Introduction
Navigating the complexities of medical device regulation presents a significant challenge for researchers, particularly in comprehending the extensive FDA Devices Database. This vital resource catalogs thousands of authorized medical instruments, playing a pivotal role in ensuring compliance and enhancing research outcomes. Given the rapid evolution of regulatory frameworks and the overwhelming volume of information available, researchers must ask: how can they effectively leverage this database to drive innovation and ensure patient safety? This guide provides a step-by-step approach to mastering the FDA Devices Database, equipping researchers with essential tools to streamline their inquiries and make informed decisions within the fast-paced MedTech landscape.
Understand the FDA Devices Database
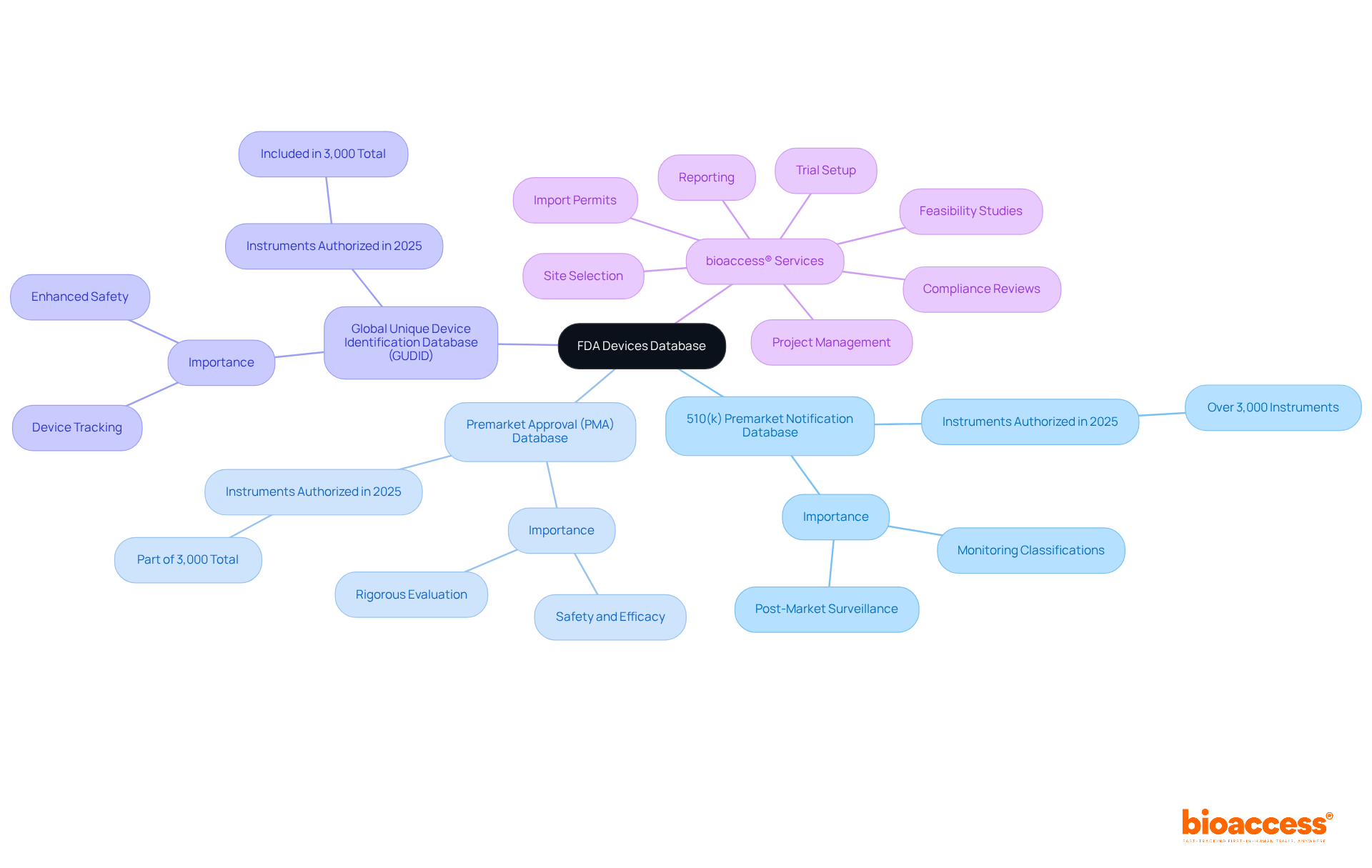
The FDA Equipment Registry serves as a vital resource, cataloging medical instruments authorized for use in the United States. This registry includes several key databases, such as:
- The
- The
- The
Each of these databases is crucial for . Notably, in 2025 alone, the FDA authorized over 3,000 medical instruments, highlighting the database’s . A thorough understanding of the structure and function of these components is essential for researchers, as it empowers them to efficiently locate and interpret device information relevant to their studies.
As industry leaders emphasize, navigating the transcends mere compliance; it involves leveraging this information to . Familiarity with the ultimately enables researchers to meet compliance requirements while advancing their research objectives.
Moreover, partnering with can offer comprehensive , encompassing:
- Feasibility studies
- Site selection
- Compliance reviews
- Trial setup
- Import permits
- Project management
- Reporting
With over 20 years of experience overseeing Early-Feasibility Studies (EFS), , Pilot Studies, Pivotal Studies, and Post-Market Clinical Follow-Up Studies (PMCF), equips researchers with the essential tools and knowledge to adeptly navigate the complexities of clinical trials and regulatory environments, thereby enhancing their ability to utilize the effectively.
Access the FDA Devices Database
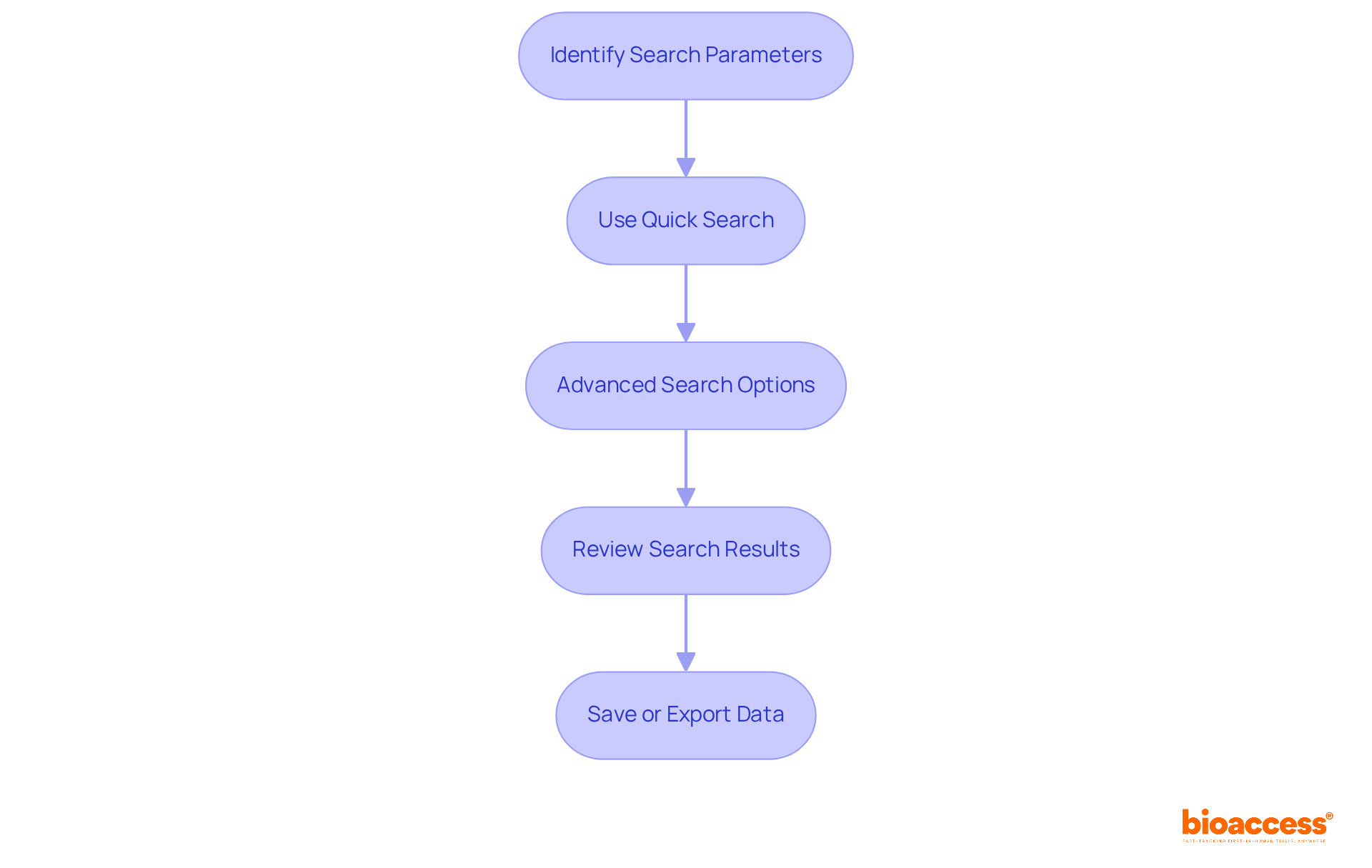
To access the , follow these steps:
- Visit the : Navigate to the official at www.fda.gov.
- Navigate to : Click on the ” tab located in the main menu.
- Select Databases: Under the ‘Device Advice’ section, locate the ” link.
- Choose the Appropriate Resource: Depending on your research needs, select from sources such as Devices@FDA, the , or the GUDID.
- Create an Account (if necessary): Some databases may require account creation for full access. Follow the prompts to register.
- Explore the System: Once logged in, you can start looking for particular items or details using the different search options provided.
Navigating the FDA Equipment Database can be complicated, particularly given that governance structures for are continually changing. Understanding the most accessed databases, particularly the , can streamline your research; for instance, the 510(k) database is frequently utilized for its comprehensive information on device clearances. Experts in the field, such as Ana Criado, Director of Compliance Affairs and a professor with vast experience in biomedical engineering and health economics, stress that “; it’s a continuous commitment to and product quality.” This perspective underscores the importance of mastering the FDA’s resources to ensure adherence to evolving regulatory frameworks.
. Follow the arrows to move from one step to the next, making the process straightforward. Each box represents a step you need to take to access the FDA Devices Database. Follow the arrows to move from one step to the next, making the process straightforward.](https://images.tely.ai/telyai/nqwedjpo-each-box-represents-a-step-you-need-to-take-to-access-the-fda-devices-database-follow-the-arrows-to-move-from-one-step-to-the-next-making-the-process-straightforward.webp)
Utilize Search Features for Device Information
To effectively utilize the search features in the , it is essential to follow these steps:
- Identify : Begin by determining the specific information you require, such as the equipment name, , or manufacturer details.
- Use Quick Search: For a broad overview, employ the by entering relevant keywords related to the equipment. This method enables rapid recognition of items that meet your criteria.
- : For more precise results, switch to the Advanced Search feature. Here, you can filter results based on various parameters, including classification, submission date, or type of equipment, enhancing the relevance of your findings.
- : Carefully . Each entry generally offers a summary, approval status, and links to comprehensive information, which are essential for grasping the apparatus’s compliance status.
- Save or Export Data: If necessary, save or export the search results for further analysis or reporting, facilitating easier access to critical information.
Researchers frequently employ an average of five to seven when navigating these databases, highlighting the complexity of the governing landscape. It is crucial to keep in mind that the includes new items in the 510(k) database around the 5th of each month for products approved in the previous month when organizing your searches. Given that the current regulatory search process is time-consuming and prone to error, a streamlined approach to searching can significantly reduce the time spent on , allowing for more efficient decision-making. By mastering these search features, you can enhance your understanding of the FDA’s classification processes and improve your product development strategies.
Interpret Device Data and Regulatory Information
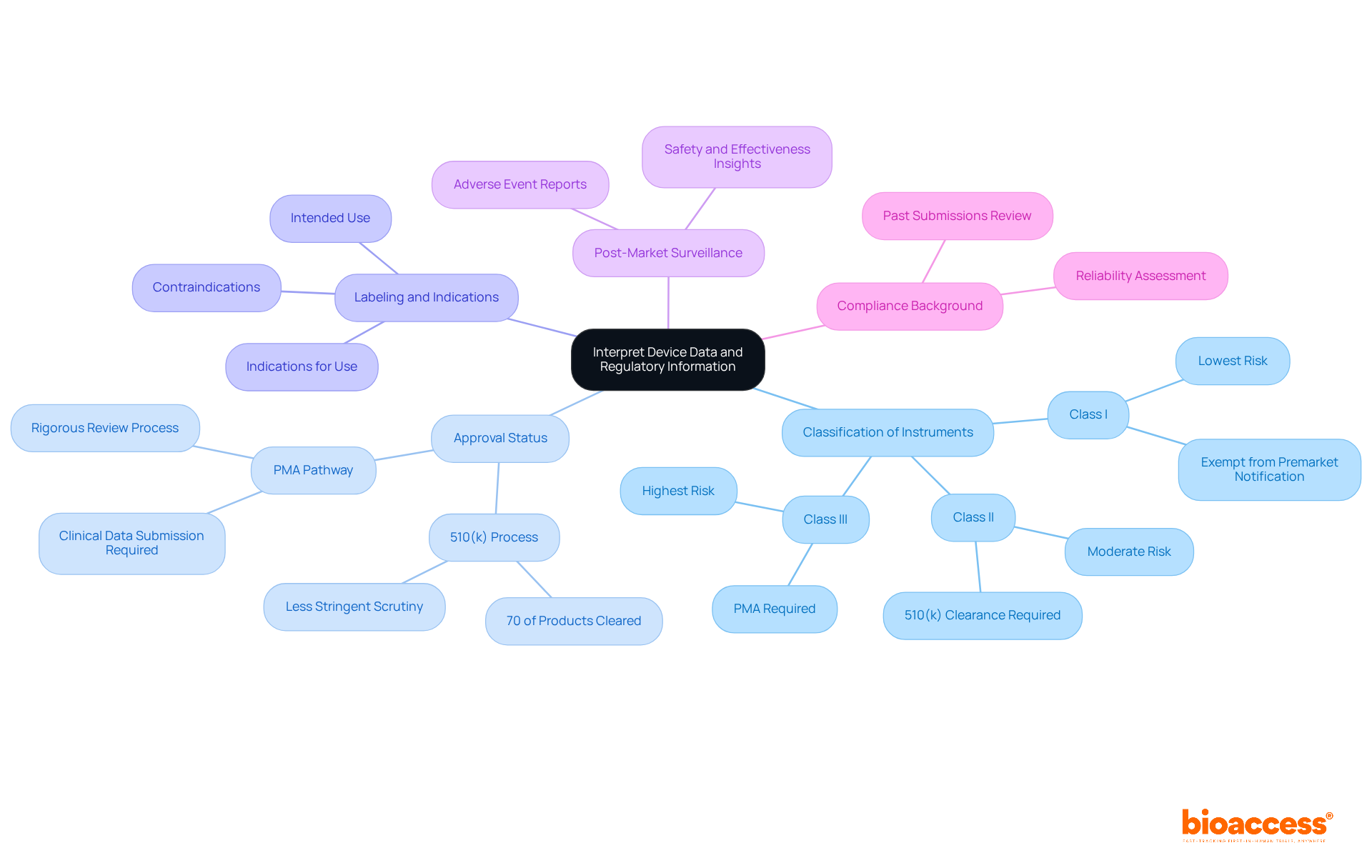
Interpreting device data from the is essential for understanding the complexities of medical instruments and their regulatory landscape.
: (I, II, or III) based on their risk levels. Class I products generally pose the lowest risk and are often exempt from premarket notification, while Class III items, which support or sustain human life, require a more rigorous (PMA) process due to their higher risk. Familiarizing yourself with these classifications is essential for assessing the regulatory requirements listed in the .
: It is crucial to determine whether a product has been cleared through the 510(k) process or approved via the PMA pathway. In 2025, approximately 70% of products submitted to the were cleared through the 510(k) process, indicating that a substantial number of items undergo less stringent scrutiny compared to those requiring PMA.
Labeling and Indications: , which outlines its intended use, indications for use, and any contraindications. This information is crucial for comprehending the extent of the application’s use and ensuring found in the .
Post-Market Surveillance: Investigate any or adverse event reports related to the for the product. These reports can offer valuable insights into the product’s safety and effectiveness, highlighting any potential risks that may arise after market entry.
: Grasping the of a product can assist in assessing its reliability and the FDA’s trust in its performance. A thorough review of past submissions and approvals can inform future development strategies and risk assessments.
Stay Informed on Database Updates and Changes
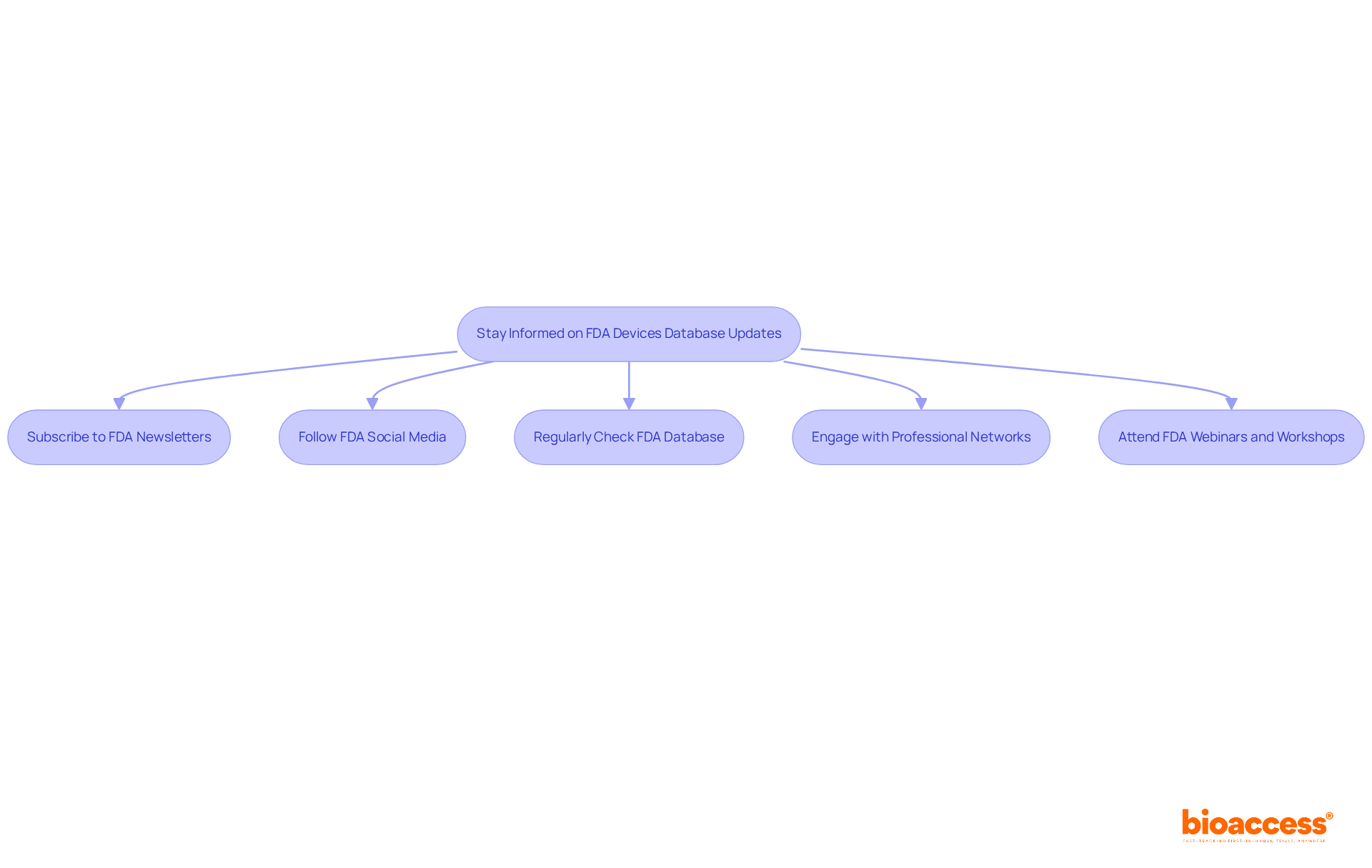
To stay informed about updates and changes to the , consider implementing the following strategies:
- Subscribe to : Enroll in email notifications from the FDA to receive timely updates on new products, regulatory changes, and guidance documents. This approach ensures you remain informed about the latest developments directly from the source.
- Follow FDA Social Media: Engage with the FDA on social media platforms for real-time updates and announcements. This channel has experienced significant engagement, with researchers increasingly utilizing these platforms to stay informed. Notably, approximately 454,3 device-associated adverse events have been reported in recent years, underscoring the importance of staying updated on the .
- Regularly check the : Establish a routine to periodically review the for new entries or changes to existing equipment information. This proactive approach can help you identify important updates that may impact your research.
- Engage with Professional Networks: Join professional organizations or forums focused on . These networks provide valuable insights and discussions on industry trends and compliance changes, fostering a collaborative environment for knowledge sharing.
- Attend : to gain deeper insights into . These events present excellent opportunities to inquire and clarify any uncertainties regarding regulations for the equipment.
By actively engaging with these resources, researchers can enhance their understanding of FDA regulations and ensure compliance, ultimately contributing to improved patient outcomes and device safety.
Conclusion
Mastering the FDA Devices Database is not merely an asset; it is an essential skill for researchers engaged in medical device innovation and regulatory compliance. This comprehensive guide elucidates the significance of the database, detailing its various components and the pivotal role it plays in facilitating informed decision-making and enhancing research outcomes. By adeptly navigating this resource, researchers can ensure compliance while leveraging invaluable insights that propel innovation in the MedTech sector.
Key arguments discussed throughout the article underscore the necessity of understanding the structure of the FDA Devices Database, accessing it correctly, utilizing its search features, interpreting device data, and remaining informed about updates. Each step outlined—from visiting the FDA website to employing advanced search techniques—equips researchers with the essential tools needed to efficiently gather and analyze critical information. Furthermore, the discourse on regulatory classifications and approval processes emphasizes the imperative of a thorough understanding of compliance requirements, ultimately fostering the development of safer and more effective medical devices.
In conclusion, the FDA Devices Database stands as a cornerstone for researchers striving to navigate the complexities of medical device regulation and development. By actively engaging with the database and employing the strategies outlined, researchers can deepen their understanding of regulatory frameworks, ensuring compliance while contributing to advancements in patient safety and device efficacy. Embracing this knowledge not only empowers individual research efforts but also fortifies the overall integrity of the medical device industry.
Frequently Asked Questions
What is the FDA Devices Database?
The FDA Devices Database is a vital resource that catalogs medical instruments authorized for use in the United States. It includes several key databases such as the 510(k) Premarket Notification Database, the Premarket Approval (PMA) Database, and the Global Unique Device Identification Database (GUDID).
Why is the FDA Devices Database important for researchers?
The FDA Devices Database is important for researchers as it helps them monitor classifications, approvals, and post-market surveillance of medical devices. It also empowers them to efficiently locate and interpret device information relevant to their studies, ultimately enhancing research outcomes and driving innovation in the MedTech field.
How can one access the FDA Devices Database?
To access the FDA Devices Database, visit the official FDA website at www.fda.gov, click on the ‘Medical Devices’ tab, select ‘Medical Device Databases’ under the ‘Device Advice’ section, and choose the appropriate resource. Some databases may require account creation for full access.
What types of services does bioaccess® provide for clinical trials?
Bioaccess® offers comprehensive clinical trial management services, including feasibility studies, site selection, compliance reviews, trial setup, import permits, project management, and reporting.
What experience does bioaccess® have in clinical trials?
Bioaccess® has over 20 years of experience overseeing Early-Feasibility Studies (EFS), First-In-Human Studies (FIH), Pilot Studies, Pivotal Studies, and Post-Market Clinical Follow-Up Studies (PMCF), equipping researchers with essential tools and knowledge to navigate clinical trials and regulatory environments effectively.
What is the significance of the 510(k) database?
The 510(k) database is frequently utilized for its comprehensive information on device clearances, making it a key resource for researchers and industry professionals navigating the FDA Devices Database.
How does understanding the FDA Devices Database contribute to compliance?
Mastering the FDA Devices Database is essential for ensuring adherence to evolving regulatory frameworks, as compliance is viewed as a continuous commitment to patient safety and product quality, rather than just a hurdle to overcome.
List of Sources
- Understand the FDA Devices Database
- Global Medical Device Podcast powered by Greenlight Guru (https://podcasts.apple.com/us/podcast/global-medical-device-podcast-powered-by-greenlight-guru/id1036394532)
- Access the FDA Devices Database
- The Top 5 Challenges in Medical Device Regulatory Compliance and How to Overcome Them – Nerac (https://nerac.com/the-top-5-challenges-in-medical-device-regulatory-compliance-and-how-to-overcome-them)
- Utilize Search Features for Device Information
- essenvia.com (https://essenvia.com/blog/what-do-regulatory-specialists-need-to-know-about-searching-the-fda-510(k)-database-for-regulatory-strategy)
- fda.gov (https://fda.gov/medical-devices/classify-your-medical-device/how-determine-if-your-product-medical-device)
- array.aami.org (https://array.aami.org/doi/full/10.2345/0899-8205-54.3.178)
- Interpret Device Data and Regulatory Information
- cohenhealthcarelaw.com (https://cohenhealthcarelaw.com/understanding-the-medical-device-classification-and-approval-process-part-one)
- registrarcorp.com (https://registrarcorp.com/blog/medical-devices/medical-device-registration/understanding-fda-medical-device-classifications)
- fda.gov (https://fda.gov/about-fda/cdrh-transparency/overview-medical-device-classification-and-reclassification)
- Stay Informed on Database Updates and Changes
- bioaccessla.com (https://bioaccessla.com/blog/us-fda-medical-device-approval-steps-for-success)
- Postmarket Surveillance of Medical Devices: A Comparison of Strategies in the US, EU, Japan, and China – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC3815401)
- bioaccessla.com (https://bioaccessla.com/blog/master-the-fda-product-code-for-medical-device-success)

Leave a Reply