Introduction
Brazil’s recent overhaul of clinical research regulations has positioned the country as a frontrunner for innovative trials, significantly reducing approval times and aligning with international standards. This transformation opens a gateway for sponsors to conduct FDA accepted clinical trials with enhanced efficiency and compliance. Navigating this new regulatory environment poses significant challenges for stakeholders, but those who adapt swiftly can leverage Brazil’s strategic advantages in early-stage clinical studies.
Understand Brazil’s New Clinical Research Regulations
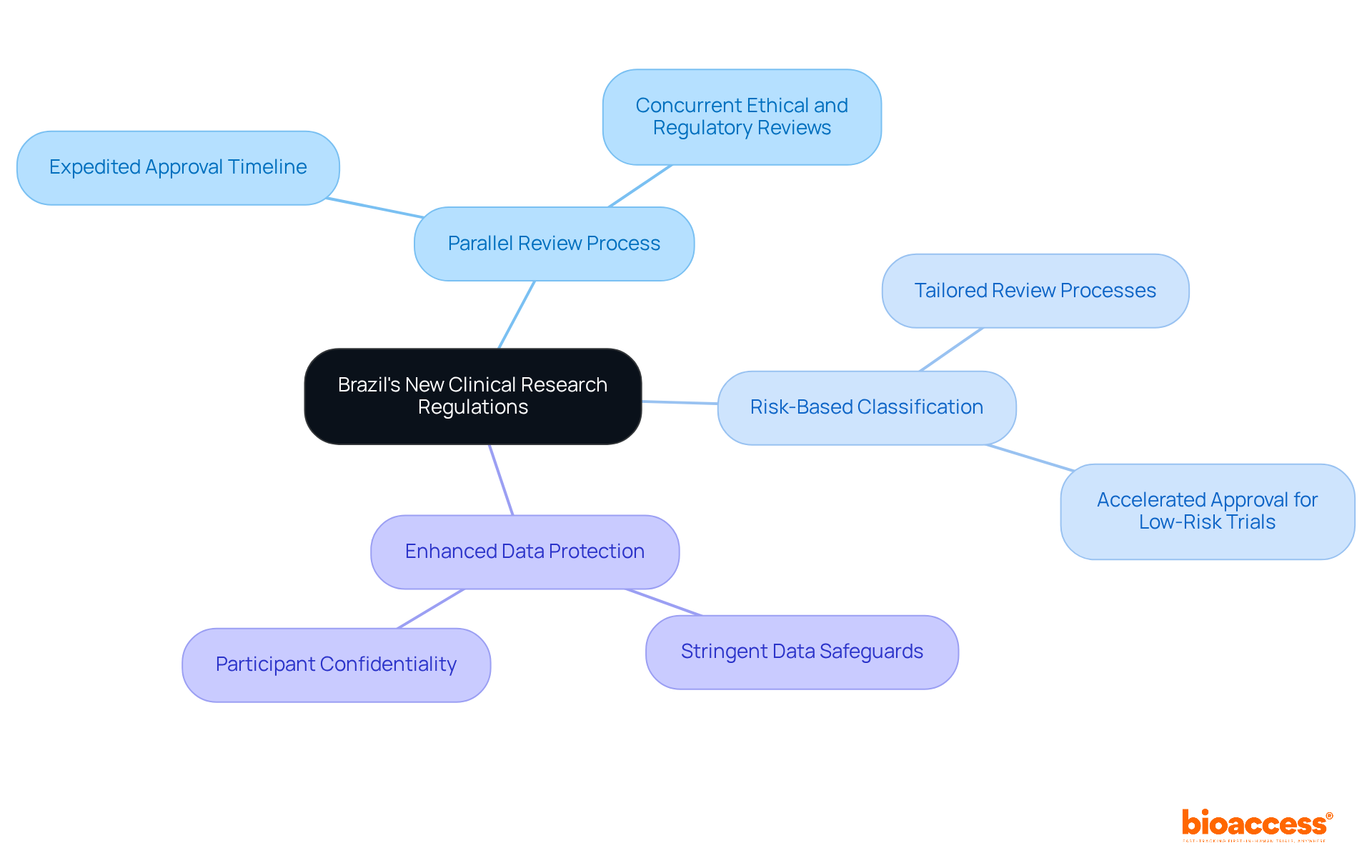
Brazil’s Law No. 14,874/2024 has revolutionized the clinical research landscape, drastically cutting approval times and aligning with global standards. This law not only aligns with international standards but also mandates compliance with ICH-GCP, setting a new benchmark for global quality in clinical research.
Key features of the new regulations include:
- Parallel Review Process: Ethical reviews are now conducted concurrently with regulatory assessments by ANVISA, significantly expediting the overall approval timeline.
- Risk-Based Classification: Trials are categorized based on their risk levels, allowing for tailored review processes that can further accelerate approval times.
- Enhanced Data Protection: The law enforces stringent data protection measures, safeguarding participant confidentiality and ensuring data integrity throughout the research process.
These regulatory advancements are critical for sponsors who must strategically plan and execute an FDA accepted clinical trial in Brazil, ensuring compliance and accelerating market entry for groundbreaking innovations. Comprehending these modifications is crucial for utilizing Brazil’s strategic benefits in early-stage clinical studies, such as rapidity, cost-effectiveness, and efficient patient recruitment pathways. By utilizing bioaccess®’s Global Trial Accelerators™, MedTech startups can maximize these benefits, achieving significant cost savings of up to $25K per patient while ensuring a streamlined path to market.
Prioritize Participant Safety and Ethical Standards
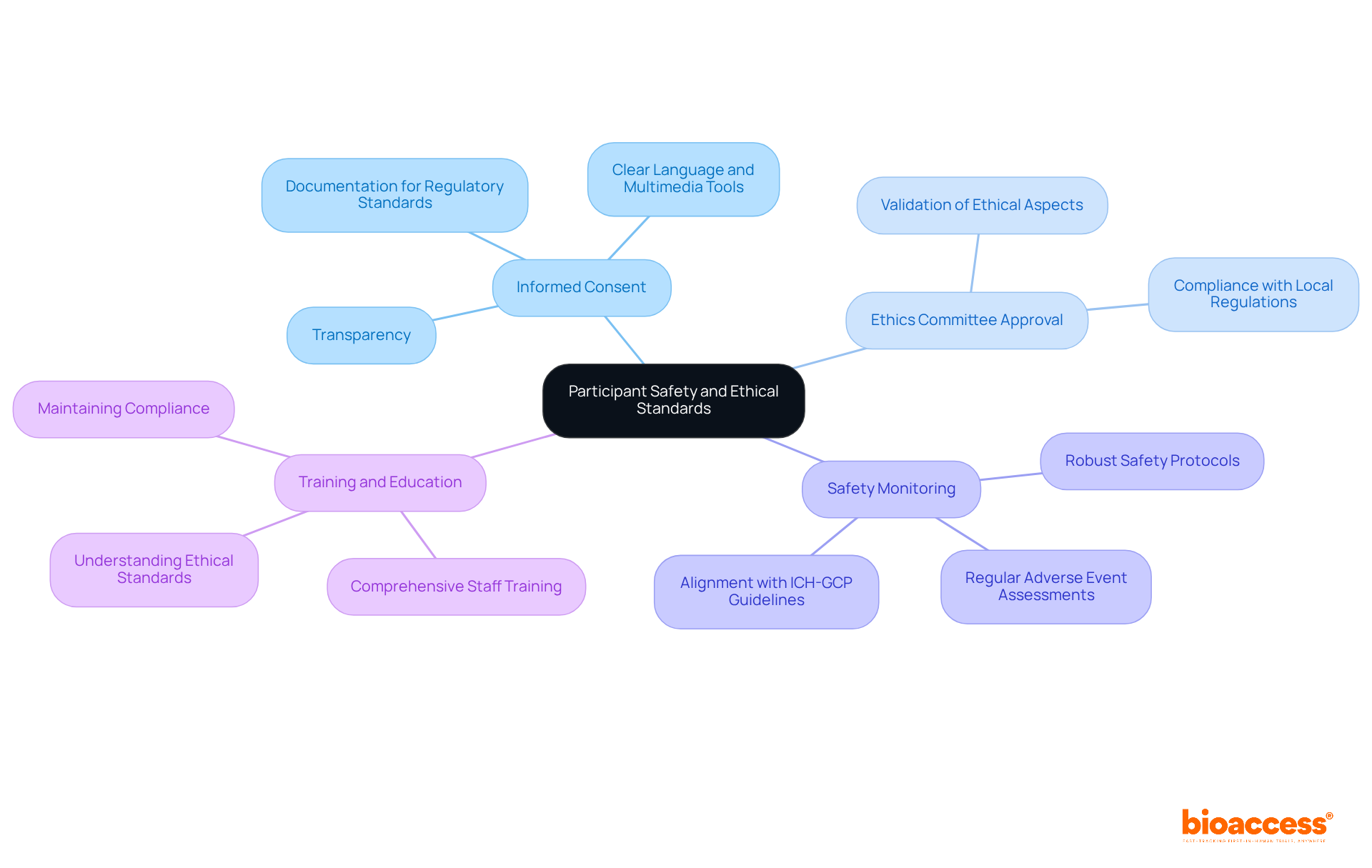
In the realm of clinical research, the stakes are high, and the safety of participants must never be compromised. Emphasizing participant safety and adhering to ethical standards is crucial, especially in first-in-human studies. Here are best practices to ensure compliance:
- Informed Consent: All participants must provide informed consent, fully understanding the risks and benefits of the trial. This process should be transparent, utilizing clear language and multimedia tools to enhance comprehension, and meticulously documented to meet regulatory standards.
- Ethics Committee Approval: Getting this approval is vital; it not only validates the ethical aspects of your study but also ensures you’re in line with local regulations like those from ANVISA in Brazil.
- Safety Monitoring: Implement robust safety monitoring protocols, including regular assessments of adverse events and a clear plan for reporting these incidents to regulatory authorities. This should align with ICH-GCP guidelines to ensure participant safety and data integrity.
- Training and Education: Provide comprehensive training for all staff involved in the study on ethical standards and participant safety protocols. This guarantees that everyone comprehends their duties and the significance of maintaining compliance throughout the study.
Implementing these practices not only enhances participant safety but also fortifies ethical standards, ultimately securing approval and fostering public trust in clinical research.
Leverage Regulations to Attract Clinical Research Investment
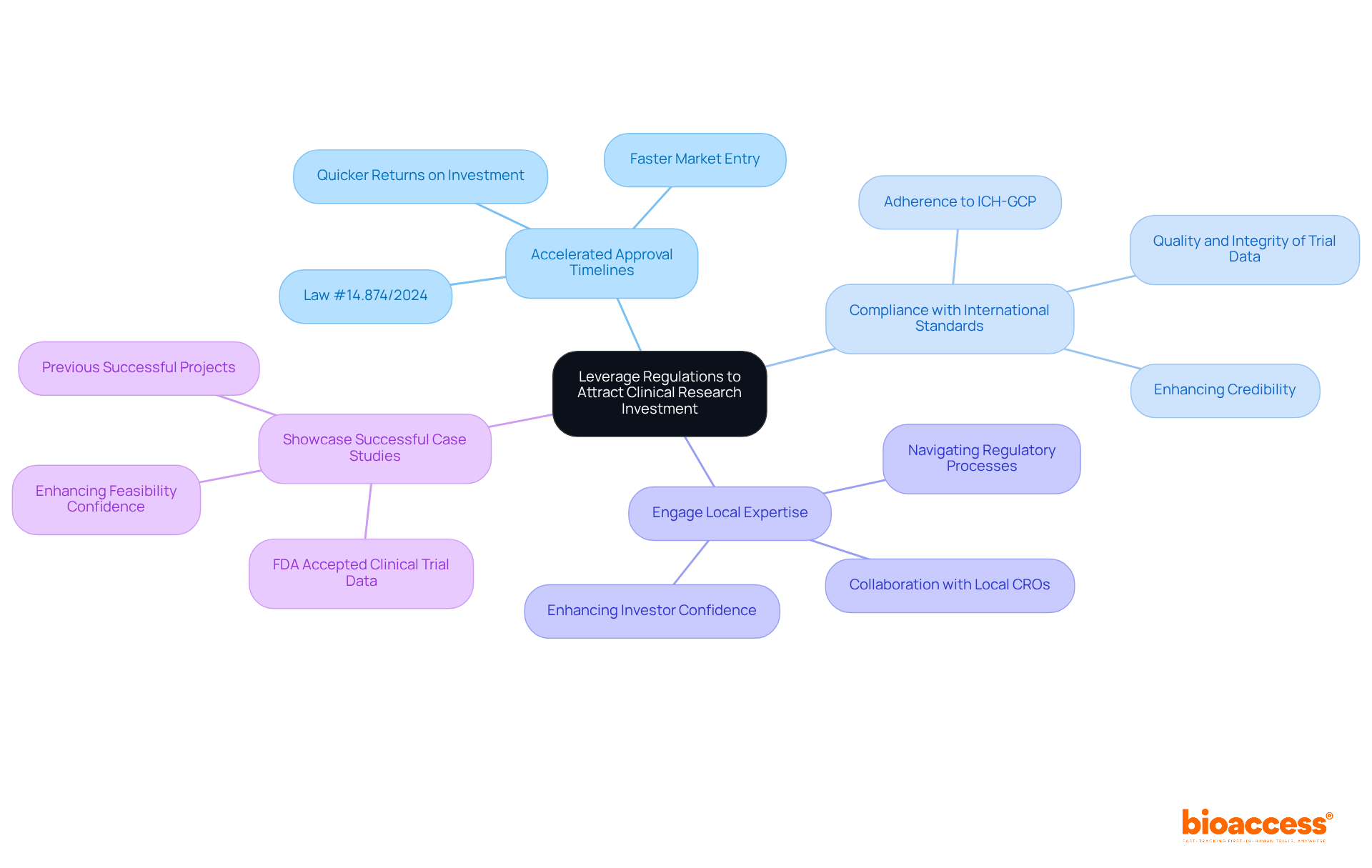
To secure funding for an FDA accepted clinical trial in Brazil, sponsors must navigate a rapidly evolving compliance landscape that presents both challenges and opportunities. Here are key strategies to enhance appeal:
- Highlight Accelerated Approval Timelines: Emphasize the reduced approval timelines under recent regulations, such as Law #14.874/2024, which streamlines ethical approval processes. This showcases the potential for faster market entry and quicker returns on investment, making Brazil an attractive option for sponsors interested in FDA accepted clinical trials in Brazil.
- Demonstrate Compliance with International Standards: Clearly communicate adherence to ICH-GCP and other relevant guidelines, reassuring investors about the quality and integrity of trial data. Compliance with these standards is crucial for an FDA accepted clinical trial Brazil and can significantly enhance credibility.
- Engage Local Expertise: Collaborate with local CROs and compliance consultants who possess in-depth knowledge of the Brazilian market. Their expertise can facilitate smoother navigation through the regulatory process, ensuring compliance and enhancing investor confidence.
- Showcase Successful Case Studies: Present case studies of previous successful projects conducted in Brazil, particularly those that led to significant market approvals or acquisitions. For example, studies that led to FDA accepted clinical trial Brazil data acceptance can enhance investor confidence in the feasibility of new investigations.
By embracing these strategies, sponsors not only enhance their appeal but also position themselves for success in a competitive market.
Implement Effective Operational Strategies for Compliance
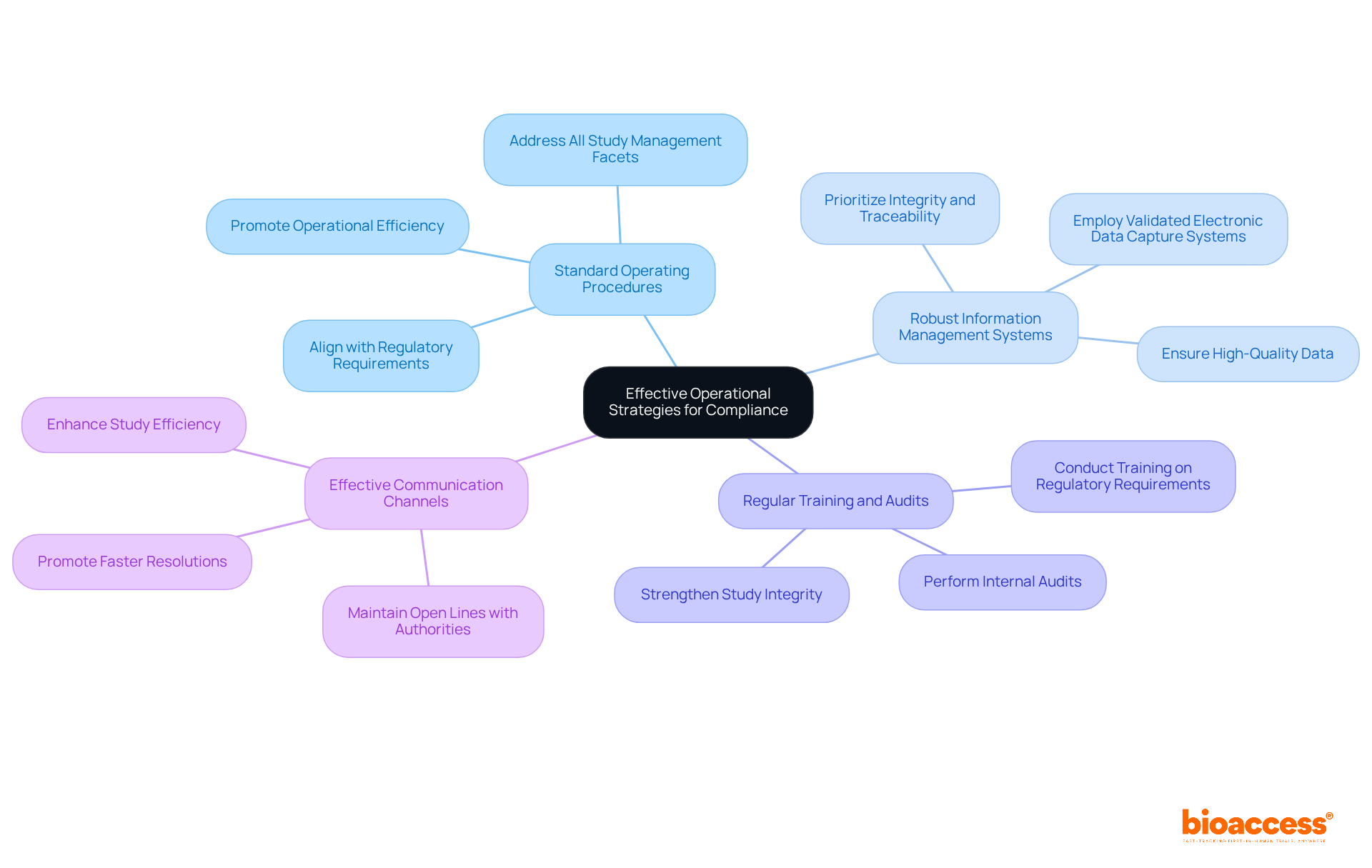
Navigating the complexities of regulatory compliance can be daunting for sponsors, often leading to delays and inefficiencies in clinical trials. To address these challenges, sponsors should adopt the following strategies:
- Standard Operating Procedures (SOPs): Develop and implement SOPs that align with regulatory requirements such as ICH-GCP and local regulations from authorities like ANVISA and INVIMA. These SOPs should thoroughly address all facets of management for the study, including site selection, patient enrollment, information handling, and adverse event reporting.
- Robust Information Management Systems: Establish management systems that prioritize integrity and traceability. Employing validated electronic data capture (EDC) systems that adhere to regulatory standards is crucial for ensuring high-quality data throughout the study process.
- Regular Training and Audits: Conduct regular training sessions for staff on regulatory requirements, ensuring that all team members are well-versed in the latest regulations and best practices. Moreover, conducting internal audits can assist in detecting and correcting any regulatory gaps, strengthening the integrity of the study.
- Effective Communication Channels: Maintain open lines of communication with regulatory authorities, ethics committees, and stakeholders. This clarity can promote faster resolutions to any adherence issues that emerge, ultimately boosting the study’s efficiency.
By prioritizing these operational strategies, sponsors not only enhance compliance but also position themselves for success in the competitive landscape of clinical research in Latin America.
Conclusion
Brazil’s regulatory reforms have reshaped the clinical trial landscape, presenting both challenges and opportunities for sponsors. Law No. 14,874/2024 has reduced approval timelines significantly. It has also established a robust framework that aligns with international standards. This evolution underscores the importance of understanding and leveraging these regulations to enhance compliance and market entry strategies.
This article has highlighted key strategies for navigating Brazil’s clinical research environment successfully. From the parallel review process that expedites approvals to the emphasis on participant safety and ethical standards, these insights are crucial for sponsors aiming to execute efficient and compliant clinical trials. Furthermore, adopting operational strategies like robust information management systems and regular training can significantly enhance compliance and operational efficiency, ultimately leading to successful trial outcomes.
To thrive in this dynamic environment, sponsors must act with urgency and strategic foresight. By embracing the new regulations, prioritizing participant safety, and implementing effective operational strategies, sponsors can enhance their appeal to investors and position themselves for success in a competitive market. With rapid patient recruitment and cost efficiencies, Brazil stands out as an attractive destination for clinical research, reinforcing the need for sponsors to act decisively and strategically in this evolving landscape.
Frequently Asked Questions
What is the significance of Brazil’s Law No. 14,874/2024 for clinical research?
Brazil’s Law No. 14,874/2024 has transformed the clinical research landscape by drastically reducing approval times and aligning the regulatory framework with international standards, including compliance with ICH-GCP.
What are the key features of the new clinical research regulations in Brazil?
The key features include a parallel review process for ethical and regulatory assessments, a risk-based classification system for trials, and enhanced data protection measures to safeguard participant confidentiality and ensure data integrity.
How does the parallel review process benefit clinical trial approval in Brazil?
The parallel review process allows ethical reviews to occur simultaneously with regulatory assessments by ANVISA, significantly expediting the overall approval timeline for clinical trials.
What is the risk-based classification system in the context of Brazil’s new regulations?
The risk-based classification system categorizes clinical trials according to their risk levels, enabling tailored review processes that can further accelerate approval times.
What measures are in place to protect participant data under the new regulations?
The new law enforces stringent data protection measures to ensure participant confidentiality and maintain data integrity throughout the clinical research process.
Why is it important for sponsors to understand these regulatory modifications in Brazil?
Understanding these modifications is crucial for sponsors to strategically plan and execute FDA-accepted clinical trials in Brazil, ensuring compliance and facilitating quicker market entry for innovative products.
How can MedTech startups benefit from Brazil’s early-stage clinical trial environment?
Brazil offers advantages such as rapid approval timelines, cost-effectiveness, and efficient patient recruitment pathways, making it an attractive location for early-stage clinical studies.
What financial benefits can be achieved by utilizing bioaccess®’s Global Trial Accelerators™ in Brazil?
By using bioaccess®’s Global Trial Accelerators™, MedTech startups can achieve significant cost savings of up to $25K per patient while ensuring a streamlined path to market.
List of Sources
- Understand Brazil’s New Clinical Research Regulations
- New regulations for clinical research in Brazil | Licks Attorneys (https://lickslegal.com/post/new-regulations-for-clinical-research-in-brazil)
- Brazil Just Changed The Rules For Clinical Trial Approvals — What Sponsors Need To Know | bioaccess® (https://bioaccessla.com/blog/brazil-anvisa-parallel-review-clinical-trial-approvals-2026)
- Brazil Cuts Clinical Trial Approval Timeline to 90 Days | bioaccess®: Fast-Tracking First-in-Human Trials, Anywhere posted on the topic | LinkedIn (https://linkedin.com/posts/bioaccess_clinicaltrials-brazil-anvisa-activity-7447976698958106627-98MW)
- Brazil Regulates Clinical Research Act: A Complete Review of Decree #12,651/2025 – Part. 1 (https://lexology.com/library/detail.aspx?g=b1b655f4-963a-4be7-876c-81afb05caef6)
- Prioritize Participant Safety and Ethical Standards
- Informed Consent in Clinical Trials | Sterling IRB (https://sterlingirb.com/resources/thinking-outside-the-consent-form-health-literacy-informed-consent-in-clinical-trials)
- Statistical Considerations for Premarketing Risk Assessment (https://fda.gov/drugs/news-events-human-drugs/statistical-considerations-premarketing-risk-assessment-05162024)
- Clinical Trial Statistics: Inside The Clinical Trial Lifecycle (https://clinmax.com/clinical-trial-statistics)
- Best Quotes of Informed Consent with Page Numbers By Tawdra Kandle (https://bookey.app/book/informed-consent/quote)
- Ensuring Participant Safety and Trial Integrity with Clinical Trials Oversight – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC3916962)
- Leverage Regulations to Attract Clinical Research Investment
- Women in Clinical Trials: 5 Quotes To Inspire Action (https://finance.yahoo.com/news/women-clinical-trials-5-quotes-161500798.html)
- Brazil Clinical Trials Market Size & Outlook, 2033 (https://grandviewresearch.com/horizon/outlook/clinical-trials-market/brazil)
- Unlocking Brazil’s Clinical Trial Opportunity: A Strategic Roadmap for Healthcare Leaders (https://lek.com/insights/life-sciences-pharma/unlocking-brazils-clinical-trial-opportunity-strategic-roadmap)
- TOP 10 CLINICAL TRIALS QUOTES | A-Z Quotes (https://azquotes.com/quotes/topics/clinical-trials.html)
- pharmaceuticalmanufacturer.media (https://pharmaceuticalmanufacturer.media/pharmaceutical-industry-insights/10-inspirational-quotes-for-the-pharma-sector)
- Implement Effective Operational Strategies for Compliance
- FDA Guidance on Statistical Methods for Clinical Trials – ECA Academy (https://gmp-compliance.org/gmp-news/fda-guidance-on-statistical-methods-for-clinical-trials)
- Sponsor-Level Compliance with ClinicalTrials.gov Reporting Requirements: A Comprehensive Analysis (https://publichealth.realclearjournals.org/research-articles/2025/09/sponsor-level-compliance-with-clinicaltrials-gov-reporting-requirements-a-comprehensive-analysis)
- Standard Operating Procedures (SOPs) in Clinical Trials | Credevo Articles (https://credevo.com/articles/2023/01/05/standard-operating-procedures-sops-in-clinical-trials)
- The Importance of Standard Operating Procedures (SOPs) in Clinical Research: Enhancing Quality and Compliance (https://infiuss.com/insights/the-importance-of-standard-operating-procedures-sops-in-clinical-research-enhancing-quality-and-compliance)
- Clinical Trials Quotes (2 quotes) (https://goodreads.com/quotes/tag/clinical-trials)

Leave a Reply