Introduction
Reducing clinical trial costs in the MedTech sector has emerged as a critical focus for organizations aiming to maximize efficiency while upholding high standards of research integrity. This article explores four best practices that not only streamline the design and execution of clinical trials but also harness technology and partnerships to achieve substantial cost savings. With the increasing complexities in trial management and participant engagement, MedTech companies must consider:
- How can they effectively navigate these challenges to ensure both financial viability and successful outcomes?
Design Efficient Clinical Trials
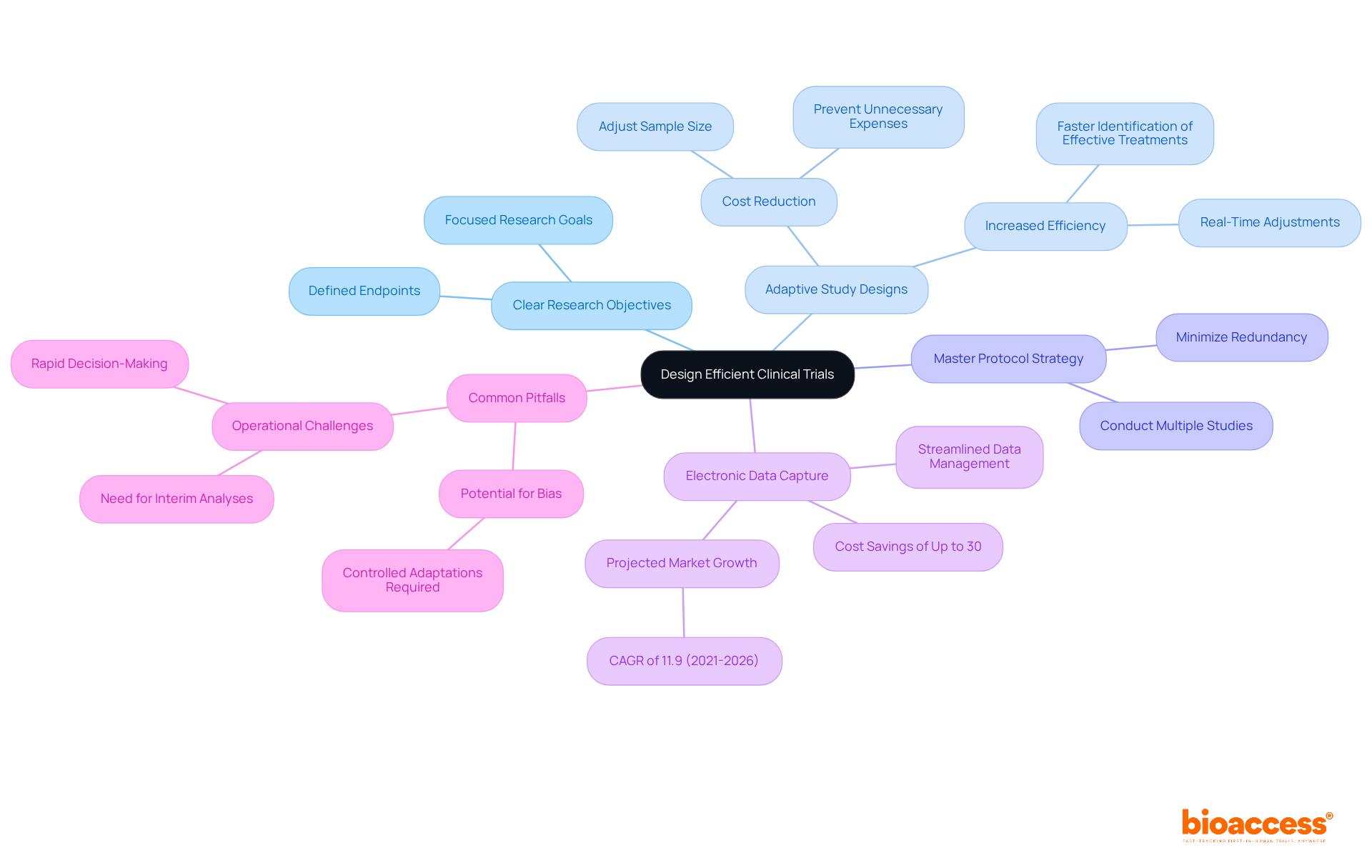
To create effective clinical studies, starting with a clear definition of research objectives and endpoints is essential. Implementing can help reduce by allowing modifications based on interim results, significantly saving time and resources. For instance, a trial that adjusts its sample size in response to early efficacy signals can prevent unnecessary expenses linked to enrolling more participants than needed.
Moreover, utilizing a master protocol strategy enables multiple studies to be conducted under one comprehensive protocol, which is essential to reduce by minimizing redundancy and related expenses. The integration of electronic data capture (EDC) systems streamlines data collection and management, leading to substantial savings. Research shows that studies using EDC can help reduce by achieving total study cost reductions of up to 30%, highlighting the financial advantages of these innovative methodologies.
Additionally, bioaccess offers , realizing $25K savings per patient and enabling through pre-qualified networks. The is projected to expand at a CAGR of 11.9% from 2021 to 2026, underscoring the growing reliance on EDC systems in medical studies. The FDA’s supports the use of , facilitating their application in medical research. A recent study identified 267 medical experiments utilizing , showcasing their increasing acceptance in the industry.
However, it is crucial to be aware of common pitfalls, such as the potential for biases if adaptations are not carefully controlled, to ensure the integrity of the results. As you consider your own challenges in clinical research, think about how these strategies could enhance your studies.
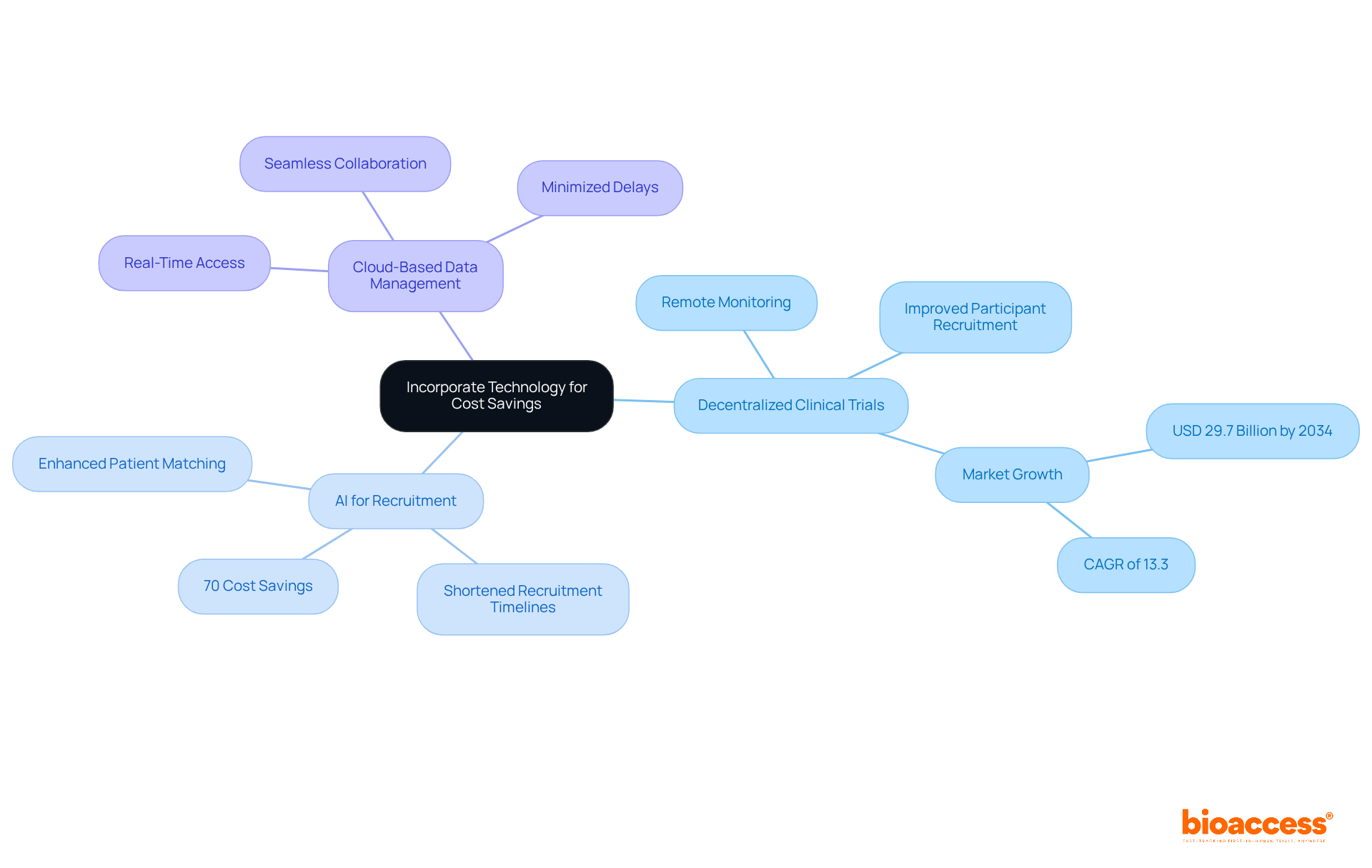
Incorporate Technology for Cost Savings
Incorporating technology into medical studies is not just a trend; it’s a game changer that can significantly reduce and boost operational efficiency. , for example, enable remote participant monitoring and data collection, reducing the need for physical site visits. This approach not only enhances participant recruitment by making involvement more accessible and convenient but also helps to . Industry leaders have observed that DCTs can and improve diversity within participant cohorts, which is vital for the generalizability of trial results.
Moreover, can significantly , with studies indicating potential savings of up to 70%. AI-driven tools enhance patient matching and outreach, significantly shortening recruitment timelines and tackling common bottlenecks in drug development. Additionally, the implementation of allows for real-time data access and seamless collaboration among stakeholders, which can help reduce by minimizing delays and miscommunication.
As the research trial landscape evolves, these technological advancements are becoming essential for looking to navigate the complexities of trial execution efficiently. bioaccess® provides designed to , saving up to $25K per patient and achieving 50% faster enrollment through its pre-qualified networks and pre-negotiated site contracts. This positions bioaccess as an ideal partner for startups eager to accelerate data generation while benefiting from the regulatory advantages and efficient processes available in Latin America.
The decentralized research market is projected to reach USD 29.7 billion by 2034, expanding at a CAGR of 13.3% from 2025 to 2034. This growth underscores the increasing importance of these models in the sector, making it clear that embracing these innovations is not just beneficial but necessary for success.
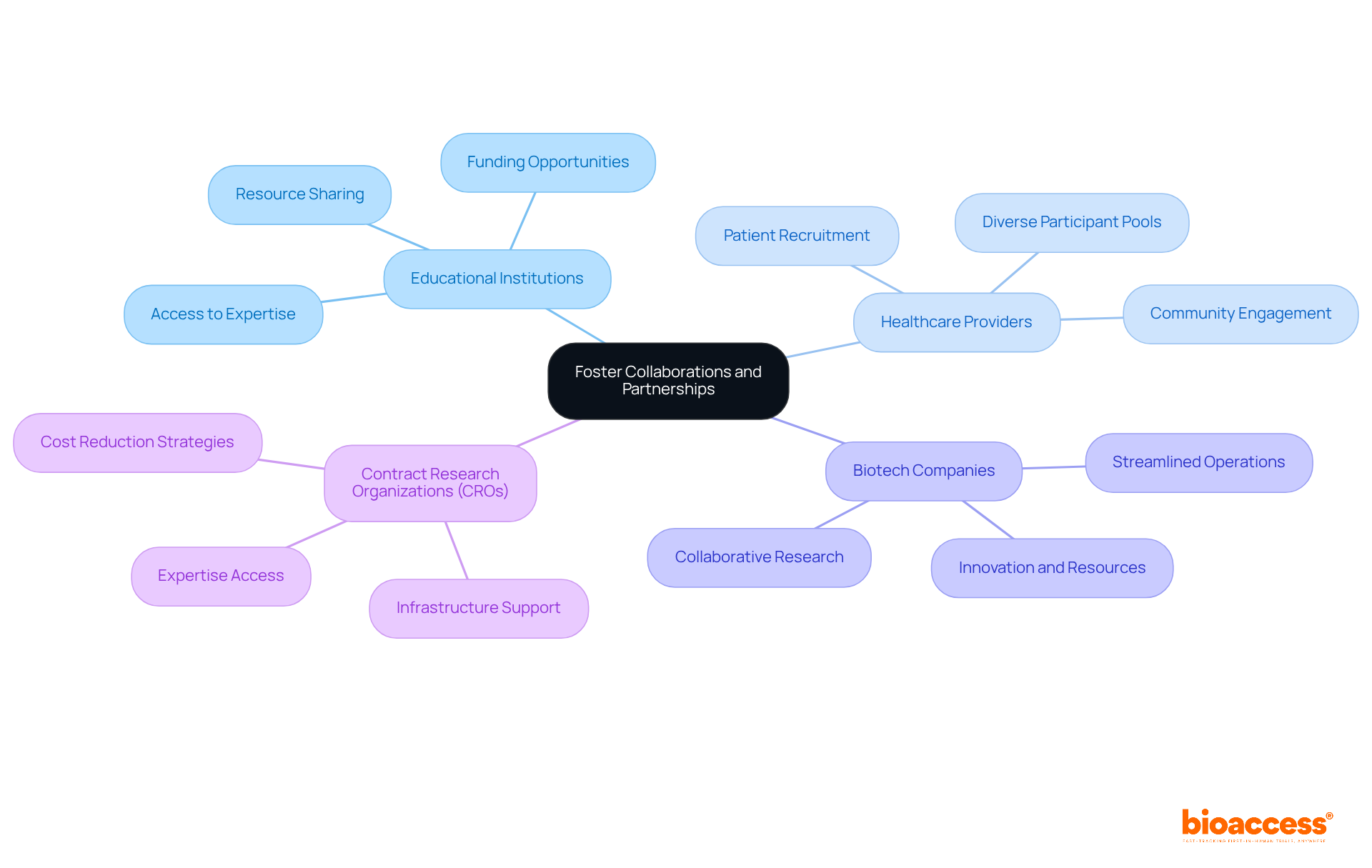
Foster Collaborations and Partnerships
such as educational institutions, healthcare providers, and other biotech companies are essential to reduce . These collaborations enable the , including , which can significantly ease . For example, partnering with nearby hospitals can enhance and provide access to treatment-naive groups, thereby accelerating enrollment and lowering expenses.
Additionally, working with that specialize in offers startups invaluable expertise and infrastructure that would otherwise be prohibitively expensive to develop independently. Notably, joint initiatives have demonstrated a 20-30% reduction in total research expenses, highlighting the to in the medical research landscape.
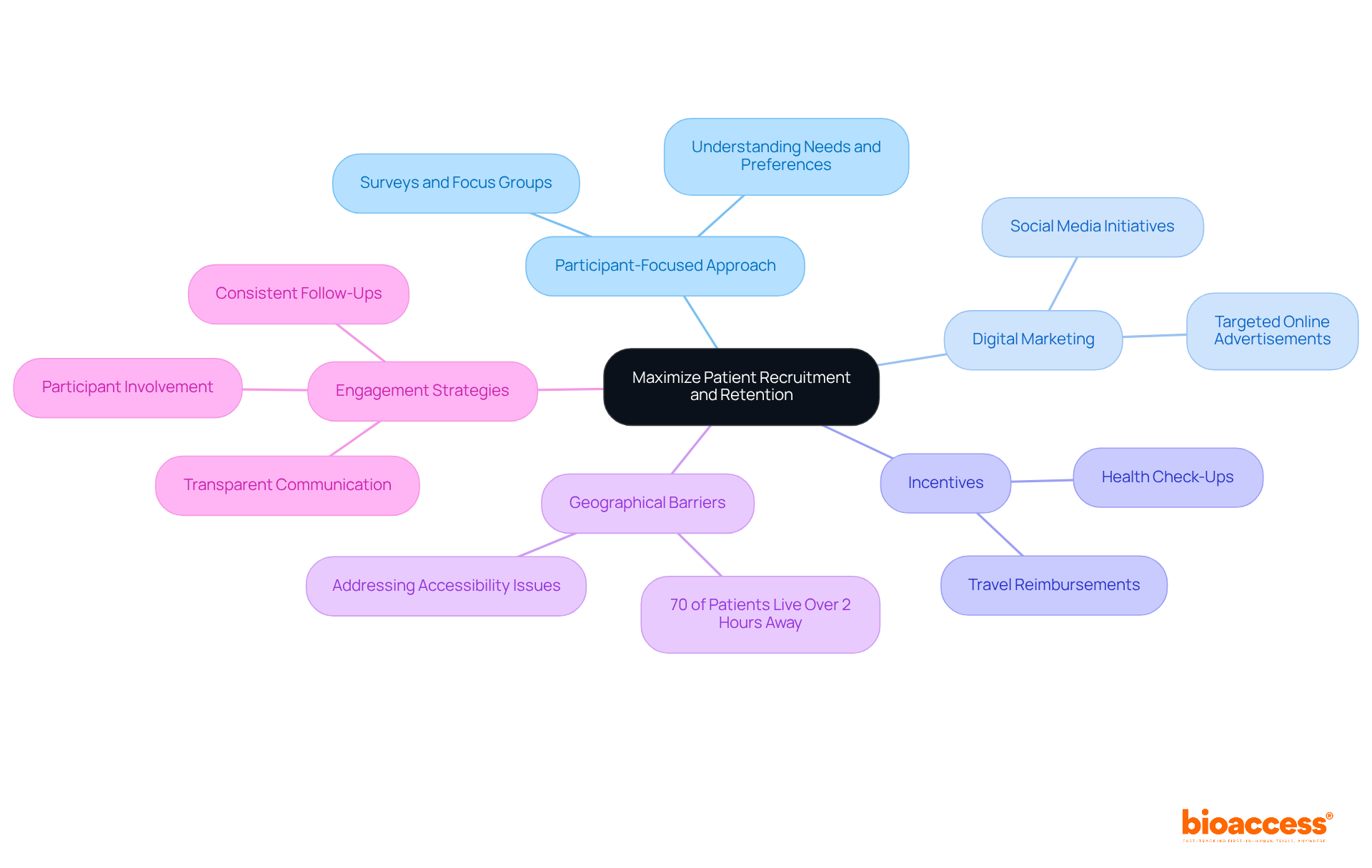
Maximize Patient Recruitment and Retention
To optimize , adopting a is essential. Understanding the can be achieved through surveys and focus groups. Given that up to 85% of specific types of clinical studies encounter , employing – such as social media initiatives and targeted online advertisements – is vital. and addressing any concerns can significantly enhance participant involvement.
Offering incentives, like travel reimbursements or health check-ups, is crucial, especially since financial constraints can hinder participation. Research indicates that studies utilizing can see a 30% increase in retention, underscoring the importance of these methods. Consistent follow-ups and updates on study progress keep participants informed and motivated, fostering a sense of involvement and commitment to the research.
Moreover, it’s important to recognize that 70% of patients eligible for a clinical trial in the United States live more than 2 hours away from an investigation center. This highlights the need to address , ensuring that all potential participants have the opportunity to engage in .
Conclusion
Implementing effective strategies to reduce clinical trial costs in MedTech is crucial for enhancing the efficiency and viability of medical research. By focusing on innovative study designs, leveraging technology, fostering collaborations, and maximizing patient recruitment and retention, organizations can navigate the financial challenges inherent in clinical trials. These practices not only streamline processes but also ensure that critical research progresses without unnecessary financial burdens.
Key insights presented include:
- The importance of adaptive trial designs, which allow for flexibility based on interim results.
- The integration of electronic data capture systems that can cut costs significantly.
- How decentralized clinical trials and the use of AI can enhance participant recruitment while reducing expenses.
- Strategic partnerships with CROs and healthcare institutions can lead to shared resources, ultimately lowering financial pressures and improving study outcomes.
In conclusion, embracing these best practices is not just about cutting costs; it is about fostering a more efficient and inclusive clinical research environment. As the MedTech landscape continues to evolve, organizations must prioritize these strategies to remain competitive and ensure that vital innovations reach the market swiftly and effectively. Taking action today can lead to a more sustainable future for clinical trials, benefiting researchers, patients, and the healthcare industry as a whole.
Frequently Asked Questions
What is essential for creating effective clinical studies?
Starting with a clear definition of research objectives and endpoints is essential for creating effective clinical studies.
How can adaptive study designs benefit clinical trials?
Adaptive study designs can help reduce clinical trial costs by allowing modifications based on interim results, saving time and resources.
What is an example of how adaptive designs can reduce costs?
A trial that adjusts its sample size in response to early efficacy signals can prevent unnecessary expenses linked to enrolling more participants than needed.
What is a master protocol strategy, and how does it help in clinical trials?
A master protocol strategy enables multiple studies to be conducted under one comprehensive protocol, reducing redundancy and related expenses in clinical trials.
How do electronic data capture (EDC) systems contribute to cost savings in clinical trials?
EDC systems streamline data collection and management, leading to total study cost reductions of up to 30%.
What are the financial benefits of using bioaccess in clinical research?
Bioaccess offers affordable research solutions in Latin America, realizing $25K savings per patient and enabling 50% quicker enrollment through pre-qualified networks.
What is the projected growth rate of the Electronic Data Capture Market?
The Electronic Data Capture Market is projected to expand at a CAGR of 11.9% from 2021 to 2026.
How does the FDA support adaptive trial designs?
The FDA’s Complex Innovative Trial Design Paired Meeting Program supports the use of adaptive designs, facilitating their application in medical research.
What should researchers be cautious of when using adaptive designs?
Researchers should be aware of common pitfalls, such as the potential for biases if adaptations are not carefully controlled, to ensure the integrity of the results.
List of Sources
- Design Efficient Clinical Trials
- Electronic Data Capture Market Share, Size and Industry Growth Analysis 2021-2026 (https://industryarc.com/Research/Electronic-Data-Capture-Market-Research-508558)
- QUOTES | Quantification and Optimization of Trial Expectations Simulator by Berry (https://berryconsultants.com/software/quotes)
- Adaptive trial designs will increase clinical trial speed, safety, and effectiveness (https://statnews.com/2024/07/12/adaptive-trial-designs-increase-speed-safety-effectiveness)
- Adaptive designs in clinical trials: a systematic review-part I – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC11451232)
- Incorporate Technology for Cost Savings
- Decentralized Clinical Trials Market Analysis & Forecast, 2034 (https://gminsights.com/industry-analysis/decentralized-clinical-trials-market)
- AI in Clinical Trials Poised for Rapid Growth with a 22.6% (https://bccresearch.com/pressroom/phm/ai-in-clinical-trials-poised-for-rapid-growth-with-a-226?srsltid=AfmBOoooeDYqjxUGmv3Utw3_UQN-I6Kqpt1L7KzcQi_WXDg6NzdMohQf)
- 20 hospital execs’ most thought-provoking quotes on health IT in 2021 – Becker’s Hospital Review | Healthcare News & Analysis (https://beckershospitalreview.com/healthcare-information-technology/innovation/20-hospital-execs-most-thought-provoking-quotes-on-health-it-in-2021)
- 30 Quotes About the Future of Healthcare: Expert Takes (https://deliberatedirections.com/quotes-future-of-healthcare)
- Top 10 Expert Quotes That Redefine the Future of AI Technology (https://nisum.com/nisum-knows/top-10-thought-provoking-quotes-from-experts-that-redefine-the-future-of-ai-technology)
- Foster Collaborations and Partnerships
- 6 Benefits of Clinical Research Collaboration and Partnerships (https://vccrn.org/benefits-clinical-research-collaboration-and-partnerships)
- Transforming the Economics of Clinical Trials – NAM (https://nam.edu/perspectives/transforming-the-economics-of-clinical-trials)
- Registry-based trials: a potential model for cost savings? – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC7682727)
- Clinical trial success depends on strong partnerships – here’s why – Siron Clinical (https://sironclinical.com/clinical-trial-success-depends-on-strong-partnerships-heres-why)
- Maximize Patient Recruitment and Retention
- Clinical studies: the challenge of patient recruitment (https://servier.com/en/newsroom/clinical-studies-patient-recruitment)
- A Primer on the Importance of Recruitment and Retention in Clinical Trials – ACRP (https://acrpnet.org/2023/04/18/a-primer-on-the-importance-of-recruitment-and-retention-in-clinical-trials)
- Patient Recruitment and Retention in Clinical Trials: Strategies and Challenges (https://mdgroup.com/blog/patient-recruitment-and-retention-in-clinical-trials-strategies-and-challenges)
- Patient Recruitment for Clinical Trials: Strategies That Actually Work (https://kapsuletech.com/blog/patient-recruitment-clinical-trials)
- Patient Engagement Statistics: Data That Proves Impact (https://nclusiv.co.uk/blog/f/patient-engagement-statistics-data-that-proves-impact)





. Follow the arrows to move from one step to the next, making the process straightforward. Each box represents a step you need to take to access the FDA Devices Database. Follow the arrows to move from one step to the next, making the process straightforward.](https://images.tely.ai/telyai/nqwedjpo-each-box-represents-a-step-you-need-to-take-to-access-the-fda-devices-database-follow-the-arrows-to-move-from-one-step-to-the-next-making-the-process-straightforward.webp)