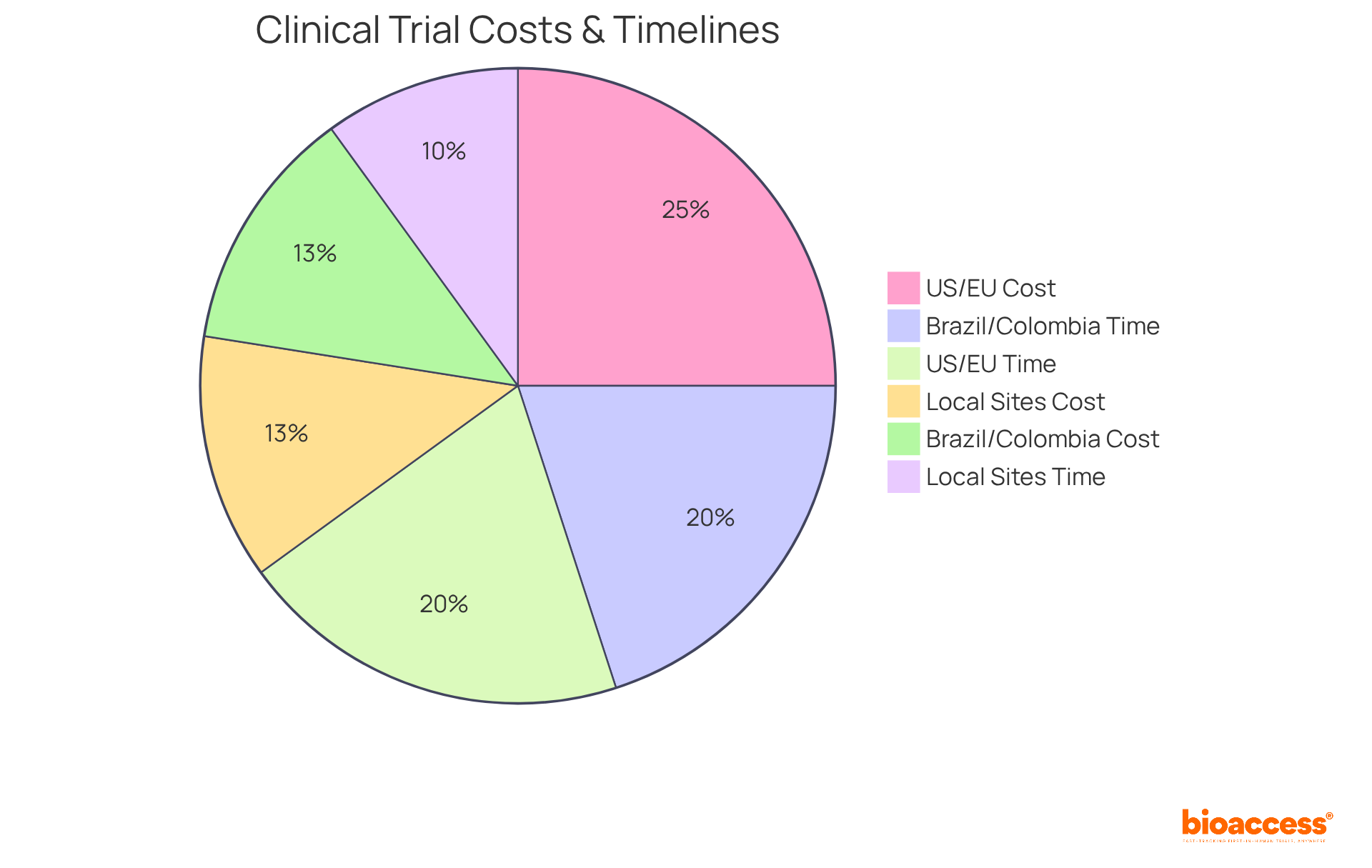
For sponsors embarking on first-in-human studies, local experimental sites in the region present a compelling advantage. These sites have established relationships with local regulatory authorities, such as the Instituto de Salud Pública (ISP) and the Agencia Nacional de Medicamentos (ANAMED), along with a deep understanding of regional patient demographics. This knowledge can significantly expedite patient recruitment and retention. Did you know that in this region, the approval process for medical device clinical evaluations takes just 4 to 8 weeks? Compare that to the six to nine months it takes in Brazil and Colombia.
Additionally, conducting studies in the region is cost-effective; per-patient expenses range from $15,000 to $35,000, significantly lower than the $40,000 to $75,000 typically observed in the U.S. and EU. Ethics committee fees in Chile also reflect this trend, ranging from $500 to $3,000, which is considerably less than the fees in developed markets. When sponsors choose pre-qualified sites that meet ICH-GCP standards, they can conduct their studies efficiently and ethically, ensuring high-quality data integrity.
Choosing local sites speeds up study execution. It also boosts the quality of the clinical data collected. With site readiness attainable in as little as 30 days, sponsors can take advantage of the swift activation timelines that the region provides. This method not only accelerates the evaluation process but also establishes Chile as a rising center for medical device research in Latin America, highlighting its ability to facilitate a variety of complex medical device studies. Additionally, sponsors can potentially save $25K per patient and benefit from an overall trial cost reduction of 30%, allowing for reinvestment into R&D or future funding milestones. By leveraging these local advantages, sponsors can not only enhance their research outcomes but also position themselves strategically in the evolving Medtech landscape.
Conclusion
Navigating the complexities of regulatory frameworks and patient recruitment can be daunting for sponsors in cardiovascular clinical trials in Chile. By capitalizing on Chile’s distinct advantages, sponsors can optimize their research initiatives, ensuring both ethical compliance and scientific integrity. Emphasizing the importance of local regulations, such as Law No. 20.120, and adherence to ICH-GCP standards lays the groundwork for robust and trustworthy clinical research.
It’s clear that early feasibility studies are crucial for refining trial designs, and targeted patient recruitment strategies play a vital role in overcoming enrollment challenges. By utilizing local clinical trial sites, sponsors can significantly reduce costs and expedite study execution, thereby enhancing the overall efficiency of their clinical trials. The integration of these best practices not only streamlines the approval process but also fosters a conducive environment for innovative medical research.
In conclusion, by embracing these strategies, sponsors can not only navigate challenges but also position themselves at the forefront of medical innovation in Latin America. Prioritizing regulatory compliance, optimizing trial designs through early feasibility studies, and implementing effective recruitment strategies will transform potential obstacles into opportunities for groundbreaking advancements in healthcare.
Frequently Asked Questions
What is the primary law governing clinical trials in Chile?
The primary law governing clinical trials in Chile is Law No. 20.120, which regulates research involving human subjects.
Which organization oversees the approval process for clinical trials in Chile?
The Instituto de Salud Pública (ISP) oversees the approval process for clinical trials in Chile.
How long does the approval process for clinical trials typically take in Chile?
The approval process for clinical trials in Chile typically takes between 30 to 90 days.
What standards must be adhered to when conducting clinical trials in Chile?
Adherence to the International Council for Harmonisation Good Clinical Practice (ICH-GCP) standards is vital for conducting clinical trials ethically and scientifically sound in Chile.
What documentation is required for submitting a clinical trial in Chile?
Necessary documentation for submitting a clinical trial in Chile includes the study protocol and informed consent forms.
How does understanding the regulatory framework benefit clinical trial sponsors?
A clear understanding of the regulatory framework accelerates approvals and enhances the trustworthiness of research data, making it more acceptable to international authorities like the FDA.
What improvements has Law No. 20.120 brought to the research landscape in Chile?
Law No. 20.120 has led to significant improvements, including enhanced protections for participants and a focus on aligning studies with local health needs.
Why is mastering the regulatory framework important for sponsors conducting research in Latin America?
Mastering the regulatory framework allows sponsors to unlock the full potential of their research initiatives, facilitating faster patient recruitment and more efficient clinical trial operations in Latin America.
List of Sources
- Understand Regulatory Frameworks for Clinical Trials in Chile
- Trends in clinical trials performed in Chile (https://scielo.cl/article_plus.php?pid=S0034-98872021000100110&tlng=en&lng=es)
- 4 Best Practices for First in Human Biopharma Trials in Chile | bioaccess® (https://bioaccessla.com/blog/4-best-practices-for-first-in-human-biopharma-trials-in-chile)
- Best Practices for Selecting Investigator Sites in Chile for Clinical Trials | bioaccess® (https://bioaccessla.com/blog/best-practices-for-selecting-investigator-sites-in-chile-for-clinical-trials)
- leaux.net (https://leaux.net/URLS/Synopses/F29314493B6746358A05ACFE4345F9B1.html)
- Implement Early Feasibility Studies to Optimize Trial Design
- Early feasibility studies on devices: “doing it sooner” to avoid trial failure | Meditrial (https://meditrial.net/2022/09/early-feasibility-studies-on-devices-doing-it-sooner-to-avoid-trial-failure)
- Clinical trials by start date Chile 1999-2024| Statista (https://statista.com/statistics/1560150/chile-number-new-clinical-trials?srsltid=AfmBOopE4ubow09F75P45cmMxVErUhyMTgNDIvHT3KC8SVNJd6uBlU3y)
- Early Feasibility Studies (EFS) Program (https://fda.gov/medical-devices/investigational-device-exemption-ide/early-feasibility-studies-efs-program)
- Should You Conduct Your Medical Device Clinical Trial In Latin America? – Journal for Clinical Studies (https://journalforclinicalstudies.com/conduct-medical-device-clinical-trial-latin-america)
- Master Early Phase Clinical Trials in Chile: Key Strategies and Insights | bioaccess® (https://bioaccessla.com/blog/master-early-phase-clinical-trials-in-chile-key-strategies-and-insights)
- Develop Targeted Patient Recruitment Strategies for Effective Participation
- Seven Best Practices For Clinical Trial Patient Recruitment (https://openclinica.com/blog/seven-patient-recruitment-best-practices-used-by-leading-research-studies)
- Patient Recruitment Strategies for Trials | CCRPS (https://ccrps.org/clinical-research-blog/patient-recruitment-strategies-for-clinical-trials)
- Patient Recruitment for Clinical Trials: Strategies That Actually Work (https://kapsuletech.com/blog/patient-recruitment-clinical-trials)
- Successes and Challenges in Clinical Trial Recruitment: The Experience of a New Study Team – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC11348161)
- 3 Patient Recruitment Strategies in Clinical Trials (https://clariness.com/resource/3-patient-recruitment-strategies-in-clinical-trials)
- Leverage Local Clinical Trial Sites for Faster Execution
- Clinical Trials Quotes (2 quotes) (https://goodreads.com/quotes/tag/clinical-trials)
- How Chile Is Shaping Medical Device Clinical Trials In Latin America (https://meddeviceonline.com/doc/how-chile-is-shaping-medical-device-clinical-trials-in-latin-america-0001)
- Costs & Timelines | bioaccess® (https://bioaccessla.com/costs-and-timelines)
- Chile Clinical Trial Supply & Logistics Market Size & Outlook, 2030 (https://grandviewresearch.com/horizon/outlook/clinical-trial-supply-logistics-market/chile)

Leave a Reply